Risk Comparison of the Diarrheal and Emetic Type of Bacillus cereus in Tofu
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains
2.2. Effect of Temperature on the Growth and Survival Characteristics of B. cereus in Tofu
2.3. Biofilm Formation
2.4. Transfer of Biofilm Cells on Stainless Steel Coupon to Tofu and Regrowth
2.5. Viability of the Transferred Biofilm Cells
2.6. Spore Formation
2.7. B. cereus Survival during Heat Treatment
2.8. Calculation of D-values
2.9. Statistical Analysis
3. Results and Discussion
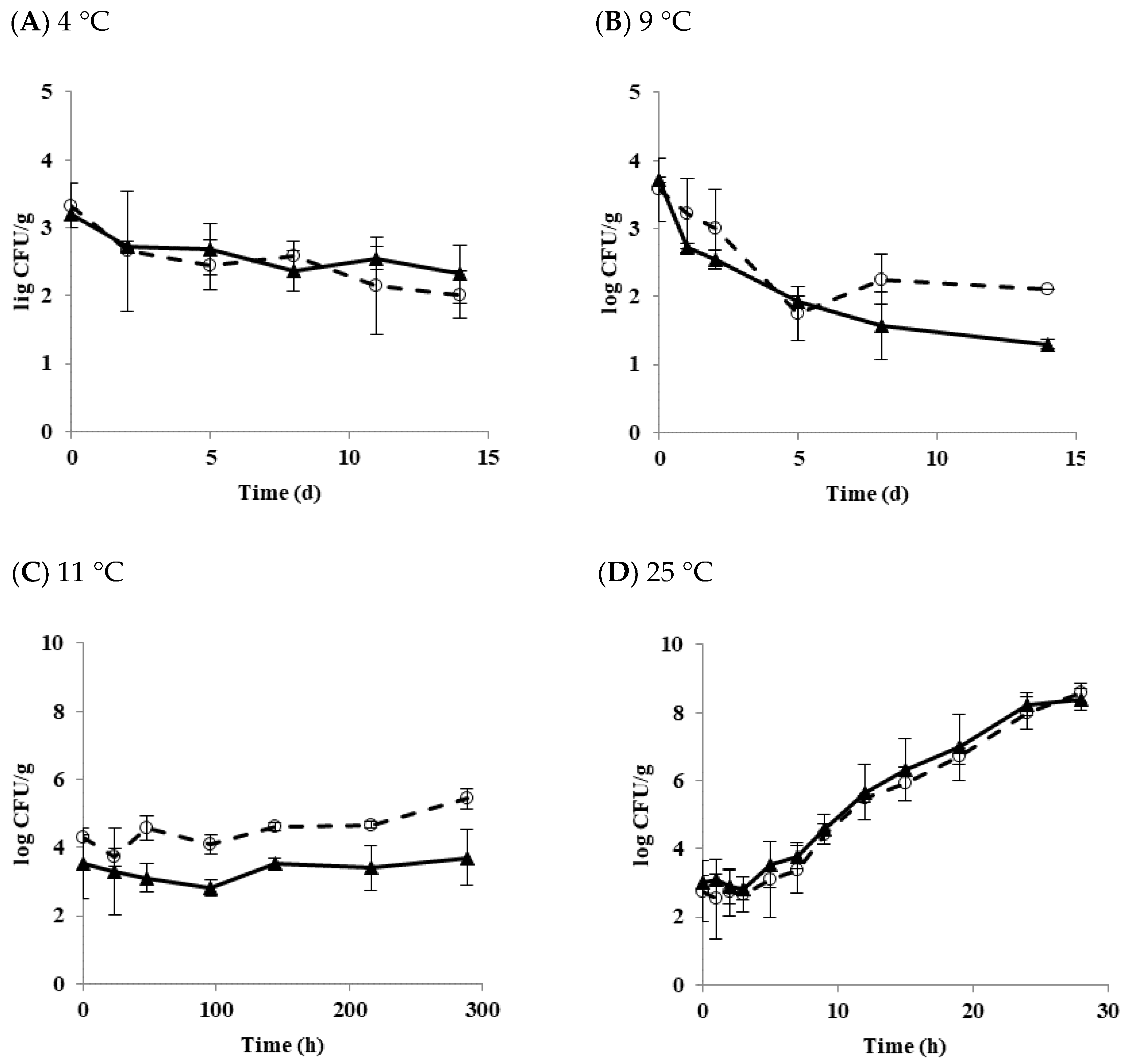
3.1. Effect of Temperature on the Growth and Survival Characteristics of B. cereus Planktonic Cells in Tofu
3.2. Biofilm Formation and Transfer of Biofilm Cells
3.3. Viability of Transferred Biofilm Cells
3.4. Growth of Transferred Biofilm Cells in Tofu
3.5. Effect of Temperature on Vegetable Cells and Spores of B. cereus and D Value in Tofu
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Préstamo, G.; Fontecha, J. High pressure treatment on the tofu fatty acids and acylglycerols content. Innov. Food Sci. Emerg. Technol. 2007, 8, 188–191. [Google Scholar] [CrossRef]
- Shin, I.S.; Han, J.S.; Choi, K.D.; Chung, D.H.; Choi, G.P.; Ahn, J.H. Effect of isothiocyanates from horseradish (Armoracia rusticana) on the quality and shelf life of tofu. Food Control. 2010, 21, 1081–1086. [Google Scholar] [CrossRef]
- Ananchaipattana, C.; Hosotani, Y.; Kawasaki, S.; Pongswat, S.; Latiful, B.M.; Isobe, S.; Inatsu, Y. Bacterial contamination of soybean curd (tofu) sold in Thailand. Food Sci. Technol. Res. 2012, 18, 843–848. [Google Scholar] [CrossRef][Green Version]
- Lee, D.Y.; Kwon, K.H.; Chai, C.H.; Oh, S.W. Microbial contamination of tofu in Korea and growth characteristics of Bacillus cereus isolates in Tofu. Lwt-Food Sci. Technol. 2017, 78, 63–69. [Google Scholar] [CrossRef]
- Sonenshein, A.L.; Hoch, J.A.; Losick, R. Bacillus Subtilis and Its Closest Relatives; ASM Press: Washinngton, DC, USA, 2002. [Google Scholar]
- Setlow, P. Spores of Bacillus subtilis: Their resistance to and killing by radiation, heat and chemicals. J. Appl. Microbiol. 2006, 101, 514–525. [Google Scholar] [CrossRef]
