Mycobacterium tuberculosis Rv0341 Promotes Mycobacterium Survival in In Vitro Hostile Environments and within Macrophages and Induces Cytokines Expression
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains, Cells Line, and Culture Conditions
2.2. Cloning of Rv0341 and Construction of Recombinant Strains
2.3. Detection of the Expression of Rv0341
2.4. In Vitro Growth Kinetic Analysis
2.5. Colony Morphology and Sliding Motility Analysis
2.6. Assessments of Recombinant Strains Survival under In Vitro Harsh Environments
2.7. Cell Wall Permeability Analysis
2.8. M. smegmatis Strains Infection of Macrophages
2.9. Analysis of Cytokine Expression
2.10. Data Analysis
3. Results
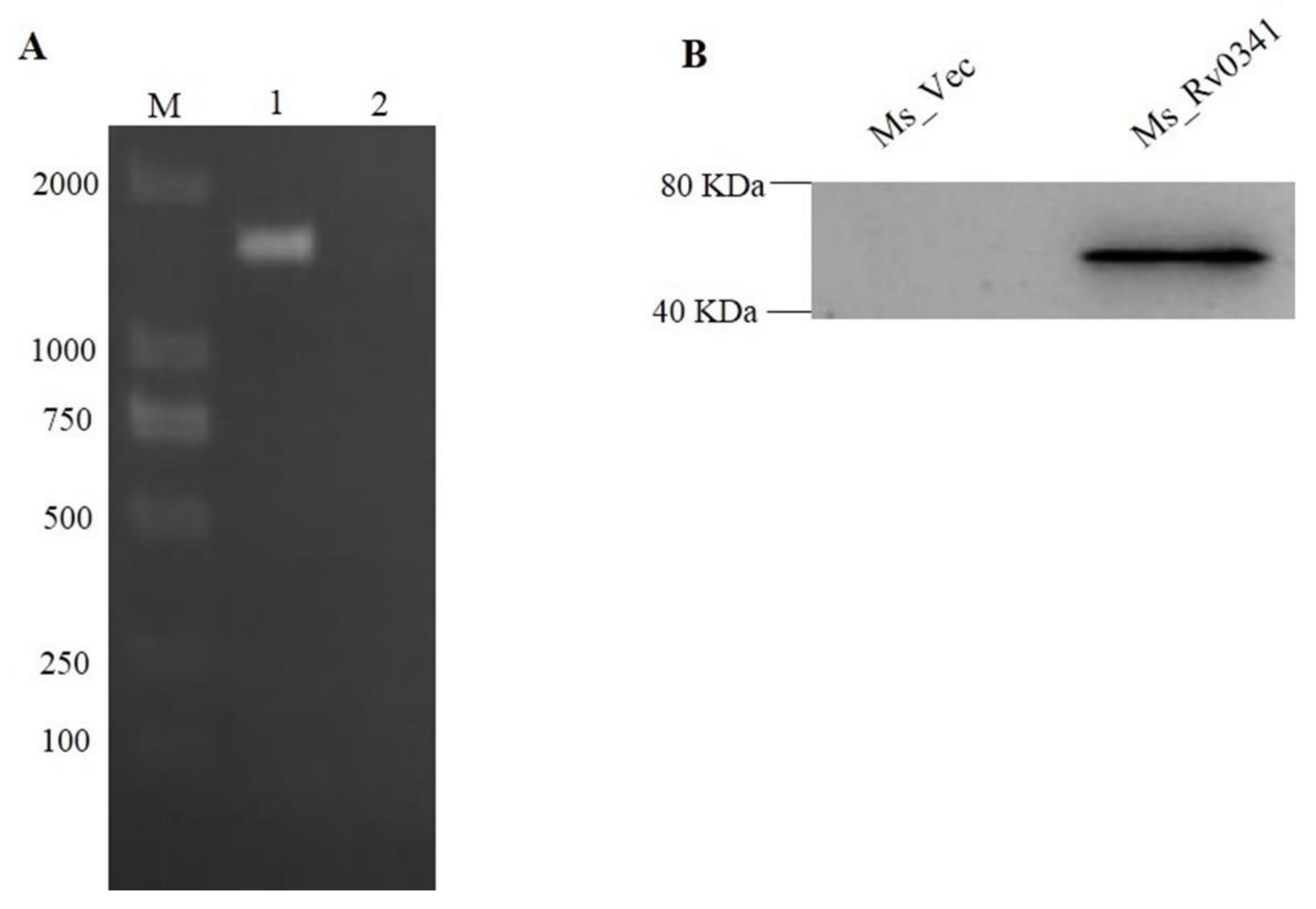
3.1. M. tuberculosis Rv0341 Protein Ectopically Expressed in M. smegmatis
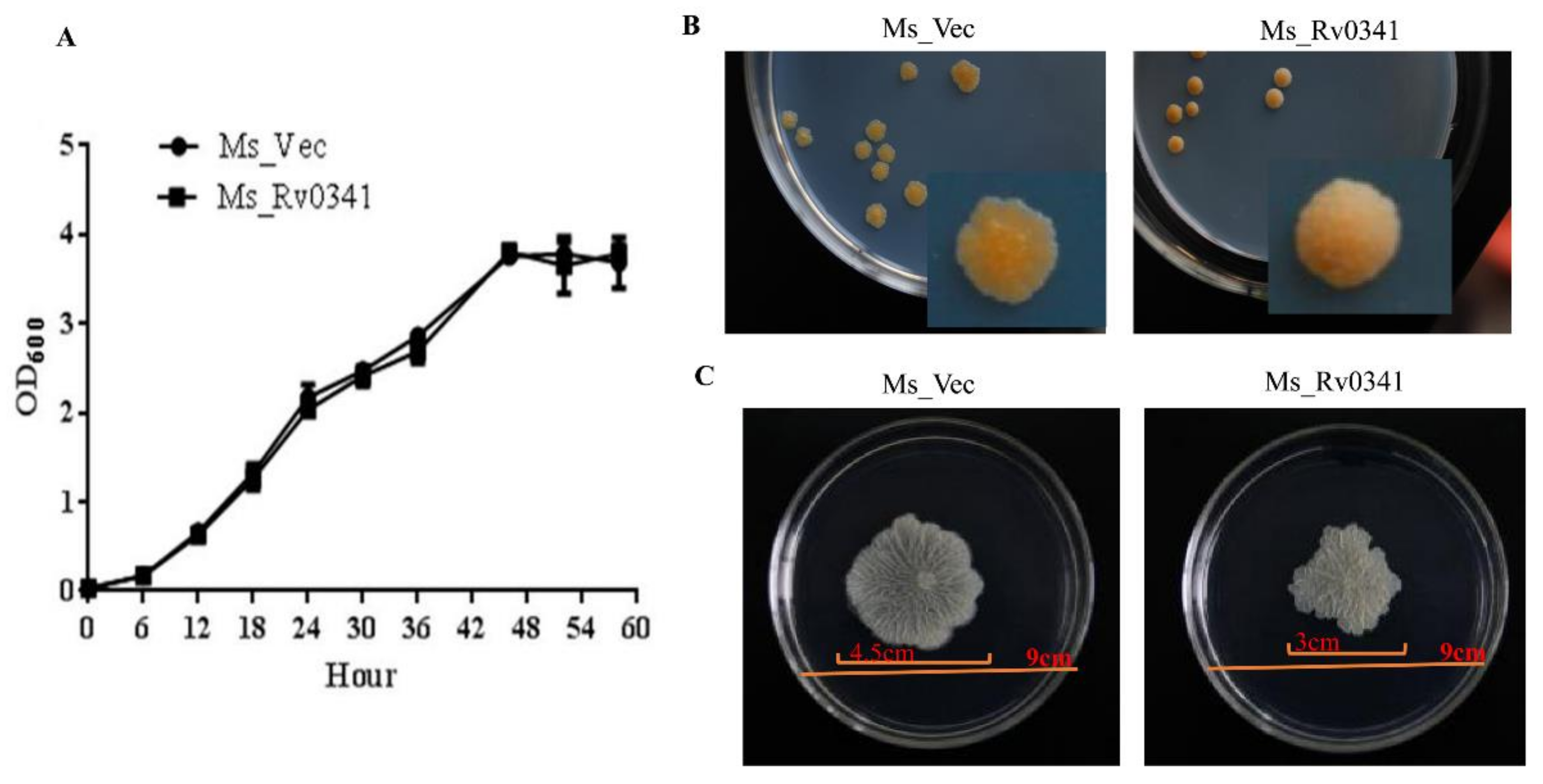
3.2. Rv0341 Alters the Colony Morphology and Sliding Motility of M. smegmatis
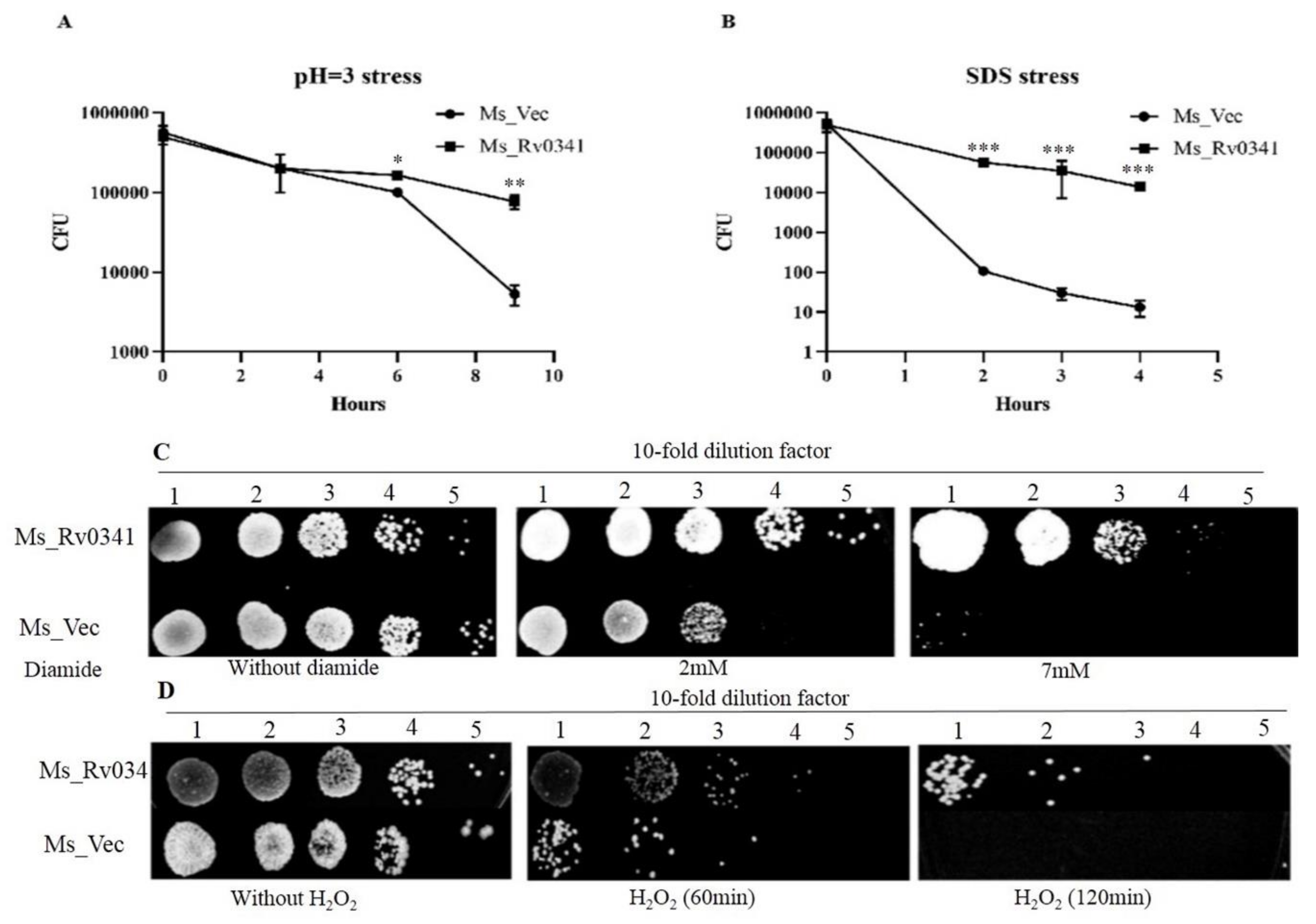
3.3. Rv0341 Mediates Mycobacterium Resistance to Harsh Environments
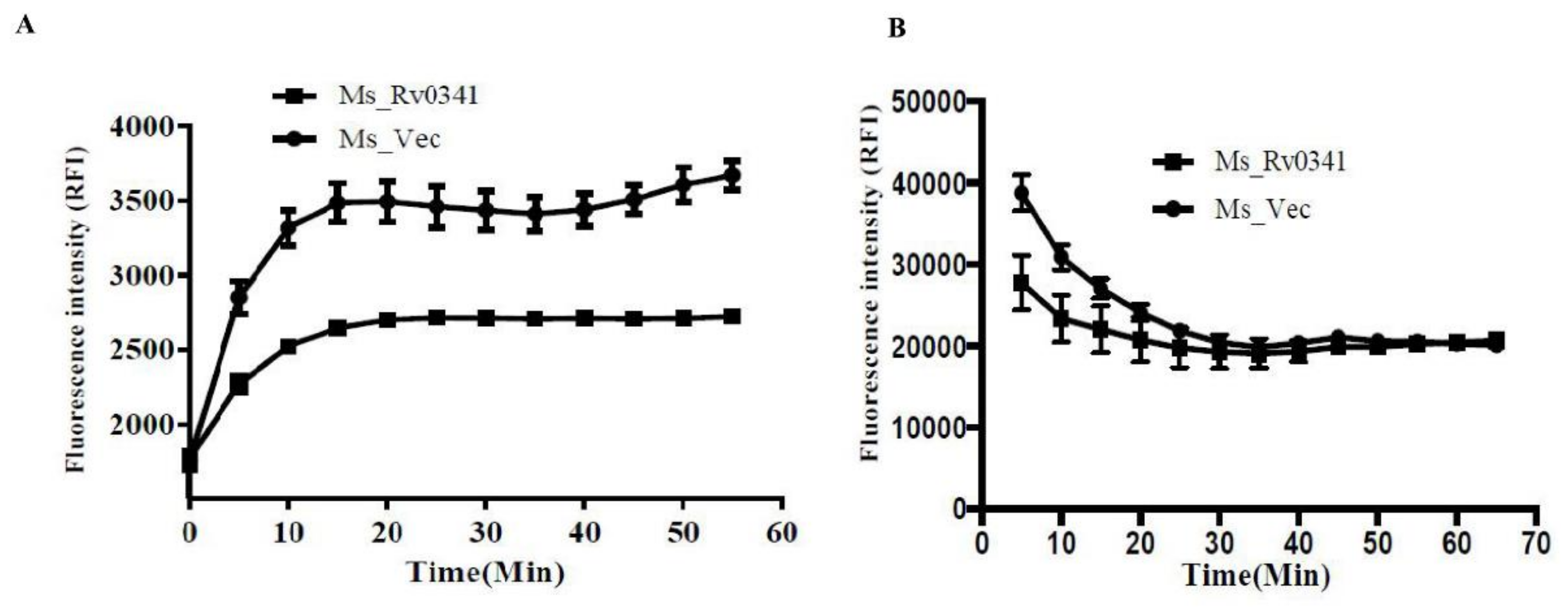
3.4. Rv0341 Decreases the Cell Wall Permeability of Mycobacterium
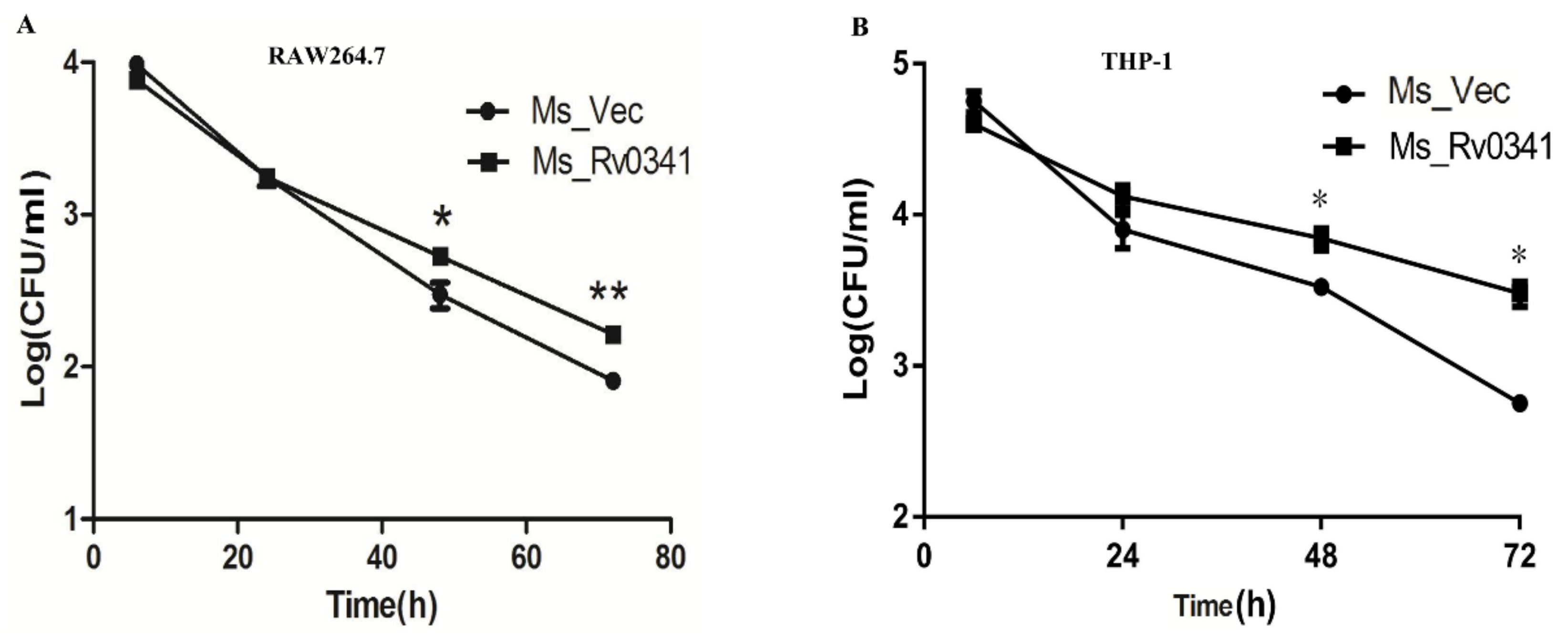
3.5. Rv0341 Expression Increases Mycobacterium Survival in Macrophages
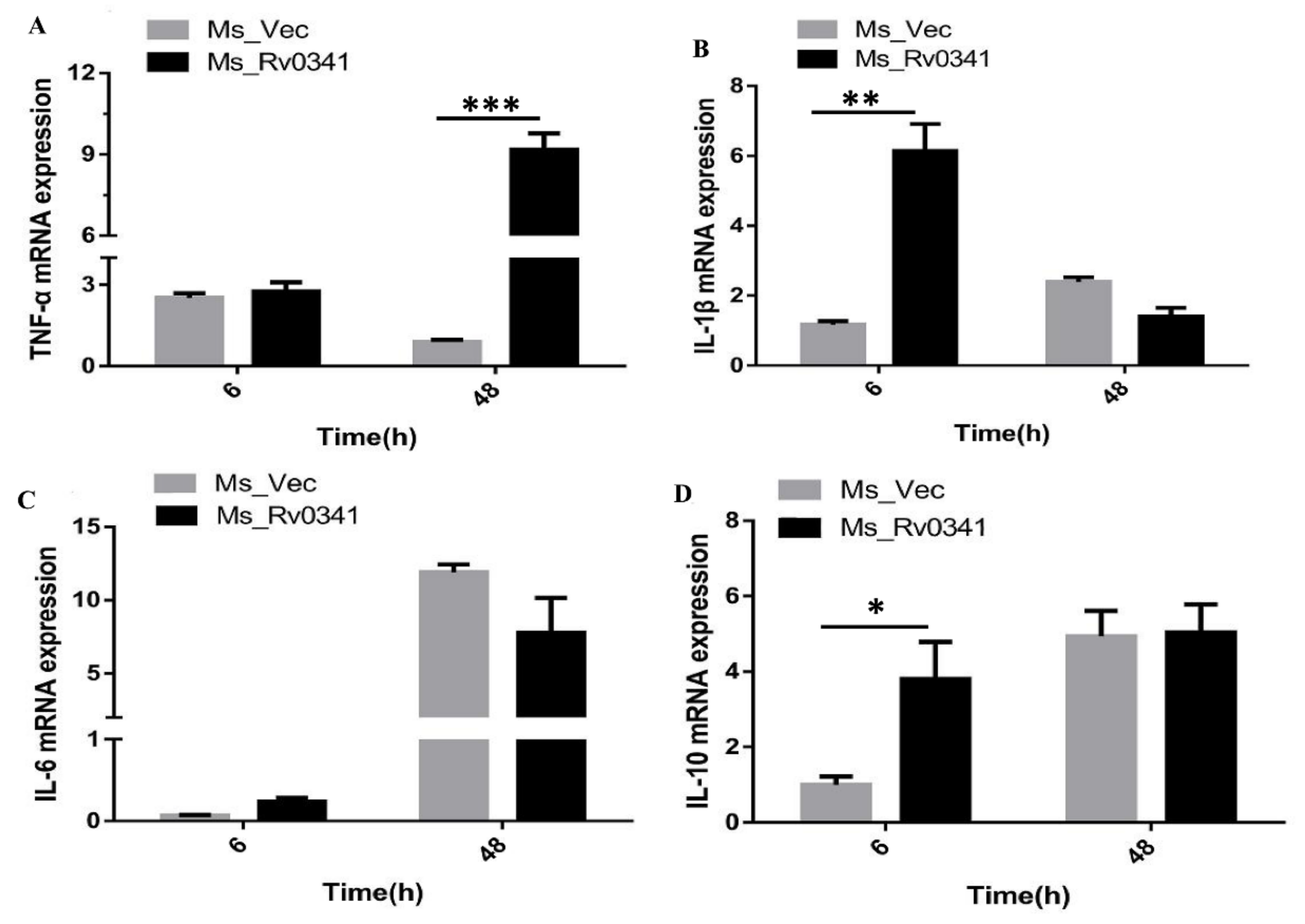
3.6. Rv0341 Induced Innate Cytokines Expression
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Tuberculosis Report 2019; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Bhat, K.H.; Mukhopadhyay, S. Macrophage takeover and the host–bacilli interplay during tuberculosis. Future Microbiol. 2015, 10, 853–872. [Google Scholar] [CrossRef] [PubMed]
