Probiotic Effects of a Novel Strain, Acinetobacter KU011TH, on the Growth Performance, Immune Responses, and Resistance against Aeromonas hydrophila of Bighead Catfish (Clarias macrocephalus Günther, 1864)
Abstract
1. Introduction
2. Materials and Methods
2.1. Cultivation and Preparation of the Probiotic Bacterium
2.2. Analyses of Probiotic Shelf Life and Its Efficacy Against Fish Pathogens
2.3. Safety and Pathogenicity Assessments of Acinetobacter KU011TH on C. macrocephalus
2.4. Preparation of the Acinetobacter KU011TH-Supplemented Diets and Water-Soluble Bacteria
2.5. Experimental Fish and Conditions
2.6. Effects of Acinetobacter KU011TH Application on Growth Performance after 120 Days of Trial and on Innate Immune Responses, Immune-Related Gene Expression, and Skin Physical Characteristics of C. macrocephalus after 30 Days of Trial
2.6.1. Experimental Designs
2.6.2. Collection of Peripheral Blood Lymphocytes (PBLs) and Fish Tissues
2.6.3. Effects of Acinetobacter KU011TH on the Growth Performance of Bighead Catfish at 120 Days
2.6.4. Effects of Acinetobacter KU011TH on the Innate Immune Response of Bighead Catfish
Lysozyme Activity Analysis
Respiratory Burst Reaction Analysis
Hemolytic Activity by the Alternative Complement Pathway (ACH50) Assay
Bactericidal Activity Analysis
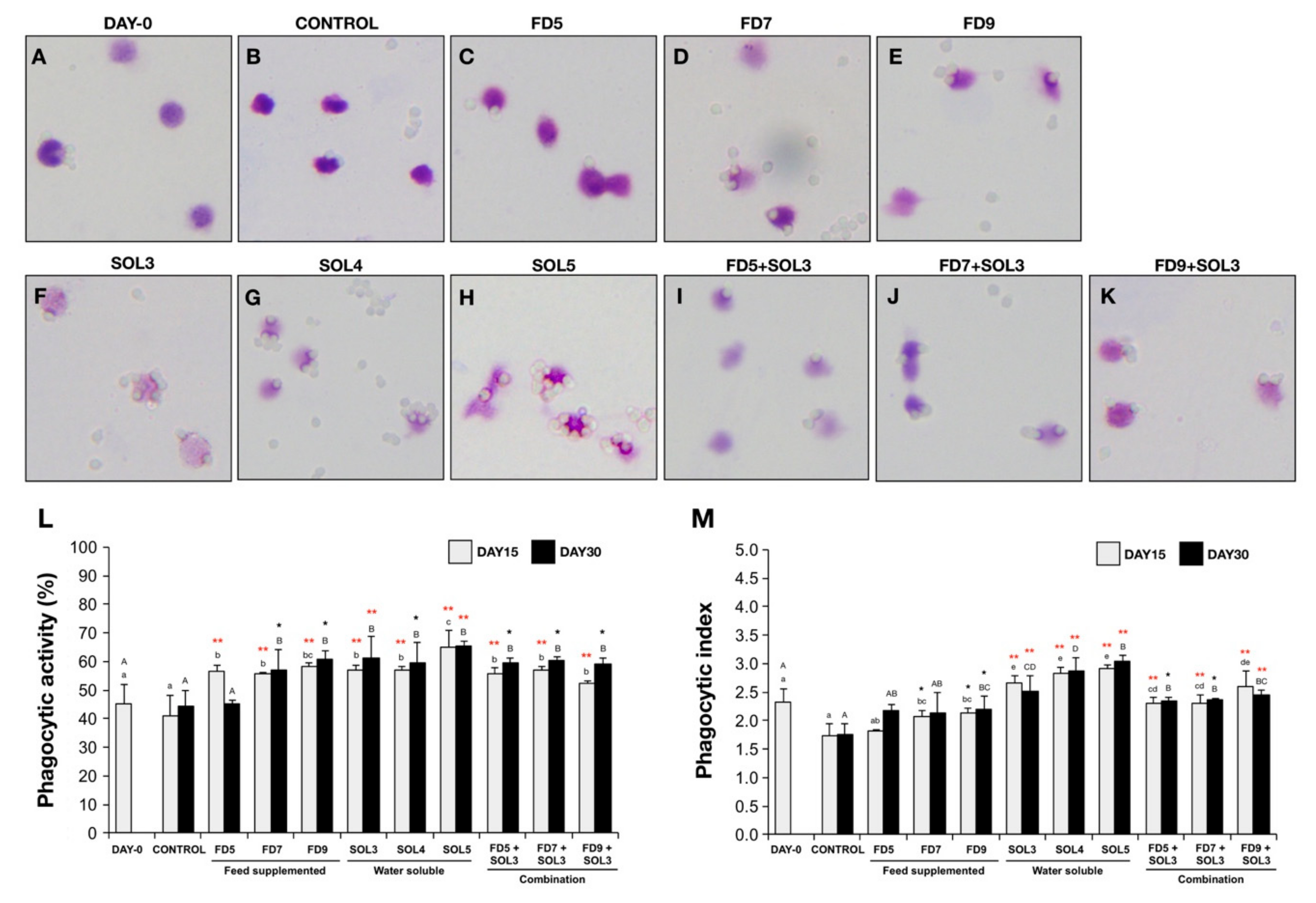
Phagocytic Activity Analysis
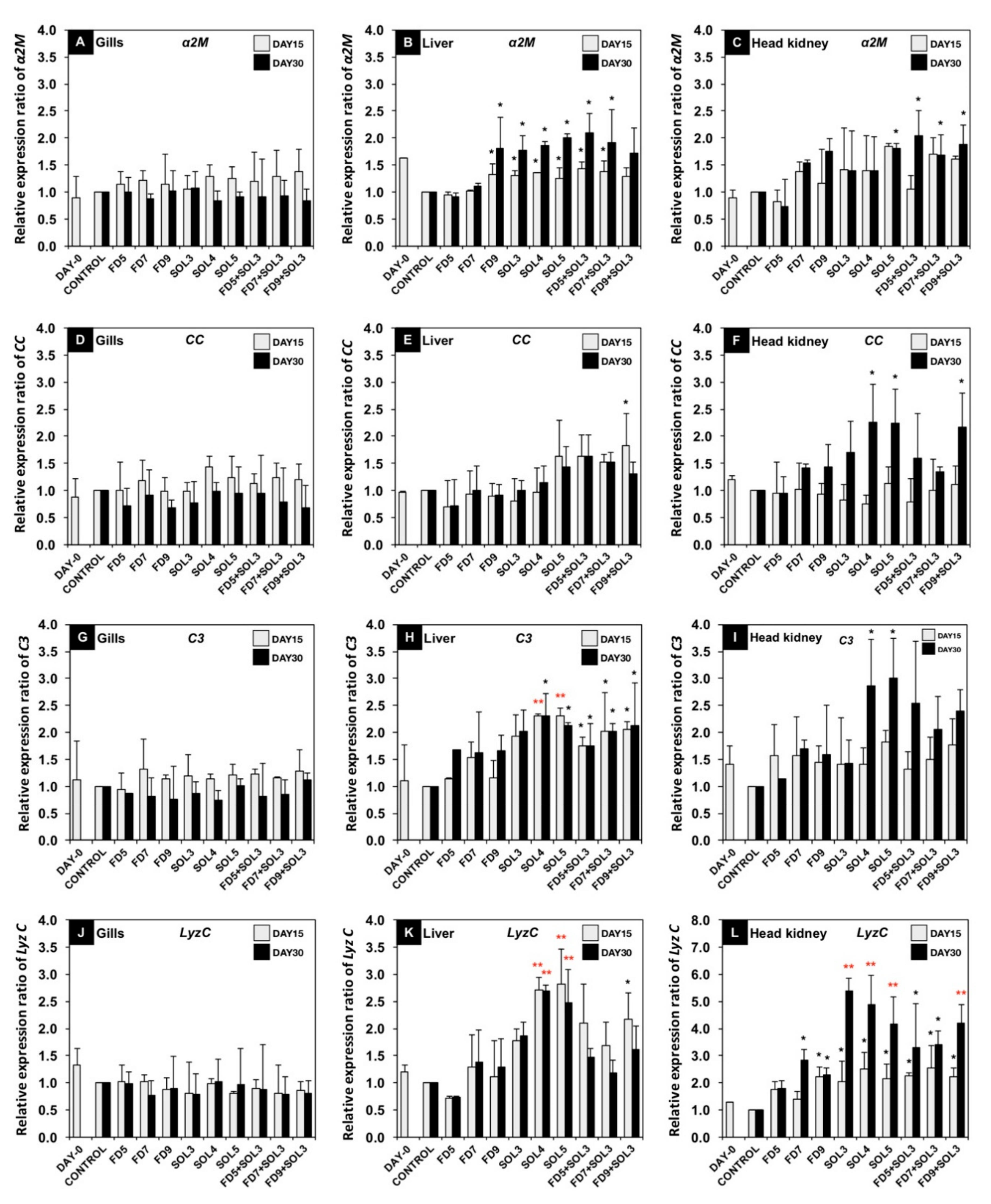
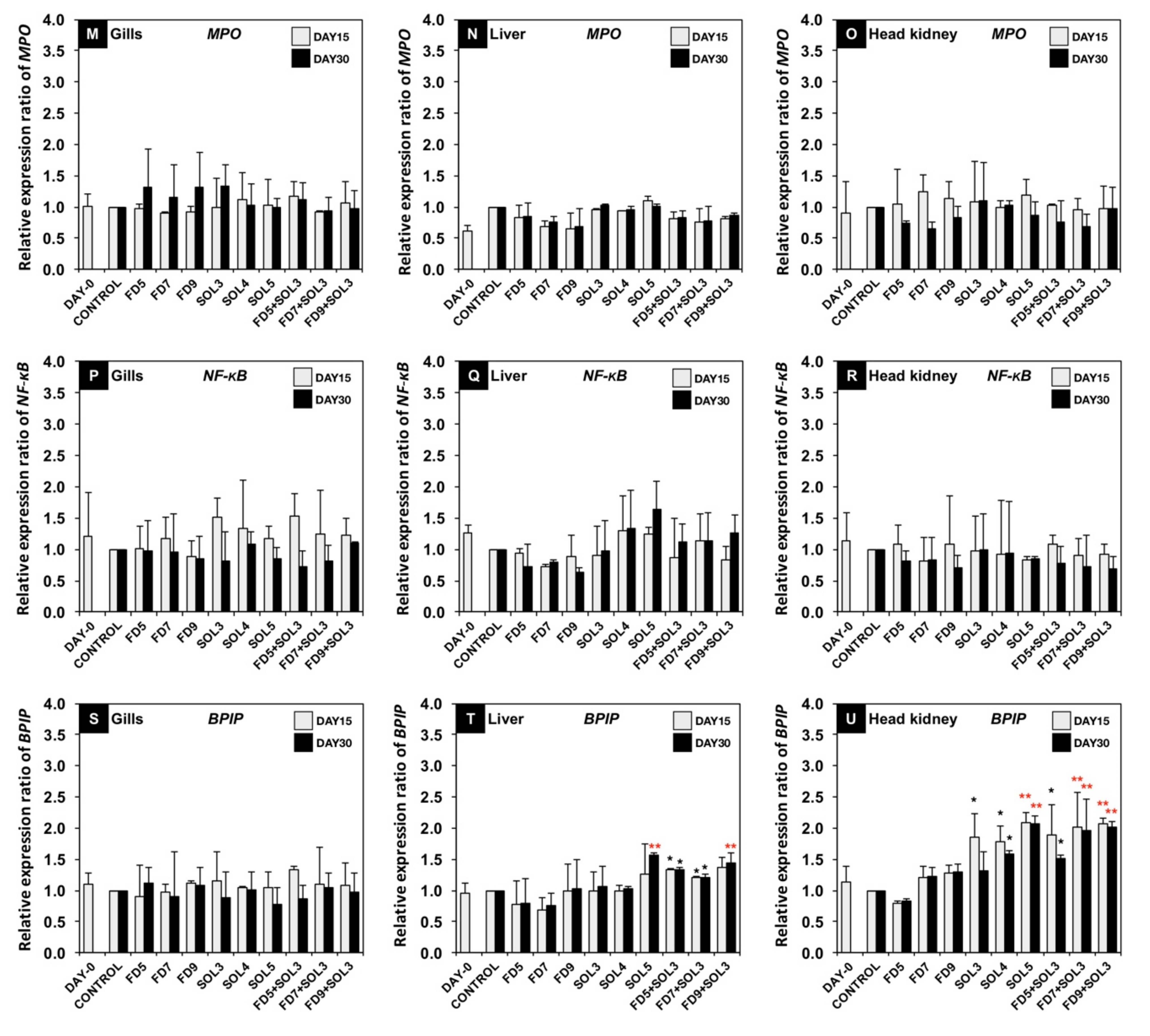
2.6.5. Effects of Acinetobacter KU011TH on Immune-Related Gene Expression of Bighead Catfish
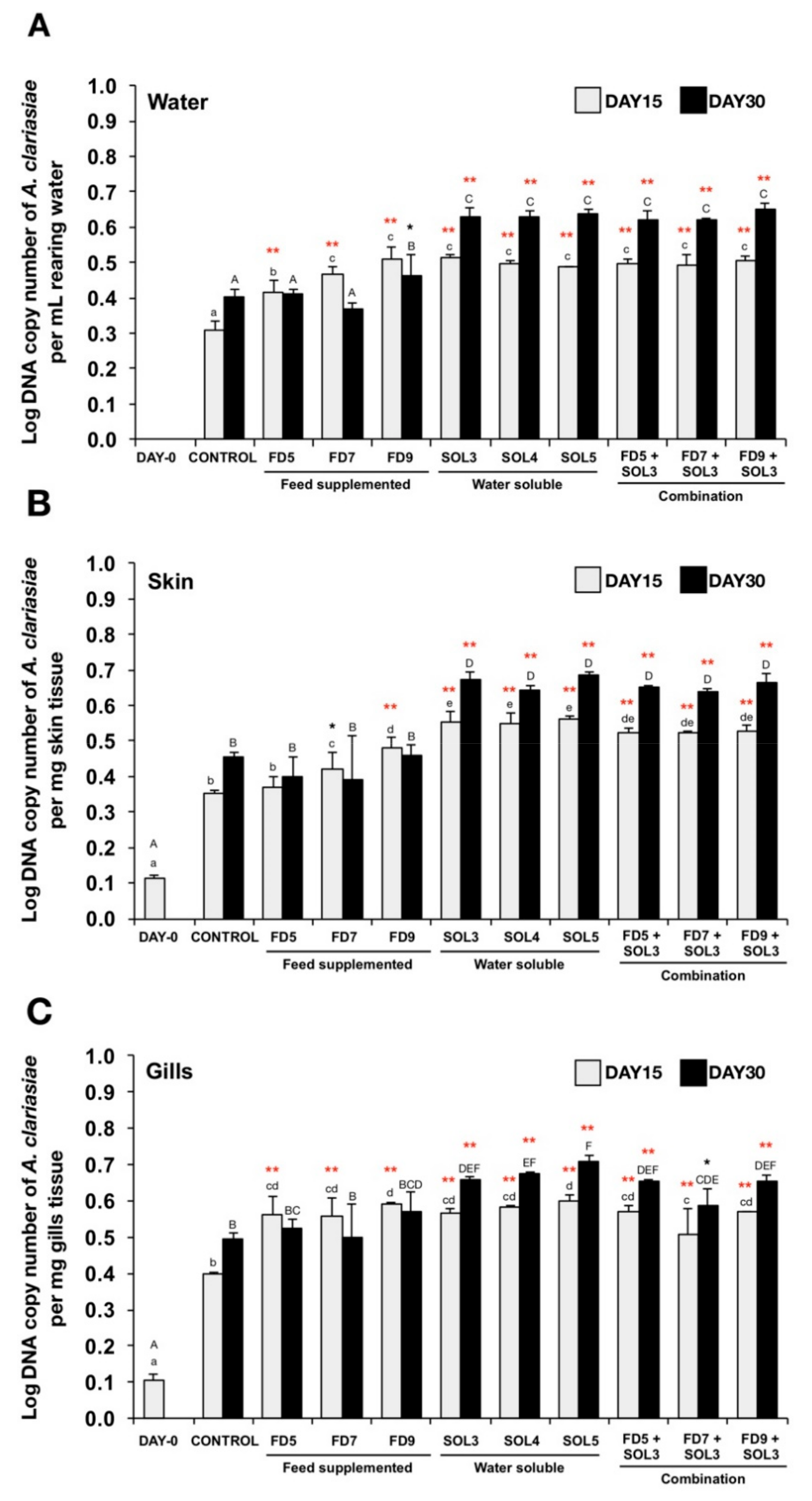
2.6.6. Quantification of Acinetobacter KU011TH in the Rearing Water, Skin, Gills and Intestine of Bighead Catfish
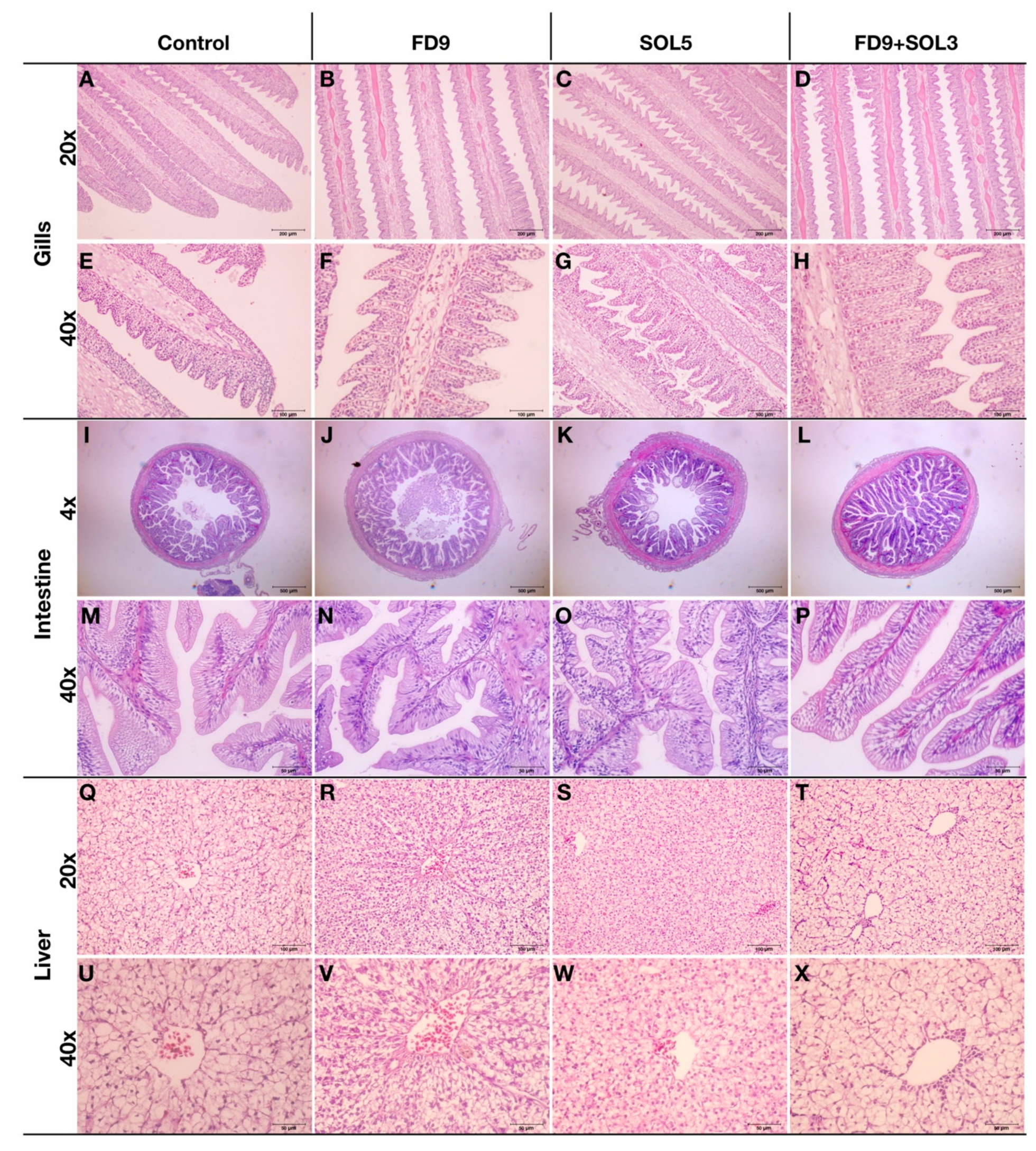
2.6.7. Effects of Acinetobacter KU011TH on the Histopathology of the Gills, Skin, Intestine and Liver of Bighead Catfish
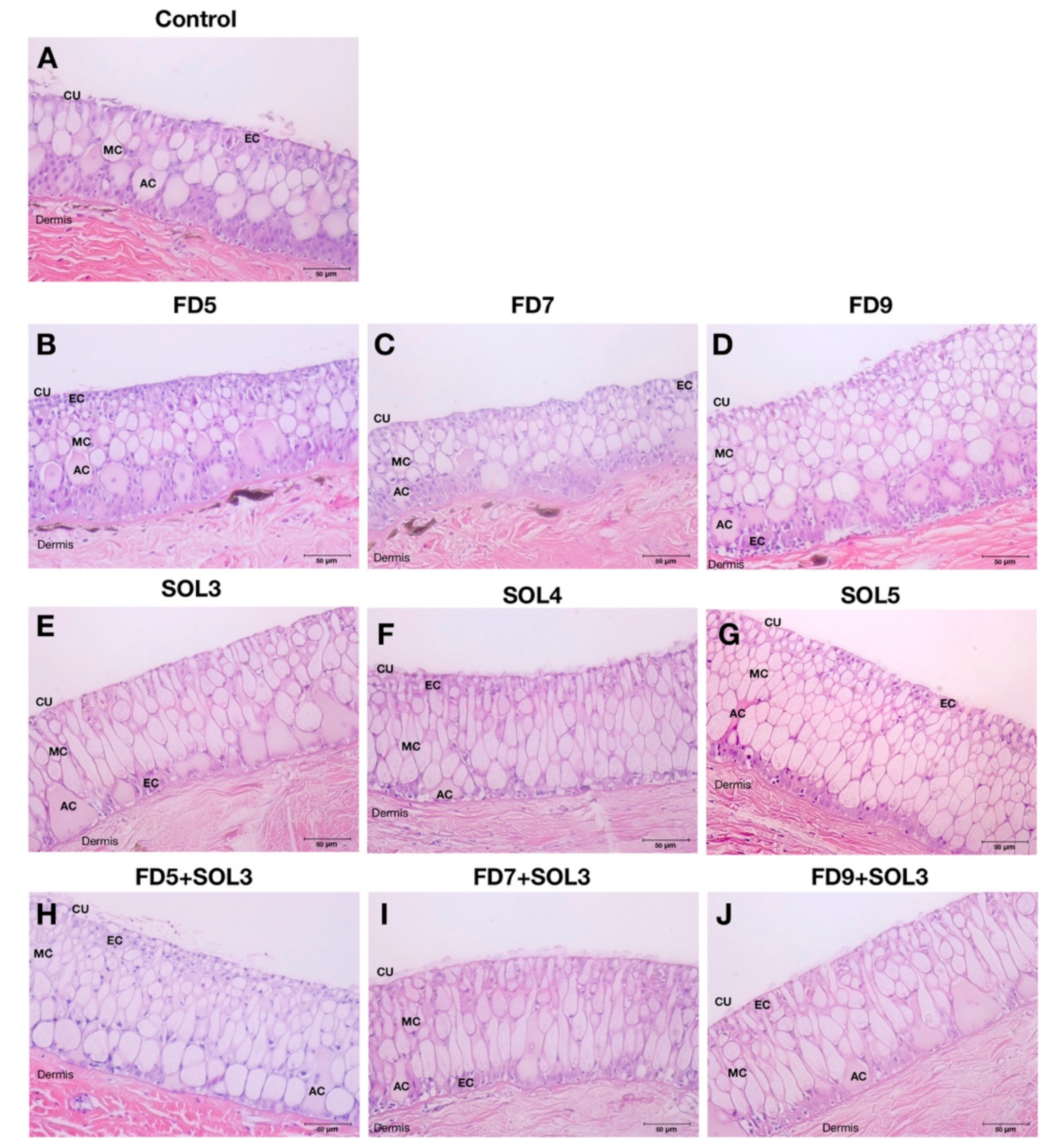
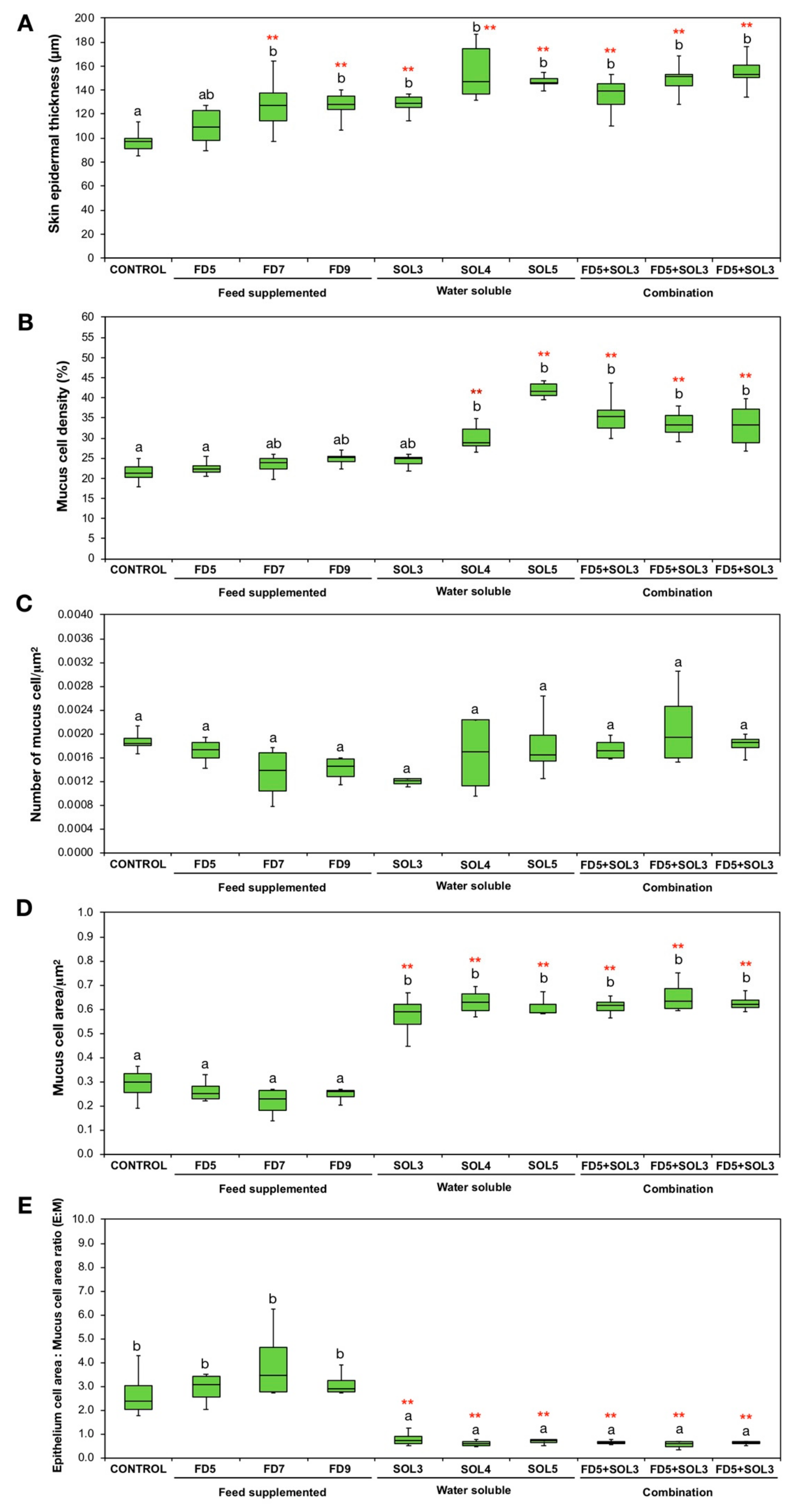
2.6.8. Effects of Acinetobacter KU011TH on the Skin Epithelium and Skin Mucus Cells of Bighead Catfish
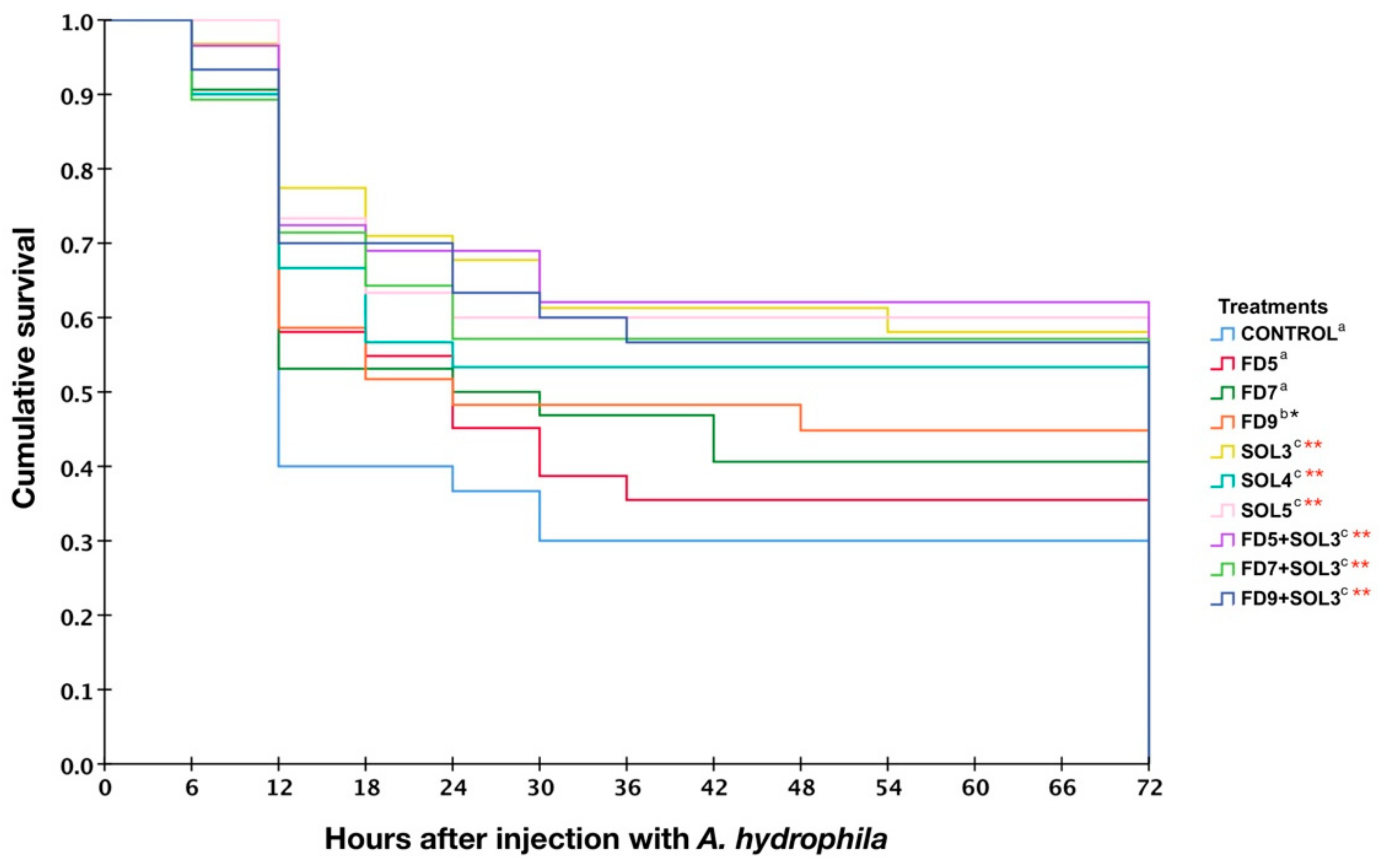
2.6.9. Effects of Acinetobacter KU011TH on Disease Resistance of Bighead Catfish against A. hydrophila
2.6.10. Statistical and Data Analysis
3. Results
3.1. Analysis of the Bacterial Shelf Life of the Probiotic and its Efficacy Against Pathogens
3.2. Safety and Pathogenicity Assessments of Acinetobacter KU011TH on C. macrocephalus
3.3. Effect of a Probiotic Bacterium on Growth Performance in a 120-Day Trial
3.4. Effect of a Probiotic Bacterium on Innate Immune Responses, Immune-Related Gene Expression, and Skin Characteristics of C. macrocephalus
3.4.1. Humoral and Cellular Immune Responses of Innate Immunity
3.4.2. mRNA Expression of Immune-Related Genes
3.4.3. Quantification of Acinetobacter KU011TH in Water, Skin, Gills and Intestine
3.4.4. Skin Epithelial and Mucus Cells
3.4.5. Histopathology of the Skin, Gills, Intestine and Liver
3.4.6. Resistance against A. hydrophila
