Abstract
Interferons (IFN) are crucial for the innate immune response. Slightly more than two decades ago, a new type of IFN was discovered: the lambda IFN (type III IFN). Like other IFN, the type III IFN display antiviral activity against a wide variety of infections, they induce expression of antiviral, interferon-stimulated genes (MX1, OAS, IFITM1), and they have immuno-modulatory activities that shape adaptive immune responses. Unlike other IFN, the type III IFN signal through distinct receptors is limited to a few cell types, primarily mucosal epithelial cells. As a consequence of their greater and more durable production in nasal and respiratory tissues, they can determine the outcome of respiratory infections. This review is focused on the role of IFN-λ in the pathogenesis of respiratory viral infections, with influenza as a prime example. The influenza virus is a major public health problem, causing up to half a million lethal infections annually. Moreover, the virus has been the cause of four pandemics over the last century. Although IFN-λ are increasingly being tested in antiviral therapy, they can have a negative influence on epithelial tissue recovery and increase the risk of secondary bacterial infections. Therefore, IFN-λ expression deserves increased scrutiny as a key factor in the host immune response to infection.
1. Introduction
1.1. Virus Entry Triggers Host Signaling Responses
In viral infection, the protective barriers are host skin and mucous membranes. In the initial stages of a viral infection, quick activation of a non-specific immune response occurs in response to the infiltration in a cell (Figure 1). Cells utilize various pattern recognition receptors (PRR) to detect viral particles. Toll-like receptors (TLR) are one such sensor. RIG-I-like receptors (RLR), Nod-like receptors, and cytosolic nucleic acid sensors become involved when viral particles enter the cytoplasm [1]. To date, ten TLR types have been found in humans. It is well known that TLR-3, TLR-7, TLR-8, and TLR-9 are localized in endosomes, while others reside on the outer surface of the cytoplasmic membrane [1,2]. Signaling through TLR requires the use of various combinations of the following protein adapters: MyD88, Mal, TRIF, and TRAM [2]. Activation of molecular adapters is aimed at regulating the activity of the NFκB, IRF-3, and IRF-7 transcription factors, and activation of MAPK-dependent signaling pathways. The combined action of these transcription factors with the AP-1 protein effectively induces the expression of target genes [1,3].
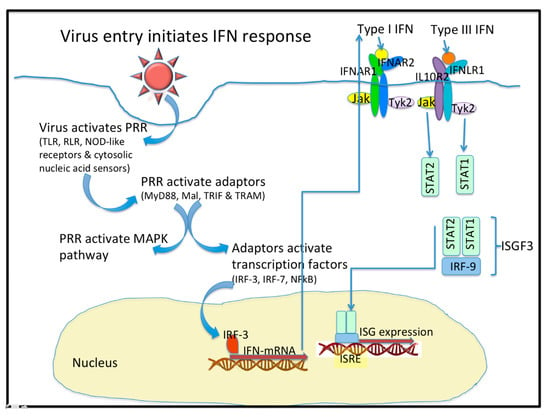
Figure 1.
Virus entry initiates the interferon (IFN) response. Upon entering a cell, virus activates Pattern Recognition Receptors (PRR) that include the Toll-like receptors (TLR), the RIG-I-like receptors (RLR), the NOD-like receptors, and some cytosolic nucleic acid receptors. These PRR activate adaptor molecules like MyD88, Mal, TRIF, and TRAM. PRR also activate the MAPK pathway. The Adaptor molecules activate Transcription factors like IRF-3, IRF-7 and NF-kB. IFN mRNA are transcribed and they express the type I, type II (not shown), and type III IFN. Type I and Type III IFN bind to distinct receptors that activate similar signaling pathways and transcriptional responses. The heterodimeric IFN receptors signal through the JAK/STAT pathways to form a complex with IRF-9 to initiate the expression of hundreds of Interferon stimulated genes (ISG). The ISG are peptidic antivirals that interfere with virus replication.
Cells are capable of TLR-independent responses to pathogen infiltration, and such responses are mediated through cytosolic sensors. The most important sensors are RNA helicases that belong to the RLR family: RIG-I, MDA-5 and LGP-2 [1]. The activated multimeric forms of RIG-I or MDA-5 are able to interact with the MAVS protein adapter located on the outer mitochondrial membrane or in peroxisomes [1]. Viral dsRNA activates both RIG-I and MDA-5. RNA containing a 5′-triphosphate end, without a cap structure, can also activate RIG-I. The MAVS protein adapter plays the role of a scaffold protein and is involved in the recruitment of signaling cascade components aimed at activating both NFκB and IRF-3 [3].
