Clinical Course and Gross Pathological Findings in Wild Boar Infected with a Highly Virulent Strain of African Swine Fever Virus Genotype II
Abstract
:1. Introduction
2. Results
2.1. Clinical Evaluation
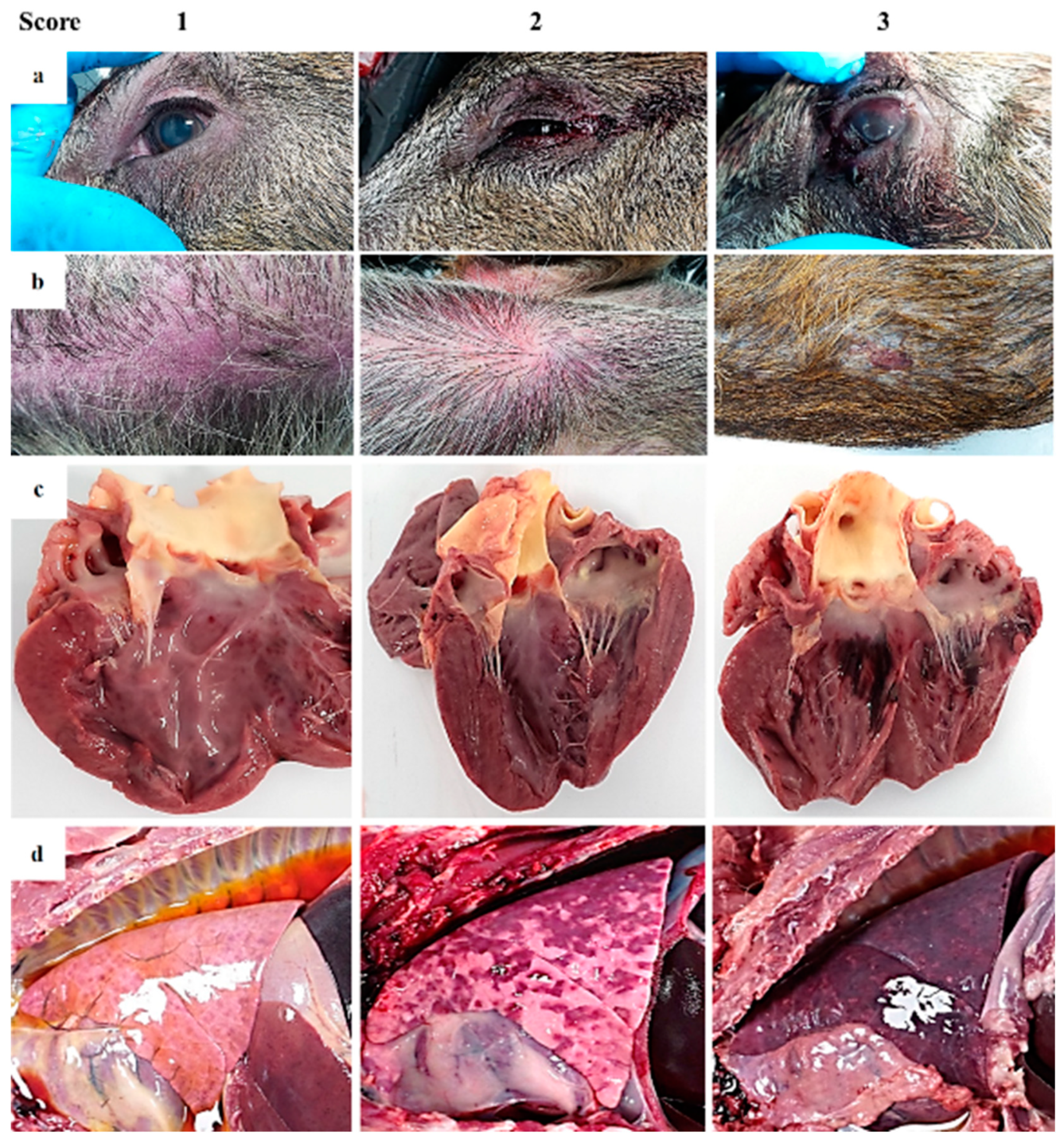
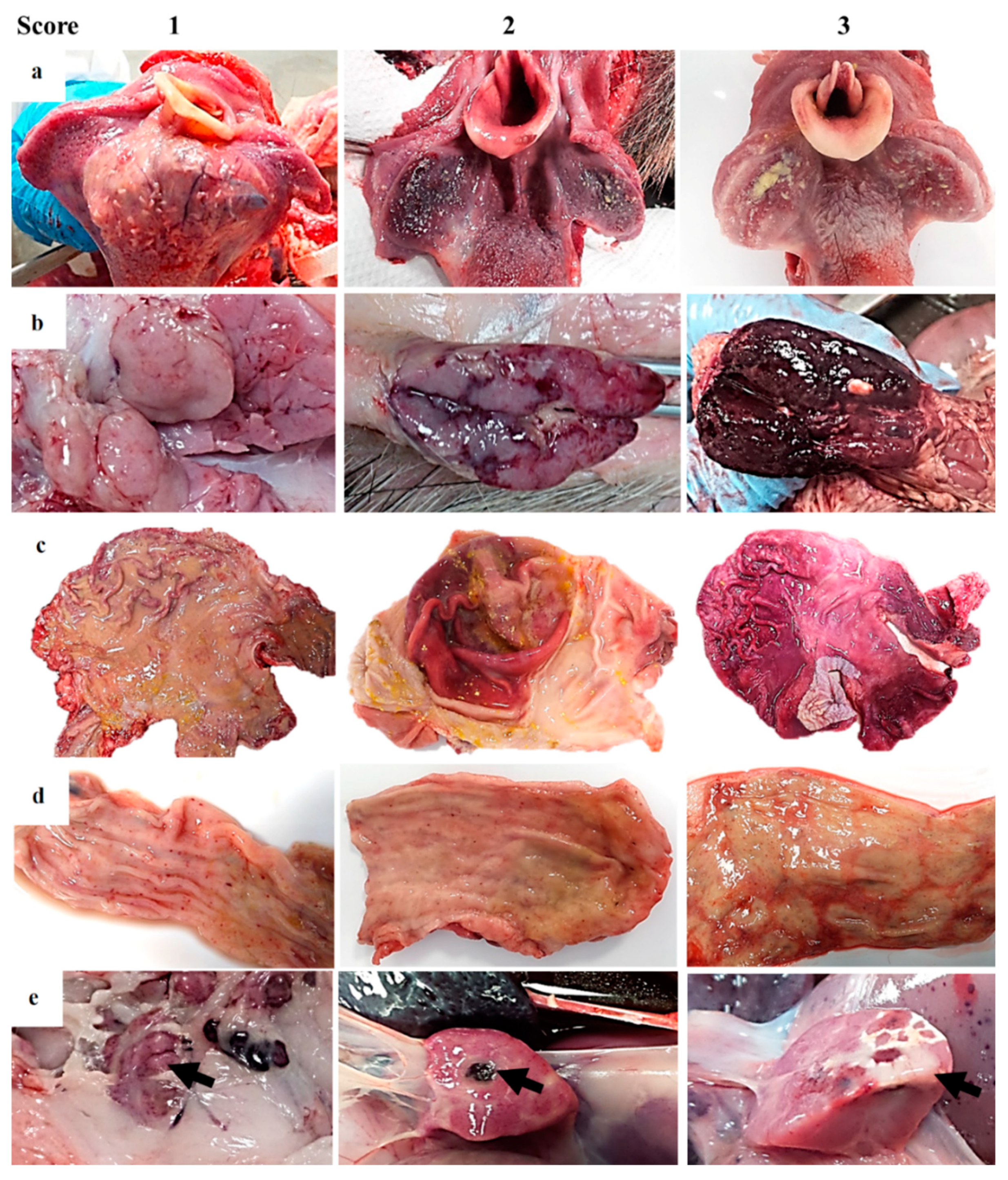
2.2. Postmortem Evaluation
2.3. Tissue Sample Analysis
3. Discussion
4. Materials and Methods
4.1. Virus
4.2. Animals
4.3. Study Design
4.4. Clinical Evaluation
4.5. Postmortem Evaluation
4.6. Tissue Sample Collection and Analysis
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Arias, M.; Jurado, C.; Gallardo, C.; Fernández-Pinero, J.; Sánchez-Vizcaíno, J.M. Gaps in African swine fever: Analysis and priorities. Transbound. Emerg. Dis. 2018, 65, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Cordón, P.J.; Montoya, M.; Reis, A.L.; Dixon, L.K. African swine fever: A re-emerging viral disease threatening the global pig industry. Vet. J. 2018, 233, 41–48. [Google Scholar] [CrossRef] [PubMed]
- OIE WAHIS. World Animal Health Information System. Available online: http://www.oie.int/wahis_2/public/wahid.php/Diseaseinformation/reportarchive (accessed on 15 July 2020).
