Fermented Soy Products and Their Potential Health Benefits: A Review
Abstract
1. Introduction
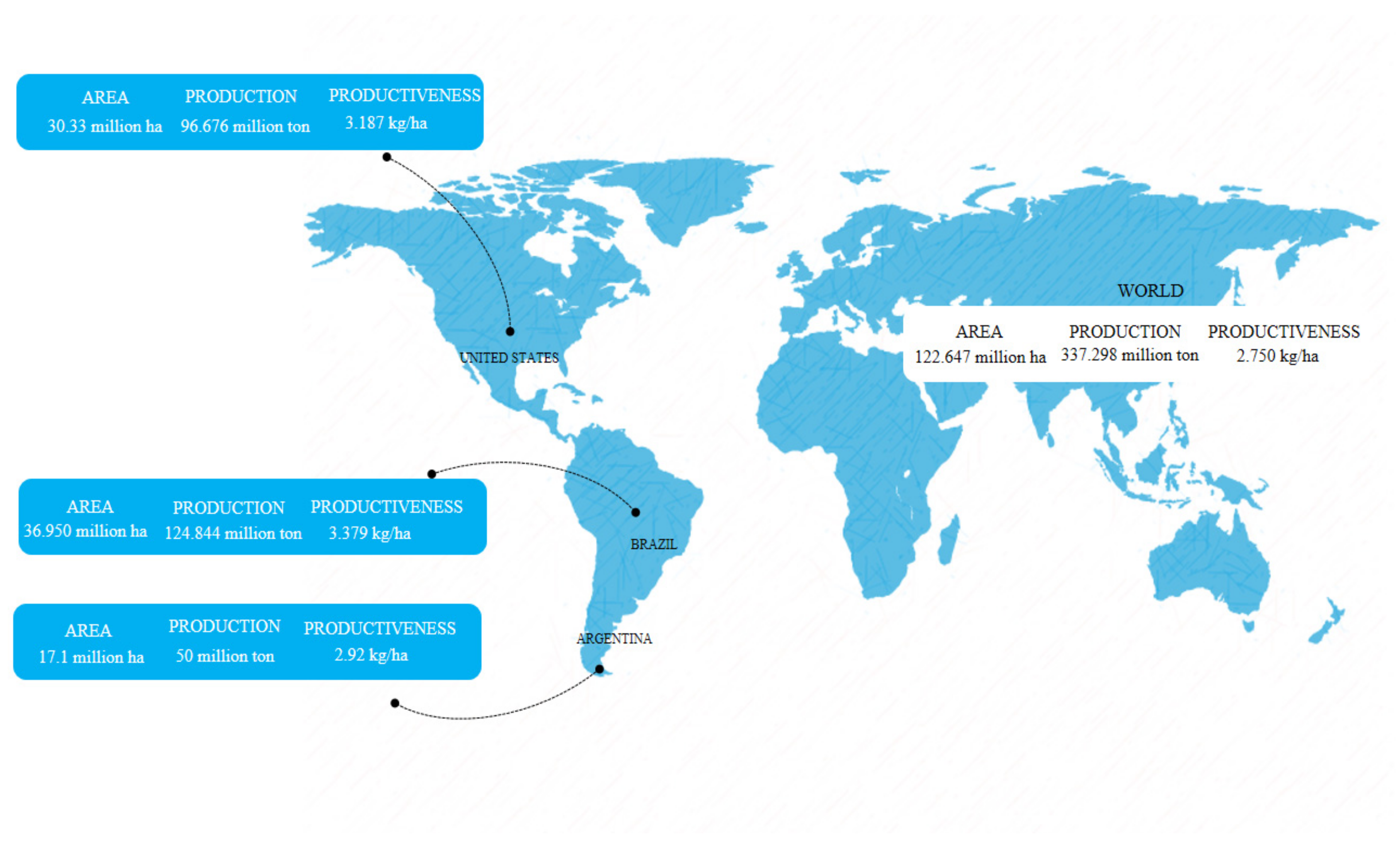
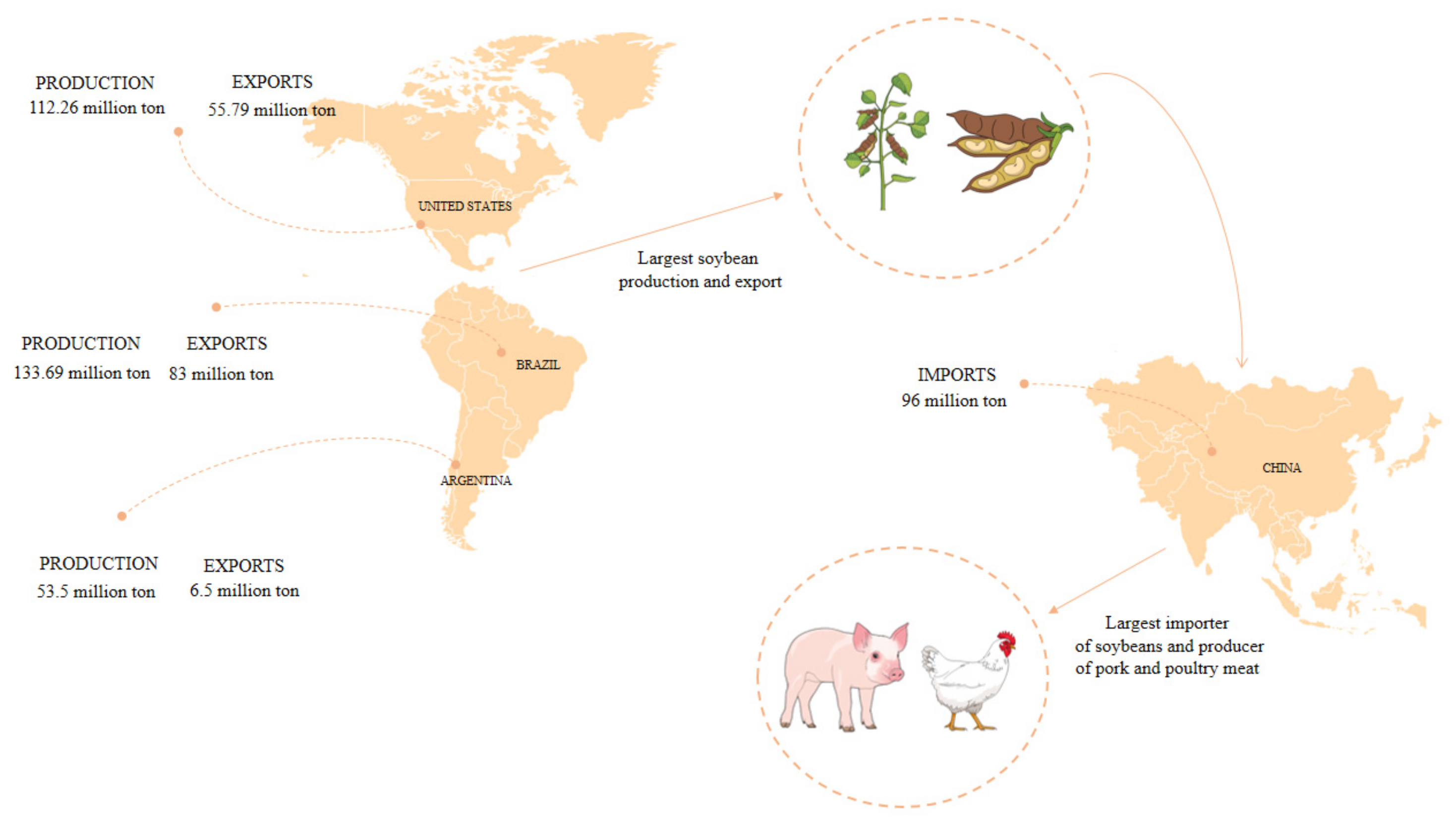
2. Soybean
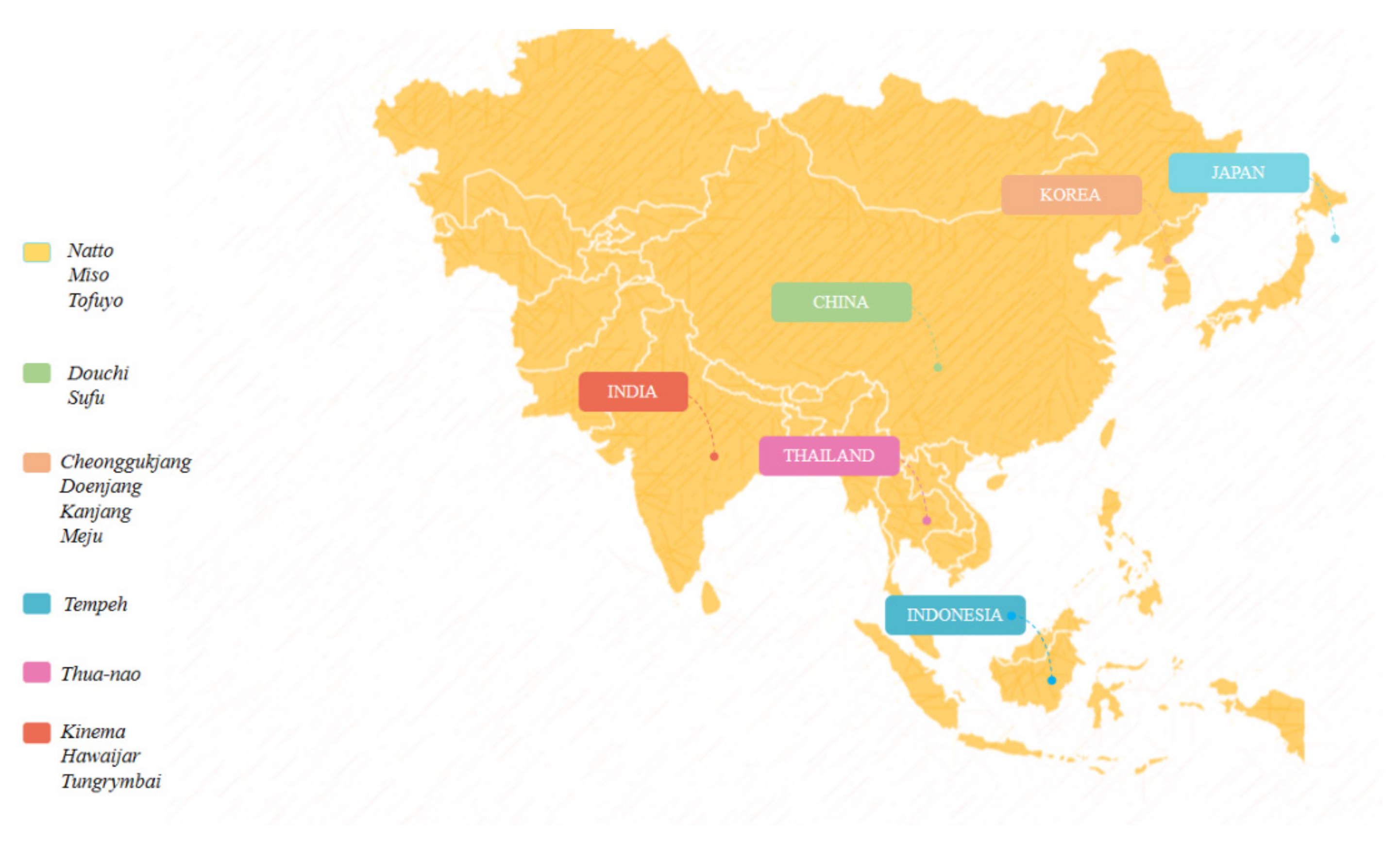
3. Fermented Soybean Products
4. Nutritional Changes in Fermented Soybean Products
5. Potential Beneficial Health Effect of Soybean Fermented Product
5.1. Antioxidant Effect
5.2. Anticancer Effect
5.3. Anti-Obesity and Antidiabetic Effect
5.4. Anti-Inflammatory Effect
5.5. Preventive Effect against Cardiovascular Disease
5.6. Neuroprotective Effect
5.7. Anti-Aging Effect
6. SARS-CoV-2
7. Bioactive Compounds in Soybean Fermented Product
| Soybean Products | Bioactive Compounds | Health Benefits | References |
|---|---|---|---|
| Tempeh | Isoflavone aglycone | Antioxidant properties | [183] |
| Tempeh | Isoflavone aglycone | Protection of HepG2 cells from oxidative stress | [176] |
| Tempeh | Genistein | Immunomodulatory Function | [184] |
| Tempeh | Trans-cinnamic acid | Antioxidant properties | [59] |
| Chungkookjang | Poly-γ-glutamic acid (γ-PGA) | Prevention of memory loss from Alzheimer’s and cerebral ischemia | [147] |
| Chungkookjang | Daidzein | Anti-diabetic property | [177] |
| Chungkookjang | Poly-γ-glutamic acid | Anti-obesity effect | [185] |
| Chungkookjang | Bacillomycin D and surfactin | Antimicrobial activity | [186] |
| Natto | Nattokinase (NK) | Anti-thrombotic and anti-coagulant activities | [179] |
| Natto | Vitamin K2 | Reducing osteoporotic fracture risk | [187] |
| Natto | Bacillopeptidase F | Anti-thrombotic and blood pressure-lowering | [188] |
| Natto | Nattokinase (NK) | Fibrinolytic activity | [189] |
| Miso | Isoflavones aglycones | Anti-tumoral activity | [190] |
| Miso | Isoflavones aglycones | Protective effects against stroke | [191] |
| Miso | Isoflavones aglycones | Sympathetic nerve activity | [192] |
| Doenjang | Linoleic acid and Genistein | Antimutagenic active | [182] |
| Doenjang | Genistein | Antimutagenic and anticancer activities | [193] |
| Doenjang | Genistein | Anti-obesity effects | [194] |
| Kinema | Poly-γ-glutamic acid (γ-PGA) | Suppression of post prandial hyperglycemia | [195] |
| Kinema | Isoflavones aglycones | Antioxidant properties | [42] |
| Kinema | Group B saponins | Prevention of dietary hypercholesterolemia | [196] |
| Douchi | β-glucosidase and protease | Antioxidant activity | [197] |
| Sufu | Isoflavones aglycones | Enhancement of the physiological function | [198] |
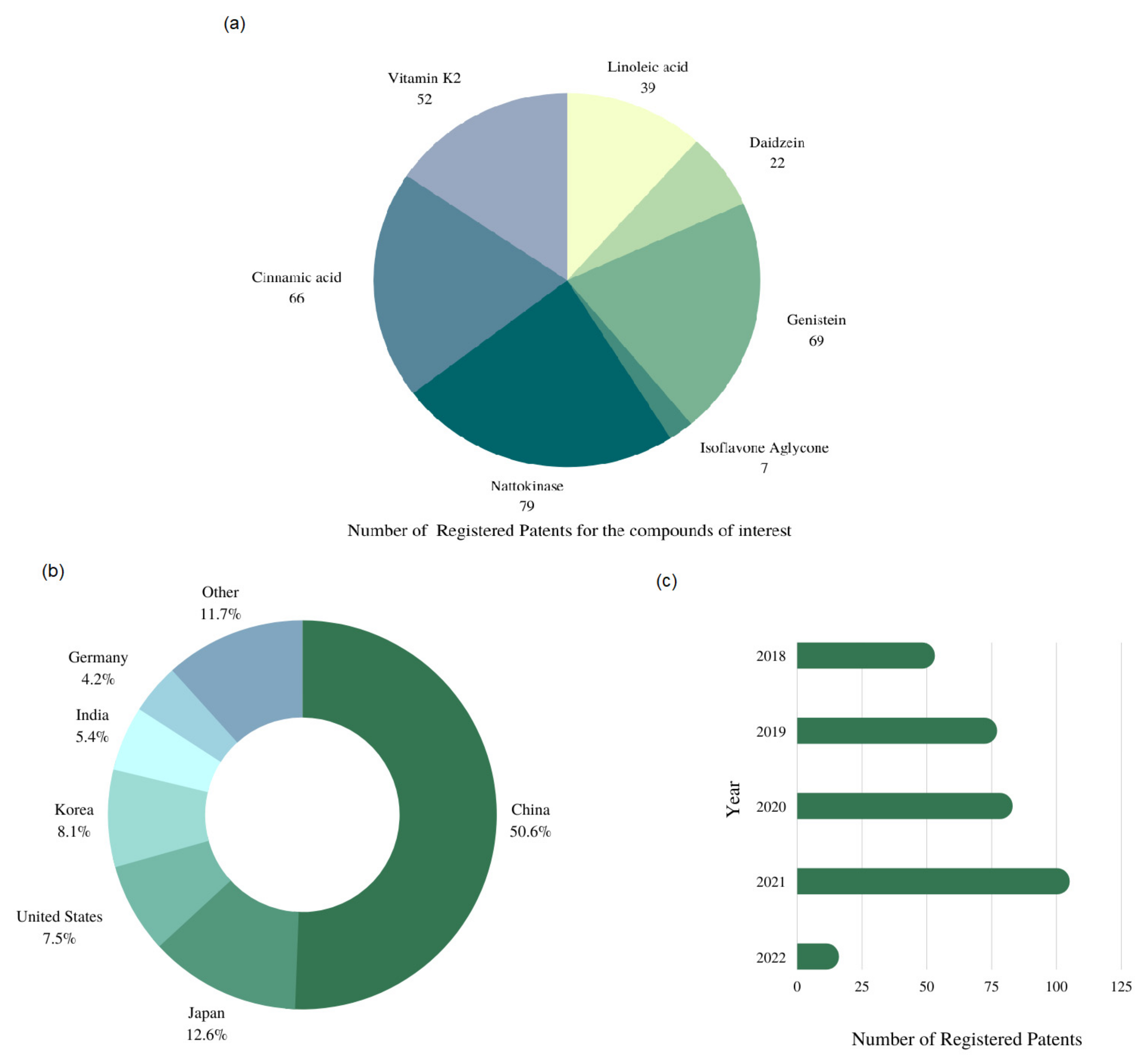
8. Recent Patents and Innovations on Bioactive Compounds in Soybean Fermented Product
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ahmad, A.; Ramasamy, K.; Bakar, A.; Majeed, A.; Ahmad, A.; Ramasamy, K.; Bakar, A.; Majeed, A.; Mani, V. Enhancement of β-Secretase Inhibition and Antioxidant Activities of Tempeh, a Fermented Soybean Cake through Enrichment of Bioactive Aglycones. Pharm. Biol. 2015, 53, 758–766. [Google Scholar] [CrossRef]
- Kangari, P.; Farahany, T.Z.; Golchin, A.; Ebadollahzadeh, S.; Salmaninejad, A.; Mahboob, S.A.; Nourazarian, A. Enzymatic Antioxidant and Lipid Peroxidation Evaluation in the Newly Diagnosed Breast Cancer Patients in Iran. Asian Pac. J. Cancer Prev. 2018, 19, 3511–3515. [Google Scholar] [CrossRef] [PubMed]
- Nematgorgani, S.; Agah, S.; Shidfar, F.; Gohari, M. Effects of Urtica Dioica Leaf Extract on Inflammation, Oxidative Stress, ESR, Blood Cell Count and Quality of Life in Patients with in Fl Ammatory Bowel Disease. J. Herb. Med. 2017, 9, 32–41. [Google Scholar] [CrossRef]
