Cows Get Crohn’s Disease and They’re Giving Us Diabetes
Abstract
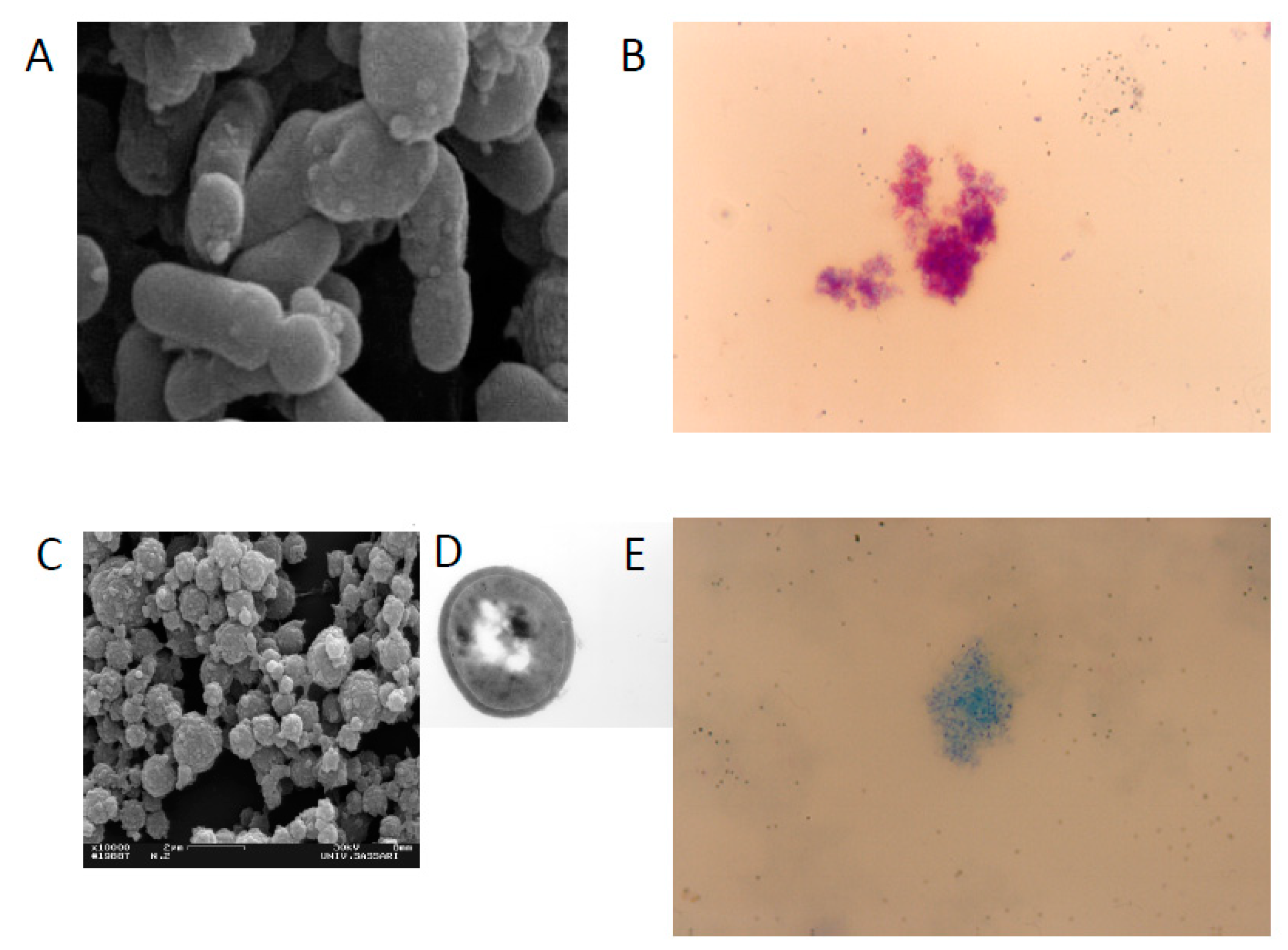
1. Introduction
2. MAP and Human Exposure
3. MAP and Crohn’s
4. MAP and Diabetes
5. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Agrawal, G.; Borody, T.J.; Chamberlin, W. ‘Global warming’ to Mycobacterium avium subspecies paratuberculosis. Future Microbiol. 2014, 9, 829–832. [Google Scholar] [CrossRef] [PubMed]
- Sechi, L.A.; Dow, C.T. Mycobacterium avium ss. paratuberculosis Zoonosis–The Hundred Year War–Beyond Crohn’s Disease. Front. Immunol. 2015, 6, 96. [Google Scholar] [CrossRef] [PubMed]
- Chamberlin, W.; Borody, W.T.; Naser, S. MAP-associated Crohn’s disease: MAP, Koch’s postulates, causality and Crohn’s disease. Digest. Liver Dis. 2007, 39, 792–794. [Google Scholar] [CrossRef] [PubMed]
- Hansen, R.; Thomson, J.M.; El-Omar, E.M.; Hold, G.L. The role of infection in the aetiology of inflammatory bowel disease. J. Gastroenterol. 2010, 45, 266–276. [Google Scholar] [CrossRef]
- Van Kruiningen, H.J.; Chiodini, R.J.; Thayer, W.R.; Coutu, J.A.; Merkal, R.S.; Runnels, P.L. Experimental disease in infant goats induced by a Mycobacterium isolated from a patient with Crohn’s disease. A preliminary report. Dig. Dis. Sci. 1986, 31, 1351–1360. [Google Scholar] [CrossRef]
- Balseiro, A.; Marín, J.G.; Solano, P.; Garrido, J.; Prieto, J. Histopathological Classification of Lesions Observed in Natural Cases of Paratuberculosis in Free-ranging Fallow Deer (Dama dama). J. Comp. Pathol. 2008, 138, 180–188. [Google Scholar] [CrossRef][Green Version]
- Vazquez, P.; Garrido, J.M.; Molina, E.; Geijo, M.V.; Gomez, N.; Pérez, V.; Sevilla, I.A.; Alonso-Hearn, M.; Cortes, A.; Juste, R.A. Latent infections are the most frequent form of paratuberculosis in slaughtered Friesian cattle. Span. J. Agric. Res. 2014, 12, 1049. [Google Scholar] [CrossRef]
- Perez, V.; Marín, J.G.; Badiola, J. Description and classification of different types of lesion associated with natural paratuberculosis infection in sheep. J. Comp. Pathol. 1996, 114, 107–122. [Google Scholar] [CrossRef]
- Corpa, J.; Garrido, J.; Marin, J.G.; Perez, V.; Arenas, J.M.C. Classification of Lesions Observed in Natural Cases of Paratuberculosis in Goats. J. Comp. Pathol. 2000, 122, 255–265. [Google Scholar] [CrossRef]
- González, J.; Geijo, M.; García-Pariente, C.; Verna, A.; Corpa, J.; Reyes, L.; Ferreras, M.; Juste, R.; Marín, J.G.; Perez, V.; et al. Histopathological Classification of Lesions associated with Natural Paratuberculosis Infection in Cattle. J. Comp. Pathol. 2005, 133, 184–196. [Google Scholar] [CrossRef]
- Corbett, E.L.; Watt, C.J.; Walker, N.; Maher, D.; Williams, B.G.; Raviglione, M.C.; Dye, C. The growing burden of tuberculosis: Global trends and interactions with the HIV epidemic. Arch. Intern. Med. 2003, 163, 1009–1021. [Google Scholar] [CrossRef] [PubMed]
- Hines, M.E.; Styer, E.L. Preliminary characterization of chemically generated Mycobacterium avium subsp. paratuberculosis cell wall deficient forms (spheroplasts). Vet. Microbiol. 2003, 95, 247–258. [Google Scholar] [CrossRef]
- Lamont, E.A.; Bannantine, J.P.; Armien, A.; Ariyakumar, D.S.; Sreevatsan, S. Identification and Characterization of a Spore-Like Morphotype in Chronically Starved Mycobacterium avium Subsp. Paratuberculosis Cultures. PLoS ONE 2012, 7, e30648. [Google Scholar] [CrossRef] [PubMed]
