Antibacterial and Anti-Inflammatory Activities of Thymus vulgaris Essential Oil Nanoemulsion on Acne Vulgaris
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Their Maintenance
2.2. Essential Oils (EOs)
2.3. Animals
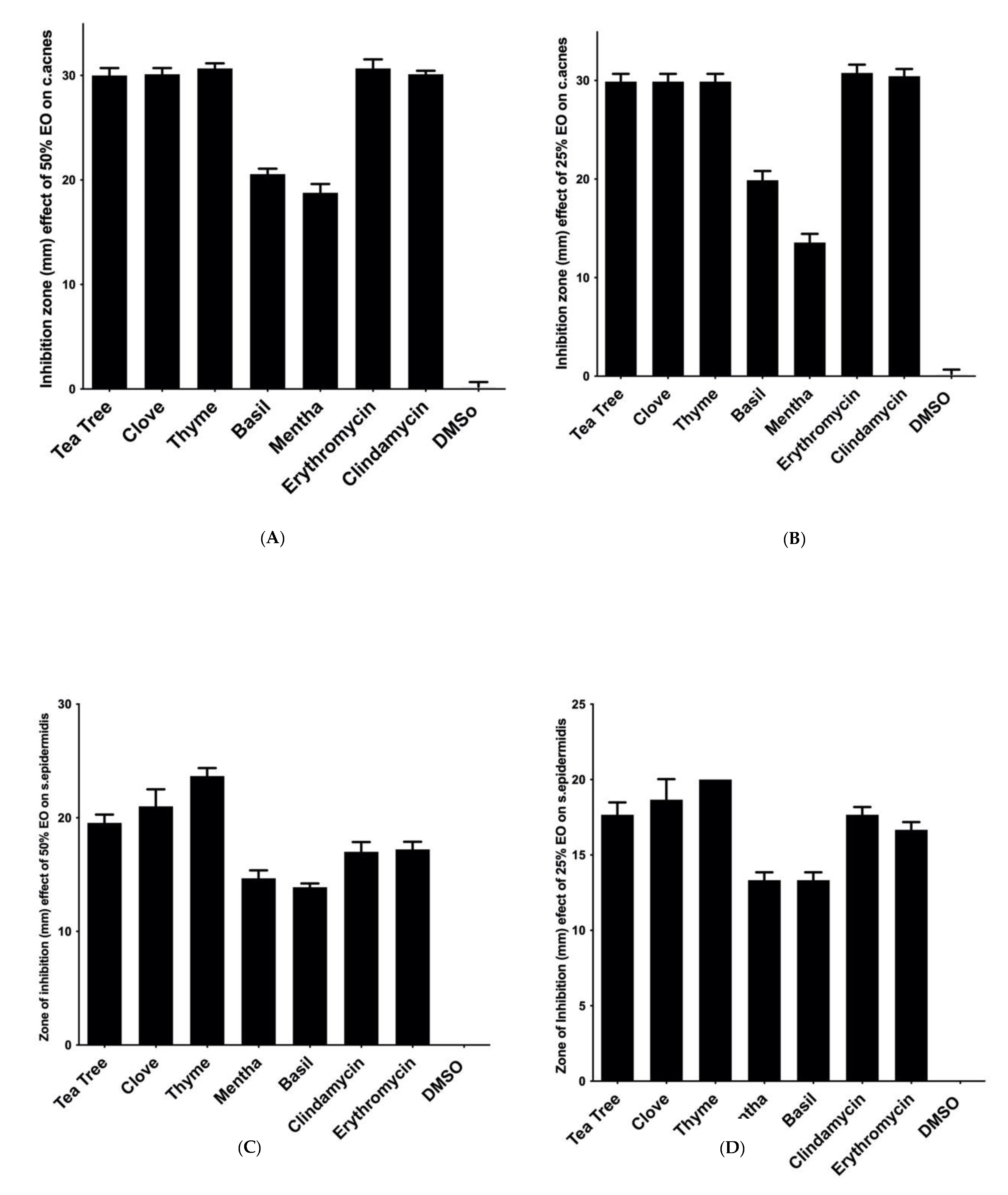
2.4. Screening of Antimicrobial Activity of the Selected EOs by Disc-Diffusion Method
2.5. Determination of MIC and MBC of the Screened EOs against C. acnes and S. epidermidis
2.6. Determination of the Chemical Composition of the Most Effective EOs by Gas Chromatography–Mass Spectroscopy
2.7. Determination of Minimum Biofilm Inhibitory Concentration
2.8. Determination of the Minimum Biofilm Eradication Concentration
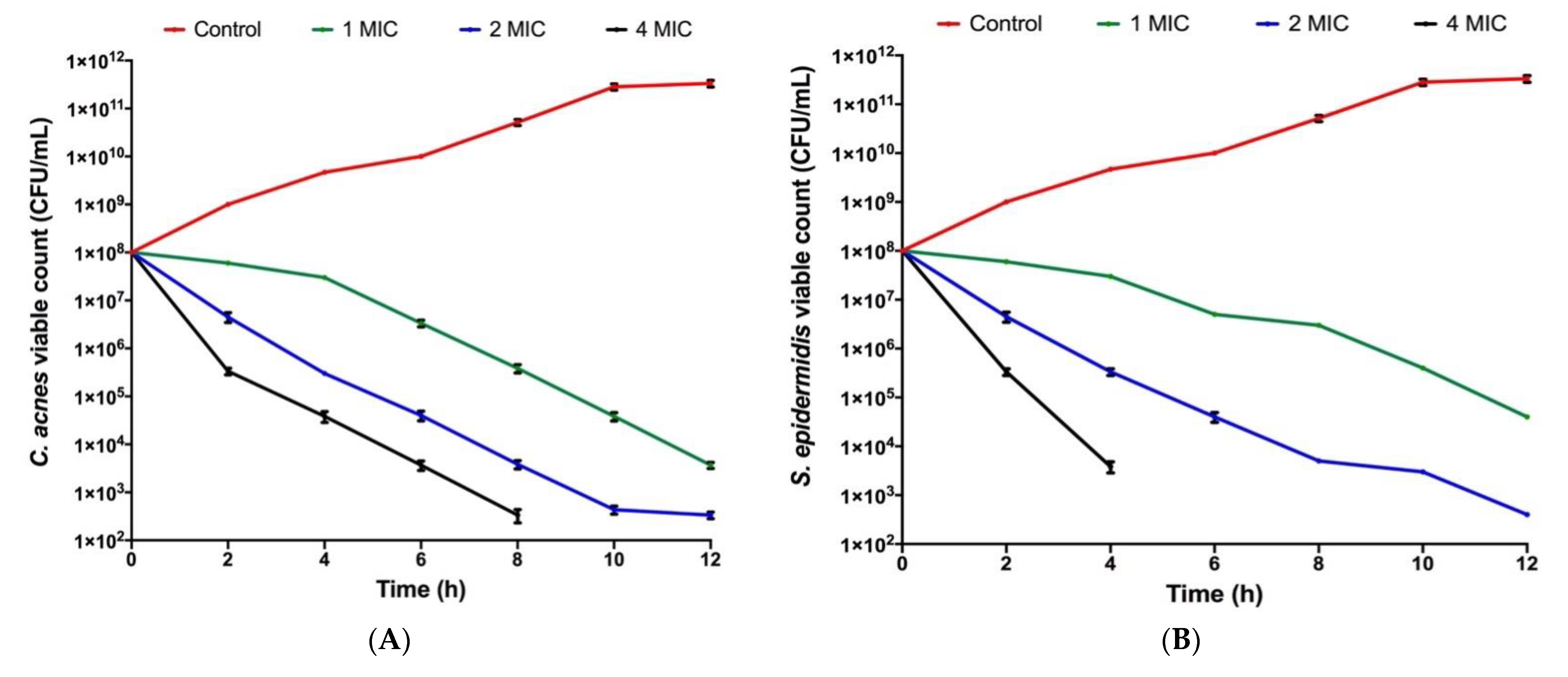
2.9. Determination of Time–Kill Kinetics of Selected EO
2.10. Assessment of Possible Mechanisms of Action of Selected EO against Acne Associated Microbes
2.10.1. Observation of Morphological Alteration of C. acnes and S. epidermidis Treated with Selected EO
Visualization of the Effect of the Selected EO on Biofilm by Scanning Electron Microscopy
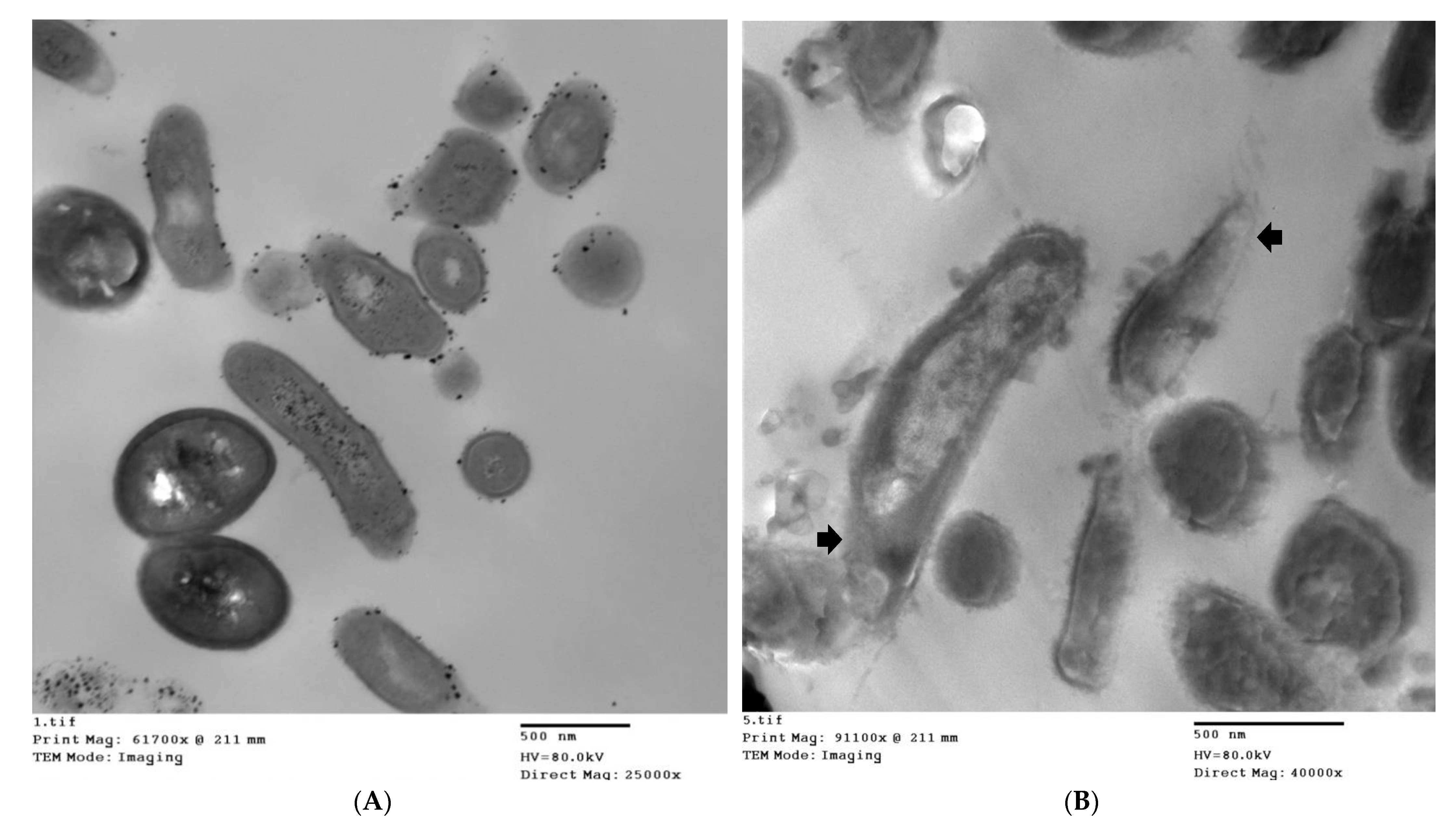
Visualization of the Effect of the Selected EO on Bacterial Cells by Transmission Electron Microscopy
2.10.2. The Effect of the Selected EO on Bacterial Membrane Integrity
The Effect of the Selected EO on Potassium Ions Permeability
The Effect of the Selected EO on 260 nm Absorbing Material (Nucleic Acids)
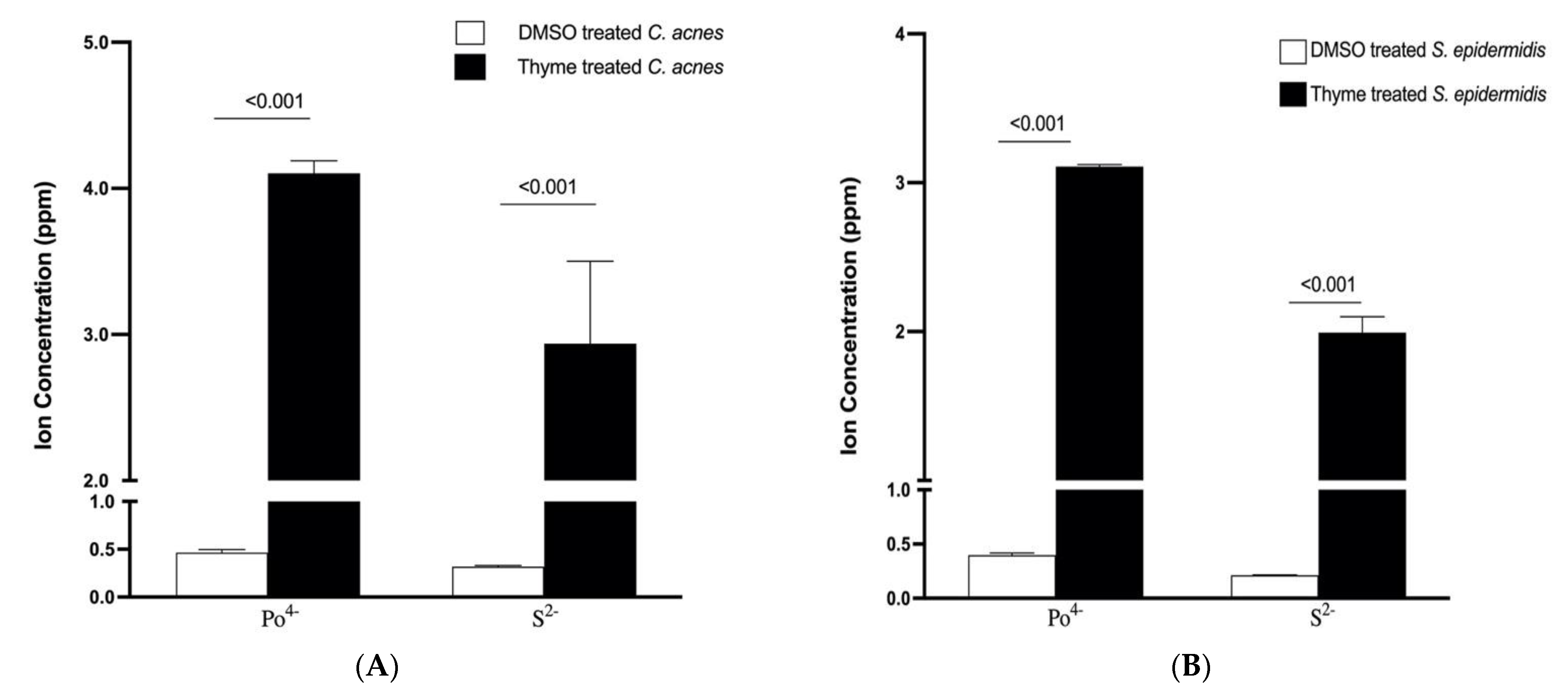
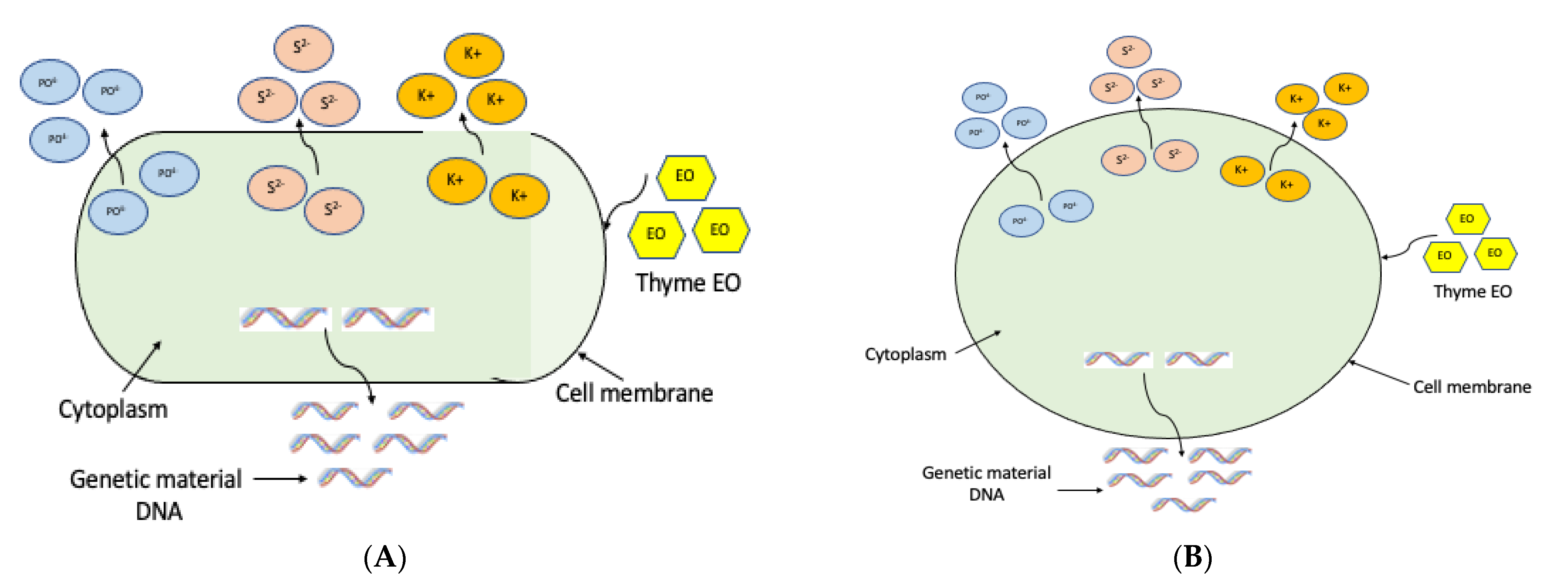
The Effect of the Selected EO on Leakage of Intracellular Ions
2.11. Development and Characterization of the Selected EO Nanoemulsion
2.11.1. Development of the Selected EO Nanoemulsion
2.11.2. Characterization of EO Nanoemulsion
Determination of the Particle Size and Polydispersity Index
Nanoemulsion Morphology Using TEM
2.12. In Vivo Acne Animal Model for Assessment of EO Nanoemulsion Efficacy
2.12.1. Assessment of the Irritability of the EO Nanoemulsion
2.12.2. Experimental Design
2.12.3. Assessment of the Anti-Inflammatory Activity of the Selected EO Nanoemulsion
2.12.4. Assessment of the Anti-Inflammatory Activity of the Selected EO Nanoemulsion on Inflammatory Mediators