- Hmama, Z.; Peña-Díaz, S.; Joseph, S.; Av-Gay, Y. Immunoevasion and immunosuppression of the macrophage by mycobacterium tuberculosis. Immunol. Rev. 2015, 264, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Dorhoi, A.; Kaufmann, S.H. Perspectives on host adaptation in response to mycobacterium tuberculosis: Modulation of inflammation. Semin. Immunol. 2014, 26, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Smith, I. Mycobacterium tuberculosis pathogenesis and molecular determinants of virulence. Clin. Microbiol. Rev. 2003, 16, 463–496. [Google Scholar] [CrossRef] [PubMed]
- Madacki, J.; Fiol, G.M.; Brosch, R. Update on the virulence factors of the obligate pathogen mycobacterium tuberculosis and related tuberculosis-causing mycobacteria. Infect. Genet. Evol. 2019, 72, 67–77. [Google Scholar] [CrossRef]
- Wang, M.; Guo, X.; Yang, X.; Zhang, B.; Ren, J.; Liu, A.; Ran, Y.; Yan, B.; Chen, F.; Guddat, L.W. Mycobacterial dynamin-like protein inia mediates membrane fission. Nat. Commun. 2019, 10, 1–13. [Google Scholar] [CrossRef]
- Alland, D.; Steyn, A.J.; Weisbrod, T.; Aldrich, K.; Jacobs, W.R. Characterization of the mycobacterium tuberculosis inibac promoter, a promoter that responds to cell wall biosynthesis inhibition. J. Bacteriol. 2000, 182, 1802–1811. [Google Scholar] [CrossRef]
- Bogatcheva, E.; Hanrahan, C.; Chen, P.; Gearhart, J.; Sacksteder, K.; Einck, L.; Nacy, C.; Protopopova, M. Discovery of dipiperidines as new antitubercular agents. Bioorg. Med. Chem. Lett. 2010, 20, 201–205. [Google Scholar] [CrossRef]
- Fenhalls, G.; Stevens, L.; Moses, L.; Bezuidenhout, J.; Betts, J.C.; van Helden, P.; Lukey, P.T.; Duncan, K. In situ detection of mycobacterium tuberculosis transcripts in human lung granulomas reveals differential gene expression in necrotic lesions. Infect. Immun. 2002, 70, 6330–6338. [Google Scholar] [CrossRef]
- Flyer, D.C.; Ramakrishna, V.; Miller, C.; Myers, H.; McDaniel, M.; Root, K.; Flournoy, C.; Engelhard, V.H.; Canaday, D.H.; Marto, J.A. Identification by mass spectrometry of cd8+-t-cell mycobacterium tuberculosis epitopes within the rv0341 gene product. Infect. Immun. 2002, 70, 2926–2932. [Google Scholar] [CrossRef]