- Sarrıas, J.A.; Valero, M.; Salmeron, M.C. Elimination of Bacillus cereus contamination in raw rice by electron beam irradiation. Food Microbiol. 2003, 20, 327–332. [Google Scholar] [CrossRef]
- Hood, S.K.; Zottola, E.A. Biofilms in food processing. Food Control. 1995, 6, 9–18. [Google Scholar] [CrossRef]
- Lee, S.H.; Park, Y.S. Analysis of Microflora Profile in the Manufacturing Process of Commercial Tofu. Food Eng. Prog. 2012, 16, 270–277. [Google Scholar]
- Rossi, F.; Felis, G.E.; Martinelli, A.; Calcavecchia, B.; Torriani, S. Microbiological characteristics of fresh tofu produced in small industrial scale and identification of specific spoiling microorganisms (SSO). Lwt-Food Sci. Technol. 2016, 70, 280–285. [Google Scholar] [CrossRef]
- Oh, K.Y.; Ahn, S.C.; Oh, S.M. A study of shelf-life and antimicrobial activity on putrefactive microorganisms related to soybean curd of Persicaria hydropiper L. extracts. Culin. Sci. Hosp. Res. 2016, 22, 198–211. [Google Scholar]
- Park, K.N.; Park, L.Y.; Kim, D.G.; Park, G.S.; Lee, S.H. Effect of turmeric (Curcuma aromatica Salab.) on shelf life of tofu. Korean J. Food Preserv. 2007, 14, 136–141. [Google Scholar]
- Gibson, A.M.; Bratchell, N.; Roberts, T.A. The effect of sodium chloride and temperature on the rate and extent of growth of Clostridium botulinum type A in pasteurized pork slurry. J. Appl. Bacteriol. 1987, 62, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Geeraerd, A.H.; Valdramidis, V.P.; Van Impe, J.F. GInaFiT, a freeware tool to assess non-log-linear microbial survivor curves. Int. J. Food Microbiol. 2005, 102, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.R.; Kwon, M.J.; Yoon, K.S. Control of Listeria innocua biofilms on food contact surfaces with slightly acidic electrolyzed water and the risk of biofilm cells transfer to duck meat. J. Food Prot. 2018, 81, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Antoniou, K.; Frank, J.F. Removal of Pseudomonas putida biofilm and associated extracellular polymeric substances from stainless steel by alkali cleaning. J. Food Prot. 2005, 68, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Sadekuzzaman, M.; Yang, S.; Mizan, M.F.R.; Kim, H.S.; Ha, S.D. Effectiveness of a phage cocktail as a biocontrol agent against L. monocytogenes biofilms. Food Control. 2017, 78, 256–263. [Google Scholar] [CrossRef]
