Zoonotic Implications of Onchocerca Species on Human Health
Abstract
1. Introduction
2. How Many Definitive Hosts Are Parasitized by Onchocerca spp.?
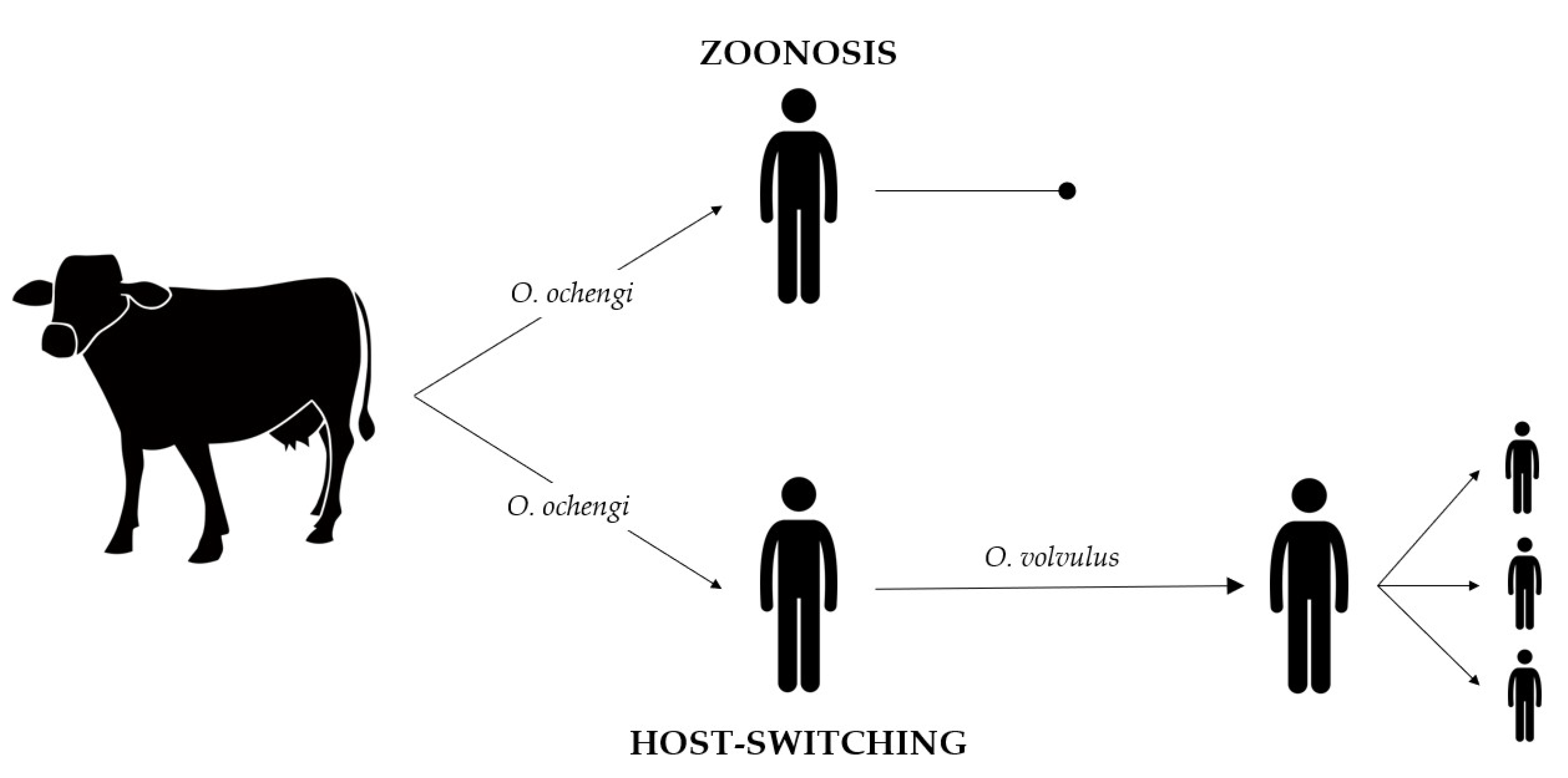
3. Does Onchocerciasis Have a Zoonotic Character?
4. Which Factors Are Involved in the Transmission of Animal Onchocerca Species to Humans?
5. What Are the Lesions and Clinical Signs that Onchocerca Species Infecting Animals Cause in Humans?
5.1. Is It Possible That Onchocerca Species Infecting Animals Produce Clinical Symptoms Such as Blindness in Humans?
5.2. Is It Possible That Onchocerca Species Infecting Animals Produce Clinical Disorders Such as Epilepsy in Humans?
6. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Centers for Disease Control and Prevention (CDC). Parasites–Onchocerciasis (Also Known as River Blindness). Available online: https://www.cdc.gov/parasites/onchocerciasis/ (accessed on 11 May 2020).
- Muller, R. Identification of Onchocerca. Symposia of the British Society for Parasitology. In Problems in the Identification of Parasites and Their Vectors, 2nd ed.; Taylor, A.E.R., Mullers, R., Eds.; Blackwell Scientific: London, UK, 1979; Volume 17, pp. 175–206. [Google Scholar]
- Ogbogu, V.C.; Bablis, J.M.; Ajanusi, O.J. Prevalence of Microfilariae in Cattle at Slaughter in Zaria, Nigeria. Vet. Parasitol. 1990, 36, 171–175. [Google Scholar] [CrossRef]
- Lefoulon, E.; Giannelli, A.; Makepeace, B.L.; Mutafchiev, Y.; Townson, S.; Uni, S.; Verocai, G.G.; Otranto, D.; Martin, C. Whence river blindness? The domestication of mammals and host-parasite co-evolution in the nematode genus Onchocerca. Int. J. Parasitol. 2017, 47, 457–470. [Google Scholar] [CrossRef] [PubMed]
- Boijsen, B.; Uhlhorn, H.; Ågren, E.; Höglund, J. Nodular onchocercosis in red deer (Cervus elaphus) in Sweden. Int. J. Parasitol. Parasites. Wildl. 2017, 6, 340–343. [Google Scholar] [CrossRef] [PubMed]
- Gumisiriza, N.; Mubiru, F.; Siewe Fodjo, J.N.; Mbonye Kayitale, M.; Hotterbeekx, A.; Idro, R.; Makumbi, I.; Lakwo, T.; Opar, B.; Kaducu, J.; et al. Prevalence and incidence of nodding syndrome and other forms of epilepsy in onchocerciasis-endemic areas in northern Uganda after the implementation of onchocerciasis control measures. Infect. Dis. Poverty 2020, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Verocai, G.G.; Nelson, K.J.; Callahan, R.T.; Wekesa, J.W.; Hassan, H.K.; Hoberg, E.P. A cryptic species of Onchocerca (Nematoda: Onchocercidae) in blackflies (Simulium spp.) from southern California, USA. Parasites Vectors 2018, 11, 547. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, M.R.; Martínez, A.; Carreño, R.A.; González, S.; Ferreras, M.C.; Díez, N. Levels of infection, pathology and nodule size of Onchocerca flexuosa (Nematoda: Onchocercidae) in red deer (Cervus elaphus) from northern Spain. J. Helminthol. 2015, 89, 326–334. [Google Scholar] [CrossRef]
- Verocai, G.G.; Lejeune, M.; Beckmen, K.B.; Kashivakura, C.K.; Veitch, A.M.; Popko, R.A.; Fuentealba, C.; Hoberg, E.P.; Kutz, S.J. Defining parasite biodiversity at high latitudes of North America: New host and geographic records for Onchocerca cervipedis (Nematoda: Onchocercidae) in moose and caribou. Parasites Vectors 2012, 5, 242. [Google Scholar] [CrossRef]
- Neary, J.M.; Trees, A.J.; Ekale, D.D.; Tanya, V.N.; Hetzel, U.; Makepeace, B.L. Onchocerca armillata contains the endosymbiotic bacterium Wolbachia and elicits a limited inflammatory response. Vet. Parasitol. 2010, 174, 267–276. [Google Scholar] [CrossRef]
- ElBihari, S.; Hussein, H.S. The distribution and redescription of the microfilariae of Onchocerca armillata Railliet and Henry, 1909. Sudan J. Vet. Sci. Anim. Husb. 1976, 17, 77–85. [Google Scholar]
- Hussein, H.S.; Atta El Mannan, A.M.; El Sinnary, K. Onchocerca armillata Railliet and Henry, 1909 and Onchocerca gutturosa (Neumann, 1910) in camels (Camelus dromedarius L.) in the Sudan. Vet. Res. Commun. 1988, 12, 475–480. [Google Scholar] [CrossRef]
- Chodnik, K.S. Aortic onchocerciasis due to Onchocerca armillata in cattle in Ghana, with special reference to the morphology of the parasite. Ann. Trop. Med. Parasitol. 1957, 51, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Cheema, A.H.; Ivoghli, B. Bovine onchocerciasis caused by Onchocerca armillata and O. gutturosa. Vet. Pathol. 1978, 15, 495–505. [Google Scholar] [CrossRef]
- Vassiliades, G.; Delbove, P.; Bain, O. Onchocercoses bovines au Senegal. Note preliminaire. Rev. Elev. Med. Vet. Pays. Trop. 1983, 36, 351–353. [Google Scholar]
- Trees, A.J.; McCall, P.J.; Davies, J.B. On the possibility of bovine Onchocerca species infecting Simulium damnosum s.l. in the forest zone of Sierra Leone. I. Parasitological aspects. Ann. Trop. Med. Parasitol. 1989, 83, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Mtei, B.J.; Sanga, H.J. Aortic onchocercosis and elaeophorosis in traditional TSZ-cattle in Tabora (Tanzania): Prevalence and pathology. Vet. Parasitol. 1990, 36, 165–170. [Google Scholar] [CrossRef]
- Begam, R.; Islam, S.; Saikia, M.; Kalita, A.; Bulbul, K.M.; Bam, J.; Pathak, P. Prevalence of aortic onchocerciasis in cattle of Assam. Vet. Pract. 2015, 16, 225–227. [Google Scholar]
- Lia, R.P.; Mutafchiev, Y.; Veneziano, V.; Giannelli, A.; Abramo, F.; Santoro, M.; Latrofa, M.S.; Cantacessi, C.; Martin, C.; Otranto, D.; et al. Filarial infection caused by Onchocerca boehmi (Supperer, 1953) in a horse from Italy. Parasitol. Res. 2017, 116, 191–198. [Google Scholar] [CrossRef]
