Molecular Evolution of Far-Red Light-Acclimated Photosystem II
Abstract
1. Introduction
2. Materials and Methods
2.1. Phylogenetic Tree Construction
2.2. Homology Modeling
3. Results
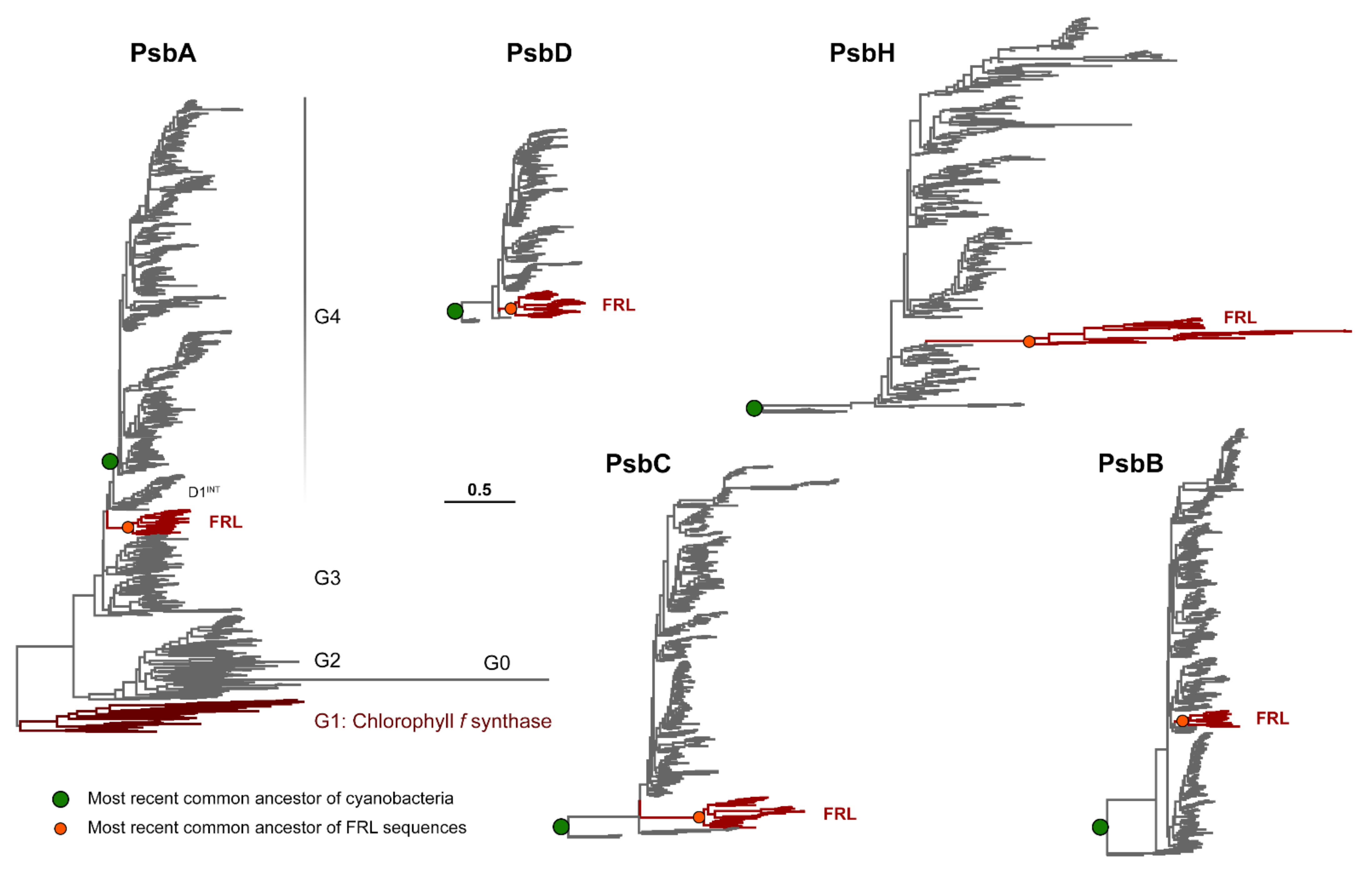
3.1. Phylogenetic Analysis
3.2. Sequence Annotations and Structural View
3.3. Conserved Features of FRL-PsbA
3.4. Conserved Features of FRL-PsbD
3.5. Conserved Features of FRL-PsbC
3.6. Conserved Features of FRL-PsbB
3.7. Conserved Features of FRL-PsbH
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Blankenship, R.E. Molecular Mechanisms of Photosynthesis, 3rd ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2021; ISBN 978-1-119-80001-9. [Google Scholar]
- Björn, L.O.; Papageorgiou, G.C.; Blankenship, R.E. Govindjee A Viewpoint: Why Chlorophyll a? Photosynth. Res. 2009, 99, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Gan, F.; Zhang, S.; Rockwell, N.C.; Martin, S.S.; Lagarias, J.C.; Bryant, D.A. Extensive Remodeling of a Cyanobacterial Photosynthetic Apparatus in Far-Red Light. Science 2014, 345, 1312–1317. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.-Y.; Niedzwiedzki, D.M.; MacGregor-Chatwin, C.; Gerstenecker, G.; Hunter, C.N.; Blankenship, R.E.; Bryant, D.A. Extensive Remodeling of the Photosynthetic Apparatus Alters Energy Transfer among Photosynthetic Complexes When Cyanobacteria Acclimate to Far-Red Light. Biochim. Biophys. Acta Bioenerg. 2020, 1861, 148064. [Google Scholar] [CrossRef] [PubMed]
- Nürnberg, D.J.; Morton, J.; Santabarbara, S.; Telfer, A.; Joliot, P.; Antonaru, L.A.; Ruban, A.V.; Cardona, T.; Krausz, E.; Boussac, A.; et al. Photochemistry beyond the Red Limit in Chlorophyll f–Containing Photosystems. Science 2018, 360, 1210–1213. [Google Scholar] [CrossRef]
- Bryant, D.A.; Hunter, C.N.; Warren, M.J. Biosynthesis of the Modified Tetrapyrroles-the Pigments of Life. J. Biol. Chem. 2020, 295, 6888–6925. [Google Scholar] [CrossRef]
- Chen, M.; Blankenship, R.E. Expanding the Solar Spectrum Used by Photosynthesis. Trends Plant Sci. 2011, 16, 427–431. [Google Scholar] [CrossRef]
- Chen, M. Chlorophyll Modifications and Their Spectral Extension in Oxygenic Photosynthesis. Annu. Rev. Biochem. 2014, 83, 317–340. [Google Scholar] [CrossRef]
- Gan, F.; Bryant, D.A. Adaptive and Acclimative Responses of Cyanobacteria to Far-Red Light. Environ. Microbiol. 2015, 17, 3450–3465. [Google Scholar] [CrossRef]
