Traditional Fermented Foods and Beverages from around the World and Their Health Benefits
Abstract
:1. Introduction
2. Microorganisms Found in Traditional Fermented Foods and Beverages
2.1. Africa
2.2. America
2.3. Asia
2.4. Europe
3. Interventions Involving the Use of Traditional Fermented Foods and Beverages and Their Association with Benefits on Human Health
3.1. Related Studies in America
3.2. Related Studies in Asia
3.3. Related Studies in Africa
3.4. Related Studies in Europe
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McGovern, P.E.; Zhang, J.; Tang, J.; Zhang, Z.; Hall, G.R.; Moreau, R.A.; Nunez, A.; Butrym, E.D.; Richards, M.P.; Wang, C.-S.; et al. Fermented Beverages of Pre- and Proto-Historic China. Proc. Natl. Acad. Sci. USA 2004, 101, 17593–17598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dimidi, E.; Cox, S.R.; Rossi, M.; Whelan, K. Fermented Foods: Definitions and Characteristics, Impact on the Gut Microbiota and Effects on Gastrointestinal Health and Disease. Nutrients 2019, 11, 1806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamang, J.P.; Watanabe, K.; Holzapfel, W.H. Review: Diversity of Microorganisms in Global Fermented Foods and Beverages. Front. Microbiol. 2016, 7, 377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamang, J.P.; Shin, D.-H.; Jung, S.-J.; Chae, S.-W. Functional Properties of Microorganisms in Fermented Foods. Front. Microbiol. 2016, 7, 578. [Google Scholar] [CrossRef] [Green Version]
- Stanton, C.; Fitzgerald, G.F.; Ross, R.P. Abstract: Overview of Technology Developments in Probiotic Field. Microb. Ecol. Health Dis. 2012, 23, 18561. [Google Scholar]
- Chilton, S.N.; Burton, J.P.; Reid, G. Inclusion of Fermented Foods in Food Guides around the World. Nutrients 2015, 7, 390–404. [Google Scholar] [CrossRef] [Green Version]
- Coton, M.; Pawtowski, A.; Taminiau, B.; Burgaud, G.; Deniel, F.; Coulloumme-Labarthe, L.; Fall, A.; Daube, G.; Coton, E. Unraveling Microbial Ecology of Industrial-Scale Kombucha Fermentations by Metabarcoding and Culture-Based Methods. FEMS Microbiol. Ecol. 2017, 93, fix048. [Google Scholar] [CrossRef]
- Rezac, S.; Kok, C.R.; Heermann, M.; Hutkins, R. Fermented Foods as a Dietary Source of Live Organisms. Front. Microbiol. 2018, 9, 1785. [Google Scholar] [CrossRef]
- Xiang, H.; Sun-Waterhouse, D.; Waterhouse, G.; Cui, C.; Ruan, Z. Fermentation-Enabled Wellness Foods: A Fresh Perspective. Food Sci. Hum. Wellness 2019, 8, 203–243. [Google Scholar] [CrossRef]
- Konstantinidi, M.; Koutelidakis, A.E. Functional Foods and Bioactive Compounds: A Review of Its Possible Role on Weight Management and Obesity’s Metabolic Consequences. Medicines 2019, 6, 94. [Google Scholar] [CrossRef] [Green Version]
- Chen, M.; Sun, Q.; Giovannucci, E.; Mozaffarian, D.; Manson, J.E.; Willett, W.C.; Hu, F.B. Dairy Consumption and Risk of Type 2 Diabetes: 3 Cohorts of Us Adults and an Updated Meta-Analysis. BMC Med. 2014, 12, 215. [Google Scholar] [CrossRef] [Green Version]
- Dongmo, S.N.; Procopio, S.; Sacher, B.; Becker, T. Flavor of Lactic Acid Fermented Malt Based Beverages: Current Status and Perspectives. Trends Food Sci. Technol. 2016, 54, 37–51. [Google Scholar] [CrossRef]
- Eussen, S.J.P.M.; van Dongen, M.C.J.M.; Wijckmans, N.; den Biggelaar, L.; Oude Elferink, S.J.W.H.; Singh-Povel, C.M.; Schram, M.T.; Sep, S.J.S.; van der Kallen, C.J.; Koster, A.; et al. Consumption of Dairy Foods in Relation to Impaired Glucose Metabolism and Type 2 Diabetes Mellitus: The Maastricht Study. Br. J. Nutr. 2016, 115, 1453–1461. [Google Scholar] [CrossRef] [Green Version]
- Tapsell, L.C. Fermented Dairy Food and CVD Risk. Br. J. Nutr. 2015, 113, S131–S135. [Google Scholar] [CrossRef] [Green Version]
- Tillisch, K.; Labus, J.; Kilpatrick, L.; Jiang, Z.; Stains, J.; Ebrat, B.; Guyonnet, D.; Legrain-Raspaud, S.; Trotin, B.; Naliboff, B.; et al. Consumption of Fermented Milk Product with Probiotic Modulates Brain Activity. Gastroenterology 2013, 144, 1394–1401.e4. [Google Scholar] [CrossRef] [Green Version]
- Zhang, K.; Dai, H.; Liang, W.; Zhang, L.; Deng, Z. Fermented Dairy Foods Intake and Risk of Cancer. Int. J. Cancer 2019, 144, 2099–2108. [Google Scholar] [CrossRef]
- Lilly, D.M.; Stillwell, R.H. Probiotics: Growth-Promoting Factors Produced by Microorganisms. Science 1965, 147, 747–748. [Google Scholar] [CrossRef]
- FAO/WHO. Evaluation of Health and Nutritional Properties of Powder Milk and Live Lactic Acid Bacteria. 2001. Available online: https://www.scirp.org/%28S%28351jmbntvnsjt1aadkposzje%29%29/reference/referencespapers.aspx?referenceid=1456307 (accessed on 24 April 2022).
