Comparative Effects of Cigarette Smoke and Heated Tobacco Product Aerosols on Biofilm Production by Respiratory Pathogens
Abstract
1. Introduction
1.1. Biofilm Formation in the Pathogenesis of Acute and Chronic Lung Infections
1.2. Smoking and COPD
1.3. Conventional Cigarette Smoke Effect on Biofilm Formation by COPD and Pneumonia Associated Pathogens
1.4. Emergence of Heated Tobacco Products (HTPs) Such as iQOS™ and Glo™
1.5. HTP Substitution in COPD
1.6. Gaps in Knowledge About the Effect of Cigarette Smoke and HTPs on Biofilm Formation
1.7. Aim
2. Materials and Methods
2.1. Bacterial Strains
2.2. Preparation of Smoke and Vapor Extracts
2.3. Evaluating Biofilm Density: Biofilm Culture and Crystal Violet Staining
2.4. Evaluating Metabolic Activity of Planktonic Bacteria
2.5. Evaluating Biofilm Metabolic Activity
2.6. Fluorescent Staining and Microscopy
2.7. Statistical Analysis
3. Results
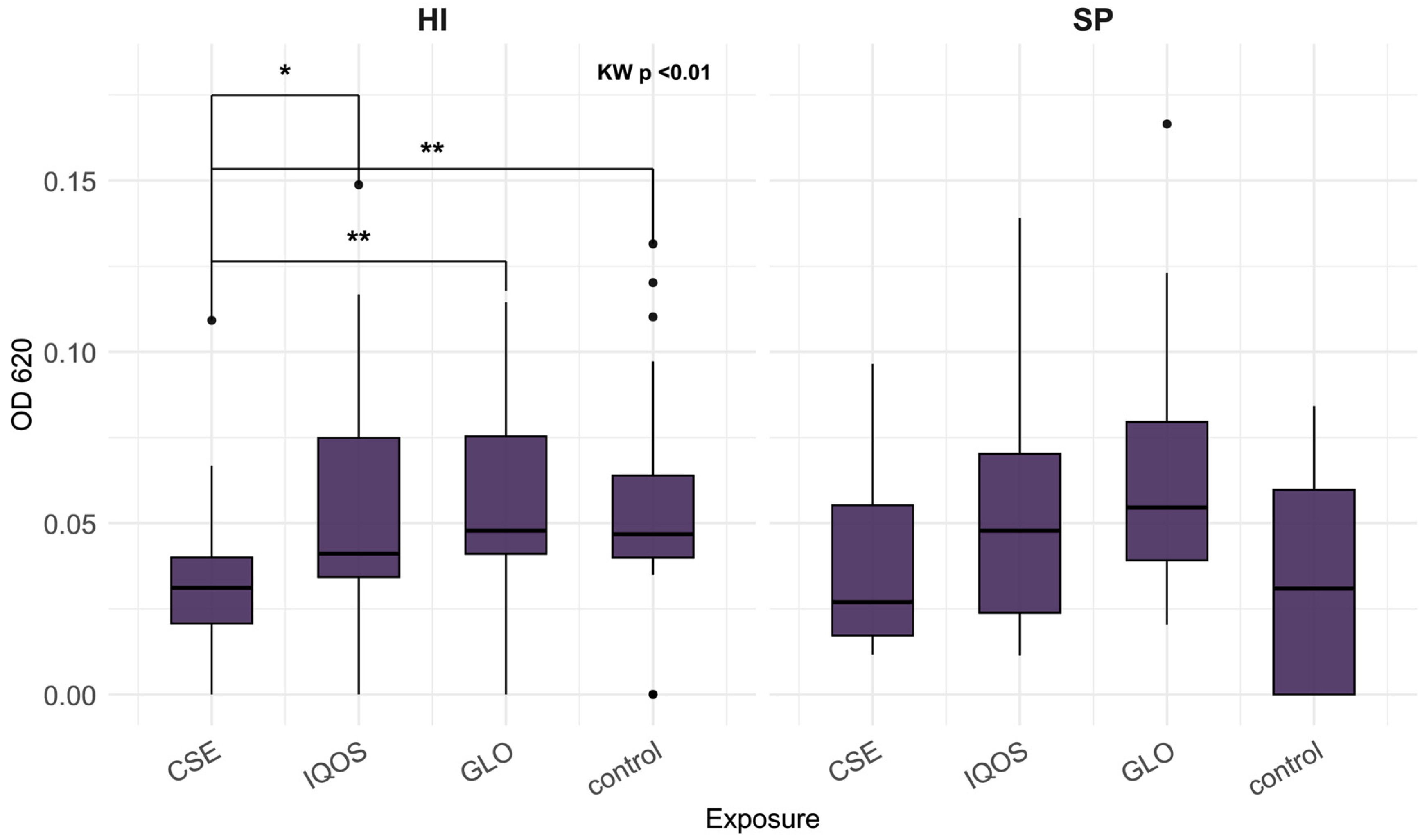
3.1. Effect of CSE and HTP Extract on Biofilm Density (Crystal Violet Assay)
3.2. Effect of CSE and HTP Extract on Bacterial Metabolic Activity (MTT Assay)
3.3. Effect of CSE and HTP Extract on Biofilm Metabolic Activity (Biofilm MTT Assay)
3.4. Biofilm Structure and Matrix Composition
4. Discussion
4.1. Biofilm Density
4.2. Biofilm Metabolic Activity
4.3. Growth and Metabolic Activity of Planktonic Cultures
4.4. Exposure to Tobbaco Extracts Modulates Biofilm Matrix Composition
4.5. Explanations for the Reduced HTP Effect
4.6. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ATCC | American Type Culture Collection |
| BHI | Brain Heart Infusion |
| CFU | Colony Forming Units |
| COPD | Chronic Obstructive Pulmonary Disease |
| ConA | Concanavalin A |
| CSC | Cigarette Smoke Condensate |
| CSE | Cigarette Smoke Extract |
| CV | Crystal Violet |
| DMSO | Dimethyl Sulfoxide |
| ECVE | Electronic Cigarette Vapor Extract |
| EPS | Extracellular Polysaccharides |
| FDR | False Discovery Rate |
| HPHC | Harmful and Potentially Harmful Constituent |
| HTP | Heated Tobacco Product |
| KW | Kruskal–Wallis |
| MIC | Minimum Inhibitory Concentration |
| MBC | Minimum Bactericidal Concentration |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NAD | Nicotinamide Adenine Dinucleotide |
| OD | Optical Density |
| PBS | Phosphate-Buffered Saline |
| PMMA | Polymethyl Methacrylate |
| RPM | Rotations Per Minute |