- Agrawal, P.; Miryala, S.; Varshney, U. Use of mycobacterium smegmatis deficient in adp-ribosyltransferase as surrogate for mycobacterium tuberculosis in drug testing and mutation analysis. PLoS ONE 2015, 10, e0122076. [Google Scholar] [CrossRef] [PubMed]
- Altaf, M.; Miller, C.H.; Bellows, D.S.; O’Toole, R. Evaluation of the mycobacterium smegmatis and bcg models for the discovery of mycobacterium tuberculosis inhibitors. Tuberculosis 2010, 90, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Rao, R.N.; Reddy, J.R.C.; Prasad, R.; Kotturu, S.K.; Ghosh, S.; Mukhopadhyay, S. Pe11, a pe/ppe family protein of mycobacterium tuberculosis is involved in cell wall remodeling and virulence. Sci. Rep. 2016, 6, 21624. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, Z.-Y.; Schittenhelm, J.; Wu, Y.; Meyermann, R.; Schluesener, H.J. Parenchymal accumulation of cd163+ macrophages/microglia in multiple sclerosis brains. J. Neuroimmunol. 2011, 237, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhao, Q.; Deng, W.; Chen, T.; Liu, M.; Xie, J. Mycobacterium tuberculosis rv3402c enhances mycobacterial survival within macrophages and modulates the host pro-inflammatory cytokines production via nf-kappa b/erk/p38 signaling. PLoS ONE 2014, 9, e94418. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Luo, H.; Liu, M.; Zeng, J.; Abdalla, A.E.; Duan, X.; Li, Q.; Xie, J. The effect of mycobacterium tuberculosis crispr-associated cas2 (rv2816c) on stress response genes expression, morphology and macrophage survival of mycobacterium smegmatis. Infect. Genet. Evol. 2016, 40, 295–301. [Google Scholar] [CrossRef]
- Luo, H.; Zeng, J.; Huang, Q.; Liu, M.; Abdalla, A.E.; Xie, L.; Wang, H.; Xie, J. Mycobacterium tuberculosis rv1265 promotes mycobacterial intracellular survival and alters cytokine profile of the infected macrophage. J. Biomol. Struct. Dyn. 2016, 34, 585–599. [Google Scholar] [CrossRef]
