Endophytes in Agriculture: Potential to Improve Yields and Tolerances of Agricultural Crops
Abstract
1. Introduction
2. Requirements for Increased Crop Productivity: Climate Change and Population Growth
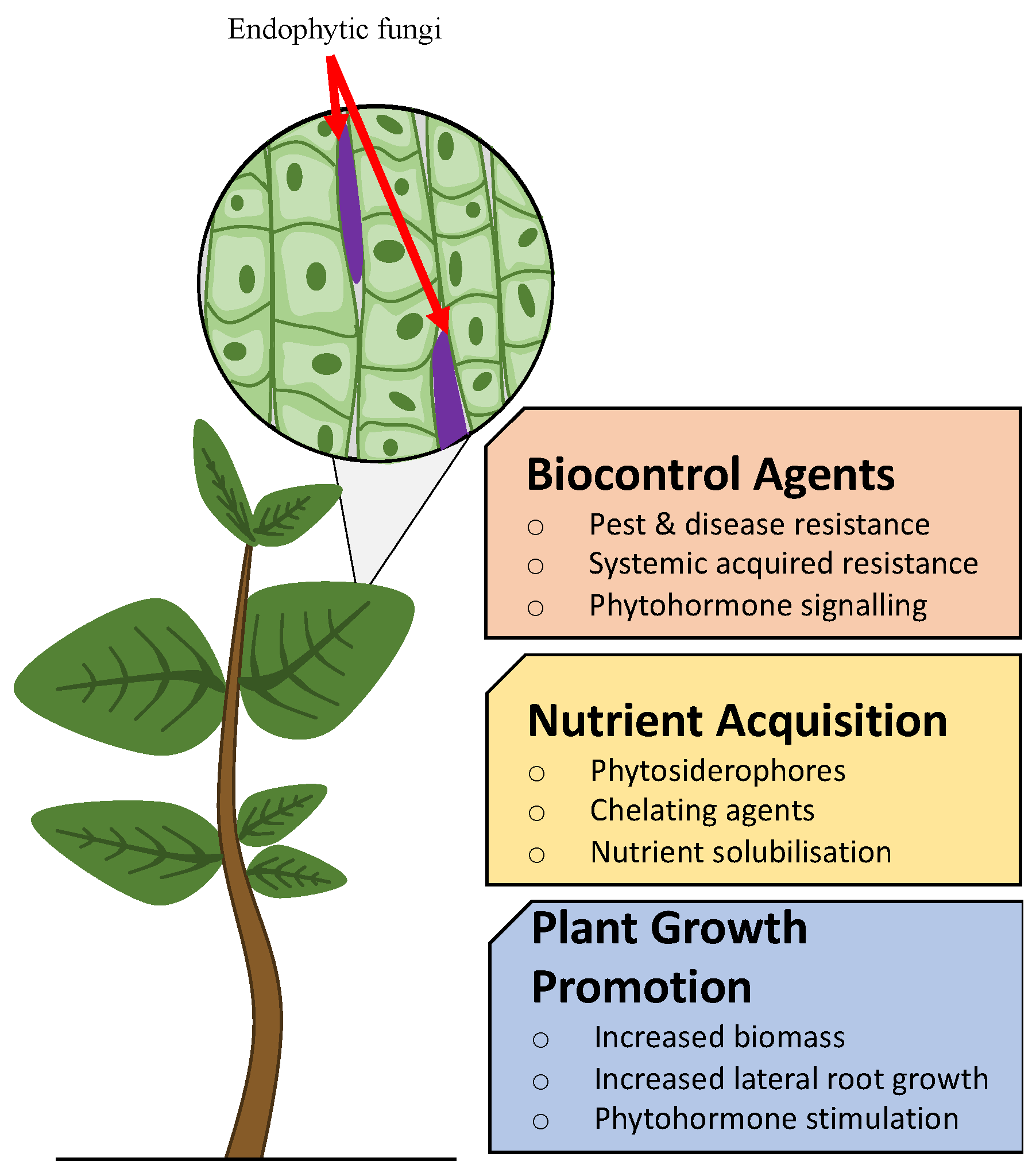
3. Agricultural Potential of Endophytes
| Endophyte | Host Plant * | Effect | Reference | Type |
|---|---|---|---|---|
| Acremonium sp. Lp1,2 | Lolium perenne | Antifungal | [37] | Fungi |
| Aspergillus terreus AM2 | Moringa oleifera | Antifungal, IAA production | [38,39] | Fungi |
| Trichoderma harzianum | Hardwood bark | Antifungal, P solubilisation | [40,41,42] | Fungi |
| Epichloe festucae Fr1, 11, E365 | Lolium perenne | Antifungal | [37] | Fungi |
| Epichloe sp. | Not reported | Biotic stress tolerance | [43] | Fungi |
| Paraphaeosphaeria sporulosa | Actinidia deliciosa | Biotic stress tolerance | [44,45] | Fungi |
| Cochliobolus sp. 23-1 | Panicum coloratum, Chloris gayana | Ca accumulation | [46] | Fungi |
| Ophiosphaerella sp. 15-2 | Panicum coloratum, Chloris gayana | Ca accumulation | [46] | Fungi |
| Penicillium bilaii | Elaeis guineensis | Ca accumulation | [47] | Fungi |
| Penicillium oxalicum P4 | Elaeis guineensis | Ca accumulation | [47] | Fungi |
| Setosphaeria rostrata GR1A | Panicum coloratum, Chloris gayana | Ca accumulation | [46] | Fungi |
| Fusarium fujikuroi IMI58289 | Manihot esuclenta, Oryza sativa | GA production | [48,49,50] | Fungi |
| Gibberella fujikuroi | Manihot esculenta | GA production | [48,49] | Fungi |
| A. lipoferum | Zea mays | GA production | [51] | Fungi |
| Penicillium citrinum IR-3-3 | Ixeris repenes | GA production | [52] | Fungi |
| Penicillium commune KNU5379 | Seasamum indicum | GA production | [49] | Fungi |
| Penicillium funiculosum | Glycine max | GA production | [53] | Fungi |
| A. brasilense | Zea mays | GA production | [51] | Fungi |
| Sphaceloma manihoticola Lu949 | Manihot esuclenta | GA production | [48,49] | Fungi |
| Aspergillus fumigatus LHL06 | Glycine max, Zea mays | GA production, pathogen resistance | [52,54] | Fungi |
| Beauveria bassiana | Vitis vinifera, wheat | General growth, pest resistance | [55,56,57,58] | Fungi |
| Colletotrichum tofieldiae CT04_08450 | Arabidopsis thaliana | General growth, hormone production | [59] | Fungi |
| Diaporthe sp. | Festuca rubra | General growth, IAA production, N, Ca, Mg, Fe accumulation | [60] | Fungi |
| Neotyphodium lolii NEA4 | Lolium perenne | General growth, Pest deterrence, pathogen resistance excl. staggers | [61] | Fungi |
| Acremonium coenophialum | Festuca arundinacea | Growth, pest resistance, pathogen resistance | [62] | Fungi |
| Aspergillus flavus | Euphorbia geniculata | IAA production, pathogen resistance | [63] | Fungi |
| Piriformospora indica | Multiple | IAA production, salt tolerance | [64,65] | Fungi |
| Gilmaniella sp. AL12 | Atractylodes lancea | Jasmonic acid production | [66] | Fungi |
| Phomopsis liquidambari | Bischofia polycarpa | N & P accumulation | [67] | Fungi |
| Heteroconium chaetospira | Chinese cabbage | Nitrogen metabolism, general growth | [68] | Fungi |
| Aspergillus niger AP5, P85 | Arachis hypogaea | P solubilisation, IAA production, Ca accumulation | [47,69] | Fungi |
| Glomus mosseae | Not reported | P, K, Mg, Cu, Zn, Mn accumulation | [70] | Fungi |
| Trichoderma arundinaceum | Not reported | Jasmonic acid production | [71] | Fungi |
| Trichoderma harzianum TRI5 | Not reported | Biotic stress tolerance | [72] | Fungi |
| Trichoderma virens IB 119/12 | Glycine max | Pathogen resistance | [73] | Fungi |
| Cladosporium cladosporioides BOU1 | Solanum melongena | Pest resistance | [74] | Fungi |
| Epichloe coenphiala AR584 | Tall fescue | Pest resistance | [75] | Fungi |
| Fusarium oxysporum 24o, V5W2 | Musa spp. | Pest resistance | [76,77] | Fungi |
| Metarhizium anisopliae QS155 | Not reported | Pest resistance | [41] | Fungi |
| Epichloe gansuensis | Achnatherum inebrians | Salicylic acid production | [78] | Fungi |
| Bacillus subtillis 26D | Not reported | Biotic stress tolerance | [79] | Bacteria |
| Streptomyces sp. 11E | Vigna radiata | Auxin production, N fixation, salt tolerance, siderophore production | [80,81] | Bacteria |
| Azospirillum brasilense B510 | Oryza sativa | IAA production, N fixation | [82,83] | Bacteria |
| Paenibacillus sp. ANM59, ANM76 | Cicer arietinum | IAA production, P solubilisation, salt tolerance | [84] | Bacteria |
| Bacillus thuringiensis | Zea mays | Insect deterrent | [85] | Bacteria |
| Ewingella americana EU-M4ARAct | Zea mays | K accumulation | [86] | Bacteria |
| Pantoea agglomerans EU-E1RT3-1 | Zea mays | K accumulation | [86] | Bacteria |
| Pseudomonas brenneri EU-A2SK1 | Zea mays | K accumulation | [86] | Bacteria |
| Mesorhizobium ciceri BRM5 | Cicer arietinum | N fixation, IAA production, salt tolerance | [87,88] | Bacteria |
| Azospirillum brasilense Cd, Az39 | Triticum aestivum | N fixation, P solubilisation, auxin production | [89,90] | Bacteria |
| Bacillus sp. 13E | Vigna radiata | N fixation, P solubilisation, auxin production | [80] | Bacteria |
| Bacillus endophyticus 14E | Vigna unguiculata, soybean | N fixation, P solubilisation, auxin production, salt tolerance | [80] | Bacteria |
| Bacillus altitudinis Q7 | Ginkgo biloba | Pathogen resistance | [91] | Bacteria |
| Bacillus polymyxa L6 | Not reported | Pathogen resistance | [92] | Bacteria |
| Serratia plymuthica HRO-C48 | Not reported | Pest resistance | [93] | Bacteria |
| Burkholderia sp. SSG | Beta vulgaris | S metabolism, N fixation, K accumulation, IAA production | [94,95] | Bacteria |
| Azotobacter chroococcum Avi2 | Not reported | Salt tolerance, drought stress, general growth | [96] | Bacteria |