- Zimmerman, J.F.; Karriker, L.A.; Ramírez, A.; Schwartz, K.J.; Stevenson, G.W. Diseases of Swine, 10th ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2012; pp. 396–404. [Google Scholar]
- Sánchez-Vizcaíno, J.M.; Mur, L.; Gomez-Villamandos, J.C.; Carrasco, L. An update on the epidemiology and pathology of African swine fever. J. Comp. Pathol. 2015, 152, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Pikalo, J.; Schoder, M.-E.; Sehl, J.; Breithaupt, A.; Tignon, M.; Cay, A.B.; Gager, A.M.; Fisher, M.; Beer, M.; Blome, S. The African swine fever virus isolate Belgium 2018/1 shows high virulence in European wild boar. Transbound. Emerg. Dis. 2020, 67, 1654–1659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Massei, G.; Kindberg, J.; Licoppe, A.; Gačić, D.; Šprem, N.; Kamler, J.; Baubet, E.; Hohmann, U.; Monaco, A.; Ozoliņš, J.; et al. Wild boar populations up, numbers of hunters down? A review of trends and implications for Europe. Pest Manag. Sci. 2015, 71, 492–500. [Google Scholar] [CrossRef]
- Olesen, A.S.; Lohse, L.; Hansen, M.F.; Boklund, A.; Halasa, T.; Belsham, G.J.; Rasmussen, T.B.; Bøtner, A.; Bødker, R. Infection of pigs with African swine fever virus via ingestion of stable flies (Stomoxys calcitrans). Transbound. Emerg. Dis. 2018, 65, 1152–1157. [Google Scholar] [CrossRef] [Green Version]
- Probst, C.; Globig, A.; Knoll, B.; Conraths, F.J.; Depner, K. Behaviour of free ranging wild boar towards their dead fellows: Potential implications for the transmission of African swine fever. R. Soc. Open Sci. 2017, 4, 170054. [Google Scholar] [CrossRef] [Green Version]
- Guinat, C.; Gogin, A.; Blome, S.; Keil, G.; Pollin, R.; Pfeiffer, D.U.; Dixon, L. Transmission routes of African swine fever virus to domestic pigs: Current knowledge and future research directions. Vet. Rec. 2016, 178, 262–267. [Google Scholar] [CrossRef] [Green Version]
- Nurmoja, I.; Mõtus, K.; Kristian, M.; Niine, T.; Schulz, K.; Depner, K.; Viltrop, A. Epidemiological analysis of the 2015–2017 African swine fever outbreaks in Estonia. Prev. Vet. Med. 2018, 181, 104556. [Google Scholar] [CrossRef]
- Cadenas-Fernández, E.; Sánchez-Vizcaíno, J.M.; Pintore, A.; Denurra, D.; Cherchi, M.; Jurado, C.; Vicente, J.; Barasona, J.A. Free-ranging domestic pig and wild boar interactions in an endemic area of African swine fever. Front. Vet. Sci. 2019, 6, 376. [Google Scholar] [CrossRef]
- Chenais, E.; Ståhl, E.; Guberti, V.; Depner, K. Identification of wild boar–habitat epidemiologic cycle in African swine fever epizootic. Emerg. Infect. Dis. 2018, 24, 810–812. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barasona, J.A.; Gallardo, C.; Cadenas-Fernández, E.; Jurado, C.; Rivera, B.; Rodríguez-Bertos, A.; Arias, M.; Sánchez-Vizcaíno, J.M. First oral vaccination of Eurasian wild boar against African swine fever virus genotype II. Front. Vet. Sci. 2019, 6, 137. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, C.; Sánchez, E.G.; Pérez-Núñez, D.; Nogal, M.; de León, P.; Carrascosa, A.L.; Nieto, R.; Soler, A.; Arias, M.L.; Revilla, Y. African swine fever virus (ASFV) protection mediated by NH/P68 and NH/P68 recombinant live-attenuated viruses. Vaccine 2018, 36, 2694–2704. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, C.; Nieto, R.; Soler, A.; Pelayo, V.; Fernández-Pinero, J.; Markowska-Daniel, I.; Pridotkas, G.; Nurmoja, I.; Granta, R.; Simón, A.; et al. Assessment of African swine fever diagnostic techniques as a response to the epidemic outbreaks in eastern European Union countries: How to improve surveillance and control programs. J. Clin. Microbiol. 2015, 53, 2555–2565. [Google Scholar] [CrossRef] [Green Version]
- Sunwoo, S.-Y.; Pérez-Núñez, D.; Morozov, I.; Sánchez, E.; Gaudreault, N.; Trujillo, J.; Mur, L.; Nogal, M.; Madden, D.; Urbaniak, K.; et al. DNA-protein vaccination strategy does not protect from challenge with African swine fever virus Armenia 2007 strain. Vaccines 2019, 7, 12. [Google Scholar] [CrossRef] [Green Version]
- Razmaite, V.; Kerziene, S.; Jatkauskiene, V. Body and carcass measurements and organ weights of Lithuaninan indigenous pigs and their wild boar hybrids. Anim. Sci. Pap. Rep. 2009, 27, 331–342. [Google Scholar]
- Gabriel, C.; Blome, S.; Malogolovkin, A.; Parilov, S.; Kolbasov, D.; Teifke, J.P.; Beer, M. Characterization of African swine fever virus Caucasus isolate in European wild boars. Emerg. Infect. Dis. 2011, 17, 2342–2345. [Google Scholar] [CrossRef]
- Sánchez-Cordón, P.J.; Nunez, A.; Neimanis, A.; Wikström-Lassa, E.; Montoya, M.; Crooke, H.; Gavier-Widén, D. African swine fever: Disease dynamics in wild boar experimentally infected with ASFV isolates belonging to genotype I and II. Viruses 2019, 11, 852. [Google Scholar] [CrossRef] [Green Version]
- Blome, S.; Gabriel, C.; Beer, M. Pathogenesis of African swine fever in domestic pigs and European wild boar. Virus Res. 2013, 173, 122–130. [Google Scholar] [CrossRef]
- Nurmoja, I.; Petrov, A.; Breidenstein, C.; Zani, L.; Forth, J.H.; Beer, M.; Kristian, M.; Viltrop, A.; Blome, S. Biological characterization of African swine fever virus genotype II strains from north-eastern Estonia in European wild boar. Transbound. Emerg. Dis. 2017, 64, 2034–2041. [Google Scholar] [CrossRef]
- Pietschmann, J.; Guinat, C.; Beer, M.; Pronin, V.; Tauscher, K.; Petrov, A.; Keil, G.; Blome, S. Course and transmission characteristics of oral low-dose infection of domestic pigs and European wild boar with a Caucasian African swine fever virus isolate. Arch. Virol. 2015, 160, 1657–1667. [Google Scholar] [CrossRef] [PubMed]
- Olesen, A.S.; Lohse, L.; Boklund, A.; Halasa, T.; Gallardo, C.; Pejsak, Z.; Belsham, G.J.; Rasmussen, T.B.; Bøtner, A. Transmission of African swine fever virus from infected pigs by direct contact and aerosol routes. Vet. Microbiol. 2017, 211, 92–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zani, L.; Forth, J.H.; Forth, L.; Nurmoja, I.; Leidenberger, S.; Henke, J.; Carlson, J.; Breidenstein, C.; Viltrop, A.; Höper, D.; et al. Deletion at the 5′-end of Estonian ASFV strains associated with an attenuated phenotype. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Llario, P.; Parra, A.; Cerrato, R.; Hermosa de Mendoza, J. Spleen size variations and reproduction in a Mediterranean population of wild boar (Sus scrofa). Eur. J. Wild Res. 2004, 50, 13–17. [Google Scholar]