- Deng, W. Pe11 (rv1169c) selectively alters fatty acid components of mycobacterium smegmatis and host cell interleukin-6 level accompanied with cell death. Front. Microbiol. 2015, 6, 613. [Google Scholar] [CrossRef]
- Li, X.; Wu, J.; Han, J.; Hu, Y.; Mi, K. Distinct responses of mycobacterium smegmatis to exposure to low and high levels of hydrogen peroxide. PLoS ONE 2015, 10, e0134595. [Google Scholar] [CrossRef]
- Viswanathan, G.; Joshi, S.V.; Sridhar, A.; Dutta, S.; Raghunand, T.R. Identifying novel mycobacterial stress associated genes using a random mutagenesis screen in mycobacterium smegmatis. Gene 2015, 574, 20–27. [Google Scholar] [CrossRef]
- Rodrigues, L.; Ramos, J.; Couto, I.; Amaral, L.; Viveiros, M. Ethidium bromide transport across mycobacterium smegmatis cell-wall: Correlation with antibiotic resistance. BMC Microbiol. 2011, 11, 35. [Google Scholar] [CrossRef] [PubMed]
- Chuang, Y.-M.; Bandyopadhyay, N.; Rifat, D.; Rubin, H.; Bader, J.S.; Karakousis, P.C. Deficiency of the novel exopolyphosphatase rv1026/ppx2 leads to metabolic downshift and altered cell wall permeability in mycobacterium tuberculosis. MBio 2015, 6, e02428. [Google Scholar] [CrossRef] [PubMed]
- Hett, E.C.; Rubin, E.J. Bacterial growth and cell division: A mycobacterial perspective. Microbiol. Mol. Biol. Rev. 2008, 72, 126–156. [Google Scholar] [CrossRef] [PubMed]
- Horváti, K.; Pályi, B.; Henczkó, J.; Balka, G.; Szabó, E.; Farkas, V.; Biri-Kovács, B.; Szeder, B.; Fodor, K. A convenient synthetic method to improve immunogenicity of mycobacterium tuberculosis related t-cell epitope peptides. Vaccines 2019, 7, 101. [Google Scholar] [CrossRef]
- Bussi, C.; Gutierrez, M.G. Mycobacterium tuberculosis infection of host cells in space and time. FEMS Microbiol. Rev. 2019, 43, 341–361. [Google Scholar] [CrossRef]