- Barengolts, E. Gut Microbiota, Prebiotics, Probiotics, and Synbiotics in Management of Obesity and Prediabetes: Review of Randomized Controlled Trials. Endocr. Pract. 2016, 22, 1224–1234. [Google Scholar] [CrossRef] [Green Version]
- Fijan, S. Microorganisms with Claimed Probiotic Properties: An Overview of Recent Literature. Int. J. Environ. Res. Public Health 2014, 11, 4745–4767. [Google Scholar] [CrossRef]
- Anlier, N.; Gökcen, B.B.; Sezgin, A.C. Health Benefits of Fermented Foods. Crit. Rev. Food Sci. Nutr. 2019, 59, 506–527. [Google Scholar] [CrossRef]
- Vitellio, P.; Celano, G.; Bonfrate, L.; Gobbetti, M.; Portincasa, P.; De Angelis, M. Effects of Bifidobacterium longum and Lactobacillus rhamnosus on Gut Microbiota in Patients with Lactose Intolerance and Persisting Functional Gastrointestinal Symptoms: A Randomised, Double-Blind, Cross-Over Study. Nutrients 2019, 11, 886. [Google Scholar] [CrossRef] [Green Version]
- Wieërs, G.; Verbelen, V.; Driessche, M.V.D.; Melnik, E.; Vanheule, G.; Marot, J.-C.; Cani, P.D. Do Probiotics During In-Hospital Antibiotic Treatment Prevent Colonization of Gut Microbiota with Multi-Drug-Resistant Bacteria? A Randomized Placebo-Controlled Trial Comparing Saccharomyces to a Mixture of Lactobacillus, Bifidobacterium, and Saccharomyces. Front. Public Health 2021, 8, 1039. [Google Scholar] [CrossRef]
- Cerdó, T.; García-Santos, J.A.; Mercedes, G.B.; Campoy, C. The Role of Probiotics and Prebiotics in the Prevention and Treatment of Obesity. Nutrients 2019, 11, 635. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.-F.; Tseng, K.-C.; Chiang, S.-S.; Lee, B.-H.; Hsu, W.-H.; Pan, T.-M. Immunomodulatory and Antioxidant Potential of Lactobacillus Exopolysaccharides. J. Sci. Food Agric. 2011, 91, 2284–2291. [Google Scholar] [CrossRef]
- Patten, D.A.; Laws, A.P. Lactobacillus-Produced Exopolysaccharides and Their Potential Health Benefits: A Review. Benef. Microbes 2015, 6, 457–471. [Google Scholar] [CrossRef]
- Gibson, G.R.; Probert, H.M.; Van Loo, J.; Rastall, R.A.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Updating the concept of prebiotics. Nutr. Res. Rev. 2004, 17, 259–275. [Google Scholar] [CrossRef] [Green Version]
- Holmes, Z.C.; Silverman, J.D.; Dressman, H.K.; Wei, Z.; Dallow, E.P.; Armstrong, S.C.; Seed, P.C.; Rawls, J.F.; David, L.A. Short-Chain Fatty Acid Production by Gut Microbiota from Children with Obesity Differs According to Prebiotic Choice and Bacterial Community Composition. mBio 2020, 11, e00914-20. [Google Scholar] [CrossRef]
- D’Angelo, S.; Motti, M.L.; Meccariello, R. ω-3 and ω-6 Polyunsaturated Fatty Acids, Obesity and Cancer. Nutrients 2020, 12, 2751. [Google Scholar] [CrossRef]
- Padilla-Camberos, E.; Barragán-Álvarez, C.P.; Diaz-Martinez, N.E.; Rathod, V.; Flores-Fernández, J.M. Effects of Agave Fructans (Agave Tequilana Weber var. Azul) on Body Fat and Serum Lipids in Obesity. Plant Foods Hum. Nutr. 2018, 73, 34–39. [Google Scholar] [CrossRef]
- Yang, H.-Y.; Yang, S.-C.; Chao, J.C.-J.; Chen, J.-R. Beneficial Effects of Catechin-Rich Green Tea and Inulin on the Body Composition of Overweight Adults. Br. J. Nutr. 2011, 107, 749–754. [Google Scholar] [CrossRef]
- Buendia, J.R.; Li, Y.; Hu, F.B.; Cabral, H.J.; Bradlee, M.L.; Quatromoni, P.A.; Singer, M.R.; Curhan, G.C.; Moore, L.L. Regular Yogurt Intake and Risk of Cardiovascular Disease Among Hypertensive Adults. Am. J. Hypertens. 2018, 31, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Ditano-Vázquez, P.; Torres-Peña, J.D.; Galeano-Valle, F.; Pérez-Caballero, A.I.; Demelo-Rodríguez, P.; Lopez-Miranda, J.; Katsiki, N.; Delgado-Lista, J.; Alvarez-Sala-Walther, L.A. The Fluid Aspect of the Mediterranean Diet in the Prevention and Management of Cardiovascular Disease and Diabetes: The Role of Polyphenol Content in Moderate Consumption of Wine and Olive Oil. Nutrients 2019, 11, 2833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, J.; Astrup, A.; Lovegrove, J.A.; Gijsbers, L.; Givens, D.I.; Soedamah-Muthu, S.S. Milk and Dairy Consumption and Risk of Cardiovascular Diseases and All-Cause Mortality: Dose–Response Meta-Analysis of Prospective Cohort Studies. Eur. J. Epidemiol. 2017, 32, 269–287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohamadshahi, M.; Veissi, M.; Haidari, F.; Shahbazian, H.; Kaydani, G.-A.; Mohammadi, F. Effects of Probiotic Yogurt Consumption on Inflammatory Biomarkers in Patients with Type 2 Diabetes. BioImpacts 2014, 4, 83–88. [Google Scholar] [CrossRef]