2.12.5. Histopathological Examination of Ears after Treatment with EO Nanoemulsion
2.12.6. Assessment of the Antimicrobial Activity of EO Nanoemulsion
2.13. Statistical Analysis
3. Results
3.1. Screening of the Antimicrobial Activity of the Tested EOs by Disc- Diffusion Method
3.2. Determination of the Minimum Inhibition Concentration and the Minimum Bactericidal Concentration of the Screened EOs
3.3. Determination of Chemical Composition of the Most Effective EOs by Gas Chromatography–Mass Spectroscopy
3.4. Determination of the Minimum Biofilm Inhibitory and Eradication Concentration of the Most Potent EOs
3.5. Determination of Time–Kill Kinetics of Selected EO
3.6. Assessment of Possible Mechanisms of Action of Thyme EO on Acne-Associated Microbes
3.6.1. Observation of Morphological Alternations of C. acnes and S. epidermidis Treated with Thyme EO
Visualization of the Effect of the Thyme EO on Bacterial Cells by Transmission Electron Microscopy
Visualization of the Effect of Thyme EO on Biofilm Formation by Scanning Electron Microscopy
3.6.2. Effect of Thyme EO on Bacterial Membrane Integrity
Effect of Thyme EO on Potassium Ion (K+) Permeability
Effect of Thyme EO on 260 nm Absorbing Material (Nucleic Acids)
3.6.3. Effect of Thyme EO on the Leakage of Intracellular Ions
3.7. Development and Characterization of Thyme EO Nanoemulsion
3.7.1. Determination of the Particle Size and Polydispersity Index (PDI)
3.7.2. Nanoemulsion Particles Morphology Using TEM
3.8. In Vivo Acne Animal Model for Assessment of EO Nanoemulsion Efficacy
3.8.1. Assessment of the Irritability of the Thyme Nanoemulsion
3.8.2. Morphological and Histopathological Assessment of the Healing Activity of the Nanoemulsion
3.8.3. Assessment of Anti-Inflammatory Activity of the Thyme Nanoemulsion
3.8.4. Assessment of Anti-Inflammatory Activity of the Thyme Nanoemulsion by Measuring NF-κB Levels
3.8.5. Assessment of the Antibacterial Activity of the Prepared Nanoemulsion
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tuchayi, S.; Makrantonaki, E.; Ganceviciene, R.; Feldman, S.R.; Zouboulis, C.C. Acne vulgaris. Nat. Rev. Dis. Primers 2015, 1, 15029. [Google Scholar] [CrossRef] [PubMed]