- Kreske, A.C.; Ryu, J.H.; Pettigrew, C.A.; Beuchat, L.R. Lethality of chlorine, chlorine dioxide, and a commercial produce sanitizer to Bacillus cereus and Pseudomonas in a liquid detergent, on stainless steel, and in biofilm. J. Food Prot. 2006, 69, 2621–2634. [Google Scholar] [CrossRef]
- Midelet, G.; Kobilinsky, A.; Carpentier, B. Construction and analysis of fractional multifactorial designs to study attachment strength and transfer of Listeria monocytogenes from pure or mixed biofilms after contact with a solid model food. Appl. Environ. Microbiol. 2006, 72, 2313–2321. [Google Scholar] [CrossRef]
- Montville, R.; Chen, Y.; Schaffner, D.W. Glove barriers to bacterial cross-contamination between hands to food. J. Food Prot. 2001, 64, 845–849. [Google Scholar] [CrossRef]
- Wang, W.; Tao, R.; Tong, Z.; Ding, Y.; Kuang, R.; Zhai, S.; Liu, J.; Ni, L. Effect of a novel antimicrobial peptide chrysophsin-1 on oral pathogens and Streptococcus mutans biofilms. Peptides 2012, 33, 212–219. [Google Scholar] [CrossRef]
- Mazas, M.; Gonzalez, I.; Lopez, M.; González, J.; Sarmiento, R.M. Effects of sporulation media and strain on thermal resistance of Bacillus cereus spores. Int. J. Food Sci. Tech. 1995, 30, 71–78. [Google Scholar] [CrossRef]
- Kim, N.H.; Koo, J.M.; Rhee, M.S. Thermal resistance characteristics of bacillus cereus, Escherichia coli O157: H7, and listeria monocytogenes in a multi-grain soy milk product. Korean J. Food Nutr. 2015, 47, 593–598. [Google Scholar]
- Albert, I.; Mafart, P. A modified Weibull model for bacterial inactivation. Int. J. Food Microbiol. 2005, 100, 197–211. [Google Scholar] [CrossRef] [PubMed]
- MFDS. Food Code, Preservation and Distribution Standards. Available online: http://www.foodsafetykorea.go.kr/foodcode/01_03.jsp?idx=13 (accessed on 2 May 2018).