- Gaschignard, J.; Grant, A.V.; Thuc, N.V.; Orlova, M.; Cobat, A.; Huong, N.T.; Ba, N.N.; Thai, V.H.; Abel, L.; Schurr, E.; et al. Pauci- and Multibacillary Leprosy: Two Distinct, Genetically Neglected Diseases. PLoS Negl. Trop. Dis. 2016, 10, e0004345.26984977. [Google Scholar] [CrossRef] [PubMed]
- Dunn, J.J.; Starke, J.R.; Revell, P.A. Laboratory Diagnosis of Mycobacterium tuberculosis Infection and Disease in Children. J. Clin. Microbiol. 2016, 54, 1434–1441. [Google Scholar] [CrossRef]
- Hanna, S.E.; Connor, C.J.; Wang, H.H. Real-time polymerase chain reaction for the food microbiologist: Technologies, applications, and limitations. J. Food Sci. 2005, 70, 49–53. [Google Scholar] [CrossRef]
- Sung, J.S.; Chang, Y.-F.; Huang, C.; Zhu, J.; Huang, L.; Han, S.Y.; Shin, K.-S.; Stehman, S.; Shin, S.J.; Torres, A. Development of a Polymerase Chain Reaction Test to Confirm Mycobacterium Avium Subsp. Paratuberculosis in Culture. J. Veter Diagn. Investig. 2004, 16, 116–120. [Google Scholar] [CrossRef]
- Naser, S.; Ghobrial, G.; Romero, C.; Valentine, J. Culture of Mycobacterium avium subspecies paratuberculosis from the blood of patients with Crohn’s disease. Lancet 2004, 364, 1039–1044. [Google Scholar] [CrossRef]
- Naser, S.A.; Collins, M.T.; Crawford, J.T.; Valentine, J.F. Culture of Mycobacterium avium subspecies paratuberculosis (MAP) from the Blood of Patients with Crohn’s disease: A Follow-Up Blind Multi Center Investigation. Open Inflamm. J. 2009, 2, 22–23. [Google Scholar] [CrossRef]
- Sechi, L.A.; Scanu, A.M.; Molicotti, P.; Cannas, S.; Mura, M.; Dettori, G.; Fadda, G.; Zanetti, S. Detection and Isolation of Mycobacterium avium subspecies paratuberculosis from intestinal mucosal biopsies of patients with and without Crohn’s disease in Sardinia. Am. J. Gastroenterol. 2005, 100, 1529–1536. [Google Scholar] [CrossRef]
- Naser, S.A.; Schwartz, D.; Shafran, I. Isolation of Mycobacterium avium subsp paratuberculosis from breast milk of Crohn’s disease patients. Am. J. Gastroenterol. 2000, 95, 1094–1095. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, J.L.; San-Pedro, A.; Culebras, E.; Cíes, R.; Taxonera, C.; Lana, R.; Urcelay, E.; De La Torre, F.; Picazo, J.J.; Díaz-Rubio, M. High prevalence of viable Mycobacterium avium subspecies paratuberculosis in Crohn’s disease. World J. Gastroenterol. 2010, 16, 4558–4563. [Google Scholar] [CrossRef] [PubMed]
- Davis, W.; Madsen-Bouterse, S. Crohn’s disease and Mycobacterium avium subsp. paratuberculosis: The need for a study is long overdue. Vet. Immunol. Immunopathol. 2012, 145, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Dow, C.T.; Ellingson, J.L.E. Detection of Mycobacterium avium ss. Paratuberculosis in Blau Syndrome Tissues. Autoimmune Dis. 2010, 2010, 1–5. [Google Scholar] [CrossRef]
- Sechi, L.A.; Gazouli, M.; Ikonomopoulos, J.; Lukas, J.C.; Scanu, A.M.; Ahmed, N.; Fadda, G.; Zanetti, S. Mycobacterium avium subsp. paratuberculosis, genetic susceptibility to Crohn’s disease, and Sardinians: The way ahead. J. Clin. Microbiol. 2005, 43, 5275–5277. [Google Scholar] [CrossRef]
- Dow, C.T.M. paratuberculosis Heat Shock Protein 65 and Human Diseases: Bridging Infection and Autoimmunity. Autoimmune Dis. 2012, 2012, 1–6. [Google Scholar] [CrossRef]
- Wang, H.; Yuan, F.F.; Dai, Z.W.; Wang, B.; Ye, D.Q. Association between rheumatoid arthritis and genetic variants of natural resistance-associated macrophage protein 1 gene: A meta-analysis. Int. J. Rheum Dis. 2018, 21, 1651–1658. [Google Scholar] [CrossRef]