| Serratia marcescens AL2-16 | Achyranthes aspera | Siderophore production, IAA production, ammonia production, general growth | [97,98,99] | Bacteria |
| Pseudomonas fluorescens L228, L111, L321 | Not reported | Siderophore production, P solubilisation, general growth, pest resistance | [100] | Bacteria |
| Enterobacter sp. SA187 | Indigofera argentea | Sulphur metabolism, reduced ROS accumulation, salt tolerance | [101] | Bacteria |
| Burkholderia phytofirmans PsJN | Not reported | Zn accumulation | [102] | Bacteria |
| Staphylococcus hominis 7E | Vigna radiata | Zn, P accumulation, hormone production, antifungal | [80] | Bacteria |
4. Plant-Endophyte Relationship
5. Role of Endophytes in Plant Health
5.1. Biotic Stressors
5.1.1. Pathogens
5.1.2. Secondary Metabolites
5.1.3. Pests
5.2. Phytohormones
5.2.1. Auxins (Indole-3-Acetic Acid)
5.2.2. Gibberellins
5.2.3. Stress Response Hormones
Abscisic Acid
Salicylic Acid
Jasmonic Acid
Cytokinin’s
Reactive Oxygen Species
5.3. Nutrient Limitations
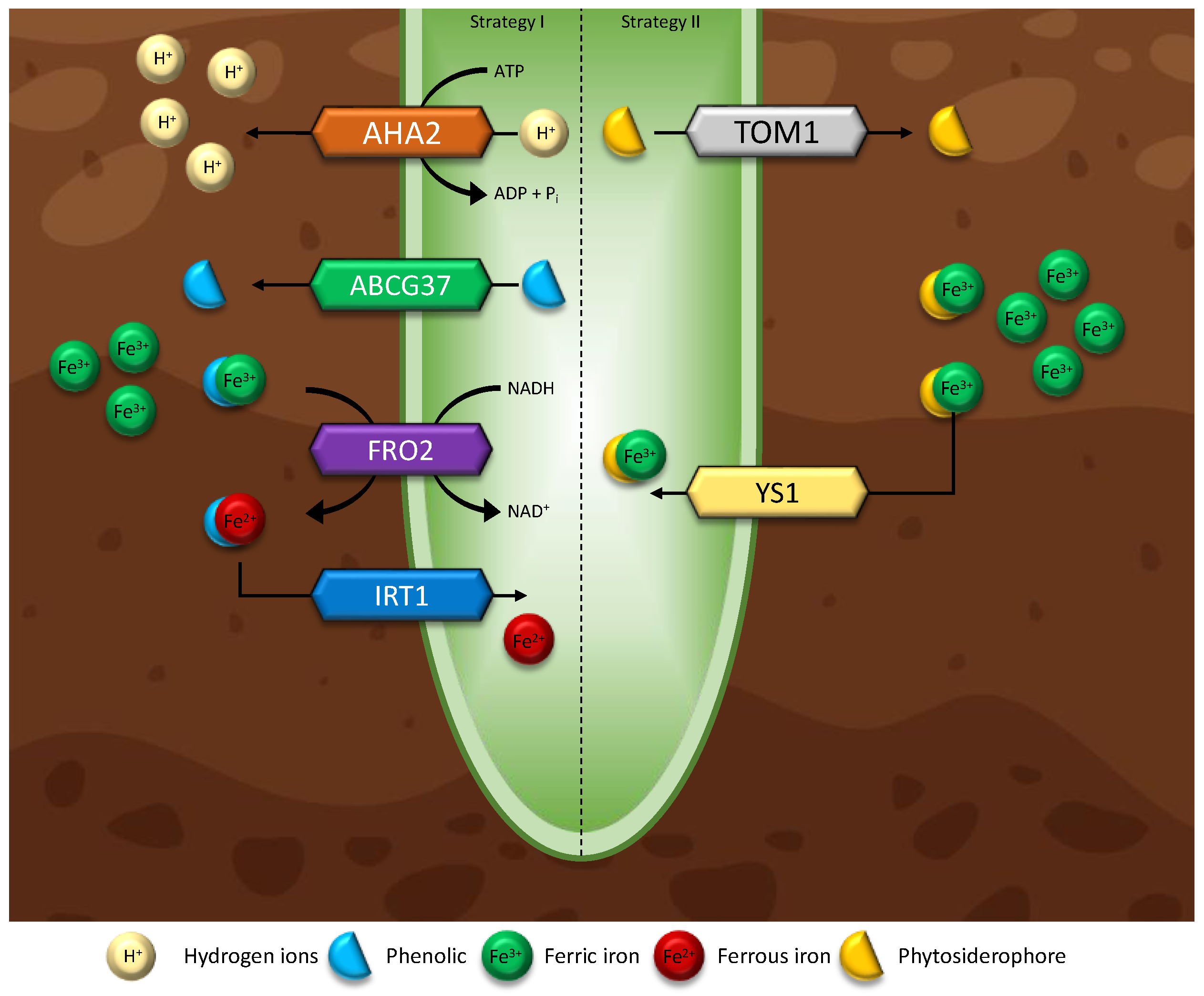
5.3.1. Iron
5.3.2. Zinc
5.3.3. Nitrogen
5.3.4. Phosphorus
5.3.5. Potassium
5.3.6. Sulphur
5.3.7. Calcium
5.3.8. Magnesium
| Nutrient | Mechanism of Endophyte Intervention | Effect on Plant | Reference |
|---|---|---|---|
| Iron | Phytosiderophore production—reducing the energy requirements to transport Fe into roots. | Reduces ROS accumulation. Key roles in photosynthesis and the electron transport chain. | [28,36,197,202,203,218] |
| Zinc | Phytosiderophore production—as above. | Enzymatic co-factor in root growth. Component of chlorophyll. | [28,36,204,205,209] |
| Nitrogen | Atmospheric fixation when soil N is low (mainly bacteria due to fungi lacking nitrogenase). | Key factor in amino compounds. Increases proline concentration. | [67,71,82,95,186,210,238] |
| Phosphorus | Organic acid production & soil acidification to increase solubilisation, and increased proteins involved in sugar metabolism. | Root growth stimulation. Key component in DNA and RNA. | [67,215,216,218,219,222] |
| Potassium | Improving flow of free K+ into roots. However, remains limited by available soil K. | Metabolic regulation, photosynthesis, stress signalling, and maintaining redox homeostasis. | [25,219,224,225] |
| Sulphur | Protein production to utilise sulphate and alkane sulfonates. | Detoxification of ROS. Key structural component of proteins. | [94,101,229] |
| Calcium | Tied to P and Mg solubilisation due to complex formation | Maintains redox homeostasis. Key structural component of cell walls. | [53,120,148,230,231] |
| Magnesium | Not determined | Key co-factor to enzymes involved in a diverse range of metabolic functions. | [234,235,237] |
6. Large-Scale Agricultural Application
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Deshmukh, S.; Verekar, S.; Bhave, S. Endophytic fungi: A reservoir of antibacterials. Front. Microbiol. 2015, 5, 715. [Google Scholar] [CrossRef] [PubMed]
- Strobel, G.; Daisy, B. Bioprospecting for microbial endophytes and their natural products. MMBR 2003, 67, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Yadav, G.; Meena, M. Bioprospecting of endophytes in medicinal plants of Thar Desert: An attractive resource for biopharmaceuticals. Biotechnol. Rep. 2021, 30, e00629. [Google Scholar] [CrossRef]
- Manganyi, M.C.; Ateba, C.N. Untapped potentials of endophytic fungi: A review of novel bioactive compounds with biological applications. Microorganisms 2020, 8, 1934. [Google Scholar] [CrossRef]
- Schulz, B.; Boyle, C. The endophytic continuum. Mycol. Res. 2005, 109, 661–686. [Google Scholar] [CrossRef]
- Baron, N.C.; Rigobelo, E.C. Endophytic fungi: A tool for plant growth promotion and sustainable agriculture. Mycology 2022, 13, 39–55. [Google Scholar] [CrossRef] [PubMed]
- Singh, O.V. Food Borne Pathogens and Antibiotic Resistance; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- AURA. Fourth Australian Report on Antimicrobial Use and Resistance in Human Health; AURA: Sydney, NSW, Australia, 2021; ISBN 978-1-922563-21-7. [Google Scholar]
- Quinn, G.A.; Banat, A.M.; Abdelhameed, A.M.; Banat, I.M. Streptomyces from traditional medicine: Sources of new innovations in antibiotic discovery. J. Med. Microbiol. 2020, 69, 1040–1048. [Google Scholar] [CrossRef]
- United Nations, Department of Economic and Social Affairs, Population Division. World Population Prospects 2022: Summary of Results; United Nations: New York, NY, USA, 2022. [Google Scholar]
- Putri, R.; Naufal, M.; Nandini, M.; Dwiputra, D.; Wibirama, S.; Sumantyo, J. The Impact of Population Pressure on Agricultural Land towards Food Sufficiency (Case in West Kalimantan Province, Indonesia). In Proceedings of the IOP Conference Series: Earth and Environmental Science, Yogyakarta, Indonesia, 22–23 October 2019; p. 012050. [Google Scholar]
- Anggraini, D.P.; Sutrisno, J.; Barokah, U. The impact analysis of rice field conversion on food security in Sragen Regency. Proc. E3S Web Conf. 2021, 316, 02035. [Google Scholar] [CrossRef]
- Patel, S.K.; Sharma, A.; Singh, G.S. Traditional agricultural practices in India: An approach for environmental sustainability and food security. Energy Ecol. Environ. 2020, 5, 253–271. [Google Scholar] [CrossRef]
- FAO; IFAD; UNICEF; WFP; WHO. The State of Food Security and Nutrition in the World 2021; FAO: Rome, Italy, 2021. [Google Scholar]
- Kraemer, K.; Gilbert, C. Do vitamin A deficiency and undernutrition still matter? Community Eye Health 2013, 26, 61–63. [Google Scholar]
- Roser, M.; Ritchie, H. Hunger and Undernourishment. Available online: https://ourworldindata.org/hunger-and-undernourishment (accessed on 24 October 2022).