- Viola, S.; Roseby, W.; Santabarabara, S.; Nürnberg, D.; Assunção, R.; Dau, H.; Sellés, J.; Boussac, A.; Fantuzzi, A.; Rutherford, A.W. Impact of Energy Limitations on Function and Resilience in Long-Wavelength Photosystem II. bioRxiv 2022. [Google Scholar] [CrossRef]
- Wolf, B.M.; Blankenship, R.E. Far-Red Light Acclimation in Diverse Oxygenic Photosynthetic Organisms. Photosynth. Res. 2019, 142, 349–359. [Google Scholar] [CrossRef]
- Hu, C.; Nawrocki, W.J.; Croce, R. Long-Term Adaptation of Arabidopsis thaliana to Far-Red Light. Plant. Cell Environ. 2021. [Google Scholar] [CrossRef] [PubMed]
- Gan, F.; Shen, G.; Bryant, D.A. Occurrence of Far-Red Light Photoacclimation (FaRLiP) in Diverse Cyanobacteria. Life 2014, 5, 4–24. [Google Scholar] [CrossRef] [PubMed]
- Antonaru, L.A.; Cardona, T.; Larkum, A.W.D.; Nürnberg, D.J. Global Distribution of a Chlorophyll f Cyanobacterial Marker. ISME J. 2020, 14, 2275–2287. [Google Scholar] [CrossRef] [PubMed]
- Averina, S.; Velichko, N.; Senatskaya, E.; Pinevich, A. Far-Red Light Photoadaptations in Aquatic Cyanobacteria. Hydrobiologia 2018, 813, 1–17. [Google Scholar] [CrossRef]
- Ohkubo, S.; Miyashita, H. A Niche for Cyanobacteria Producing Chlorophyll f within a Microbial Mat. ISME J. 2017, 11, 2368–2378. [Google Scholar] [CrossRef]
- Behrendt, L.; Brejnrod, A.; Schliep, M.; Sørensen, S.J.; Larkum, A.W.D.; Kühl, M. Chlorophyll f-Driven Photosynthesis in a Cavernous Cyanobacterium. ISME J. 2015, 9, 2108–2111. [Google Scholar] [CrossRef]
- Behrendt, L.; Trampe, E.L.; Nord, N.B.; Nguyen, J.; Kühl, M.; Lonco, D.; Nyarko, A.; Dhinojwala, A.; Hershey, O.S.; Barton, H. Life in the Dark: Far-Red Absorbing Cyanobacteria Extend Photic Zones Deep into Terrestrial Caves. Environ. Microbiol. 2019. [Google Scholar] [CrossRef]
- Kühl, M.; Trampe, E.; Mosshammer, M.; Johnson, M.; Larkum, A.W.D.; Frigaard, N.-U.; Koren, K. Substantial Near-Infrared Radiation-Driven Photosynthesis of Chlorophyll f-Containing Cyanobacteria in a Natural Habitat. Elife 2020, 9, e50871. [Google Scholar] [CrossRef]