- Sechi, L.A.; Gazouli, M.; Sieswerda, L.E.; Molicotti, P.; Ahmed, N.; Ikonomopoulos, J.; Scanu, A.M.; Paccagnini, D.; Zanetti, S. Relationship between Crohn’s disease, infection withMycobacterium aviumsubspeciesparatuberculosisandSLC11A1gene polymorphisms in Sardinian patients. World J. Gastroenterol. 2006, 12, 7161–7164. [Google Scholar] [CrossRef]
- Dow, C.T.M. paratuberculosis and Parkinson’s disease—Is this a trigger. Med. Hypotheses 2014, 83, 709–712. [Google Scholar] [CrossRef]
- Härtlova, A.; Herbst, S.; Peltier, J.; Rodgers, A.; Bilkei-Gorzo, O.; Fearns, A.; Dill, B.D.; Lee, H.; Flynn, R.; Cowley, S.A.; et al. LRRK2 is a negative regulator of Mycobacterium tuberculosis phagosome maturation in macrophages. EMBO J. 2018, 37, e98694. [Google Scholar] [CrossRef]
- Sharp, R.C.; Beg, S.A.; Naser, S.A. Polymorphisms in Protein Tyrosine Phosphatase Non-receptor Type 2 and 22 (PTPN2/22) Are Linked to Hyper-Proliferative T-Cells and Susceptibility to Mycobacteria in Rheumatoid Arthritis. Front. Microbiol. 2018, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Dow, C.T. Paratuberculosis and Type I diabetes: Is this the trigger? Med. Hypotheses. 2006, 67, 782–785. [Google Scholar] [CrossRef] [PubMed]
- Cossu, D.; Masala, S.; Cocco, E.; Paccagnini, D.; Tranquilli, S.; Frau, J.; Marrosu, M.G.; Sechi, L.A. Association of Mycobacterium avium subsp. paratuberculosis and SLC11A1 polymorphisms in Sardinian multiple sclerosis patients. J. Infect. Dev. Ctries. 2013, 7, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Gong, L.; Liu, B.; Wang, J.; Pan, H.; Qi, A.; Zhang, S.; Wu, J.; Yang, P.; Wang, B. Novel missense mutation in PTPN22 in a Chinese pedigree with Hashimoto’s thyroiditis. BMC Endocr. Disord. 2018, 18, 76. [Google Scholar] [CrossRef] [PubMed]
- D’Amore, M.; Lisi, S.; Sisto, M.; Cucci, L.; Dow, C.T. Molecular identification of Mycobacterium avium subspecies paratuberculosis in an Italian patient with Hashimoto’s thyroiditis and Melkersson-Rosenthal syndrome. J. Med. Microbiol. 2010, 59, 137–139. [Google Scholar] [CrossRef]
- Sisto, M.; Cucci, L.; D’Amore, M.; Dow, T.C.; Mitolo, V.; Lisi, S. Proposing a relationship between Mycobacterium avium subspecies paratuberculosis infection and Hashimoto’s thyroiditis. Scand. J. Infect. Dis. 2010, 42, 787–790. [Google Scholar] [CrossRef]
- Arru, G.; Caggiu, E.; Paulus, K.; Sechi, G.P.; Mameli, G.; Sechi, L.A. Is there a role for Mycobacterium avium subspecies paratuberculosis in Parkinson’s disease? J. Neuroimmunol. 2016, 293, 86–90. [Google Scholar] [CrossRef]
- Bo, M.; Erre, G.L.; Niegowska, M.; Piras, M.; Taras, L.; Longu, M.G.; Passiu, G.; Sechi, L.A. Interferon regulatory factor 5 is a potential target of autoimmune response triggered by Epstein-barr virus and Mycobacterium avium subsp. paratuberculosis in rheumatoid arthritis, investigating a mechanism of molecular mimicry. Clin. Exp. Rheumatol. 2018, 36, 376–381. [Google Scholar]
- Dow, C.T. Detection of M. paratuberculosis Bacteremia in a Child with Lupus Erythematosus and Sjogren’s Syndrome. Autoimmun. Infect. Dis. 2016, 2. [Google Scholar] [CrossRef]
- Paccagnini, D.; Sieswerda, L.; Rosu, V.; Masala, S.; Pacifico, A.; Gazouli, M.; Ikonomopoulos, J.; Ahmed, N.; Zanetti, S.; Sechi, L.A. Linking Chronic Infection and Autoimmune Diseases: Mycobacterium avium Subspecies paratuberculosis, SLC11A1 Polymorphisms and Type-1 Diabetes Mellitus. PLoS ONE 2009, 4, e7109. [Google Scholar] [CrossRef]