- Tehrani, S.S.; Karimian, A.; Parsian, H.; Majidinia, M.; Yousefi, B. Multiple Functions of Long Non-Coding RNAs in Oxidative Stress, DNA Damage Response and Cancer Progression. J. Cell. Biochem. 2018, 119, 223–236. [Google Scholar] [CrossRef] [PubMed]
- Taleb, A.; Ahmad, K.A.; Ihsan, A.U.; Qu, J.; Lin, N.; Hezam, K.; Koju, N.; Hui, L.; Qilong, D. Antioxidant Effects and Mechanism of Silymarin in Oxidative Stress Induced Cardiovascular Diseases. Biomed. Pharmacother. 2018, 102, 689–698. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Panahi, Y.; Javadi, B.; Sahebkar, A. The Underlying Role of Oxidative Stress in Neurodegeneration: A Mechanistic Review. CNS Neurol. Disord.-Drug Targets 2018, 17, 207–215. [Google Scholar] [CrossRef]
- Bakhtiari, A.; Hajian-Tilaki, K.; Omidvar, S.; Amiri, F.N. Association of Lipid Peroxidation and Antioxidant Status with Metabolic Syndrome in Iranian Healthy Elderly Women. Biomed. Rep. 2017, 7, 331–336. [Google Scholar] [CrossRef]
- Lemasters, J.J.; Jaeschke, H. Oxidative Stress and Inflammation in the Liver. Liver 2020, 2, 714–727. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Munekata, P.E.S. Phenolic Compounds of Green Tea: Health Benefits and Technological Application in Food. Asian Pac. J. Trop. Biomed. 2016, 6, 709–719. [Google Scholar] [CrossRef]
- Khan, Y.; Khan, S.M.; Haq, I.u.; Farzana, F.; Abdullah, A.; Mehmood, A.A.; Alamri, S.; Hashem, M.; Sakhi, S.; Asif, M.; et al. Antioxidant Potential in the Leaves of Grape Varieties (Vitis vinifera L.) Grown in Different Soilcompositions. Arab. J. Chem. 2021, 14, 103412. [Google Scholar] [CrossRef]
- Duan, Y.; Santiago, F.E.M.; dos Reis, A.R.; de Figueiredo, M.A.; Zhou, S.; Thannhauser, T.W.; Li, L. Genotypic Variation of Flavonols and Antioxidant Capacity in Broccoli. Food Chem. 2021, 385, 127997. [Google Scholar] [CrossRef]
- Hariram, S.; Nile, A.; Oh, J.; Kai, G. Soybean Processing Waste: Potential Antioxidant, Cytotoxic and Enzyme Inhibitory Activities. Food Biosci. 2020, 38, 100778. [Google Scholar] [CrossRef]
- Moreno-Montoro, M.; Jauregi, P.; Navarro-Alarcón, M.; Olalla-Herrera, M.; Giménez-Martínez, R.; Amigo, L.; Miralles, B. Bioaccessible Peptides Released by in Vitro Gastrointestinal Digestion of Fermented Goat Milks. Anal. Bioanal. Chem. 2018, 410, 3597–3606. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Zhang, Y.Y.; He, M.D.; Li, C.Y.; Zhou, C.X.; Hong, P.Z.; Qian, Z. Antioxidant Peptide Purified from Enzymatic Hydrolysates of Isochrysis Zhanjiangensis and Its Protective Effect against Ethanol Induced Oxidative Stress of HepG2 Cells. Biotechnol. Bioprocess Eng. 2019, 317, 308–317. [Google Scholar] [CrossRef]
- Tamang, J.P.; Shin, D.H.; Jung, S.J.; Chae, S.W. Functional Properties of Microorganisms in Fermented Foods. Front. Microbiol. 2016, 7, 578. [Google Scholar] [CrossRef]
- Daliri, E.B.M.; Tyagi, A.; Ofosu, F.K.; Chelliah, R.; Kim, J.H.; Kim, J.R.; Yoo, D.; Oh, D.H. A Discovery-Based Metabolomic Approach Using UHPLC Q-TOF MS/MS Unveils a Plethora of Prospective Antihypertensive Compounds in Korean Fermented Soybeans. LWT 2021, 137, 110399. [Google Scholar] [CrossRef]
- Incir, S.; Bolayirli, I.M.; Inan, O.; Ayd, M.S.; Bilgin, I.A.; Sayan, I. The Effects of Genistein Supplementation on Fructose Induced Insulin Resistance, Oxidative Stress and Inflammation. Life Sci. 2016, 158, 57–62. [Google Scholar] [CrossRef]