- Supperer, R. Filarosen der Pferde in Österreich. Wien. Tierärztliche Mon. 1953, 40, 193–220. [Google Scholar]
- Mirzayans, A.; Maghsoodloo, H. Filarial infection of Equidae in the Tehran area of Iran. Trop. Anim. Health Prod. 1977, 9, 19–20. [Google Scholar] [CrossRef]
- Mellor, P.S. Studies on Onchocerca cervicalis Railliet and Henry 1910: I. Onchocerca cervicalis in British horses. J. Helminthol. 1973, 47, 97–110. [Google Scholar] [CrossRef]
- Foil, L.D.; Kleir, T.R.; Miller, R.I.; Foil, C.S.; French, D.D.; Smith, J.N. Seasonal changes in density and tissue distribution of Onchocerca cervicalis microfilariae in ponies and related changes in Culicoides variipennis populations in Louisiana. J. Parasitol. 1987, 73, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Riek, R.F. Studies on allergic dermatitis (Queensland itch) of the horse. I. Description, distribution, symptoms and pathology. Aust. Vet. J. 1953, 7, 177–184. [Google Scholar] [CrossRef]
- Monahan, C.M.; Chapman, M.R.; French, D.D.; Klei, T.R. Efficacy of moxidectin oral gel against Onchocerca cervicalis microfilariae. J. Parasitol. 1995, 81, 117–118. [Google Scholar] [CrossRef] [PubMed]
- Burr, W.E., Jr.; Brown, M.F.; Eberhard, M.L. Zoonotic Onchocerca (Nematoda:Filarioidea) in the cornea of a Colorado resident. Ophthalmology 1998, 105, 1494–1497. [Google Scholar] [CrossRef]
- Radwan, A.M.; Ahmed, N.E.; Elakabawy, L.M.; Ramadan, M.Y.; Elmadawy, R.S. Prevalence and pathogenesis of some filarial nematodes infecting donkeys in Egypt. Vet. World 2016, 9, 888–892. [Google Scholar] [CrossRef]
- Sato, T.; Hayashi, S.; Tanaka, K. Studies on Onchocerca gutturosa Neumann, 1910 as a causative parasite of bovine dermatitis ‘Wahi’ and Onchocerca cervicalis Railliet et Henry, 1910 as a causative parasite of equine dermatitis ‘Kasen’. Jpn. J. Parasitol. 1954, 2, 199–206. [Google Scholar]
- Lutz, J.E.; Boersema, J.H.; Németh, F. Preliminary studies on the incidence of Filaziae in equidae in the Netherlands. Tijdschr. Diergeneeskd. 1975, 100, 321–326. [Google Scholar]
- Lees, M.J.; Kleider, N.; Tuddenham, T.J. Cutaneous onchocerciasis in the horse: Five cases in southwestern british columbia. Can. Vet. J. 1983, 24, 3–5. [Google Scholar]
- French, D.D.; Klei, T.R.; Foil, C.S.; Miller, R.I.; Foil, L.D.; Chapman, M.R.; McClure, J.J. Efficacy of ivermectin in paste and injectable formulations against microfilariae of Onchocerca cervicalis and resolution of associated dermatitis in horses. Am. J. Vet. Res. 1988, 49, 1550–1554. [Google Scholar]
- Marques, S.M.T.; Scroferneker, M.L. Onchocerca cervicalis in horses from southern Brazil. Trop. Anim. Health Prod. 2004, 36, 633–636. [Google Scholar] [CrossRef]
- Franck, M.T.; Colombet, J.; Hugnet, C.; Ducos de Lahitte, J.; Desmaizières, L.M.; Delverdier, M.; Franc, M. Research of skin microfilariae on 160 horses from Poland, France and Spain. Revue. Méd. Vét. 2006, 157, 323–325. [Google Scholar]
- Pledger, D.J. Black flies (Diptera, Simuliidae) of the Swan Hills, Alberta, as possible vectors of Onchocerca cervipedis Wehr Dikmans 1935 (Nematoda; Onchocercidae) in moose (Alces alces Linnaeus). Ph.D. Thesis, University of Alberta, Edmonton, AB, Canada, 1978. [Google Scholar]
- Wehr, E.E.; Dikmans, G. New nematodes (Filariidae) from North America ruminants. Zool. Anz. 1935, 110, 202–208. [Google Scholar]
- Carreno, R.A.; Durden, L.A.; Brooks, D.R.; Abrams, A.; Hoberg, E.P. Parelaphostrongylus tenuis (Nematoda: Protostrongylidae) and other parasites of white-tailed deer (Odocoileus virginianus) in Costa Rica. Comp. Parasitol. 2001, 68, 177–184. [Google Scholar]
- Bain, O.; Ramachandran, C.P.; Petter, F.; Mak, J.W. Description d’Onchocerca dewittei n. sp. (Filarioidea) chez Sus scrofa en Malaisie. Ann. Parasitol. Hum. Comp. 1977, 52, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, M.; Otsuka, Y.; Uni, S.; Bain, O.; Takaoka, H. Molecular identification of infective larvae of three species of Onchocerca found in wild-caught females of Simulium bidentatum in Japan. Parasite 2010, 17, 39–45. [Google Scholar] [CrossRef]
- Takaoka, H.; Fukuda, M.; Otsuka, Y.; Aoki, C.; Uni, S.; Bain, O. Blackfly vectors of zoonotic onchocerciasis in Japan. Med. Vet. Entomol. 2012, 26, 372–378. [Google Scholar] [CrossRef]
- Uni, S.; Bain, O.; Takaoka, H.; Miyashita, M.; Suzuki, Y. Onchocerca dewittei japonica n. subsp., a common parasite from wild boar in Kyushu Island, Japan. Parasite 2001, 8, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, H.; Bain, O.; Uni, S.; Korenaga, M.; Shirasaka, C.; Aoki, C.; Otsuka, Y.; Fukuda, M.; Eshita, Y.; Daa, T. Zoonotic onchocerciasis caused by a parasite from wild boar in Oita, Japan. A comprehensive analysis of morphological characteristics of the worms for its diagnosis. Parasite 2004, 11, 285–292. [Google Scholar] [CrossRef][Green Version]
- Wahl, G.; Ekale, D.; Enyong, P.; Renz, A. The development of Onchocerca dukei and O. ochengi microfilariae to infective-stage larvae in Simulium damnosum s.l. and in members of the S. medusaeforme group, following intra-thoracic injection. Ann. Trop. Med. Parasitol. 1991, 85, 329–337. [Google Scholar] [CrossRef]
- Wahl, G.; Achu-Kwi, M.D.; Mbah, D.; Dawa, O.; Renz, A. Bovine onchocercosis in north Cameroon. Vet. Parasitol. 1994, 52, 297–311. [Google Scholar] [CrossRef]
- Beaver, P.C.; Hira, P.R.; Patel, B.G. Onchocerciasis in Zambia: Report of O. volvulus in a child and its differentiation from O. dukei in cattle. Trans. R. Soc. Trop. Med. Hyg. 1983, 77, 162–166. [Google Scholar] [CrossRef]