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations (FAO). Fishstat Plus Rome 2017. In: FAO Fisheries and Aquaculture Department. Rome. Updated 14 September 2017. Available online: http://www.fao.org/fishery/statistics/software/fishstat/en (accessed on 1 December 2018).
- Na-Nakorn, U.; Kamonrat, W.; Ngamsiri, T. Genetic diversity of walking catfish, Clarias macrocephalus, in Thailand and evidence of genetic introgression from introduced farmed C. gariepinus. Aquaculture 2004, 240, 145–163. [Google Scholar] [CrossRef]
- Chatchaiphan, S.; Srisapoome, P.; Kim, J.H.; Devlin, R.H.; Na-Nakorn, U. De novo transcriptome characterization and growth-related gene expression profiling of diploid and triploid bighead catfish (Clarias macrocephalus Gunther, 1864). Mar. Biotechnol. (Ny) 2017, 19, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Suanyuk, N.; Rogge, M.; Thune, R.; Watthanaphiromsakul, M.; Champhat, N.; Wiangkum, W. Mortality and pathology of hybrid catfish, Clarias macrocephalus (Gunther) x Clarias gariepinus (Burchell), associated with Edwardsiella ictaluri infection in southern Thailand. J. Fish. Dis. 2014, 37, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Na-Nakorn, U.; Chantsawang, S.; Tarnchalanukit, W. Response to mass selection for disease resistance in walking catfish, Clarias macrocephalus. J. Appl. Aquac. 1995, 4, 65–74. [Google Scholar] [CrossRef]
- Hoseinifar, S.H.; Dadar, M.; Ringø, E. Modulation of nutrient digestibility and digestive enzyme activities in aquatic animals: The functional feed additives scenario. Aquac. Res. 2017, 48, 3987–4000. [Google Scholar] [CrossRef]
- Nawaz, A.; Bakhsh javaid, A.; Irshad, S.; Hoseinifar, S.H.; Xiong, H. The functionality of prebiotics as immunostimulant: Evidences from trials on terrestrial and aquatic animals. Fish Shellfish Immunol. 2018, 76, 272–278. [Google Scholar] [CrossRef]
- Fuller, R. Probiotics in man and animals. J. Appl. Bacteriol. 1989, 66, 365–378. [Google Scholar]
- Merrifield, D.L.; Dimitroglou, A.; Foey, A.; Davies, S.; Baker, R.T.M.; Bøgwald, J.; Castex, M.; Ringø, E. The current status and future focus of probiotic and prebiotic applications for salmonids. Aquaculture 2010, 302, 1–18. [Google Scholar] [CrossRef]
- Martínez Cruz, P.; Ibáñez, A.L.; Monroy Hermosillo, O.A.; Ramírez Saad, H.C. Use of probiotics in aquaculture. ISRN Microbiol. 2012, 2012, 916845. [Google Scholar] [CrossRef]
- Van Doan, H.; Hoseinifar, S.H.; Ringø, E.; Ángeles Esteban, M.; Dadar, M.; Dawood, M.A.O.; Faggio, C. Host-Associated Probiotics: A Key Factor in Sustainable Aquaculture. Rev. Fish. Sci. Aquac. 2019, 1–27. [Google Scholar] [CrossRef]
- Bunnoy, A.; Na-Nakorn, U.; Kayansamruaj, P.; Srisapoome, P. Acinetobacter strain KUO11TH, a unique organism related to Acinetobacter pittii and isolated from the skin mucus of healthy bighead catfish and its efficacy against several fish pathogens. Microorganisms 2019, 7, 459. [Google Scholar] [CrossRef] [PubMed]
- Chinedu, E.; Arome, D.; Ameh, F.S. A new method for determining acute toxicity in animal models. Int. J. Toxicol. 2013, 20, 224–226. [Google Scholar] [CrossRef] [PubMed]
