Phosphate-Solubilizing Bacteria Isolated from Phosphate Solid Sludge and Their Ability to Solubilize Three Inorganic Phosphate Forms: Calcium, Iron, and Aluminum Phosphates
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation and Screening of Phosphate Solubilizing Bacteria
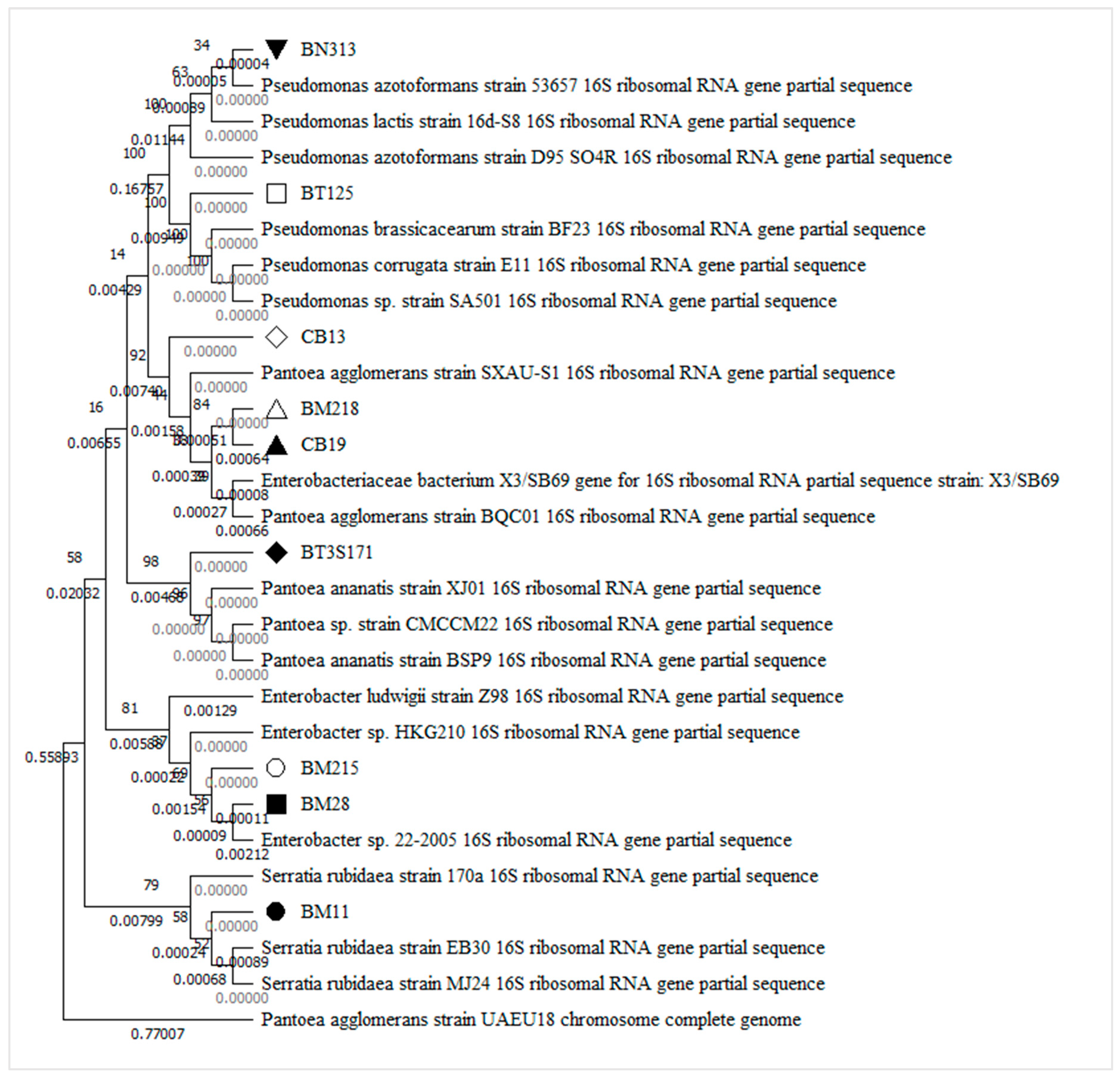
2.2. Molecular Characterization of Selected PSB
2.3. Morphological, Biochemical Characterization
2.4. Plant Growth-Promoting Traits of PSB
2.5. Qualitative Analyses of Potassium (K) Solubilization
2.6. Organic Acid Analysis by GC-MS
2.7. Inoculum Preparation
2.8. Solubilization Test of the Three Forms of Phosphates
2.9. Statistical Analysis
3. Results
3.1. Isolation and Screening of PSB Strains
3.2. Morphological, Biochemical Characterization, and Molecular Characterization of Selected PBS
3.3. Plant Growth-Promoting Traits of PSB
3.4. Qualitative K Solubilization by PSB
3.5. Quantification of Organic Acids Produced by PSB Strains
3.6. Solubilization Test of the Three Forms of Phosphates
4. Discussions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, Y.P.; Rekha, P.D.; Arun, A.B.; Shen, F.T.; Lai, W.-A.; Young, C.C. Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl. Soil Ecol. 2006, 34, 33–41. [Google Scholar] [CrossRef]
- Hegyi, A.; Bao, T.; Nguyen, K.; Posta, K. Metagenomic analysis of bacterial communities in agricultural soils from Vietnam with special attention to phosphate solubilizing bacteria. Microorganisms 2021, 9, 1796. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Cai, K.; Li, M.; Zheng, J.; Han, Y. Plant-growth-promoting potential of PGPE isolated from Dactylis glomerata L. Microorganisms 2022, 10, 731. [Google Scholar] [CrossRef] [PubMed]
- Madrid-Delgado, G.; Orozco-Miranda, M.; Cruz-Osorio, M.; Hernández-Rodríguez, O.A.; Rodriguez-Heredia, R.; Roa-Huerta, M.; Avila-Quezada, G.D. Pathways of phosphorus absorption and early signaling between the mycorrhizal fungi and plants. Phyton 2021, 90, 1321–1338. [Google Scholar] [CrossRef]
- Zou, X.; Binkley, D.; Doxtader, K.G. A new method for estimating gross phosphorus mineralization and immobilization rates in soils. Plant Soil 1992, 147, 243–250. [Google Scholar] [CrossRef]
- Spagnoletti, F.N.; Tobar, N.E.; Di Pardo, A.F.; Chiocchio, V.M.; Lavado, R.S. Dark septate endophytes present different potential to solubilize calcium, iron and aluminum phosphates. Appl. Soil Ecol. 2016, 111, 25–32. [Google Scholar] [CrossRef]
- Holtan, H.; Stuanes, A. Phosphorus in soil, water and sediment: An overview. In Proceedings of the 1988 Phosphorus in Freshwater Ecosystems Symposium, Uppsala, Sweden, 25–28 September 1985; Volume 34, pp. 19–34. [Google Scholar]
- Bashan, Y.; Kamnev, A.A.; De-Bashan, L.E. Tricalcium phosphate is inappropriate as a universal selection factor for isolating and testing phosphate-solubilizing bacteria that enhance plant growth: A proposal for an alternative procedure. Biol. Fertil. Soils 2012, 49, 465–479. [Google Scholar] [CrossRef]