- Denke, A.M. The prevalence of Onchocerca spp. in cattle in northern Togo in 1979. Trop. Med. Parasitol. 1986, 37, 46–48. [Google Scholar]
- Fukuda, M.; Takaoka, H.; Uni, S.; Bain, O. Infective larvae of five Onchocerca species from experimentally infected Simulium species in an area of zoonotic onchocerciasis in Japan. Parasite 2008, 15, 111–119. [Google Scholar] [CrossRef]
- Uni, S.; Bain, O.; Agatsuma, T.; Harada, M.; Torii, H.; Fukuda, M.; Takaoka, H. Onchocerca eberhardi n. sp. (Nematoda: Filarioidea) from sika deer in Japan; relationships between species parasitic in cervids and bovids in the Holarctic region. Parasite 2007, 14, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Yu, Z.C.; Luo, X.P.; Chen, L.J.; Shao, G.Y.; Yang, X.Y. Survey of infection and determination of the transmission vector of Onchocerca fasciata in camels (Camelus bactrianus) in Inner Mongolia, China. Med. Vet. Entomol. 2020, 34, 59–68. [Google Scholar] [CrossRef]
- Cheema, A.H.; El-Bihari, S.; Ashour, N.A.; Ali, A.S. Onchocerciasis in camels (Camelus dromedarius) in Saudi Arabia. J. Helminthol. 1984, 58, 279–285. [Google Scholar] [CrossRef]
- Chhabra, M.B.; Gupta, S.K. Parasitic diseases of camels. J. Camel. Pract. Res. 2006, 13, 81–87. [Google Scholar]
- El-Massry, A.A.; Derbala, A.A. Evidence of Onchocerca fasciata (Filaroidea: Onchocercidae) in camels (Camelus dromedarius): I- prevalence, nodular lesions appearance and parasite morphology. Vet. Parasitol. 2000, 88, 305–312. [Google Scholar] [CrossRef]
- Al-Rawashdeh, O.F.; Al-Ani, F.K.; Sharrif, L.A.; Al-Qudah, K.M.; Al-Hami, Y.; Frank, N. A survey of camel (Camelus dromedarius) diseases in Jordan. J. Zoo Wildl. Med. 2000, 31, 335–338. [Google Scholar] [PubMed]
- Krueger, A.; Fischer, P.; Morales-Hojas, R. Molecular phylogeny of the filaria genus Onchocerca with special emphasis on Afrotropical human and bovine parasites. Acta Trop. 2007, 101, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sazmand, A.; Joachim, A. Parasitic diseases of camels in Iran (1931–2017)—A literature review. Parasite 2017, 24, 21. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.C.; Zhang, W.; Li, B.; Luo, X.P.; Wang, R.; Yang, X.Y. A survey of Onchocerca fasciata infection in camels (Camelus bactrianus) with notes on morphology. PLoS ONE 2019, 14, e0214477. [Google Scholar] [CrossRef] [PubMed]
- Dykova, I. Results of studies on the potential vectors of microfilariae of Onchocerca flesuosa (Weld, 1856) and Wehrdikmansia cervipedis (Wehr et Dikmans, 1935). Acta Vet. 1972, 41, 203–206. [Google Scholar]
- Dikmans, G. Onchocerca flexuosa from the subcutaneous tissues of an antelope and subcutaneous abscesses of a deer. J. Parasitol. 1933, 19, 246. [Google Scholar]
- Bain, O.; Schulz-Key, H. Les onchocerques du cerf européen: Redescription d’O. flexuosa (Wedl, 1856) et description d’O. tubingensis n.sp. et O. tarsicola n.sp. Tropenmed. Parasitol. 1974, 25, 437–449. [Google Scholar]
- Demiaszkiewicz, A.W. Occurrence of tissue Filarioidea in ruminants in Poland. Wiad. Parazytol. 1989, 35, 315–321. [Google Scholar]
- Wildenburg, G.; Plenge-Bönig, A.; Renz, A.; Fischer, P.; Büttner, D.W. Distribution of mast cells and their correlation with inflammatory cells around Onchocerca gutturosa, O. tarsicola, O. ochengi, and O. flexuosa. Parasitol. Res. 1997, 83, 109–120. [Google Scholar] [CrossRef]
- McNulty, S.N.; Fischer, K.; Curtis, K.C.; Weil, G.J.; Brattig, N.W.; Fischer, P.U. Localization of Wolbachia-like gene transcripts and peptides in adult Onchocerca flexuosa worms indicates tissue specific expression. Parasites Vectors 2013, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Barbušinová, E.; Iglódyová, A.; Čurlík, J.; Lazar, P.; Mravcová, K.; Štrkolcová, G.; Mucha, R.; Karolová, R. First molecular detection of Onchocerca flexuosa (Wedl, 1856) in red deer in Slovakia. Parasitol. Res. 2020, 119, 985–990. [Google Scholar] [CrossRef]
- El Sinnary, K.; Hussein, H.S. Culicoides kingi, Austen: A vector of Onchocerca gutturosa (Neumann, 1910) in the Sudan. Ann. Trop. Med. Parasitol. 1980, 74, 655–656. [Google Scholar] [CrossRef]
- Holdsworth, P.A.; Moorhouse, D.E. Onchocerca gutturosa in an Australian camel. Aust. Vet. J. 1985, 62, 201–202. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.W.; Miller, W.H. Equine Dermatology; Elsevier: St. Louis, MO, USA, 2003; pp. 242–245. [Google Scholar]
- Wright, R.W.; Neafie, R.C.; McLean, M.; Markman, A.W. Zoonotic onchocerciasis of the shoulder. A case report. J. Bone Joint Surg. Am. 2002, 84, 627–629. [Google Scholar] [CrossRef] [PubMed]
- Beytut, E.; Akca, A.; Bain, O. Teat onchocercosis in cows with reference to prevalence, species involved and pathology. Res. Vet. Sci. 2005, 78, 45–51. [Google Scholar] [CrossRef]
- Wesolowska, M.; Zając-Pytrus, H.; Masny, A.; Pytrus, W.; Knysz, B.; Golab, E.; Sałamatin, R. Onchocerca jakutensis ocular infection in Poland: A new vector-borne human health risk? Parasites Vectors 2020, 13, 61. [Google Scholar] [CrossRef]
- Morandi, F.; Krueger, A.; Panarese, S.; Sarli, G.; Verin, R.; Nicoloso, S.; Benazzi, C.; Galuppi, R. First description of nodular onchocercosis (Onchocerca jakutensis) in free-ranging Italian red deer (Cervus elaphus). J. Wildl. Dis. 2011, 47, 963–967. [Google Scholar] [CrossRef]