- Lombard, J.E.; Gardner, I.A.; Jafarzadeh, S.R.; Fossler, C.P.; Harris, B.; Capsel, B.R. Herd-level prevalence of Mycobacterium avium subsp. paratuberculosis infection in United States dairy herds in 2007. Prev. Vet. Med. 2013, 108, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Millar, D.; Ford, J.; Sanderson, J.; Withey, S.; Tizard, M.; Doran, T.; Hermon-Taylor, J. IS900 PCR to detect Mycobacterium paratuberculosis in retail supplies of whole pasteurized cows’ milk in England and Wales. Appl. Environ. Microbiol. 1996, 62, 3446–3452. [Google Scholar] [PubMed]
- Ellingson, J.L.; Anderson, J.L.; Koziczkowski, J.J.; Radcliff, R.P.; Sloan, S.J.; Allen, S.E. Detection of viable Mycobacterium avium subsp. paratuberculosis in retail pasteurized whole milk by two culture methods and PCR. J. Food Prot. 2005, 68, 966–972. [Google Scholar] [CrossRef] [PubMed]
- Hruska, K.; Bartos, M.; Kralik, P.; Pavlik, I. Mycobacterium avium subsp. paratuberculosis in powdered infant milk: Paratuberculosis in cattle – the public health problem to be solved. Vet. Med. Czech. 2005, 50, 327–335. [Google Scholar] [CrossRef]
- Pickup, R.W.; Rhodes, G.; Arnott, S.; Sidi-Boumedine, K.; Bull, T.J.; Weightman, A. Mycobacterium avium subsp. paratuberculosis in the catchment area and water of the River Taff in South Wales, United Kingdom, and its potential relationship to clustering of Crohn’s disease cases in the city of Cardiff. Appl. Environ. Microbiol. 2003, 71, 2130–2139. [Google Scholar] [CrossRef]
- Whan, L.; Ball, H.J.; Grant, I.R.; Rowe, M.T. Occurrence of Mycobacterium avium subsp. paratuberculosis in untreated water in Northern Ireland. Appl. Environ. Microbiol. 2006, 71, 7107–7112. [Google Scholar] [CrossRef]
- Pickup, R.W.; Rhodes, G.; Bull, T.J.; Arnott, S.; Sidi-Boumedine, K.; Hurley, M.; Hermon-Taylor, J. Mycobacterium avium subsp. paratuberculosis in Lake Catchments, in River Water Abstracted for Domestic Use, and in Effluent from Domestic Sewage Treatment Works: Diverse Opportunities for Environmental Cycling and Human Exposure. Appl. Environ. Microbiol. 2006, 72, 4067–4077. [Google Scholar] [CrossRef]
- Richardson, H.; Rhodes, G.; Henrys, P.; Sedda, L.; Weightman, A.J.; Pickup, R.W. Presence of Mycobacterium avium Subspecies paratuberculosis Monitored Over Varying Temporal and Spatial Scales in River Catchments: Persistent Routes for Human Exposure. Microorganisms 2019, 7, 136. [Google Scholar] [CrossRef]
- Grewal, S.K.; Rajeev, S.; Sreevatsan, S.; Michel, F.C., Jr. Persistence of Mycobacterium avium subsp. paratuberculosis and other zoonotic pathogens during simulated composting, manure packing, and liquid storage of dairy manure. Appl. Environ. Microbiol. 2003, 72, 565–574. [Google Scholar] [CrossRef]
- Di Cerbo, A.; Murru, N.; Coppola, R.; Aponte, M. Persistence of bacterial indicators and zoonotic pathogens in contaminated cattle wastes. BMC Microbiol. 2016, 16, 87. [Google Scholar]
- Collins, M.T. International Handbook of Foodborne Pathogens; Marianne, D.M., Jeffrey, W., Eds.; Bier CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Beumer, A.; King, D.; Donohue, M.; Mistry, J.; Covert, T.; Pfaller, S. Detection of Mycobacterium avium subsp. paratuberculosis in drinking water and biofilms by quantitative PCR. Appl. Environ. Microb. 2010, 76, 7367–7370. [Google Scholar] [CrossRef]
- Falkinham, J.O., III. Factors influencing the chlorine susceptibility of Mycobacterium avium, Mycobacterium intracellulare, and Mycobacterium scrofulaceum. Appl. Environ. Microbiol. 2003, 69, 5685–5689. [Google Scholar] [CrossRef]