- Shakeri, H.; Hadaegh, H.; Abedi, F.; Tajabadi-Ebrahimi, M.; Mazroii, N.; Ghandi, Y.; Asemi, Z. Consumption of Synbiotic Bread Decreases Triacylglycerol and VLDL Levels While Increasing HDL Levels in Serum from Patients with Type-2 Diabetes. Lipids 2014, 49, 695–701. [Google Scholar] [CrossRef]
- Simon, M.-C.; Strassburger, K.; Nowotny, B.; Kolb, H.; Nowotny, P.; Burkart, V.; Zivehe, F.; Hwang, J.-H.; Stehle, P.; Pacini, G.; et al. Intake of Lactobacillus reuteri Improves Incretin and Insulin Secretion in Glucose-Tolerant Humans: A Proof of Concept. Diabetes Care 2015, 38, 1827–1834. [Google Scholar] [CrossRef] [Green Version]
- Chrysohoou, C.; Stefanadis, C. Longevity and Diet. Myth or Pragmatism? Maturitas 2013, 76, 303–307. [Google Scholar] [CrossRef]
- Mihatsch, W.A.; Braegger, C.P.; Decsi, T.; Kolacek, S.; Lanzinger, H.; Mayer, B.; Moreno, L.A.; Pohlandt, F.; Puntis, J.; Shamir, R.; et al. Critical Systematic Review of the Level of Evidence for Routine Use of Probiotics for Reduction of Mortality and Prevention of Necrotizing Enterocolitis and Sepsis in Preterm Infants. Clin. Nutr. 2012, 31, 6–15. [Google Scholar] [CrossRef]
- Soedamah-Muthu, S.S.; Masset, G.; Verberne, L.; Geleijnse, J.M.; Brunner, E.J. Consumption of Dairy Products and Associations with Incident Diabetes, CHD and Mortality in the Whitehall II Study. Br. J. Nutr. 2013, 109, 718–726. [Google Scholar] [CrossRef] [Green Version]
- Tamang, J.P.; Cotter, P.D.; Endo, A.; Han, N.S.; Kort, R.; Liu, S.Q.; Mayo, B.; Westerik, N.; Hutkins, R. Fermented Foods in a Global Age: East meets West. Compr. Rev. Food Sci. Food Saf. 2020, 19, 184–217. [Google Scholar] [CrossRef] [Green Version]
- Afolayan, A.O.; Ayeni, F.A.; Ruppitsch, W. Antagonistic and Quantitative Assessment of Indigenous Lactic acid Bacteria in Different Varieties of Ogi against Gastrointestinal Pathogens. Pan Afr. Med. J. 2017, 27, 22. [Google Scholar] [CrossRef]
- Mokoena, M.P.; Mutanda, T.; Olaniran, A.O. Perspectives on the Probiotic Potential of Lactic Acid Bacteria from African Traditional Fermented Foods and Beverages. Food Nutr. Res. 2016, 60, 29630. [Google Scholar] [CrossRef] [Green Version]
- Johansen, P.G.; Owusu-Kwarteng, J.; Parkouda, C.; Padonou, S.W.; Jespersen, L. Occurrence and Importance of Yeasts in Indigenous Fermented Food and Beverages Produced in Sub-Saharan Africa. Front. Microbiol. 2019, 10, 1789. [Google Scholar] [CrossRef] [Green Version]
- Wassie, M.; Wassie, T. Isolation, Characterization and Identification of Lactic Acid Bacteria from Ready to Consume Shamita: Ethiopian Traditional Fermented Beverage. Int. J. Life Sci. Technol. 2016, 9, 51–55. [Google Scholar]
- Phiri, S.; Schoustra, S.E.; Van Den Heuvel, J.; Smid, E.J.; Shindano, J.; Linnemann, A. Fermented Cereal-Based Munkoyo Beverage: Processing Practices, Microbial Diversity and Aroma Compounds. PLoS ONE 2019, 14, e0223501. [Google Scholar] [CrossRef] [Green Version]
- Pswarayi, F.; Gänzle, M.G. Composition and Origin of the Fermentation Microbiota of Mahewu, a Zimbabwean Fermented Cereal Beverage. Appl. Environ. Microbiol. 2019, 85, e03130-18. [Google Scholar] [CrossRef] [Green Version]
- Agyei, D.; Owusu-Kwarteng, J.; Akabanda, F.; Akomea-Frempong, S. Indigenous African Fermented Dairy Products: Processing Technology, Microbiology and Health Benefits. Crit. Rev. Food Sci. Nutr. 2020, 60, 991–1006. [Google Scholar] [CrossRef]
- Nyambane, B.; Thari, W.M.; Wangoh, J.; Njage, P.M.K. Lactic Acid Bacteria and Yeasts Involved in the Fermentation of Amabere Amaruranu, a Kenyan Fermented Milk. Food Sci. Nutr. 2014, 2, 692–699. [Google Scholar] [CrossRef]
- Simatende, P.; Siwela, M.; Gadaga, T.H. Identification of Lactic Acid Bacteria and Determination of Selected Biochemical Properties in Emasi and Emahewu. S. Afr. J. Sci. 2019, 115, 1–7. [Google Scholar] [CrossRef]
- Moodley, S.S.; Dlamini, N.R.; Steenkamp, L.; Buys, E. Bacteria and Yeast Isolation and Characterisation from a South African Fermented Beverage. S. Afr. J. Sci. 2019, 115, 1–6. [Google Scholar] [CrossRef]
- Attchelouwa, C.K.; Aka-Gbézo, S.; N’Guessan, F.K.; Kouakou, C.A.; Djè, M.K. Biochemical and Microbiological Changes during the Ivorian Sorghum Beer Deterioration at Different Storage Temperatures. Beverages 2017, 3, 43. [Google Scholar] [CrossRef] [Green Version]
- Hlangwani, E.; Adebiyi, J.; Doorsamy, W.; Adebo, O. Processing, Characteristics and Composition of Umqombothi (a South African Traditional Beer). Processes 2020, 8, 1451. [Google Scholar] [CrossRef]
- Atter, A.; Obiri-Danso, K.; Amoa-Awua, W.K. Microbiological and Chemical Processes Associated with the Production of Burukutu a Traditional Beer in Ghana. Int. Food Res. J. 2014, 21, 1769–1776. [Google Scholar]
