Efficacy of a Protein Vaccine and a Conjugate Vaccine Against Co-Colonization with Vaccine-Type and Non-Vaccine Type Pneumococci in Mice
Abstract
1. Introduction
2. Results
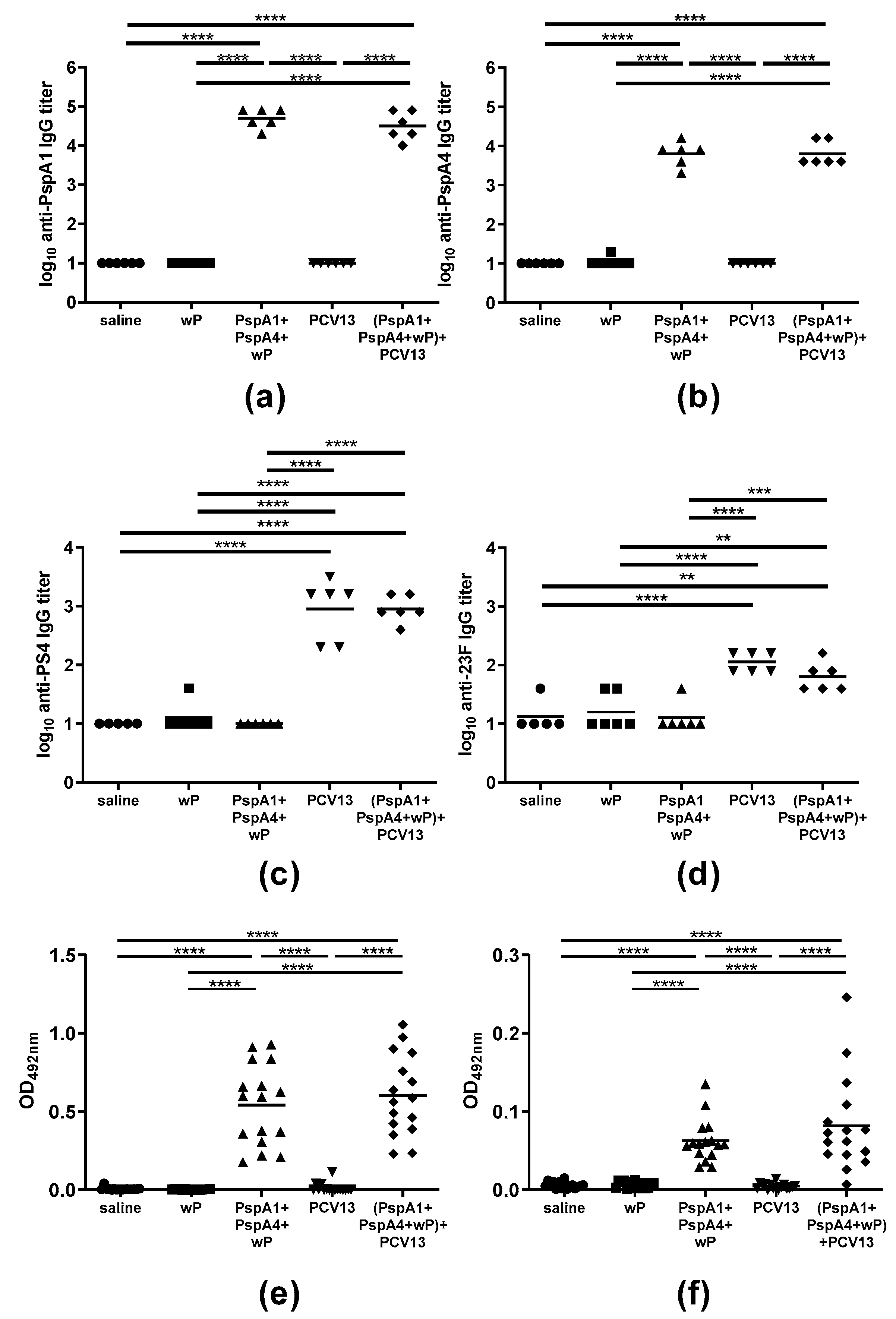
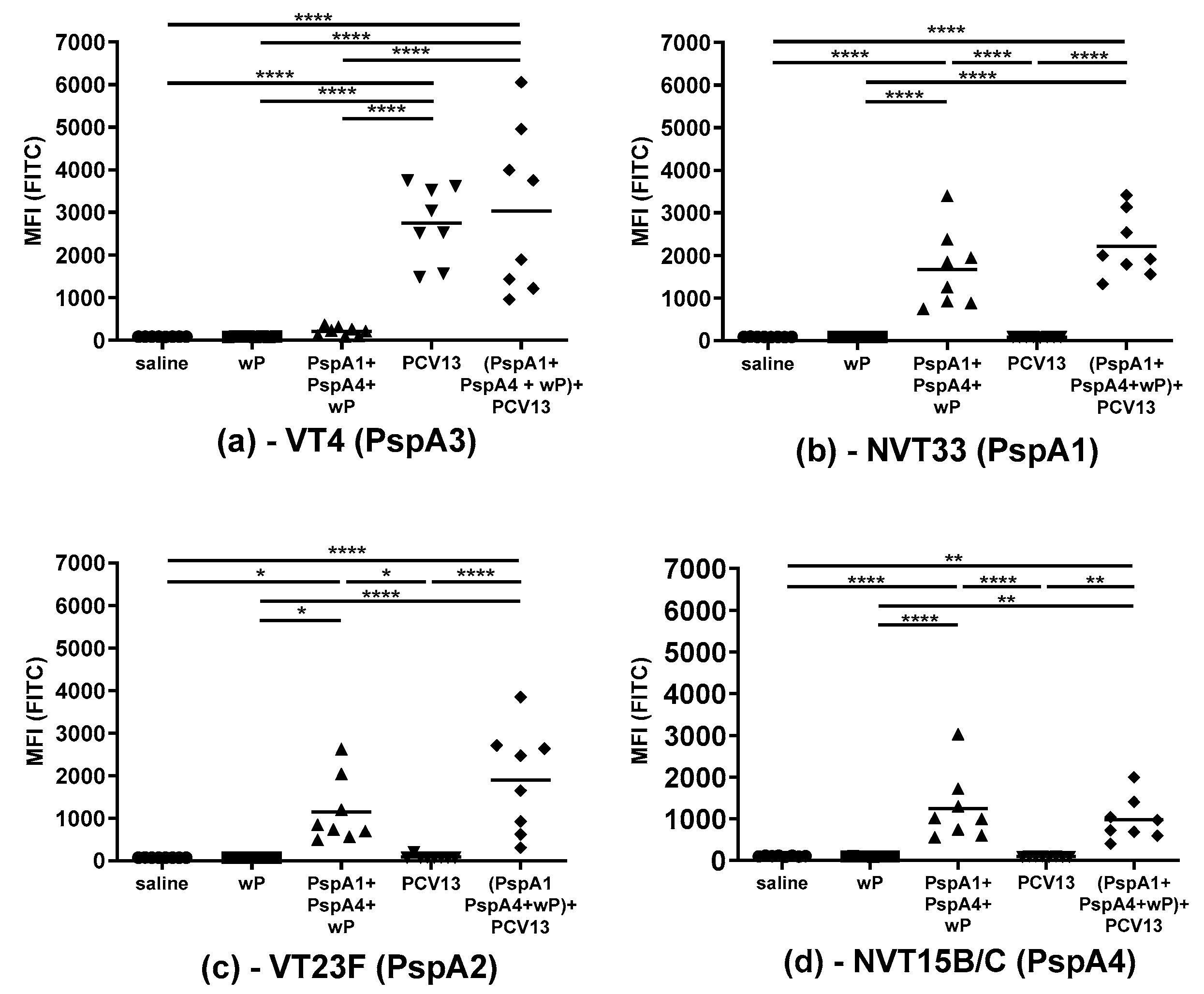
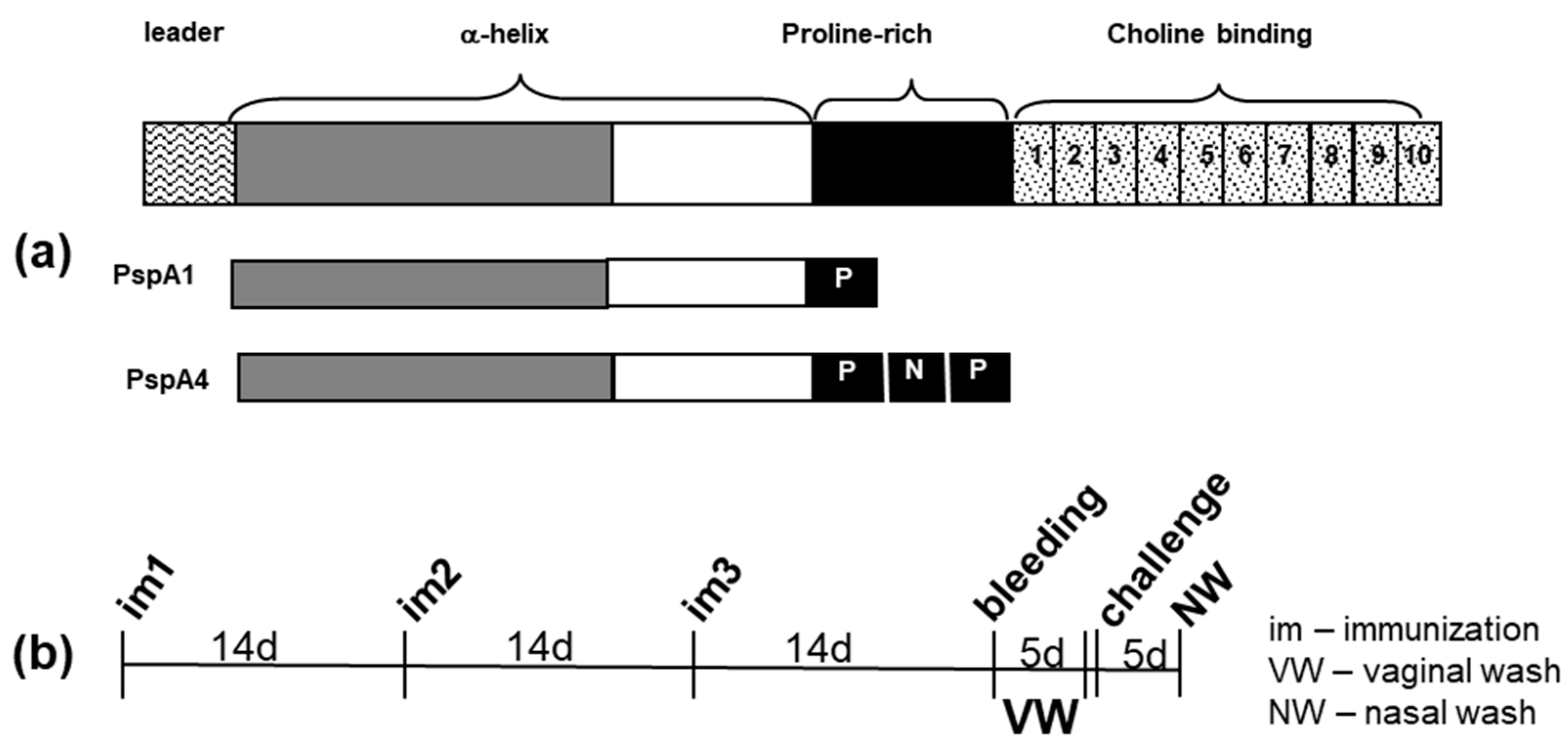
2.1. Antibody Response Induced by Immunization
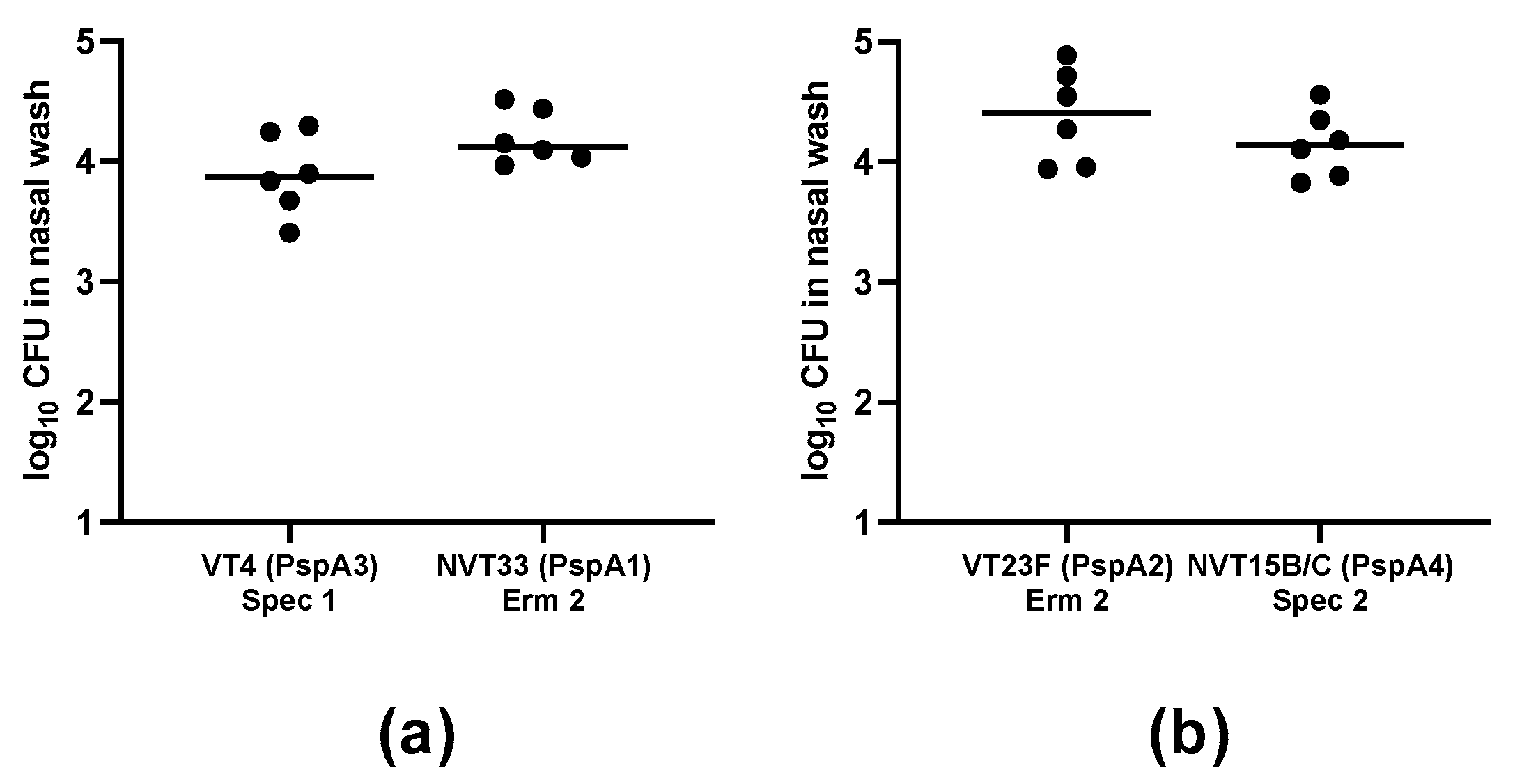
2.2. Establishment of Co-Colonization Models using VT and NVT Isolates
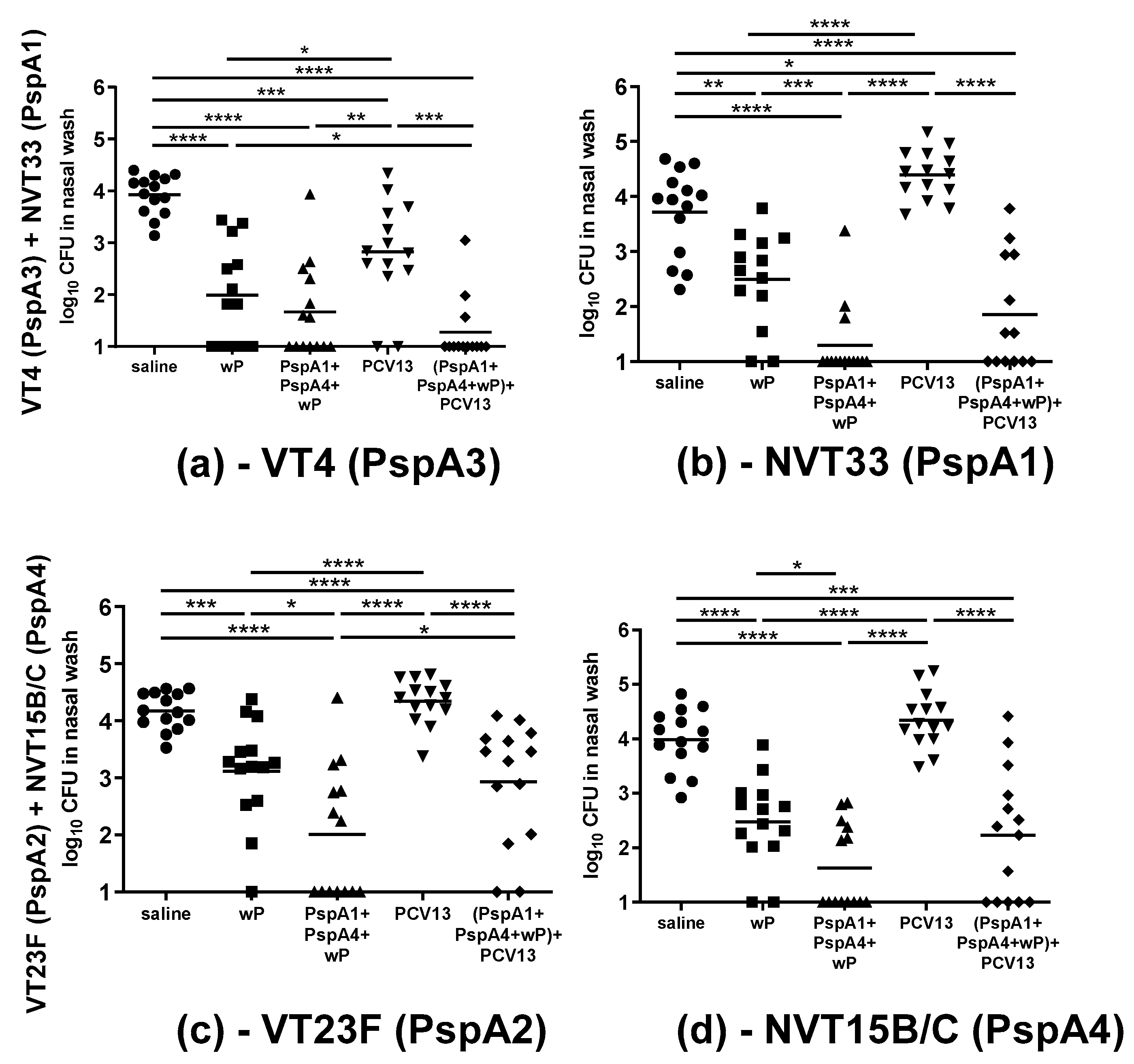
2.3. Co-Colonization Challenge
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Pneumococcal Strains
4.3. Construction of the Strains Resistant to Erm and Spec
4.4. Co-Colonization Challenge
4.5. Immunization
4.6. Measurement of Anti-PspA Antibodies by ELISA
4.7. Measurement of Anti-PS Antibodies by ELISA
4.8. Binding of Antibodies to Intact Bacteria
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Adegbola, R.A.; DeAntonio, R.; Hill, P.C.; Roca, A.; Usuf, E.; Hoet, B.; Greenwood, B.M. Carriage of Streptococcus pneumoniae and other respiratory bacterial pathogens in low and lower-middle income countries: A systematic review and meta-analysis. PLoS ONE 2014, 9, e103293. [Google Scholar] [CrossRef] [PubMed]
- Wyllie, A.L.; Chu, M.L.; Schellens, M.H.; Van Engelsdorp Gastelaars, J.; Jansen, M.D.; van der Ende, A.; Bogaert, D.; Sanders, E.A.; Trzcinski, K. Streptococcus pneumoniae in saliva of Dutch primary school children. PLoS ONE 2014, 9, e102045. [Google Scholar] [CrossRef] [PubMed]
- Turner, P.; Hinds, J.; Turner, C.; Jankhot, A.; Gould, K.; Bentley, S.D.; Nosten, F.; Goldblatt, D. Improved detection of nasopharyngeal cocolonization by multiple pneumococcal serotypes by use of latex agglutination or molecular serotyping by microarray. J. Clin. Microbiol. 2011, 49, 1784–1789. [Google Scholar] [CrossRef] [PubMed]