- Rippka, R.; Deruelles, J.; Waterbury, J.B.; Herdman, M.; Stanier, R.Y. Generic Assignments, Strain Histories and Properties of Pure Cultures of Cyanobacteria. Microbiology 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Gisriel, C.J.; Shen, G.; Ho, M.-Y.; Kurashov, V.; Flesher, D.A.; Wang, J.; Armstrong, W.H.; Golbeck, J.H.; Gunner, M.R.; Vinyard, D.J.; et al. Structure of a Monomeric Photosystem II Core Complex from a Cyanobacterium Acclimated to Far-Red Light Reveals the Functions of Chlorophylls d and f. J. Biol. Chem. 2022, 298, 101424. [Google Scholar] [CrossRef]
- Soulier, N.; Laremore, T.N.; Bryant, D.A. Characterization of Cyanobacterial Allophycocyanins Absorbing Far-Red Light. Photosynth. Res. 2020, 145, 189–207. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.-T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A Fast and Effective Stochastic Algorithm for Estimating Maximum-Likelihood Phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Duvaud, S.; Gabella, C.; Lisacek, F.; Stockinger, H.; Ioannidis, V.; Durinx, C. Expasy, the Swiss Bioinformatics Resource Portal, as Designed by Its Users. Nucleic Acids Res. 2021, 49, W216–W227. [Google Scholar] [CrossRef]
- Sievers, F.; Wilm, A.; Dineen, D.; Gibson, T.J.; Karplus, K.; Li, W.; Lopez, R.; McWilliam, H.; Remmert, M.; Söding, J.; et al. Fast, Scalable Generation of High Quality Protein Multiple Sequence Alignments Using Clustal Omega. Mol. Syst. Biol. 2011, 7, 1–6. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast Model Selection for Accurate Phylogenetic Estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; von Haeseler, A.; Lanfear, R. IQ-TREE 2: New Models and Efficient Methods for Phylogenetic Inference in the Genomic Era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef]
- Minh, B.Q.; Nguyen, M.A.T.; von Haeseler, A. Ultrafast Approximation for Phylogenetic Bootstrap. Mol. Biol. Evol. 2013, 30, 1188–1195. [Google Scholar] [CrossRef]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the Ultrafast Bootstrap Approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef]
- Anisimova, M.; Gascuel, O. Approximate Likelihood-Ratio Test for Branches: A Fast, Accurate, and Powerful Alternative. Syst. Biol. 2006, 55, 539–552. [Google Scholar] [CrossRef]
- Huson, D.H.; Scornavacca, C. Dendroscope 3: An Interactive Tool for Rooted Phylogenetic Trees and Networks. Syst. Biol. 2012, 61, 1061–1067. [Google Scholar] [CrossRef]
- The PyMOL Molecular Graphics System, Version 2.0 Schrödinger, LLC. Available online: https://pymol.org/2/ (accessed on 26 May 2022).
