A Cross-Sectional Study of Seroprevalence of Strongyloidiasis in Pregnant Women (Peruvian Amazon Basin)
Abstract
1. Introduction
2. Methods
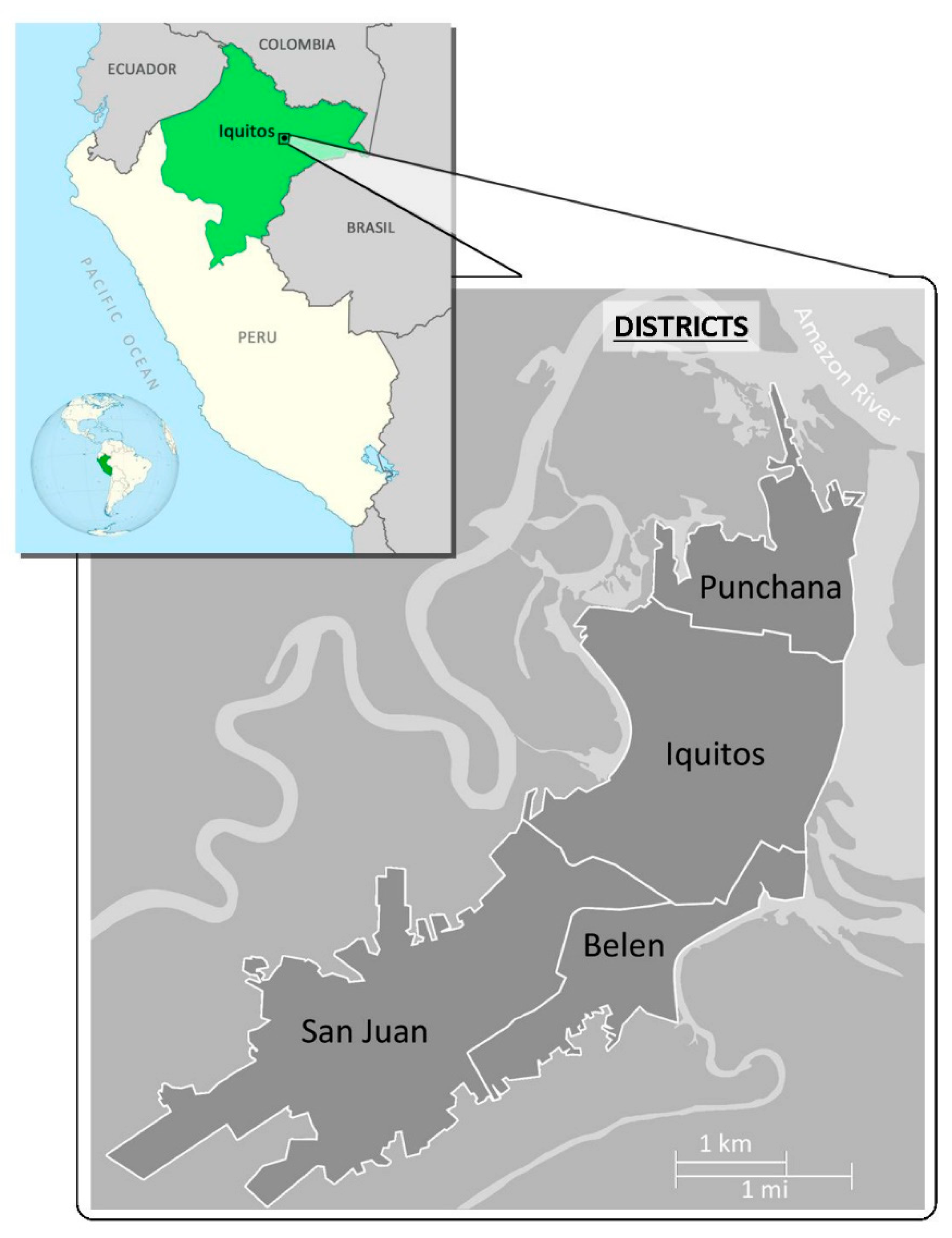
2.1. Study Population
2.2. Procedures
2.3. Stool Examination for S. stercoralis Infection
2.4. Serology
2.5. Stool Examination for Other Intestinal Helminthic Infestations
2.6. Data Analysis
2.7. Ethical Considerations
3. Results
3.1. Study Population
3.2. Stool Examination of S. stercoralis Infection
3.3. Intestinal Helminthic Infestations in Stool
3.4. Co-Infections of S. stercoralis and Other Helminths
3.5. Serology of S. stercoralis Infection
3.6. Participant and Housing Characteristics and Their Relation to S. stercoralis Infection
3.7. Serology Versus Stool Examination for Detecting S. stercoralis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Krolewiecki, A.; Nutman, T.B. Strongyloidiasis: A Neglected Tropical Disease. Infect. Dis. Clin. N. Am. 2019, 33, 135–151. [Google Scholar] [CrossRef]
- Schär, F.; Trostdorf, U.; Giardina, F.; Khieu, V.; Muth, S.; Marti, H.; Vounatsou, P.; Odermatt, P. Strongyloides stercoralis: Global Distribution and Risk Factors. PLoS Negl. Trop. Dis. 2013, 7, e2288. [Google Scholar] [CrossRef] [PubMed]
- Bisoffi, Z.; Buonfrate, D.; Montresor, A.; Requena-Mendez, A.; Munoz, J.; Krolewiecki, A.J.; Gotuzzo, E.; Mena, M.A.; Chiodini, P.L.; Anselmi, M.; et al. Strongyloides stercoralis: A plea for action. PLoS Negl. Trop. Dis. 2013, 7, e2214. [Google Scholar] [CrossRef] [PubMed]
- Van De, N.; Minh, P.N.; Van Duyet, L.; Mas-Coma, S. Strongyloidiasis in northern Vietnam: Epidemiology, clinical characteristics and molecular diagnosis of the causal agent. Parasit. Vectors 2019, 12, 515. [Google Scholar] [CrossRef] [PubMed]
- Guevara, A.G.; Anselmi, M.; Bisoffi, Z.; Prandi, R.; Márquez, M.; Silva, R.; Vicuña, Y.; Calvopiña, M.; Cevallos, W.; Pérez, J.; et al. Mapping the Prevalence of Strongyloides stercoralis Infection in Ecuador: A Serosurvey. Am. J. Trop. Med. Hyg. 2019, 102, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Buresch, A.M.; Judge, N.E.; Dayal, A.K.; Garry, D.J. A Fatal Case of Strongyloidiasis in Pregnancy. Obstet. Gynecol. 2015, 126, 87–89. [Google Scholar] [CrossRef] [PubMed]