- Basu, P.; Maier, C. Phytoestrogens and Breast Cancer: In Vitro Anticancer Activities of Iso Fl Avones, Lignans, Coumestans, Stilbenes and Their Analogs and Derivatives. Biomed. Pharmacother. 2018, 107, 1648–1666. [Google Scholar] [CrossRef]
- Zujko, M.E.; Witkowska, A.M. Antioxidant Potential and Polyphenol Content of Selected Food.Pdf. Int. J. Food Prop. 2008, 14, 300–308. [Google Scholar] [CrossRef]
- Nachvak, S.M.; Moradi, S.; Anjom-Shoae, J.; Rahmani, J.; Nasiri, M.; Maleki, V.; Sadeghi, O. Soy, Soy Isoflavones, and Protein Intake in Relation to Mortality from All Causes, Cancers, and Cardiovascular Diseases: A Systematic Review and Dose-Response Meta-Analysis of Prospective Cohort Studies. J. Acad. Nutr. Diet. 2019, 119, 1483–1500.e17. [Google Scholar] [CrossRef]
- Simões, M.J.; Sasso, G.R.S.; Carbonel, A.A.F. Soy Isoflavones Protect against Oxidative Stress and Diminish Apoptosis in Ovary of Middle-Aged Female Rats. Gynecol. Endocrinol. 2019, 35, 586–590. [Google Scholar] [CrossRef]
- Jaskulski, S.; Jung, A.Y.; Rudolph, A.; Johnson, T.; Thöne, K.; Herpel, E.; Sinn, P.; Chang-Claude, J. Genistein and Enterolactone in Relation to Ki-67 Expression and HER2 Status in Postmenopausal Breast Cancer Patients. Mol. Nutr. Food Res. 2017, 61, 1700449. [Google Scholar] [CrossRef]
- Abernathy, L.M.; Ms, M.D.F.; Bs, S.E.R.; David, J.M.; Bs, C.K.Y.; Rakowski, J.; Lonardo, F.; Joiner, M.C.; Hillman, G.G. Soy Isoflavones Promote Radioprotection of Normal Lung Tissue by Inhibition of Radiation-Induced Activation of. J. Thorac. Oncol. 2015, 10, 1703–1712. [Google Scholar] [CrossRef]
- Setchell, K.; Brown, N.M.; Zhao, X.; Lindley, S.L.; Heubi, J.E.; King, E.C.; Messina, M.J. Soy Isoflavone Phase II Metabolism Differs between Rodents and Humans: Implications for the Effect on Breast Cancer Risk. Am. J. Clin. Nutr. 2011, 94, 1284–1294. [Google Scholar] [CrossRef] [PubMed]
- Taku, K.; Melby, M.K.; Nishi, N.; Omori, T.; Kurzer, M.S. Soy Isoflavones for Osteoporosis An Evidence-Based Approach. Maturitas 2011, 70, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Jia, F.; Peng, S.; Green, J.; Koh, L.; Chen, X. Soybean Supply Chain Management and Sustainability: A Systematic Literature Review. J. Clean. Prod. 2020, 255, 120254. [Google Scholar] [CrossRef]
- Reddy, K.N.; Duke, S.O. Soybean Mineral Composition and Glyphosate Use. In Processing and Impact on Active Components in Food; Elsevier Inc.: Amsterdam, The Netherlands, 2015; ISBN 9780124047099. [Google Scholar]
- Medic, J.; Atkinson, C.; Hurburgh, C.R. Current Knowledge in Soybean Composition. JAOCS J. Am. Oil Chem. Soc. 2014, 91, 363–384. [Google Scholar] [CrossRef]
- De Lima, D.P.; Fioriolli, J.C.; Padula, A.D.; Pumi, G. The Impact of Chinese Imports of Soybean on Port Infrastructure in Brazil: A Study Based on the Concept of the “Bullwhip Effect”. J. Commod. Mark. 2018, 9, 55–76. [Google Scholar] [CrossRef]
- Pessoa, F.L.P.; Villardi, H.; Calixto, E.E.S.; Vieira, E.D.; Souza, A.L.B.; Machado, B.A.S. Integrated Soybean Biorefinery. In Biorefinery Concepts, Energy and Products; IntechOpen: London, UK, 2019. [Google Scholar]
- USDA United States Department of Agriculture. Available online: https://www.fas.usda.gov/data/world-agricultural-production (accessed on 2 February 2022).
- Conab. Acompanhamento Da Safra Brasileira. 2022; 8. Available online: https://www.conab.gov.br/info-agro/safras (accessed on 2 February 2022).
- USDA United States Department of Agriculture. Available online: https://usdabrazil.org.br/ (accessed on 2 February 2022).