| sBHI | Supplemented Brain Heart Infusion (with hemin and β-NAD) |
| TSB | Tryptone Soy Broth |
| WGA | Wheat Germ Agglutinin |
| WHO | World Health Organization |
| HI | Haemophilus influenzae |
| KP | Klebsiella pneumoniae |
| PSD | Pseudomonas aeruginosa |
| SA | Staphylococcus aureus |
| SP | Streptococcus pneumoniae |
References
- Høiby, N.; Bjarnsholt, T.; Givskov, M.; Molin, S.; Ciofu, O. Antibiotic Resistance of Bacterial Biofilms. Int. J. Antimicrob. Agents 2010, 35, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.; Chan, R.; Lam, K.; Costerton, J.W. Production of Mucoid Microcolonies by Pseudomonas Aeruginosa within Infected Lungs in Cystic Fibrosis. Infect. Immun. 1980, 28, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Hassett, D.J.; Borchers, M.T.; Panos, R.J. Chronic Obstructive Pulmonary Disease (COPD): Evaluation from Clinical, Immunological and Bacterial Pathogenesis Perspectives. J. Microbiol. 2014, 52, 211–226. [Google Scholar] [CrossRef] [PubMed]
- Høiby, N.; Bjarnsholt, T.; Moser, C.; Jensen, P.Ø.; Kolpen, M.; Qvist, T.; Aanæs, K.; Pressler, T.; Skov, M.; Ciofu, O. Diagnosis of Biofilm Infections in Cystic Fibrosis Patients. APMIS 2017, 125, 339–343. [Google Scholar] [CrossRef]
- Kolpen, M.; Kragh, K.N.; Enciso, J.B.; Faurholt-Jepsen, D.; Lindegaard, B.; Egelund, G.B.; Jensen, A.V.; Ravn, P.; Mathiesen, I.H.M.; Gheorge, A.G.; et al. Bacterial Biofilms Predominate in Both Acute and Chronic Human Lung Infections. Thorax 2022, 77, 1015–1022. [Google Scholar] [CrossRef]
- Bjarnsholt, T.; Jensen, P.Ø.; Fiandaca, M.J.; Pedersen, J.; Hansen, C.R.; Andersen, C.B.; Pressler, T.; Givskov, M.; Høiby, N. Pseudomonas aeruginosa Biofilms in the Respiratory Tract of Cystic Fibrosis Patients: Pseudomonas aeruginosa Biofilms in the Cystic Fibrosis Lung. Pediatr. Pulmonol. 2009, 44, 547–558. [Google Scholar] [CrossRef]
- Boisvert, A.-A.; Cheng, M.P.; Sheppard, D.C.; Nguyen, D. Microbial Biofilms in Pulmonary and Critical Care Diseases. Ann. Am. Thorac. Soc. 2016, 13, 1615–1623. [Google Scholar] [CrossRef]
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2024 Report); Global Initiative for Chronic Obstructive Lung Disease: Deer Park, IL, USA, 2024. [Google Scholar]
- Kohansal, R.; Martinez-Camblor, P.; Agustí, A.; Buist, A.S.; Mannino, D.M.; Soriano, J.B. The Natural History of Chronic Airflow Obstruction Revisited. Am. J. Respir. Crit. Care Med. 2009, 180, 3–10. [Google Scholar] [CrossRef]