- Wahl, B.; O’Brien, K.L.; Greenbaum, A.; Majumder, A.; Liu, L.; Chu, Y.; Luksic, I.; Nair, H.; McAllister, D.A.; Campbell, H.; et al. Burden of Streptococcus pneumoniae and Haemophilus influenzae type b disease in children in the era of conjugate vaccinges: Global, regional, and national estimates for 2000-15. Lancet Glob. Health 2018, 6, e744–e757. [Google Scholar] [CrossRef]
- Geno, K.A.; Gilbert, G.L.; Song, J.Y.; Skovsted, I.C.; Klugman, K.P.; Jones, C.; Konradsen, H.B.; Nahm, M.H. Pneumococcal Capsules and Their Types: Past, Present, and Future. Clin. Microbiol. Rev. 2015, 28, 871–899. [Google Scholar] [CrossRef]
- Frazao, N.; Brito-Avo, A.; Simas, C.; Saldanha, J.; Mato, R.; Nunes, S.; Sousa, N.G.; Carrico, J.A.; Almeida, J.S.; Santos-Sanches, I.; et al. Effect of the seven-valent conjugate pneumococcal vaccine on carriage and drug resistance of Streptococcus pneumoniae in healthy children attending day-care centers in Lisbon. Pediatr. Infect. Dis. J. 2005, 24, 243–252. [Google Scholar] [CrossRef]
- Huang, S.S.; Platt, R.; Rifas-Shiman, S.L.; Pelton, S.I.; Goldmann, D.; Finkelstein, J.A. Post-PCV7 changes in colonizing pneumococcal serotypes in 16 Massachusetts communities, 2001 and 2004. Pediatrics 2005, 116, e408–e413. [Google Scholar] [CrossRef]
- Hausdorff, W.P.; Hanage, W.P. Interim results of an ecological experiment—Conjugate vaccination against the pneumococcus and serotype replacement. Human Vaccines Immunother. 2016, 12, 358–374. [Google Scholar] [CrossRef]