- Savary, S.; Willocquet, L. Simulation modeling in botanical epidemiology and crop loss analysis. Plant Health Instr. 2014, 147. [Google Scholar]
- Cerda, R.; Avelino, J.; Gary, C.; Tixier, P.; Lechevallier, E.; Allinne, C. Primary and Secondary Yield Losses Caused by Pests and Diseases: Assessment and Modeling in Coffee. PLoS ONE 2017, 12, e0169133. [Google Scholar] [CrossRef]
- Smith, H.M.; Samach, A. Constraints to obtaining consistent annual yields in perennial tree crops. I: Heavy fruit load dominates over vegetative growth. Plant Sci. 2013, 207, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Blair, A.; Ritz, B.; Wesseling, C.; Beane Freeman, L. Pesticides and human health. Occup. Environ. Med. 2015, 72, 81. [Google Scholar] [CrossRef]
- Gill, S.S.; Gill, R.; Trivedi, D.K.; Anjum, N.A.; Sharma, K.K.; Ansari, M.W.; Ansari, A.A.; Johri, A.K.; Prasad, R.; Pereira, E.; et al. Piriformospora indica: Potential and Significance in Plant Stress Tolerance. Front. Microbiol. 2016, 7, 332. [Google Scholar] [CrossRef] [PubMed]
- Le Cocq, K.; Gurr, S.J.; Hirsch, P.R.; Mauchline, T.H. Exploitation of endophytes for sustainable agricultural intensification. Mol. Plant Pathol. 2017, 18, 469–473. [Google Scholar] [CrossRef] [PubMed]
- Pammi, M.; Aghaeepour, N.; Neu, J. Multiomics, artificial intelligence, and precision medicine in perinatology. Pediatr. Res. 2022, 93, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Govindaraj, M.; Masilamani, P.; Albert, V.A.; Bhaskaran, M. Effect of physical seed treatment on yield and quality of crops: A review. Agric. Rev. 2017, 38, 1–14. [Google Scholar] [CrossRef]
- Hoffmann, C.; Stockfisch, N.; Koch, H.-J. Influence of sulphur supply on yield and quality of sugar beet (Beta vulgaris L.)—Determination of a threshold value. Eur. J. Agron. 2004, 21, 69–80. [Google Scholar] [CrossRef]
- Sheng, M.; Tang, M.; Chen, H.; Yang, B.; Zhang, F.; Huang, Y. Influence of arbuscular mycorrhizae on photosynthesis and water status of maize plants under salt stress. Mycorrhiza 2008, 18, 287–296. [Google Scholar] [CrossRef]
- Su, Z.-z.; Wang, T.; Shrivastava, N.; Chen, Y.-y.; Liu, X.; Sun, C.; Yin, Y.; Gao, Q.-k.; Lou, B.-g. Piriformospora indica promotes growth, seed yield and quality of Brassica napus L. Microbiol. Res. 2017, 199, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Geat, N.; Rajawat, M.V.S.; Prasanna, R.; Kar, A.; Singh, A.M.; Saxena, A.K. Prospecting endophytes from different Fe or Zn accumulating wheat genotypes for their influence as inoculants on plant growth, yield, and micronutrient content. Ann. Microbiol. 2018, 68, 815–833. [Google Scholar] [CrossRef]
- Mei, C.; Flinn, B.S. The use of beneficial microbial endophytes for plant biomass and stress tolerance improvement. Recent Pat. Biotechnol. 2010, 4, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Khaksar, G.; Treesubsuntorn, C.; Thiravetyan, P. Effect of endophytic Bacillus cereus ERBP inoculation into non-native host: Potentials and challenges for airborne formaldehyde removal. Plant Physiol. Biochem. 2016, 107, 326–336. [Google Scholar] [CrossRef]
- Gaiero, J.R.; McCall, C.A.; Thompson, K.A.; Day, N.J.; Best, A.S.; Dunfield, K.E. Inside the root microbiome: Bacterial root endophytes and plant growth promotion. Am. J. Bot. 2013, 100, 1738–1750. [Google Scholar] [CrossRef]
- Nelson, E.B. Microbial dynamics and interactions in the spermosphere. Annu. Rev. Phytopathol. 2004, 42, 271–309. [Google Scholar] [CrossRef]
- Robinson, R.J.; Fraaije, B.A.; Clark, I.M.; Jackson, R.W.; Hirsch, P.R.; Mauchline, T.H. Wheat seed embryo excision enables the creation of axenic seedlings and Koch’s postulates testing of putative bacterial endophytes. Sci. Rep. 2016, 6, 25581. [Google Scholar] [CrossRef]
- Wakeel, A.; Farooq, M.; Bashir, K.; Ozturk, L. Micronutrient Malnutrition and Biofortification: Recent Advances and Future Perspectives. In Plant Micronutrient Use Efficiency; Academic Press: Cambridge, MA, USA, 2018; pp. 225–243. [Google Scholar]
- Römheld, V. The role of phytosiderophores in acquisition of iron and other micronutrients in graminaceous species: An ecological approach. In Iron Nutrition and Interactions in Plants; Springer: Berlin/Heidelberg, Germany, 1991; pp. 159–166. [Google Scholar]
- Prasad, R.; Shivay, Y.S.; Mandi, S. Chapter 21—Phytosiderophores and absorption of iron and other cations by plants. In Cation Transporters in Plants; Upadhyay, S.K., Ed.; Academic Press: Cambridge, MA, USA, 2022; pp. 385–399. [Google Scholar]
- Christensen, M.J. Antifungal activity in grasses infected with Acremonium and Epichloe endophytes. Australas. Plant Pathol. 1996, 25, 186–191. [Google Scholar] [CrossRef]
- Hashem, A.H.; Shehabeldine, A.M.; Abdelaziz, A.M.; Amin, B.H.; Sharaf, M.H. Antifungal Activity of Endophytic Aspergillus terreus Extract Against Some Fungi Causing Mucormycosis: Ultrastructural Study. Appl. Biochem. Biotechnol. 2022, 194, 3468–3482. [Google Scholar] [CrossRef]
- Vassileva, M.; Malusá, E.; Eichler-Löbermann, B.; Vassilev, N. Aspegillus terreus: From Soil to Industry and Back. Microorganisms 2020, 8, 1655. [Google Scholar] [CrossRef]
- Dabiré, T.G.; Bonzi, S.; Somda, I.; Legrève, A. Evaluation of the potential of Trichoderma harzianum as a plant growth promoter and biocontrol agent against Fusarium damping-off in onion in Burkina Faso. Asian J. Plant Pathol. 2016, 10, 49–60. [Google Scholar] [CrossRef]
- Shah, S.; Ash, G.J.; Wilson, B.A. Resporulation of Metarhizium anisopliae granules on soil and mortality of Tenebrio molitor: Implications for wireworm management in sweetpotato. Ann. Appl. Biol. 2023, 182, 65–76. [Google Scholar] [CrossRef]
- Vinale, F.; Flematti, G.; Sivasithamparam, K.; Lorito, M.; Marra, R.; Skelton, B.W.; Ghisalberti, E.L. Harzianic acid, an antifungal and plant growth promoting metabolite from Trichoderma harzianum. J. Nat. Prod. 2009, 72, 2032–2035. [Google Scholar] [CrossRef] [PubMed]
- Xia, C.; Li, N.; Zhang, X.; Feng, Y.; Christensen, M.J.; Nan, Z. An Epichloe endophyte improves photosynthetic ability and dry matter production of its host Achnatherum inebrians infected by Blumeria graminis under various soil water conditions. Fungal Ecol. 2016, 22, 26–34. [Google Scholar] [CrossRef]
- Chen, Q.; Yu, J.-J.; He, J.; Feng, T.; Liu, J.-K. Isobenzofuranones and isocoumarins from kiwi endophytic fungus Paraphaeosphaeria sporulosa and their antibacterial activity against Pseudomonas syringae pv. actinidiae. Phytochemistry 2022, 195, 113050. [Google Scholar] [CrossRef]
- Xin, X.-F.; Kvitko, B.; He, S.Y. Pseudomonas syringae: What it takes to be a pathogen. Nat. Rev. Microbiol. 2018, 16, 316–328. [Google Scholar] [CrossRef]
- Spagnoletti, F.N.; Tobar, N.E.; Fernández Di Pardo, A.; Chiocchio, V.M.; Lavado, R.S. Dark septate endophytes present different potential to solubilize calcium, iron and aluminum phosphates. Appl. Soil Ecol. 2017, 111, 25–32. [Google Scholar] [CrossRef]
- Yin, Z.; Shi, F.; Jiang, H.; Roberts, D.P.; Chen, S.; Fan, B. Phosphate solubilization and promotion of maize growth by Penicillium oxalicum P4 and Aspergillus niger P85 in a calcareous soil. Can. J. Microbiol 2015, 61, 913–923. [Google Scholar] [CrossRef]
- Bömke, C.; Rojas, M.C.; Gong, F.; Hedden, P.; Tudzynski, B. Isolation and characterization of the gibberellin biosynthetic gene cluster in Sphaceloma manihoticola. Appl. Environ. Microbiol. 2008, 74, 5325–5339. [Google Scholar] [CrossRef]
- Choi, W.-Y.; Rim, S.-O.; Lee, J.-H.; Lee, J.-M.; Lee, I.-J.; Cho, K.-J.; Rhee, I.-K.; Kwon, J.-B.; Kim, J.-G. Isolation of gibberellins-producing fungi from the root of several Sesamum indicum plants. J. Microbiol. Biotechnol. 2005, 15, 22–28. [Google Scholar]
- Rademacher, W.; Graebe, J.E. Gibberellin A4 produced by Sphacelomamanihoticola, the cause of the superelongation disease of cassava (Manihotesculenta). Biochem. Biophys. Res. Commun. 1979, 91, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Lucangeli, C.; Bottini, R. Effects of Azospirillum spp. on endogenous gibberellin content and growth of maize (Zea mays L.) treated with uniconazole. Symbiosis 1997, 23, 63–72. [Google Scholar]
- Khan, S.A.; Hamayun, M.; Yoon, H.; Kim, H.-Y.; Suh, S.-J.; Hwang, S.-K.; Kim, J.-M.; Lee, I.-J.; Choo, Y.-S.; Yoon, U.-H. Plant growth promotion and Penicillium citrinum. BMC Microbiol. 2008, 8, 231. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.L.; Hamayun, M.; Kim, Y.-H.; Kang, S.-M.; Lee, I.-J. Ameliorative symbiosis of endophyte (Penicillium funiculosum LHL06) under salt stress elevated plant growth of Glycine max L. Plant Physiol. Biochem. 2011, 49, 852–861. [Google Scholar] [CrossRef]
- Abdelaziz, A.M.; El-Wakil, D.A.; Attia, M.S.; Ali, O.M.; AbdElgawad, H.; Hashem, A.H. Inhibition of Aspergillus flavus Growth and Aflatoxin Production in Zea mays L. Using Endophytic Aspergillus fumigatus. J. Fungi 2022, 8, 482. [Google Scholar] [CrossRef]
- Mantzoukas, S.; Lagogiannis, I.; Mpousia, D.; Ntoukas, A.; Karmakolia, K.; Eliopoulos, P.A.; Poulas, K. Beauveria bassiana Endophytic Strain as Plant Growth Promoter: The Case of the Grape Vine Vitis vinifera. J. Fungi 2021, 7, 142. [Google Scholar] [CrossRef]
- Saragih, M.; Trizelia; Nurbailis; Yusniwati. Endophytic Colonization and Plant Growth Promoting Effect by Entomopathogenic fungus, Beauveria bassiana to Red Chili (Capsicum annuum L.) with Different Inoculation Methods. IOP Conf. Ser. Earth Environ. Sci. 2019, 305, 012070. [Google Scholar] [CrossRef]