- Zu Bentrup, K.H.; Russell, D.G. Mycobacterial persistence: Adaptation to a changing environment. Trends Microbiol. 2001, 9, 597–605. [Google Scholar] [CrossRef]
- Boot, M.; van Winden, V.J.; Sparrius, M.; van de Weerd, R.; Speer, A.; Ummels, R.; Rustad, T.; Sherman, D.R.; Bitter, W. Cell envelope stress in mycobacteria is regulated by the novel signal transduction atpase inir in response to trehalose. PLoS Genet. 2017, 13, e1007131. [Google Scholar] [CrossRef]
- Mehra, S.; Dutta, N.K.; Mollenkopf, H.-J.; Kaushal, D. Mt2816 encodes a key mycobacterium tuberculosis stress-response regulator. J. Infect. Dis. 2010, 202, 943. [Google Scholar] [CrossRef]
- Maan, P.; Kumar, A.; Kaur, J.; Kaur, J. Rv1288, a two domain, cell wall anchored, nutrient stress inducible carboxyl-esterase of mycobacterium tuberculosis, modulates cell wall lipid. Front. Cell. Infect. Microbiol. 2018, 8, 421. [Google Scholar] [CrossRef]
- Campodónico, V.L.; Rifat, D.; Chuang, Y.-M.; Ioerger, T.R.; Karakousis, P.C. Altered mycobacterium tuberculosis cell wall metabolism and physiology associated with rpob mutation h526d. Front. Microbiol. 2018, 9, 494. [Google Scholar] [CrossRef]
- Kumari, B.; Saini, V.; Kaur, J.; Kaur, J. Rv2037c, a stress induced conserved hypothetical protein of mycobacterium tuberculosis, is a phospholipase: Role in cell wall modulation and intracellular survival. Int. J. Biol. Macromol. 2020, 153, 817–835. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, L.M.; Mahaffey, S.B.; Kruh, N.A.; Dobos, K.M. Proteomic definition of the cell wall of mycobacterium tuberculosis. J. Proteome Res. 2010, 9, 5816–5826. [Google Scholar] [CrossRef] [PubMed]
- Raman, S.; Hazra, R.; Dascher, C.C.; Husson, R.N. Transcription regulation by the mycobacterium tuberculosis alternative sigma factor sigd and its role in virulence. J. Bacteriol. 2004, 186, 6605–6616. [Google Scholar] [CrossRef] [PubMed]