1.2. IFN Are Class II Cytokines
Class II cytokines are an extensive family of protein mediators that have similar gene structure, receptor structure and common signaling pathways. Four types of cytokines are commonly assigned to this family: “IL-10-like” cytokines; and the type I, II, and III interferons (IFN) [4]. Class II cytokine receptors are heterodimers and consist of a subunit featuring high ligand affinity (usually referred to as R1) and a low-affinity subunit (R2). Both subunits, however, are necessary for signal transmission [4]. IFN play a crucial role in the immune response. They inhibit the spread of viral infection in the early stages of illness and form the first line of defense in mammals against viral infections [5,6]. All IFN have an α-helical structure. According to the amino acid sequence and the type of receptor through which signal transmission is mediated, IFN are divided into three groups [3,6,7].
The most studied are type I IFN. In humans, a number of genes have been identified: 13 genes encoding different IFN-α subtypes; 1 gene encoding IFN-β; and other genes encoding IFN-ω, IFN-τ, IFN-ε, IFN-δ and IFN-κ [8]. Despite the wide variety of type I IFN, their action is mediated through the ubiquitous, heterodimeric IFNα receptor (IFNαR); such action is aimed at the induction of interferon-stimulated genes (ISG) [6]. The biological activity of type I IFN depends on their affinity for receptor subunits and the density of these subunits on the cell surface [9]. For example, all IFN-α subtypes are characterized by a non-optimal affinity for the subunit of the receptor [4], which leads to differences in biological activity between α and β IFN [10]. Therefore, the effects of type I IFN may vary in duration and intensity [9].
IFN induce activation of defense mechanisms and prepare cells for possible viral invasion. Induction of IFN production is closely associated with PRR activation. Generally, a cell first synthesizes IFN-β in response to signs of infection. Activation of the transcription factors NFκB and IRF-3 is required to this end. IFN-β stimulates the production of other IFN through its autocrine action associated with activation of IRF-7. IRF-7, in turn, binds to IFN-β and IFN-α gene promoters, enhancing the synthesis of those cytokines [3,11].
Type I IFN interact with the heterodimeric IFNα receptors. Ligand binding causes dimerization of receptor subunits and activation of tyrosine kinases JAK1 and Tyk2, which phosphorylate the transcription factors STAT1 and STAT2 [3,12]. Due to interaction with IRF-9, the ISGF3 heterotrimeric complex is formed. The complex is bound by the ISRE regulatory element that is located on the promoters of most ISG. Consequently, type I IFN enhance the transcription of hundreds of genes and contribute to the cell’s antiviral response [3,13]. It is important to note that the expression of an entire ensemble of genes is necessary to limit viral replication; expression of single genes alone is not capable of providing a sufficient antiviral response [3]. The signaling pathways that are affected by the actions of type III IFN are generally similar to those of type I IFN [14].
It has been shown that other signaling pathways can affect the induction of IFN-dependent transcription. The JAK-STAT signaling pathway is known to be associated with the phosphotidylinositol-3-dependent signaling cascade [12,15]. Moreover, the effects of type I IFN are associated with increased activity of MAPK-dependent signaling cascades [3,16]. In addition to direct antiviral effects, type I IFN have immuno-modulatory properties [3,9,17,18,19].
Cells have mechanisms for inhibiting the effects of type I IFN. These mechanisms may be focused on the attenuation of JAK-STAT-dependent signaling cascades. For example, it has been shown in vivo that injections of IFN-α cause activation of negative regulators, such as SOCS-1 and SOCS-3 [20,21]. Moreover, a prolonged effect of the USP18/UBP43 inhibitor ISG-encoded isopeptidase has been described [20]. The USP18/UBP43 protein is an ISG15-specific protease [22]. Knockout of USP18/UBP43 in mice results in hypersensitivity to type I IFN. This protein inhibits JAK-STAT signaling pathways by binding to the IFNαR2 subunit and blocking the interaction between the JAK1 kinase and the heterodimeric receptor [23]. Pre-incubation with type I or type III IFN has been shown to cause desensitization of cells to the stimulatory effects of IFN-α. The degree of desensitization depends on the expression level of USP18/UBP43. Thus, there is a negative feedback loop attenuating both the type I and type III IFN [9].