- Dunn, L.K.; O’Neill, J.L.; Feldman, S.R. Acne in adolescents: Quality of life, self-esteem, mood and psychological disorders. Dermatol. Online J. 2011, 17, 1. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.M.; Zhao, H.J.; Guo, D.Y.; Zhu, P.Q.; Zhang, C.L.; Jiang, W. Gut microbiota alterations in moderate to severe acne vulgaris patients. J. Dermatol. 2018, 45, 1166–1171. [Google Scholar] [CrossRef] [PubMed]
- Nagy, I.; Pivarcsi, A.; Kis, K.; Koreck, A.; Bodai, L.; McDowell, A.; Kemény, L. Propionibacterium acnes and lipopolysaccharide induce the expression of antimicrobial peptides and proinflammatory cytokines/chemokines in human sebocytes. Microbes Infect. 2006, 8, 2195–2205. [Google Scholar] [CrossRef] [PubMed]
- Fournière, M.; Latire, T.; Souak, D.; Feuilloley, M.G.J.; Bedoux, G. Staphylococcus epidermidis and Cutibacterium acnes: Two major sentinels of skin microbiota and the influence of cosmetics. Microorganisms 2020, 8, 1752. [Google Scholar] [CrossRef] [PubMed]
- Zaenglein, A.L.; Pathy, A.L.; Schlosser, B.J.; Alikhan, A.; Baldwin, H.E.; Berson, D.S.; Bhushan, R. Guidelines of care for the management of acne vulgaris. J. Am. Acad. Dermatol. 2016, 74, 945–973. [Google Scholar] [CrossRef] [PubMed]
- Nazzaro, F.; Fratianni, F.; Martino, D.L.; Coppola, R.; Feo, D.V. Effect of essential oils on pathogenic bacteria. Pharmaceuticals 2013, 6, 1451–1474. [Google Scholar] [CrossRef]
- Miguel, M.G.; Duarte, J.; Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G. Thymus carnosus Boiss.: Effect of harvesting period, collection site and type of plant material on essential oil composition. J. Essent. Oil Res. 2005, 17, 22–426. [Google Scholar] [CrossRef]
- Dorman, H.D.; Deans, S.G. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Pretsch, A.; Nagl, M.; Schwendinger, K.; Kreiseder, B.; Wiederstein, M.; Pretsch, D.; Genov, M.; Hollaus, R.; Zinssmeister, D.; Debbab, A.; et al. Antimicrobial and anti-inflammatory activities of endophytic fungi Talaromyces wortmannii extracts against acne-inducing bacteria. PLoS ONE 2014, 9, e97929. [Google Scholar] [CrossRef]