- Tatchou-Nyamsi-Konig, J.A.; Dailloux, M.; Block, J.C. Survival of Mycobacterium avium attached to polyethylene terephtalate (PET) water bottles. J. Appl. Microbiol. 2009, 106, 825–832. [Google Scholar] [CrossRef]
- Falkinham, J.O., III; Norton, C.D.; LeChevallier, M.W. Factors influencing numbers of Mycobacterium avium, Mycobacterium intracellulare, and other Mycobacteria in drinking water distribution systems. Appl. Environ. Microbiol. 2001, 67, 1225–1231. [Google Scholar] [CrossRef]
- Vaerewijck, M.J.; Huys, G.; Palomino, J.C.; Swings, J.; Portaels, F. Mycobacteria in drinking water distribution systems: Ecology and significance for human health. FEMS Microbiol. Rev. 2005, 29, 911–934. [Google Scholar] [CrossRef]
- Grant, I.; Foddai, A.; Kunkel, B.; Collins, M.T. Detection of Viable Mycobacterium avium subsp. paratuberculosis (MAP) in Infant Formula. In Proceedings of the 12th International Colloquium on Paratuberculosis, Parma, Italy, 22–26 June 2014. [Google Scholar]
- Davis, W.C.; Park, K.T. Progress Towards Control of a Mycobacterial Pathogen, Mycobacterium avium subsp. paratuberculosis, the Causative Agent of Johne’s Disease in Cattle and Humans. J. Food Hyg. Saf. 2018, 33, 221–228. [Google Scholar] [CrossRef]
- Balseiro, A.; Perez, V.; Juste, R.A. Chronic regional intestinal inflammatory disease: A trans-species slow infection? Comp. Immunol. Microbiol. Infect. Dis. 2019, 62, 88–100. [Google Scholar] [CrossRef]
- Davis, W.C.; Kuenstner, J.T.; Singh, S.V. Resolution of Crohn’s (Johne’s) disease with antibiotics: What are the next steps? Expert Rev. Gastroenterol. Hepatol. 2017, 21, 1–4. [Google Scholar] [CrossRef][Green Version]
- Chaubey, K.K.; Singh, S.V.; Gupta, S.; Singh, M.; Sohal, J.S.; Kumar, N.; Singh, M.K.; Bhatia, A.K.; Dhama, K. Mycobacterium avium subspecies paratuberculosis—An important food borne pathogen of high public health significance with special reference to India: An update. Vet. Q. 2017, 37, 282–299. [Google Scholar] [CrossRef]
- Kirkwood, C.D.; Wagner, J.; Boniface, K.; Vaughan, J.; Michalski, W.P.; Catto-Smith, A.G.; Cameron, D.J.; Bishop, R.F. Mycobacterium avium subspecies paratuberculosis in children with early-onset Crohn’s disease. Inflamm. Bowel. Dis. 2009, 15, 1643–1655. [Google Scholar] [CrossRef]
- Feller, M.; Huwiler, K.; Stephan, R.; Altpeter, E.; Shang, A.; Furrer, H.; Pfyffer, G.E.; Jemmi, T.; Baumgartner, A.; Egger, M. Mycobacterium avium subspecies paratuberculosis and Crohn’s disease: A systematic review and meta-analysis. Lancet Infect. Dis. 2007, 7, 607–613. [Google Scholar] [CrossRef]
- Abubakar, I.; Myhill, D.; Aliyu, S.H.; Hunter, P.R. Detection of Mycobacterium avium subspecies paratuberculosis from patients with Crohn’s disease using nucleic acid-based techniques: A systematic review and meta-analysis. Inflamm. Bowel. Dis. 2008, 14, 401–410. [Google Scholar] [CrossRef]
- McNees, A.L.; Markesich, D.; Zayyani, N.R.; Graham, D.Y. Mycobacterium paratuberculosis as a cause of Crohn’s disease. Expert. Rev. Gastroenterol. Hepatol. 2015, 9, 1523–1534. [Google Scholar] [CrossRef]
- Chamberlin, W.; Borody, T.J.; Campbell, J. Primary treatment of Crohn’s disease: Combined antibiotics taking center stage. Expert Rev. Clin. Immunol. 2011, 7, 751–760. [Google Scholar] [CrossRef]
- Open Label Efficacy and Safety of Anti-MAP (Mycobacterium avium subsp. paratuberculosis) Therapy in Adult Crohn’s Disease (MAPUS2). Available online: https://clinicaltrials.gov/ct2/show/NCT03009396 (accessed on 24 May 2019).