- Wu, F.; Geng, Y.; Zhang, Y.; Ji, C.; Chen, Y.; Sun, L.; Xie, W.; Ali, T.; Fujita, T. Assessing Sustainability of Soybean Supply in China: Evidence from Provincial Production and Trade Data. J. Clean. Prod. 2020, 244, 119006. [Google Scholar] [CrossRef]
- Garrett, R.D.; Rausch, L.L. Green for Gold: Social and Ecological Tradeoffs Influencing the Sustainability of the Brazilian Soy Industry. J. Peasant Stud. 2016, 43, 461–493. [Google Scholar] [CrossRef]
- Bourdichon, F.; Casaregola, S.; Farrokh, C.; Frisvad, J.C.; Gerds, M.L.; Hammes, W.P.; Harnett, J.; Huys, G.; Laulund, S.; Ouwehand, A.; et al. Food Fermentations: Microorganisms with Technological Beneficial Use. Int. J. Food Microbiol. 2012, 154, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Guilpart, N.; Iizumi, T.; Makowski, D. Data-Driven Yield Projections Suggest Large Opportunities to Improve Europe’s Soybean Self-Sufficiency under Climate Change. Nat. Food 2020, 3, 255–265. [Google Scholar] [CrossRef]
- Assefa, Y.; Purcell, L.C.; Salmeron, M.; Naeve, S.; Casteel, S.N.; Kovács, P.; Archontoulis, S.; Licht, M.; Below, F.; Kandel, H.; et al. Assessing Variation in US Soybean Seed Composition (Protein and Oil). Front. Plant Sci. 2019, 10, 298. [Google Scholar] [CrossRef] [PubMed]
- Perkins, E.G. Composition of Soybeans and Soybean Products. In Practical Handbook of Soybean Processing and Utilization; AOCS Press: Urbana, IL, USA, 1995; Volume I. [Google Scholar]
- Mani, V.; Ming, L.C. Tempeh and Other Fermented Soybean Products Rich in Isoflavones. In Fermented Foods in Health and Disease Prevention; Elsevier Inc.: Amsterdam, The Netherlands, 2017; ISBN 9780128023099. [Google Scholar]
- Giordano, E.; Dávalos, A.; Crespo, M.C.; Tomé-Carneiro, J.; Gómez-Coronado, D.; Visioli, F. Soy Isoflavones in Nutritionally Relevant Amounts Have Varied Nutrigenomic Effects on Adipose Tissue. Molecules 2015, 20, 2310–2322. [Google Scholar] [CrossRef]
- Sanjukta, S.; Rai, A.K. Production of Bioactive Peptides during Soybean Fermentation and Their Potential Health Bene. Trends Food Sci. Technol. 2016, 50, 1–10. [Google Scholar] [CrossRef]
- Mah, J. Fermented Soybean Foods: Significance of Biogenic Amines. Austin J. Nutr. Food Sci. 2015, 3, 1058. [Google Scholar]
- Wu, A.; David, W.; George, E.; Wu, Z.; Schenk-Hamlin, D.; Zhan, W.; Ragsdale, D.W. The Soybean Aphid in China: A Historical Review. Entomol. Soc. Am. 2004, 97, 209–218. [Google Scholar] [CrossRef]
- Park, Y.K.; Lee, J.H.; Mah, J. Occurrence and Reduction of Biogenic Amines in Traditional Asian Fermented Soybean Foods: A Review. Food Chem. 2019, 278, 1–9. [Google Scholar] [CrossRef]
- Rai, A.K.; Kumaraswamy, J. Health Benefits of Functional Proteins in Fermented Foods. Health Benefits Fermented Foods Beverages 2015, 4, 455–474. [Google Scholar] [CrossRef]
- Difo, H.V.; Onyike, E.; Ameh, D.A.; Ndidi, U.S.; Njoku, G.C. Chemical Changes during Open and Controlled Fermentation of Cowpea (Vigna unguiculata) Flour. Int. J. Food Nutr. Saf. 2014, 5, 1–10. [Google Scholar]
- Steinkraus, K.H.; Hwa, Y.B.; Van Buren, J.P.; Provvidenti, M.I.; Hand, D.B. Studies on Tempeh. An Indonesian Fermented Soybean Food. Food Res. 1959, 25, 777–788. [Google Scholar] [CrossRef]
- Jung, J.Y.; Lee, S.H.; Jeon, C.O. Microbial Community Dynamics during Fermentation of Doenjang-Meju, Traditional Korean Fermented Soybean. Int. J. Food Microbiol. 2014, 185, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, M. Fermented Tofu, Tofuyo. In Soybean-Biochemistry, Chemistry and Physiology; IntechOpen: London, UK, 2011. [Google Scholar]
- Cao, X.-H.; Liao, Z.-Y.; Wang, C.-L.; Yang, W.-Y.; Lu, M.-F. Evaluation of a Lipopeptide Biosurfactant from Bacillus Natto TK-1 as a Potential Source of Anti-Adhesive, Antimicrobial and Antitumor Activities. Braz. J. Microbiol. 2009, 40, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Thakur, K.; Feng, J.; Cai, J.; Zhang, J.; Hu, F. B-Vitamin Enriched Fermented Soymilk: A Novel Strategy for Soy-Based Functional Foods Development. Trends Food Sci. Technol. 2020, 105, 43–55. [Google Scholar] [CrossRef]
- Dai, S.; Pan, M.; El-nezami, H.S.; Wan, J.M.F.; Wang, M.F.; Habimana, O.; Lee, J.C.Y.; Louie, J.C.Y.; Shah, N.P. Effects of Lactic Acid Bacteria-Fermented Soymilk on Isoflavone Metabolites and Short-Chain Fatty Acids Excretion and Their Modulating Effects on Gut Microbiota. J. Food Sci. 2019, 84, 1854–1863. [Google Scholar] [CrossRef]
- Cao, Z.; Green-Johnson, J.M.; Buckley, N.D.; Lin, Q. Bioactivity of Soy-Based Fermented Foods: A Review. Biotechnol. Adv. 2019, 37, 223–238. [Google Scholar] [CrossRef]
- Handa, C.L.; de Lima, F.S.; Guelfi, M.F.G.; da Silva Fernandes, M.; Georgetti, S.R.; Ida, E.I. Parameters of the Fermentation of Soybean Flour by Monascus purpureus or Aspergillus oryzae on the Production of Bioactive Compounds and Antioxidant Activity. Food Chem. 2019, 271, 274–283. [Google Scholar] [CrossRef]
- Zhang, B.; Yang, Z.; Huang, W. Isoflavone Aglycones Enrichment in Soybean Sourdough Bread Fermented by Lactic Acid Bacteria Strains Isolated from Traditional Qu Starters: Effects on in Vitro Gastrointestinal Digestion, Nutritional, and Baking Properties. Cereal Chem. 2019, 96, 129–141. [Google Scholar] [CrossRef]
- Xiao, Y.; Wang, L.; Rui, X.; Li, W.; Chen, X.; Jiang, M.; Dong, M. Enhancement of the Antioxidant Capacity of Soy Whey by Fermentation with Lactobacillus plantarum B1-6. J. Funct. Foods 2015, 12, 33–44. [Google Scholar] [CrossRef]
- Chen, M.; Rao, Y.; Zheng, Y.; Wei, S.; Li, Y.; Guo, T.; Yin, P. Association between Soy Isoflavone Intake and Breast Cancer Risk for Pre- and Post-Menopausal Women: A Meta-Analysis of Epidemiological Studies. PLoS ONE 2014, 9, e89288. [Google Scholar] [CrossRef]
- do Prado, F.G.; Miyaoka, M.F.; de Melo Pereira, G.V.; Pagnoncelli, M.G.B.; Prado, M.R.M.; Bonatto, S.J.R.; Spier, M.R.; Soccol, C.R. Fungal-Mediated Biotransformation of Soybean Supplemented with Different Cereal Grains into a Functional Compound with Antioxidant, Anti- Inflammatory and Antitumoral Activities. Biointerface Res. Appl. Chem. 2021, 11, 8018–8033. [Google Scholar]
- Mai, C.; Technology, A.; Rai, C. Analysis and Characterisation of Amino Acid Contents of Thua Nao, a Traditionally Fermented Soybean Food of Northern Thailand. Int. Food Res. J. 2011, 599, 595–599. [Google Scholar]
- Sanjukta, S.; Rai, A.K.; Muhammed, A.; Jeyaram, K.; Talukdar, N.C. Enhancement of Antioxidant Properties of Two Soybean Varieties of Sikkim Himalayan Region by Proteolytic Bacillus subtilis Fermentation. J. Funct. Foods 2015, 14, 650–658. [Google Scholar] [CrossRef]
- Man, K.; Hwan, J.; Dae, H.; Yong, B.; Kim, H.; Taek, W. Changes of Phytochemical Constituents (Isoflavones, Flavanols, and Phenolic Acids) during Cheonggukjang Soybeans Fermentation Using Potential Probiotics Bacillus subtilis CS90. J. Food Compos. Anal. 2011, 24, 402–410. [Google Scholar] [CrossRef]
- Manabe, H. D-Amino Acids in Viscous Parts of Natto. J. Integr. Study Diet. Habits 2011, 14, 200–206. [Google Scholar] [CrossRef]
- Sato, K.; Miyasaka, S.; Tsuji, A.; Tachi, H. Isolation and Characterization of Peptides with Dipeptidyl Peptidase IV (DPPIV) Inhibitory Activity from Natto Using DPPIV from Aspergillus oryzae. Food Chem. 2018, 261, 51–56. [Google Scholar] [CrossRef]
- Inomata, N.; Miyakawa, M.; Aihara, M. Surfing as a Risk Factor for Sensitization to Poly(γ-Glutamic Acid) in Fermented Soybeans, Natto, Allergy. Allergol. Int. 2018, 67, 341–346. [Google Scholar] [CrossRef]
- Wang, S.; Tamura, T.; Kyouno, N.; Liu, X.; Zhang, H.; Akiyama, Y.; Yu Chen, J. Effect of the Chemical Composition of Miso (Japanese Fermented Soybean Paste) Upon the Sensory Evaluation. Anal. Lett. 2019, 52, 1813–1827. [Google Scholar] [CrossRef]
- Inoue, Y.; Kato, S.; Saikusa, M.; Suzuki, C.; Otsubo, Y.; Tanaka, Y.; Watanabe, H.; Hayase, F. Analysis of the Cooked Aroma and Odorants That Contribute to Umami Aftertaste of Soy Miso (Japanese Soybean Paste). Food Chem. 2016, 213, 521–528. [Google Scholar] [CrossRef]
- Ogasawara, M.; Yamada, Y.; Egi, M. Taste Enhancer from the Long-Term Ripening of Miso (Soybean Paste). Food Chem. 2006, 99, 736–741. [Google Scholar] [CrossRef]