- Meena, L.S. Survival mechanisms of pathogenic mycobacterium tuberculosis h37rv. FEBS J. 2010, 277, 2416–2427. [Google Scholar] [CrossRef]
- Purdy, G.E.; Niederweis, M.; Russell, D.G. Decreased outer membrane permeability protects mycobacteria from killing by ubiquitin-derived peptides. Mol. Microbiol. 2009, 73, 844–857. [Google Scholar] [CrossRef]
- Deng, W.; Long, Q.; Zeng, J.; Li, P.; Yang, W.; Chen, X.; Xie, J. Mycobacterium tuberculosis pe_pgrs41 enhances the intracellular survival of m. Smegmatis within macrophages via blocking innate immunity and inhibition of host defense. Sci. Rep. 2017, 7, 46716. [Google Scholar] [CrossRef]
- Li, Y.; Zeng, J.; Shi, J.; Wang, M.; Rao, M.; Xue, C.; Du, Y.; He, Z.-G. A proteome-scale identification of novel antigenic proteins in mycobacterium tuberculosis toward diagnostic and vaccine development. J. Proteome Res. 2010, 9, 4812–4822. [Google Scholar] [CrossRef]
- Marinho, F.V.; Fahel, J.S.; Scanga, C.A.; Gomes, M.T.R.; Guimarães, G.; Carvalho, G.R.; Morales, S.V.; Báfica, A.; Oliveira, S.C. Lack of il-1 receptor–associated kinase-4 leads to defective th1 cell responses and renders mice susceptible to mycobacterial infection. J. Immunol. 2016, 197, 1852–1863. [Google Scholar] [CrossRef]
- Zhang, G.; Zhou, B.; Li, S.; Yue, J.; Yang, H.; Wen, Y.; Zhan, S.; Wang, W.; Liao, M.; Zhang, M. Allele-specific induction of il-1β expression by c/ebpβ and pu. 1 contributes to increased tuberculosis susceptibility. PLoS Pathog. 2014, 10, e1004426. [Google Scholar] [CrossRef]
- Tram, T.T.; Nhung, H.N.; Vijay, S.; Hai, H.T.; Thu, D.D.; Ha, V.T.; Dinh, T.D.; Ashton, P.M.; Hanh, N.T.; Phu, N.H. Virulence of mycobacterium tuberculosis clinical isolates is associated with sputum pre-treatment bacterial load, lineage, survival in macrophages, and cytokine response. Front. Cell. Infect. Microbiol. 2018, 8, 417. [Google Scholar] [CrossRef]