- Late Breaking Abstracts. Available online: https://journals.sagepub.com/doi/full/10.1177/2050640618812015#_i15 (accessed on 24 May 2019).
- Proof of Concept Study of RHB-104 as Add-On Therapy to Interferon Beta-1a in Relapsing Remitting Multiple Sclerosis (RRMS) (CEASE-MS). Available online: https://clinicaltrials.gov/ct2/show/NCT01717664 (accessed on 24 May 2019).
- Atkinson, M.A. The Pathogenesis and Natural History of Type 1 Diabetes. Cold Spring Harb. Perspect. Med. 2012, 2, a007641. [Google Scholar] [CrossRef]
- Gale, E.A. The rise of childhood type 1 diabetes in the 20th century. Diabetes 2002, 51, 3353–3361. [Google Scholar] [CrossRef]
- The 64 K question in diabetes. Lancet 1990, 336, 597–598. [CrossRef]
- Baekkeskov, S.; Nielsen, J.H.; Marner, B.; Bilde, T.; Ludvigsson, J.; Lernmark, Å. Autoantibodies in newly diagnosed diabetic children immunoprecipitate human pancreatic islet cell proteins. Nature 1982, 298, 167–169. [Google Scholar] [CrossRef]
- Jones, D.B.; Hunter, N.R.; Duff, G.W. Heat-shock protein 65 as a beta cell antigen of insulin-dependent diabetes. Lancet 1990, 336, 583. [Google Scholar] [CrossRef]
- Naser, S.A.; Thanigachalam, S.; Dow, C.T.; Collins, M.T. Exploring the role of Mycobacterium avium subspecies paratuberculosis in the pathogenesis of type 1 diabetes mellitus: A pilot study. Gut Pathog. 2013, 5, 14. [Google Scholar] [CrossRef]
- Scheinin, T.; Minh, N.-N.T.; Tuomi, T.; Miettinen, A.; Kontiainen, S. Islet cell and glutamic acid decarboxylase antibodies and heat-shock protein 65 responses in children with newly diagnosed insulin-dependent diabetes mellitus. Immunol. Lett. 1996, 49, 123–126. [Google Scholar] [CrossRef]
- Kennedy, D. International Efforts at Paratuberculosis Control. Veter Clin. N. Am. Food Anim. Pr. 2011, 27, 647–654. [Google Scholar] [CrossRef]
- Bakker, D.; Willemsen, P.; Van Zijderveld, F. Paratuberculosis recognized as a problem at last: A review. Veter Q. 2000, 22, 200–204. [Google Scholar] [CrossRef]
- Gerstein, H.C. Cow’s milk exposure and type I diabetes mellitus. A critical overview of the clinical literature. Diabetes Care 1994, 17, 13–19. [Google Scholar] [CrossRef]
- Writing Group for the TRIGR Study Group; Knip, M.; Åkerblom, H.K.; Al Taji, E.; Becker, D.; Bruining, J. Effect of Hydrolyzed Infant Formula vs Conventional Formula on Risk of Type 1 Diabetes: The TRIGR Randomized Clinical Trial. JAMA 2018, 319, 38–48. [Google Scholar]
- Sechi, L.A.; Rosu, V.; Pacifico, A.; Fadda, G.; Ahmed, N.; Zanetti, S. Humoral immune responses of type 1 diabetes patients to Mycobacterium avium subsp. paratuberculosis lend support to the infectious trigger hypothesis. Clin. Vaccine Immunol. 2008, 15, 320–326. [Google Scholar] [CrossRef]
- Sechi, L.A.; Paccagnini, D.; Salza, S.; Pacifico, A.; Ahmed, N.; Zanetti, S. Mycobacterium avium subspecies paratuberculosis bacteremia in type 1 diabetes mellitus: An infectious trigger? Clin. Infect. Dis. 2008, 46, 148–149. [Google Scholar] [CrossRef]
- Cossu, A.; Rosu, V.; Paccagnini, D.; Cossu, D.; Pacifico, A.; Sechi, L.A. MAP3738c and MptD are specific tags of Mycobacterium avium subsp. paratuberculosis infection in type I diabetes mellitus. Clin. Immunol. 2011, 141, 49–57. [Google Scholar] [CrossRef]
- Songini, M.; Mannu, C.; Targhetta, C.; Bruno, G. Type 1 diabetes in Sardinia: Facts and hypotheses in the context of worldwide epidemiological data. Acta Diabetol. 2017, 54, 9–17. [Google Scholar] [CrossRef]
- Rosu, V.; Ahmed, N.; Paccagnini, D.; Pacifico, A.; Zanetti, S.; Sechi, L.A. Mycobacterium avium subspecies paratuberculosis is not associated with Type-2 Diabetes Mellitus. Ann. Clin. Microbiol. Antimicrob. 2008, 7, 9. [Google Scholar] [CrossRef]