- Rodrı́guez, H.; Fraga, R. Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol. Adv. 1999, 17, 319–339. [Google Scholar] [CrossRef]
- Aliyat, F.Z.; Maldani, M.; El Guilli, M.; Nassiri, L.; Ibijbijen, J. Isolation and characterization of phosphate solubilizing bacteria from phosphate solid sludge of the Moroccan phosphate mines. Open Agric. J. 2020, 14, 16–24. [Google Scholar] [CrossRef]
- Behera, B.C.; Yadav, H.; Singh, S.K.; Mishra, R.R.; Sethi, B.K.; Dutta, S.K.; Thatoi, H.N. Phosphate solubilization and acid phosphatase activity of Serratia sp. isolated from mangrove soil of Mahanadi River delta, Odisha, India. J. Genet. Eng. Biotechnol. 2017, 15, 169–178. [Google Scholar] [CrossRef]
- Chawngthu, L.; Hnamte, R.; Lalfakzuala, R. Isolation and characterization of rhizospheric phosphate solubilizing bacteria from wetland paddy field of Mizoram, India. Geomicrobiol. J. 2020, 37, 366–375. [Google Scholar] [CrossRef]
- Kaur, R.; Kaur, S. Variation in the phosphate solubilizing bacteria from virgin and the agricultural soils of Punjab. Curr. Microbiol. 2020, 77, 2118–2127. [Google Scholar] [CrossRef] [PubMed]
- Manzoor, M.; Abbasi, M.K.; Sultan, T. Isolation of phosphate solubilizing bacteria from maize rhizosphere and their potential for rock phosphate solubilization–mineralization and plant growth promotion. Geomicrobiol. J. 2017, 34, 81–95. [Google Scholar] [CrossRef]
- Rfaki, A.; Zennouhi, O.; Aliyat, F.Z.; Nassiri, L.; Ibijbijen, J. Isolation, selection and characterization of root-associated rock phosphate solubilizing bacteria in Moroccan wheat (Triticum aestivum L.). Geomicrobiol. J. 2020, 37, 230–241. [Google Scholar] [CrossRef]
- Premono, M.E.; Moawad, A.M.; Vlek, P.L.G. Effect of Phosphate-Solubilizing Pseudomonas Putida on the Growth of Maize and Its Survival in the Rhizosphere; Centro Internacional de Mejoramiento de Maíz y Trigo, (CIMMYT): Veracruz, Mexico, 1996. [Google Scholar]
- Ma, J.; Bei, Q.; Wang, X.; Lan, P.; Liu, G.; Lin, X.; Liu, Q.; Lin, Z.; Liu, B.; Zhang, Y.; et al. Impacts of Mo application on biological nitrogen fixation and diazotrophic communities in a flooded rice-soil system. Sci. Total Environ. 2019, 649, 686–694. [Google Scholar] [CrossRef]
- Nautiyal, C.S. An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol. Lett. 1999, 170, 265–270. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [Green Version]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef]
- Gordon, S.A.; Weber, R.P. Colorimetric estimation of indoleacetic acid. Plant Physiol. 1951, 26, 192–195. [Google Scholar] [CrossRef] [Green Version]
- Schwyn, B.; Neilands, J.B. Universal chemical assay for the detection and determination of siderophores. Anal. Biochem. 1987, 160, 47–56. [Google Scholar] [CrossRef]
- Bakker, A.W.; Schippers, B. Microbial cyanide production in the rhizosphere in relation to potato yield reduction and Pseudomonas SPP-mediated plant growth-stimulation. Soil Biol. Biochem. 1987, 19, 451–457. [Google Scholar] [CrossRef]