- Boyum, A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand. J. Clin. Lab. Invest. Suppl. 1968, 97, 77–89. [Google Scholar] [PubMed]
- Bernet, D.; Schmidt, H.; Meier, W.; Burkhardt-Holm, P.; Wahli, T. Histopathology in fish: proposal for a protocol to assess aquatic pollution. J. Fish. Dis. 1999, 22, 25–34. [Google Scholar] [CrossRef]
- Lugert, V.; Thaller, G.; Tetens, J.; Schulz, C.; Krieter, J. A review on fish growth calculation: multiple functions in fish production and their specific application. Rev. Aquac. 2016, 8, 30–42. [Google Scholar] [CrossRef]
- Ito, Y.; Yamada, H.; Imoto, T. Colorimetric assay for lysozyme using Micrococcus luteus labeled with a blue dye, Remazol brilliant blue R, as a substrate. Chem. Pharm. Bull. (Tokyo) 1992, 40, 1523–1526. [Google Scholar] [CrossRef] [PubMed]
- Segal, A.W.; Levi, A.J. Cell damage and dye reduction in the quantitative nitroblue tetrazolium (NBT) test. Clin. Exp. Immunol. 1975, 19, 309–318. [Google Scholar]
- Nayak, S.; Portugal, I.; Zilberg, D. Analyzing complement activity in the serum and body homogenates of different fish species, using rabbit and sheep red blood cells. Vet. Immunol. Immunopathol. 2018, 199, 39–42. [Google Scholar] [CrossRef]
- Guo, L.; Zhang, F.; Wang, X.; Chen, H.; Wang, Q.; Guo, J.; Cao, X.; Wang, L. Antibacterial activity and action mechanism of questin from marine Aspergillus flavipes HN4-13 against aquatic pathogen Vibrio harveyi. 3 Biotech. 2019, 9, 1–14. [Google Scholar] [CrossRef]
- Koenigsknecht, J.; Landreth, G. Microglial phagocytosis of fibrillar beta-amyloid through a beta1 integrin-dependent mechanism. J. Neurosci. 2004, 24, 9838–9846. [Google Scholar] [CrossRef]
- Panigrahi, A.; Kiron, V.; Kobayashi, T.; Puangkaew, J.; Satoh, S.; Sugita, H. Immune responses in rainbow trout Oncorhynchus mykiss induced by a potential probiotic bacteria Lactobacillus rhamnosus JCM 1136. Vet. Immunol. Immunopathol. 2004, 102, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Hai, N.V. Research findings from the use of probiotics in tilapia aquaculture: A review. Fish Shellfish Immunol. 2015, 45, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Hoseinifar, S.H.; Hosseini, M.; Paknejad, H.; Safari, R.; Jafar, A.; Yousefi, M.; Van Doan, H.; Torfi Mozanzadeh, M. Enhanced mucosal immune responses, immune related genes and growth performance in common carp (Cyprinus carpio) juveniles fed dietary Pediococcus acidilactici MA18/5M and raffinose. Dev. Comp. Immunol. 2019, 94, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-H.; Chiu, C.-H.; Wang, S.-W.; Cheng, W. Dietary administration of the probiotic, Bacillus subtilis E20, enhances the growth, innate immune responses, and disease resistance of the grouper, Epinephelus coioides. Fish Shellfish Immunol. 2012, 33, 699–706. [Google Scholar] [CrossRef] [PubMed]