- Briles, D.E.; Ades, E.; Paton, J.C.; Sampson, J.S.; Carlone, G.M.; Huebner, R.C.; Virolainen, A.; Swiatlo, E.; Hollingshead, S.K. Intranasal immunization of mice with a mixture of the pneumococcal proteins PsaA and PspA is highly protective against nasopharyngeal carriage of Streptococcus pneumoniae. Infect. Immun. 2000, 68, 796–800. [Google Scholar] [CrossRef]
- Ogunniyi, A.D.; Folland, R.L.; Briles, D.E.; Hollingshead, S.K.; Paton, J.C. Immunization of mice with combinations of pneumococcal virulence proteins elicits enhanced protection against challenge with Streptococcus pneumoniae. Infect. Immun. 2000, 68, 3028–3033. [Google Scholar] [CrossRef]
- Briles, D.E.; Hollingshead, S.K.; Paton, J.C.; Ades, E.W.; Novak, L.; van Ginkel, F.W.; Benjamin, W.H., Jr. Immunizations with pneumococcal surface protein A and pneumolysin are protective against pneumonia in a murine model of pulmonary infection with Streptococcus pneumoniae. J. Infect. Dis. 2003, 188, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Hollingshead, S.K.; Becker, R.; Briles, D.E. Diversity of PspA: Mosaic genes and evidence for past recombination in Streptococcus pneumoniae. Infect. Immun. 2000, 68, 5889–5900. [Google Scholar] [CrossRef]
- Nabors, G.S.; Braun, P.A.; Herrmann, D.J.; Heise, M.L.; Pyle, D.J.; Gravenstein, S.; Schilling, M.; Ferguson, L.M.; Hollingshead, S.K.; Briles, D.E.; et al. Immunization of healthy adults with a single recombinant pneumococcal surface protein A (PspA) variant stimulates broadly cross-reactive antibodies to heterologous PspA molecules. Vaccine 2000, 18, 1743–1754. [Google Scholar] [CrossRef]
- Moreno, A.T.; Oliveira, M.L.; Ferreira, D.M.; Ho, P.L.; Darrieux, M.; Leite, L.C.; Ferreira, J.M., Jr.; Pimenta, F.C.; Andrade, A.L.; Miyaji, E.N. Immunization of mice with single PspA fragments induces antibodies capable of mediating complement deposition on different pneumococcal strains and cross-protection. Clin. Vaccine Immunol. 2010, 17, 439–446. [Google Scholar] [CrossRef]