- Dessinioti, C.; Katsambas, A. Propionibacterium acnes and antimicrobial resistance in acne. Clin. Dermatol. 2017, 35, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Han, R.; Blencke, H.M.; Cheng, H.; Li, C. The antimicrobial effect of CEN1HC-Br against Propionibacterium acnes and its therapeutic and anti-inflammatory effects on acne vulgaris. Peptides 2018, 99, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Wang, J.Q.; Song, H.Y.; Zhang, Q.; Liu, G.F. Chemical analysis and in vitro antimicrobial effects and mechanism of action of Trachyspermum copticum essential oil against Escherichia coli. Asian Pac. J. Trop. Med. 2017, 10, 663–669. [Google Scholar] [CrossRef]
- O’Toole, G.A. Microtiter dish biofilm formation assay. J. Vis. Exp. JoVE 2011, 30, e2437. [Google Scholar] [CrossRef]
- Purwasena, I.A.; Aditiawati, P.; Siwi, I.K.; Fauziyyah, N.A. The effect of cymbopogon citratus essential oil on community dynamics of biofilm-forming bacteria isolated from brine water of oil reservoir in South Sumatra. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2019; Volume 299, p. 012057. [Google Scholar]
- Avila, J.G.; de Liverant, J.G.; Martinez, A.; Martinez, G.; Munoz, J.L.; Arciniegas, A.; de Vivar, A.R. Mode of action of Buddleja cordata verbascoside against Stphylococcus aureus. J. Ethnopharmacol. 1999, 66, 75–78. [Google Scholar] [CrossRef]
- Clemente, I.; Aznar, M.; Silva, F.; Nerín, C. Antimicrobial properties and mode of action of mustard and cinnamon essential oils and their combination against foodborne bacteria. Innov. Food Sci. Emerg. Technol. 2016, 36, 26–33. [Google Scholar] [CrossRef]
- Weigel, D.; Glazebrook, J. Transmission electron microscopy (TEM) freeze substitution of plant tissues. Cold Spring Harb. Protoc. 2010, 2010, pdb.prot4959. [Google Scholar] [CrossRef]
- Lambert, R.; Skandamis, P.N.; Coote, P.J.; Nychas, G.J. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J. Appl. Microbiol. 2001, 91, 453–462. [Google Scholar] [CrossRef]
- Wilschefski, S.C.; Baxter, M.R. Inductively coupled plasma mass spectrometry: Introduction to analytical aspects. Clin. Biochem. Rev. 2019, 40, 115–133. [Google Scholar] [CrossRef] [PubMed]