- Duscher, G.G.; Leschnik, M.; Fuehrer, H.P.; Joachim, A. Wildlife reservoirs for vector-borne canine, feline and zoonotic infections in Austria. Int. J. Parasitol. Parasites Wildl. 2014, 4, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Bosch, F.; Manzanell, R.; Mathis, A. First description of Onchocerca jakutensis (Nematoda: Filarioidea) in red deer (Cervus elaphus) in Switzerland. Int. J. Parasitol. Parasites. Wildl. 2016, 5, 192–197. [Google Scholar] [CrossRef]
- Latrofa, M.S.; Annoscia, G.; Colella, V.; Cavalera, M.A.; Maia, C.; Martin, C.; Slapeta, J.; Otranto, D. A real-time PCR tool for the surveillance of zoonotic Onchocerca lupi in dogs, cats and potential vectors. PLoS Negl. Trop. Dis. 2018, 12, ce0006402. [Google Scholar] [CrossRef]
- Rodonaja, T.E. A New Species of Nematode, Onchocerca lupi n. sp., from Canis Lupus Cubanensis; Soobshchenyia Akad Nauk Gruzinoskoy SSR: Tbilisi, Georgia, 1967; Volume 45, pp. 715–719. [Google Scholar]
- Sréter, T.; Széll, Z. Onchocercosis: A newly recognized disease in dogs. Vet. Parasitol. 2008, 151, 1–13. [Google Scholar] [CrossRef]
- Maia, C.; Annoscia, G.; Latrofa, M.S.; Pereira, A.; Giannelli, A.; Pedroso, L.; Otranto, D. Onchocerca lupi Nematode in Cat, Portugal. Emerg. Infect. Dis. 2015, 21, 2252–2254. [Google Scholar] [CrossRef]
- Otranto, D.; Sakru, N.; Testini, G.; Gürlü, V.P.; Yakar, K.; Lia, R.P.; Dantas-Torres, F.; Bain, O. Case report: First evidence of human zoonotic infection by Onchocerca lupi (Spirurida, Onchocercidae). Am. J. Trop. Med. Hyg. 2011, 84, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Széll, Z.; Sréter, T.; Erdélyi, I.; Varga, I. Ocular onchocercosis in dogs: Aberrant infection in an accidental host or lupi onchocercosis? Vet. Parasitol. 2001, 101, 115–125. [Google Scholar] [CrossRef]
- Komnenou, A.; Eberhard, M.L.; Kaldrymidou, E.; Tsalie, E.; Dessiris, A. Subconjunctival filariasis due to Onchocerca sp. in dogs: Report of 23 cases in Greece. Vet. Ophthalmol. 2002, 5, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Sréter-Lancz, Z.; Széll, Z.; Sréter, T. Molecular genetic comparison of Onchocerca sp. infecting dogs in Europe with other spirurid nematodes including Onchocerca lienalis. Vet. Parasitol. 2007, 148, 365–370. [Google Scholar] [CrossRef]
- Otranto, D.; Dantas-Torres, F.; Cebeci, Z.; Yeniad, B.; Buyukbabani, N.; Boral, O.B.; Gustinelli, A.; Mounir, T.; Mutafchiev, Y.; Bain, O. Human ocular filariasis: Further evidence on the zoonotic role of Onchocerca lupi. Parasites Vectors 2012, 5, 84. [Google Scholar] [CrossRef]
- Ilhan, H.D.; Yaman, A.; Morishima, Y.; Sugiyama, H.; Muto, M.; Yamasaki, H.; Hasegawa, H.; Lebe, B.; Bajin, M.S. Onchocerca lupi infection in Turkey: A unique case of a rare human parasite. Acta Parasitol. 2013, 58, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Mowlavi, G.; Farzbod, F.; Kheirkhah, A.; Mobedi, I.; Bowman, D.D.; Naddaf, S.R. Human ocular onchocerciasis caused by Onchocerca lupi (Spirurida, Onchocercidae) in Iran. J. Helminthol. 2014, 88, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Otranto, D.; Giannelli, A.; Latrofa, M.S.; Dantas-Torres, F.; Scotty Trumble, N.; Chavkin, M.; Kennard, G.; Eberhard, M.L.; Bowman, D.D. Canine Infections with Onchocerca lupi Nematodes, United States, 2011–2014. Emerg. Infect. Dis. 2015, 21, 868–871. [Google Scholar] [CrossRef] [PubMed]
- Miró, G.; Montoya, A.; Checa, R.; Gálvez, R.; Mínguez, J.J.; Marino, V.; Otranto, D. First detection of Onchocerca lupi infection in dogs in southern Spain. Parasites Vectors 2016, 9, 290. [Google Scholar]
- Achukwi, M.D.; Harnett, W.; Renz, A. Onchocerca ochengi transmission dynamics and the correlation of O. ochengi microfilaria density in cattle with the transmission potential. Vet. Res. 2000, 31, 611–621. [Google Scholar] [CrossRef]
- Bussieras, J.; Amagee, E.; Barn, O. Les onchocercoses des bovins togolais a O. dukei et O. dermata. Rev. Elev. Med. Vet. Pays. Trop. 1974, 27, 189–194. (In French) [Google Scholar] [CrossRef] [PubMed]
- Séchan, Y. Développement d’onchocerques animales chez le vecteur de l’onchocercose humaine Simulium sirbanum Vajime et Dunbar, 1975 (Diptera: Simuliidae) en zone subsahélienne du Mali, Afrique de L’Ouest. Cahiers. ORSTOM 1984, 178, 1–234. (In French) [Google Scholar]
- Doyle, S.R.; Armoo, S.; Renz, A.; Taylor, M.J.; Osei-Atweneboana, M.Y.; Grant, W.N. Discrimination between Onchocerca volvulus and O. ochengi filarial larvae in Simulium damnosum (s.l.) and their distribution throughout central Ghana using a versatile high-resolution speciation assay. Parasites Vectors 2016, 9, 536. [Google Scholar] [CrossRef] [PubMed]
- Verocai, G.G.; Hassan, H.K.; Lakwo, T.; Habomugisha, P.; Katabarwa, M.N.; Begumisa, S.; Clouds, P.; Katamanywa, J.; Nahabwe, C.; Unnasch, T.R. Molecular Identification of Onchocerca spp. Larvae in Simulium damnosum sensu lato Collected in Northern Uganda. Am. J. Trop. Med. Hyg. 2017, 97, 1843–1845. [Google Scholar] [CrossRef]
- Bain, O.; Wahl, G.; Renz, A. Onchocerca ramachandrini n. sp. from the warthog in Cameroon. Ann. Parasitol. Hum. Comp. 1993, 68, 139–143. [Google Scholar] [CrossRef]
- Moignoux, J.B. Culicoides nubeculosus Meig (Diptera Ceratopogonida), possible intermediary host of the filaria Onchocerca reticulata Dies. in Camargue. C R Hebd. Seances. Acad. Sci. 1951, 233, 102–103. [Google Scholar]
- Riek, R.F. A note on the occurrence of Onchocerca Reticulata Diesing 1841 in the horse in Queensland. Vet. J. 1954, 30, 178–181. [Google Scholar] [CrossRef]