- Meidong, R.; Khotchanalekha, K.; Doolgindachbaporn, S.; Nagasawa, T.; Nakao, M.; Sakai, K.; Tongpim, S. Evaluation of probiotic Bacillus aerius B81e isolated from healthy hybrid catfish on growth, disease resistance and innate immunity of Pla-mong Pangasius bocourti. Fish Shellfish Immunol. 2018, 73, 1–10. [Google Scholar] [CrossRef]
- Truong Thy, H.T.; Tri, N.N.; Quy, O.M.; Fotedar, R.; Kannika, K.; Unajak, S.; Areechon, N. Effects of the dietary supplementation of mixed probiotic spores of Bacillus amyloliquefaciens 54A, and Bacillus pumilus 47B on growth, innate immunity and stress responses of striped catfish (Pangasianodon hypophthalmus). Fish Shellfish Immunol. 2017, 60, 391–399. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Fischer, A.H.; Jacobson, K.A.; Rose, J.; Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. Csh. Protoc. 2008, 49, 86. [Google Scholar] [CrossRef]
- Torrecillas, S.; Montero, D.; Caballero, M.J.; Pittman, K.A.; Custodio, M.; Campo, A.; Sweetman, J.; Izquierdo, M. Dietary mannan oligosaccharides: counteracting the side effects of soybean meal oil inclusion on European sea bass (Dicentrarchus labrax) gut health and skin mucosa mucus production? Front. Immunol. 2015, 6, 397. [Google Scholar] [CrossRef]
- Sosa, J.M.; Huber, D.E.; Welk, B.; Fraser, H.L. Development and application of MIPAR™: a novel software package for two- and three-dimensional microstructural characterization. IMMI 2014, 3, 10. [Google Scholar] [CrossRef]
- Kent, M.L.; Feist, S.W.; Harper, C.; Hoogstraten-Miller, S.; Law, J.M.; Sánchez-Morgado, J.M.; Tanguay, R.L.; Sanders, G.E.; Spitsbergen, J.M.; Whipps, C.M. Recommendations for control of pathogens and infectious diseases in fish research facilities. Comp. Biochem. Physiol. Toxicol. Pharm. 2009, 149, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Bewick, V.; Cheek, L.; Ball, J. Statistics review 12: survival analysis. Crit Care 2004, 8, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Kim, P.S.; Shin, N.R.; Kim, J.Y.; Yun, J.H.; Hyun, D.W.; Bae, J.W. Acinetobacter apis sp. nov., isolated from the intestinal tract of a honey bee, Apis mellifera. J. Microbiol. 2014, 52, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, Y.; Ruan, Z.; Ma, K.; Wu, B.; Xu, Y.; Wang, J.; You, Y.; He, M.; Hu, G. Acinetobacter larvae sp. nov., isolated from the larval gut of Omphisa fuscidentalis. Int. J. Syst. Evol. Microbiol. 2017, 67, 806–811. [Google Scholar] [CrossRef]
- O’Sullivan, M.G.; Thornton, G.; O’Sullivan, G.C.; Collins, J.K. Probiotic bacteria: myth or reality? Trends Food Sci. Technol. 1992, 3, 309–314. [Google Scholar] [CrossRef]
- Brunt, J.; Austin, B. Use of a probiotic to control lactococcosis and streptococcosis in rainbow trout, Oncorhynchus mykiss (Walbaum). J. Fish. Dis. 2005, 28, 693–701. [Google Scholar] [CrossRef]
- Salinas, I.; Cuesta, A.; Esteban, M.A.; Meseguer, J. Dietary administration of Lactobacillus delbrueckii and Bacillus subtilis, single or combined, on gilthead seabream cellular innate immune responses. Fish Shellfish Immunol. 2005, 19, 67–77. [Google Scholar] [CrossRef]