- Guex, N.; Peitsch, M.C.; Schwede, T. Automated Comparative Protein Structure Modeling with SWISS-MODEL and Swiss-PdbViewer: A Historical Perspective. Electrophoresis 2009, 30, S162–S173. [Google Scholar] [CrossRef]
- Gisriel, C.J.; Wang, J.; Liu, J.; Flesher, D.A.; Reiss, K.M.; Huang, H.-L.; Yang, K.R.; Armstrong, W.H.; Gunner, M.R.; Batista, V.S.; et al. High-Resolution Cryo-EM Structure of Photosystem II from the Mesophilic Cyanobacterium, Synechocystis sp. PCC 6803. Proc. Natl. Acad. Sci. USA 2022, 119, e2116765118. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, K.J.; Duncan, E.J.; Eaton-Rye, J.J.; Summerfield, T.C. The Diversity and Distribution of D1 Proteins in Cyanobacteria. Photosynth. Res. 2020, 145, 111–128. [Google Scholar] [CrossRef] [PubMed]
- Cardona, T.; Murray, J.W.; Rutherford, A.W. Origin and Evolution of Water Oxidation before the Last Common Ancestor of the Cyanobacteria. Mol. Biol. Evol. 2015, 32, 1310–1328. [Google Scholar] [CrossRef] [PubMed]
- Cardona, T.; Sánchez-Baracaldo, P.; Rutherford, A.W.; Larkum, A.W. Early Archean Origin of Photosystem II. Geobiology 2019, 17, 127–150. [Google Scholar] [CrossRef] [PubMed]
- Oliver, T.; Sánchez-Baracaldo, P.; Larkum, A.W.; Rutherford, A.W.; Cardona, T. Time-Resolved Comparative Molecular Evolution of Oxygenic Photosynthesis. Biochim. Biophys. Acta Bioenerg. 2021, 1862, 148400. [Google Scholar] [CrossRef]
- Ho, M.Y.; Shen, G.; Canniffe, D.P.; Zhao, C.; Bryant, D.A. Light-Dependent Chlorophyll f Synthase Is a Highly Divergent Paralog of PsbA of Photosystem II. Science 2016, 353, aaf9178. [Google Scholar] [CrossRef]
- Trinugroho, J.P.; Bečková, M.; Shao, S.; Yu, J.; Zhao, Z.; Murray, J.W.; Sobotka, R.; Komenda, J.; Nixon, P.J. Chlorophyll f Synthesis by a Super-Rogue Photosystem II Complex. Nat. Plants 2020, 6, 238–244. [Google Scholar] [CrossRef]
- Shen, G.; Canniffe, D.P.; Ho, M.-Y.; Kurashov, V.; van der Est, A.; Golbeck, J.H.; Bryant, D.A. Characterization of Chlorophyll f Synthase Heterologously Produced in Synechococcus Sp. PCC 7002. Photosynth. Res. 2019, 140, 77–92. [Google Scholar] [CrossRef]
- Saw, J.H.; Cardona, T.; Montejano, G. Complete Genome Sequencing of a Novel Gloeobacter Species from a Waterfall Cave in Mexico. Genome Biol. Evol. 2021, 13, evab264. [Google Scholar] [CrossRef]
- Shih, P.M.; Wu, D.; Latifi, A.; Axen, S.D.; Fewer, D.P.; Talla ECalteau, A.; Cai, F.; de Marsac, N.T.; Rippka, R. Improving the Coverage of the Cyanobacterial Phylum Using Diversity-Driven Genome Sequencing. Proc. Natl. Acad. Sci. USA 2013, 110, 1053–1058. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.Y.; Gan, F.; Shen, G.; Bryant, D.A. Far-Red Light Photoacclimation (FaRLiP) in Synechococcus sp. PCC 7335. II. Characterization of Phycobiliproteins Produced during Acclimation to Far-Red Light. Photosynth. Res. 2017, 131, 187–202. [Google Scholar] [CrossRef] [PubMed]