- Barasona, J.; López-Olvera, J.; Beltrán-Beck, B.; Gortázar, C.; Vicente, J. Trap-effectiveness and response to tiletamine-zolazepam and medetomidine anaesthesia in Eurasian wild boar captured with cage and corral traps. BMC Vet. Res. 2013, 9, 107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salguero, F.J. Comparative pathology and pathogenesis of African Swine Fever infection in swine. Front. Vet. Sci. 2020, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cadenas-Fernández, E.; Sánchez-Vizcaíno, J.M.; Kosowska, A.; Rivera, B.; Mayoral-Alegre, F.; Rodríguez-Bertos, A.; Yao, J.; Bray, J.; Lokhandwala, S.; Mwangi, W.; et al. Adenovirus-vectored African swine fever virus antigens cocktail is not protective against virulent Arm07 isolate in Eurasian wild boar. Pathogens 2020, 9, 171. [Google Scholar] [CrossRef]
- Galindo-Cardiel, I.; Ballester, M.; Solanes, D.; Nofrarías, M.; López-Soria, S.; Argilaguet, J.M.; Lacasta, A.; Accensi, F.; Rodríguez, F.; Segalés, J. Standardization of pathological investigations in the framework of experimental ASFV infections. Virus Res. 2013, 173, 180–190. [Google Scholar] [CrossRef]
- Maxie, G. Jubb, Kennedy & Palmer’s Pathology of Domestic Animals, 6th ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2015. [Google Scholar]
- King, D.P.; Reid, S.M.; Hutchings, G.H.; Grierson, S.S.; Wilkinson, P.J.; Dixon, L.K.; Bastos, A.D.; Drew, T.W. Development of a TaqMan® PCR assay with internal amplification control for the detection of African swine fever virus. J. Virol. Methods 2003, 107, 53–61. [Google Scholar] [CrossRef]



| Tissue/Pathological Finding | n Animals Affected | ASFV CT Values 1 | |||||
|---|---|---|---|---|---|---|---|
| 0 | Score 1 | Score 2 | Score 3 | IM | In-Contact | ||
| External examination | |||||||
| Body condition | 0/17 | 14/17 | 3/17 | 0/17 | |||
| Eyes/conjunctiva | 0/17 | 8/17 | 4/17 | 5/17 | |||
| Skin | 0/17 | 4/17 | 12/17 | 1/17 | |||
| Thoracic cavity | |||||||
| Cardiorespiratory system | Thoracic effusion | 0/17 | 3/17 | 6/17 | 8/17 | ||
| Heart | 25 ± 7 | 23 ± 3 | |||||
| a) Hydropericardium | 0/17 | 9/17 | 6/17 | 2/17 | |||
| b) Cardiac muscles | 2/17 | 9/17 | 3/17 | 3/17 | |||
| c) Edematous valve | 0/17 | 9/17 | 6/17 | 2/17 | |||
| Lung | 22 ± 3 | 22 ± 2 | |||||
| a) Collapse | 0/17 | 0/17 | 5/17 | 12/17 | |||
| b) Congestion/hemorrhage | 0/17 | 11/17 | 3/17 | 3/17 | |||
| c) Edema | 0/17 | 1/17 | 8/17 | 8/17 | |||
| d) Pneumonia | 0/17 | 7/17 | 6/17 | 4/17 | |||
| e) Pleura | 4/17 | 9/17 | 2/17 | 2/17 | |||
| Abdominal cavity | |||||||
| Ascites (yellowish/reddish fluid) | 0/17 | 7/17 | 7/17 | 3/17 | |||
| Alimentary system | Stomach | 0/17 | 10/17 | 5/17 | 2/17 | ||
| Small intestine | 0/17 | 3/17 | 12/17 | 2/17 | |||
| Large intestine | 0/17 | 6/17 | 7/17 | 4/17 | |||
| Liver | 0/17 | 2/17 | 11/17 | 4/17 | 23 ± 7 | 20 ± 2 | |
| Gallbladder | 0/17 | 7/17 | 10/17 | 0/17 | |||
| Pancreas | 1/17 | 7/17 | 6/17 | 3/17 | |||