- Celli, B.R.; Christenson, S.A.; Rabe, K.F.; Han, M.K.; van den Berge, M.; Criner, G.J.; Soler, X.; Djandji, M.; Radwan, A.; Rowe, P.J.; et al. Current Smoker: A Clinical Chronic Obstructive Pulmonary Disease Phenotype Affecting Disease Progression and Response to Therapy. Am. J. Respir. Crit. Care Med. 2025, 211, 729–736. [Google Scholar] [CrossRef]
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2025 Report); Global Initiative for Chronic Obstructive Lung Disease: Deer Park, IL, USA, 2025; Available online: https://goldcopd.org/2025-gold-report/ (accessed on 20 September 2025).
- Hunter, L.C.; Lee, R.J.; Butcher, I.; Weir, C.J.; Fischbacher, C.M.; McAllister, D.; Wild, S.H.; Hewitt, N.; Hardie, R.M. Patient Characteristics Associated with Risk of First Hospital Admission and Readmission for Acute Exacerbation of Chronic Obstructive Pulmonary Disease (COPD) Following Primary Care COPD Diagnosis: A Cohort Study Using Linked Electronic Patient Records. BMJ Open 2016, 6, e009121. [Google Scholar] [CrossRef]
- Moghoofei, M.; Azimzadeh Jamalkandi, S.; Moein, M.; Salimian, J.; Ahmadi, A. Bacterial Infections in Acute Exacerbation of Chronic Obstructive Pulmonary Disease: A Systematic Review and Meta-Analysis. Infection 2020, 48, 19–35. [Google Scholar] [CrossRef]
- Messous, S.; Elargoubi, A.; Pillet, S.; Rajoharison, A.; Hoffmann, J.; Trabelsi, I.; Grissa, M.H.; Boukef, R.; Beltaief, K.; Mastouri, M.; et al. Bacterial and Viral Infection in Patients Hospitalized for Acute Exacerbation of Chronic Obstructive Pulmonary Disease: Implication for Antimicrobial Management and Clinical Outcome. COPD J. Chronic Obstr. Pulm. Dis. 2021, 18, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, R.; Caskey, J.; Singh, S.K.; Paudel, S.; Baral, P.; Schexnayder, M.; Kim, J.; Kim, N.; Kosmider, B.; Ratner, A.J.; et al. Cigarette Smoke Extract–Exposed Methicillin-Resistant Staphylococcus Aureus Regulates Leukocyte Function for Pulmonary Persistence. Am. J. Respir. Cell Mol. Biol. 2016, 55, 586. [Google Scholar] [CrossRef] [PubMed]
- Goldstein-Daruech, N.; Cope, E.K.; Zhao, K.-Q.; Vukovic, K.; Kofonow, J.M.; Doghramji, L.; González, B.; Chiu, A.G.; Kennedy, D.W.; Palmer, J.N.; et al. Tobacco Smoke Mediated Induction of Sinonasal Microbial Biofilms. PLoS ONE 2011, 6, e15700. [Google Scholar] [CrossRef] [PubMed]