- Llansola-Portoles, M.J.; Li, F.; Xu, P.; Streckaite, S.; Ilioaia, C.; Yang, C.; Gall, A.; Pascal, A.A.; Croce, R.; Robert, B. Tuning Antenna Function through Hydrogen Bonds to Chlorophyll a. Biochim. Biophys. Acta Bioenerg. 2020, 1861, 148078. [Google Scholar] [CrossRef]
- Umena, Y.; Kawakami, K.; Shen, J.-R.; Kamiya, N. Crystal Structure of Oxygen-Evolving Photosystem II at a Resolution of 1.9 Å. Nature 2011, 473, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Gisriel, C.J.; Huang, H.-L.; Reiss, K.M.; Flesher, D.A.; Batista, V.S.; Bryant, D.A.; Brudvig, G.W.; Wang, J. Quantitative Assessment of Chlorophyll Types in Cryo-EM Maps of Photosystem I Acclimated to Far-Red Light. BBA Adv. 2021, 1, 100019. [Google Scholar] [CrossRef]
- Gisriel, C.J.; Wang, J.; Brudvig, G.W.; Bryant, D.A. Opportunities and Challenges for Assigning Cofactors in Cryo-EM Density Maps of Chlorophyll-Containing Proteins. Commun. Biol. 2020, 3, 408. [Google Scholar] [CrossRef] [PubMed]
- Gisriel, C.J.; Shen, G.; Kurashov, V.; Ho, M.-Y.; Zhang, S.; Williams, D.; Golbeck, J.H.; Fromme, P.; Bryant, D.A. The Structure of Photosystem I Acclimated to Far-Red Light Illuminates an Ecologically Important Acclimation Process in Photosynthesis. Sci. Adv. 2020, 6, eaay6415. [Google Scholar] [CrossRef]
- Gisriel, C.J.; Flesher, D.A.; Shen, G.; Wang, J.; Ho, M.-Y.; Brudvig, G.W.; Bryant, D.A. Structure of a Photosystem I-Ferredoxin Complex from a Marine Cyanobacterium Provides Insights into Far-Red Light Photoacclimation. J. Biol. Chem. 2022, 298, 101408. [Google Scholar] [CrossRef]
- Komenda, J.; Tichý, M.; Eichacker, L.A. The PsbH Protein Is Associated with the Inner Antenna CP47 and Facilitates D1 Processing and Incorporation into PSII in the Cyanobacterium Synechocystis PCC 6803. Plant Cell Physiol. 2005, 46, 1477–1483. [Google Scholar] [CrossRef]
- Komenda, J.; Lupínková, L.; Kopecký, J. Absence of the PsbH Gene Product Destabilizes Photosystem II Complex and Bicarbonate Binding on Its Acceptor Side in Synechocystis PCC 6803. Eur. J. Biochem. 2002, 269, 610–619. [Google Scholar] [CrossRef]
- D’Haene, S.E.; Sobotka, R.; Bučinská, L.; Dekker, J.P.; Komenda, J. Interaction of the PsbH Subunit with a Chlorophyll Bound to Histidine 114 of CP47 Is Responsible for the Red 77K Fluorescence of Photosystem II. Biochim. Biophys. Acta Bioenerg. 2015, 1847, 1327–1334. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zabret, J.; Bohn, S.; Schuller, S.K.; Arnolds, O.; Möller, M.; Meier-Credo, J.; Liauw, P.; Chan, A.; Tajkhorshid, E.; Langer, J.D.; et al. Structural Insights into Photosystem II Assembly. Nat. Plants 2021, 7, 524–538. [Google Scholar] [CrossRef] [PubMed]