| Urinary system | Kidney (hemorrhages) | 24 ± 5 | 22 ± 2 | ||||
| a) Medullo-cortical pattern | 0/17 | 5/17 | 8/17 | 4/17 | |||
| b) Cortical-medullar pattern | 0/17 | 7/17 | 8/17 | 2/17 | |||
| Urinary bladder | 1/17 | 10/17 | 6/17 | 0/17 | 26 ± 6 | 26 ± 3 | |
| Lymphoid system | Spleen | 0/17 | 0/17 | 4/17 | 13/17 | 22 ± 6 | 20 ± 4 |
| Lymph nodes | 25 ± 5 2 | 23 ± 2 2 | |||||
| a) Lymphadenomegaly | 0/17 | 0/17 | 1/17 | 16/17 | |||
| b) Congestion/hemorrhages | 0/17 | 3/17 | 4/17 | 10/17 | |||
| c) Affected lymph nodes | 0/17 | 1/17 | 5/17 | 11/17 | |||
| Tonsils | 0/17 | 5/17 | 5/17 | 7/17 | 25 ± 8 | 22 ± 7 | |
| Bone marrow | 0/17 | 17/17 | 0/17 | 0/17 | 24 ± 8 | 22 ± 4 | |
| Endocrine system—Adrenal glands | 4/17 | 8/17 | 3/17 | 2/17 | |||
| Central nervous system—Brain | 0/17 | 6/17 | 9/17 | 2/17 | 25 ± 7 | 23 ± 2 | |
| Blood | 23 ± 7 | 18 ± 2 | |||||
| Tissue/Pathological Finding | Score | |||
|---|---|---|---|---|
| 1 (Slight) | 2 (Moderate) | 3 (Intense) | ||
| External examination | ||||
| Body condition | Slightly thin | Thin | Very thin | |
| Eyes/conjunctiva | Slight congestion | Congestion, swelling of eyelids and serosanguinous ocular discharge | Congested ocular mucosa with hemorrhages, inflamed eyelids and hyphemia | |
| Skin | Areas with skin erythema (ears, flanks, abdomen) | Erythema of the skin, multiple hemorrhages | Erythema of the skin, multifocal to coalescent hemorrhages. Subcutaneous edema. Occasionally necrotic areas | |
| Thoracic cavity | ||||
| Cardiorespiratory system | Thoracic effusion | Moderate hydrothorax (with serosanguinous fluid) | Severe hydrothorax | Hemothorax |
| Heart | ||||
| a) Hydropericardium | Slight | Moderate with fibrinous adhesions | Intense with fibrinous adhesions | |
| b) Cardiac muscles | Petechiae on epicardium | Multifocal hemorrhages on the cardiac muscle and valvular endocardium | Extensive coalescent hemorrhages on the cardiac muscle and valvular endocardium | |
| c) Edematous valve | Slight thickening of the endocardium of the atrioventricular valves | Moderate thickening of the endocardium of the atrioventricular valves | Severe thickening of the endocardium of the atrioventricular and semilunar valves | |
| Lung | ||||
| a) Collapse | Mild lack of collapse with no rib impressions | Moderate lack of collapse with mild or scarce rib impressions | Marked presence of foamy material in the trachea and intense distension of interlobular walls | |
| b) Congestion or hemorrhage | Mild diffuse/patchy congestion of parenchyma. No hemorrhages | Variable degree of congestion. Multifocal to coalescent randomly distributed petechiae or ecchymosis | Variable degree of congestion. Multifocal to coalescent random hemorrhages in pulmonary parenchyma | |
| c) Edema | Scarce or no presence of foamy material in trachea/bronchus (alveolar edema) and minimal (interstitial edema) | Mild to moderate presence of foamy material in trachea/bronchus and moderate distension of interlobular walls | Marked presence of foamy material in trachea/bronchus and intense distension of interlobular walls | |