- Kang, K.A.; Kim, Y.W.; Yoon, K.S. Development of predictive growth models for Staphylococcus aureus and Bacillus cereus on various food matrices consisting of ready-to-eat (RTE) foods. Food Sci. Anim. Resour. 2010, 30, 730–738. [Google Scholar] [CrossRef]
- Liu, J.G.; Lin, T.S.; Lin, W.Y. Evaluating the growth of Listeria monocytogenes that has been inoculated into tofu containing background microflora. Food Control. 2010, 21, 1764–1768. [Google Scholar] [CrossRef]
- Auger, S.; Ramarao, N.; Faille, C.; Fouet, A.; Aymerich, S.; Gohar, M. Biofilm formation and cell surface properties among pathogenic and nonpathogenic strains of the Bacillus cereus group. Appl. Environ. Microbiol. 2009, 75, 6616–6618. [Google Scholar] [CrossRef]
- Oliveira, R.; Melo, L.F.; Oliveira, A.; Salgueiro, R. Polysaccharide production and biofilm formation by Pseudomonas fluorescens: Effects of pH and surface material. Colloids Surf. B Biointerfaces 1994, 2, 41–46. [Google Scholar] [CrossRef]
- Pagedar, A.; Singh, J.; Batish, V.K. Surface hydrophobicity, nutritional contents affect Staphylococcus aureus biofilms and temperature influences its survival in preformed biofilms. J. Basic Microbiol. 2010, 50, 98–106. [Google Scholar] [CrossRef]
- Garrett, T.R.; Bhakoo, M.; Zhang, Z. Bacterial adhesion and biofilms on surfaces. Prog. Nat. Sci. 2008, 18, 1049–1056. [Google Scholar] [CrossRef]
- Jensen, D.A.; Friedrich, L.M.; Harris, L.J.; Danyluk, M.D.; Schaffner, D.W. Quantifying transfer rates of Salmonella and Escherichia coli O157: H7 between fresh-cut produce and common kitchen surfaces. J. Food Prot. 2013, 76, 1530–1538. [Google Scholar] [CrossRef]
- Rodriguez, A.; McLandsborough, L.A. Evaluation of the transfer of Listeria monocytogenes from stainless steel and high-density polyethylene to Bologna and American cheese. J. Food Prot. 2007, 70, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Toyofuku, M.; Inaba, T.; Kiyokawa, T.; Obana, N.; Yawata, Y.; Nomura, N. Environmental factors that shape biofilm formation. Biosci. Biotechnol. Biochem. 2016, 80, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.; Fratamico, P.M.; Uhlich, G. Molecular mechanisms involved in biofilm formation by food-associated bacteria. In Biofilms in the Food and Beverage Industries; Elsevier: Amsterdam, The Netherlands, 2009; pp. 42–98. [Google Scholar]
- Simoes, M.; Simoes, L.C.; Vieira, M.J. Species association increases biofilm resistance to chemical and mechanical treatments. Water Res. 2009, 43, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Hingston, P.A.; Stea, E.C.; Knøchel, S.; Hansen, T. Role of initial contamination levels, biofilm maturity and presence of salt and fat on desiccation survival of Listeria monocytogenes on stainless steel surfaces. Food Microbiol. 2013, 36, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Baweja, R.B.; Zaman, M.S.; Mattoo, A.R.; Sharma, K.; Tripathi, V.; Aggarwal, A.; Dubey, G.P.; Kurupati, R.K.; Ganguli, M.; Chaudhury, N.K. Properties of Bacillus anthracis spores prepared under various environmental conditions. Arch. Microbiol. 2008, 189, 71–79. [Google Scholar] [CrossRef]