- Briles, D.E.; Hollingshead, S.K.; Nabors, G.S.; Paton, J.C.; Brooks-Walter, A. The potential for using protein vaccines to protect against otitis media caused by Streptococcus pneumoniae. Vaccine 2000, 19, S87–S95. [Google Scholar] [CrossRef]
- Lipsitch, M.; Dykes, J.K.; Johnson, S.E.; Ades, E.W.; King, J.; Briles, D.E.; Carlone, G.M. Competition among Streptococcus pneumoniae for intranasal colonization in a mouse model. Vaccine 2000, 18, 2895–2901. [Google Scholar] [CrossRef]
- Trzcinski, K.; Li, Y.; Weinberger, D.M.; Thompson, C.M.; Cordy, D.; Bessolo, A.; Malley, R.; Lipsitch, M. Effect of Serotype on Pneumococcal Competition in a Mouse Colonization Model. MBio 2015, 6, e00902–e00915. [Google Scholar] [CrossRef]
- Tostes, R.O.; Rodrigues, T.C.; da Silva, J.B.; Schanoski, A.S.; Oliveira, M.L.; Miyaji, E.N. Protection Elicited by Nasal Immunization with Recombinant Pneumococcal Surface Protein A (rPspA) Adjuvanted with Whole-Cell Pertussis Vaccine (wP) against Co-Colonization of Mice with Streptococcus pneumoniae. PLoS ONE 2017, 12, e0170157. [Google Scholar] [CrossRef]
- Johansson, E.L.; Wassen, L.; Holmgren, J.; Jertborn, M.; Rudin, A. Nasal and vaginal vaccinations have differential effects on antibody responses in vaginal and cervical secretions in humans. Infect. Immun. 2001, 69, 7481–7486. [Google Scholar] [CrossRef]
- Janoff, E.N.; Rubins, J.B.; Fasching, C.; Charboneau, D.; Rahkola, J.T.; Plaut, A.G.; Weiser, J.N. Pneumococcal IgA1 protease subverts specific protection by human IgA1. Mucosal Immunol. 2014, 7, 249–256. [Google Scholar] [CrossRef]