- Lee, M.-R.; Chang, L.-Y.; Chang, C.-H.; Yan, B.-S.; Wang, J.-Y.; Lin, W.-H. Differed il-1 beta response between active tb and ltbi cases by ex vivo stimulation of human monocyte-derived macrophage with tb-specific antigen. Dis. Markers 2019, 2019, 7869576. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, A.E.; Lambert, N.; Duan, X.; Xie, J. Interleukin-10 family and tuberculosis: An old story renewed. Int. J. Biol. Sci. 2016, 12, 710. [Google Scholar] [CrossRef] [PubMed]
- Redford, P.S.; Boonstra, A.; Read, S.; Pitt, J.; Graham, C.; Stavropoulos, E.; Bancroft, G.J.; O’Garra, A. Enhanced protection to mycobacterium tuberculosis infection in il-10-deficient mice is accompanied by early and enhanced th1 responses in the lung. Eur. J. Immunol. 2010, 40, 2200–2210. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, S.; O’Sullivan, M.P.; Keane, J. Il-10 blocks phagosome maturation in mycobacterium tuberculosis–infected human macrophages. Am. J. Respir. Cell Mol. Biol. 2011, 45, 172–180. [Google Scholar] [CrossRef]
- Lin, P.L.; Plessner, H.L.; Voitenok, N.N.; Flynn, J.L. Tumor Necrosis Factor and Tuberculosis. J. Investig. Dermatol. Symp. Proc. 2007, 12, 22–25. [Google Scholar] [CrossRef]
- Kumar, A.; Lewin, A.; Rani, P.S.; Qureshi, I.A.; Devi, S.; Majid, M.; Kamal, E.; Marek, S.; Hasnain, S.E.; Ahmed, N. Dormancy associated translation inhibitor (datin/rv0079) of mycobacterium tuberculosis interacts with tlr2 and induces proinflammatory cytokine expression. Cytokine 2013, 64, 258–264. [Google Scholar] [CrossRef]
- Dutta, N.K.; Illei, P.B.; Jain, S.K.; Karakousis, P.C. Characterization of a novel necrotic granuloma model of latent tuberculosis infection and reactivation in mice. Am. J. Pathol. 2014, 184, 2045–2055. [Google Scholar] [CrossRef]
