Detection of Antibodies against Mycobacterium bovis in Oral Fluid from Eurasian Wild Boar
Abstract
1. Introduction
2. Results
2.1. Descriptive Analysis
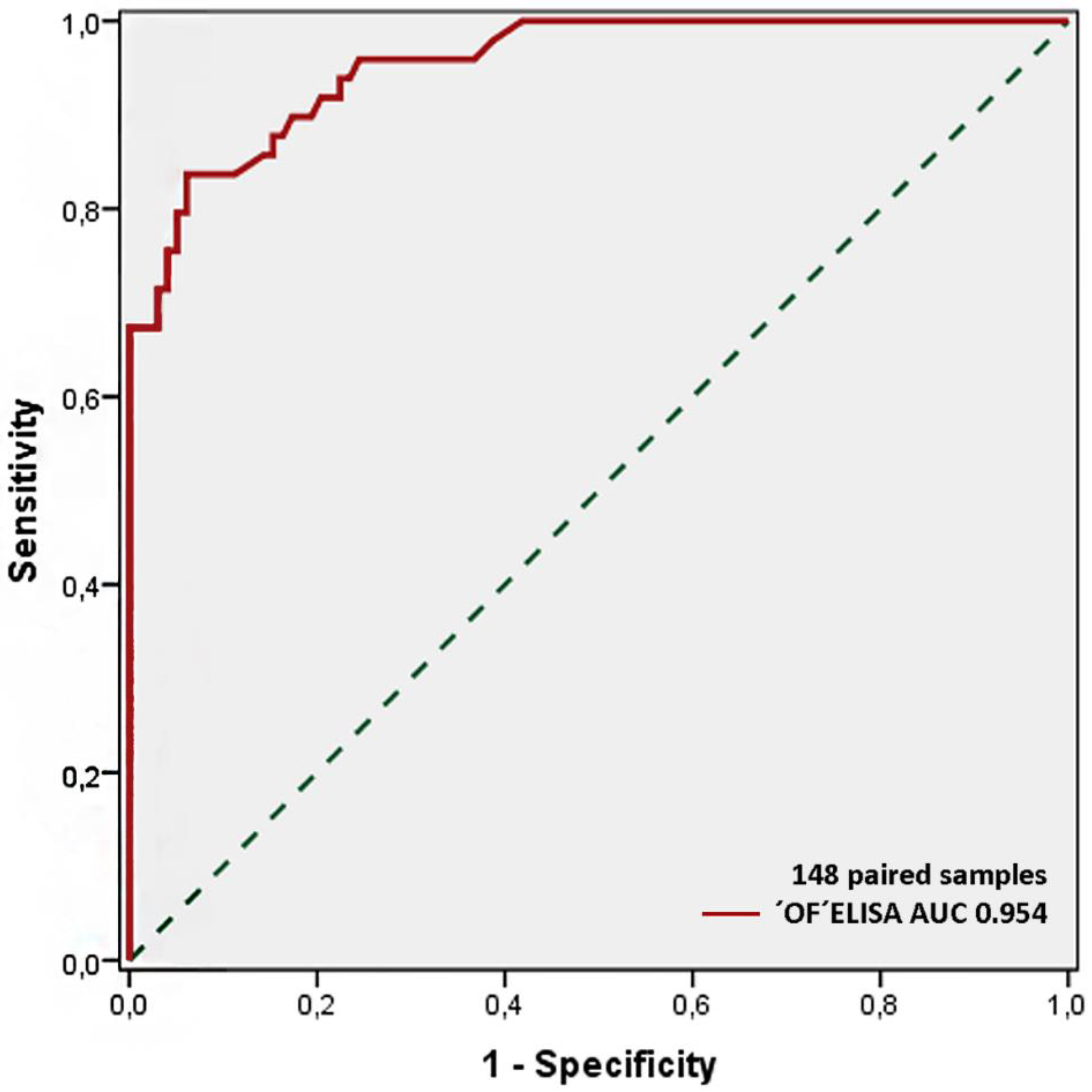
2.2. OF ELISA Validation
3. Discussion
4. Material and Methods
4.1. Animal Sampling
4.2. Study Design
4.3. Serum Sample Collection and Testing (Serum ELISA)
4.4. Oral Fluid Sample Collection and Testing (OF ELISA)
4.5. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brosch, R.; Gordon, S.V.; Marmiesse, M.; Brodin, P.; Buchrieser, C.; Eiglmeier, K.; Garnier, T.; Gutierrez, C.; Hewinson, G.; Kremer, K.; et al. A new evolutionary scenario for the Mycobacterium tuberculosis complex. Proc. Natl. Acad. Sci. USA 2002, 99, 3684–3689. [Google Scholar] [CrossRef]
- Fitzgerald, S.D.; Kaneene, J.B. Wildlife reservoirs of bovine tuberculosis worldwide: Hosts, pathology, surveillance, and control. Vet. Pathol. 2013, 50, 488–499. [Google Scholar] [CrossRef]
- Cowie, C.E.; Hutchings, M.R.; Barasona, J.A.; Gortázar, C.; Vicente, J.; White, P.C.L. Interactions between four species in a complex wildlife: Livestock disease community: Implications for Mycobacterium bovis maintenance and transmission. Eur. J. Wildl. Res. 2016, 62, 51–64. [Google Scholar] [CrossRef]
- Kukielka, E.; Barasona, J.A.; Cowie, C.E.; Drewe, J.A.; Gortazar, C.; Cotarelo, I.; Vicente, J. Spatial and temporal interactions between livestock and wildlife in South Central Spain assessed by camera traps. Prev. Vet. Med. 2013, 112, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Gortázar, C.; Delahay, R.J.; McDonald, R.A.; Boadella, M.; Wilson, G.J.; Gavier-Widen, D.; Acevedo, P. The status of tuberculosis in European wild mammals. Mammal Rev. 2012, 42, 193–206. [Google Scholar] [CrossRef]
- Hardstaff, J.L.; Marion, G.; Hutchings, M.R.; White, P.C. Evaluating the tuberculosis hazard posed to cattle from wildlife across Europe. Res. Vet. Sci. 2014, 97, S86–S93. [Google Scholar] [CrossRef]
- Rodríguez-Prieto, V.; Martinez-Lopez, B.; Barasona, J.; Acevedo, P.; Romero, B.; Rodriguez-Campos, S.; Gortázar, C.; Sánchez-Vizcaíno, J.; Vicente, J. A Bayesian approach to study the risk variables for tuberculosis occurrence in domestic and wild ungulates in South Central Spain. BMC Vet. Res. 2012, 8, 148. [Google Scholar] [CrossRef]