- Rosu, V.; Ahmed, N.; Paccagnini, D.; Gerlach, G.; Fadda, G.; Hasnain, S.E.; Zanetti, S.; Sechi, L.A. Specific Immunoassays Confirm Association of Mycobacterium avium Subsp. paratuberculosis with Type-1 but Not Type-2 Diabetes Mellitus. PLoS ONE 2009, 4, e4386. [Google Scholar] [CrossRef]
- Bitti, M.L.M.; Masala, S.; Capasso, F.; Rapini, N.; Piccinini, S.; Angelini, F.; Pierantozzi, A.; Lidano, R.; Pietrosanti, S.; Paccagnini, D.; et al. Mycobacterium avium subsp. paratuberculosis in an Italian Cohort of Type 1 Diabetes Pediatric Patients. Clin. Dev. Immunol. 2012, 2012, 1–5. [Google Scholar] [CrossRef][Green Version]
- Cossu, A.; Ferrannini, E.; Fallahi, P.; Antonelli, A.; Sechi, L.A. Antibodies recognizing specific Mycobacterium avium subsp. paratuberculosis’s MAP3738c protein in type 1 diabetes mellitus children are associated with serum Th1 (CXCL10) chemokine. Cytokine 2013, 61, 337–339. [Google Scholar] [CrossRef]
- Masala, S.; Zedda, M.A.; Cossu, D.; Ripoli, C.; Palermo, M.; Sechi, L.A. Zinc Transporter 8 and MAP3865c Homologous Epitopes are Recognized at T1D Onset in Sardinian Children. PLoS ONE 2013, 8, e63371. [Google Scholar] [CrossRef]
- Masala, S.; Paccagnini, D.; Cossu, D.; Brezar, V.; Pacifico, A.; Ahmed, N.; Mallone, R.; Sechi, L.A. Antibodies Recognizing Mycobacterium avium paratuberculosis Epitopes Cross-React with the Beta-Cell Antigen ZnT8 in Sardinian Type 1 Diabetic Patients. PLoS ONE 2011, 6, 26931. [Google Scholar] [CrossRef]
- Scotto, M.; Afonso, G.; Larger, E.; Raverdy, C.; Lemonnier, F.A.; Carel, J.C.; Dubois-Laforgue, D.; Baz, B.; Levy, D.; Gautier, J.F.; et al. Zinc transporter (ZnT)8(186–194) is an immunodominant CD8+ T cell epitope in HLA-A2+ type 1 diabetic patients. Diabetologia 2012, 55, 2026–2031. [Google Scholar] [CrossRef]
- Niegowska, M.; Paccagnini, D.; Mannu, C.; Targhetta, C.; Songini, M.; Sechi, L.A. Recognition of ZnT8, Proinsulin, and Homologous MAP Peptides in Sardinian Children at Risk of T1D Precedes Detection of Classical Islet Antibodies. J. Diabetes Res. 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Masala, S.; Cossu, D.; Piccinini, S.; Rapini, N.; Massimi, A.; Porzio, O.; Pietrosanti, S.; Lidano, R.; Bitti, M.L.M.; Sechi, L.A. Recognition of zinc transporter 8 and MAP3865c homologous epitopes by new-onset type 1 diabetes children from continental Italy. Acta Diabetol. 2014, 51, 577–585. [Google Scholar] [CrossRef]
- Masala, S.; Cossu, D.; Piccinini, S.; Rapini, N.; Mameli, G.; Bitti, M.L.M.; Sechi, L.A. Proinsulin and MAP3865c homologous epitopes are a target of antibody response in new-onset type 1 diabetes children from continental Italy. Pediatr. Diabetes 2015, 16, 189–195. [Google Scholar] [CrossRef]
- Greenig, M. HERVs, immunity, and autoimmunity: Understanding the connection. PeerJ 2019, 7, e6711. [Google Scholar] [CrossRef]
- Levet, S.; Medina, J.; Joanou, J.; Demolder, A.; Queruel, N.; Réant, K.; Normand, M.; Seffals, M.; Dimier, J.; Germi, R.; et al. An ancestral retroviral protein identified as a therapeutic target in type-1 diabetes. JCI Insight 2017, 2, 94387. [Google Scholar] [CrossRef]
- Niegowska, M.; Wajda-Cuszlag, M.; Stępień-Ptak, G.; Trojanek, J.; Michałkiewicz, J.; Szalecki, M.; Sechi, L.A. Anti-HERV-WEnv antibodies are correlated with seroreactivity against Mycobacterium avium subsp. paratuberculosis in children and youths at T1D risk. Sci. Rep. 2019, 9, 6282. [Google Scholar] [CrossRef] [PubMed]
- Rani, P.S.; Doddam, S.N.; Agrawal, S.; Hasnain, S.E.; Sechi, L.A.; Kumar, A.; Ahmed, N. Mycobacterium avium subsp. paratuberculosis is not discerned in diabetes mellitus patients in Hyderabad, India. Int. J. Med. Microbiol. 2014, 304, 620–625. [Google Scholar] [CrossRef] [PubMed]