- Nakahara, Y.; Shimura, S.; Ueno, C.; Kanamori, Y.; Mita, K.; Kiuchi, M.; Kamimura, M. Purification and characterization of silkworm hemocytes by flow cytometry. Dev. Comp. Immunol. 2009, 33, 439–448. [Google Scholar] [CrossRef]
- Wang, X.; Radwan, M.M.; Taráwneh, A.H.; Gao, J.; Wedge, D.E.; Rosa, L.H.; Cutler, H.G.; Cutler, S.J. Antifungal activity against plant pathogens of metabolites from the endophytic fungus Cladosporium cladosporioides. J. Agric. Food Chem. 2013, 61, 4551–4555. [Google Scholar] [CrossRef]
- Hiruma, K.; Gerlach, N.; Sacristán, S.; Nakano, R.T.; Hacquard, S.; Kracher, B.; Neumann, U.; Ramírez, D.; Bucher, M.; O’Connell, R.J.; et al. Root Endophyte Colletotrichum tofieldiae Confers Plant Fitness Benefits that Are Phosphate Status Dependent. Cell 2016, 165, 464–474. [Google Scholar] [CrossRef]
- Huang, W.; Cai, Y.; Hyde, K.; Corke, H.; Sun, M. Biodiversity of endophytic fungi associated with 29 traditional Chinese medicinal plants. Fungal Divers. 2008, 33, 61–75. [Google Scholar]
- Barenbrug, A. Barenbrug Autumn 2022 Product Guide; Barenbrug Agriculture: Melbourne, Australia, 2022; pp. 1–12. [Google Scholar]
- Christensen, M.J.; Latch, G.C.M. Variation among isolates of Acremonium endophytes (A. coenophialum and possibly A. typhinum) from tall fescue (Festuca arundinacea). Mycol. Res. 1991, 95, 1123–1126. [Google Scholar] [CrossRef]
- Abdel-Motaal, F.; Kamel, N.; El-Zayat, S.; Abou-Ellail, M. Early blight suppression and plant growth promotion potential of the endophyte Aspergillus flavus in tomato plant. Ann. Agric. Sci. 2020, 65, 117–123. [Google Scholar] [CrossRef]
- Oelmüller, R.; Sherameti, I.; Tripathi, S.; Varma, A. Piriformospora indica, a cultivable root endophyte with multiple biotechnological applications. Symbiosis 2009, 49, 1–17. [Google Scholar] [CrossRef]
- Sirrenberg, A.; Göbel, C.; Grond, S.; Czempinski, N.; Ratzinger, A.; Karlovsky, P.; Santos, P.; Feussner, I.; Pawlowski, K. Piriformospora indica affects plant growth by auxin production. Physiol. Plant. 2007, 131, 581–589. [Google Scholar] [CrossRef]
- Ren, C.-G.; Dai, C.-C. Jasmonic acid is involved in the signaling pathway for fungal endophyte-induced volatile oil accumulation of Atractylodes lancea plantlets. BMC Plant Biol. 2012, 12, 128. [Google Scholar] [CrossRef]
- Tang, M.-J.; Zhu, Q.; Zhang, F.-M.; Zhang, W.; Yuan, J.; Sun, K.; Xu, F.-J.; Dai, C.-C. Enhanced nitrogen and phosphorus activation with an optimized bacterial community by endophytic fungus Phomopsis liquidambari in paddy soil. Microbiol. Res. 2019, 221, 50–59. [Google Scholar] [CrossRef]
- Usuki, F.; Narisawa, K. A mutualistic symbiosis between a dark septate endophytic fungus, Heteroconium chaetospira, and a nonmycorrhizal plant, Chinese cabbage. Mycologia 2007, 99, 175–184. [Google Scholar] [CrossRef]
- Abdelgawad, M.A.; Hamed, A.A.; Nayl, A.A.; Badawy, M.; Ghoneim, M.M.; Sayed, A.M.; Hassan, H.M.; Gamaleldin, N.M. The Chemical Profiling, Docking Study, and Antimicrobial and Antibiofilm Activities of the Endophytic fungi Aspergillus sp. AP5. Molecules 2022, 27, 1704. [Google Scholar] [CrossRef]
- Chen, M.; Yang, G.; Sheng, Y.; Li, P.; Qiu, H.; Zhou, X.; Huang, L.; Chao, Z. Glomus mosseae Inoculation Improves the Root System Architecture, Photosynthetic Efficiency and Flavonoids Accumulation of Liquorice under Nutrient Stress. Front. Plant Sci. 2017, 8, 931. [Google Scholar] [CrossRef]
- Mengistu, A.A. Endophytes: Colonization, Behaviour, and Their Role in Defense Mechanism. Int. J. Microbiol. 2020, 2020, 6927219. [Google Scholar] [CrossRef] [PubMed]
- Taylor, L.; Gutierrez, S.; McCormick, S.P.; Bakker, M.G.; Proctor, R.H.; Teresi, J.; Kurtzman, B.; Hao, G.; Vaughan, M.M. Use of the volatile trichodiene to reduce Fusarium head blight and trichothecene contamination in wheat. Microb. Biotechnol. 2022, 15, 513–527. [Google Scholar] [CrossRef] [PubMed]
- Haddad, P.E.; Leite, L.G.; Lucon, C.M.M.; Harakava, R. Selection of Trichoderma spp. strains for the control of Sclerotinia sclerotiorum in soybean. Pesqui. Agropecu. Bras. 2017, 52, 1140–1148. [Google Scholar] [CrossRef]
- Islam, T.; Gupta, D.R.; Surovy, M.Z.; Mahmud, N.U.; Mazlan, N.; Islam, T. Identification and application of a fungal biocontrol agent Cladosporium cladosporioides against Bemisia tabaci. Biotechnol. Bioteq. 2019, 33, 1698–1705. [Google Scholar] [CrossRef]
- Popay, A.J.; Jensen, J.G.; Mace, W.J. Root Herbivory: Grass Species, Epichloe Endophytes and Moisture Status Make a Difference. Microorganisms 2020, 8, 997. [Google Scholar] [CrossRef]
- Waweru, B.; Turoop, L.; Kahangi, E.; Coyne, D.; Dubois, T. Non-pathogenic Fusarium oxysporum endophytes provide field control of nematodes, improving yield of banana (Musa sp.). Biol. Control 2014, 74, 82–88. [Google Scholar] [CrossRef]
- Lopes, R.; Tsui, S.; Gonçalves, P.J.R.O.; De Queiroz, M.V. A look into a multifunctional toolbox: Endophytic Bacillus species provide broad and underexploited benefits for plants. World J. Microbiol. Biotechnol. 2018, 34, 94. [Google Scholar] [CrossRef]
- Kou, M.-Z.; Bastías, D.A.; Christensen, M.J.; Zhong, R.; Nan, Z.-B.; Zhang, X.-X. The Plant Salicylic Acid Signalling Pathway Regulates the Infection of a Biotrophic Pathogen in Grasses Associated with an Epichloe Endophyte. J. Fungi 2021, 7, 633. [Google Scholar] [CrossRef]
- Chakraborty, M.; Mahmud, N.U.; Gupta, D.R.; Tareq, F.S.; Shin, H.J.; Islam, T. Inhibitory Effects of Linear Lipopeptides From a Marine Bacillus subtilis on the Wheat Blast Fungus Magnaporthe oryzae Triticum. Front. Microbiol. 2020, 11, 665. [Google Scholar] [CrossRef]
- Bakhtiyarifar, M.; Enayatizamir, N.; Mehdi Khanlou, K. Biochemical and molecular investigation of non-rhizobial endophytic bacteria as potential biofertilisers. Arch. Microbiol. 2021, 203, 513–521. [Google Scholar] [CrossRef]
- Nonthakaew, N.; Panbangred, W.; Songnuan, W.; Intra, B. Plant growth-promoting properties of Streptomyces spp. isolates and their impact on mung bean plantlets’ rhizosphere microbiome. Front. Microbiol. 2022, 13, 967415. [Google Scholar] [CrossRef] [PubMed]
- Naher, K.; Miwa, H.; Okazaki, S.; Yasuda, M. Effects of Different Sources of Nitrogen on Endophytic Colonization of Rice Plants by Azospirillum sp. B510. Microbes Environ. 2018, 33, 301–308. [Google Scholar] [CrossRef]
- Molina, R.; Rivera, D.; Mora, V.; López, G.; Rosas, S.; Spaepen, S.; Vanderleyden, J.; Cassán, F. Regulation of IAA Biosynthesis in Azospirillum brasilense Under Environmental Stress Conditions. Curr. Microbiol. 2018, 75, 1408–1418. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Naseer, I.; Hussain, A.; Zahid Mumtaz, M.; Mustafa, A.; Hilger, T.H.; Ahmad Zahir, Z.; Minggang, X. Appraising Endophyte–Plant Symbiosis for Improved Growth, Nodulation, Nitrogen Fixation and Abiotic Stress Tolerance: An Experimental Investigation with Chickpea (Cicer arietinum L.). Agronomy 2019, 9, 621. [Google Scholar] [CrossRef]
- Xiao, Y.; Wu, K. Recent progress on the interaction between insects and Bacillus thuringiensis crops. Philos. Trans. R. Soc. B 2019, 374, 20180316. [Google Scholar] [CrossRef] [PubMed]
- Rana, K.L.; Kour, D.; Kaur, T.; Devi, R.; Yadav, A.; Yadav, A.N. Bioprospecting of endophytic bacteria from the Indian Himalayas and their role in plant growth promotion of maize (Zea mays L.). J. Appl. Biol. Biotechnol. 2021, 9, 41–50. [Google Scholar]
- Shahid, M.; Khan, M.S.; Syed, A.; Marraiki, N.; Elgorban, A.M. Mesorhizobium ciceri as biological tool for improving physiological, biochemical and antioxidant state of Cicer aritienum (L.) under fungicide stress. Sci. Rep. 2021, 11, 9655. [Google Scholar] [CrossRef]
- Vijayabharathi, R.; Gopalakrishnan, S.; Sathya, A.; Srinivas, V.; Sharma, M. Deciphering the tri-dimensional effect of endophytic Streptomyces sp. on chickpea for plant growth promotion, helper effect with Mesorhizobium ciceri and host-plant resistance induction against Botrytis cinerea. Microb. Pathog. 2018, 122, 98–107. [Google Scholar] [CrossRef]
- Cassán, F.; Diaz-Zorita, M. Azospirillum sp. in current agriculture: From the laboratory to the field. Soil Biol. Biochem. 2016, 103, 117–130. [Google Scholar] [CrossRef]
- Perrig, D.; Boiero, M.L.; Masciarelli, O.A.; Penna, C.; Ruiz, O.A.; Cassán, F.D.; Luna, M.V. Plant-growth-promoting compounds produced by two agronomically important strains of Azospirillum brasilense, and implications for inoculant formulation. Appl. Microbiol. Biot. 2007, 75, 1143–1150. [Google Scholar] [CrossRef]
- Sun, M.; Ye, S.; Xu, Z.; Wan, L.; Zhao, Y. Endophytic Bacillus altitudinis Q7 from Ginkgo biloba inhibits the growth of Alternaria alternata in vitro and its inhibition mode of action. Biotechnol. Biotechnol. Equip. 2021, 35, 880–894. [Google Scholar] [CrossRef]
- Bent, E.; Chanway, C.P. The growth-promoting effects of a bacterial endophyte on lodgepole pine are partially inhibited by the presence of other rhizobacteria. Can. J. Microbiol 1998, 44, 980–988. [Google Scholar] [CrossRef]
- Kshetri, L.; Naseem, F.; Pandey, P. Role of Serratia sp. as biocontrol agent and plant growth stimulator, with prospects of biotic stress management in plant. In Plant Growth Promoting Rhizobacteria for Sustainable Stress Management: Volume 2: Rhizobacteria in Biotic Stress Management; Springer: Singapore, 2019; pp. 169–200. [Google Scholar]