- Naranjo, V.; Gortazar, C.; Vicente, J.; de la Fuente, J. Evidence of the role of European wild boar as a reservoir of Mycobacterium tuberculosis complex. Vet. Microbiol. 2008, 127, 1–9. [Google Scholar] [CrossRef]
- Barasona, J.A.; Gortazar, C.; de la Fuente, J.; Vicente, J. Host richness increases tuberculosis disease risk in game-managed areas. Microorganisms 2019, 7, 182. [Google Scholar] [CrossRef]
- Acevedo, P.; Escudero, M.A.; Muńoz, R.; Gortázar, C. Factors affecting wild boar abundance across an environmental gradient in Spain. Acta Theriol. 2006, 51, 327–336. [Google Scholar] [CrossRef]
- Gortázar, C.; Herrero, J.; Villafuerte, R.; Marco, J. Historical examination of the status of large mammals in Aragon, Spain. Mammalia 2000, 64, 411–422. [Google Scholar] [CrossRef]
- Massei, G.; Kindberg, J.; Licoppe, A.; Gacic, D.; Sprem, N.; Kamler, J.; Baubet, E.; Hohmann, U.; Monaco, A.; Ozolins, J.; et al. Wild boar populations up, numbers of hunters down? A review of trends and implications for Europe. Pest Manag. Sci. 2015, 71, 492–500. [Google Scholar] [CrossRef] [PubMed]
- Triguero-Ocana, R.; Barasona, J.A.; Carro, F.; Soriguer, R.C.; Vicente, J.; Acevedo, P. Spatio-temporal trends in the frequency of interspecific interactions between domestic and wild ungulates from Mediterranean Spain. PLoS ONE 2019, 14, e0211216. [Google Scholar] [CrossRef] [PubMed]
- Boadella, M.; Lyashchenko, K.; Greenwald, R.; Esfandiari, J.; Jaroso, R.; Carta, T.; Garrido, J.; Vicente, J.; de la Fuente, J.; Gortázar, C. Serologic tests for detecting antibodies against mycobacterium bovis and mycobacterium avium subspecies paratuberculosis in Eurasian wild boar (Sus Scrofa Scrofa). J. Vet. Diagn. Investig. 2011, 23, 77–83. [Google Scholar] [CrossRef]
- Ministerio de Agricultura, Pesca y Alimentacion. Manual Práctico de Operaciones en el Control de las Enfermedades de la Fauna Silvestre; Ministerio de Agricultura, Pesca y Alimentacion: Madrid, Spain, 2019.