- Ratnaningrum, D.; Budiwati, T.A.; Darsini, T.; Mawarda, P.C. The Production of Corn Kernel Miso Based on Rice-Koji Fermented by Aspergillus oryzae and Rhizopus oligosporus. J. Trop. Biodivers. Biotechnol. 2018, 3, 8. [Google Scholar] [CrossRef]
- Nuryani, H.; Kinjo, A.; Yasuda, S.; Kuba-Miyara, M. Taste and Chemical Characteristics of Low Molecular Weight Fractions from Tofuyo–Japanese Fermented Soybean Curd. Food Chem. 2018, 252, 265–270. [Google Scholar] [CrossRef]
- Kuba, M.; Tanaka, K.; Tawata, S.; Takeda, Y.; Yasuda, M. Angiotensin I-Converting Enzyme Inhibitory Peptides Isolated from Tofuyo Fermented Soybean Food. Biosci. Biotechnol. Biochem. 2003, 67, 1278–1283. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, M.; Tachibana, S.; Kuba-Miyara, M. Biochemical Aspects of Red Koji and Tofuyo Prepared Using Monascus Fungi. Appl. Microbiol. Biotechnol. 2012, 94, 49–60. [Google Scholar] [CrossRef]
- Chen, T.; Wang, M.; Li, S.; Wu, Q.; Wei, H. Molecular Identification of Microbial Community in Surface and Undersurface Douchi During Postfermentation. J. Food Sci. 2014, 79, 653–658. [Google Scholar] [CrossRef]
- He, G.; Huang, J.; Liang, R.; Wu, C.; Zhou, R. Comparing the Differences of Characteristic FLavour Betweennatural Maturation and Starter Culture for Mucor-Type Douchi. Int. J. Food Sci. Technol. 2016, 51, 1252–1259. [Google Scholar] [CrossRef]
- Wang, D.; Wang, L.-J.; Zhu, F.-X.; Zhu, J.-Y.; Chen, X.D.; Zou, L.; Saito, M.; Li, L. te In Vitro and in Vivo Studies on the Antioxidant Activities of the Aqueous Extracts of Douchi (a Traditional Chinese Salt-Fermented Soybean Food). Food Chem. 2008, 107, 1421–1428. [Google Scholar] [CrossRef]
- Tan, Y.; Zhang, R.; Chen, G.; Wang, S.; Li, C.; Xu, Y.; Kan, J. Effect of Different Starter Cultures on the Control of Biogenic Amines and Quality Change of Douchi by Rapid Fermentation. LWT-Food Sci. Technol. 2019, 109, 395–405. [Google Scholar] [CrossRef]
- Huang, X.; Yu, S.; Han, B.; Chen, J. Bacterial Community Succession and Metabolite Changes during Sufu Fermentation. LWT-Food Sci. Technol. 2018, 97, 537–545. [Google Scholar] [CrossRef]
- Yang, B.; Tan, Y.; Kan, J. Determination and Mitigation of Chemical Risks in Sufu by NaCl and Ethanol Addition during Fermentation. J. Food Compos. Anal. 2021, 98, 103820. [Google Scholar] [CrossRef]
- Xu, L.; Du, B.; Xu, B. A Systematic, Comparative Study on the Beneficial Health Components and Antioxidant Activities of Commercially Fermented Soy Products Marketed in China. Food Chem. 2015, 174, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Li, D.; Li, Z.; Kang, L.; Jiang, Y.; Liu, X.; Chi, Y.; Li, Y.; Wang, J. Effects of Bacillus Fermentation on the Protein Microstructure and Anti-Nutritional Factors of Soybean Meal. Lett. Appl. Microbiol. 2017, 65, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Kumari, R.; Sanjukta, S.; Sahoo, D. Production of Bioactive Protein Hydrolysate Using the Yeasts Isolated from Soft Chhurpi. Bioresour. Technol. 2016, 219, 239–245. [Google Scholar] [CrossRef]
- Florindo, R.N.; Souza, V.P.; Mutti, H.S.; Camilo, C.; Regina, L.; Marana, S.R.; Polikarpov, I.; Nascimento, A.S. Structural Insights into β -Glucosidase Transglycosylation Based on Biochemical, Structural and Computational Analysis of Two GH1 Enzymes from Trichoderma Harzianum. New Biotechnol. 2018, 40, 218–227. [Google Scholar] [CrossRef]
- Guadamuro, L.; Flórez, A.B.; Alegría, Á.; Vázquez, L.; Mayo, B. Characterization of Four β -Glucosidases Acting on Iso Fl Avone-Glycosides from Bi Fi Dobacterium Pseudocatenulatum IPLA 36007. Food Res. Int. 2017, 100, 522–528. [Google Scholar] [CrossRef]
- Raimondi, S.; Roncaglia, L.; De Lucia, M.; Amaretti, A.; Leonardi, A.; Pagnoni, U.M.; Rossi, M. Bioconversion of Soy Isoflavones Daidzin and Daidzein by Bifidobacterium Strains. Appl. Microb. Cell Physiol. 2009, 81, 943–950. [Google Scholar] [CrossRef]
- Kuligowski, M.; Pawłowska, K.; Jasińska-Kuligowska, I. Isoflavone Composition, Polyphenols Content and Antioxidative Activity of Soybean Seeds during Tempeh Fermentation. CyTA-J. Food 2017, 15, 27–33. [Google Scholar] [CrossRef]
- Kamao, M.; Suhara, Y.; Tsugawa, N.; Uwano, M.; Yamaguchi, N.; Uenishi, K.; Ishida, K.; Sasaki, S.; Okano, T. Vitamin K Content of Foods and Dietary Vitamin K Intake in Japanese Young Women. J. Nutr. Sci. Vitaminol. 2007, 53, 464–470. [Google Scholar] [CrossRef]
- Mo, H.; Kariluoto, S.; Piironen, V.; Zhu, Y.; Sanders, M.G.; Vincken, J.; Wolkers-Rooijackers, J.; Nout, M.J.R. Effect of Soybean Processing on Content and Bioaccessibility of Folate, Vitamin B12 and Isoflavones in Tofu and Tempe. Food Chem. 2013, 141, 2418–2425. [Google Scholar] [CrossRef]
- Xu, L.; Cai, W.X.; Xu, B.J. A Systematic Assesmet on Vitamins (B2, B12) an GABA Profiles in Fermented Soy Products Marketed in China. J. Food Process. Preserv. 2017, 41, e13126. [Google Scholar] [CrossRef]
- Young, D.; Ph, K.; Mee, S.; Hong, M.S.; Sung, I.; Ahn, M.S.; Jung, M.; Kim, M.S.; Jeong, H.; Yang, M.S.; et al. Isoflavonoids and Peptides from Meju, Long-Term Fermented Soybeans, Increase Insulin Sensitivity and Exert Insulinotropic Effects in Vitro. Nutrition 2011, 27, 244–252. [Google Scholar] [CrossRef]
- Chan, W.; Liu, S. Biovalorisation of Okara (Soybean Residue) for Food and Nutrition. Trends Food Sci. Technol. 2016, 52, 139–147. [Google Scholar] [CrossRef]
- Meinlschmidt, P.; Sussmann, D.; Schweiggert-Weisz, U.; Eisner, P. Enzymatic Treatment of Soy Protein Isolates: Effects on the Potential Allergenicity, Technofunctionality, and Sensory Properties. Food Sci. Nutr. 2015, 4, 11–23. [Google Scholar] [CrossRef]
- Rai, A.K.; Sanjukta, S.; Jeyaram, K. Production of Angiotensin I Converting Enzyme Inhibitory (ACE-I) Peptides during Milk Fermentation and Their Role in Reducing Hypertension. Crit. Rev. Food Sci. Nutr. 2017, 57, 2789–2800. [Google Scholar] [CrossRef] [PubMed]
- He, R.; Ma, H.; Zhao, W.; Qu, W.; Zhao, J.; Luo, L.; Zhu, W. Modeling the QSAR of ACE-Inhibitory Peptides with ANN and Its Applied Illustration. Int. J. Pept. 2012, 2012, 620609. [Google Scholar] [CrossRef] [PubMed]
- Dastmalchi, N.; Baradaran, B.; Latifi-Navid, S.; Safaralizadeh, R. Antioxidants with Two Faces toward Cancer. Life Sci. 2020, 258, 118–186. [Google Scholar] [CrossRef]
- Shukla, S.; Park, J.; Kim, D.H.; Hong, S.Y.; Lee, J.S.; Kim, M. Total Phenolic Content, Antioxidant, Tyrosinase and α-Glucosidase Inhibitory Activities of Water Soluble Extracts of Noble Starter Culture Doenjang, a Korean Fermented Soybean Sauce Variety. Food Control 2016, 59, 854–861. [Google Scholar] [CrossRef]
- Wang, D.; Zhao, H.; Zhu, H.; Wen, L.; Yu, J.; Li, L.; Chen, L.; Geng, Y. A Novel Method for Highly e Ffi Cient Biotransformation and Separation of Iso Fl Avone Aglycones from Soybean with High-Speed Counter-Current Chromatography. Ind. Crops Prod. 2019, 129, 224–230. [Google Scholar] [CrossRef]
- Kimura, T. East Meets West: Ethnic Differences in Prostate Cancer Epidemiology between East Asians and Caucasians. Chin. J. Cancer 2012, 31, 421–429. [Google Scholar] [CrossRef]
- Lee, H.P.; Lee, J.; Gourley, L.; Duffy, S.W.; Day, N.E.; Estève, J. Dietary Effects on Breast-Cancer Risk in Singapore. Lancet 1991, 337, 1197–1200. [Google Scholar] [CrossRef]
- Khosravi, A.; Razavi, S.H. Therapeutic Effects of Polyphenols in Fermented Soybean and Black Soybean Products. J. Funct. Foods 2021, 81, 104467. [Google Scholar] [CrossRef]
- Singh, P.B.; Yadav, D.; Vij, S. Soybean Bioactive Molecules: Current Trend and Future Prospective. In Bioactive Molecules in Food; Springer: Cham, Switzerland, 2019; pp. 267–294. [Google Scholar]
- Calvello, R.; Aresta, A.; Trapani, A.; Zambonin, C.; Cianciulli, A.; Salvatore, R.; Lisa, M.; Corbo, F.; Franchini, C.; Antonietta, M. Bovine and Soybean Milk Bioactive Compounds: Effects on Inflammatory Response of Human Intestinal Caco-2 Cells. Food Chem. 2016, 210, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.; Jeong, D. Korean Traditional Fermented Soybean Products: Jang. J. Ethn. Foods 2015, 2, 2–7. [Google Scholar] [CrossRef]