- Kono, M.; Zafar, M.A.; Zuniga, M.; Roche, A.M.; Hamaguchi, S.; Weiser, J.N. Single Cell Bottlenecks in the Pathogenesis of Streptococcus pneumoniae. PLoS Pathog. 2016, 12, e1005887. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.K.; Lipsitch, M.; Hanage, W.P. Carriage burden, multiple colonization and antibiotic pressure promote emergence of resistant vaccine escape pneumococci. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20140342. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.L.; Miyaji, E.N.; Ferreira, D.M.; Moreno, A.T.; Ferreira, P.C.; Lima, F.A.; Santos, F.L.; Sakauchi, M.A.; Takata, C.S.; Higashi, H.G.; et al. Combination of pneumococcal surface protein A (PspA) with whole cell pertussis vaccine increases protection against pneumococcal challenge in mice. PLoS ONE 2010, 5, e10863. [Google Scholar] [CrossRef]
- Malley, R.; Morse, S.C.; Leite, L.C.; Areas, A.P.; Ho, P.L.; Kubrusly, F.S.; Almeida, I.C.; Anderson, P. Multiserotype protection of mice against pneumococcal colonization of the nasopharynx and middle ear by killed nonencapsulated cells given intranasally with a nontoxic adjuvant. Infect. Immun. 2004, 72, 4290–4292. [Google Scholar] [CrossRef] [PubMed]
- Kuipers, K.; Diavatopoulos, D.A.; van Opzeeland, F.; Simonetti, E.; van den Kieboom, C.H.; Kerstholt, M.; Borczyk, M.; van IngenSchenau, D.; Brandsma, E.T.; Netea, M.G.; et al. Antigen-Independent Restriction of Pneumococcal Density by Mucosal Adjuvant Cholera Toxin Subunit B. J. Infect. Dis. 2016. [Google Scholar] [CrossRef] [PubMed]
- Mitsi, E.; Roche, A.M.; Reine, J.; Zangari, T.; Owugha, J.T.; Pennington, S.H.; Gritzfeld, J.F.; Wright, A.D.; Collins, A.M.; van Selm, S.; et al. Agglutination by anti-capsular polysaccharide antibody is associated with protection against experimental human pneumococcal carriage. Mucosal Immunol. 2017, 10, 385–394. [Google Scholar] [CrossRef]