- Angelov, A.; Petrova, G.; Angelov, A.; Stefanova, P.; Bokossa, I.Y.; Tchekessi, C.; Marco, M.L.; Gotcheva, V. Molecular Identification of Yeasts and Lactic Acid Bacteria Involved in the Production of Beninese Fermented Food Degue. Open Biotechnol. J. 2017, 11, 94–104. [Google Scholar] [CrossRef]
- Rizo, J.; Guillén, D.; Farrés, A.; Díaz-Ruiz, G.; Sánchez, S.; Wacher, C.; Rodríguez-Sanoja, R. Omics in Traditional Vegetable Fermented Foods and Beverages. Crit. Rev. Food Sci. Nutr. 2018, 60, 791–809. [Google Scholar] [CrossRef] [PubMed]
- Bassi, D.; Orrù, L.; Vasquez, J.C.; Cocconcelli, P.S.; Fontana, C. Peruvian chicha: A Focus on the Microbial Populations of This Ancient Maize-Based Fermented Beverage. Microorganisms 2020, 8, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leite, A.M.O.; Miguel, M.A.L.; Peixoto, R.S.; Ruas-Madiedo, P.; Paschoalin, V.M.F.; Mayo, B.; Delgado, S. Probiotic Potential of Selected Lactic Acid Bacteria Strains Isolated from Brazilian Kefir Grains. J. Dairy Sci. 2015, 98, 3622–3632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pérez-Cataluña, A.; Elizaquível, P.; Carrasco, P.; Espinosa, J.; Reyes, D.; Wacher, C.; Aznar, R. Diversity and Dynamics of Lactic Acid Bacteria in Atole Agrio, a Traditional Maize-Based Fermented Beverage from South-Eastern Mexico, Analysed by High Throughput Sequencing and Culturing. Antonie Van Leeuwenhoek 2017, 111, 385–399. [Google Scholar] [CrossRef]
- de la Fuente-Salcido, N.M.; Castañeda-Ramírez, J.C.; García-Almendárez, B.E.; Bideshi, D.K.; Salcedo-Hernández, R.; Barboza-Corona, J.E. Isolation and Characterization of Bacteriocinogenic Lactic Bacteria from M-Tuba and Tepache, Two Traditional Fermented Beverages in México. Food Sci. Nutr. 2015, 3, 434–442. [Google Scholar] [CrossRef]
- Romero-Luna, H.E.; Hernández-Sánchez, H.; Dávila-Ortiz, G. Traditional Fermented Beverages from Mexico as a Potential Probiotic Source. Ann. Microbiol. 2017, 67, 577–586. [Google Scholar] [CrossRef]
- Velázquez-López, A.; Covatzin-Jirón, D.; Toledo-Meza, M.D.; Vela-Gutiérrez, G. Bebida Fermentada Elaborada con Bacterias ácido Lácticas Aisladas del Pozol Tradicional Chiapaneco. CienciaUAT 2018, 13, 165. [Google Scholar] [CrossRef] [Green Version]
- Ojeda-Linares, C.I.; Vallejo, M.; Lappe-Oliveras, P.; Casas, A. Traditional Management of Microorganisms in Fermented Beverages from Cactus Fruits in Mexico: An Ethnobiological Approach. J. Ethnobiol. Ethnomed. 2020, 16, 1. [Google Scholar] [CrossRef]
- Ramírez-Guzmán, K.N.; Torres-León, C.; Martinez-Medina, G.A.; de la Rosa, O.; Hernández-Almanza, A.; Alvarez-Perez, O.B.; Araujo, R.; González, L.R.; Londoño, L.; Ventura, J.; et al. Traditional Fermented Beverages in Mexico. In Fermented Beverages; Grumezescu, A.M., Holban, A.M., Eds.; Woodhead Publishing: Sawston, UK, 2019; pp. 605–635. [Google Scholar] [CrossRef]
- Escalante, A.; López Soto, D.R.; Velázquez Gutiérrez, J.E.; Giles-Gómez, M.; Bolívar, F.; López-Munguía, A. Pulque, a Traditional Mexican Alcoholic Fermented Beverage: Historical, Microbiological, and Technical Aspects. Front. Microbiol. 2016, 7, 1026. [Google Scholar] [CrossRef] [Green Version]
- Terasaki, M.; Nishida, H. Bacterial DNA Diversity among Clear and Cloudy Sakes, and Sake-kasu. Open Bioinform. J. 2020, 13, 74–82. [Google Scholar] [CrossRef]
- Yang, Z.-W.; Ji, B.-P.; Zhou, F.; Li, B.; Luo, Y.; Yang, L.; Li, T. Hypocholesterolaemic and Antioxidant Effects of Kombucha Tea in High-Cholesterol Fed Mice. J. Sci. Food Agric. 2009, 89, 150–156. [Google Scholar] [CrossRef]
- Anal, A.K. Quality Ingredients and Safety Concerns for Traditional Fermented Foods and Beverages from Asia: A Review. Fermentation 2019, 5, 8. [Google Scholar] [CrossRef] [Green Version]
- Han, K.; Bose, S.; Wang, J.-H.; Kim, B.-S.; Kim, M.J.; Kim, E.-J.; Kim, H. Contrasting Effects of Fresh and Fermented Kimchi Consumption on Gut Microbiota Composition and Gene Expression Related to Metabolic Syndrome in Obese Korean Women. Mol. Nutr. Food Res. 2015, 59, 1004–1008. [Google Scholar] [CrossRef]
- Kim, H.-Y.; Park, K.-Y. Clinical Trials of Kimchi Intakes on the Regulation of Metabolic Parameters and Colon Health in Healthy Korean Young Adults. J. Funct. Foods 2018, 47, 325–333. [Google Scholar] [CrossRef]
- Swain, M.R.; Anandharaj, M.; Ray, R.C.; Rani, R.P. Fermented Fruits and Vegetables of Asia: A Potential Source of Probiotics. Biotechnol. Res. Int. 2014, 2014, 250424. [Google Scholar] [CrossRef]