- Palmer, M.V. Mycobacterium bovis: Characteristics of wildlife reservoir hosts. Transbound. Emerg. Dis. 2013, 60 (Suppl. 1), 1–13. [Google Scholar] [CrossRef] [PubMed]
- Che’ Amat, A.; Gonzalez-Barrio, D.; Ortiz, J.A.; Diez-Delgado, I.; Boadella, M.; Barasona, J.A.; Bezos, J.; Romero, B.; Armenteros, J.A.; Lyashchenko, K.P.; et al. Testing Eurasian wild boar piglets for serum antibodies against Mycobacterium bovis. Prev. Vet. Med. 2015, 121, 93–98. [Google Scholar] [CrossRef]
- Gortazar, C.; Diez-Delgado, I.; Barasona, J.A.; Vicente, J.; De La Fuente, J.; Boadella, M. The wild side of disease control at the wildlife-livestock-human interface: A review. Front. Vet. Sci. 2015, 1, 27. [Google Scholar] [CrossRef]
- OIE. Bovine Tuberculosis; OIE: Paris, France, 2009. [Google Scholar]
- Gavin, W.; Jennifer, B.; Mark, C.; Richard, C.-H.; Tim, C.; de la Fuente, J.; Richard, D.; Dolores, G.-W.; Christian, G.; Glyn, H.; et al. Scientific review on Tuberculosis in wildlife in the EU. EFSA Support. Publ. 2009, 6, 12E. [Google Scholar] [CrossRef]
- Barasona, J.A.; López-Olvera, J.R.; Beltrán-Beck, B.; Gortázar, C.; Vicente, J. Trap-effectiveness and response to tiletamine-zolazepam and medetomidine anaesthesia in Eurasian wild boar captured with cage and corral traps. BMC Vet. Res. 2013, 9, 107. [Google Scholar] [CrossRef]
- Fournier-Chambrillon, C.; Fournier, P.; Vié, J.-C. Immobilization of Wild Collared Anteaters with Ketamine- and Xylazine-Hydrochloride. J. Wildl. Dis. 1997, 33, 795–800. [Google Scholar] [CrossRef]
- Mouchantat, S.; Globig, A.; Bohle, W.; Petrov, A.; Strebelow, H.G.; Mettenleiter, T.C.; Depner, K. Novel rope-based sampling of classical swine fever shedding in a group of wild boar showing low contagiosity upon experimental infection with a classical swine fever field strain of genotype 2.3. Vet. Microbiol. 2014, 170, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Mouchantat, S.; Haas, B.; Bohle, W.; Globig, A.; Lange, E.; Mettenleiter, T.C.; Depner, K. Proof of principle: Non-invasive sampling for early detection of foot-and-mouth disease virus infection in wild boar using a rope-in-a-bait sampling technique. Vet. Microbiol. 2014, 172, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Henao-Diaz, A.; Giménez-Lirola, L.; Magtoto, R.; Ji, J.; Zimmerman, J. Evaluation of three commercial porcine reproductive and respiratory syndrome virus (PRRSV) oral fluid antibody ELISAs using samples of known status. Res. Vet. Sci. 2019, 125, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Prickett, J.R.; Kim, W.; Simer, R.; Yoon, K.J.; Zimmerman, J. Oral-fluid samples for surveillance of commercial growing pigs for porcine reproductive and respiratory syndrome virus and porcine circovirus type 2 infections. J. Swine Health Prod. 2008, 16, 86–91. [Google Scholar]