- Bertoldo, G.; Della Lucia, M.C.; Squartini, A.; Concheri, G.; Broccanello, C.; Romano, A.; Ravi, S.; Cagnin, M.; Baglieri, A.; Stevanato, P. Endophytic Microbiome Responses to Sulfur Availability in Beta vulgaris (L.). Int. J. Mol. Sci. 2021, 22, 7184. [Google Scholar] [CrossRef]
- Kong, P.; Hong, C. Endophytic Burkholderia sp. SSG as a potential biofertilizer promoting boxwood growth. PeerJ 2020, 8, e9547. [Google Scholar] [CrossRef]
- Kumar, U.; Kaviraj, M.; Rout, S.; Chakraborty, K.; Swain, P.; Nayak, P.K.; Nayak, A.K. Combined application of ascorbic acid and endophytic N-fixing Azotobacter chroococcum Avi2 modulates photosynthetic efficacy, antioxidants and growth-promotion in rice under moisture deficit stress. Microbiol. Res. 2021, 250, 126808. [Google Scholar] [CrossRef] [PubMed]
- Devi, K.A.; Pandey, P.; Sharma, G.D. Plant Growth-Promoting Endophyte Serratia marcescens AL2-16 Enhances the Growth of Achyranthes aspera L., a Medicinal Plant. HAYATI J. Biosci. 2016, 23, 173–180. [Google Scholar] [CrossRef]
- David, B.V.; Chandrasehar, G.; Selvam, P.N. Pseudomonas fluorescens: A plant-growth-promoting rhizobacterium (PGPR) with potential role in biocontrol of pests of crops. In Crop Improvement through Microbial Biotechnology; Elsevier: Amsterdam, The Netherlands, 2018; pp. 221–243. [Google Scholar]
- Oni, F.E.; Kieu Phuong, N.; Höfte, M. Recent advances in Pseudomonas biocontrol. In Bacteria-Plant Interactions: Advanced Research and Future Trends; Caister Academic Press: Poole, UK, 2015; pp. 167–198. [Google Scholar]
- Lally, R.D.; Galbally, P.; Moreira, A.S.; Spink, J.; Ryan, D.; Germaine, K.J.; Dowling, D.N. Application of Endophytic Pseudomonas fluorescens and a Bacterial Consortium to Brassica napus Can Increase Plant Height and Biomass under Greenhouse and Field Conditions. Front. Plant Sci. 2017, 8, 2193. [Google Scholar] [CrossRef]
- Andrés-Barrao, C.; Alzubaidy, H.; Jalal, R.; Mariappan, K.G.; de Zélicourt, A.; Bokhari, A.; Artyukh, O.; Alwutayd, K.; Rawat, A.; Shekhawat, K. Coordinated bacterial and plant sulfur metabolism in Enterobacter sp. SA187–induced plant salt stress tolerance. Proc. Natl. Acad. Sci. USA 2021, 118, e2107417118. [Google Scholar] [CrossRef]
- Zhao, S.; Wei, H.; Lin, C.-Y.; Zeng, Y.; Tucker, M.P.; Himmel, M.E.; Ding, S.-Y. Burkholderia phytofirmans Inoculation-Induced Changes on the Shoot Cell Anatomy and Iron Accumulation Reveal Novel Components of Arabidopsis-Endophyte Interaction that Can Benefit Downstream Biomass Deconstruction. Front. Plant Sci. 2016, 7, 24. [Google Scholar] [CrossRef]
- Chen, Z.; Jin, Y.; Yao, X.; Chen, T.; Wei, X.; Li, C.; White, J.F.; Nan, Z. Fungal Endophyte Improves Survival of Lolium perenne in Low Fertility Soils by Increasing Root Growth, Metabolic Activity and Absorption of Nutrients. Plant Soil 2020, 452, 185–206. [Google Scholar] [CrossRef]
- Khan, Z.; Rho, H.; Firrincieli, A.; Hung, S.H.; Luna, V.; Masciarelli, O.; Kim, S.-H.; Doty, S.L. Growth enhancement and drought tolerance of hybrid poplar upon inoculation with endophyte consortia. Curr. Plant Biol. 2016, 6, 38–47. [Google Scholar] [CrossRef]
- Rana, K.L.; Kour, D.; Sheikh, I.; Yadav, N.; Yadav, A.N.; Kumar, V.; Singh, B.P.; Dhaliwal, H.S.; Saxena, A.K. Biodiversity of Endophytic Fungi from Diverse Niches and Their Biotechnological Applications. In Advances in Endophytic Fungal Research: Present Status and Future Challenges; Singh, B.P., Ed.; Springer International Publishing: Cham, Switzerland, 2019; pp. 105–144. [Google Scholar]
- Rodriguez, R.J.; White, J.F., Jr.; Arnold, A.E.; Redman, R.S. Fungal endophytes: Diversity and functional roles. New Phytol. 2009, 182, 314–330. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Kumar, A.; Singh, R.; Pandey, K.D. Endophytic bacteria: A new source of bioactive compounds. Biotech 2017, 7, 315. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, K.R. Diversity, Ecology, and Significance of Fungal Endophytes. In Endophytes and Secondary Metabolites; Jha, S., Ed.; Springer International Publishing: Cham, Switzerland, 2019; pp. 61–100. [Google Scholar]
- Tonial, F.; Falcão De Macedo Nava, F.; Luisa Gayger, A.; Bernardon Mar, T. Endophytes Potential Use in Crop Production; IntechOpen: London, UK, 2020. [Google Scholar]
- Yadav, A.; Yadav, K. Exploring the potential of endophytes in agriculture: A minireview. Adv. Plants Agric. Res. 2017, 6, 102–106. [Google Scholar] [CrossRef]
- Singh, S.; Kumar, V.; Dhanjal, D.S.; Sidhu, G.K.; Datta, S.; Kumar, S.; Singh, J. Endophytic microbes in abiotic stress management. In Microbial Endophytes; Elsevier: Amsterdam, The Netherlands, 2020; pp. 91–123. [Google Scholar]
- Liu, D.; Zhang, P.; Chen, D.; Howell, K. From the Vineyard to the Winery: How Microbial Ecology Drives Regional Distinctiveness of Wine. Front. Microbiol. 2019, 10, 2679. [Google Scholar] [CrossRef]
- Saikkonen, K.; Wäli, P.; Helander, M.; Faeth, S.H. Evolution of endophyte–plant symbioses. Trends Plant Sci. 2004, 9, 275–280. [Google Scholar] [CrossRef]
- Rizzo, D.M.; Lichtveld, M.; Mazet, J.A.; Togami, E.; Miller, S.A. Plant health and its effects on food safety and security in a One Health framework: Four case studies. One Health Outlook 2021, 3, 6. [Google Scholar] [CrossRef]
- Zappia, R.E.; Hüberli, D.; Hardy, G.E.S.J.; Bayliss, K.L. Fungi and oomycetes in open irrigation systems: Knowledge gaps and biosecurity implications. Plant Pathol. 2014, 63, 961–972. [Google Scholar] [CrossRef]
- Agrios, G.N. Plant Pathogens and Disease: General Introduction. In Encyclopedia of Microbiology, 3rd ed.; Schaechter, M., Ed.; Academic Press: Oxford, UK, 2009; pp. 613–646. [Google Scholar]
- Delgado-Baquerizo, M.; Guerra, C.A.; Cano-Díaz, C.; Egidi, E.; Wang, J.-T.; Eisenhauer, N.; Singh, B.K.; Maestre, F.T. The proportion of soil-borne pathogens increases with warming at the global scale. Nat. Clim. Chang. 2020, 10, 550–554. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhu, S.; Zhang, F.; Zhao, Z.; Christensen, M.J.; Nan, Z.; Zhang, X. Transcriptomic Analyses Reveals Molecular Regulation of Photosynthesis by Epichloe endophyte in Achnatherum inebrians under Blumeria graminis Infection. J. Fungi 2022, 8, 1201. [Google Scholar] [CrossRef]
- Li, X.Z.; Simpson, W.R.; Song, M.L.; Bao, G.S.; Niu, X.L.; Zhang, Z.H.; Xu, H.F.; Liu, X.; Li, Y.L.; Li, C.J. Effects of seed moisture content and Epichloe endophyte on germination and physiology of Achnatherum inebrians. S. Afr. J. Bot. 2020, 134, 407–414. [Google Scholar] [CrossRef]
- Thor, K. Calcium—Nutrient and Messenger. Front. Plant Sci. 2019, 10, 440. [Google Scholar] [CrossRef] [PubMed]
- Dodds, P.N.; Rathjen, J.P. Plant immunity: Towards an integrated view of plant–pathogen interactions. Nat. Rev. Genet. 2010, 11, 539–548. [Google Scholar] [CrossRef]
- Vadassery, J.; Ranf, S.; Drzewiecki, C.; Mithöfer, A.; Mazars, C.; Scheel, D.; Lee, J.; Oelmüller, R. A cell wall extract from the endophytic fungus Piriformospora indica promotes growth of Arabidopsis seedlings and induces intracellular calcium elevation in roots. Plant J. 2009, 59, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Wicaksono, W.A.; Jones, E.E.; Casonato, S.; Monk, J.; Ridgway, H.J. Biological control of Pseudomonas syringae pv. actinidiae (Psa), the causal agent of bacterial canker of kiwifruit, using endophytic bacteria recovered from a medicinal plant. Biol. Control 2018, 116, 103–112. [Google Scholar]
- Carrieri, R.; Borriello, G.; Piccirillo, G.; Lahoz, E.; Sorrentino, R.; Cermola, M.; Censi, S.B.; Grauso, L.; Mangoni, A.; Vinale, F. Antibiotic Activity of a Paraphaeosphaeria sporulosa-Produced Diketopiperazine against Salmonella enterica. J. Fungi 2020, 6, 83. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, M.; Abraham, W.R. Antimicrobial and Biofilm Inhibiting Diketopiperazines. Curr. Med. Chem. 2012, 19, 3564–3577. [Google Scholar] [CrossRef]
- Tembo, B.; Mulenga, R.M.; Sichilima, S.; M’siska, K.K.; Mwale, M.; Chikoti, P.C.; Singh, P.K.; He, X.; Pedley, K.F.; Peterson, G.L. Detection and characterization of fungus (Magnaporthe oryzae pathotype Triticum) causing wheat blast disease on rain-fed grown wheat (Triticum aestivum L.) in Zambia. PLoS ONE 2020, 15, e0238724. [Google Scholar] [CrossRef]
- di Menna, M.E.; Finch, S.C.; Popay, A.J.; Smith, B.L. A review of the Neotyphodium lolii/Lolium perenne symbiosis and its associated effects on animal and plant health, with particular emphasis on ryegrass staggers. N. Z. Vet. J. 2012, 60, 315–328. [Google Scholar] [CrossRef]
- Şahin, M.; Bayram, G.K.; Can, M.; Ayan, İ.; Acar, Z. Perennıal Ryegrass (Lolium perenne L.). Ph.D. Thesis, Oregon State University, Corvallis, OR, USA, 2022. [Google Scholar]
- Zabaleta, G.; Lee, S.T.; Cook, D.; Aguilar, M.; Iannone, L.J.; Robles, C.; Martinez, A. Indole-diterpenes alkaloid profiles of native grasses involved in tremorgenic syndromes in the Argentine Patagonia. Toxicon 2022, 217, 107–111. [Google Scholar] [CrossRef]
- Currie, A.F.; Gange, A.C.; Ab Razak, N.; Ellison, C.A.; Maczey, N.; Wood, S.V. Endophytic fungi in the invasive weed Impatiens glandulifera: A barrier to classical biological control? Weed Res. 2020, 60, 50–59. [Google Scholar] [CrossRef]