- Vina, I.; Semjonovs, P.; Linde, R.; Denina, I. Current Evidence on Physiological Activity and Expected Health Effects of Kombucha Fermented Beverage. J. Med. Food 2014, 17, 179–188. [Google Scholar] [CrossRef] [Green Version]
- Kushida, M.; Okouchi, R.; Iwagaki, Y.; Asano, M.; Du, M.X.; Yamamoto, K.; Tsuduki, T. Fermented Soybean Suppresses Visceral Fat Accumulation in Mice. Mol. Nutr. Food Res. 2018, 62, 1701054. [Google Scholar] [CrossRef]
- Soni, S.; Dey, G. Perspectives on Global Fermented Foods. Br. Food J. 2014, 116, 1767–1787. [Google Scholar] [CrossRef]
- Baschali, A.; Tsakalidou, E.; Kyriacou, A.; Karavasiloglou, N.; Matalas, A.-L. Traditional Low-Alcoholic and Non-alcoholic Fermented Beverages Consumed in European Countries: A Neglected Food Group. Nutr. Res. Rev. 2017, 30, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Rosa, D.D.; Dias, M.M.S.; Grześkowiak, Ł.M.; Reis, S.A.; Conceição, L.L.; Maria do Carmo, G.P. Milk Kefir: Nutritional, Microbiological and Health Benefits. Nutr. Res. Rev. 2017, 30, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Medici, M.; Vinderola, C.G.; Perdigón, G. Gut Mucosal Immunomodulation by Probiotic Fresh Cheese. Int. Dairy J. 2004, 14, 611–618. [Google Scholar] [CrossRef]
- Songisepp, E.; Kullisaar, T.; Hütt, P.; Elias, P.; Brilene, T.; Zilmer, M.; Mikelsaar, M. A New Probiotic Cheese with Antioxidative and Antimicrobial Activity. J. Dairy Sci. 2004, 87, 2017–2023. [Google Scholar] [CrossRef] [Green Version]
- Franz, C.M.; Huch, M.; Mathara, J.M.; Abriouel, H.; Benomar, N.; Reid, G.; Galvez, A.; Holzapfel, W.H. African Fermented Foods and Probiotics. Int. J. Food Microbiol. 2014, 190, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Diaz, M.; Kellingray, L.; Akinyemi, N.; Adefiranye, O.O.; Olaonipekun, A.B.; Bayili, G.R.; Ibezim, J.; Du Plessis, A.S.; Houngbédji, M.; Kamya, D.; et al. Comparison of the Microbial Composition of African Fermented Foods Using Amplicon Sequencing. Sci. Rep. 2019, 9, 13863. [Google Scholar] [CrossRef] [Green Version]
- Bernal Castro, C.A.; Díaz-Moreno, C.; Gutiérrez-Cortés, C. Probióticos y Prebióticos en Matrices de Origen Vegetal: Avances en el Desarrollo de Bebidas de Frutas. Rev. Chil. Nutr. 2017, 44, 383–392. [Google Scholar] [CrossRef] [Green Version]
- Lau, E.; Carvalho, D.; Pina-Vaz, C.; Barbosa, J.-A.; Freitas, P.; Stagi, S.; Iurato, C.; Lapi, E.; Cavalli, L.; Brandi, M.L.; et al. Beyond Gut Microbiota: Understanding Obesity and Type 2 Diabetes. Hormones 2015, 14, 358–369. [Google Scholar] [CrossRef] [Green Version]
- Mota de Carvalho, N.; Costa, E.M.; Silva, S.; Pimentel, L.; Fernandes, T.H.; Pintado, M.E. Fermented Foods and Beverages in Human Diet and Their Influence on Gut Microbiota and Health. Fermentation 2018, 4, 90. [Google Scholar] [CrossRef] [Green Version]
- Başar Gökcen, B.; Sezgin, A. Health Benefits of Fermented Foods. Crit. Rev. Food Sci. Nutr. 2017, 59, 506–527. [Google Scholar]
- Ivey, K.L.; Hodgson, J.M.; Kerr, D.A.; Thompson, P.L.; Stojceski, B.; Prince, R.L. The Effect of Yoghurt and Its Probiotics on Blood Pressure and Serum Lipid Profile; A Randomised Controlled Trial. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 46–51. [Google Scholar] [CrossRef] [Green Version]
- Rajkumar, H.; Mahmood, N.; Kumar, M.; Varikuti, S.R.; Challa, H.R.; Myakala, S.P. Effect of Probiotic (VSL#3) and Omega-3 on Lipid Profile, Insulin Sensitivity, Inflammatory Markers, and Gut Colonization in Overweight Adults: A Randomized, Controlled Trial. Mediat. Inflamm. 2014, 2014, 348959. [Google Scholar] [CrossRef] [Green Version]
- Mitrea, L.; Nemeş, S.-A.; Szabo, K.; Teleky, B.-E.; Vodnar, D.-C. Guts Imbalance Imbalances the Brain: A Review of Gut Microbiota Association with Neurological and Psychiatric Disorders. Front. Med. 2022, 9, 813204. [Google Scholar]
- Yilmaz, B.; Bangar, S.P.; Echegaray, N.; Suri, S.; Tomasevic, I.; Lorenzo, J.M.; Melekoglu, E.; Rocha, J.M.; Ozogul, F. The Impacts of Lactiplantibacillus plantarum on the Functional Properties of Fermented Foods: A Review of Current Knowledge. Microorganisms 2022, 10, 826. [Google Scholar] [CrossRef]
- Márquez-Aguirre, A.L.; Camacho-Ruíz, R.M.; Mercado, Y.K.G.; Padilla-Camberos, E.; González-Ávila, M.; Gálvez-Gastélum, F.J.; Díaz-Martínez, N.E.; Ortuño-Sahagún, D. Fructans from Agave tequilana with a Lower Degree of Polymerization Prevent Weight Gain, Hyperglycemia and Liver Steatosis in High-Fat Diet-Induced Obese Mice. Plant Foods Hum. Nutr. 2016, 71, 416–421. [Google Scholar] [CrossRef] [Green Version]