| d) Pneumonia | Minimal to mild cranio-ventral (uni/bilateral) consolidation (bronchopneumonia) | Moderate cranio-ventral (uni/bilateral) consolidation (bronchopneumonia) | Marked-extended cranio-ventral (uni/bilateral) consolidation (bronchopneumonia) (with red and grey hepatization of lobules) | |
| e) Pleura | Fibrinous adhesions | Focal fibrinous pleuritis/pleural adhesions | Diffuse fibrinous pleuritis with or without pleural edema | |
| Abdominal cavity | ||||
| Alimentary system | Ascites (yellowish/reddish fluid) | Slight | Moderate | Intense |
| Stomach | Acute catarrhal gastritis | Acute catarrhal gastritis with numerous petechiae/ecchymosis on the surface | Hemorrhagic gastritis (more than 50% affected) with occasional erosions | |
| Small intestine 1 | Acute catarrhal enteritis with some petechiae on serosa surface | Acute catarrhal enteritis with numerous petechiae/ecchymosis on serosa surface | Hemorrhagic enteritis with occasionally multifocal erosive lesions | |
| Large intestine 1 | Acute catarrhal colitis/typhlitis/proctitis with some petechiae on serosa surface | Acute catarrhal colitis/typhlitis/proctitis with numerous petechiae/ecchymosis on serosa surface. GALT hyperplasia. | Hemorrhagic colitis/typhlitis/proctitis with occasionally multifocal necrotizing lesions associated with GALT hyperplasia | |
| Alimentary system (cont.) | Liver | Mild multifocal parenchymal color changes with slight hepatomegaly | Moderate hepatomegaly, moderate congestion and presence multifocal red areas and hemorrhages. Mild diffuse lobular pattern | Intense hepatomegaly, intense congestion and presence of intra-parenchymal coalescent red zones. Moderate pan-lobular pattern |
| Gallbladder | Mild to moderate edema affecting gallbladder wall/cystic duct. Normal bile | Edema affecting gallbladder wall/cystic duct with hemorrhages on serosa/submucosa surface. Bile with reddish aspect | Severe edema affecting gallbladder wall/cystic duct with severe hemorrhages. Bile content display red appearance with blood clots | |
| Pancreas | Multifocal hemorrhages on the surface | Focal necrosis | Extensive necrosis | |
| Urinary system | Kidney (hemorrhages) | |||
| a) Medullo-cortical pattern | Mild multifocal cortical and medullar petechiae with multifocal vascular angiectasia | Moderate multifocal cortical (petechiae) and medullar (dark red band) hemorrhages with moderate pelvic dilation | Marked diffuse cortical-medullar hemorrhages with diffuse general renal dark red, marked pelvic dilation and extensive sub-capsular hemorrhages | |
| b) Cortical-medullar pattern | Minimal to mild multifocal cortical and medullar petechiae | Moderate multifocal cortical and medullar hemorrhages (petechiae) with preponderance of cortical affectation. Perirenal edema | Marked multifocal cortical-medullar hemorrhages more frequent on the cortical layer with/without multifocal of moderate dark red pelvic areas. Perirenal edema | |
| Urinary bladder | Mild multifocal petechiae on mucosa surface without/with mild color changes in the urinary bladder wall | Multifocal petechiae and ecchymosis on urinary bladder wall with edema. Turbid urine | Multifocal to coalescent hemorrhages on the mucosal surface. Intense intramural edema. Blood-stained urine with clots. | |