- Mur, L.; Gallardo, C.; Soler, A.; Zimmermman, J.; Pelayo, V.; Nieto, R.; Sanchez-Vizcaino, J.M.; Arias, M. Potential use of oral fluid samples for serological diagnosis of African swine fever. Vet. Microbiol. 2013, 165, 135–139. [Google Scholar] [CrossRef]
- Kaufman, E.; Lamster, I.B. The diagnostic applications of saliva—A review. Crit. Rev. Oral Biol. Med. 2002, 13, 197–212. [Google Scholar] [CrossRef]
- Pol, F.; Dorenlor, V.; Eono, F.; Eudier, S.; Eveno, E.; Liégard-Vanhecke, D.; Rose, N.; Fablet, C. Individual and pen-based oral fluid sampling: A welfare-friendly sampling method for group-housed gestating sows. Prev. Vet. Med. 2017, 147, 58–65. [Google Scholar] [CrossRef]
- Rotolo, M.L.; Sun, Y.; Wang, C.; Gimenez-Lirola, L.; Baum, D.H.; Gauger, P.C.; Harmon, K.M.; Hoogland, M.; Main, R.; Zimmerman, J.J. Sampling guidelines for oral fluid-based surveys of group-housed animals. Vet. Microbiol. 2017, 209, 20–29. [Google Scholar] [CrossRef]
- Barasona, J.A.; Torres, M.J.; Aznar, J.; Gortázar, C.; Vicente, J. DNA detection reveals mycobacterium tuberculosis complex shedding routes in its wildlife reservoir the eurasian wild boar. Transbound. Emerg. Dis. 2017, 64, 906–915. [Google Scholar] [CrossRef]
- Fredriksson-Ahomaa, M. Wild boar: A reservoir of foodborne zoonoses. Foodborne Pathog. Dis. 2019, 16, 153–165. [Google Scholar] [CrossRef]
- Chambers, M.A. Review of the diagnosis and study of tuberculosis in non-bovine wildlife species using immunological methods. Transbound. Emerg. Dis. 2009, 56, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Cardoso-Toset, F.; Luque, I.; Carrasco, L.; Jurado-Martos, F.; Risalde, M.A.; Venteo, A.; Infantes-Lorenzo, J.A.; Bezos, J.; Rueda, P.; Tapia, I.; et al. Evaluation of five serologic assays for bovine tuberculosis surveillance in domestic free-range pigs from southern Spain. Prev. Vet. Med. 2017, 137, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Garrido, J.M.; Vicente, J.; Carrasco-Garcia, R.; Galindo, R.C.; Minguijon, E.; Ballesteros, C.; Aranaz, A.; Romero, B.; Sevilla, I.; Juste, R.; et al. Experimental infection of Eurasian wild boar with Mycobacterium avium subsp. avium. Vet. Microbiol. 2010, 144, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Crawshaw, T.R.; Griffiths, I.B.; Clifton-Hadley, R.S. Comparison of a standard and a detailed postmortem protocol for detecting Mycobacterium bovis in badgers. Vet. Rec. 2008, 163, 473–477. [Google Scholar] [CrossRef]