- Hu, X.; Chen, J.; Guo, J. Two phosphate-and potassium-solubilizing bacteria isolated from Tianmu mountain, Zhejiang, China. World J. Microbiol. Biotechnol. 2006, 22, 983–990. [Google Scholar] [CrossRef]
- Vazquez, P.; Holguin, G.; Puente, M.E.; Lopez-Cortes, A.; Bashan, Y. Phosphate-solubilizing microorganisms associated with the rhizosphere of mangroves in a semiarid coastal lagoon. Biol. Fertil. Soils 2000, 30, 460–468. [Google Scholar] [CrossRef]
- McFaland, J. The nephelometer: An instrument for media used for estimating the number of bacteria in suspensions used for calculating the opsonic index and for vaccines. J. Am. Med. Assoc. 1907, 49, 1176–1178. [Google Scholar] [CrossRef] [Green Version]
- Murphy, J.A.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Banik, S.; Dey, B. Phosphate-solubilizing potentiality of the microorganisms capable of utilizing aluminium phosphate as a sole phosphate source. Zent. Mikrobiol. 1983, 138, 17–23. [Google Scholar] [CrossRef]
- Gadagi, R.S.; Sa, T. New isolation method for microorganisms solubilizing iron and aluminum phosphates using dyes. Soil Sci. Plant Nutr. 2002, 48, 615–618. [Google Scholar] [CrossRef]
- Panhwar, Q.A.; Radziah, O.; Sariah, M.; Ismail, M.R. Solubilization of different phosphate forms by phosphate solubilizing bacteria isolated from aerobic rice. Int. J. Agric. Biol. 2009, 11, 667–673. [Google Scholar]
- Devi, Y.B.; Thakuria, D. Diversity of multifunctional phosphorus solubilizing bacteria in acid soils of diverse hill rice ecosystems. J. Indian Soc. Soil Sci. 2021, 69, 306–318. [Google Scholar] [CrossRef]
- Reyes, I.; Bernier, L.; Simard, R.R.; Tanguay, P.; Antoun, H. Characteristics of phosphate solubilization by an isolate of a tropical Penicillium rugulosum and two UV-induced mutants. FEMS Microbiol. Ecol. 1999, 28, 291–295. [Google Scholar] [CrossRef]
- Kang, S.C.; Ha, C.G.; Lee, T.G.; Maheshwari, D.K. Solubilization of insoluble inorganic phosphates by a soil-inhabiting Fungus fomitopsis sp. PS 102. Curr. Sci. 2002, 82, 439–442. [Google Scholar]
- Maharana, R.; Dhal, N.K. Solubilization of rock phosphate by phosphate solubilizing bacteria isolated from effluent treatment plant sludge of a fertilizer plant. Folia Microbiol. 2022, 67, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.K.; Vyas, R.V.; Shelat, H.N. Selective enrichment method for isolation of efficient phosphate solubilizing bacteria from soil. Commun. Soil Sci. Plant Anal. 2022, 53, 1–10. [Google Scholar] [CrossRef]
- Liu, X.; Chen, C.; Wang, J.; Zou, S.; Long, X. Phosphorus solubilizing bacteria Bacillus thuringiensis and Pantoea ananatis simultaneously promote soil inorganic phosphate dissolution and soil Pb immobilization. Rhizosphere 2021, 20, 100448. [Google Scholar] [CrossRef]