- Reimer, R.A.; Willis, H.J.; Tunnicliffe, J.M.; Park, H.; Madsen, K.L.; Soto-Vaca, A. Inulin-Type Fructans and Whey Protein Both Modulate Appetite but Only Fructans Alter Gut Microbiota in Adults with Overweight/Obesity: A Randomized Controlled Trial. Mol. Nutr. Food Res. 2017, 61, 1700484. [Google Scholar] [CrossRef]
- López-Velázquez, G.; Parra-Ortiz, M.; Mora, I.D.L.M.-D.L.; García-Torres, I.; Flores, S.E.; Alcántara-Ortigoza, M.A.; Angel, A.G.-D.; Velázquez-Aragón, J.; Ortiz-Hernández, R.; Cruz-Rubio, J.M.; et al. Effects of Fructans from Mexican Agave in Newborns Fed with Infant Formula: A Randomized Controlled Trial. Nutrients 2015, 7, 8939–8951. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Abundis, E.; Barrera-Durán, C.; González-Ortiz, M.; Hernández-Salazar, E. Effect of Simvastatin Plus Inulin in Comparison with Simvastatin Plus Ezetimibe on the Treatment of Mixed Dyslipidemia. Gaceta Médica de México 2016, 152, 88–95. [Google Scholar]
- Contreras-Haro, B.; Robles-Cervantes, J.A.; Gonzalez-Ortiz, M.; Martinez-Abundis, E.; Espinel-Bermudez, C.; Gallegos-Arreola, M.P.; Morgado-Castillo, K.C. The Effect of Agave tequilana Weber Inulin on Postprandial Ghrelin Concentration in Obese Patients. J. Med. Food 2017, 20, 197–199. [Google Scholar] [CrossRef]
- Beltrán-Barrientos, L.M.; González-Córdova, A.F.; Hernández-Mendoza, A.; Torres-Inguanzo, E.H.; Astiazarán-García, H.; Esparza-Romero, J.; Vallejo-Cordoba, B. Randomized Double-Blind Controlled Clinical Trial of the Blood Pressure–Lowering Effect of Fermented Milk with Lactococcus Lactis: A Pilot Study. J. Dairy Sci. 2018, 101, 2819–2825. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Figueroa, J.; González-Córdova, A.; Astiazaran-García, H.; Hernandez-Mendoza, A.; Vallejo-Cordoba, B. Antihypertensive and Hypolipidemic Effect of Milk Fermented by Specific Lactococcus Lactis Strains. J. Dairy Sci. 2013, 96, 4094–4099. [Google Scholar] [CrossRef] [Green Version]
- Rahat-Rozenbloom, S.; Fernandes, J.; Cheng, J.; Gloor, G.B.; Wolever, T.M.S. The Acute Effects of Inulin and Resistant Starch on Postprandial Serum Short-Chain Fatty Acids and Second-Meal Glycemic Response in Lean and Overweight Humans. Eur. J. Clin. Nutr. 2017, 71, 227–233. [Google Scholar] [CrossRef] [Green Version]
- Sai Ram, M.; Anju, B.; Pauline, T.; Dipti, P.; Kain, A.K.; Mongia, S.S.; Sharma, S.K.; Singh, B.; Singh, R.; Ilavazhagan, G.; et al. Effect of Kombucha Tea on Chromate(VI)-Induced Oxidative Stress in Albino Rats. J. Ethnopharmacol. 2000, 71, 235–240. [Google Scholar] [CrossRef]
- Aloulou, A.; Hamden, K.; Elloumi, D.; Ali, M.B.; Hargafi, K.; Jaouadi, B.; Ayadi, F.; Elfeki, A.; Ammar, E. Hypoglycemic and Antilipidemic Properties of Kombucha Tea in Alloxan-Induced Diabetic Rats. BMC Complement. Altern. Med. 2012, 12, 63. [Google Scholar] [CrossRef] [Green Version]
- Byun, M.-S.; Yu, O.-K.; Cha, Y.-S.; Park, T.-S. Korean Traditional Chungkookjang Improves Body Composition, Lipid Profiles and Atherogenic Indices in Overweight/Obese Subjects: A Double-Blind, Randomized, Crossover, Placebo-Controlled Clinical Trial. Eur. J. Clin. Nutr. 2016, 70, 1116–1122. [Google Scholar] [CrossRef]
- Tjasa Subandi, S.; Kartawidjajaputra, F.; Silo, W.; Yogiara, Y.; Suwanto, A. Tempeh Consumption Enhanced Beneficial Bacteria in the Human Gut. Food Res. 2018, 3, 57–63. [Google Scholar]
- Helmyati, S.; Shanti, K.; Sari, F.; Sari, M.; Atmaka, D.; Pratama, R.; Wigati, M.; Wisnusanti, S.; Nisa’, F.; Rahayu, E. Synbiotic Fermented Milk with Double Fortification (Fe-Zn) as a Strategy to Address Stunting: A Randomized Controlled Trial among Children under Five in Yogyakarta, Indonesia. Processes 2021, 9, 543. [Google Scholar] [CrossRef]
- Amoutzopoulos, B.; Löker, G.B.; Samur, G.; Çevikkalp, S.A.; Yaman, M.; Köse, T.; Pelvan, E. Effects of a Traditional Fermented Grape-Based Drink ‘Hardaliye’ on Antioxidant Status of Healthy Adults: A Randomized Controlled Clinical Trial. J. Sci. Food Agric. 2013, 93, 3604–3610. [Google Scholar] [CrossRef]
- Donmez, N.; Kısadere, I.; Balaban, C.; Kadiralieva, N. Effects of Traditional Homemade Koumiss on Some Hematological and Biochemical Characteristics in Sedentary Men Exposed to Exercise. Biotech. Histochem. 2014, 89, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Herrera, A.; Mulder, K.; Bouritius, H.; Rubio, R.; Muñoz, A.; Agosti, M.; Lista, G.; Corvaglia, L.; Ludwig, T.; Abrahamse-Berkeveld, M.; et al. Gastrointestinal Tolerance, Growth and Safety of a Partly Fermented Formula with Specific Prebiotics in Healthy Infants: A Double-Blind, Randomized, Controlled Trial. Nutrients 2019, 11, 1530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bogovič Matijašić, B.; Obermajer, T.; Lipoglavšek, L.; Sernel, T.; Locatelli, I.; Kos, M.; Šmid, A.; Rogelj, I. Effects of Synbiotic Fermented Milk Containing Lactobacillus acidophilus La-5 and Bifidobacterium animalis ssp. lactis BB-12 on the Fecal Microbiota of Adults with Irritable Bowel Syndrome: A Randomized Double-Blind, Placebo-Controlled Trial. J. Dairy Sci. 2016, 99, 5008–5021. [Google Scholar] [CrossRef] [PubMed]