- Corner, L.A.; Trajstman, A.C.; Lund, K. Determination of the optimum concentration of decontaminants for the primary isolation of Mycobacterium bovis. N. Z. Vet. J. 1995, 43, 129–133. [Google Scholar] [CrossRef]
- Jaroso, R.; Vicente, J.; Fernandez-de-Mera, I.G.; Aranaz, A.; Gortazar, C. Eurasian wild boar response to skin-testing with mycobacterial and non-mycobacterial antigens. Prev. Vet. Med. 2010, 96, 211–217. [Google Scholar] [CrossRef]
- Wisselink, H.J.; van Solt-Smits, C.B.; Oorburg, D.; van Soolingen, D.; Overduin, P.; Maneschijn-Bonsing, J.; Stockhofe-Zurwieden, N.; Buys-Bergen, H.; Engel, B.; Urlings, B.A.; et al. Serodiagnosis of Mycobacterium avium infections in pigs. Vet. Microbiol. 2010, 142, 401–407. [Google Scholar] [CrossRef]
- Kittawornrat, A.; Panyasing, Y.; Goodell, C.; Wang, C.; Gauger, P.; Harmon, K.; Rauh, R.; Desfresne, L.; Levis, I.; Zimmerman, J. Porcine reproductive and respiratory syndrome virus (PRRSV) surveillance using pre-weaning oral fluid samples detects circulation of wild-type PRRSV. Vet. Microbiol. 2014, 168, 331–339. [Google Scholar] [CrossRef]
- Prickett, J.R.; Zimmerman, J.J. The development of oral fluid-based diagnostics and applications in veterinary medicine. Anim. Health Res. Rev. 2010, 11, 207–216. [Google Scholar] [CrossRef]
- Dietze, K.; Tucakov, A.; Engel, T.; Wirtz, S.; Depner, K.; Globig, A.; Kammerer, R.; Mouchantat, S. Rope-based oral fluid sampling for early detection of classical swine fever in domestic pigs at group level. BMC Vet. Res. 2017, 13, 5. [Google Scholar] [CrossRef]
- Ramirez, A.; Wang, C.; Prickett, J.R.; Pogranichniy, R.; Yoon, K.J.; Main, R.; Johnson, J.K.; Rademacher, C.; Hoogland, M.; Hoffmann, P.; et al. Efficient surveillance of pig populations using oral fluids. Prev. Vet. Med. 2012, 104, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Beltrán-Beck, B.; Romero, B.; Boadella, M.; Casal, C.; Bezos, J.; Mazariegos, M.; Martín, M.; Galindo, R.C.; Pérez de la Lastra, J.M.; Villar, M.; et al. Tonsils of the soft palate do not mediate the response of pigs to oral vaccination with heat-inactivated Mycobacterium bovis. Clin. Vaccine Immunol. 2014, 21, 1128–1136. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.; Infantes-Lorenzo, J.A.; Moreno, I.; Cano-Terriza, D.; de Juan, L.; García-Bocanegra, I.; Domínguez, L.; Domínguez, M.; Gortázar, C.; Risalde, M.A. Validation of a new serological assay for the identification of Mycobacterium tuberculosis complex-specific antibodies in pigs and wild boar. Prev. Vet. Med. 2019, 162, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Leeflang, M.M.G.; Bossuyt, P.M.M.; Irwig, L. Diagnostic test accuracy may vary with prevalence: Implications for evidence-based diagnosis. J. Clin. Epidemiol. 2009, 62, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Vicente, J.; Barasona, J.A.; Acevedo, P.; Ruiz-Fons, J.F.; Boadella, M.; Diez-Delgado, I.; Beltran-Beck, B.; González-Barrio, D.; Queirós, J.; Montoro, V.; et al. Temporal trend of tuberculosis in wild ungulates from Mediterranean Spain. Transbound. Emerg. Dis. 2013, 60 (Suppl. 1), 92–103. [Google Scholar] [CrossRef] [PubMed]
- Olsen, C.; Wang, C.; Christopher-Hennings, J.; Doolittle, K.; Harmon, K.; Abate, S.; Kittawornrat, A.; Lizano, S.; Main, R.; Nelson, E.; et al. Probability of detecting Porcine reproductive and respiratory syndrome virus infection using pen-based swine oral fluid specimens as a function of within-pen prevalence. J. Vet. Diagn. Investig. 2013, 25. [Google Scholar] [CrossRef] [PubMed]
- Parikh, R.; Mathai, A.; Parikh, S.; Chandra Sekhar, G.; Thomas, R. Understanding and using sensitivity, specificity and predictive values. Indian J. Ophthalmol. 2008, 56, 45–50. [Google Scholar] [CrossRef]
- Che’Amat, A.; Armenteros, J.A.; Gonzalez-Barrio, D.; Lima, J.F.; Diez-Delgado, I.; Barasona, J.A.; Romero, B.; Lyashchenko, K.P.; Ortiz, J.A.; Gortazar, C. Is targeted removal a suitable means for tuberculosis control in wild boar? Prev. Vet. Med. 2016, 135, 132–135. [Google Scholar] [CrossRef]
- LaHue, N.P.; Banos, J.V.; Acevedo, P.; Gortazar, C.; Martinez-Lopez, B. Spatially explicit modeling of animal tuberculosis at the wildlife-livestock interface in Ciudad Real province, Spain. Prev. Vet. Med. 2016, 128, 101–111. [Google Scholar] [CrossRef]
- Martinez-Lopez, B.; Barasona, J.A.; Gortazar, C.; Rodriguez-Prieto, V.; Sanchez-Vizcaino, J.M.; Vicente, J. Farm-level risk factors for the occurrence, new infection or persistence of tuberculosis in cattle herds from South-Central Spain. Prev. Vet. Med. 2014, 116, 268–278. [Google Scholar] [CrossRef]
- Gonçalves, M.S.S.; Gil-Delgado, J.A.; Gosálvez, R.U.; López-Iborra, G.M.; Ponz, A.; Velasco, Á. Seasonal differences in drivers of species richness of waders in inland wetlands of La Mancha Húmeda Biosphere Reserve. Aquat. Conserv. Mar. Freshw. Ecosyst. 2018, 28, 1414–1423. [Google Scholar] [CrossRef]