- Byrne, B.; Dunne, G.; Bolton, D.J. Thermal inactivation of Bacillus cereus and Clostridium perfringens vegetative cells and spores in pork luncheon roll. Food Microbiol. 2006, 23, 803–808. [Google Scholar] [CrossRef]
- Kim, Y.S.; Ahn, Y.S.; Jeong, D.Y.; Shin, D.H. Isolation and identification of Bacillus cereus from fermented red pepper-soybean paste (kochujang), and its heat resistance characteristics. Food Sci. Biotechnol. 2008, 17, 123–129. [Google Scholar]
- Marquis, R.E.; Shin, S.Y. Mineralization and responses of bacterial spores to heat and oxidative agents. Fems Microbiol. Rev. 1994, 14, 375–379. [Google Scholar] [CrossRef]
- Oteiza, J.M.; Giannuzzi, L.; Califano, A.N. Thermal inactivation of Escherichia coli O157: H7 and Escherichia coli isolated from morcilla as affected by composition of the product. Food Res. Int. 2003, 36, 703–712. [Google Scholar] [CrossRef]
- Kim, G.W.; Kim, G.H.; Kim, J.S.; An, H.Y.; Hu, G.W.; Son, J.K.; Kim, O.S.; Cho, S.Y. Quality of tofu prepared with deep seawater as coagulant. Korean J. Fish. Aquat. Sci. 2008, 41, 77–83. [Google Scholar]
- Choma, C.; Guinebretiere, M.H.; Carlin, F.; Schmitt, P.; Velge, P.; Granum, P.E.; Nguyen-The, C. Prevalence, characterization and growth of Bacillus cereus in commercial cooked chilled foods containing vegetables. J. Appl. Microbiol. 2000, 88, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Casadei, M.A.; Ingram, R.; Hitchings, E.; Archer, J.; Gaze, J.E. Heat resistance of Bacillus cereus, Salmonella typhimurium and Lactobacillus delbrueckii in relation to pH and ethanol. Int. J. Food Microbiol. 2001, 63, 125–134. [Google Scholar] [CrossRef]
- Gaillard, S.; Leguérinel, I.; Mafart, P. Model for combined effects of temperature, pH and water activity on thermal inactivation of Bacillus cereus spores. J. Food Sci. 1998, 63, 887–889. [Google Scholar] [CrossRef]
- Humpheson, L.; Adams, M.R.; Anderson, W.A.; Cole, M.B. Biphasic thermal inactivation kinetics in Salmonella enteritidis PT4. Appl. Environ. Microbiol. 1998, 64, 459–464. [Google Scholar] [PubMed]
- Cho, Y.S.; Jung, E.Y.; Lee, M.K.; Yang, C.Y.; Shin, D.B. Survival, isolation and characterization of Bacillus cereus from Sunshik. J. Food Hyg. Saf. 2008, 23, 343–347. [Google Scholar]
- Desai, S.V.; Varadaraj, M.C. Behavioural pattern of vegetative cells and spores of Bacillus cereus as affected by time-temperature combinations used in processing of Indian traditional foods. J. Food Sci. Technol. 2010, 47, 549–556. [Google Scholar] [CrossRef][Green Version]
- MFDS. Tofu HACCP. Available online: http://www.mfds.go.kr/index.do?mid=1486&seq=11933&cmd=v (accessed on 15 May 2018).