- Roche, A.M.; Richard, A.L.; Rahkola, J.T.; Janoff, E.N.; Weiser, J.N. Antibody blocks acquisition of bacterial colonization through agglutination. Mucosal Immunol. 2015, 8, 176–185. [Google Scholar] [CrossRef]
- Lu, Y.J.; Gross, J.; Bogaert, D.; Finn, A.; Bagrade, L.; Zhang, Q.; Kolls, J.K.; Srivastava, A.; Lundgren, A.; Forte, S.; et al. Interleukin-17A mediates acquired immunity to pneumococcal colonization. PLoS Pathog. 2008, 4, e1000159. [Google Scholar] [CrossRef]
- Zhang, Z.; Clarke, T.B.; Weiser, J.N. Cellular effectors mediating Th17-dependent clearance of pneumococcal colonization in mice. J. Clin. Investig. 2009, 119, 1899–1909. [Google Scholar] [CrossRef]
- Jochems, S.P.; Marcon, F.; Carniel, B.F.; Holloway, M.; Mitsi, E.; Smith, E.; Gritzfeld, J.F.; Solorzano, C.; Reine, J.; Pojar, S.; et al. Inflammation induced by influenza virus impairs human innate immune control of pneumococcus. Nat. Immunol. 2018, 19, 1299–1308. [Google Scholar] [CrossRef]
- Jochems, S.P.; de Ruiter, K.; Solorzano, C.; Voskamp, A.; Mitsi, E.; Nikolaou, E.; Carniel, B.F.; Pojar, S.; German, E.L.; Reine, J.; et al. Innate and adaptive nasal mucosal immune responses following experimental human pneumococcal colonization. J. Clin. Investig. 2019, 130, 4523–4538. [Google Scholar] [CrossRef] [PubMed]
- Ren, B.; Szalai, A.J.; Hollingshead, S.K.; Briles, D.E. Effects of PspA and antibodies to PspA on activation and deposition of complement on the pneumococcal surface. Infect. Immun. 2004, 72, 114–122. [Google Scholar] [CrossRef]