- Carson, C.F.; Mee, B.J.; Riley, T.V. Mechanism of action of melaleuca alternifolia (tea tree) oil on Staphylococcus aureus determined by time-kill, lysis, leakage, and salt tolerance assays and electron microscopy. Antimicrob. Agents Chemother. 2002, 46, 1914–1920. [Google Scholar] [CrossRef] [PubMed]
- Ostertag, F.; Weiss, J.; McClements, D.J. Low-energy formation of edible nanoemulsions: Factors influencing droplet size produced by emulsion phase inversion. J. Colloid Interface Sci. 2012, 388, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, P.M.; Felício, M.R.; Santos, N.C.; Gonçalves, S.; Domingues, M.M. Application of light scattering techniques to nanoparticle characterization and development. Front. Chem. 2018, 6, 237. [Google Scholar] [CrossRef]
- Robledo, N.; Vera, P.; López, L.; Yazdani-Pedram, M.; Tapia, C.; Abugoch, L. Thymol nanoemulsions incorporated in quinoa protein/chitosan edible films; antifungal effect in cherry tomatoes. Food Chem. 2018, 246, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Taleb, M.H.; Abdeltawab, N.F.; Shamma, R.N.; Abdelgayed, S.S.; Mohamed, S.S.; Farag, M.A.; Ramadan, M.A. Origanum vulgare L. essential oil as a potential anti-acne topical nanoemulsion—In vitro and in vivo study. Molecules 2018, 23, 2164. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Lv, X.-D.; Wang, G.-E.; Li, Y.-F.; Kurihara, H.; He, R.-R. Anti-inflammatory effects of anthocyanins-rich extract from bilberry (Vaccinium myrtillus L.) on croton oil-induced ear edema and Propionibacterium acnes plus LPS-induced liver damage in mice. Int. J. Food Sci. Nutr. 2014, 65, 594–601. [Google Scholar] [CrossRef]
- Budhiraja, A.; Dhingra, G. Development and characterization of a novel antiacne niosomal gel of rosmarinic acid. Drug Deliv. 2015, 22, 723–730. [Google Scholar] [CrossRef]
- Chafin, D.; Theiss, A.; Roberts, E.; Borlee, G.; Otter, M.; Baird, G.S. Rapid two-temperature formalin fixation. PLoS ONE 2013, 8, e54138. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Byrd, A.L.; Deming, C.; Conlan, S.; NISC Comparative Sequencing Program; Kong, H.H.; Segre, J.A. Biogeography and individuality shape function in the human skin metagenome. Nature 2014, 514, 59–64. [Google Scholar] [CrossRef]