- Aly, S.M.; Abdel-Galil Ahmed, Y.; Abdel-Aziz Ghareeb, A.; Mohamed, M.F. Studies on Bacillus subtilis and Lactobacillus acidophilus, as potential probiotics, on the immune response and resistance of Tilapia nilotica (Oreochromis niloticus) to challenge infections. Fish Shellfish Immunol. 2008, 25, 128–136. [Google Scholar] [CrossRef]
- Azimirad, M.; Meshkini, S.; Ahmadifard, N.; Hoseinifar, S.H. The effects of feeding with synbiotic (Pediococcus acidilactici and fructooligosaccharide) enriched adult Artemia on skin mucus immune responses, stress resistance, intestinal microbiota and performance of angelfish (Pterophyllum scalare). Fish Shellfish Immunol. 2016, 54, 516–522. [Google Scholar] [CrossRef]
- Irianto, A.; Austin, B. Use of probiotics to control furunculosis in rainbow trout, Oncorhynchus mykiss (Walbaum). J. Fish. Dis. 2002, 25, 333–342. [Google Scholar] [CrossRef]
- Aly, S.M.; Mohamed, M.F.; John, G. Effect of probiotics on the survival, growth and challenge infection in Tilapia nilotica (Oreochromis niloticus). Aquac. Res. 2008, 39, 647–656. [Google Scholar] [CrossRef]
- Nayak, S.K.; Swain, P.; Mukherjee, S.C. Effect of dietary supplementation of probiotic and vitamin C on the immune response of Indian major carp, Labeo rohita (Ham.). Fish Shellfish Immunol. 2017, 23, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Thurlow, C.M.; Williams, M.A.; Carrias, A.; Ran, C.; Newman, M.; Tweedie, J.; Allison, E.; Jescovitch, L.N.; Wilson, A.E.; Terhune, J.S.; et al. Bacillus velezensis AP193 exerts probiotic effects in channel catfish (Ictalurus punctatus) and reduces aquaculture pond eutrophication. Aquaculture 2019, 503, 347–356. [Google Scholar] [CrossRef]
- Mohammadian, T.; Nasirpour, M.; Tabandeh, M.R.; Heidary, A.A.; Ghanei-Motlagh, R.; Hosseini, S.S. Administrations of autochthonous probiotics altered juvenile rainbow trout Oncorhynchus mykiss health status, growth performance and resistance to Lactococcus garvieae, an experimental infection. Fish Shellfish Immunol. 2019, 86, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Moniruzzaman, M.; Lee, S.; Hong, J.; Won, S.; Lee, J.M.; Yun, H.; Kim, K.-W.; Ko, D.; Bai, S.C. Comparison of the effects of dietary single and multi-probiotics on growth, non-specific immune responses and disease resistance in starry flounder, Platichthys stellatus. Fish Shellfish Immunol. 2016, 59, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Van Doan, H.; Hoseinifar, S.H.; Khanongnuch, C.; Kanpiengjai, A.; Unban, K.; Van Kim, V.; Srichaiyo, S. Host-associated probiotics boosted mucosal and serum immunity, disease resistance and growth performance of Nile tilapia (Oreochromis niloticus). Aquaculture 2018, 491, 94–100. [Google Scholar] [CrossRef]
- Butel, M.J. Probiotics, gut microbiota and health. Med. Mal. Infect. 2014, 44, 1–8. [Google Scholar] [CrossRef]
- Di Gioia, D.; Aloisio, I.; Mazzola, G.; Biavati, B. Bifidobacteria: their impact on gut microbiota composition and their applications as probiotics in infants. Appl. Microbiol. Biotechnol. 2014, 98, 563–577. [Google Scholar] [CrossRef]