- Sharma, A.; Kaushik, N.; Sharma, A.; Marzouk, T.; Djébali, N. Exploring the potential of endophytes and their metabolites for bio-control activity. 3 Biotech 2022, 12, 277. [Google Scholar] [CrossRef] [PubMed]
- Caruso, D.J.; Palombo, E.A.; Moulton, S.E.; Zaferanloo, B. Exploring the Promise of Endophytic Fungi: A Review of Novel Antimicrobial Compounds. Microorganisms 2022, 10, 1990. [Google Scholar] [CrossRef] [PubMed]
- Dubey, A.; Malla, M.A.; Kumar, A.; Dayanandan, S.; Khan, M.L. Plants endophytes: Unveiling hidden agenda for bioprospecting toward sustainable agriculture. Crit. Rev. Biotechnol. 2020, 40, 1210–1231. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Kenawy, A.; Dailin, D.J.; Abo-Zaid, G.A.; Malek, R.A.; Ambehabati, K.K.; Zakaria, K.H.N.; Sayyed, R.; El Enshasy, H.A. Biosynthesis of antibiotics by PGPR and their roles in biocontrol of plant diseases. In Plant Growth Promoting Rhizobacteria for Sustainable Stress Management: Volume 2: Rhizobacteria in Biotic Stress Management; Springer: Singapore, 2019; pp. 1–35. [Google Scholar]
- Julian, W.T.; Vasilchenko, A.V.; Shpindyuk, D.D.; Poshvina, D.V.; Vasilchenko, A.S. Bacterial-derived plant protection metabolite 2, 4-diacetylphloroglucinol: Effects on bacterial cells at inhibitory and subinhibitory concentrations. Biomolecules 2020, 11, 13. [Google Scholar] [CrossRef] [PubMed]
- Aleti, G. Secondary Metabolite Biosynthetic Gene Diversity–Bacillales Non-Ribosomal Peptides and Polyketides in Plant-Microbe Interactions. Ph.D. Thesis, Austrian Institute of Technology, Vienna, Austria, 2016. [Google Scholar]
- Eid, A.M.; Salim, S.S.; Hassan, S.E.-D.; Ismail, M.A.; Fouda, A. Role of endophytes in plant health and abiotic stress management. In Microbiome in Plant Health and Disease; Springer: Singapore, 2019; pp. 119–144. [Google Scholar]
- Pandey, P.K.; Samanta, R.; Yadav, R.N.S. Inside the plant: Addressing bacterial endophytes in biotic stress alleviation. Arch. Microbiol. 2019, 201, 415–429. [Google Scholar] [CrossRef]
- Brosset, A.; Blande, J.D. Volatile-mediated plant–plant interactions: Volatile organic compounds as modulators of receiver plant defence, growth, and reproduction. J. Exp. Bot. 2022, 73, 511–528. [Google Scholar] [CrossRef]
- Bastías, D.A.; Martínez-Ghersa, M.A.; Newman, J.A.; Card, S.D.; Mace, W.J.; Gundel, P.E. Jasmonic acid regulation of the anti-herbivory mechanism conferred by fungal endophytes in grasses. J. Ecol. 2018, 106, 2365–2379. [Google Scholar] [CrossRef]
- Zhu, J.; He, Y.; Yan, X.; Liu, L.; Guo, R.; Xia, X.; Cheng, D.; Mi, X.; Samarina, L.; Liu, S.; et al. Duplication and transcriptional divergence of three Kunitz protease inhibitor genes that modulate insect and pathogen defenses in tea plant (Camellia sinensis). Hortic. Res. 2019, 6, 126. [Google Scholar] [CrossRef]
- Datta, D.; Behera, L.; Chaudhary, V.T.; Kumar, S.; Bisen, K. Endophytes: Rendering Systemic Resistance to Plants. In Rhizosphere Microbes: Biotic Stress Management; Singh, U.B., Sahu, P.K., Singh, H.V., Sharma, P.K., Sharma, S.K., Eds.; Springer Nature Singapore: Singapore, 2022; pp. 175–195. [Google Scholar]
- Mastan, A.; Rane, D.; Dastager, S.G.; Babu, C.V. Development of low-cost plant probiotic formulations of functional endophytes for sustainable cultivation of Coleus forskohlii. Microbiol. Res. 2019, 227, 126310. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Peng, H.; Li, W.; Cheng, P.; Gong, M. The Toxins of Beauveria bassiana and the Strategies to Improve Their Virulence to Insects. Front. Microbiol. 2021, 12, 705343. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.; Shang, Y.; Cen, K.; Wang, C. Fungal biosynthesis of the bibenzoquinone oosporein to evade insect immunity. Proc. Natl. Acad. Sci. USA 2015, 112, 11365–11370. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, B.J.; Short, S.M.; Dimopoulos, G. Chapter 15—Employing the Mosquito Microflora for Disease Control. In Genetic Control of Malaria and Dengue; Adelman, Z.N., Ed.; Academic Press: Boston, MA, USA, 2016; pp. 335–362. [Google Scholar]
- Vitorino, L.C.; Silva, F.G.; Soares, M.A.; Souchie, E.L.; Costa, A.C.; Lima, W.C. Solubilization of calcium and iron phosphate and in vitro production of Indoleacetic acid by Endophytic isolates of Hyptis marrubioides Epling (Lamiaceae). Int. Res. J. Biotechnol. 2012, 3, 47–54. [Google Scholar]
- Nemhauser, J.L. Back to basics: What is the function of an Aux/IAA in auxin response? New Phytol. 2018, 218, 1295–1297. [Google Scholar] [CrossRef]
- Khan, A.L.; Hussain, J.; Al-Harrasi, A.; Al-Rawahi, A.; Lee, I.-J. Endophytic fungi: Resource for gibberellins and crop abiotic stress resistance. Crit. Rev. Biotechnol. 2015, 35, 62–74. [Google Scholar]
- Liu, R.; Yang, L.; Zou, Y.; Wu, Q. Root-associated endophytic fungi modulate endogenous auxin and cytokinin levels to improve plant biomass and root morphology of trifoliate orange. Hortic. Plant J. 2022. [Google Scholar] [CrossRef]
- Vanderstraeten, L.; Van Der Straeten, D. Accumulation and Transport of 1-Aminocyclopropane-1-Carboxylic Acid (ACC) in Plants: Current Status, Considerations for Future Research and Agronomic Applications. Front Plant Sci 2017, 8, 38. [Google Scholar] [CrossRef]
- Gupta, S.; Pandey, S. ACC deaminase producing bacteria with multifarious plant growth promoting traits alleviates salinity stress in French bean (Phaseolus vulgaris) plants. Front. Microbiol 2019, 10, 1506. [Google Scholar] [CrossRef]
- Roy Choudhury, A.; Choi, J.; Walitang, D.I.; Trivedi, P.; Lee, Y.; Sa, T. ACC deaminase and indole acetic acid producing endophytic bacterial co-inoculation improves physiological traits of red pepper (Capsicum annum L.) under salt stress. J. Plant Physiol. 2021, 267, 153544. [Google Scholar] [CrossRef]
- Canellas, L.P.; Olivares, F.L. Physiological responses to humic substances as plant growth promoter. Chem. Biol. Technol. Agric. 2014, 1, 3. [Google Scholar] [CrossRef]
- Sponsel, V.M. Gibberellins. In Encyclopedia of Hormones; Henry, H.L., Norman, A.W., Eds.; Academic Press: New York, NY, USA, 2003; pp. 29–40. [Google Scholar]
- Gao, X.; Zhang, Y.; He, Z.; Fu, X. 4—Gibberellins. In Hormone Metabolism and Signaling in Plants; Li, J., Li, C., Smith, S.M., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 107–160. [Google Scholar]
- Mukherjee, A.; Gaurav, A.K.; Singh, S.; Yadav, S.; Bhowmick, S.; Abeysinghe, S.; Verma, J.P. The Bioactive potential of phytohormones: A review. Biotechnol. Rep. 2022, 35, e00748. [Google Scholar] [CrossRef]
- Leitão, A.L.; Enguita, F.J. Gibberellins in Penicillium strains: Challenges for endophyte-plant host interactions under salinity stress. Microbiol. Res. 2016, 183, 8–18. [Google Scholar] [CrossRef]
- Muñoz-Espinoza, V.A.; López-Climent, M.F.; Casaretto, J.A.; Gómez-Cadenas, A. Water stress responses of tomato mutants impaired in hormone biosynthesis reveal abscisic acid, jasmonic acid and salicylic acid interactions. Front. Plant Sci. 2015, 6, 997. [Google Scholar] [CrossRef]
- Wang, X.-M.; Yang, B.; Ren, C.-G.; Wang, H.-W.; Wang, J.-Y.; Dai, C.-C. Involvement of abscisic acid and salicylic acid in signal cascade regulating bacterial endophyte-induced volatile oil biosynthesis in plantlets of Atractylodes lancea. Physiol. Plant. 2015, 153, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.; Tsuda, K. Salicylic acid and jasmonic acid crosstalk in plant immunity. Essays Biochem. 2022, 66, 647–656. [Google Scholar]
- Cui, X.; He, W.; Christensen, M.J.; Yue, J.; Zeng, F.; Zhang, X.; Nan, Z.; Xia, C. Abscisic Acid May Play a Critical Role in the Moderating Effect of Epichloe Endophyte on Achnatherum inebrians under Drought Stress. J. Fungi 2022, 8, 1140. [Google Scholar] [CrossRef] [PubMed]
- Waqas, M.; Khan, A.L.; Kamran, M.; Hamayun, M.; Kang, S.-M.; Kim, Y.-H.; Lee, I.-J. Endophytic Fungi Produce Gibberellins and Indoleacetic Acid and Promotes Host-Plant Growth during Stress. Molecules 2012, 17, 10754–10773. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Awasthi, L.P.; Verma, H.N. Chapter 42—Systemic resistance inducers from plants—An ecofriendly approach for the management of viral diseases of crops. In Applied Plant Virology; Awasthi, L.P., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 603–617. [Google Scholar]
- Lefevere, H.; Bauters, L.; Gheysen, G. Salicylic Acid Biosynthesis in Plants. Front. Plant Sci. 2020, 11, 338. [Google Scholar] [CrossRef]
- Jisha, S.; Sabu, K.; Manjula, S. Multifunctional aspects of Piriformospora indica in plant endosymbiosis. Mycology 2019, 10, 182–190. [Google Scholar] [CrossRef]
- Mollah, M.M.I.; Choi, H.W.; Yeam, I.; Lee, J.M.; Kim, Y. Salicylic Acid, a Plant Hormone, Suppresses Phytophagous Insect Immune Response by Interrupting HMG-Like DSP1. Front. Physiol. 2021, 12, 744272. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.; Raikhy, G.; Kumar, D. Chemical elicitors of systemic acquired resistance—Salicylic acid and its functional analogs. Curr. Plant Biol. 2019, 17, 48–59. [Google Scholar] [CrossRef]
- Zhou, Q.; Meng, Q.; Tan, X.; Ding, W.; Ma, K.; Xu, Z.; Huang, X.; Gao, H. Protein Phosphorylation Changes during Systemic Acquired Resistance in Arabidopsis thaliana. Front Plant Sci. 2021, 12, 748287. [Google Scholar] [CrossRef] [PubMed]
- Haq, S.K.; Atif, S.M.; Khan, R.H. Protein proteinase inhibitor genes in combat against insects, pests, and pathogens: Natural and engineered phytoprotection. Arch. Biochem. Biophys. 2004, 431, 145–159. [Google Scholar] [CrossRef]