- Zúñiga-Silgado, D.; Rivera-Leyva, J.C.; Coleman, J.J.; Sánchez-Reyez, A.; Valencia-Díaz, S.; Serrano, M.; De-Bashan, L.E.; Folch-Mallol, J.L. Soil type affects organic acid production and phosphorus solubilization efficiency mediated by several native fungal strains from Mexico. Microorganisms 2020, 8, 1337. [Google Scholar] [CrossRef]
- Mažylytė, R.; Kaziūnienė, J.; Orola, L.; Valkovska, V.; Lastauskienė, E.; Gegeckas, A. Phosphate solubilizing microorganism Bacillus sp. MVY-004 and its significance for biomineral fertilizers’ development in agrobiotechnology. Biology 2022, 11, 254. [Google Scholar] [CrossRef]
- Arenas, F.; López-García, Á.; Berná, L.M.; Morte, A.; Navarro-Ródenas, A. Desert truffle mycorrhizosphere harbors organic acid releasing plant growth–promoting rhizobacteria, essentially during the truffle fruiting season. Mycorrhiza 2022, 32, 193–202. [Google Scholar] [CrossRef]
- Barin, M.; Asadzadeh, F.; Hashemnejad, F.; Vetukuri, R.R.; Kushwaha, S. Optimization of culture conditions for zinc phosphate solubilization by Aspergillus sp. using response surface methodology. J. Soil Sci. Plant Nutr. 2022, 22, 1009–1018. [Google Scholar] [CrossRef]
- Cardoso, P.; Alves, A.; Silveira, P.; Sá, C.; Fidalgo, C.; Freitas, R.; Figueira, E. Bacteria from nodules of wild legume species: Phylogenetic diversity, plant growth promotion abilities and osmotolerance. Sci. Total Environ. 2018, 645, 1094–1102. [Google Scholar] [CrossRef]
- Nahas, E. Factors determining rock phosphate solubilization by microorganisms isolated from soil. World J. Microbiol. Biotechnol. 1996, 12, 567–572. [Google Scholar] [CrossRef]
- Anand, K.; Kumari, B.; Mallick, M.A. Phosphate solubilizing microbes: An effective and alternative approach as biofertilizers. Int. J. Pharm. Sci. 2016, 8, 37–40. [Google Scholar]
- Omar, S.A. The role of rock-phosphate-solubilizing fungi and vesicular–arbusular-mycorrhiza (VAM) in growth of wheat plants fertilized with rock phosphate. World J. Microbiol. Biotechnol. 1997, 14, 211–218. [Google Scholar] [CrossRef]
- Chatli, A.S.; Beri, V.; Sidhu, B.S. Isolation and characterisation of phosphate solubilising microorganisms from the cold desert habitat of Salix alba Linn. in trans Himalayan region of Himachal Pradesh. Indian J. Microbiol. 2008, 48, 267–273. [Google Scholar] [CrossRef] [Green Version]
- Vasseur-Coronado, M.; Vlassi, A.; du Boulois, H.; Schuhmacher, R.; Parich, A.; Pertot, I.; Puopolo, G. Ecological role of volatile organic compounds emitted by Pantoea agglomerans as interspecies and interkingdom signals. Microorganisms 2021, 9, 1186. [Google Scholar] [CrossRef] [PubMed]
- Mahidi, S.S.; Hassan, G.I.; Hussain, A.; Rasool, F. Phosphorus availability issue—Its fixation and role of phosphate solubilizing bacteria in phosphate solubilization—Case study. Agric. Sci. Res. J. 2011, 2, 174–179. [Google Scholar]
- Elfiati, D.; Delvian, D.; Hanum, H.; Susilowati, A.; Rachmat, H.H. Potential of phosphate solubilizing fungi isolated from peat soils as inoculant biofertilizer. Biodiversitas J. Biol. Divers. 2021, 22, 220605. [Google Scholar] [CrossRef]