- Moignoux, J.B. Experimental study of the evolutive cycle of microfilaria Onchocerca reticulata Dies. in Camargue. C R Seances. Soc. Biol. Fil. 1951, 145, 1572–1573. [Google Scholar]
- Yagi, K.; Bain, O.; Shoho, C. Onchocerca suzukii n. sp. and O. skrjabini (= O. tarsicola) from a relict bovid, Capricornis crispus, in Japan. Parasite 1994, 1, 349–356. [Google Scholar] [CrossRef][Green Version]
- Saeung, A.; Srisuka, W.; Aupalee, K.; Fukuda, M.; Otsuka, Y.; Taai, K.; Maleewong, W.; Takaoka, H. Natural infections with larvae of Onchocerca species type I in the human-biting black fly, Simulium nigrogilvum (Diptera: Simuliidae), in western Thailand. Acta Trop. 2020, 204, 105344. [Google Scholar] [CrossRef]
- Uni, S.; Fukuda, M.; Otsuka, Y.; Hiramatsu, N.; Yokobayashi, K.; Takahashi, H.; Murata, S.; Kusatake, K.; Morita, E.; Maruyama, H.; et al. New zoonotic cases of Onchocerca dewittei japonica (Nematoda: Onchocercidae) in Honshu, Japan. Parasites Vectors 2015, 8, 59. [Google Scholar] [CrossRef] [PubMed]
- Baranwal, V.K.; Shyamsundar, K.; Kabuyaya, V.; Biswas, J.; Vannadil, H. Study of onchocerciasis-related visual impairment in North Kivu province of the Democratic Republic of Congo in Africa. Indian J. Ophthalmol. 2020, 68, 890–894. [Google Scholar]
- Crainey, J.L.; Silva, T.R.; Encinas, F.; Marín, M.A.; Vicente, A.C.; Luz, S.L. The mitogenome of Onchocerca volvulus from the Brazilian Amazonia focus. Mem. Inst. Oswaldo Cruz. 2016, 111, 79–81. [Google Scholar] [CrossRef]
- Kazura, J.W. More Progress in Eliminating Transmission of Onchocerca volvulus and Wuchereria bancrofti in the Americas: A Portent of Global Eradication. Am. J. Trop. Med. Hyg. 2015, 93, 1128–1129. [Google Scholar] [CrossRef]
- Lagoro, D.K.; Arony, D.A. Nodding syndrome (NS) and Onchocerca Volvulus (OV) in Northern Uganda. Pan. Afr. Med. J. 2017, 28, 1. [Google Scholar] [CrossRef]
- Hendy, A.; Krüger, A.; Pfarr, K.; De Witte, J.; Kibweja, A.; Mwingira, U.; Dujardin, J.-C.; Post, R.; Colebunders, R.; O’Neill, S.; et al. The blackfly vectors and transmission of Onchocerca volvulus in Mahenge, south eastern Tanzania. Acta Trop. 2018, 181, 50–59. [Google Scholar] [CrossRef]
- Prince-Guerra, J.L.; Cama, V.A.; Wilson, N.; Thiele, E.A.; Likwela, J.; Ndakala, N.; Muzinga Wa Muzinga, J.; Ayebazibwe, N.; Ndjakani, Y.D.; Pitchouna, N.A.; et al. Comparison of PCR Methods for Onchocerca volvulus Detection in Skin Snip Biopsies from the Tshopo Province, Democratic Republic of the Congo. Am. J. Trop. Med. Hyg. 2018, 98, 1427–1434. [Google Scholar] [CrossRef] [PubMed]
- Hotterbeekx, A.; Namale Ssonko, V.; Oyet, W.; Lakwo, T.; Idro, R. Neurological manifestations in Onchocerca volvulus infection: A review. Brain Res. Bull. 2019, 145, 39–44. [Google Scholar] [CrossRef]
- Morales-Hojas, R. Molecular systematics of filarial parasites, with an emphasis on groups of medical and veterinary importance, and its relevance for epidemiology. Infect. Genet. Evol. 2009, 9, 748–759. [Google Scholar] [CrossRef]
- Chabaud, A.G.; Bain, O. The evolutionary expansion of the Spirurida. Int. J. Parasitol. 1994, 24, 1179–1201. [Google Scholar] [CrossRef]
- Bain, O. Le genre Onchocerca: Hypothèses sur son évolution et clé dichotomique des espèces. Ann. Parasitol. Hum. Comp. 1981, 56, 503–526. [Google Scholar] [CrossRef] [PubMed]
- Demiaszkiewicz, A.W.; Filip, K.J. Microfilariae Onchocerca alcis Bain et Rehbinder, 1986—A new parasite of moose Alces alces (L.) in Poland. Ann. Parasitol. 2018, 64, 89–91. [Google Scholar] [PubMed]
- Demiaszkiewicz, A.W. Redescription of Onchocerca jakutensis (gubanov, 11964) (nematoda, filarioidea). Acta Parasitol. 1993, 38, 124–127. [Google Scholar]
- Schulz-Key, H. Investigations on the filariidae of the cervidae in Southern Germany. 1. Development of the nodule, finding of the sexes and production of the microfilariae in Onchocerca flexuosa (Wedl, 1856), in the red deer (Cervus elaphus). Tropenmed. Parasitol. 1975, 26, 60–69. [Google Scholar]
- Pascoe, R.R.R.; Knottenbelt, D.C. Onchocercal dermatitis (onchocercosis microfilariasis). Manual Equine Dermatol. 1999, 141–143. [Google Scholar]
- Otranto, D.; Dantas-Torres, F.; Giannelli, A.; Abramo, F.; Ignjatovic Cupina, A.; Petric, D.; Cardoso, L.; Mutafchiev, Y.; Cortes, H. Cutaneous distribution and circadian rhythm of Onchocerca lupi microfilariae in dogs. PLoS Negl. Trop. Dis. 2013, 7, e2585. [Google Scholar] [CrossRef]
- Labelle, A.L.; Daniels, J.B.; Dix, M.; Labelle, P. Onchocerca lupi causing ocular disease in two cats. Vet. Ophthalmol. 2011, 14, 105–110. [Google Scholar] [CrossRef]
- Rodríguez-Pérez, M.A.; Unnasch, T.R.; Real-Najarro, O. Assessment and monitoring of onchocerciasis in Latin America. Adv. Parasitol. 2011, 77, 175–226. [Google Scholar]
- König, R.; Nassri, A.; Meindl, M.; Matuja, W.; Kidunda, A.R.; Siegmund, V.; Bretzel, G.; Löscher, T.; Jilek-Aall, L.; Schmutzhard, E.; et al. The role of Onchocerca volvulus in the development of epilepsy in a rural area of Tanzania. Parasitology 2010, 137, 1559–1568. [Google Scholar] [CrossRef]
- Foltz, J.L.; Makumbi, I.; Sejvar, J.J.; Malimbo, M.; Ndyomugyenyi, R.; Atai-Omoruto, A.D.; Alexander, L.N.; Abang, B.; Melstrom, P.; Kakooza, A.M.; et al. An Epidemiologic Investigation of Potential Risk Factors for Nodding Syndrome in Kitgum District, Uganda. PLoS ONE 2013, 8, e66419. [Google Scholar] [CrossRef] [PubMed]