- Malézieux-Picard, A.; Saint-Paul, M.C.; Dellamonica, J.; Courjon, J.; Tieulié, N.; Marty, P.; Fuzibet, J.G.; Collomp, R.; Marinho, J.A.; Queyrel, V. Occlusion Intestinale à Strongyloides Stercoralis chez une Femme Enceinte. Med. Mal. Infect. 2017, 47, 429–431. [Google Scholar] [CrossRef]
- Beltrán Rosel, A.; Sanjoaquín Conde, I.; Pérez Muñoz, A.; Alcalá, P.M.; Marta, C.B.; von Sierakowski, A.I. Strongyloides stercoralis: A rare and severe presentation in a pregnant woman. New Microbiol. 2019, 43, 44–46. [Google Scholar]
- Hays, R.; McDermott, R. Strongyloides stercoralis infection and antenatal care. In Medical Journal of Australia; Australasian Medical Publishing Co. Ltd.: Sydney, Australia, 2015; pp. 1–2. [Google Scholar] [CrossRef]
- Buonfrate, D.; Perandin, F.; Formenti, F.; Bisoffi, Z. A retrospective study comparing agar plate culture, indirect immunofluorescence and real-time PCR for the diagnosis of Strongyloides stercoralis infection. Parasitology 2017, 144, 812–816. [Google Scholar] [CrossRef]
- Buonfrate, D.; Requena-Mendez, A.; Angheben, A.; Cinquini, M.; Cruciani, M.; Fittipaldo, A.; Giorli, G.; Gobbi, F.; Piubelli, C.; Bisoffi, Z. Accuracy of molecular biology techniques for the diagnosis of Strongyloides stercoralis infection—A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2018, 12, e0006229. [Google Scholar] [CrossRef]
- Marcos, R.; Luis, A.; Canales, M.; Terashima, A. Métodos de diagnóstico para en el Perú. Rev. Peru Parasitol. 2010, 18, e6–e9. [Google Scholar]
- Buonfrate, D.; Formenti, F.; Perandin, F.; Bisoffi, Z. Novel approaches to the diagnosis of Strongyloides stercoralis infection. Clin. Microbiol. Infect. 2015, 21, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Bisoffi, Z.; Buonfrate, D.; Sequi, M.; Mejia, R.; Cimino, R.O.; Krolewiecki, A.J.; Albonico, M.; Gobbo, M.; Bonafini, S.; Angheben, A.; et al. Diagnostic Accuracy of Five Serologic Tests for Strongyloides stercoralis Infection. PLoS Negl. Trop. Dis. 2014, 8, 38. [Google Scholar] [CrossRef] [PubMed]
- Ministerio de Salud de Peru. Prevalence, Intestinal helminths in Perù: Analysis of (1981–2001); Oficina General de Epidemiologia, Ed.; Oficina de Epidemiología: Lima, Peru, 2003.