- Hocking, P.J. Organic acids exuded from roots in phosphorus uptake and aluminum tolerance of plants in acid soils. Adv. Agron. 2001, 74, 63–97. [Google Scholar] [CrossRef]
- Hou, X.; Han, H.; Cai, L.; Liu, A.; Ma, X.; Zhou, C.; Wang, G.; Meng, F. Pb stress effects on leaf chlorophyll fluorescence, antioxidative enzyme activities, and organic acid contents of Pogonatherum crinitum seedlings. Flora Morphol. Distrib. Funct. Ecol. Plants 2018, 240, 82–88. [Google Scholar] [CrossRef]
- Rawat, P.; Das, S.; Shankhdhar, D.; Shankhdhar, S.C. Phosphate-solubilizing microorganisms: Mechanism and their role in phosphate solubilization and uptake. J. Soil Sci. Plant Nutr. 2020, 21, 49–68. [Google Scholar] [CrossRef]
- Biswas, S.; Shivaprakash, M.K. Effect of co inoculation of potassium solubilizing, mobilizing and phosphorus solubilizing bacteria on growth, yield and nutrient uptake of radish (Raphanus sativus L). Int. J. Adv. Res. Biol. Sci. 2021, 8, 108–113. [Google Scholar]
- Sulbarán, M.; Pérez, E.; Ball, M.M.; Bahsas, A.; Yarzábal, L.A. Characterization of the mineral phosphate-solubilizing activity of Pantoea aglomerans MMB051 isolated from an iron-rich soil in southeastern Venezuela (Bolívar state). Curr. Microbiol. 2009, 58, 378–383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benjelloun, I.; Thami Alami, I.; El Khadir, M.; Douira, A.; Udupa, S.M. Co-inoculation of Mesorhizobium ciceri with either Bacillus sp. or Enterobacter aerogenes on chickpea improves growth and productivity in phosphate-deficient soils in dry areas of a Mediterranean region. Plants 2021, 10, 571. [Google Scholar] [CrossRef]
- Adhikari, P.; Jain, R.; Sharma, A.; Pandey, A. Plant growth promotion at low temperature by phosphate-solubilizing Pseudomonas spp. isolated from high-altitude Himalayan soil. Microb. Ecol. 2021, 82, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Wang, Z.; He, S.; Shi, L.; Song, Y.; Lou, X.; He, D. Endogenous hormone levels and activities of IAA-modifying enzymes during adventitious rooting of tree peony cuttings and grafted scions. Hortic. Environ. Biotechnol. 2019, 60, 187–197. [Google Scholar] [CrossRef]
- Sultana, S.; Alam, S.; Karim, M.M. Screening of siderophore-producing salt-tolerant rhizobacteria suitable for supporting plant growth in saline soils with iron limitation. J. Agric. Food Res. 2021, 4, 100150. [Google Scholar] [CrossRef]
- Rajkumar, M.; Ae, N.; Prasad, M.N.V.; Freitas, H. Potential of siderophore-producing bacteria for improving heavy metal phytoextraction. Trends Biotechnol. 2010, 28, 142–149. [Google Scholar] [CrossRef]
- Raji, M.; Thangavelu, M. Isolation and screening of potassium solubilizing bacteria from saxicolous habitat and their impact on tomato growth in different soil types. Arch. Microbiol. 2021, 203, 3147–3161. [Google Scholar] [CrossRef]
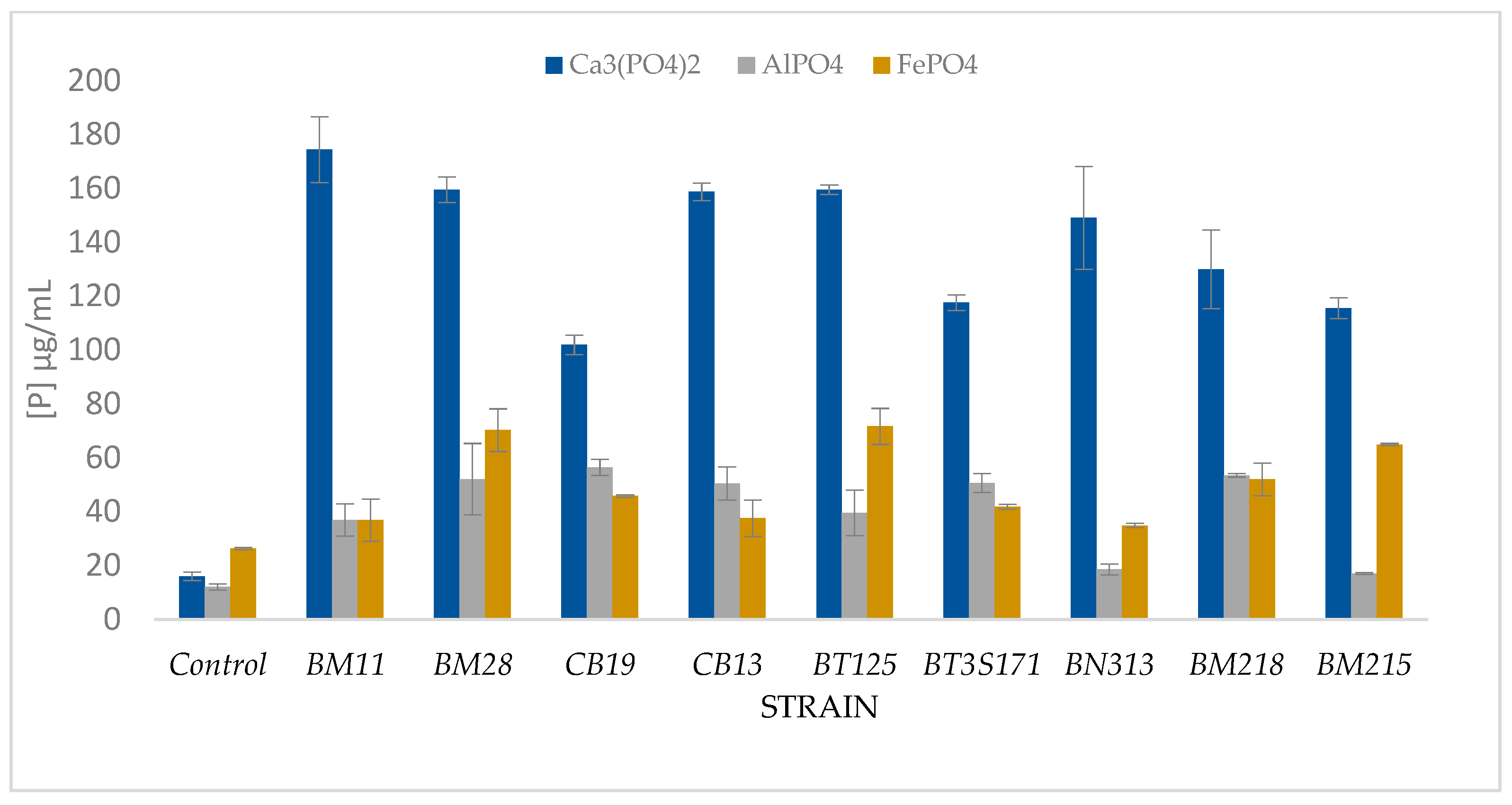
| Strains | Colony Diameter (cm) | Halo Zone Diameter (cm) | PSI = C + H/C |