- De Leo, F.; Volpicella, M.; Licciulli, F.; Liuni, S.; Gallerani, R.; Ceci, L.R. PLANT-PIs: A database for plant protease inhibitors and their genes. Nucleic Acids Res. 2002, 30, 347–348. [Google Scholar] [CrossRef]
- He, Y.; Chen, T.; Zhang, H.; White, J.F.; Li, C. Fungal Endophytes Help Grasses to Tolerate Sap-Sucking Herbivores through a Hormone-Signaling System. J. Plant Growth Regul. 2022, 41, 2122–2137. [Google Scholar] [CrossRef]
- Lyons, R.; Manners, J.M.; Kazan, K. Jasmonate biosynthesis and signaling in monocots: A comparative overview. Plant Cell Rep. 2013, 32, 815–827. [Google Scholar] [CrossRef] [PubMed]
- Rynkiewicz, M.J.; Cane, D.E.; Christianson, D.W. Structure of trichodiene synthase from Fusarium sporotrichioides provides mechanistic inferences on the terpene cyclization cascade. Proc. Natl. Acad. Sci. USA 2001, 98, 13543–13548. [Google Scholar] [CrossRef]
- Grosskinsky, D.K.; Naseem, M.; Abdelmohsen, U.R.; Plickert, N.; Engelke, T.; Griebel, T.; Zeier, J.; Novák, O.; Strnad, M.; Pfeifhofer, H.; et al. Cytokinins mediate resistance against Pseudomonas syringae in tobacco through increased antimicrobial phytoalexin synthesis independent of salicylic acid signaling. Plant Physiol. 2011, 157, 815–830. [Google Scholar] [CrossRef]
- Choi, J.; Huh, S.U.; Kojima, M.; Sakakibara, H.; Paek, K.-H.; Hwang, I. The Cytokinin-Activated Transcription Factor ARR2 Promotes Plant Immunity via TGA3/NPR1-Dependent Salicylic Acid Signaling in Arabidopsis. Dev. Cell 2010, 19, 284–295. [Google Scholar] [CrossRef]
- Großkinsky, D.K.; Tafner, R.; Moreno, M.V.; Stenglein, S.A.; García De Salamone, I.E.; Nelson, L.M.; Novák, O.; Strnad, M.; Van Der Graaff, E.; Roitsch, T. Cytokinin production by Pseudomonas fluorescens G20-18 determines biocontrol activity against Pseudomonas syringae in Arabidopsis. Sci. Rep. 2016, 6, 23310. [Google Scholar] [CrossRef] [PubMed]
- Jameson, P. Cytokinins and auxins in plant-pathogen interactions–An overview. Plant Growth Regul. 2000, 32, 369–380. [Google Scholar] [CrossRef]
- Babosha, A.V. Regulation of resistance and susceptibility in wheat–powdery mildew pathosystem with exogenous cytokinins. J. Plant Physiol. 2009, 166, 1892–1903. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Anand, G.; Pizarro, L.; Bar-Yosef, D.L.; Kovetz, N.; Sela, N.; Yehuda, T.; Gazit, E.; Bar, M. Cytokinin Inhibits Fungal Development and Virulence by Targeting the Cytoskeleton and Cellular Trafficking. mBio 2021, 12, e03068-20. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhou, J.; Xu, R.-S.; Meng, M.-Y.; Yu, X.; Dai, C.-C. Auxin, Cytokinin, and Ethylene Involved in Rice N Availability Improvement Caused by Endophyte Phomopsis liquidambari. J. Plant Growth Regul. 2018, 37, 128–143. [Google Scholar] [CrossRef]
- Sachdev, S.; Ansari, S.A.; Ansari, M.I.; Fujita, M.; Hasanuzzaman, M. Abiotic Stress and Reactive Oxygen Species: Generation, Signaling, and Defense Mechanisms. Antioxidants 2021, 10, 277. [Google Scholar] [CrossRef] [PubMed]
- Mhamdi, A.; Van Breusegem, F. Reactive oxygen species in plant development. Development 2018, 145, dev164376. [Google Scholar] [CrossRef] [PubMed]
- Bela, K.; Horváth, E.; Gallé, Á.; Szabados, L.; Tari, I.; Csiszár, J. Plant glutathione peroxidases: Emerging role of the antioxidant enzymes in plant development and stress responses. J. Plant Physiol. 2015, 176, 192–201. [Google Scholar] [CrossRef]
- Tomar, R.S.; Kataria, S.; Jajoo, A. Behind the scene: Critical role of reactive oxygen species and reactive nitrogen species in salt stress tolerance. J. Agron. Crop Sci. 2021, 207, 577–588. [Google Scholar] [CrossRef]
- Hereme, R.; Morales-Navarro, S.; Ballesteros, G.; Barrera, A.; Ramos, P.; Gundel, P.E.; Molina-Montenegro, M.A. Fungal Endophytes Exert Positive Effects on Colobanthus quitensis Under Water Stress but Neutral Under a Projected Climate Change Scenario in Antarctica. Front. Microbiol. 2020, 11, 264. [Google Scholar] [CrossRef]
- Mahgoub, H.A.M.; Fouda, A.; Eid, A.M.; Ewais, E.E.-D.; Hassan, S.E.-D. Biotechnological application of plant growth-promoting endophytic bacteria isolated from halophytic plants to ameliorate salinity tolerance of Vicia faba L. Plant Biotechnol. Rep. 2021, 15, 819–843. [Google Scholar] [CrossRef]
- Khan, M.A.; Asaf, S.; Khan, A.L.; Ullah, I.; Ali, S.; Kang, S.-M.; Lee, I.-J. Alleviation of salt stress response in soybean plants with the endophytic bacterial isolate Curtobacterium sp. SAK1. Ann. Microbiol. 2019, 69, 797–808. [Google Scholar] [CrossRef]
- Iqbal, N.; Umar, S.; Khan, N. Nitrogen availability regulates proline and ethylene production and alleviates salinity stress in mustard. J. Plant Physiol. 2015, 178, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Harter, K.; Theologis, A. Protein-protein interactions among the Aux/IAA proteins. Proc. Natl. Acad. Sci. USA 1997, 94, 11786–11791. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Zhang, H.-T.; Wang, Y.; Jia, W.-S.; Xu, X.-F.; Zhang, X.-Z.; Han, Z.H. Induction of root Fe(lll) reductase activity and proton extrusion by iron deficiency is mediated by auxin-based systemic signalling in Malus xiaojinensis. J. Exp. Bot. 2011, 63, 859–870. [Google Scholar] [CrossRef]
- Higgins, T.J.; Jacobsen, J.V.; Zwar, J.A. Gibberellic acid and abscisic acid modulate protein synthesis and mRNA levels in barley aleurone layers. Plant Mol. Biol. 1982, 1, 191–215. [Google Scholar] [CrossRef]
- Raghavendra, A.S.; Gonugunta, V.K.; Christmann, A.; Grill, E. ABA perception and signalling. Trends Plant Sci. 2010, 15, 395–401. [Google Scholar] [CrossRef]
- Fadiji, A.E.; Babalola, O.O. Elucidating Mechanisms of Endophytes Used in Plant Protection and Other Bioactivities With Multifunctional Prospects. Front. Bioeng. Biotechnol. 2020, 8, 467. [Google Scholar] [CrossRef]
- Shahid, M.A.; Sarkhosh, A.; Khan, N.; Balal, R.M.; Ali, S.; Rossi, L.; Gómez, C.; Mattson, N.; Nasim, W.; Garcia-Sanchez, F. Insights into the physiological and biochemical impacts of salt stress on plant growth and development. Agronomy 2020, 10, 938. [Google Scholar] [CrossRef]
- Schmidt, W.; Thomine, S.; Buckhout, T.J. Iron nutrition and interactions in plants. Front. Plant Sci. 2020, 10, 1670. [Google Scholar] [CrossRef]
- Murata, Y.; Itoh, Y.; Iwashita, T.; Namba, K. Transgenic Petunia with the Iron(III)-Phytosiderophore Transporter Gene Acquires Tolerance to Iron Deficiency in Alkaline Environments. PLoS ONE 2015, 10, e0120227. [Google Scholar] [CrossRef] [PubMed]
- Cesco, S.; Rombolà, A.D.; Tagliavini, M.; Varanini, Z.; Pinton, R. Phytosiderophores released by graminaceous species promote 59Fe-uptake in citrus. Plant Soil 2006, 287, 223–233. [Google Scholar] [CrossRef]
- Banakar, R.; Alvarez Fernandez, A.; Díaz-Benito, P.; Abadia, J.; Capell, T.; Christou, P. Phytosiderophores determine thresholds for iron and zinc accumulation in biofortified rice endosperm while inhibiting the accumulation of cadmium. J. Exp. Bot. 2017, 68, 4983–4995. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Rajkumar, M.; Zhang, C.; Freitas, H. Beneficial role of bacterial endophytes in heavy metal phytoremediation. J. Environ. Manag. 2016, 174, 14–25. [Google Scholar] [CrossRef]
- Du, Y.-C.; Kong, L.-J.; Cao, L.-S.; Zhang, W.; Zhu, Q.; Ma, C.-Y.; Sun, K.; Dai, C.-C. Endophytic Fungus Phomopsis liquidambaris Enhances Fe Absorption in Peanuts by Reducing Hydrogen Peroxide. Front. Plant Sci. 2022, 13, 872242. [Google Scholar] [CrossRef] [PubMed]
- Long, L.; Persson, D.P.; Duan, F.; Jørgensen, K.; Yuan, L.; Schjoerring, J.K.; Pedas, P.R. The iron-regulated transporter 1 plays an essential role in uptake, translocation and grain-loading of manganese, but not iron, in barley. New Phytol. 2018, 217, 1640–1653. [Google Scholar] [CrossRef]
- Nielsen, F.H. History of zinc in agriculture. Adv. Nutr. 2012, 3, 783–789. [Google Scholar] [CrossRef]
- Umair Hassan, M.; Aamer, M.; Umer Chattha, M.; Haiying, T.; Shahzad, B.; Barbanti, L.; Nawaz, M.; Rasheed, A.; Afzal, A.; Liu, Y. The critical role of zinc in plants facing the drought stress. Agriculture 2020, 10, 396. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, B.; Raigond, P.; Sahu, C.; Mishra, U.N.; Sharma, S.; Lal, M.K. Phytic acid: Blessing in disguise, a prime compound required for both plant and human nutrition. Food Res. Int. 2021, 142, 110193. [Google Scholar] [CrossRef]
- Wang, S.; Li, L.; Ying, Y.; Wang, J.; Shao, J.F.; Yamaji, N.; Whelan, J.; Ma, J.F.; Shou, H. A transcription factor OsbHLH156 regulates Strategy II iron acquisition through localising IRO2 to the nucleus in rice. New Phytol. 2020, 225, 1247–1260. [Google Scholar] [CrossRef]
- Broadley, M.R.; White, P.J.; Hammond, J.P.; Zelko, I.; Lux, A. Zinc in plants. New Phytol. 2007, 173, 677–702. [Google Scholar] [CrossRef] [PubMed]
- Jahandideh Mahjen Abadi, V.A.; Sepehri, M.; Khatabi, B.; Rezaei, M. Alleviation of zinc deficiency in wheat inoculated with root endophytic fungus Piriformospora indica and rhizobacterium Pseudomonas putida. Rhizosphere 2021, 17, 100311. [Google Scholar] [CrossRef]
- Padda, K.P.; Puri, A.; Chanway, C. Endophytic nitrogen fixation—A possible ‘hidden’ source of nitrogen for lodgepole pine trees growing at unreclaimed gravel mining sites. FEMS Microbiol. Ecol. 2019, 95, fiz172. [Google Scholar] [CrossRef] [PubMed]