- Yori, P.P.; Kosek, M.; Gilman, R.H.; Cordova, J.; Bern, C.; Chavez, C.B.; Olortegui, M.P.; Montalvan, C.; Sanchez, G.M.; Worthen, B.; et al. Seroepidemiology of strongyloidiasis in the Peruvian Amazon. Am. J. Trop. Med. Hyg. 2006, 74, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Ivoke, N.; Ikpor, N.; Ivoke, O.; Ekeh, F.; Ezenwaji, N.; Odo, G.; Iyaji, F.; Onoja, U.; Eyo, J. Geophagy as risk behaviour for gastrointestinal nematode infections among pregnant women attending antenatal clinics in a humid tropical zone of Nigeria. Afr. Health Sci. 2017, 17, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Ojurongbe, O.; Okorie, P.N.; Opatokun, R.L.; Ojurongbe, T.A.; Mabayoje, V.O.; Olowe, O.A.; Adeyeba, O.A. Prevalence and associated factors of Plasmodium falciparum and soil transmitted helminth infections among pregnant women in Osun state, Nigeria. Afr. Health Sci. 2018, 18, 542–551. [Google Scholar] [CrossRef]
- Hailegebriel, T.; Petros, B.; Endeshaw, T. Evaluation of Parasitological Methods for the Detection of Strongyloides Stercoralis among Individuals in Selected Health Institutions In Addis Ababa, Ethiopia. Ethiop. J. Health Sci. 2017, 27, 515–522. [Google Scholar] [CrossRef]
- Speich, B.; Utzinger, J.; Marti, H.; Ame, S.M.; Ali, S.M.; Albonico, M.; Keiser, J. Comparison of the Kato-Katz method and ether-concentration technique for the diagnosis of soil-transmitted helminth infections in the framework of a randomised controlled trial. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 815–822. [Google Scholar] [CrossRef]
- Newcombe, R.G. Two-sided confidence intervals for the single proportion: Comparison of seven methods. Stat Med. 1998, 17, 857–872. [Google Scholar] [CrossRef]
- Dotters-Katz, S.; Kuller, J.; Heine, R.P. Parasitic infections in pregnancy. Obstet. Gynecol. Surv. 2011, 66, 515–525. [Google Scholar] [CrossRef]
- Mpairwe, H.; Tweyongyere, R.; Elliott, A. Pregnancy and helminth infections. Parasite Immunol. 2014, 36, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Errea, R.A.; Vasquez-Rios, G.; Calderon, M.L.; Siu, D.; Duque, K.R.; Juarez, L.H.; Gallegos, R.; Uriol, C.; Rondon, C.R.; Baca, K.P.; et al. Soil-Transmitted Helminthiasis in Children from a Rural Community Taking Part in a Periodic Deworming Program in the Peruvian Amazon. Am. J. Trop. Med. Hyg. 2019, 101, 636–640. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, J.; Calderón, J. Intestinal parasitosis in pre-school children from Tarapoto. Rev. Gastroenterol. Peru 1991, 11, 153–160. [Google Scholar] [PubMed]
- Egido, J.M.; De Diego, J.A.; Penin, P. The prevalence of enteropathy due to strongyloidiasis in Puerto Maldonado (Peruvian Amazon). Braz. J. Infect. Dis. 2001, 5, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Lau Chong, C.; Samalvides Cuba, F.; Terashima Iwashita, A. Evaluación de técnicas parasitológicas en el diagnóstico de estrongiloidiasis por Strongyloides stercoralis. Rev. Med. Hered. 2005, 16, 11–18. [Google Scholar] [CrossRef]