- Cockeran, R.; Herbert, J.A.; Mitchell, T.J.; Dix-Peek, T.; Dickens, C.; Anderson, R.; Feldman, C. Exposure of a 23F Serotype Strain of Streptococcus Pneumoniae to Cigarette Smoke Condensate Is Associated with Selective Upregulation of Genes Encoding the Two-Component Regulatory System 11 (TCS11). BioMed Res. Int. 2014, 2014, 976347. [Google Scholar] [CrossRef]
- Mutepe, N.D.; Cockeran, R.; Steel, H.C.; Theron, A.J.; Mitchell, T.J.; Feldman, C.; Anderson, R. Effects of Cigarette Smoke Condensate on Pneumococcal Biofilm Formation and Pneumolysin. Eur. Respir. J. 2013, 41, 392–395. [Google Scholar] [CrossRef]
- Kulkarni, R.; Antala, S.; Wang, A.; Amaral, F.E.; Rampersaud, R.; Larussa, S.J.; Planet, P.J.; Ratner, A.J. Cigarette Smoke Increases Staphylococcus Aureus Biofilm Formation via Oxidative Stress. Infect. Immun. 2012, 80, 3804–3811. [Google Scholar] [CrossRef]
- McEachern, E.K.; Hwang, J.H.; Sladewski, K.M.; Nicatia, S.; Dewitz, C.; Mathew, D.P.; Nizet, V.; Crotty Alexander, L.E. Analysis of the Effects of Cigarette Smoke on Staphylococcal Virulence Phenotypes. Infect. Immun. 2015, 83, 2443–2452. [Google Scholar] [CrossRef]
- Antunes, M.B.; Chi, J.J.; Liu, Z.; Goldstein-Daruech, N.; Palmer, J.N.; Zhu, J.; Cohen, N.A. Molecular Basis of Tobacco-Induced Bacterial Biofilms: An In Vitro Study. Otolaryngol. Neck Surg. 2012, 147, 876–884. [Google Scholar] [CrossRef]
- Gilpin, D.F.; McGown, K.-A.; Gallagher, K.; Bengoechea, J.; Dumigan, A.; Einarsson, G.; Elborn, J.S.; Tunney, M.M. Electronic Cigarette Vapor Increases Virulence and Inflammatory Potential of Respiratory Pathogens. Respir. Res. 2019, 20, 267. [Google Scholar] [CrossRef]
- Davis, B.; Williams, M.; Talbot, P. iQOS: Evidence of Pyrolysis and Release of a Toxicant from Plastic. Tob. Control 2019, 28, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Auer, R.; Concha-Lozano, N.; Jacot-Sadowski, I.; Cornuz, J.; Berthet, A. Heat-Not-Burn Tobacco Cigarettes: Smoke by Any Other Name. JAMA Intern. Med. 2017, 177, 1050–1052. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Study Group on Tobacco Product Regulation: Report on the Scientific Basis of Tobacco Product Regulation: Eighth Report of a WHO Study Group; WHO Technical Report Series; World Health Organization: Geneva, Switzerland, 2021; Available online: https://www.who.int/publications/i/item/9789240022720 (accessed on 26 September 2025).