- Vieira, A.T.; Teixeira, M.M.; Martins, F.S. The role of probiotics and prebiotics in inducing gut immunity. Front. Immunol. 2013, 4, 445. [Google Scholar] [CrossRef]
- Sun, Y.Z.; Yang, H.L.; Ma, R.L.; Lin, W.Y. Probiotic applications of two dominant gut Bacillus strains with antagonistic activity improved the growth performance and immune responses of grouper Epinephelus coioides. Fish Shellfish Immunol. 2010, 29, 803–809. [Google Scholar] [CrossRef]
- Ramesh, D.; Souissi, S. Effects of potential probiotic Bacillus subtilis KADR1 and its subcellular components on immune responses and disease resistance in Labeo rohita. Aquac. Res. 2018, 49, 367–377. [Google Scholar] [CrossRef]
- Silva-Gomes, S.; Decout, A.; Nigou, J. Pathogen-associated molecular patterns (PAMPs). In Encyclopedia of Inflammatory Diseases; Parnham, M., Ed.; Springer Basel: Basel, Switzerland, 2015; pp. 1–16. [Google Scholar]
- Lazado, C.C.; Caipang, C.M.A. Mucosal immunity and probiotics in fish. Fish Shellfish Immunol. 2014, 39, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.M.; Kemp, G.D.; Molle, M.G.; Smith, V.J. Anti-microbial properties of histone H2A from skin secretions of rainbow trout, Oncorhynchus mykiss. Biochem. J. 2002, 368, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Noga, E.J.; Silphaduang, U. Piscidins: a novel family of peptide antibiotics from fish. Drug News Perspect. 2003, 16, 87–92. [Google Scholar] [CrossRef]
- Rakers, S.; Gebert, M.; Uppalapati, S.; Meyer, W.; Maderson, P.; Sell, A.F.; Kruse, C.; Paus, R. ‘Fish matters’: the relevance of fish skin biology to investigative dermatology. Exp. Derm. 2010, 19, 313–324. [Google Scholar] [CrossRef]
- Rakers, S.; Niklasson, L.; Steinhagen, D.; Kruse, C.; Schauber, J.; Sundell, K.; Paus, R. Antimicrobial peptides (AMPs) from fish epidermis: perspectives for investigative dermatology. J. Investig. Derm. 2013, 133, 1140–1149. [Google Scholar] [CrossRef]
- Subramanian, S.; MacKinnon, S.L.; Ross, N.W. A comparative study on innate immune parameters in the epidermal mucus of various fish species. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2007, 148, 256–263. [Google Scholar] [CrossRef]
- Loganathan, K.; Muniyan, M.; Arul Prakash, A.; Senthil Raja, P.; Prakash, M. Studies on the role of mucus from Clarias batrachus (Linn) against selected microbes. IJPA 2011, 2, 202–206. [Google Scholar]
- Modanloo, M.; Soltanian, S.; Akhlaghi, M.; Hoseinifar, S.H. The effects of single or combined administration of galactooligosaccharide and Pediococcus acidilactici on cutaneous mucus immune parameters, humoral immune responses and immune related genes expression in common carp (Cyprinus carpio) fingerlings. Fish Shellfish Immunol. 2017, 70, 391–397. [Google Scholar] [CrossRef]
- Pittman, K.; Pittman, A.; Karlson, S.; Cieplinska, T.; Sourd, P.; Redmond, K.; Ravnoy, B.; Sweetman, E. Body site matters: an evaluation and application of a novel histological methodology on the quantification of mucous cells in the skin of Atlantic salmon, Salmo salar L. J. Fish. Dis. 2013, 36, 115–127. [Google Scholar] [CrossRef]