- O’Neill, A.M.; Gallo, R.L. Host-microbiome interactions and recent progress into understanding the biology of acne vulgaris. Microbiome 2018, 6, 177. [Google Scholar] [CrossRef]
- Gollnick, H.; Cunliffe, W.; Berson, D.; Dreno, B.; Finlay, A.; Leyden, J.J.; Shalita, A.R.; Thiboutot, D. Management of acne: A report from a global alliance to improve outcomes in acne. J. Am. Acad. Dermatol. 2003, 49, S1–S37. [Google Scholar] [CrossRef]
- Thiboutot, D.; Gollnick, H.; Bettoli, V.; Dréno, B.; Kang, S.; Leyden, J.J.; Shalita, A.R.; Lozada, V.T.; Berson, D.; Finlay, A. New insights into the management of acne: An update from the global alliance to improve outcomes in acne group. J. Am. Acad. Dermatol. 2009, 60, S1–S50. [Google Scholar] [CrossRef]
- Patel, M.; Bowe, W.P.; Heughebaert, C.; Shalita, A.R. The development of antimicrobial resistance due to the antibiotic treatment of acne vulgaris: A review. J. Drugs Dermatol. 2010, 9, 655–664. [Google Scholar]
- Tongnuanchan, P.; Benjakul, S. Essential oils: Extraction, bioactivities, and their uses for food preservation. J. Food Sci. 2014, 79, R1231–R1249. [Google Scholar] [CrossRef]
- Warnke, P.H.; Becker, S.T.; Podschun, R.; Sivananthan, S.; Springer, N.; Russo, P.A.J.; Wiltfang, J.; Fickenscher, H.; Sherry, E. The battle against multi-resistant strains: Renaissance of antimicrobial essential oils as a promising force to fight hospital-acquired infections. J. Cranio-Maxillofac. Surg. 2009, 37, 392–397. [Google Scholar] [CrossRef]
- Thombare, S.; Mukundan, U. Control of Propionibacterium acnes using essential oil. Advis. Board Prof. John Cairns Asian J. Exp. Sci. 2012, 26, 1. [Google Scholar]
- Gulluce, M.; Karadayi, M.; Guvenalp, Z.; Ozbek, H.; Arasoglu, T.; Baris, O. Isolation of some active compounds from Origanum vulgare L. ssp. vulgare and determination of their genotoxic potentials. Food Chem. 2012, 130, 248–253. [Google Scholar] [CrossRef]