- Graça, A.T.; Hall, M.; Persson, K.; Schröder, W.P. High-Resolution Model of Arabidopsis Photosystem II Reveals the Structural Consequences of Digitonin-Extraction. Sci. Rep. 2021, 11, 15534. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Hamaguchi, T.; Nakajima, Y.; Kato, K.; Kawakami, K.; Akita, F.; Yonekura, K.; Shen, J.-R. Cryo-EM Structure of Monomeric Photosystem II at 2.78 Å Resolution Reveals Factors Important for the Formation of Dimer. Biochim. Biophys. Acta Bioenerg. 2021, 1862, 148471. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Xiao, Y.; Pi, X.; Zhao, L.; Zhu, Q.; Wang, W.; Kuang, T.; Han, G.; Sui, S.-F.; Shen, J.-R. Structural Insights into a Dimeric Psb27-Photosystem II Complex from a Cyanobacterium Thermosynechococcus vulcanus. Proc. Natl. Acad. Sci. USA 2021, 118, e2018053118. [Google Scholar] [CrossRef]
- Bryant, D.A.; Shen, G.; Turner, G.M.; Soulier, N.; Laremore, T.N.; Ho, M.-Y. Far-Red Light Allophycocyanin Subunits Play a Role in Chlorophyll d Accumulation in Far-Red Light. Photosynth. Res. 2020, 143, 81–95. [Google Scholar] [CrossRef]
- Carpenter, S.D.; Charite, J.; Eggers, B.; Vermaas, W.F.J. The PsbC Start Codon in Synechocystis sp. PCC 6803. FEBS Lett. 1990, 260, 135–137. [Google Scholar] [CrossRef]
- Hessa, T.; Meindl-Beinker, N.M.; Bernsel, A.; Kim, H.; Sato, Y.; Lerch-Bader, M.; Nilsson, I.; White, S.H.; von Heijne, G. Molecular Code for Transmembrane-Helix Recognition by the Sec61 Translocon. Nature 2007, 450, 1026–1030. [Google Scholar] [CrossRef]
- Emsley, P.; Lohkamp, B.; Scott, W.G.; Cowtan, K. Features and Development of Coot. Acta Crystallogr. Sect. D Biol. Crystallogr. 2010, 66, 486–501. [Google Scholar] [CrossRef]







| FRL Sequence from Synechococcus 7335 | FRL-Specific Positions Conserved in the Ancestral Sequence (%) | FRL-Specific Positions Conserved Only in the Extant Sequences (%) |
|---|---|---|
| FRL-PsbA | 3.3 | 3.3 |
| FRL-PsbB | 2.4 | 6.3 |
| FRL-PsbC | 3.9 | 3.9 |
| FRL-PsbD | 1.7 | 1.7 |
| FRL-PsbH | 6.1 | 7.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gisriel, C.J.; Cardona, T.; Bryant, D.A.; Brudvig, G.W. Molecular Evolution of Far-Red Light-Acclimated Photosystem II. Microorganisms 2022, 10, 1270. https://doi.org/10.3390/microorganisms10071270
Gisriel CJ, Cardona T, Bryant DA, Brudvig GW. Molecular Evolution of Far-Red Light-Acclimated Photosystem II. Microorganisms. 2022; 10(7):1270. https://doi.org/10.3390/microorganisms10071270
Chicago/Turabian StyleGisriel, Christopher J., Tanai Cardona, Donald A. Bryant, and Gary W. Brudvig. 2022. "Molecular Evolution of Far-Red Light-Acclimated Photosystem II" Microorganisms 10, no. 7: 1270. https://doi.org/10.3390/microorganisms10071270
APA StyleGisriel, C. J., Cardona, T., Bryant, D. A., & Brudvig, G. W. (2022). Molecular Evolution of Far-Red Light-Acclimated Photosystem II. Microorganisms, 10(7), 1270. https://doi.org/10.3390/microorganisms10071270