- Dambuza, I.; Keeton, R.; Allie, N.; Hsu, N.-J.; Randall, P.; Sebesho, B.; Fick, L.; Quesniaux, V.J.; Jacobs, M. Reactivation of m. Tuberculosis infection in trans-membrane tumour necrosis factor mice. PLoS ONE 2011, 6, e25121. [Google Scholar] [CrossRef]
| Target Gene | Primers | Sequence (5′→3′) |
|---|---|---|
| IL-1β | F | TTCAGGCAGGCCGCGTCAGTTGT |
| R | TGTGAGTCCCGGAGCGTGCAGTT | |
| IL-6 | F | GCCTTCGGTCCAGTTGCCTTCT |
| R | TGCCAGTGCCTCTTTGCTGCTTT | |
| IL-10 | F | ACCTGGGTTGCCAAGCCTTGT |
| R | GCTCCACGGCCTTGCTCTTGTTT | |
| TNFα | F | GGCGGTGCTTGTTCCT |
| R | GCTACAGGCTTGTCACTCG | |
| β-actin | F | GTGACGTTGACATCCGTAAAGA |
| R | GTGACGTTGACATCCGTAAAGA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdalla, A.E.; Yan, S.; Zeng, J.; Deng, W.; Xie, L.; Xie, J. Mycobacterium tuberculosis Rv0341 Promotes Mycobacterium Survival in In Vitro Hostile Environments and within Macrophages and Induces Cytokines Expression. Pathogens 2020, 9, 454. https://doi.org/10.3390/pathogens9060454
Abdalla AE, Yan S, Zeng J, Deng W, Xie L, Xie J. Mycobacterium tuberculosis Rv0341 Promotes Mycobacterium Survival in In Vitro Hostile Environments and within Macrophages and Induces Cytokines Expression. Pathogens. 2020; 9(6):454. https://doi.org/10.3390/pathogens9060454
Chicago/Turabian StyleAbdalla, Abualgasim Elgaili, Shuangquan Yan, Jie Zeng, Wanyan Deng, Longxiang Xie, and Jianping Xie. 2020. "Mycobacterium tuberculosis Rv0341 Promotes Mycobacterium Survival in In Vitro Hostile Environments and within Macrophages and Induces Cytokines Expression" Pathogens 9, no. 6: 454. https://doi.org/10.3390/pathogens9060454
APA StyleAbdalla, A. E., Yan, S., Zeng, J., Deng, W., Xie, L., & Xie, J. (2020). Mycobacterium tuberculosis Rv0341 Promotes Mycobacterium Survival in In Vitro Hostile Environments and within Macrophages and Induces Cytokines Expression. Pathogens, 9(6), 454. https://doi.org/10.3390/pathogens9060454