- Smith, M.R.; Clark, B.; Lüdicke, F.; Schaller, J.-P.; Vanscheeuwijck, P.; Hoeng, J.; Peitsch, M.C. Evaluation of the Tobacco Heating System 2.2. Part 1: Description of the System and the Scientific Assessment Program. Regul. Toxicol. Pharmacol. 2016, 81, S17–S26. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.T.-H.; Grigg, J.; Filippidis, F.T. European Respiratory Society Statement on Novel Nicotine and Tobacco Products, Their Role in Tobacco Control and “Harm Reduction”. Eur. Respir. J. 2024, 63, 2301808. [Google Scholar] [CrossRef]
- Tattan-Birch, H.; Hartmann-Boyce, J.; Kock, L.; Simonavicius, E.; Brose, L.; Jackson, S.; Shahab, L.; Brown, J. Heated Tobacco Products for Smoking Cessation and Reducing Smoking Prevalence. Cochrane Database Syst. Rev. 2022, 1, CD013790. [Google Scholar] [CrossRef]
- World Health Organization. Heated Tobacco Products: A Brief; World Health Organization: Geneva, Switzerland, 2020; p. 11. Available online: https://www.who.int/europe/publications/i/item/WHO-EURO-2020-4571-44334-62636 (accessed on 16 September 2024).
- Sun, T.; Anandan, A.; Lim, C.C.W.; East, K.; Xu, S.S.; Quah, A.C.K.; Rutherford, B.N.; Johnson, B.; Qi, Y.; Stjepanovic, D.; et al. Global Prevalence of Heated Tobacco Product Use, 2015–2022: A Systematic Review and Meta-Analysis. Addiction 2023, 118, 1430–1444. [Google Scholar] [CrossRef]
- Polosa, R.; Morjaria, J.B.; Prosperini, U.; Busà, B.; Pennisi, A.; Gussoni, G.; Rust, S.; Maglia, M.; Caponnetto, P. Health Outcomes in COPD Smokers Using Heated Tobacco Products: A 3-Year Follow-Up. Intern. Emerg. Med. 2021, 16, 687–696. [Google Scholar] [CrossRef]
- Adamson, J.; Kanitscheider, C.; Prasad, K.; Camacho, O.M.; Beyerlein, E.; Bhagavan, Y.K.; Proctor, C.; Murphy, J. Results from a 2018 Cross-Sectional Survey in Tokyo, Osaka and Sendai to Assess Tobacco and Nicotine Product Usage after the Introduction of Heated Tobacco Products (HTPs) in Japan. Harm. Reduct. J. 2020, 17, 32. [Google Scholar] [CrossRef]
- Høiby, N.; Bjarnsholt, T.; Moser, C.; Bassi, G.L.; Coenye, T.; Donelli, G.; Hall-Stoodley, L.; Holá, V.; Imbert, C.; Kirketerp-Møller, K.; et al. ESCMID∗ Guideline for the Diagnosis and Treatment of Biofilm Infections 2014. Clin. Microbiol. Infect. 2015, 21, S1–S25. [Google Scholar] [CrossRef]
- Comer, D.M.; Elborn, J.S.; Ennis, M. Comparison of Nasal and Bronchial Epithelial Cells Obtained from Patients with COPD. PLoS ONE 2012, 7, e32924. [Google Scholar] [CrossRef]
- Stepanović, S.; Vuković, D.; Hola, V.; Bonaventura, G.D.; Djukić, S.; Ćirković, I.; Ruzicka, F. Quantification of Biofilm in Microtiter Plates: Overview of Testing Conditions and Practical Recommendations for Assessment of Biofilm Production by Staphylococci. APMIS 2007, 115, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing, version 4.4.2; R foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Posit Team. RStudio: Integrated Development Environment for R, version 2025.05.01+513; Posit Software, PBC: Boston, MA, USA, 2025. [Google Scholar]
- Bagale, K.; Kulkarni, R. A Systematic Review of the Literature Examining the Effects of Cigarette Smoke and E-Cigarette Vapor on the Virulence of Human Pathogenic Bacteria. Int. J. Environ. Res. Public Health 2022, 19, 12518. [Google Scholar] [CrossRef] [PubMed]
- Hunt, B.C.; Stanford, D.; Xu, X.; Li, J.; Gaggar, A.; Rowe, S.M.; Raju, S.V.; Swords, W.E. Haemophilus Influenzae Persists in Biofilm Communities in a Smoke-Exposed Ferret Model of COPD. ERJ Open Res. 2020, 6, 00200–02020. [Google Scholar] [CrossRef]
- Lacoma, A.; Edwards, A.M.; Young, B.C.; Domínguez, J.; Prat, C.; Laabei, M. Cigarette Smoke Exposure Redirects Staphylococcus Aureus to a Virulence Profile Associated with Persistent Infection. Sci. Rep. 2019, 9, 10798. [Google Scholar] [CrossRef]