- Habets, M.N.; van Selm, S.; van der Gaast-de Jongh, C.E.; Diavatopoulos, D.A.; de Jonge, M.I. A novel flow cytometry-based assay for the quantification of antibody-dependent pneumococcal agglutination. PLoS ONE 2017, 12, e0170884. [Google Scholar] [CrossRef]
- Usuf, E.; Bottomley, C.; Bojang, E.; Cox, I.; Bojang, A.; Gladstone, R.; Kampmann, B.; Hill, P.C.; Roca, A. Persistence of Nasopharyngeal Pneumococcal Vaccine Serotypes and Increase of Nonvaccine Serotypes Among Vaccinated Infants and Their Mothers 5 Years After Introduction of Pneumococcal Conjugate Vaccine 13 in The Gambia. Clin. Infect. Dis. 2019, 68, 1512–1521. [Google Scholar] [CrossRef] [PubMed]
- Marks, L.R.; Reddinger, R.M.; Hakansson, A.P. High levels of genetic recombination during nasopharyngeal carriage and biofilm formation in Streptococcus pneumoniae. MBio 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Pai, R.; Gertz, R.E.; Beall, B. Sequential multiplex PCR approach for determining capsular serotypes of Streptococcus pneumoniae isolates. J. Clin. Microbiol. 2006, 44, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Aida, Y.; Pabst, M.J. Removal of endotoxin from protein solutions by phase separation using Triton X-114. J. Immunol. Methods 1990, 132, 191–195. [Google Scholar] [CrossRef]
- Teodorowicz, M.; Perdijk, O.; Verhoek, I.; Govers, C.; Savelkoul, H.F.; Tang, Y.; Wichers, H.; Broersen, K. Optimized Triton X-114 assisted lipopolysaccharide (LPS) removal method reveals the immunomodulatory effect of food proteins. PLoS ONE 2017, 12, e0173778. [Google Scholar] [CrossRef]
- Caro-Aguilar, I.; Indrawati, L.; Kaufhold, R.M.; Gaunt, C.; Zhang, Y.; Nawrocki, D.K.; Giovarelli, C.; Winters, M.A.; Smith, W.J.; Heinrichs, J.; et al. Immunogenicity differences of a 15-valent pneumococcal polysaccharide conjugate vaccine (PCV15) based on vaccine dose, route of immunization and mouse strain. Vaccine 2017, 35, 865–872. [Google Scholar] [CrossRef]