- Dove, C.R.; Alworth, L.C. Blood collection from the orbital sinus of swine. Lab. Anim. (N. Y.) 2015, 44, 383–384. [Google Scholar] [CrossRef] [PubMed]
- Fawcett, T. Introduction to ROC analysis. Pattern Recognit. Lett. 2006, 27, 861–874. [Google Scholar] [CrossRef]
- Youden, W.J. Index for rating diagnostic tests. Cancer 1950, 3, 32–35. [Google Scholar] [CrossRef]
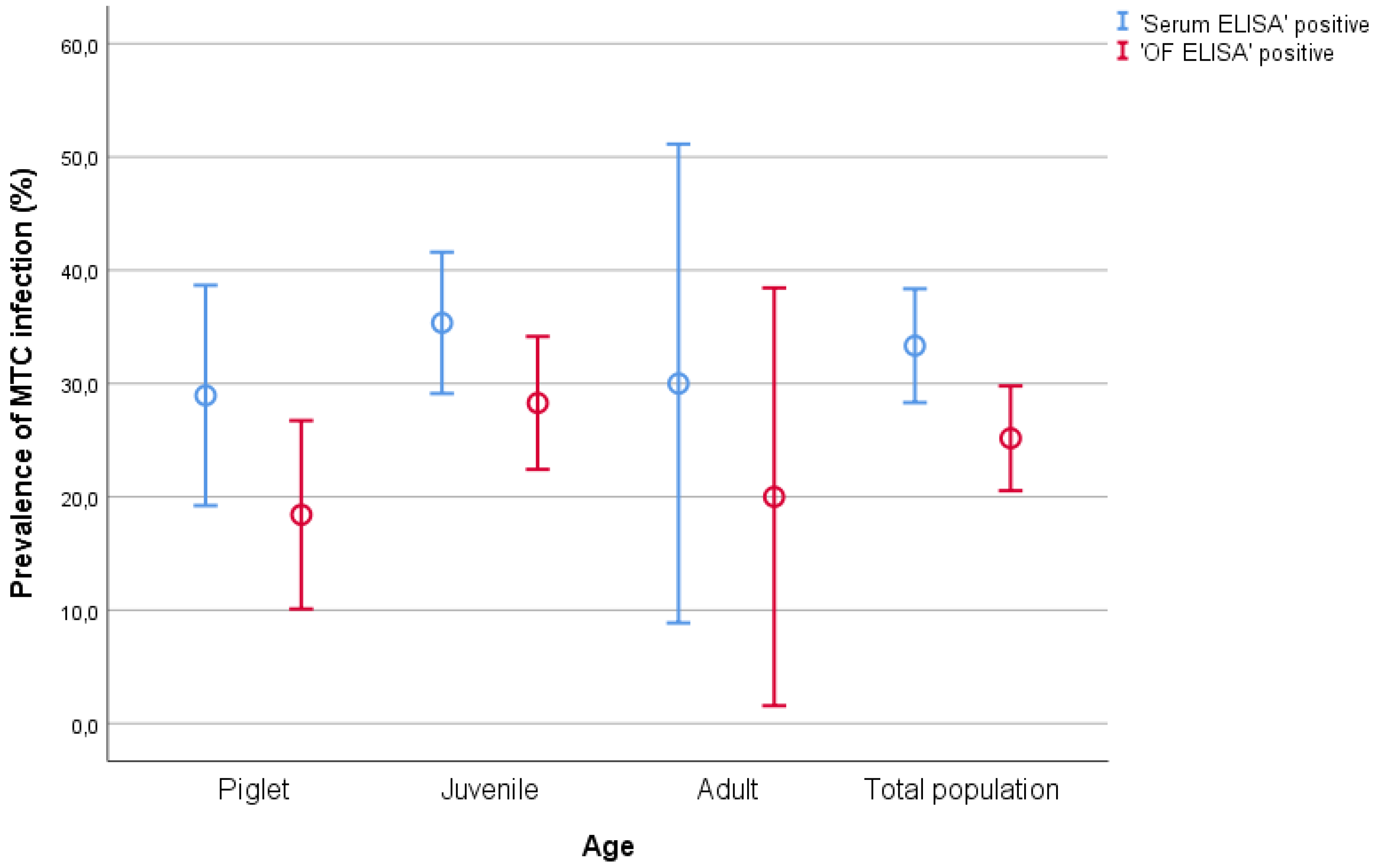
| Age | Serum Test Positive (Prevalence) | OF Test Positive (Prevalence) | Total Sampled |
|---|---|---|---|
| Piglet | 11 (28.2%) | 7 (17.9%) | 39 |
| Juvenile | 35 (35.3%) | 28 (28.8%) | 99 |
| Adult | 3 (30%) | 2 (20%) | 10 |
| Total population | 49 (33.1%) | 37 (25%) | 148 |
| Sample Type | Reference Serum Positive | Reference Serum Negative | Total Test Results |
|---|---|---|---|
| Oral fluid ELISA test positive | 37 (TP) | 0 (FP) | 37 (total test positives) |
| Oral fluid ELISA test negative | 12 (FN) | 99 (TN) | 111 (total test negatives) |
| Total samples analyzed | 49 | 99 | 148 (total population) |
| Area | Sampling Period | Type of Sample | No. of Animals | Total No. of Animals |
|---|---|---|---|---|
| TB-endemic site (Sevilla) | February 2017 | Serum | 24 | 133 |
| Oral fluid | ||||
| June 2017 | Serum | 66 | ||
| Oral fluid | ||||
| November 2017 | Serum | 43 | ||
| Oral fluid | ||||
| TB-free site (Guadiana Valley) | June 2017 | Serum Oral fluid | 15 | 15 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barasona, J.A.; Barroso-Arévalo, S.; Rivera, B.; Gortázar, C.; Sánchez-Vizcaíno, J.M. Detection of Antibodies against Mycobacterium bovis in Oral Fluid from Eurasian Wild Boar. Pathogens 2020, 9, 242. https://doi.org/10.3390/pathogens9040242
Barasona JA, Barroso-Arévalo S, Rivera B, Gortázar C, Sánchez-Vizcaíno JM. Detection of Antibodies against Mycobacterium bovis in Oral Fluid from Eurasian Wild Boar. Pathogens. 2020; 9(4):242. https://doi.org/10.3390/pathogens9040242
Chicago/Turabian StyleBarasona, Jose A., Sandra Barroso-Arévalo, Belén Rivera, Christian Gortázar, and Jose M. Sánchez-Vizcaíno. 2020. "Detection of Antibodies against Mycobacterium bovis in Oral Fluid from Eurasian Wild Boar" Pathogens 9, no. 4: 242. https://doi.org/10.3390/pathogens9040242
APA StyleBarasona, J. A., Barroso-Arévalo, S., Rivera, B., Gortázar, C., & Sánchez-Vizcaíno, J. M. (2020). Detection of Antibodies against Mycobacterium bovis in Oral Fluid from Eurasian Wild Boar. Pathogens, 9(4), 242. https://doi.org/10.3390/pathogens9040242