- Sundaram, M.K.; Ansari, M.Z.; Mutery, A.A.; Maryam, A.; Nasab, R.; Rai, S.; Rais, N.; Hussain, A. Genistein Induces Alterations of Epigenetic Modulatory Signatures in Human Cervical Cancer Cells. Anti-Cancer Agents Med. Chem. 2018, 18, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Bilir, B.; Sharma, N.V.; Lee, J.; Hammarstrom, B.; Svindland, A.; Kucuk, O.; Moreno, C.S. Effects of Genistein Supplementation on Genome-Wide DNA Methylation and Gene Expression in Patients with Localized Prostate Cancer. Int. J. Oncol. 2017, 51, 223–234. [Google Scholar] [CrossRef]
- Hsiao, Y.H.; Ho, C.T.; Pan, M.H. Bioavailability and Health Benefits of Major Isoflavone Aglycones and Their Metabolites. J. Funct. Foods 2020, 74, 104164. [Google Scholar] [CrossRef]
- Wang, S.; Wang, Y.; Pan, M.H.; Ho, C.T. Anti-Obesity Molecular Mechanism of Soy Isoflavones: Weaving the Way to New Therapeutic Routes. Food Funct. 2017, 8, 3831–3846. [Google Scholar] [CrossRef]
- Rosas-Villegas, A.; Sánchez-Tapia, M.; Avila-Nava, A.; Ramírez, V.; Tovar, A.R.; Torres, N. Differential Effect of Sucrose and Fructose in Combination with a High Fat Diet on Intestinal Microbiota and Kidney Oxidative Stress. Nutrients 2017, 9, 393. [Google Scholar] [CrossRef]
- Gaman, L.; Stoian, I.; Atanasiu, V. Can Ageing Be Slowed ? Hormetic and Redox Perspectives. J. Med. Life 2011, 4, 346–351. [Google Scholar]
- Sharma, A.; Baluja, Z. Therapeutic Effects of Glycine Max (Soybean): A Summary. Int. J. Res. Pharm. Biosci. 2015, 2, 22–27. [Google Scholar]
- Huang, C.; Pang, D.; Luo, Q.; Chen, X.; Gao, Q.; Shi, L.; Liu, W.; Zou, Y.; Li, L.; Chen, Z. Soy Isoflavones Regulate Lipid Metabolism through an AKT/MTORC1 Pathway in Diet-Induced Obesity (DIO) Male Rats. Molecules 2016, 21, 586. [Google Scholar] [CrossRef]
- Squadrito, F.; Marini, H.; Bitto, A.; Altavilla, D.; Polito, F.; Adamo, E.B.; D’Anna, R.; Arcoraci, V.; Burnett, B.P.; Minutoli, L.; et al. Genistein in the Metabolic Syndrome: Results of a Randomized Clinical Trial. J. Clin. Endocrinol. Metab. 2013, 98, 3366–3374. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Ling, W. The Update of Anthocyanins on Obesity and Type 2 Diabetes: Experimental Evidence and Clinical Perspectives. Rev. Endocr. Metab. Disord. 2015, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Vieira, C.P.; Cabral, C.C.; da Costa Lima, B.R.C.; Paschoalin, V.M.F.; Leandro, K.C.; Conte-Junior, C.A. Lactococcus Lactis Ssp. Cremoris MRS47, a Potential Probiotic Strain Isolated from Kefir Grains, Increases Cis-9, Trans-11-CLA and PUFA Contents in Fermented Milk. J. Funct. Foods 2017, 31, 172–178. [Google Scholar] [CrossRef]
- Rains, J.L.; Jain, S.K. Oxidative Stress, Insulin Signaling, and Diabetes. Free Radic. Biol. Med. 2011, 50, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; Shirihai, O.S.; Gentil, B.J.; Burelle, Y. Mitochondrial Morphology Transitions and Functions: Implications for Retrograde Signaling? Am. J. Physiol. Integr. Comp. Physiol. 2013, 304, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Behloul, N.; Wu, G. Genistein: A Promising Therapeutic Agent for Obesity and Diabetes Treatment. Eur. J. Pharmacol. 2013, 698, 31–38. [Google Scholar] [CrossRef]
- Lei, H.; Dong, H.; Xu, L.; Wang, J. Genistein Reverses Free Fatty Acid-Induced Insulin Resistance in HepG2 Hepatocytes through Targeting JNK. J. Huazhong Univ. Sci. Technol. Med. Sci. 2011, 31, 185–189. [Google Scholar] [CrossRef]
- Choksomngam, Y.; Pattanakuhar, S.; Chattipakorn, N.; Chattipakorn, S.C. The Metabolic Role of Spermidine in Obesity: Evidence from Cells to Community. Obes. Res. Clin. Pract. 2021, 15, 315–326. [Google Scholar] [CrossRef]
- Eisenberg, T.; Knauer, H.; Schauer, A.; Büttner, S.; Ruckenstuhl, C.; Carmona-Gutierrez, D.; Ring, J.; Schroeder, S.; Magnes, C.; Antonacci, L.; et al. Induction of Autophagy by Spermidine Promotes Longevity. Nat. Cell Biol. 2009, 11, 1305–1314. [Google Scholar] [CrossRef]
- Soda, K.; Kano, Y.; Chiba, F. Food Polyamine and Cardiovascular Disease -An Epidemiological Study-.Pdf. Glob. J. Health Sci. 2012, 4, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Kraus, D.; Yang, Q.; Kong, D.; Banks, A.S.; Zhang, L.; Rodgers, J.T.; Pirinen, E.; Pulinilkunnil, T.C.; Gong, F.; Wang, Y.C.; et al. Nicotinamide N-Methyltransferase Knockdown Protects against Diet-Induced Obesity. Nature 2014, 508, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Lei, C.; Teng, H.; Xie, Z.; Cao, H.; Cheang, W.S.; Skalicka-Woniak, K.; Georgiev, M.I.; Xiao, J. Modifications of Dietary Flavonoids towards Improved Bioactivity: An Update on Structure–Activity Relationship. Crit. Rev. Food Sci. Nutr. 2018, 58, 513–527. [Google Scholar] [CrossRef]
- Yusof, H.M.; Ali, N.M.; Yeap, S.K.; Ho, W.Y.; Beh, B.K.; Koh, S.P. Anti-Inflammatory, Analgesic and Acute Toxicity Effects of Fermented Soybean. BMC Complement. Altern. Med. 2019, 2, 373. [Google Scholar] [CrossRef] [PubMed]
- Szliszka, E.; Skaba, D.; Czuba, Z.P.; Krol, W. Inhibition of Inflammatory Mediators by Neobavaisoflavone in Activated RAW264.7 Macrophages. Molecules 2011, 16, 3701–3712. [Google Scholar] [CrossRef]
- Liao, C.; Huang, H.; Sheen, L.; Chou, C. Anti-Inflammatory Activity of Soymilk and Fermented Soymilk Prepared with Lactic Acid Bacterium and Bifidobacterium Anti-Inflammatory Activity of Soymilk and Fermented Soymilk Prepared with Lactic Acid Bacterium and Bifidobacterium. J. Food Drug Anal. 2010, 18, 5. [Google Scholar] [CrossRef]
- Chen, L.; Teng, H.; Jia, Z.; Battino, M.; Miron, A.; Yu, Z. Intracellular Signaling Pathways of Inflammation Modulated by Dietary Flavonoids: The Most Recent Evidence. Crit. Rev. Food Sci. Nutr. 2018, 58, 2908–2924. [Google Scholar] [CrossRef]
- Yang, X.; Nakamoto, M.; Shuto, E.; Hata, A.; Aki, N.; Shikama, Y.; Bando, Y.; Ichihara, T.; Minamigawa, T.; Kuwamura, Y.; et al. Associations between Intake of Dietary Fermented Soy Food and Concentrations of Inflammatory Markers: A cross-sectional study in Japanese workers. J. Med. Investig. 2018, 65, 74–80. [Google Scholar] [CrossRef]
- Nagata, C.; Wada, K.; Tamura, T.; Konishi, K.; Goto, Y.; Koda, S.; Kawachi, T.; Tsuji, M. Dietary Soy and Natto Intake and Cardiovascular Disease Mortality in Japanese Adults: The Takayama Study 1. Am. J. Clin. Nutr. 2017, 105, 426–431. [Google Scholar] [CrossRef]
- Dabbagh, F.; Negahdaripour, M.; Berenjian, A.; Behfar, A.; Mohammadi, F.; Zamani, M.; Irajie, C.; Ghasemi, Y. Nattokinase: Production and Application. Appl. Microbiol. Biotechnol. 2014, 98, 9199–9206. [Google Scholar] [CrossRef]
- Yatagai, C.; Maruyama, M.; Kawahara, T.; Sumi, H. Nattokinase-Promoted Tissue Plasminogen Activator Release from Human Cells. Pathophysiol. Haemost. Thromb. 2008, 36, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Takabayashi, T.; Imoto, Y.; Sakashita, M.; Kato, Y.; Tokunaga, T.; Yoshida, K.; Narita, N.; Ishizuka, T.; Fujieda, S. Nattokinase, Profibrinolytic Enzyme, Effectively Shrinks the Nasal Polyp Tissue and Decreases Viscosity of Mucus. Allergol. Int. 2017, 66, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Kurosawa, Y.; Nirengi, S.; Homma, T.; Esaki, K.; Ohta, M. A Single-Dose of Oral Nattokinase Potentiates Thrombolysis and Anti-Coagulation Profiles. Sci. Rep. 2015, 5, 11601. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.J.; Seo, J.Y.; Cho, K.M.; Lee, C.K.; Kim, J.H.; Kim, J. Antioxidant and Neuroprotective Effects of Doenjang Prepared with Rhizopus, Pichia, and Bacillus. Prev. Nutr. Food Sci. 2016, 21, 221–226. [Google Scholar] [CrossRef]
- Deng, Y.Y.; Lu, J.; Ling, E.-A.; Kaur, C. Role of Microglia in the Process of Inflammation in the Hypoxic Developing Brain. Front. Biosci. 2011, 3, 884–900. [Google Scholar] [CrossRef]
- Dias, G.P.; Cavegn, N.; Nix, A.; Stangl, D.; Syahrul, M.; Zainuddin, A.; Nardi, A.E.; Gardino, P.F.; Thuret, S. The Role of Dietary Polyphenols on Adult Hippocampal Neurogenesis: Molecular Mechanisms and Behavioural Effects on Depression and Anxiety. Oxid. Med. Cell. Longev. 2012, 2012, 541971. [Google Scholar] [CrossRef]
- Lara, J.; Michael, J.S.; Margaret, R. Aging and Anti-Aging. Maturitas 2016, 93, 1–3. [Google Scholar] [CrossRef]
- De Magalhães, J.P.; Stevens, M.; Thornton, D. The Business of Anti-Aging Science. Trends Biotechonol. 2017, 35, 1062–1073. [Google Scholar] [CrossRef]