- Anselmi, M.; Guevara, A.; Vicuña, Y.; Vivero, S.; Prandi, R.; Caicedo, C.; Marquez, M.; Bisoffi, Z.; Buonfrate, D. Community Epidemiology Approach to Parasitic Infection Screening in a Remote Community in Ecuador. Am. J. Trop. Med. Hyg. 2019, 101, 650–653. [Google Scholar] [CrossRef]
- Conway, D.J.; Atkins, N.S.; Lillywhite, J.E.; Bailey, J.W.; Robinson, R.D.; Lindo, J.F.; Bundy, D.A.; Bianco, A.E. Immunodiagnosis of Strongyloides stercoralis infection: A method for increasing the specificity of the indirect ELISA. Trans. R. Soc. Trop. Med. Hyg. 1993, 87, 173–176. [Google Scholar] [CrossRef]
- Casado, L.; Rodriguez-Guardado, A.; Boga, J.A.; Fernández-Suarez, J.; Martínez-Camblor, P.; Rodríguez-Perez, M.; García-Pérez, A.; Vazquez, F.; Gascon, J. Use of Serology in a Systematic Screening Programme for Strongyloidiasis in an Immigrant Population. Int. J. Infect. Dis. 2019, 88, 60–64. [Google Scholar] [CrossRef]
- Requena-Méndez, A.; Chiodini, P.; Bisoffi, Z.; Buonfrate, D.; Gotuzzo, E.; Muñoz, J. The laboratory diagnosis and follow up of strongyloidiasis: A systematic review. PLoS Negl. Trop. Dis. 2013, 7, e2002. [Google Scholar] [CrossRef]
- Krolewiecki, A.J.; Ramanathan, R.; Fink, V.; McAuliffe, I.; Cajal, S.P.; Won, K.; Juarez, M.; Di Paolo, A.; Tapia, L.; Acosta, N.; et al. Improved diagnosis of Strongyloides stercoralis using recombinant antigen-based serologies in a community-wide study in northern Argentina. Clin. Vaccine Immunol. 2010, 17, 1624–1630. [Google Scholar] [CrossRef]
- Echazú, A.; Juarez, M.; Vargas, P.A.; Cajal, S.P.; Cimino, R.O.; Heredia, V.; Caropresi, S.; Paredes, G.; Arias, L.M.; Abril, M.; et al. Albendazole and ivermectin for the control of soil-transmitted helminths in an area with high prevalence of Strongyloides stercoralis and hookworm in northwestern Argentina: A community-based pragmatic study. PLoS Negl. Trop. Dis. 2017, 11, e0006003. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, M.J.; Brown, J.D. Human strongyloidiasis in Hawaii: A retrospective review of enzyme-linked immunosorbent assay serodiagnostic testing. Am. J. Trop. Med. Hyg. 2018, 99, 370–374. [Google Scholar] [CrossRef] [PubMed]
- Diep, N.T.N.; Thai, P.Q.; Trang, N.N.M.; Jaeger, J.; Fox, A.; Horby, P.; Phuong, H.V.M.; Anh, D.D.; LE THI, Q.M.; Van Doorn, H.R.; et al. Strongyloides stercoralis seroprevalence in Vietnam. Epidemiol. Infect. 2017, 145, 3214–3218. [Google Scholar] [CrossRef] [PubMed]
- Zueter, A.M.; Mohamed, Z.; Abdullah, A.D.; Mohamad, N.; Arifin, N.; Othman, N.; Noordin, R. Detection of Strongyloides stercoralis infection among cancer patients in a major hospital in Kelantan, Malaysia. Singap. Med. J. 2014, 55, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.F.; Hadip, F.; Ngui, R.; Lim, Y.A.L.; Mahmud, R. Serological and molecular detection of Strongyloides stercoralis infection among an Orang Asli community in Malaysia. Parasitol. Res. 2013, 112, 2811–2816. [Google Scholar] [CrossRef]