- Sinha, P.; Srivastava, S.; Mishra, N.; Yadav, N.P. New perspectives on antiacne plant drugs: Contribution to modern therapeutics. BioMed Res. Int. 2014, 2014, 301304. [Google Scholar] [CrossRef] [PubMed]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Bouyahya, A.; Abrini, J.; Dakka, N.; Bakri, Y. Essential oils of Origanum compactum increase membrane permeability, disturb cell membrane integrity, and suppress quorum-sensing phenotype in bacteria. J. Pharm. Anal. 2019, 9, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Nakatsuji, T.; Shi, Y.; Zhu, W.; Huang, C.P.; Chen, Y.R.; Lee, D.Y.; Smith, J.W.; Zouboulis, C.C.; Gallo, R.L.; Huang, C.M. Bioengineering a humanized acne microenvironment model: Proteomics analysis of host responses to Propionibacterium Acnes infection in vivo. Proteomics 2008, 8, 3406–3415. [Google Scholar] [CrossRef] [PubMed]
- Shamma, R.N.; Ad-din, I.S.; Abdeltawab, N.F. Dapsone-gel as a novel platform for acne treatment: In vitro evaluation and in vivo performance and histopathological studies in acne infected mice. J. Drug Deliv. Sci. Technol. 2019, 54, 101238. [Google Scholar] [CrossRef]







| Common Name | Latin Name | Family Name | Part of Plant Used |
|---|---|---|---|
| Tea tree | Melaleuca alternifolia | Myrtaceae | Buds |
| Clove | Syzygium aromaticum | Myrtaceae | Buds |
| Thyme | Thymus vulgaris | Lamiaceae | Leaves |
| Mentha | Mentha spicate | Lamiaceae | Leaves |
| Basil | Ocimum basilicum | Lamiaceae | Leaves and flowering tops |
| Minimum Inhibitory Concentrations (MIC) | ||
|---|---|---|
| EO | C. acnes (g/mL) | S. epidermidis (g/mL) |
| Tea tree | 0.053 | 0.053 |
| Clove | 0.137 | 0.137 |
| Thyme | 0.026 | 0.053 |
| Mentha | 0.125 | 0.107 |
| Basil | 0.220 | 0.110 |
| Minimum Bactericidal Concentration (MBC) | ||
|---|---|---|
| EO | C. acnes (g/mL) | S. epidermidis (g/mL) |
| Tea tree | 0.053 | 0.053 |
| Clove | 0.137 | 0.137 |
| Thyme | 0.026 | 0.053 |
| Mentha | 0.215 | 0.107 |
| Basil | 0.220 | 0.110 |
| Compound | Thyme EO Content (%) | Retention Time (Minutes) | Retention Index |
|---|---|---|---|
| β-Myrcene | 0.38 | 9.026 | 872 |
| α-Terpinolene | 0.33 | 9.789 | 918 |