- Shi, L.; Wu, Y.; Yang, C.; Ma, Y.; Zhang, Q.; Huang, W.; Zhu, X.; Yan, Y.; Wang, J.; Zhu, T.; et al. Effect of Nicotine on Staphylococcus Aureus Biofilm Formation and Virulence Factors. Sci. Rep. 2019, 9, 20243. [Google Scholar] [CrossRef]
- Cockeran, R.; Dix-Peek, T.; Dickens, C.; Steel, H.C.; Anderson, R.; Feldman, C. Biofilm Formation and Induction of Stress Response Genes Is a Common Response of Several Serotypes of the Pneumococcus to Cigarette Smoke Condensate. J. Infect. 2020, 80, 204–209. [Google Scholar] [CrossRef]
- Manna, S.; Waring, A.; Papanicolaou, A.; Hall, N.E.; Bozinovski, S.; Dunne, E.M.; Satzke, C. The Transcriptomic Response of Streptococcus Pneumoniae Following Exposure to Cigarette Smoke Extract. Sci. Rep. 2018, 8, 15716. [Google Scholar] [CrossRef]
- Cholo, M.C.; Rasehlo, S.S.M.; Venter, E.; Venter, C.; Anderson, R. Effects of Cigarette Smoke Condensate on Growth and Biofilm Formation by Mycobacterium Tuberculosis. BioMed Res. Int. 2020, 2020, 8237402. [Google Scholar] [CrossRef]
- Chusri, S.; Sompetch, K.; Mukdee, S.; Jansrisewangwong, S.; Srichai, T.; Maneenoon, K.; Limsuwan, S.; Voravuthikunchai, S.P. Inhibition of Staphylococcus epidermidis Biofilm Formation by Traditional Thai Herbal Recipes Used for Wound Treatment. Evid. Based Complement. Alternat. Med. 2012, 2012, 159797. [Google Scholar] [CrossRef]
- He, Z.; Wang, Q.; Hu, Y.; Liang, J.; Jiang, Y.; Ma, R.; Tang, Z.; Huang, Z. Use of the Quorum Sensing Inhibitor Furanone C-30 to Interfere with Biofilm Formation by Streptococcus Mutans and Its luxS Mutant Strain. Int. J. Antimicrob. Agents 2012, 40, 30–35. [Google Scholar] [CrossRef]
- He, Z.; Huang, Z.; Zhou, W.; Tang, Z.; Ma, R.; Liang, J. Anti-Biofilm Activities from Resveratrol against Fusobacterium Nucleatum. Front. Microbiol. 2016, 7, 1065. [Google Scholar] [CrossRef] [PubMed]
- McGown, K.; Tunney, M.M.; McGrath, S.J.; Elborn, J.S.; Gilpin, D.F. S22 The Effect Of Cigarette Smoke On Important Pathogens In Copd Lung Infection. Thorax 2014, 69, A14. [Google Scholar] [CrossRef]
- Furnari, S.; Emma, R.; Caruso, M.; Furneri, P.M.; Fuochi, V. Evaluating the Risks of Heated Tobacco Products: Toxicological Effects on Two Selected Respiratory Bacteria and Human Lung Cells. Toxics 2025, 13, 70. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Li, M.; Ye, M.; Yang, K.; Xu, X.; Gregory, R.L. Effects of Nicotine on Streptococcus Gordonii Growth, Biofilm Formation, and Cell Aggregation. Appl. Environ. Microbiol. 2014, 80, 7212. [Google Scholar] [CrossRef]
- Han, Y. Effects of Cigarette Smoking on the Growth of Streptococcus Mutans Biofilms: An in Vitro Study. PLoS ONE 2021, 16, e0259895. [Google Scholar] [CrossRef]
- Upadhyay, S.; Rahman, M.; Johanson, G.; Palmberg, L.; Ganguly, K. Heated Tobacco Products: Insights into Composition and Toxicity. Toxics 2023, 11, 667. [Google Scholar] [CrossRef]
- Keyser, B.M.; Leverette, R.; McRae, R.; Wertman, J.; Shutsky, T.; Jordan, K.; Szeliga, K.; Makena, P. In Vitro Toxicological Evaluation of Glo Menthol and Non-Menthol Heated Tobacco Products. Toxicology 2024, 504, 153801. [Google Scholar] [CrossRef]
- El Shafei, S.F.; Amin, A.H.; Abdelghaffar, E.G.; Moataz, S.; Makkeyah, F.; Shamel, M.; Al Ankily, M. The Effect of Cigarette Smoking and Heated Tobacco Products on Different Denture Materials; an in Vitro Study. BMC Oral Health 2025, 25, 179. [Google Scholar] [CrossRef]
- Mišković, I.; Kuiš, D.; Špalj, S.; Pupovac, A.; Mohar-Vitezić, B.; Prpić, J. Does Exposure to Burning and Heated Tobacco Affect the Abundance of Perio-Pathogenic Species in the Subgingival Biofilm? Appl. Sci. 2024, 14, 4824. [Google Scholar] [CrossRef]
- Philip Morris Trading SRL. HEETS Își Ia Rămas Bun. Available online: https://www.iqos.ro/old-categories/iqos-originals-duo/heets-iqos-duo (accessed on 21 September 2025).