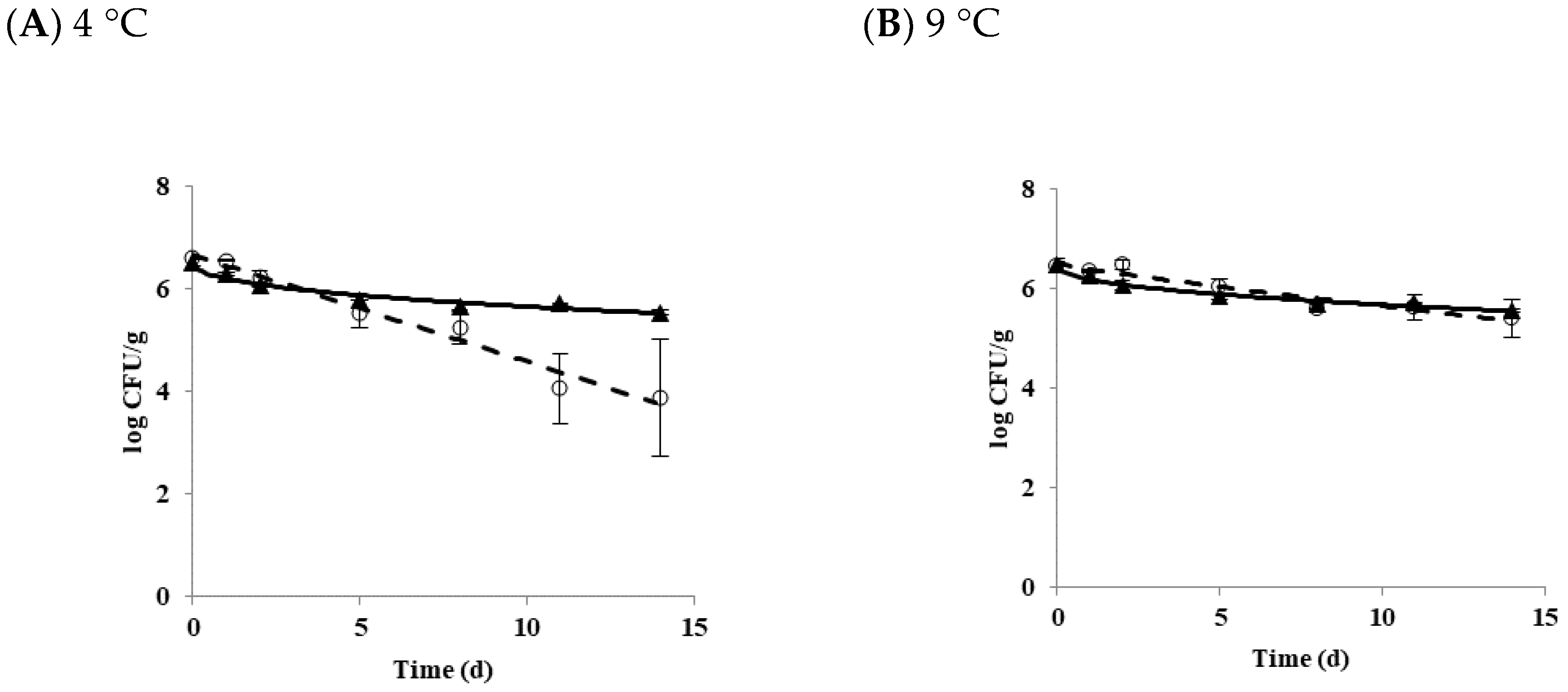
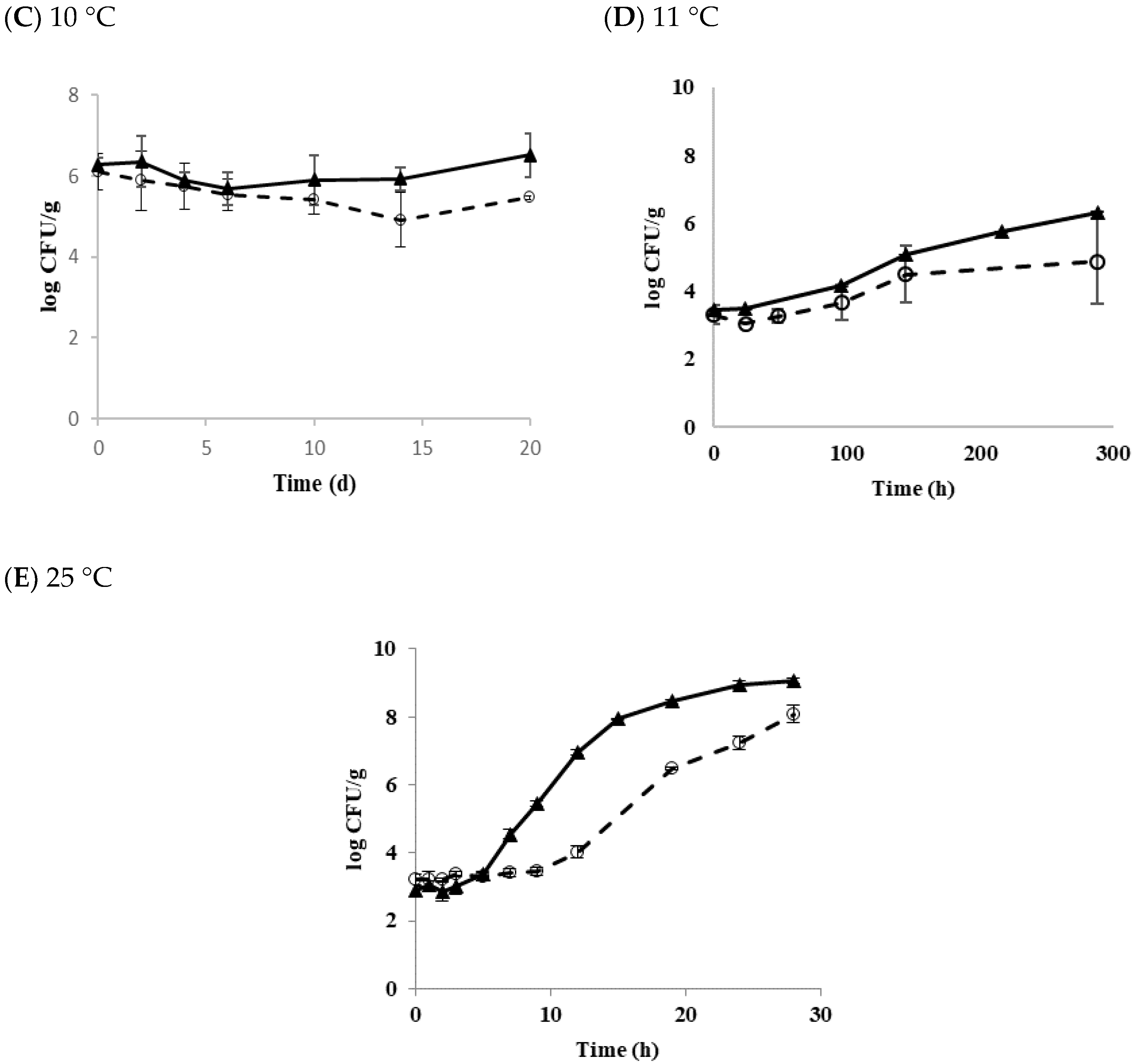
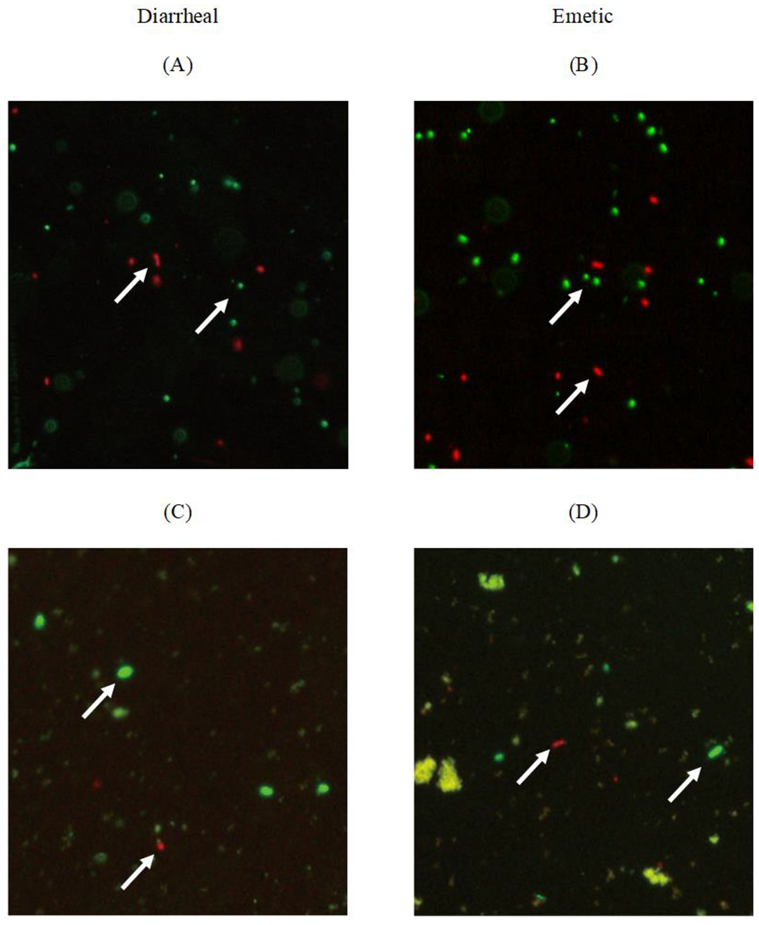
| Strain | Storage Temperature (°C) | Delta (d) | p | LT(h) | SGR (logCFU/h) |
|---|---|---|---|---|---|
| Diarrheal | 4 | 5.15 ± 1.08 * | 0.98 ± 0.02 | - | - |
| Emetic | 14.00 ± 2.69 | 0.43 ± 0.02 | |||
| Diarrheal | 9 | 12.88 ± 3.31 * | 0.88 ± 0.14 | - | - |
| Emetic | 22.59 ± 5.25 | 0.44 ± 0.05 | |||
| Diarrheal | 11 | - | - | 79.67 ± 8.43 * | 0.02 ± 0.00 |
| Emetic | 45.39 ± 8.87 | 0.02 ± 0.00 | |||
| Diarrheal | 25 | - | - | 9.53 ± 0.94 * | 0.31 ± 0.02 * |
| Emetic | 4.34 ± 0.21 | 0.55 ± 0.04 |
| Strain | Biofilm Formation (Log CFU/Coupon) | Efficiency of Transfer (EOT) |