| o-Cymene | 4.17 | 10.096 | 929 |
| Limonene | 0.18 | 10.195 | 817 |
| γ-Terpinene | 1.21 | 11.122 | 941 |
| α-Ocinene | 0.60 | 12.405 | 897 |
| Isoborneol | 0.35 | 14.465 | 856 |
| (+)-4-Carene | 0.50 | 14.812 | 820 |
| Carvacrol | 2.18 | 18.708 | 926 |
| Thymol | 75.46 | 18.299 | 939 |
| Caryophyllene | 13.40 | 21.797 | 951 |
| Humulene | 0.28 | 22.689 | 902 |
| Total identified compounds | 99.04% | ||
| Phenolic compounds | 77.64% | ||
| Terpenoid compounds | 21.4% | ||
| EO | Minimum Biofilm Inhibitory Concentration (g/mL) | Minimum Biofilm Eradication Concentration (g/mL) |
|---|---|---|
| Thyme | 0.026 | 0.053 |
| Tea tree | 0.107 | No effect |
| Clove | 0.274 | 0.274 |
| Sign of Inflammation | Blank Formula-Treated Ear Tissue | 1% Clindamycin-Treated Ear Tissue | Thyme EO-Treated Ear Tissue |
|---|---|---|---|
| Edema | ++ | + 1 | - 2 |
| Vascular congestion | + | - | - |
| Inflammatory cells infiltration | ++ | Few | Markedly reduced |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelhamed, F.M.; Abdeltawab, N.F.; ElRakaiby, M.T.; Shamma, R.N.; Moneib, N.A. Antibacterial and Anti-Inflammatory Activities of Thymus vulgaris Essential Oil Nanoemulsion on Acne Vulgaris. Microorganisms 2022, 10, 1874. https://doi.org/10.3390/microorganisms10091874
Abdelhamed FM, Abdeltawab NF, ElRakaiby MT, Shamma RN, Moneib NA. Antibacterial and Anti-Inflammatory Activities of Thymus vulgaris Essential Oil Nanoemulsion on Acne Vulgaris. Microorganisms. 2022; 10(9):1874. https://doi.org/10.3390/microorganisms10091874
Chicago/Turabian StyleAbdelhamed, Farah M., Nourtan F. Abdeltawab, Marwa T. ElRakaiby, Rehab N. Shamma, and Nayera A. Moneib. 2022. "Antibacterial and Anti-Inflammatory Activities of Thymus vulgaris Essential Oil Nanoemulsion on Acne Vulgaris" Microorganisms 10, no. 9: 1874. https://doi.org/10.3390/microorganisms10091874
APA StyleAbdelhamed, F. M., Abdeltawab, N. F., ElRakaiby, M. T., Shamma, R. N., & Moneib, N. A. (2022). Antibacterial and Anti-Inflammatory Activities of Thymus vulgaris Essential Oil Nanoemulsion on Acne Vulgaris. Microorganisms, 10(9), 1874. https://doi.org/10.3390/microorganisms10091874