- Karimi, K.; Zarei, O.; Sedighi, P.; Taheri, M.; Doosti-Irani, A.; Shokoohizadeh, L. Investigation of Antibiotic Resistance and Biofilm Formation in Clinical Isolates of Klebsiella Pneumoniae. Int. J. Microbiol. 2021, 2021, 5573388. [Google Scholar] [CrossRef]
- Seifi, K.; Kazemian, H.; Heidari, H.; Rezagholizadeh, F.; Saee, Y.; Shirvani, F.; Houri, H. Evaluation of Biofilm Formation Among Klebsiella Pneumoniae Isolates and Molecular Characterization by ERIC-PCR. Jundishapur J. Microbiol. 2016, 9, e30682. [Google Scholar] [CrossRef]
- Rodgman, A.; Perfetti, T. The Chemical Components of Tobacco and Tobacco Smoke, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2013; ISBN 978-1-4665-1548-2. [Google Scholar]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schiopu, P.; Toc, D.A.; Colosi, I.A.; Costache, C.; Panaitescu, P.-Ș.; Neculicioiu, V.S.; Gorcea, C.M.; Zăgărin, T.-I.; Murarasu, A.R.; Todea, D.A. Comparative Effects of Cigarette Smoke and Heated Tobacco Product Aerosols on Biofilm Production by Respiratory Pathogens. Microorganisms 2025, 13, 2459. https://doi.org/10.3390/microorganisms13112459
Schiopu P, Toc DA, Colosi IA, Costache C, Panaitescu P-Ș, Neculicioiu VS, Gorcea CM, Zăgărin T-I, Murarasu AR, Todea DA. Comparative Effects of Cigarette Smoke and Heated Tobacco Product Aerosols on Biofilm Production by Respiratory Pathogens. Microorganisms. 2025; 13(11):2459. https://doi.org/10.3390/microorganisms13112459
Chicago/Turabian StyleSchiopu, Pavel, Dan Alexandru Toc, Ioana Alina Colosi, Carmen Costache, Paul-Ștefan Panaitescu, Vlad Sever Neculicioiu, Codrina Mihaela Gorcea, Tudor-Ioan Zăgărin, Andreea Roxana Murarasu, and Doina Adina Todea. 2025. "Comparative Effects of Cigarette Smoke and Heated Tobacco Product Aerosols on Biofilm Production by Respiratory Pathogens" Microorganisms 13, no. 11: 2459. https://doi.org/10.3390/microorganisms13112459
APA StyleSchiopu, P., Toc, D. A., Colosi, I. A., Costache, C., Panaitescu, P.-Ș., Neculicioiu, V. S., Gorcea, C. M., Zăgărin, T.-I., Murarasu, A. R., & Todea, D. A. (2025). Comparative Effects of Cigarette Smoke and Heated Tobacco Product Aerosols on Biofilm Production by Respiratory Pathogens. Microorganisms, 13(11), 2459. https://doi.org/10.3390/microorganisms13112459