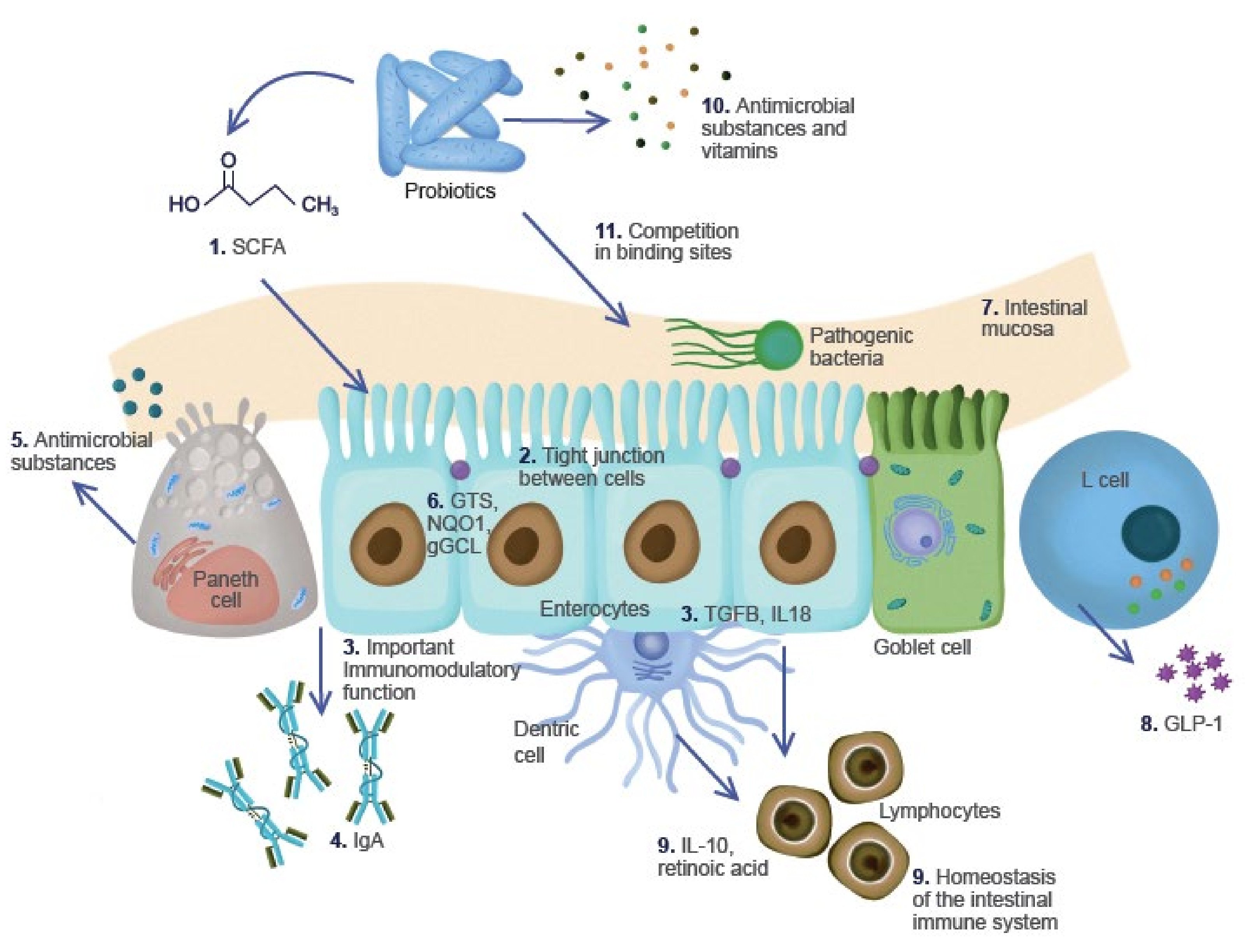
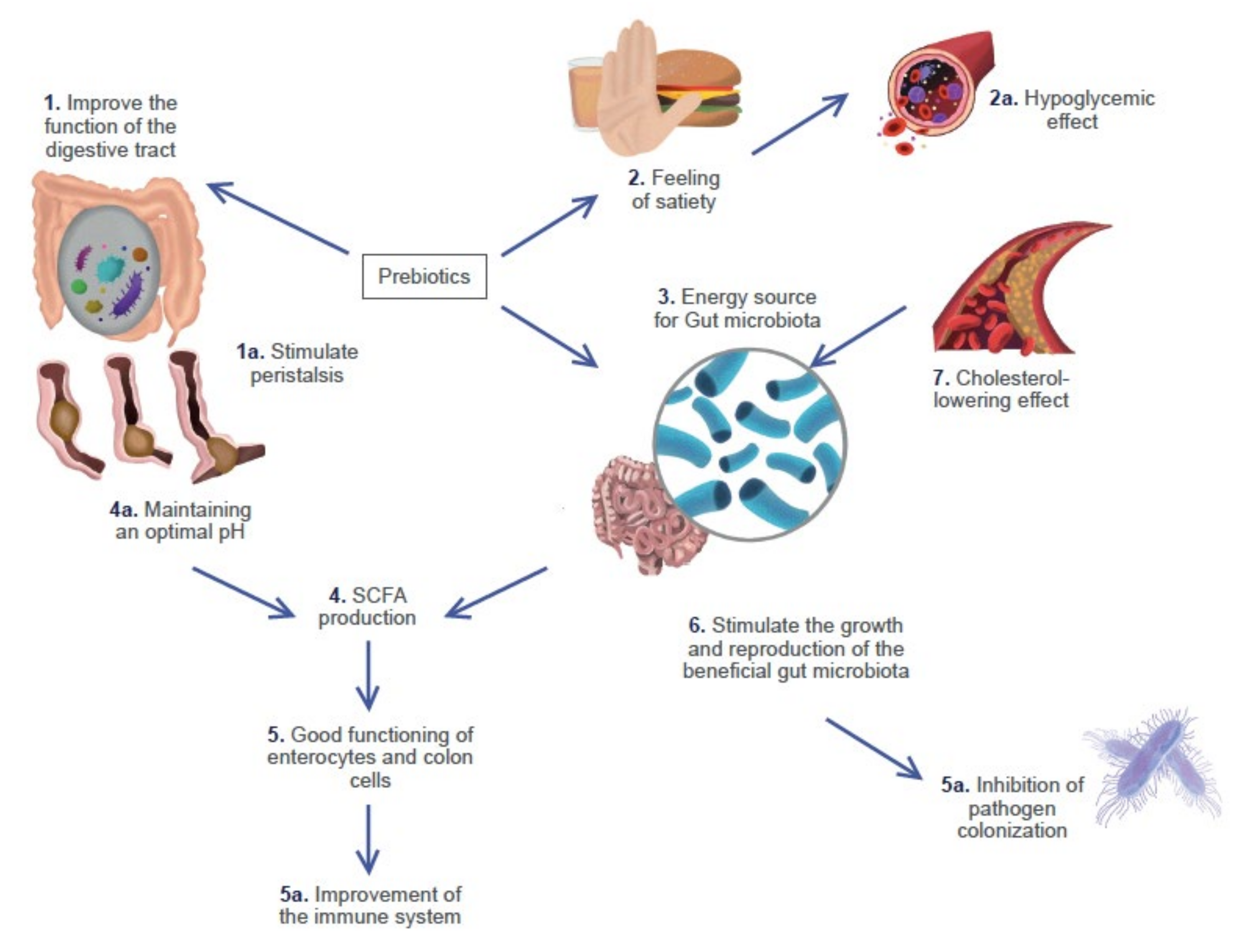
| Name of the Product | Description | Microorganisms Identified | Region of Origin | References |
|---|---|---|---|---|
| Africa | ||||
| Ogi | Fermented cereal pudding (maize, sorghum, or millet) | Candida krusei, Lactiplantibacillus plantarum, Limosilactobacillus fermentum, Saccharomyces cerevisiae, Acetobacter spp., Corynebacterium spp. | West Africa | [42] |
| Iru | Fermented locust beans (Parkia biglobosa) | Bacillus, Staphylococcus spp. | West Africa | [43] |
| Gari | Cassava | Leuconostoc mesenteroide, Lactiplantibacillus plantarum, Bacillus subtilis, Candida krusei | West Africa | [43] |
| Togwa | Cereal-based, starch-saccharified, non-alcoholic, lactic acid-containing gruel food (Cassava, maize, sorghum, millet) | Lactobacillus spp., Pediococcus pentosaceus, Weissella confusa, Issatchenkia orientalis, Saccharomyces cerevisiae, Candida pelliculosa and Candida tropicalis. | Eastern Africa | [44] |
| Bushera | Fermented cereal beverage (sorgum) | Lactobacillus spp., Streptococcus spp., Leuconostoc spp., Pediococcus spp., Weissella spp. | Eastern Africa | [43] |
| Shamita | Fermented beverage (barley) | Lactobacillus spp. | North East Africa | [45] |
| Munkoyo | Mildly fermented drink made from pounded roots mixed with bits of maize | Weisella and Lactobacillus spp. | Southern Africa | [46] |
| Mahewu | Fermented maize or sorghum beverage | Lactiplantibacillus plantarum, Limosilactobacillus fermentum | Southern Africa | [47] |
| Mabisi/Amasi | Fermented milk drink | Lactococcus, Lactobacillus and Streptococcus spp., Leuconostoc spp. | Southern Africa | [48] |
| Amabere amaruranu | Fermented milk drink | Streptococcus thermophilus, Lactiplantibacillus plantarum, Leuconostoc mesenteroides; Yeasts | Southern Africa | [49] |
| Marula/buganu | Fermented juice and pulp (marula fruit) | Yeasts, Lactobacillus spp. | Southern Africa | [50] |
| Motoho | Fermented sorghum beverage | Lactiplantibacillus plantarum, Limosilactobacillus fermentum, Lactobacillus coryniformis, Lacticaseibacillus paracasei paracasei, Candida lambica, Candida kefyr, Candida glabarata, Candida pelliculosa; Rhodotorula mucilaginosa, Geotrichum, candidum, Geotrichum silvicola | South Africa | [51] |
| Chibuku | Sorghum beer | Lactobacillus spp., Saccharomyces cerevisiae | Southern Africa | [52] |
| Umqombothi | Beer made from maize and sorghum | Lactobacillus spp., Saccharomyces cerevisiae | Southern Africa | [53] |
| Burukutu | Alcoholic beverage, brewed from the grains of Guinea corn (Sorghum bicolor) and millet (Pennisetum glaucum). | Escherichia, Staphylococcus, Bacillus, Lactobacillus, Leuconostoc, Acetobacter | Nigeria, Benin, Ghana | [54] |
| Dégué | Fermented millet dough | Escherichia, Bacillus, Lactobacillus, Enterococcus | Burkina Faso | [55] |
| America | ||||
| Champuz | Fermented maize or rice beverage | Unknow | Colombia, Peru | [56] |
| Chicha | Fermented maize beverage | Enterococcus, Lactococcus, Streptococcus, Weissella, Leuconostoc, Lactobacillus, Saccharomyces. | Colombia, Peru, Argentina | [57] |
| Kefir grain | Fermented lactoalcoholic milk | Leuconostoc mesenteroides, Lactococcus lactis sp. cremoris, Lacticaseibacillus paracasei | Brazil | [58] |
| Axokot/Atole agrio | Fermented maize beberage | Leuconostoc, Lactococcus, Lactobacillus, Pediococcus, Weisella | Southeast Mexico | [59] |
| Tepache | Fermented alcoholic pineapple drink | Lactiplantibacillus plantarum, Leuconostoc mesenteroides, Lactobacillus sp., Lactococcus lactis, Hanseniaspora, Bacillus spp., Torulopsis, Saccharomyces and Candida | Mexico | [60] |
| Tesgüiño | Maize beer | Lactobacillus spp. Candida, Saccharomyces, Hansenula | North Western and North Mexico | [61] |
| Tuba | Alcoholic beverage from coconut palm | Data not available | Western Mexico | [56] |
| Pozol | Drink based on cocoa and maize. | Lactococcus, Lactobacillus, Leuconostoc, Streptococcus, Enterococcus, Weisella, Saccharomyces, Candida, Aspergillus, Penicillum, Rhizopus | Southeast Mexico | [61,62] |