- Puri, A.; Padda, K.P.; Chanway, C.P. Evidence of endophytic diazotrophic bacteria in lodgepole pine and hybrid white spruce trees growing in soils with different nutrient statuses in the West Chilcotin region of British Columbia, Canada. For. Ecol. Manag. 2018, 430, 558–565. [Google Scholar] [CrossRef]
- Reis, I.A.; Souza, M.G.D.; Granja-Salcedo, Y.T.; Carvalho, I.P.C.D.; Porcionato, M.A.D.F.; Prados, L.F.; Siqueira, G.R.; De Resende, F.D. Effect of Post-Ruminal Urea Supply on Growth Performance of Grazing Nellore Young Bulls at Dry Season. Animals 2023, 13, 207. [Google Scholar] [CrossRef]
- Scherger, L.E.; Zanello, V.; Lexow, C. Impact of urea and ammoniacal nitrogen wastewaters on soil: Field study in a fertilizer industry (Bahía Blanca, Argentina). Bull. Environ. Contam. Toxicol. 2021, 107, 565–573. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, Y.; Wang, K.; Zhang, Y.; Yu, N.; Liu, Z. Effects of urea application on the reproduction of Pardosa pseudoannulata: Field and laboratory studies. Chemosphere 2022, 301, 134697. [Google Scholar] [CrossRef]
- Varga, T.; Hixson, K.K.; Ahkami, A.H.; Sher, A.W.; Barnes, M.E.; Chu, R.K.; Battu, A.K.; Nicora, C.D.; Winkler, T.E.; Reno, L.R.; et al. Endophyte-Promoted Phosphorus Solubilization in Populus. Front. Plant Sci. 2020, 11, 567918. [Google Scholar] [CrossRef]
- Emami, S.; Alikhani, H.A.; Pourbabaee, A.A.; Etesami, H.; Motasharezadeh, B.; Sarmadian, F. Consortium of endophyte and rhizosphere phosphate solubilizing bacteria improves phosphorous use efficiency in wheat cultivars in phosphorus deficient soils. Rhizosphere 2020, 14, 100196. [Google Scholar] [CrossRef]
- Hegedűs, M.; Tóth-Bodrogi, E.; Németh, S.; Somlai, J.; Kovács, T. Radiological investigation of phosphate fertilizers: Leaching studies. J. Environ. Radioact. 2017, 173, 34–43. [Google Scholar] [CrossRef]
- Chhabra, S.; Dowling, D.N. Endophyte-Promoted Nutrient Acquisition: Phosphorus and Iron; Springer International Publishing: Cham, Switzerland, 2017; pp. 21–42. [Google Scholar]
- Baghel, V.; Thakur, J.K.; Yadav, S.S.; Manna, M.C.; Mandal, A.; Shirale, A.O.; Sharma, P.; Sinha, N.K.; Mohanty, M.; Singh, A.B.; et al. Phosphorus and Potassium Solubilization From Rock Minerals by Endophytic Burkholderia sp. Strain FDN2-1 in Soil and Shift in Diversity of Bacterial Endophytes of Corn Root Tissue with Crop Growth Stage. Geomicrobiol. J. 2020, 37, 550–563. [Google Scholar] [CrossRef]
- Kumar, M.; Yadav, V.; Kumar, H.; Sharma, R.; Singh, A.; Tuteja, N.; Johri, A.K. Piriformospora indica enhances plant growth by transferring phosphate. Plant Signal. Behav. 2011, 6, 723–725. [Google Scholar] [CrossRef] [PubMed]
- Etesami, H.; Jeong, B.R.; Glick, B.R. Contribution of arbuscular mycorrhizal fungi, phosphate–solubilizing bacteria, and silicon to P uptake by plant. Front. Plant Sci. 2021, 12, 699618. [Google Scholar] [CrossRef] [PubMed]
- Bargaz, A.; Elhaissoufi, W.; Khourchi, S.; Benmrid, B.; Borden, K.A.; Rchiad, Z. Benefits of phosphate solubilizing bacteria on belowground crop performance for improved crop acquisition of phosphorus. Microbiol. Res. 2021, 252, 126842. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ren, A.; Han, R.; Yin, L.; Wei, M.; Gao, Y. Endophyte-Mediated Effects on the Growth and Physiology of Achnatherum sibiricum Are Conditional on Both N and P Availability. PLoS ONE 2012, 7, e48010. [Google Scholar] [CrossRef]
- Hafsi, C.; Debez, A.; Abdelly, C. Potassium deficiency in plants: Effects and signaling cascades. Acta Physiol. Plant. 2014, 36, 1055–1070. [Google Scholar] [CrossRef]
- Sakaguchi, T.; Nishizawa, N.K.; Nakanishi, H.; Yoshimura, E.; Mori, S. The role of potassium in the secretion of mugineic acids familyphytosiderophores from iron-deficient barley roots. Plant Soil. 1999, 215, 221–227. [Google Scholar] [CrossRef]
- Takagi, S.-i. Naturally occurring iron-chelating compounds in oat-and rice-root washings: I. Activity measurement and preliminary characterization. Soil Sci. Plant Nutr. 1976, 22, 423–433. [Google Scholar] [CrossRef]
- Loka, D.A.; Oosterhuis, D.M.; Baxevanos, D.; Vlachostergios, D.; Hu, W. How potassium deficiency alters flower bud retention on cotton (Gossypium hirsutum L.). Arch. Agron. Soil Sci. 2019, 65, 521–536. [Google Scholar] [CrossRef]
- Hu, W.; Liu, Y.; Loka, D.A.; Zahoor, R.; Wang, S.; Zhou, Z. Drought limits pollen tube growth rate by altering carbohydrate metabolism in cotton (Gossypium hirsutum) pistils. Plant Sci. 2019, 286, 108–117. [Google Scholar] [CrossRef]
- Shaffer, J.P.; Carter, M.E.; Spraker, J.E.; Clark, M.; Smith, B.A.; Hockett, K.L.; Baltrus, D.A.; Arnold, A.E. Transcriptional Profiles of a Foliar Fungal Endophyte (Pestalotiopsis, Ascomycota) and Its Bacterial Symbiont (Luteibacter, Gammaproteobacteria) Reveal Sulfur Exchange and Growth Regulation during Early Phases of Symbiotic Interaction. Msystems 2022, 7, e00091-22. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, E.; Galindo-Castañeda, T.; Prada, F.; Navia, M.; Romero, H.M. Phosphate-solubilizing microorganisms associated with the rhizosphere of oil palm (Elaeis guineensis Jacq.) in Colombia. Appl. Soil Ecol. 2014, 80, 26–33. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, U.M.; Manohar, M.; Gaur, V.S. Calcium transport from source to sink: Understanding the mechanism(s) of acquisition, translocation, and accumulation for crop biofortification. Acta Physiol. Plant. 2015, 37, 1722. [Google Scholar] [CrossRef]
- White, J.F.; Kingsley, K.L.; Zhang, Q.; Verma, R.; Obi, N.; Dvinskikh, S.; Elmore, M.T.; Verma, S.K.; Gond, S.K.; Kowalski, K.P. Review: Endophytic microbes and their potential applications in crop management. Pest Manag. Sci. 2019, 75, 2558–2565. [Google Scholar] [CrossRef] [PubMed]
- Shahabivand, S.; Parvaneh, A.; Aliloo, A.A. Root endophytic fungus Piriformospora indica affected growth, cadmium partitioning and chlorophyll fluorescence of sunflower under cadmium toxicity. Ecotoxicol. Environ. Saf. 2017, 145, 496–502. [Google Scholar] [CrossRef]
- Guo, W.; Nazim, H.; Liang, Z.; Yang, D. Magnesium deficiency in plants: An urgent problem. Crop J. 2016, 4, 83–91. [Google Scholar] [CrossRef]
- Tian, X.-Y.; He, D.-D.; Bai, S.; Zeng, W.-Z.; Wang, Z.; Wang, M.; Wu, L.-Q.; Chen, Z.-C. Physiological and molecular advances in magnesium nutrition of plants. Plant Soil. 2021, 468, 1–17. [Google Scholar] [CrossRef]
- Adams, N.B.; Bisson, C.; Brindley, A.A.; Farmer, D.A.; Davison, P.A.; Reid, J.D.; Hunter, C.N. The active site of magnesium chelatase. Nat. Plants 2020, 6, 1491–1502. [Google Scholar] [CrossRef]
- Prasad, D.; Verma, N.; Bakshi, M.; Narayan, O.P.; Singh, A.K.; Dua, M.; Johri, A.K. Functional characterization of a magnesium transporter of root endophytic fungus Piriformospora indica. Front. Microbiol. 2019, 9, 3231. [Google Scholar] [CrossRef]
- Song, J.; Wang, Y.; Pan, Y.; Pang, J.; Zhang, X.; Fan, J.; Zhang, Y. The influence of nitrogen availability on anatomical and physiological responses of Populus alba × P. glandulosa to drought stress. BMC Plant Biol. 2019, 19, 63. [Google Scholar] [CrossRef]
- Ku, Y.S.; Rehman, H.M.; Lam, H.M. Possible Roles of Rhizospheric and Endophytic Microbes to Provide a Safe and Affordable Means of Crop Biofortification. Agronomy 2019, 9, 764. [Google Scholar] [CrossRef]
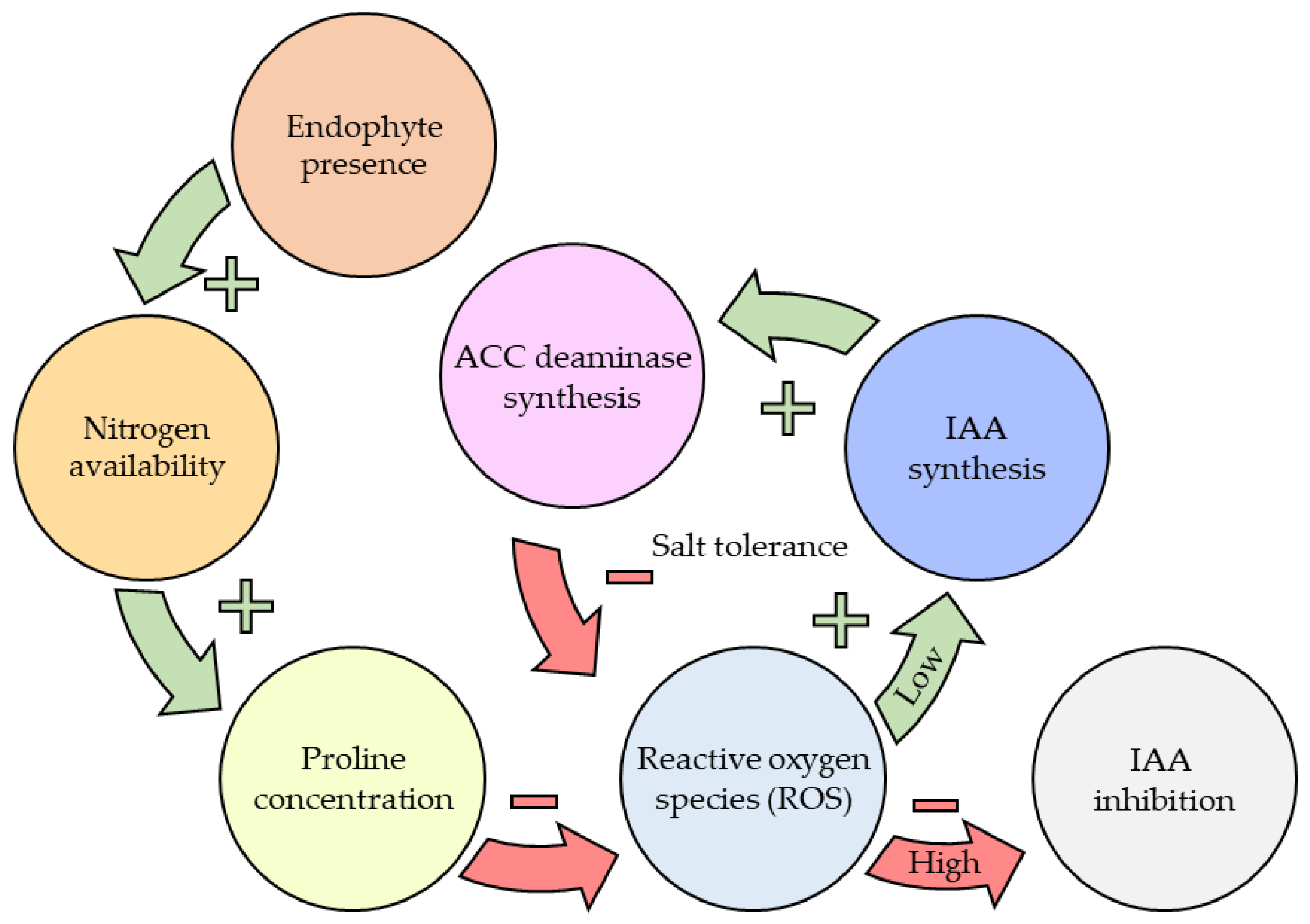
| Signalling Molecule | Effect | Reference |
|---|---|---|
| Auxins (IAA) | Promote cell elongation, root development, and apical dominance. | [83,90,149,191,192] |
| Gibberellins (GA) | Stem elongation, flowering, and leaf spread. | [48,49,150,159,193] |
| Abscisic acid (ABA) | Promote cellular conservation of water under drought and salinity stress. | [83,90,193,194] |
| Salicylic acid (SA) | Induces SAR combatting pathogen infection. | [78,160,161,166,168,169,177] |
| Jasmonic acid (JA) | Defence against pests through deterrence and elimination of pests. | [66,141,142,160,162] |
| Cytokinin’s (CK) | Promotes cell division, apical dominance, lateral root growth. | [151,176,177,178,179,180,181,182] |
| Reactive oxygen species (ROS) | Involved in stress signalling and programmed cell death. Major issues with accumulation resulting in toxification. | [104,133,140,184,186,187,195,196] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watts, D.; Palombo, E.A.; Jaimes Castillo, A.; Zaferanloo, B. Endophytes in Agriculture: Potential to Improve Yields and Tolerances of Agricultural Crops. Microorganisms 2023, 11, 1276. https://doi.org/10.3390/microorganisms11051276
Watts D, Palombo EA, Jaimes Castillo A, Zaferanloo B. Endophytes in Agriculture: Potential to Improve Yields and Tolerances of Agricultural Crops. Microorganisms. 2023; 11(5):1276. https://doi.org/10.3390/microorganisms11051276
Chicago/Turabian StyleWatts, Declan, Enzo A. Palombo, Alex Jaimes Castillo, and Bita Zaferanloo. 2023. "Endophytes in Agriculture: Potential to Improve Yields and Tolerances of Agricultural Crops" Microorganisms 11, no. 5: 1276. https://doi.org/10.3390/microorganisms11051276
APA StyleWatts, D., Palombo, E. A., Jaimes Castillo, A., & Zaferanloo, B. (2023). Endophytes in Agriculture: Potential to Improve Yields and Tolerances of Agricultural Crops. Microorganisms, 11(5), 1276. https://doi.org/10.3390/microorganisms11051276