- Kühtreiber, W.M.; Tran, L.; Kim, T.; Dybala, M.; Nguyen, B.; Plager, S.; Huang, D.; Janes, S.; DeFusco, A.; Baum, D.; et al. Long-term reduction in hyperglycemia in advanced type 1 diabetes: The value of induced aerobic glycolysis with BCG vaccinations. Npj Vaccines 2018, 3, 23. [Google Scholar] [CrossRef]
- Kühtreiber, W.M.; Faustman, D.L. BCG Therapy for Type 1 Diabetes: Restoration of Balanced Immunity and Metabolism. Trends Endocrinol. Metab. 2019, 30, 80–92. [Google Scholar] [CrossRef]
- Dow, C.T. BCG, Autoimmune Diabetes and M. Paratuberculosis. J. Diabetes Metab. Disord. 2018, 5, 24. [Google Scholar]
- Davis, W.C. On deaf ears, Mycobacterium avium paratuberculosis in pathogenesis Crohn’s and other diseases. World J. Gastroenterol. 2015, 21, 13411–13417. [Google Scholar] [CrossRef]
- Hermon-Taylor, J. Treatment with drugs active against Mycobacterium avium subspecies paratuberculosis can heal Crohn’s disease: More evidence for a neglected public health tragedy. Dig. Liver Dis. 2002, 34, 9–12. [Google Scholar] [CrossRef]
- Kuenstner, J.T.; Naser, S.; Chamberlin, W.; Borody, T.; Graham, D.Y.; McNees, A. The Consensus from the Mycobacterium avium ssp. paratuberculosis (MAP) Conference 2017. Front. Public Health 2017, 5, 208. [Google Scholar] [CrossRef]
- Bo, A.; Niegowska, M.; Erre Manchia, P.A.; Sechi, L.A.; Bo, M.; Arru, G.; Erre, G.L. Association between Lipoprotein Levels and Humoral Reactivity to Mycobacterium avium subsp. paratuberculosis in Multiple Sclerosis, Type 1 Diabetes Mellitus and Rheumatoid Arthritis. Microorganisms 2019, 7, 423. [Google Scholar] [CrossRef]
- Greenstein, R.J.; Su, L.; Juste, R.A.; Brown, S.T. On the Action of Cyclosporine A, Rapamycin and Tacrolimus on M. avium Including Subspecies paratuberculosis. PLoS ONE 2008, 3, e2496. [Google Scholar] [CrossRef] [PubMed]
- Greenstein, R.; Su, L.; Haroutunian, V.; Shahidi, A.; Brown, S. On the action of methotrexate and 6-Mercaptopurine on M. avium subspecies paratuberculosis. Inflamm. Bowel Dis. 2007, 13, 650. [Google Scholar]
- Greenstein, R.J.; Su, L.; Shahidi, A.; Brown, S.T. On the Action of 5-Amino-Salicylic Acid and Sulfapyridine on M. avium including Subspecies paratuberculosis. PLoS ONE 2007, 2, e516. [Google Scholar] [CrossRef] [PubMed]
- Qasem, A.; Naser, A.E.; Naser, S.A. The alternate effects of anti-TNFα therapeutics and their role in mycobacterial granulomatous infection in Crohn’s disease. Expert Rev. Anti-Infect. Ther. 2017, 15, 637–643. [Google Scholar] [CrossRef]
- Cao, B.L.; Qasem, A.; Sharp, R.C.; Abdelli, L.S.; Naser, S.A. Systematic review and meta-analysis on the association of tuberculosis in Crohn’s disease patients treated with tumor necrosis factor-α inhibitors (Anti-TNFα). World J. Gastroenterol. 2018, 24, 2764–2775. [Google Scholar] [CrossRef] [PubMed]
- Qasem, A.; Naser, S.A. TNFα inhibitors exacerbate Mycobacterium paratuberculosis infection in tissue culture: A rationale for poor response of patients with Crohn’s disease to current approved therapy. BMJ Open Gastroenterol. 2018, 5, e000216. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dow, C.T.; Sechi, L.A. Cows Get Crohn’s Disease and They’re Giving Us Diabetes. Microorganisms 2019, 7, 466. https://doi.org/10.3390/microorganisms7100466
Dow CT, Sechi LA. Cows Get Crohn’s Disease and They’re Giving Us Diabetes. Microorganisms. 2019; 7(10):466. https://doi.org/10.3390/microorganisms7100466
Chicago/Turabian StyleDow, Coad Thomas, and Leonardo A Sechi. 2019. "Cows Get Crohn’s Disease and They’re Giving Us Diabetes" Microorganisms 7, no. 10: 466. https://doi.org/10.3390/microorganisms7100466
APA StyleDow, C. T., & Sechi, L. A. (2019). Cows Get Crohn’s Disease and They’re Giving Us Diabetes. Microorganisms, 7(10), 466. https://doi.org/10.3390/microorganisms7100466