| Colonche | Fermented red prickly beverage | Candida valida, Saccharomyces cerevisiae, Torulopsis taboadae, Pichia fermentans | North of Mexico | [63,64] |
| Pulque | Fermented alcoholic drink (several species of Agave) | Lactobacillus, Leuconostoc, Microbacterium, Flavobacterium, Acetobacter, Gluconobacter, Zymomonas, Saccharomyces | Central Mexico | [65] |
| Asia | ||||
| Kombucha | Fermented tea beverage | Komagataeibacter xylinus, Brettanomyces bruxellensis, Acetobacter pasteurianus, Acetobacter xylinum, Acetobater aceti, Saccharomyces cerevisiae, Zygosaccharomyces bailii, Zygosaccharomyces spp., Gluconacetobacter | China | [2,66,67] |
| Resistant starch | Insoluble type of cereal fiber from grains | FOS | Japan, EUA, Europe. | [68] |
| Catechin-rich green tea | Green tea from Camellia sinensis plant | Epicatechin (EC) Epicatechin-3-gallate (ECG) Epigallocatechin (EGC) Epigallocatechin-3-gallate (EGCG) | China | [31] |
| Kimchi | Cabbage, radish, various vegetables | Leuconostoc mesenteroides, Levilactobacillus brevis, Lactiplantibacillus plantarum | Korea | [69,70] |
| Tempeh | Fermented boiled and dehulled soybeans | Enterococcus faecium, Rhizopus oryzae, Rhizopus oligoporus, Mucor indicus Mucor circinelloides, Geotrichum candidum, Aureobasidium pullulans, Alternaria alternata, Cladosporium oxysporum, Trichosporon beigelii, Clavispora lusitaniae, Candida maltosa, Candida intermedia, Yarrowia lipolytica, Lodderomyces elongisporus, Rhodotorula mucilaginosa, Candida sake, Hansenula fabiani, Candida tropicalis, Candida parapsilosis, Pichia membranefaciens, Rhodotorula rubra, Candida rugosa, Candida curvata, Hansenula anomola | Indonesia | [2] |
| Khalpi | Cucumber | Lactiplantibacillus plantarum, Levilactobacillus brevis, Leuconostoc fallax | Nepal | [71,72] |
| Chungkookjang | Fermented soybean | Bacillus subtilis, Bacillus licheniformis | Korea | [73] |
| Miso | Fermented soybean paste | Bacillus subtilis, Bacillus amyloliquefaciens, Staphylococcus gallinarum, Staphylococcus kloosii, Lactococcus sp. GM005 | Japan | [2] |
| Sake | Rice wine | Fructilactobacillus fructivorans, Lactobacillus homohiochi. | Japan | [66] |
| Burong mustala | Mustard leaf | Levilactobacillus brevis | Philippines | [71] |
| Europe | ||||
| Sourdough bread | Bread made from longer ferment | Data not available | Middle East and Europe | [2,74] |
| Fermented olives | Olives | Lactiplantibacillus plantarum, Lactobacillus pentosus, Lacticaseibacillus casei | Spain and Portugal | [74,75] |
| Salsiccia, Soppressata | Chopped pork | Micrococci, Staphylococci | Italy | [74] |
| Kefir | Fermented milk beverage | Lentilactobacillus kefiri, Lacticaseibacillus paracasei, Lactobacillus parabuchneri, Lacticaseibacillus casei, Lactobacillus lactis, Acetobacter lovaniensis, Kluyveromyces lactis, Saccharomyces cerevisiae | Russia, Europe, Middle East | [76] |
| Fermented Cheese | Milk | Lactococcus lactis, Bifidobacterium bifidum, Lactobacillus acidophilus, Lacticaseibacillus paracasei | Europe, Middle East | [77,78] |
| Sauerkraut | Fermented cabbage | Lactiplantibacillus plantarum | Germany | [6] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuamatzin-García, L.; Rodríguez-Rugarcía, P.; El-Kassis, E.G.; Galicia, G.; Meza-Jiménez, M.d.L.; Baños-Lara, M.d.R.; Zaragoza-Maldonado, D.S.; Pérez-Armendáriz, B. Traditional Fermented Foods and Beverages from around the World and Their Health Benefits. Microorganisms 2022, 10, 1151. https://doi.org/10.3390/microorganisms10061151
Cuamatzin-García L, Rodríguez-Rugarcía P, El-Kassis EG, Galicia G, Meza-Jiménez MdL, Baños-Lara MdR, Zaragoza-Maldonado DS, Pérez-Armendáriz B. Traditional Fermented Foods and Beverages from around the World and Their Health Benefits. Microorganisms. 2022; 10(6):1151. https://doi.org/10.3390/microorganisms10061151
Chicago/Turabian StyleCuamatzin-García, Leonel, Paola Rodríguez-Rugarcía, Elie Girgis El-Kassis, Georgina Galicia, María de Lourdes Meza-Jiménez, Ma. del Rocío Baños-Lara, Diego Salatiel Zaragoza-Maldonado, and Beatriz Pérez-Armendáriz. 2022. "Traditional Fermented Foods and Beverages from around the World and Their Health Benefits" Microorganisms 10, no. 6: 1151. https://doi.org/10.3390/microorganisms10061151
APA StyleCuamatzin-García, L., Rodríguez-Rugarcía, P., El-Kassis, E. G., Galicia, G., Meza-Jiménez, M. d. L., Baños-Lara, M. d. R., Zaragoza-Maldonado, D. S., & Pérez-Armendáriz, B. (2022). Traditional Fermented Foods and Beverages from around the World and Their Health Benefits. Microorganisms, 10(6), 1151. https://doi.org/10.3390/microorganisms10061151