|---|---|---|---|
| BM11 | 0.98 | 2.3 | 3.33 b |
| BM28 | 0.78 | 2.6 | 4.10 a |
| CB19 | 0.58 | 2.4 | 4.79 d |
| CB13 | 0.70 | 1.5 | 2.77 c |
| BT125 | 0.68 | 2.3 | 4.12 a |
| BT3S171 | 0.65 | 1.4 | 2.77 c |
| BN313 | 0.63 | 1.6 | 3.11 b |
| BM218 | 0.95 | 2.9 | 4.00 a |
| BM215 | 0.78 | 2.1 | 3.42 b |
| Strain | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| BM11 | BM28 | CB19 | CB13 | BT125 | BT3S171 | BN313 | BM218 | BM215 | ||
| Colony color | Red | Yellowish | Yellowish | Yellow-ish | Yellow-ish | Yellowish | Yellow-ish | Yellowish | Yellowish | |
| Gram | − | − | − | − | − | − | − | − | − | |
| β−galactosidase | + | + | + | + | − | + | − | + | + | |
| Arginine dihydrolase | + | + | − | − | − | − | + | − | + | |
| Lysine decarboxylase | − | − | − | − | − | − | − | − | − | |
| Ornithine decarboxylase | − | + | − | − | − | − | + | − | + | |
| Citrate utilization | + | + | − | − | − | − | − | − | + | |
| H2S production | − | − | − | − | − | − | − | − | − | |
| Urease | − | − | − | − | + | − | − | − | − | |
| Tryptophan deaminase | − | − | − | − | − | − | − | − | − | |
| Indole production | − | − | − | − | − | − | − | − | − | |
| Acetoin production | − | − | + | − | − | − | − | − | − | |
| Gelatinase | + | + | + | + | + | − | + | + | + | |
| Sugar fermentation | Glucose | + | + | + | + | − | + | + | + | + |
| Mannitol | + | + | − | + | − | − | − | + | + | |
| Inositol | − | − | − | − | − | − | − | − | − | |
| Sorbitol | − | − | + | − | − | − | − | − | − | |
| Rhamnose | − | + | + | + | − | − | − | − | + | |
| Sucrose | + | − | + | + | − | − | − | + | − | |
| Melibiose | + | − | + | − | − | − | + | + | − | |
| Amygdalin | + | + | + | + | − | − | − | + | + | |
| Arabinose | + | + | + | + | − | − | + | + | + | |
| PGP traits | Indole-3-acetic acid production (µg/mL) | 2.6 ± 0.0 a | 7.7 ± 0.03 b | 3.2 ± 0.03 a | 8.7 ± 0.1 c | 3.9 ± 0.4 a | 43.8 ± 0.39 d | 1.3 ± 0.09 e | 20.5 ± 0.03 f | 15.9 ± 0.2 g |
| Siderophore | + | + | + | + | ++ | ++ | ++ | ++ | ++ | |
| Hydrogen cyanide | − | − | − | − | ++ | − | − | − | − | |
| K solubilization | +++ | ++ | +++ | +++ | ++ | +++ | ++ | + | + | |
| Strain | Acetic | Formic | Propionic | Isobutyric | Butyric | Isovaleric | Caproic | Heptanoic |
|---|---|---|---|---|---|---|---|---|
| mg/L | ||||||||
| BM11 | 40.62 | 3.99 | 1.59 | 6.86 | 8.53 | 1.32 | 0.59 | 0.51 |
| BM28 | 102.98 | 1.78 | 9.14 | 5.39 | ND | 0.10 | 2.19 | ND |
| CB19 | 28.67 | 3.42 | ND | 3.89 | 2.95 | 1.21 | 7.29 | ND |
| CB13 | 27.01 | 2.93 | ND | 4.23 | 2.88 | 0.31 | 5.60 | ND |
| BT125 | 93.27 | 3.94 | 0.38 | 5.48 | ND | 0.98 | 7.87 | 0.20 |
| BT3S171 | 73.52 | 3.70 | 0.79 | 8.12 | ND | ND | 1.64 | ND |
| BN313 | 60.94 | 7.38 | 5.80 | 4.66 | 3.89 | ND | ND | ND |
| BM218 | 28.77 | 2.80 | ND | 4.55 | 2.37 | 0.90 | 4.58 | ND |
| BM215 | 144.76 | 2.71 | 8.67 | 8.01 | ND | 0.82 | 1.34 | ND |
| Strain | Hour of Incubation | |||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 48 | 96 | 144 | |||||