| Isolate | Identification | Serotype/Serogroup | VT/NVT | PspA Family | PspA Clade |
|---|---|---|---|---|---|
| TIGR4 | VT4 (PspA3) | 4 | VT | 2 | 3 |
| 368/06 | VT23F (PspA2) | 23F | VT | 1 | 2 |
| 4431/119 | NVT33 (PspA1) | 33 | NVT | 1 | 1 |
| 237/53 | NVT15B/C (PspA4) | 15B/C | NVT | 2 | 4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Colichio, G.B.C.; Oliveira, G.S.; Rodrigues, T.C.; Oliveira, M.L.S.; Miyaji, E.N. Efficacy of a Protein Vaccine and a Conjugate Vaccine Against Co-Colonization with Vaccine-Type and Non-Vaccine Type Pneumococci in Mice. Pathogens 2020, 9, 278. https://doi.org/10.3390/pathogens9040278
Colichio GBC, Oliveira GS, Rodrigues TC, Oliveira MLS, Miyaji EN. Efficacy of a Protein Vaccine and a Conjugate Vaccine Against Co-Colonization with Vaccine-Type and Non-Vaccine Type Pneumococci in Mice. Pathogens. 2020; 9(4):278. https://doi.org/10.3390/pathogens9040278
Chicago/Turabian StyleColichio, Gabriela B. C., Giuliana S. Oliveira, Tasson C. Rodrigues, Maria Leonor S. Oliveira, and Eliane N. Miyaji. 2020. "Efficacy of a Protein Vaccine and a Conjugate Vaccine Against Co-Colonization with Vaccine-Type and Non-Vaccine Type Pneumococci in Mice" Pathogens 9, no. 4: 278. https://doi.org/10.3390/pathogens9040278
APA StyleColichio, G. B. C., Oliveira, G. S., Rodrigues, T. C., Oliveira, M. L. S., & Miyaji, E. N. (2020). Efficacy of a Protein Vaccine and a Conjugate Vaccine Against Co-Colonization with Vaccine-Type and Non-Vaccine Type Pneumococci in Mice. Pathogens, 9(4), 278. https://doi.org/10.3390/pathogens9040278