- Sultana, Y.; Gilbert, G.L.; Ahmed, B.N.; Lee, R. Strongyloidiasis in a high risk community of Dhaka, Bangladesh. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 756–762. [Google Scholar] [CrossRef]
- Hays, R.; Esterman, A.; McDermott, R. Control of chronic Strongyloides stercoralis infection in an endemic community may be possible by pharmacological means alone: Results of a three-year cohort study. PLoS Negl. Trop. Dis. 2017, 11. [Google Scholar] [CrossRef]
- Kaya, F.; İnkaya, A.Ç.; Ertenli, A.İ.; Abbasoğlu, O.; Aksoy, S.; Yilmaz, Y.A.; Ergüven, S. The investigation of Strongyloides stercoralis seroprevalence in immunosupressed patients in Turkey. Turk. J. Med. Sci. 2019, 49, 16–19. [Google Scholar] [CrossRef]
- Terefe, Y.; Ross, K.; Whiley, H. Strongyloidiasis in Ethiopia: Systematic review on risk factors, diagnosis, prevalence and clinical outcomes. Infect. Dis. Poverty 2019, 8, 53. [Google Scholar] [CrossRef]
| Variables | n (%) |
|---|---|
| Age, mean (SD) | 26.7 (6.4) |
| Number of deliveries, mean (SD) | 2.9 (1.7) |
| Gestation age in days, mean (SD) | 172 (59) |
| Health center districts, n (%) | |
| San Juan | 149 (49.7) |
| Puchana | 79 (26.3) |
| Belén | 51 (17.0) |
| Iquitos | 21 (7.0) |
| Area of residence, n (%) | |
| Urban area | 166 (55.3) |
| Rural area | 134 (44.7) |
| Bowel movements/day, mean (SD) | 1.5 (0.7) |
| Pathogens | n (%) |
|---|---|
| Strongyloides stercoralis alone | 16 (53.3) |
| S. stercoralis + hookworm | 5 (16.7) |
| S. stercoralis + Trichuris trichiura | 5 (16.7) |
| S. stercoralis + Ascaris lumbricoides + T. trichiura | 1 (3.3) |
| S. stercoralis + A. lumbricoides + hookworms | 1 (3.3) |
| S. stercoralis + T. trichiura + hookworms | 1 (3.3) |
| S. stercoralis + A. lumbricoides + T. trichiura + hookworms | 1 (3.3) |
| Helminthic Parasites | S. stercoralis ELISA | p Value * | |
|---|---|---|---|
| Positive n (% row) | Negative n (% row) | ||
| Stool examination for S. stercoralis | |||
| Modified Baermann method positive (N = 15) | 14 (93.3) | 1 (6.7) | <0.001 |
| Charcoal culture-positive (N = 23) | 23 (56.5) | 10 (43.5) | 0.016 |
| Modified Baermann method and/or charcoal culture-positive (N = 30) | 19 (63.3) | 11 (36.7) | < 0.001 |
| Stool examination for other intestinal helminthic infestations | |||
| A. lumbricoides (N = 49) | 17 (34.7) | 32 (65.3) | 0.87 |
| T. trichiura (N = 27) | 27 (44.4) | 15 (55.6) | 0.21 |
| Hookworms (N = 11) | 11 (54.5) | 5 (45.5) | 0.13 |
| Hymenolepis nana (N = 1) | 1(100) | 0 (0.0) | 0.32 |
| Variables | S. stercoralis in Feces | S. stercoralis ELISA | ||||
|---|---|---|---|---|---|---|
| Positive | Negative | p Value * | Positive | Negative | p Value * | |
| Age, mean (SD) | 26.7 (6.5) | 26.9 (5.7) | 0.84 | 26.7 (6.3) | 26.7 (6.7) | 0.35 |