| [P] µg/mL | pH | [P] µg/mL | pH | [P] µg/mL | pH | [P] µg/mL | pH | |
| Control | 12.95 ± 1.39 a | 7.0 ± 0.2 a | 16.61 ± 2.62 a | 6.3 ± 0.04 a | 16.00 ± 1.57 a | 6.3 ± 0.00 a | 12.67 ± 1.46 a | 6.51 ± 0.0 a |
| BM11 | 20.65 ± 0.09 a | 7.0 ± 0.2 a | 109.07 ± 5.4 d | 4.2 ± 0.02 b | 174.33 ± 12.19 e | 4.1 ± 0.08 d | 101.30 ± 6.49 b | 4.0 ± 0.25 ef |
| BM28 | 14.85 ± 0.09 a | 7.0 ± 0.2 a | 106.83 ± 1.23 d | 4.7 ± 0.06 c | 159.48 ± 4.73 de | 4.4 ± 0.1 bc | 111.15 ± 8.79 bc | 4.6 ±0.2 ef |
| CB19 | 13.23 ± 0.73 a | 7.0 ± 0.2 a | 81.10 ± 35.36 b | 4.1 ± 0.08 b | 101.90 ± 3.55 b | 4.2 ± 0.7 cd | 112.51 ± 7.56 bc | 4.6 ±0.2 cd |
| CB13 | 17.47 ± 0.07 a | 7.0 ± 0.2 a | 110.34 ± 5.74 d | 4.2 ± 0.05 b | 158.72 ± 3.27 de | 4.1 ± 0.97 d | 107.21 ± 10.7 bc | 4.3 ± 0.3 de |
| BT125 | 15.44 ± 0.30 a | 7.0 ± 0.2 a | 107.90 ± 5.60 d | 4.2 ± 0.04 b | 159.48 ± 1.83 de | 4.1 ± 0.00 d | 117.36 ± 0.78 c | 4.1 ± 0.0 bcd |
| BT3S171 | 15.58 ± 2.03 a | 7.0 ± 0.2 a | 23.87 ± 3.38 a | 5.9 ± 0.09 e | 117.51 ± 2.92 bc | 4.0 ± 0.27 d | 111.45 ± 6.14 bc | 3.9 ± 0.0 b |
| BN313 | 20.29 ± 0.10 a | 7.0 ± 0.2 a | 88.72 ± 28.6 bc | 4.9 ± 0.28 d | 149.03 ± 19.06 | 4.5 ± 0.22 b | 114.18 ± 2.53 c | 4.3 ± 0.25 cde |
| BM218 | 12.03 ± 0.09 a | 7.0 ± 0.2 a | 104.4 ± 2.45 cd | 4.2 ± 0.05 b | 129.93 ± 14.54 c | 4.1 ± 0.05 d | 106.00 ± 8.19 bc | 4.06 ± 0.2 bc |
| BM215 | 14.21 ± 1.15 a | 7.0 ± 0.2 a | 62.06 ± 6.95 b | 4.6 ± 0.03 c | 115.54 ± 3.88 bc | 4.5 ± 0.0 b | 112.96 ± 4.59 bc | 4.7 ±0.2 f |
| Strain | Hour of Incubation | |||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 48 | 96 | 144 | |||||
| [P] µg/mL | pH | [P] µg/mL | pH | [P] µg/mL | pH | [P] µg/mL | pH | |
| Control | 24.42 ± 1.11 a | 7.0 ± 0.2 a | 27.81 ± 0.37 a | 6.9 ± 0.17 a | 26.29 ± 0.39 a | 5.8 ± 0.2 a | 29.44 ± 0.44 a | 5.1 ± 0.1 a |
| BM11 | 38.18 ± 3.43 b | 7.0 ± 0.2 a | 32.83 ± 0.78 a | 3.9 ± 0.0 c | 36.90 ± 7.82 bc | 3.8 ± 0.0 c | 33.59 ± 0.53 a | 3.8 ± 0.0 c |
| BM28 | 33.48 ± 2.74 b | 7.0 ± 0.2 a | 57.84 ± 2.07 cde | 4.1 ± 0.0 d | 70.26 ± 7.92 e | 4.0 ± 0.0 cd | 84.15 ± 5.03 e | 3.7 ± 0.0 cd |
| CB19 | 41.90 ± 2.54 b | 7.0 ± 0.2 a | 51.30 ± 0.42 cd | 3.3 ± 0.0 b | 45.81 ± 0.37 cb | 3.3 ±0.0 b | 48.43 ± 1.85 df | 3.3 ±0.0 b |
| CB13 | 31.21 ± 1.56 b | 7.0 ± 0.2 a | 61.39 ± 3.3 e | 4.1 ± 0.0 d | 37.52 ± 6.74 cd | 3.3 ± 0.2 b | 36.50 ± 0.82 a | 3.3 ± 0.12 b |
| BT125 | 36.79 ± 5.17 b | 7.0 ± 0.2 a | 40.8 ± 1.1 b | 3.7 ± 0.2 b | 71.64 ± 6.72 e | 4.0 ± 0.1 cd | 77.44 ±7.26 e | 4.0 ± 0.78 d |
| BT3S171 | 34.38 ± 1.38 b | 7.0 ± 0.2 a | 61.39 ± 3.3 cde | 3.4 ± 0.0 b | 41.85 ± 0.93 bc | 3.4 ± 0.0 b | 35.60 ±2.54 a | 3.4 ± 0.0 b |
| BN313 | 39.76 ± 0.04 b | 7.0 ± 0.2 a | 40.05 ± 2.3 b | 4.2 ± 0.0 d | 34.85 ± 0.79 ab | 4.2 ± 0.0 d | 15.86 ± 4.36 b | 4.0 ± 0.0 d |
| BM218 | 47.76 ± 1.54 b | 7.0 ± 0.2 a | 58.18 ± 6.8 de | 3.4 ± 0.0 b | 51.96 ± 6.09 d | 3.4 ± 0.0 b | 44.86 ± 3.83 d | 3.3 ± 0.0 b |
| BM215 | 33.14 ± 0.71 b | 7.0 ± 0.2 a | 50.96 ± 5.61 c | 3.9 ± 0.0 c | 64.92 ± 0.43 e | 4.2 ± 0.0 d | 51.87 ± 3.58 f | 3.8 ± 0.0 d |