| Lymphoid system | Spleen | Mild splenomegaly (minimal to mild bleeding after sectioning) | Moderate splenomegaly (moderate bleeding after sectioning) | Intense splenomegaly (marked bleeding after sectioning) |
| Lymph nodes | ||||
| a) Lymphadenomegaly | Slight | Moderate | Severe | |
| b) Congestion or hemorrhages | Moderate color changes with occasional petechiae | Petechiae and ecchymosis | Increased size, edematous and hemorrhagic | |
| c) Affected lymph nodes | Between 1–3 lymph nodes | Between 4–6 lymph nodes | 6 or more lymph nodes | |
| Tonsils | Swelling and erythematous | Swelling and erythematous with fibrin on the surface | Swelling and erythematous with fibrin and multifocal necrosis on the surface | |
| Bone marrow | Congestion | Congestion with petechiae and ecchymosis | Congestion with multifocal to coalescent hemorrhages | |
| Endocrine system—Adrenal glands | Multifocal petechiae hemorrhages in cortex and medulla | Medullar congestion. Petechiae and ecchymosis in cortex | Multifocal to coalescent hemorrhages predominantly in cortex | |
| Central nervous system—Brain | Congestion meninges/edema | Congestion with occasional petechiae | Severe vascular changes with petechiae and ecchymosis | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Bertos, A.; Cadenas-Fernández, E.; Rebollada-Merino, A.; Porras-González, N.; Mayoral-Alegre, F.J.; Barreno, L.; Kosowska, A.; Tomé-Sánchez, I.; Barasona, J.A.; Sánchez-Vizcaíno, J.M. Clinical Course and Gross Pathological Findings in Wild Boar Infected with a Highly Virulent Strain of African Swine Fever Virus Genotype II. Pathogens 2020, 9, 688. https://doi.org/10.3390/pathogens9090688
Rodríguez-Bertos A, Cadenas-Fernández E, Rebollada-Merino A, Porras-González N, Mayoral-Alegre FJ, Barreno L, Kosowska A, Tomé-Sánchez I, Barasona JA, Sánchez-Vizcaíno JM. Clinical Course and Gross Pathological Findings in Wild Boar Infected with a Highly Virulent Strain of African Swine Fever Virus Genotype II. Pathogens. 2020; 9(9):688. https://doi.org/10.3390/pathogens9090688
Chicago/Turabian StyleRodríguez-Bertos, Antonio, Estefanía Cadenas-Fernández, Agustín Rebollada-Merino, Néstor Porras-González, Francisco J. Mayoral-Alegre, Lucía Barreno, Aleksandra Kosowska, Irene Tomé-Sánchez, José A. Barasona, and José M. Sánchez-Vizcaíno. 2020. "Clinical Course and Gross Pathological Findings in Wild Boar Infected with a Highly Virulent Strain of African Swine Fever Virus Genotype II" Pathogens 9, no. 9: 688. https://doi.org/10.3390/pathogens9090688
APA StyleRodríguez-Bertos, A., Cadenas-Fernández, E., Rebollada-Merino, A., Porras-González, N., Mayoral-Alegre, F. J., Barreno, L., Kosowska, A., Tomé-Sánchez, I., Barasona, J. A., & Sánchez-Vizcaíno, J. M. (2020). Clinical Course and Gross Pathological Findings in Wild Boar Infected with a Highly Virulent Strain of African Swine Fever Virus Genotype II. Pathogens, 9(9), 688. https://doi.org/10.3390/pathogens9090688