- Ellis, A.E. Immunity to bacteria in fish. Fish Shellfish Immunol. 1999, 9, 291–308. [Google Scholar] [CrossRef]
- Kurath, G.; Adams, A. Foreword: pathogens and immune responses of fish and reptiles. Vet. Res. 2011, 42, 101. [Google Scholar] [CrossRef]
- Brunt, J.; Newaj-Fyzul, A.; Austin, B. The development of probiotics for the control of multiple bacterial diseases of rainbow trout, Oncorhynchus mykiss (Walbaum). J. Fish. Dis. 2007, 30, 573–579. [Google Scholar] [CrossRef]

| Gene | Primer Names | Nucleotide Sequences (5’-3’) | Amplicon Size (bp) | Tm (°C) | Accession Number |
|---|---|---|---|---|---|
| Acinetobacter KU011TH gyrB | Ac_gyrB_F Ac_gyrB_R | GGCGTGCGTATTGTTTTACGTGAT CAATACCGTTTTCTGTATCTGCGG | 154 | 59 | MG950236 |
| Alpha-2-macroglobulin (A2M) | Cm_A2M_F Cm_A2M_R | TACTTCTCTACAATGCCCCTACAC GATCGGCTATGAACCCTGATAAGA | 192 | 60 | SRS1284019 |
| CC chemokine (CC) | Cm_CC_F Cm_CC_R | TCTTAACAATGAAGCCTTGCTGTG ACATGAAGATGAACCGTGTGTTTT | 196 | 59 | |
| C3 complement (C3) | Cm_C3_F Cm_C3_R | GAGCAAATACTTTGGCAACATACG GTGAGGCTATCCAACACATTCAGA | 135 | 60 | |
| Lysozyme C (LyzC) | Cm_LYZc_F Cm_LYZc_R | TGCTAAACAGTATGATCGGTGTGA TATCTGGAAAATGCCGTAGTCTGT | 178 | 60 | |
| Myeloperoxide (MPO) | Cm_MPO_F Cm_MPO_ R | ACTTCCTCAACTGCAGAAGTATCC GAGGCTTCTCATTACTGGCTGAT | 124 | 60 | |
| Nuclear factor kappa B (NF-κB) | Cm_NFKb_F Cm_NFKb_R | CGTACCCTCAGGTTAAGATCTGTC TACCAAGGTTAGGGAAACTGATGG | 183 | 60 | |
| Bactericidal/permeability-increasing protein (BPIP) | Cm_BPIP_F Cm_BPIP_R | TCGACTGAACACAAAGACATTTGG GCTGAATAGCGTAAGCAGTAATGG | 170 | 59 | |
| Beta-actin (β-actin) | Cm_ βActin_F Cm_ βActin _R | GTCCGTGACATCAAGGAGAAGCTC GGACTCCATACCCAGGAAAGATGG | 189 | 59 |
| Week | Total Viable Count ( × 107 CFU/mL) | Percentage Change * | Clearance Zone Diameter (cm) |
|---|---|---|---|
| 0 (control) | 6.77 ± 0.28 d | 0 | 1.76 ± 0.03 a |
| 1 | 6.49 ± 0.42 d | −4.19% | 1.73 ± 0.03 a |
| 2 | 6.56 ± 0.38 d | −3.15% | 1.69 ± 0.09 a |
| 3 | 6.44 ± 0.23 d | −4.87% | 1.66 ± 0.03 a |
| 4 | 6.46 ± 0.42 d | - 4.53% | 1.66 ± 0.05 a |
| 5 | 5.34 ± 0.22 c | −21.17% | 1.68 ± 0.04 a |
| 6 | 4.45 ± 0.29 b | −34.22% | 1.74 ± 0.10 a |
| 7 | 3.68 ± 0.50 a | −45.69% | 1.67 ± 0.06 a |
| 8 | 3.43 ± 0.03 a | −49.29% | 1.66 ± 0.03 a |
| 9 | 3.64 ± 0.22 a | −46.28% | 1.72 ± 0.05 a |
| 10 | 3.21 ± 0.04 a | −52.63% | 1.70 ± 0.04 a |
| Growth Performance Parameters | Experimental Groups | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | Probiotic-Supplemented Diet (FD) | Water-Soluble Probiotics (SOL) | Combination of FD and SOL | |||||||
| FD5 | FD7 | FD9 | SOL3 | SOL4 | SOL5 | FD5 + SOL3 | FD7 + SOL3 | FD9 + SOL3 | ||
| 1. Body weight analysis | ||||||||||
| 1.1 AGR: WG (g /120 days) | 19.45 ± 0.57 a | 23.71 ± 1.85 a | 24.48 ± 1.24 a | 23.57 ± 5.48 a | 21.99 ± 4.13 a | 21.51 ± 4.06 a | 23.16 ± 4.17 a | 24.34 ± 1.26 a | 22.16 ± 0.81 a | 25.19 ± 0.19 b |