| Rural residence, n (%) | 16 (53.3) | 118 (43.7) | 0.31 | 56 (55.4) | 78 (39.2) | 0.007 |
| Characteristics of house, n (%) | ||||||
| Dirt floor | 8 (26.7) | 81 (30.0) | 0.70 | 30 (29.7) | 59 (29.6) | 0.99 |
| Leaf roofing | 27 (90) | 207 (76.7) | 0.094 | 86 (85.1) | 148 (74.4) | 0.033 |
| Wooden house | 29 (96.7) | 257 (95.2) | 0.71 | 98 (97.0) | 188 (94.5) | 0.32 |
| Brick house | 18 (60.0) | 171 (63.3) | 0.72 | 67 (66.3) | 122 (61.3) | 0.30 |
| Characteristics of individuals | ||||||
| Bowel movements/day, mean (SD) | 1.5 (0.8) | 1.5 (0.8) | 0.95 | 1.6 (7.8) | 1.5 (0.7) | 0.64 |
| Hemoglobin < 11 g/dL, n (%) | 12 (40) | 121 (44.8) | 0.70 | 42 (41.6) | 91 (45.7) | 0.49 |
| S. stercoralis ELISA | Modified Baermann Methods and/or Charcoal Culture | Total | |
|---|---|---|---|
| Positive | Negative | ||
| Positive | 19 | 82 | 101 |
| Negative | 11 | 188 | 199 |
| Total | 30 | 270 | 300 |
| Sensitivity: 63.3% (95% CI 43.9% to 79.4%) Specificity: 69.6% (95% CI 63.7% to 74.9%) Positive predictive value: 18.8% (95% CI 11.9% to 28.1%) Negative predictive value: 94.4% (95% CI 90.0% to 97.1%) | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortiz-Martínez, S.; Ramos-Rincón, J.-M.; Vásquez-Chasnamote, M.-E.; Alarcón-Baldeón, J.J.; Parraguez-de-la-Cruz, J.; Gamboa-Paredes, O.-N.; Schillyk-Guerra, P.; Espinoza-Venegas, L.-A.; Pinedo-Cancino, V.-V.; Perez-Tanoira, R.; et al. A Cross-Sectional Study of Seroprevalence of Strongyloidiasis in Pregnant Women (Peruvian Amazon Basin). Pathogens 2020, 9, 348. https://doi.org/10.3390/pathogens9050348
Ortiz-Martínez S, Ramos-Rincón J-M, Vásquez-Chasnamote M-E, Alarcón-Baldeón JJ, Parraguez-de-la-Cruz J, Gamboa-Paredes O-N, Schillyk-Guerra P, Espinoza-Venegas L-A, Pinedo-Cancino V-V, Perez-Tanoira R, et al. A Cross-Sectional Study of Seroprevalence of Strongyloidiasis in Pregnant Women (Peruvian Amazon Basin). Pathogens. 2020; 9(5):348. https://doi.org/10.3390/pathogens9050348
Chicago/Turabian StyleOrtiz-Martínez, Sonia, José-Manuel Ramos-Rincón, María-Esteyner Vásquez-Chasnamote, Jhonatan J. Alarcón-Baldeón, Jorge Parraguez-de-la-Cruz, Olga-Nohelia Gamboa-Paredes, Patricia Schillyk-Guerra, Luis-Alfredo Espinoza-Venegas, Viviana-Vanessa Pinedo-Cancino, Ramón Perez-Tanoira, and et al. 2020. "A Cross-Sectional Study of Seroprevalence of Strongyloidiasis in Pregnant Women (Peruvian Amazon Basin)" Pathogens 9, no. 5: 348. https://doi.org/10.3390/pathogens9050348
APA StyleOrtiz-Martínez, S., Ramos-Rincón, J.-M., Vásquez-Chasnamote, M.-E., Alarcón-Baldeón, J. J., Parraguez-de-la-Cruz, J., Gamboa-Paredes, O.-N., Schillyk-Guerra, P., Espinoza-Venegas, L.-A., Pinedo-Cancino, V.-V., Perez-Tanoira, R., Górgolas-Hernández-Mora, M., Casapía-Morales, M., & Spanish-Peruvian Chagas, HTLV and Strongyloides Network. (2020). A Cross-Sectional Study of Seroprevalence of Strongyloidiasis in Pregnant Women (Peruvian Amazon Basin). Pathogens, 9(5), 348. https://doi.org/10.3390/pathogens9050348