- Leonardi, G.R. An Overview about Oxidation in Clinical Practice of Skin Aging. An. Bras. Dermatol. 2017, 92, 367–374. [Google Scholar]
- Reviews, I.N.D. Biology of Healthy Aging and Longevity. Rev. Investig. Clin. 2016, 68, 7–16. [Google Scholar]
- Pan, M.; Lai, C.; Tsai, M.; Wu, J.C.; Ho, C.T. Molecular Mechanisms for Anti-Aging by Natural Dietary Compounds. Mol. Nutr. Food Res. 2012, 56, 88–115. [Google Scholar] [CrossRef] [PubMed]
- Sapbamrer, R.; Visavarungroj, N.; Suttajit, M. Effects of Dietary Traditional Fermented Soybean on Reproductive Hormones, Lipids, and Glucose among Postmenopausal Women in Northern Thailand. Asia Pac. J. Clin. Nutr. 2013, 22, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Das, G.; Paramithiotis, S.; Sivamaruthi, B.S.; Hanny, C.; Suharta, S.; Sanlier, N.; Shin, H.; Kumar, J. Traditional Fermented Foods with Anti-Aging Effect: A Concentric Review. Food Res. Int. 2020, 134, 109269. [Google Scholar] [CrossRef] [PubMed]
- Muñoz Fernández, S.S.; Lima Ribeiro, S.M. Nutrition and Alzheimer Disease. Clin. Geriatr. Med. 2018, 34, 677–697. [Google Scholar] [CrossRef]
- Ahmad, A.; Ramasamy, K.; Murnirah, S.; Bakar, A.; Majeed, A.; Mani, V. Total Isoflavones from Soybean and Tempeh Reversed Scopolamine-Induced Amnesia, Improved Cholinergic Activities and Reduced Neuroinflammation in Brain. Food Chem. Toxicol. 2014, 65, 120–128. [Google Scholar] [CrossRef]
- Overk, C.R.; Felder, C.C.; Tu, Y.; Schober, D.A.; Bales, K.R.; Wuu, J.; Mufson, E.J. Cortical M 1 Receptor Concentration Increases without a Concomitant Change in Function in Alzheimer’s Disease. J. Chem. Neuroanat. 2010, 40, 63–70. [Google Scholar] [CrossRef]
- Chan, Y.; Lee, I.; Wang, M.; Yeh, W.; Liang, B. Tempeh Attenuates Cognitive de Fi Cit, Antioxidant Imbalance, and Amyloid β of Senescence-Accelerated Mice by Modulating Nrf2 Expression via MAPK Pathway. J. Funct. Foods 2018, 50, 112–119. [Google Scholar] [CrossRef]
- Jeong, D.-Y.; Ryu, M.S.; Yang, H.-J.; Park, S.; Gonzalez-Manzano, S. γ-PGA-Rich Chungkookjang, Short-Term Fermented Soybeans: Prevents Memory Impairment by Modulating Brain Insulin Sensitivity, Neuro-Inflammation, and the Gut-Microbiome-Brain Axis. Foods 2021, 10, 221. [Google Scholar] [CrossRef]
- Kim, I.S.; Hwang, C.W.; Yang, W.S.; Kim, C.H. Current Perspectives on the Physiological Activities of Fermented Soybean-Derived Cheonggukjang. Int. J. Mol. Sci. 2021, 22, 5746. [Google Scholar] [CrossRef]
- Yeon, D.; Jeong, S.; Zhang, T.; Wu, X.; Yi, J.; Park, S. Chungkookjang, a Soy Food, Fermented with Bacillus amyloliquefaciens Protects Gerbils against Ishcmeic Stroke Injury, and Post-Stroke Hyperglycemia. Food Res. Int. 2020, 128, 108769. [Google Scholar] [CrossRef]
- Park, S.; Sol, D.; Kang, S.; Young, D. Ischemic Hippocampal Cell Death Induces Glucose Dysregulation by Attenuating Glucose-Stimulated Insulin Secretion Which Is Exacerbated by a High Fat Diet. Life Sci. 2011, 88, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Ibe, S.; Kumada, K.; Yoshida, K.; Otobe, K. Natto (Fermented Soybean) Extract Extends the Adult Lifespan of Caenorhabditis Elegans. Biosci. Biotechnol. Biochem. 2013, 77, 392–394. [Google Scholar] [CrossRef] [PubMed]
- Sagara, T.; Fiechter, G.; Pachner, M.; Mayer, H.K.; Vollmann, J. Soybean Spermidine Concentration: Genetic and Environmental Variation of a Potential ‘Anti-Aging’ Constituent. J. Food Compos. Anal. 2017, 56, 11–17. [Google Scholar] [CrossRef]
- Santacroce, L.; Inchingolo, F.; Topi, S.; Del, R.; Di, M.; Alexandros, I.; Montagnani, M. Potential Beneficial Role of Probiotics on the Outcome of COVID-19 Patients: An Evolving Perspective. Diabetes Metab. Syndr. Clin. Res. Rev. 2021, 15, 295–301. [Google Scholar] [CrossRef]
- OPAS. Histórico Da Pandemia de COVID-19. Available online: https://www.paho.org/pt/covid19/historico-da-pandemia-covid-19 (accessed on 2 August 2021).
- WHO. Coronavirus (COVID-19) Dashboard. World Health Organization. Available online: https://covid19.who.int/ (accessed on 2 August 2021).
- Zeng, F.; Huang, Y.; Guo, Y.; Yin, M.; Chen, X. Association of Inflammatory Markers with the Severity of COVID-19: A Meta-Analysis. Int. J. Infect. Dis. 2020, 96, 467–474. [Google Scholar] [CrossRef]
- Mahmudpour, M.; Roozbeh, J.; Keshavarz, M.; Farrokhi, S.; Nabipour, I. COVID-19 Cytokine Storm: The Anger of Inflammation. Cytokine 2020, 133, 155151. [Google Scholar] [CrossRef]
- Gouda, A.S.; Adbelruhman, F.G.; Sabbah, H.; Mégarbane, B. Theoretical Benefits of Yogurt-Derived Bioactive Peptides and Probiotics in COVID-19 Patients—A Narrative Review and Hypotheses. Saudi J. Biol. Sci. 2021, 20, 5897–5905. [Google Scholar] [CrossRef]
- Costela-Ruiz, V.J.; Illescas-montes, R.; Puerta-Puerta, J.M.; Ruiz, C.; Melguizo-Rodríguez, L. SARS-CoV-2 Infection: The Role of Cytokines in COVID-19 Disease. Cytokine Growth Factor Rev. 2020, 54, 62–75. [Google Scholar] [CrossRef]
- Huang, Q.; Wu, X.; Zheng, X.; Luo, S.; Xu, S.; Weng, J. Targeting Inflammation and Cytokine Storm in COVID-19. Pharmacol. Res. J. 2020, 159, 3–5. [Google Scholar] [CrossRef]
- Muchtaridi, M.; Fauzi, M.; Ikram, N.K.K.; Gazzali, A.M.; Wahab, H.A. Natural Flavonoids as Potential for Anti-SARS-CoV-2. Molecules 2020, 25, 3980. [Google Scholar] [CrossRef]
- CDC. Food and Coronavirus Disease 2019. Available online: https://www.cdc.gov/coronavirus/2019-ncov/daily-life-coping/food-and-COVID-19.html (accessed on 3 August 2021).
- Calder, P.C.; Carr, A.C.; Gombart, A.F.; Eggersdorfer, M. Optimal Nutritional Status for a Well-Functioning Immune System Is an Important Factor to Protect against Viral Infections. Nutrients 2020, 12, 1181. [Google Scholar] [CrossRef] [PubMed]
- Mohseni, H.; Amini, S.; Abiri, B.; Kalantar, M. Are History of Dietary Intake and Food Habits of Patients with Clinical Symptoms of COVID 19 Different from Healthy Controls? A Case-Control Study. Clin. Nutr. ESPEN 2021, 42, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Bousquet, J.; Anto, J.M.; Iaccarino, G.; Czarlewski, W.; Haahtela, T. Is Diet Partly Responsible for Differences in COVID-19 Death Rates between and within Countries? Clin. Transl. Allergy 2020, 10, 1–7. [Google Scholar] [CrossRef]
- Adem, S.; Eyupoglu, V.; Sarfraz, I.; Rasul, A.; Ali, M. Identification of Potent COVID-19 Main Protease (Mpro) Inhibitors from Natural Polyphenols: An in Silico Strategy Unveils a Hope against CORONA. Preprints 2020. [Google Scholar] [CrossRef]
- Pendyala, B.; Patras, A. In silico Screening of Food Bioactive Compounds to Predict Potential Inhibitors of COVID-19 Main protease (Mpro) and RNA-dependent RNA polymerase (RdRp). ChemRxiv 2020, 11–44. [Google Scholar] [CrossRef]
- Chourasia, R.; Padhi, S.; Chiring Phukon, L.; Abedin, M.M.; Singh, S.P.; Rai, A.K. A Potential Peptide From Soy Cheese Produced Using Lactobacillus delbrueckii WS4 for Effective Inhibition of SARS-CoV-2 Main Protease and S1 Glycoprotein. Front. Mol. Biosci. 2020, 7, 1–12. [Google Scholar] [CrossRef]
- Kuba, K.; Imai, Y.; Rao, S.; Gao, H.; Guo, F.; Guan, B.; Huan, Y.; Yang, P.; Zhang, Y.; Deng, W.; et al. A Crucial Role of Angiotensin Converting Enzyme 2 (ACE2) in SARS Coronavirus—Induced Lung Injury. Nat. Med. 2005, 11, 875–879. [Google Scholar] [CrossRef]
- Sachie, I.; Keiko, Y.; Kaoru, K.; Shigeko, T.; Tadasu, F.; Kazunori, O. Antihypertensive Effects of Natto, a Traditional Japanese Fermented Food, in Spontaneously Hypertensive Rats. Food Sci. Technol. 2009, 15, 199–202. [Google Scholar]
- Ma, Y.; Cheng, Y.; Yin, L. Effects of Processing and NaCl on Angiotensin I-Converting Enzyme Inhibitory Activity and γ-Aminobutyric Acid Content During Sufu Manufacturing. Food Bioprocess Technol. 2013, 6, 1782–1789. [Google Scholar] [CrossRef]
- Zhang, J.; Tatsumi, E.; Ding, C.; Li, L. Angiotensin I-Converting Enzyme Inhibitory Peptides in Douchi, a Chinese Traditional Fermented Soybean Product. Food Chem. 2006, 98, 551–557. [Google Scholar] [CrossRef]