- Dowell, S.F.; Sejvar, J.J.; Riek, L.; Vandemaele, K.A.; Lamunu, M.; Kuesel, A.C.; Schmutzhard, E.; Matuja, W.; Bunga, S.; Foltz, J.; et al. Nodding syndrome. Emerg. Infect. Dis. 2013, 19, 1374–1384. [Google Scholar] [CrossRef] [PubMed]
- McArthur, D.B. Emerging Infectious Diseases. Nurs. Clin. N. Am. 2019, 54, 297–311. [Google Scholar] [CrossRef]
- Bergua, A.; Hohberger, B.; Held, J.; Muntau, B.; Tannich, E.; Tappe, D. Human case of Onchocerca lupi infection, Germany, August 2014. Euro Surveill. 2015, 20, 21099. [Google Scholar] [CrossRef] [PubMed]
- Dudley, R.W.R.; Smith, C.; Dishop, M.; Mirsky, D.; Handler, M.H.; Rao, S. A cervical spine mass caused by Onchocerca lupi. Lancet 2015, 86, 1372. [Google Scholar] [CrossRef]
- Grácio, A.J.; Richter, J.; Komnenou, A.T.; Grácio, M.A. Onchocerciasis caused by Onchocerca lupi: An emerging zoonotic infection. Systematic review. Parasitol. Res. 2015, 114, 2401–2413. [Google Scholar] [CrossRef]
- Chen, T.; Moon, K.; DeMello, D.E.; Feiz-Erfan, I.; Theodore, N.; Bhardwaj, R.D. Case report of an epidural cervical Onchocerca lupi infection in a 13-year-old boy. J. Neurosurg. Pediatr. 2015, 16, 217–221. [Google Scholar] [CrossRef]
- Cantey, P.T.; Weeks, J.; Edwards, M.; Rao, S.; Ostovar, G.A.; Dehority, W.; Alzona, M.; Swoboda, S.; Christiaens, B.; Ballan, W.; et al. The Emergence of Zoonotic Onchocerca lupi Infection in the United States—A Case-Series. Clin. Infect. Dis. 2016, 62, 778–783. [Google Scholar] [CrossRef]
- Ishizawa, H.; Kato, S.; Nishimura, H.; Tanaka, R.; Fukuda, M.; Takaoka, H.; Tsutsumi, Y. Zoonotic onchocerciasis. Pathol. Int. 2015, 65, 271–273. [Google Scholar] [CrossRef]
- Fukuda, M.; Uni, S.; Otsuka, Y.; Eshita, Y.; Nakatani, J.; Ihara, K.; Yoshikawa, Y.; Goto, M.; Fujiwara, S.; Ramli, R.; et al. A new case of zoonotic onchocercosis in northern Kyushu, Japan. Parasitol. Int. 2015, 64, 519–521. [Google Scholar] [CrossRef]
- Uni, S.; Fukuda, M.; Ogawa, K.; Lim, Y.A.; Agatsuma, T.; Bunchom, N.; Saijuntha, W.; Otsuka, Y.; Bhassu, S.; Mat Udin, A.S.; et al. Zoonotic infection with Onchocerca dewittei japonica in an 11-year-old boy in Kansai Region, Western Honshu, Japan. Parasitol. Int. 2017, 66, 593–595. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, M.; Uni, S.; Igari, R.; Utsumi, Y.; Otsuka, Y.; Nakatani, J.; Uga, S.; Hará, T.; Hasegawa, H.; Takaoka, H. Human case of Onchocerca dewittei japonica infection in Fukushima, Northeastern Honshu, Japan. Parasitol. Int. 2019, 72, 101943. [Google Scholar] [CrossRef] [PubMed]
- Azarova, N.S.; Miretskii, O.I.; Sonin, M.D. The first instance of detection of nematode Onchocerca Diesing, 1841 in a person in the USSR. Med. Parazitol. 1965, 34, 156–158. [Google Scholar]
- Siegenthaler, R.; Gubler, R. Paraarticuläres Nematodengranulom (einheimische Onchocerca). Schweiz. Med. Wochenschr. 1965, 95, 1102–1104. [Google Scholar]
- Beaver, P.C.; Yoshimura, H.; Takayasu, S.; Hashimoto, H.; Little, M.D. Zoonotic Onchocerca in a Japanese Child. Am. J. Trop. Med. Hyg. 1989, 40, 298–300. [Google Scholar] [CrossRef] [PubMed]
- Pampiglione, S.; Vakalis, N.; Lyssimachou, A.; Kouppari, G.; Orihel, T.C. Subconjunctival zoonotic Onchocerca in an Albanian man. Ann. Trop. Med. Parasitol. 2001, 95, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Sallo, F.; Eberhard, M.L.; Fok, E.; Baska, F.; Hatvani, I. Zoonotic intravitreal Onchocerca in Hungary. Ophthalmology 2005, 112, 502–504. [Google Scholar] [CrossRef]
- Hira, P.R.; Al-Buloushi, A.; Khalid, N.; Iqbal, J.; Bain, O.; Eberhard, M.L. Zoonotic filariasis in the Arabian Peninsula: Autochthonous onchocerciasis and dirofilariasis. Am. J. Trop. Med. Hyg. 2008, 79, 739–741. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lai, J.H.; Walsh, N.M.; Pritt, B.S.; Sloan, L.; Gibson, L.E.; Desormeau, L.; Haldane, D.J. Cutaneous manifestations of a zoonotic Onchocerca species in an adult male, acquired in Nova Scotia, Canada. J. Clin. Microbiol. 2014, 52, 1768–1770. [Google Scholar] [CrossRef][Green Version]
- Beaver, P.C.; Horner, G.S.; Bilos, J.Z. Zoonotic onchocercosis in a resident of Illiois and observations on the identification of Onchocerca species. Am. J. Trop. Med. Hyg. 1974, 23, 595–607. [Google Scholar] [CrossRef]
- Ali-Khan, Z. Tissue pathology and comparative microanatomy of Onchocerca from a resident of Ontario and other enzootic Onchocerca species from Canada and the U.S.A. Ann. Trop. Med. Parasitol. 1977, 71, 469–482. [Google Scholar] [CrossRef]
- Takaoka, H.; Bain, O.; Tajimi, S.; Kashima, K.; Nakayama, I.; Korenaga, M.; Aoki, C.; Otsuka, Y. Second case of zoonotic Onchocerca infection in a resident of Oita in Japan. Parasite 1996, 3, 179–182. [Google Scholar] [CrossRef]
- Eberhard, M.L.; Ostovar, G.A.; Chundu, K.; Hobohm, D.; Feiz-Erfan, I.; Mathison, B.A.; Bishop, H.S.; Cantey, P.T. Case Report, Zoonotic Onchocerca lupi infection in a 22-month-old child in Arizona: First report in the United States and a review of the literature. Am. J. Trop. Med. Hyg. 2013, 88, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, H.; Yanagi, T.; Daa, T.; Anzai, S.; Aoki, C.; Fukuda, M.; Uni, S.; Bain, O. An Onchocerca species of wild boar found in the subcutaneous nodule of a resident of Oita, Japan. Parasitol. Int. 2005, 54, 91–93. [Google Scholar] [CrossRef]
- Uni, S.; Boda, T.; Daisaku, K.; Ikura, Y.; Maruyama, H.; Hasegawa, H.; Fukuda, M.; Takaoka, H.; Bain, O. Zoonotic filariasis caused by Onchocerca dewittei japonica in a resident of Hiroshima Prefecture, Honshu, Japan. Parasitol. Int. 2010, 59, 477–480. [Google Scholar] [CrossRef]