|---|---|---|
| Diarrheal | 4.01 ± 0.51 | 0.72 ± 0.05 |
| Emetic | 4.00 ± 0.71 | 0.81 ± 0.12 |
| Strain Type | D-Values (min) | |||
|---|---|---|---|---|
| 80 °C | 85 °C | 90 °C | ||
| Diarrheal | spore | A 218.30 ± 6.36 b | B 139.83 ± 10.61 b | C 70.78 ± 6.25 a |
| vegetative cell | A 50.29 ± 6.24 c | A 38.84 ± 0.19 d | A 36.91 ± 4.39 c | |
| Emetic | spore | A 288.30 ± 14.28 a | B 260.67 ± 4.40 a | C 70.12 ± 2.47 a |
| vegetative cell | A 62.25 ± 17.11 c | A 56.52 ± 0.06 c | A 56.43 ± 5.49 b | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, M.J.; Lee, C.L.; Yoon, K.S. Risk Comparison of the Diarrheal and Emetic Type of Bacillus cereus in Tofu. Microorganisms 2019, 7, 536. https://doi.org/10.3390/microorganisms7110536
Kwon MJ, Lee CL, Yoon KS. Risk Comparison of the Diarrheal and Emetic Type of Bacillus cereus in Tofu. Microorganisms. 2019; 7(11):536. https://doi.org/10.3390/microorganisms7110536
Chicago/Turabian StyleKwon, Mi Jin, Chae Lim Lee, and Ki Sun Yoon. 2019. "Risk Comparison of the Diarrheal and Emetic Type of Bacillus cereus in Tofu" Microorganisms 7, no. 11: 536. https://doi.org/10.3390/microorganisms7110536
APA StyleKwon, M. J., Lee, C. L., & Yoon, K. S. (2019). Risk Comparison of the Diarrheal and Emetic Type of Bacillus cereus in Tofu. Microorganisms, 7(11), 536. https://doi.org/10.3390/microorganisms7110536