- Oba, M.; Rongduo, W.; Saito, A.; Okabayashi, T.; Yokota, T.; Yasuoka, J.; Sato, Y.; Nishifuji, K.; Wake, H.; Nibu, Y.; et al. Natto Extract, a Japanese Fermented Soybean Food, Directly Inhibits Viral Infections Including SARS-CoV-2 in Vitro. Biochem. Biophys. Res. Commun. 2021, 570, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Xia, P.; Zhang, L.; Hu, Y.; Xie, Q.; Xiang, H. A Novel Fermented Soybean, Inoculated with Selected Bacillus, Lactobacillus and Hansenula Strains, Showed Strong Antioxidant and Anti-Fatigue Potential Activity. Food Chem. 2020, 333, 127527. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.N.G.; Kusnadib, J.; Hsu, J.-L.; Doerksen, R.J.; Huanga, T.-C. Identification of a Novel Umami Peptide in Tempeh (Indonesian Fermented Soybean) and Its Binding Mechanism to the Umami Receptor T1R. Food Chem. 2020, 333, 127411. [Google Scholar] [CrossRef]
- Surya, R.; Romulo, A. Tempeh Extract Protects HepG2 Cells against Oxidative Stress-Induced Cell Death. J. Phys. Conf. Ser. 2020, 1655, 012110. [Google Scholar] [CrossRef]
- Jeong, D.Y.; Daily, J.W.; Lee, G.H.; Ryu, M.S.; Yang, H.J.; Jeong, S.Y.; Qiu, J.Y.; Zhang, T.; Park, S. Short-Term Fermented Soybeans with Bacillus amyloliquefaciens Potentiated Insulin Secretion Capacity and Improved Gut Microbiome Diversity and Intestinal Integrity to Alleviate Asian Type 2 Diabetic Symptoms. J. Agric. Food Chem. 2020, 68, 13168–13178. [Google Scholar] [CrossRef] [PubMed]
- Katagiri, R.; Sawada, N.; Goto, A.; Yamaji, T.; Iwasaki, M.; Noda, M.; Iso, H.; Tsugane, S.; The Japan Public Health Center-Based Prospective Study Group. Agro-Economic Prospects for Expanding Soybean Production beyond Its Current Northerly Limit in Europe. BMJ 2020, 368, 126415. [Google Scholar]
- Chan, E.W.C.; Wong, S.K.; Kezuka, M.; Oshiro, N.; Chan, H.T. Natto and Miso: An Overview on Their Preparation, Bioactive Components and Health-Promoting Effects. Food Res. 2021, 5, 446–452. [Google Scholar] [CrossRef]
- Ren, N.N.; Chen, H.J.; Li, Y.; Mcgowan, G.W.; Lin, Y.G. A Clinical Study on the Effect of Nattokinase on Carotid Artery Atherosclerosis and Hyperlipidaemia. Zhonghua Yi Xue Za Zhi 2017, 97, 2038–2042. [Google Scholar]
- Hirota, A.; Inaba, M.; Chen, Y.; Abe, N.A.; Taki, S.; Yano, M.; Kwaii, S. Isolation of 8-Hydroxyglycitein and 6-Hydroxydaidzein from Soybean Miso. Biosci. Biotechnol. Biochem. 2004, 68, 1372–1374. [Google Scholar] [CrossRef]
- Park, K.Y.; Jung, K.O.; Rhee, S.H.; Choi, Y.H. Antimutagenic Effects of Doenjang (Korean Fermented Soypaste) and Its Active Compounds. Mutat. Res.-Fundam. Mol. Mech. Mutagen. 2003, 523–524, 43–53. [Google Scholar] [CrossRef]
- Romulo, A.; Surya, R. Tempe: A Traditional Fermented Food of Indonesia and Its Health Benefits. Int. J. Gastron. Food Sci. 2021, 26, 100413. [Google Scholar] [CrossRef]
- Aoki, H.; Chuma, S.; Iba, Y.; Tashiro, H.; Watanabe, N.; Oyama, H. Comparison of Bioactive Components in Tempeh Produced by Three Different Rhizopus Starters and Immunomodulatory Effect of Tempeh on Atopic Dermatitis Mice. Food Sci. Technol. Res. 2020, 26, 665–672. [Google Scholar] [CrossRef]
- Choi, J.H.; Pichiah, P.B.T.; Kim, M.J.; Cha, Y.S. Cheonggukjang, a Soybean Paste Fermented with B. licheniformis-67 Prevents Weight Gain and Improves Glycemic Control in High Fat Diet Induced Obese Mice. J. Clin. Biochem. Nutr. 2016, 59, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.H.; Lee, J.; Do Nam, Y.; Lee, J.S.; Seo, M.J.; Yi, S.H. Characterization of Antimicrobial Lipopeptides Produced by Bacillus Sp. LM7 Isolated from Chungkookjang, a Korean Traditional Fermented Soybean Food. Int. J. Food Microbiol. 2016, 221, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Kojima, A.; Ikehara, S.; Kamiya, K.; Kajita, E.; Sato, Y.; Kouda, K.; Tamaki, J.; Kagamimori, S.; Iki, M. Natto Intake Is Inversely Associated with Osteoporotic Fracture Risk in Postmenopausal Bone Mass. J. Nutr. 2019, 150, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Hitosugi, M.; Hamada, K.; Misaka, K. Effects of Bacillus subtilis Var. Natto Products on Symptoms Caused by Blood Flow Disturbance in Female Patients with Lifestyle Diseases. Int. J. Gen. Med. 2015, 8, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Pagnoncelli, M.G.B.; Fernandes, M.J.; Rodrigues, C.; Soccol, C.R. Nattokinase. In Current Developments in Biotechnology and Bioengineering—Production, Isolation and Purification of Industrial Products; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 509–526. ISBN 9780444636621. [Google Scholar]
- Abe, S.K.; Sawada, N.; Ishihara, J.; Takachi, R.; Mori, N.; Yamaji, T.; Shimazu, T.; Goto, A.; Iwasaki, M.; Inoue, M.; et al. Comparison between the Impact of Fermented and Unfermented Soy Intake on the Risk of Liver Cancer: The JPHC Study. Eur. J. Nutr. 2021, 60, 1389–1401. [Google Scholar] [CrossRef]
- Watanabe, H.; Sasatani, M.; Doi, T.; Masaki, T.; Satoh, K.; Yoshizumi, M. Protective Effects of Japanese Soybean Paste (Miso) on Stroke in Stroke-Prone Spontaneously Hypertensive Rats (SHRSP). Am. J. Hypertens. 2018, 31, 43–47. [Google Scholar] [CrossRef]
- Ito, K. Review of the Health Benefits of Habitual Consumption of Miso Soup: Focus on the Effects on Sympathetic Nerve Activity, Blood Pressure, and Heart Rate. Environ. Health Prev. Med. 2020, 25, 45. [Google Scholar] [CrossRef]
- Jung, K.O.; Park, S.Y.; Park, K.Y. Longer Aging Time Increases the Anticancer and Antimetastatic Properties of Doenjang. Nutrition 2006, 22, 539–545. [Google Scholar] [CrossRef]
- Cha, Y.; Yang, J.; Back, H.; Kim, S.; Kim, M.; Jung, S.; Song, W.O.; Chae, S. Visceral Fat and Body Weight Are Reduced in Overweight Adults by the Supplementation of Doenjang, a Fermented Soybean Paste. Nutr. Res. Pract. 2012, 6, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Chettri, R.; Bhutia, M.O.; Tamang, J.P. Poly-γ-Glutamic Acid (PGA)-Producing Bacillus Species Isolated from Kinema, Indian Fermented Soybean Food. Front. Microbiol. 2016, 7, 971. [Google Scholar] [CrossRef] [PubMed]
- Omizu, Y.; Tsukamoto, C.; Chettri, R.; Tamang, J.P. Determination of Saponin Contents in Raw Soybean and Fermented Soybean Foods of India. J. Sci. Ind. Res. 2011, 70, 533–538. [Google Scholar]
- Yang, H.; Yang, L.; Zhang, J.; Li, H.; Tu, Z.; Wang, X. Exploring Functional Core Bacteria in Fermentation of a Traditional Chinese Food, Aspergillus-Type Douchi. PLoS ONE 2019, 14, e0226965. [Google Scholar] [CrossRef] [PubMed]
- Li-Jun, Y.; Li-Te, L.; Zai-Gui, L.; Tatsumi, E.; Saito, M. Changes in Isoflavone Contents and Composition of Sufu (Fermented Tofu) during Manufacturing. Food Chem. 2004, 87, 587–592. [Google Scholar] [CrossRef]
- WIPO World Intellectual Property Organization. Available online: https://www.wipo.int/portal/en/index.html (accessed on 25 March 2022).
| Initial Stock | Production | Export | Import | Animal Consumption | Domestic Consumption | Final Stock |
|---|---|---|---|---|---|---|
| 100.27 | 362.76 | 312.80 | 158.02 | 360.73 | 161.93 | 98.39 |
| −11% | 8% | 4% | 3% | 4% | 5% | −2% |
| Components | % Grain | Chemical Composition (% Dry Weight) | |||
|---|---|---|---|---|---|
| Proteins | Lipids | Carbohydrates | Others * | ||
| Husks | 8 | 9 | 1 | 86 | 4.3 |
| Hypocotyls | 2 | 41 | 11 | 43 | 4.4 |
| Cotyledons | 90 | 43 | 23 | 29 | 5.0 |
| Total | 100 | 40 | 20 | 35 | 5.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
do Prado, F.G.; Pagnoncelli, M.G.B.; de Melo Pereira, G.V.; Karp, S.G.; Soccol, C.R. Fermented Soy Products and Their Potential Health Benefits: A Review. Microorganisms 2022, 10, 1606. https://doi.org/10.3390/microorganisms10081606
do Prado FG, Pagnoncelli MGB, de Melo Pereira GV, Karp SG, Soccol CR. Fermented Soy Products and Their Potential Health Benefits: A Review. Microorganisms. 2022; 10(8):1606. https://doi.org/10.3390/microorganisms10081606
Chicago/Turabian Styledo Prado, Fernanda Guilherme, Maria Giovana Binder Pagnoncelli, Gilberto Vinícius de Melo Pereira, Susan Grace Karp, and Carlos Ricardo Soccol. 2022. "Fermented Soy Products and Their Potential Health Benefits: A Review" Microorganisms 10, no. 8: 1606. https://doi.org/10.3390/microorganisms10081606
APA Styledo Prado, F. G., Pagnoncelli, M. G. B., de Melo Pereira, G. V., Karp, S. G., & Soccol, C. R. (2022). Fermented Soy Products and Their Potential Health Benefits: A Review. Microorganisms, 10(8), 1606. https://doi.org/10.3390/microorganisms10081606