- Koehsler, M.; Soleiman, A.; Aspöck, H.; Auer, H.; Walochnik, J. Onchocerca jakutensis filariasis in humans. Emerg. Infect. Dis. 2007, 13, 1749–1752. [Google Scholar] [CrossRef]
- Tahir, D.; Davoust, B.; Parola, P. Vector-borne nematode diseases in pets and humans in the Mediterranean Basin: An update. Vet. World 2019, 12, 1630–1643. [Google Scholar] [CrossRef]
- Hoberg, E.P.; Brooks, D.R. Evolution in action: Climate change, biodiversity dynamics and emerging infectious disease. Philos. Trans. R. Soc. B 2015, 370, 20130553. [Google Scholar] [CrossRef]
- Sréter, T.; Széll, Z.; Egyed, Z.; Varga, I. Subconjunctival zoonotic onchocerciasis in man: Aberrant infection with Onchocerca lupi? Ann. Trop. Med. Parasitol. 2002, 96, 497–502. [Google Scholar] [CrossRef]
- Osei-Atweneboana, M.Y.; Eng, J.K.; Boakye, D.; Gyapong, J.O.; Prichard, R.K. Prevalence and intensity of Onchocerca volvulus infection and efficacy of ivermectin in endemic communities in Ghana: A two-phase epidemiological study, 2021–2029. Lancet 2007, 369, 2021–2029. [Google Scholar] [CrossRef]
- Remme, J.; Dadzie, K.Y.; Rolland, A.; Thylefors, B. Ocular onchocerciasis and intensity of infection in the community. I. West African savanna. Trop. Med. Parasitol. 1989, 40, 340–347. [Google Scholar]
- Dadzie, K.Y.; Remme, J.; Baker, R.H.; Rolland, A.; Thylefors, B. Ocular onchocerciasis and intensity of infection in the community. III. West African rainforest foci of the vector Simulium sanctipauli. Trop. Med. Parasitol. 1990, 41, 376–382. [Google Scholar] [PubMed]
- Crump, A.; Morel, C.M.; Omura, S. The onchocerciasis chronicle: From the beginning to the end? Trends Parasitol. 2012, 28, 280–288. [Google Scholar] [CrossRef]
- Pearlman, E. Immunopathogenesis of Onchocerca volvulus keratitis (river blindness): A novel role for endosymbiotic Wolbachia bacteria. Med. Microbiol. Immunol. 2003, 192, 57–60. [Google Scholar] [CrossRef]
- Hawryluk, N.A. Macrofilaricides: An Unmet Medical Need for Filarial Diseases. ACS Infect. Dis. 2020, 6, 662–671. [Google Scholar] [CrossRef]
- Vinkeles Melchers, N.V.S.; Mollenkopf, S.; Colebunders, R.; Edlinger, M.; Coffeng, L.E.; Irani, J.; Zola, T.; Siewe, J.N.; de Vlas, S.J.; Winkler, A.S.; et al. Burden of onchocerciasis-associated epilepsy: First estimates and research priorities. Infect. Dis. Poverty 2018, 7, 101. [Google Scholar] [CrossRef]
- Colebunders, R.; Tepage, F.; Rood, E.; Mandro, M.; Abatih, E.N.; Musinya, G.; Mambandu, G.; Kabeya, J.; Komba, M.; Levick, B.; et al. Prevalence of River Epilepsy in the Orientale Province in the Democratic Republic of the Congo. PLoS Negl. Trop. Dis. 2016, 10, 1–8. [Google Scholar] [CrossRef]
- Chesnais, C.; Nana-Djeunga, H.C.; Njamnshi, A.K.; Lenou-Nanga, C.G.; Boullé, C.; Zoung-Kanyi Bissek, A.C.; Kamgno, J.; Colebunders, R.; Boussinesq, M. The temporal relationship between onchocerciasis and epilepsy: A prospective population-based cohort study. Lancet Infect. Dis. 2018, 18, 1278–1286. [Google Scholar] [CrossRef]
- Colebunders, R.; Carter, J.Y.; Olore, P.C.; Puok, K.; Bhattacharyya, S.; Menon, S.; Abd-Elfarag, G.; Ojok, M.; Ensoy-Musoro, C.; Lako, R.; et al. High prevalence of onchocerciasis-associated epilepsy in villages in Maridi County, Republic of South Sudan: A community-based survey. Seizures 2018, 63, 93–101. [Google Scholar] [CrossRef]
- Gumisriza, N.; Kaiser, C.; Asaba, G.; Onen, H.; Mubiru, F.; Kisembo, D.; Siewe Fodjo, J.N.; Colebunders, R. Changes in epilepsy burden after onchocerciasis elimination in a hyperendemic focus of western Uganda: A comparison of two population-based, cross-sectional studies. Lancet Infect. Dis. 2020, 20, 30122–30125. [Google Scholar]
- Chesnais, C.B.; Bizet, C.; Campillo, J.T.; Njamnshi, W.Y.; Bopda, J.; Nwane, P.; Pion, S.D.; Njamnshi, A.K.; Boussinesq, M. A Second Population-Based Cohort Study in Cameroon Confirms the Temporal Relationship Between Onchocerciasis and Epilepsy. Open Forum Infect. Dis. 2020, 7, ofaa206. [Google Scholar] [CrossRef]
- Hotterbeekx, A.; Lammens, M.; Idro, R.; Akun, P.R.; Lukande, R.; Akena, G.; Nath, A.; Taylor, J.; Olwa, F.; Kumar-Singh, S.; et al. Neuroinflammation and Not Tauopathy Is a Predominant Pathological Signature of Nodding Syndrome. J. Neuropathol. Exp. Neurol. 2019, 78, 1049–1058. [Google Scholar] [CrossRef]
- Rodger, F.C. The pathogenesis and pathology of ocular onchocerciasis. Am. J. Ophthalmol. 1960, 49, 104–135. [Google Scholar] [CrossRef]
- Johnson, T.P.; Tyagi, R.; Lee, P.R.; Lee, M.H.; Johnson, K.R.; Kowalak, J.; Elkahloun, A.; Medynets, M.; Hategan, A.; Kubofcik, J.; et al. Nodding syndrome may be an autoimmune reaction to the parasitic worm Onchocerca volvulus. Sci. Transl. Med. 2017, 9, eaaf6953. [Google Scholar] [CrossRef]
- Mellor, P.S.; Boorman, J. Multiplication of bluetongue virus in culicoides nubeculosus (Meigen) simultaneously infected with the virus and the microfilariae of Onchocerca cervicalis (Railliet Henry). Ann. Trop. Med. Parasitol. 1980, 74, 463–469. [Google Scholar]
- Johnson, T.P.; Sejvar, J.; Nutman, T.B.; Nath, A. The Pathogenesis of Nodding Syndrome. Annu. Rev. Pathol. 2020, 15, 395–417. [Google Scholar] [CrossRef]
- Hotterbeekx, A.; Raimon, S.; Abd-Elfarag, G.; Carter, Y.J.; Sebit, W.; Suliman, A.; Nelson Siewe Fodjo, J.; De Witte, P.; Yibi Logora, M.; Colebunders, R.; et al. Onchocerca volvulus is not detected in the cerebrospinal fluid of persons with onchocerciasis-associated epilepsy. Int. J. Infect. Dis. 2019, 91, 119–123. [Google Scholar] [CrossRef]