| Strain | Hour of Incubation | |||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 48 | 96 | 144 | |||||
| [P] µg/mL | pH | [P] µg/mL | pH | [P] µg/mL | pH | [P] µg/mL | pH | |
| Control | 24.42 ± 1.11 a | 7.0 ± 0.2 a | 27.81 ± 0.37 a | 6.9 ± 0.17 a | 26.29 ± 0.39 a | 5.8 ± 0.2 a | 29.44 ± 0.44 a | 5.1 ± 0.1 a |
| BM11 | 38.18 ± 3.43 b | 7.0 ± 0.2 a | 32.83 ± 0.78 a | 3.9 ± 0.0 c | 36.90 ± 7.82 bc | 3.8 ± 0.0 c | 33.59 ± 0.53 a | 3.8 ± 0.0 c |
| BM28 | 33.48 ± 2.74 b | 7.0 ± 0.2 a | 57.84 ± 2.07 cde | 4.1 ± 0.0 d | 70.26 ± 7.92 e | 4.0 ± 0.0 cd | 84.15 ± 5.03 e | 3.7 ± 0.0 cd |
| CB19 | 41.90 ± 2.54 b | 7.0 ± 0.2 a | 51.30 ± 0.42 cd | 3.3 ± 0.0 b | 45.81 ± 0.37 cb | 3.3 ±0.0 b | 48.43 ± 1.85 df | 3.3 ±0.0 b |
| CB13 | 31.21 ± 1.56 b | 7.0 ± 0.2 a | 61.39 ± 3.3 e | 4.1 ± 0.0 d | 37.52 ± 6.74 cd | 3.3 ± 0.2 b | 36.50 ± 0.82 a | 3.3 ± 0.12 b |
| BT125 | 36.79 ± 5.17 b | 7.0 ± 0.2 a | 40.8 ± 1.1 b | 3.7 ± 0.2 b | 71.64 ± 6.72 e | 4.0 ± 0.1 cd | 77.44 ±7.26 e | 4.0 ± 0.78 d |
| BT3S171 | 34.38 ± 1.38 b | 7.0 ± 0.2 a | 61.39 ± 3.3 cde | 3.4 ± 0.0 b | 41.85 ± 0.93 bc | 3.4 ± 0.0 b | 35.60 ±2.54 a | 3.4 ± 0.0 b |
| BN313 | 39.76 ± 0.04 b | 7.0 ± 0.2 a | 40.05 ± 2.3 b | 4.2 ± 0.0 d | 34.85 ± 0.79 ab | 4.2 ± 0.0 d | 15.86 ± 4.36 b | 4.0 ± 0.0 d |
| BM218 | 47.76 ± 1.54 b | 7.0 ± 0.2 a | 58.18 ± 6.8 de | 3.4 ± 0.0 b | 51.96 ± 6.09 d | 3.4 ± 0.0 b | 44.86 ± 3.83 d | 3.3 ± 0.0 b |
| BM215 | 33.14 ± 0.71 b | 7.0 ± 0.2 a | 50.96 ± 5.61 c | 3.9 ± 0.0 c | 64.92 ± 0.43 e | 4.2 ± 0.0 d | 51.87 ± 3.58 f | 3.8 ± 0.0 d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aliyat, F.Z.; Maldani, M.; El Guilli, M.; Nassiri, L.; Ibijbijen, J. Phosphate-Solubilizing Bacteria Isolated from Phosphate Solid Sludge and Their Ability to Solubilize Three Inorganic Phosphate Forms: Calcium, Iron, and Aluminum Phosphates. Microorganisms 2022, 10, 980. https://doi.org/10.3390/microorganisms10050980
Aliyat FZ, Maldani M, El Guilli M, Nassiri L, Ibijbijen J. Phosphate-Solubilizing Bacteria Isolated from Phosphate Solid Sludge and Their Ability to Solubilize Three Inorganic Phosphate Forms: Calcium, Iron, and Aluminum Phosphates. Microorganisms. 2022; 10(5):980. https://doi.org/10.3390/microorganisms10050980
Chicago/Turabian StyleAliyat, Fatima Zahra, Mohamed Maldani, Mohammed El Guilli, Laila Nassiri, and Jamal Ibijbijen. 2022. "Phosphate-Solubilizing Bacteria Isolated from Phosphate Solid Sludge and Their Ability to Solubilize Three Inorganic Phosphate Forms: Calcium, Iron, and Aluminum Phosphates" Microorganisms 10, no. 5: 980. https://doi.org/10.3390/microorganisms10050980
APA StyleAliyat, F. Z., Maldani, M., El Guilli, M., Nassiri, L., & Ibijbijen, J. (2022). Phosphate-Solubilizing Bacteria Isolated from Phosphate Solid Sludge and Their Ability to Solubilize Three Inorganic Phosphate Forms: Calcium, Iron, and Aluminum Phosphates. Microorganisms, 10(5), 980. https://doi.org/10.3390/microorganisms10050980