| 1.2 AGR: ADG (g/day) | 0.16 ± 0.00 a | 0.20 ± 0.02 a | 0.20 ± 0.01 a | 0.20 ± 0.05 a | 0.18 ± 0.03 a | 0.18 ± 0.03 a | 0.19 ± 0.03 a | 0.20 ± 0.01 a | 0.18 ± 0.01 a | 0.24 ± 0.00 b |
| 1.3 RGR (% in 120 days) | 177.37 ± 2.25 a | 205.23 ± 14.48 ab | 224.61 ± 0.56 ab | 209.72 ± 36.41 ab | 207.46 ± 45.14 ab | 195.17 ± 32.34 ab | 210.55 ± 35.08 ab | 219.20 ± 0.66 ab | 202.55 ± 12.83 ab | 240.53 ± 7.06 b |
| 1.4 SGR (%/day) | 0.369 ± 0.003 a | 0.404 ± 0.017 ab | 0.426 ± 0.001 ab | 0.408 ± 0.043 ab | 0.405 ± 0.053 ab | 0.391 ± 0.040 ab | 0.409 ± 0.041 ab | 0.420 ± 0.001 ab | 0.400 ± 0.015 ab | 0.443 ± 0.008 b |
| 1.5 FCR | 3.09 ± 0.09 a | 2.74 ± 0.20 a | 2.75 ± 0.12 a | 2.62 ± 0.6 a | 2.78 ± 0.52 a | 2.84 ± 0.54 a | 2.63 ± 0.47 a | 2.77 ± 0.13 a | 2.71 ± 0.10 a | 2.38 ± 0.02 b |
| 2. Body length analysis | ||||||||||
| 2.1 AGR: TLG (cm/120 days) | 12.65 ± 0.39 a | 10.78 ± 0.68 a | 10.09 ± 1.71 a | 12.57 ± 2.17 a | 11.13 ± 0.03 a | 10.82 ± 0.45 a | 14.16 ± 2.52 a | 12.55 ± 0.48 a | 11.71 ± 0.28 a | 12.00 ± 0.18 a |
| 2.2 AGR: ADG (cm/day) | 0.11 ± 0.00 a | 0.09 ± 0.01 a | 0.08 ± 0.01 a | 0.10 ± 0.02 a | 0.09 ± 0.00 a | 0.09 ± 0.00 a | 0.12 ± 0.02 a | 0.10 ± 0.00 a | 0.10 ± 0.00 a | 0.10 ± 0.00 a |
| 2.3 RGR (% in 120 days) | 169.04 ± 2.16 a | 159.21 ± 15.38 a | 160.03 ± 15.93 a | 163.78 ± 42.03 a | 1520.94 ± 5.61 a | 139.46 ± 8.98 a | 168.04 ± 37.60 a | 147.89 ± 16.04 a | 151.82 ± 9.92 a | 155.94 ± 2.05 a |
| 2.4 SGR (%/day) | 0.36 ± 0.00 a | 0.28 ± 0.03 a | 0.28 ± 0.03 a | 0.35 ± 0.06 a | 0.32 ± 0.01 a | 0.32 ± 0.01 a | 0.38 ± 0.05 a | 0.33 ± 0.02 a | 0.33 ± 0.01 a | 0.34 ± 0.00 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bunnoy, A.; Na-Nakorn, U.; Srisapoome, P. Probiotic Effects of a Novel Strain, Acinetobacter KU011TH, on the Growth Performance, Immune Responses, and Resistance against Aeromonas hydrophila of Bighead Catfish (Clarias macrocephalus Günther, 1864). Microorganisms 2019, 7, 613. https://doi.org/10.3390/microorganisms7120613
Bunnoy A, Na-Nakorn U, Srisapoome P. Probiotic Effects of a Novel Strain, Acinetobacter KU011TH, on the Growth Performance, Immune Responses, and Resistance against Aeromonas hydrophila of Bighead Catfish (Clarias macrocephalus Günther, 1864). Microorganisms. 2019; 7(12):613. https://doi.org/10.3390/microorganisms7120613
Chicago/Turabian StyleBunnoy, Anurak, Uthairat Na-Nakorn, and Prapansak Srisapoome. 2019. "Probiotic Effects of a Novel Strain, Acinetobacter KU011TH, on the Growth Performance, Immune Responses, and Resistance against Aeromonas hydrophila of Bighead Catfish (Clarias macrocephalus Günther, 1864)" Microorganisms 7, no. 12: 613. https://doi.org/10.3390/microorganisms7120613
APA StyleBunnoy, A., Na-Nakorn, U., & Srisapoome, P. (2019). Probiotic Effects of a Novel Strain, Acinetobacter KU011TH, on the Growth Performance, Immune Responses, and Resistance against Aeromonas hydrophila of Bighead Catfish (Clarias macrocephalus Günther, 1864). Microorganisms, 7(12), 613. https://doi.org/10.3390/microorganisms7120613