- Siewe Fodjo, J.N.; Mandro, M.; Mukendi, D.; Tepage, F.; Menon, S.; Nakato, S.; Nyisi, F.; Abhafule, G.; Wonya’rossi, D.; Anyolito, A.; et al. Onchocerciasis-associated epilepsy in the Democratic Republic of Congo: Clinical description and relationship with microfilarial density. PLoS Negl. Trop. Dis. 2019, 13, e0007300. [Google Scholar] [CrossRef]
- Idro, R.; Opar, B.; Wamala, J.; Abbo, C.; Onzivua, S.; Amos Mwaka, D.; Kakooza-Mwesige, A.; Mbonye, A. Is nodding syndrome an Onchocerca volvulus-induced neuroinflammatory disorder? Uganda’s story of research in understanding the disease. Int. J. Infect. Dis. 2016, 45, 112–117. [Google Scholar] [CrossRef]
- Colebunders, R.; Hendy, A.; Nanyunja, M.; Wamala, J.F.; van Oijen, M. Nodding syndrome-a new hypothesis and new direction for research. Int. J. Infect. Dis. 2014, 27, 74–77. [Google Scholar] [CrossRef]
- Saint Andre, A.; Blackwell, N.M.; Hall, L.R.; Hoerauf, A.; Brattig, N.W.; Volkmann, L.; Taylor, M.J.; Ford, L.; Hise, A.G.; Lass, J.H.; et al. The role of endosymbiotic Wolbachia bacteria in the pathogenesis of river blindness. Science 2002, 295, 1892–1895. [Google Scholar] [CrossRef]
| Onchocerca Species | Vectors | Hosts | Geographical Distribution |
|---|---|---|---|
| O. armillata | Unknown [10] | Cattle [11], buffaloes [11], dromedaries [12], goats [11] | Ghana [13], Iran [14], Senegal [15], Sudan [12], Sierra Leona [16], Tanzania [17], Nigeria [3], Cameroon [10], India [18] |
| O. boehmi | Unknown [19] | Horses [19] | Austria [20], Iran [21], Italy [19] |
| O. cervicalis | Culicoides nubeculosus [22], C. variipennis [23] | Horses [24], ponies [25], humans [26], donkeys [27] | Australia [24], Japan [28], United Kingdom [22], Holland [29], Canada [30], United States [31], Brazil [32], Poland [33], Spain [33], Egypt [27] |
| O. cervipedis | Simulium venustum [34] | White-tailed deer [35], black-tailed deer [35], moose [9], caribou [9] | Canada [35], Costa Rica [36], United States [9], Alaska [9] |
| O. dewittei dewittei | Unknown | Wild boar [37] | Malaysia [37] |
| O. dewittei japonica | Simulium bidentatum [38], S. arakawae, S. japonicum, S. oitanum, S. quinquestriatum, S. rufibasis [39] | Wild boar [40], humans [41] | Japan [40] |
| O. dukei | Simulium hargreavesi, S. vorax, S. damnosum s.l. [42] | Cattle [43] | Zambia [44], Togo [45], Cameroon [43] |
| O. eberhardi | Simulium arakawae, S. oitanum, S. bidentatum [46] | Sika deer [47] | Japan [47] |
| O. fasciata | Culicoides puncticollis [48] | Dromedaries [49], camels [50] | Sudan [51], Somalia [51], Jordan [52], Saudi Arabia [53], Iran [54], China [55], Mongolia [48] |
| O. flexuosa | Prosimulium nigripes, Simulium ornatum [56] | Antelope [57], reed deer [58], Roe deer [59] | Uganda [60], Germany [61], Spain [8], Sweden [5], Slovakia [62] |
| O. gutturosa | Culicoides spp., C. kingi [63] | Cattle [43], camels [64], dromedaries [12], horses [65], humans [66] | Iran [14], Senegal [15], Australia [64], Togo [45], Sudan [12], Sierra Leona [16], Cameroon [43], Turkey [67], India [18] |
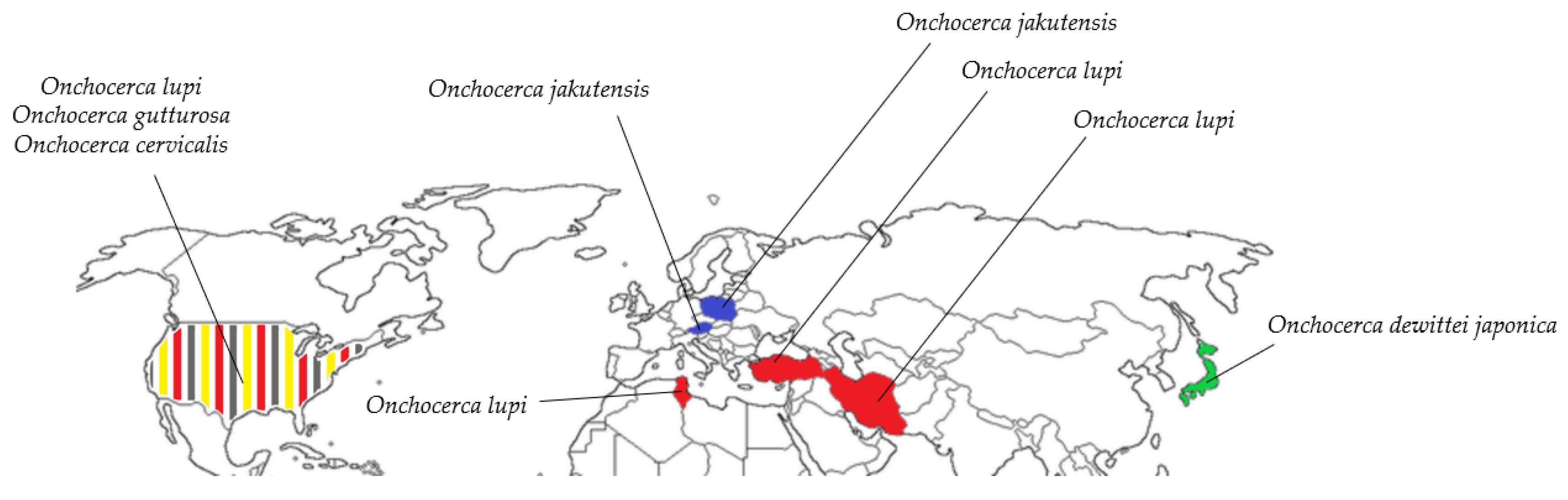
| O. jakutensis | Unknown | Red deer [58], humans [68] | Germany [53], Italy [69], Austria [70], Switzerland [71], Poland [68] |
| O. lupi | Simulium sp. [72], S. tribulatum [7] | Wolf [73], Dogs [74], Cats [75], Humans [76] | Georgia [73], Germany [77], Greece [78], Hungary [79], Tunis [80], Turkey [81], Iran [82], Portugal [75], United States [83], Spain [84] |
| O. ochengi | S. damnosum s.l. [85] | Cattle [43] | Burkina Faso [86], Senegal [15], Mali [87], Sierra Leona [16], Togo [45], Cameroon [43], Ghana [88] |
| O. ramachandrini | Simulium damnosum s.l. [89] | Warthogs [90] | Cameroon [90], Uganda [89] |
| O. reticulata | Culicoides nubeculosus Meig [91] | Horses [92], donkeys [27] | France [93], Australia [92], United States [65], Egypt [27] |
| O. skrjabini | Simulium arakawae, S. oitanum, S. bidentatum [46], S. japonicum, Prosimulium sp. [28] | Japanese serow [94], sika deer [47] | Japan [47,94] |
| Onchocerca spp. type I | Simulium bidentatum [38], S. sigrogilvum [95] | Wild boar [38], cattle [95] | Japan [38], Thailand [95] |
| O. suzukii | Simulium japonicum, Prosimulium sp. [28] | Japanese serow [94] | Japan [94] |
| O. takaokai | Simulium bidentatum [96] | Wild boar [96] | Japan [96] |
| O. volvulus | Simulium sp. [97] | Humans [97] | Brazil [98], Guatemala [99], Uganda [100], Tanzania [101], Democratic Republic of the Congo [102], Yemen [103] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cambra-Pellejà, M.; Gandasegui, J.; Balaña-Fouce, R.; Muñoz, J.; Martínez-Valladares, M. Zoonotic Implications of Onchocerca Species on Human Health. Pathogens 2020, 9, 761. https://doi.org/10.3390/pathogens9090761
Cambra-Pellejà M, Gandasegui J, Balaña-Fouce R, Muñoz J, Martínez-Valladares M. Zoonotic Implications of Onchocerca Species on Human Health. Pathogens. 2020; 9(9):761. https://doi.org/10.3390/pathogens9090761
Chicago/Turabian StyleCambra-Pellejà, Maria, Javier Gandasegui, Rafael Balaña-Fouce, José Muñoz, and María Martínez-Valladares. 2020. "Zoonotic Implications of Onchocerca Species on Human Health" Pathogens 9, no. 9: 761. https://doi.org/10.3390/pathogens9090761
APA StyleCambra-Pellejà, M., Gandasegui, J., Balaña-Fouce, R., Muñoz, J., & Martínez-Valladares, M. (2020). Zoonotic Implications of Onchocerca Species on Human Health. Pathogens, 9(9), 761. https://doi.org/10.3390/pathogens9090761