2. IFN-λ Play a Distinct Anti-Viral Role in Collaboration with Other IFN
2.1. IFN-λ Structure
Type III IFN (IFN-λ) are a group of cytokines that is related to type I IFN and elicit similar antiviral effects [1,24]. Four IFN-λ subtypes have been found in humans: IFN-λ1 (IL-29), IFN-λ2 (IL-28A), IFN-λ3 (IL-28B), and IFN-λ4. All of these proteins are encoded on the 19th chromosome, and these genes consist of five or six exons [7]. Several of these IFN (λ1, λ2, λ3) feature a high degree of amino acid conservation [25,26], which suggests the presence of a single ancestor [7,27]. IFN-λ2 and IFN-λ3, for example, are 96% identical in amino acid sequence; they differ by just seven residues. However, IFN-λ1 is only 80% identical to them in primary structure and also differs in disulfide bond configuration; IFN-λ1 does not form a third disulfide bond [4,25]. Initially, IFN-λ4 was thought to be a pseudogene. However, it is now known that humans have the IFNL4 gene, but in some populations, there is a polymorphism (ss469415590, TT/ΔG). The TT allele causes a frameshift leading to suppression of IFNL4 production, while the ΔG allele results in the functional IFNL4 gene [4,28]. Although the product of this gene is only 40.8% identical to IFN-λ3 [29], IFN-λ4 interacts with the heterodimeric receptor common to all IFN-λ. It has been revealed that N-glycosylation of IFN-λ4 is necessary for protein secretion. IFN-λ1 is also known to have a potential N-glycosylation site at asparagine residue 65 [4]. Only IFN-λ2 and IFN-λ3 have been found in mice, and murine IFN-λ1 is a pseudogene [26,30,31]. Murine IFN-λ2 and IFN-λ3 are also N-glycosylated [27]. IFN-λ are conserved in tetrapod vertebrates; and tetrapod type III IFN form a monophyletic group [32]. IFN-λ are considered to be derived from “IL-10-like” cytokine family [33]. Fundamental similarities between type III IFN systems of mammals and birds indicate that type III IFN might play a significant role in defending mucosal surfaces against viral infections in birds [34]. Two groups of IFN were discovered in fishes, but fish IFN are evolutionarily closer to type I than to type III tetrapod IFN [33].
Although type III IFN (IFN-λ) should be considered most closely related to type I IFN (according to primary structure), IFN-λ are close to IL-10 (and other “IL-10-like” cytokine family members) in their spatial structure [25,26]. The spatial structure of IFN-λ includes five α-helices (A, C, D, E, F) and an element B having a less-defined structure [4]. Helix A, helix F, and the AB loop are responsible for the interaction between lambda IFN and their receptors. Certain amino acids located in the AB loop (Lys49 and Arg51 for IFN-λ3; Arg49 and His51 for IFN-λ2) have a critical effect on affinity for the IFNλR1 subunit of the receptor. Helix D is responsible for binding to the IL-10R2 subunit (Gly95 for IFN-λ3, and Val95 for IFN-λ2, are considered to be important for the interaction). Lambda IFN differ in their receptor subunit affinities; the stabilities of their final ligand-receptor complexes also differ [29]. Hepatitis C virus (HCV) persistence is strongly associated with the expression of a functional IFNL4 gene, whereas the nonfunctional IFNL4 gene is associated with more rapid viral clearance [28]. Humans have several mechanisms to limit the expression of functional IFN-λ4 through noncoding splice variants and nonfunctional protein isoforms. Moreover, protein-coding IFN-λ4 mRNA are not loaded onto polyribosomes and lack a strong polyadenylation signal, which results in poor translation efficiency [35]. Amino-acid substitution (P70S) is also strongly associated with HCV clearance. Patients harbouring the S70 variant display lower ISG expression, better treatment response rates and better spontaneous clearance rates, compared with patients coding for the fully active P70 variant [36]. Interestingly, variant E159 (E159K substitution) of IFN-λ4, that was found in some ancient African populations, exhibit more significant antiviral activity than wild-type IFN-λ4. Thus, substitution E154K also negatively affects IFNλ4 activity by reducing its secretion and potency [37].
2.2. Expression of IFN-λ
Synthesis of type III IFN is induced by viral infection and PRR activation (TLR, RLR, Ku70), and it occurs in various tissues. For example, high IFN-λ levels are observed in the lungs and liver [25,26,38]. Many cell types are capable of producing both IFN-α and IFN-β (IFN-α/β) with IFN-λ, but there are exceptions. For example, in response to infection with influenza virus or herpes simplex virus type-2 (HSV-2), macrophages can produce only IFN-α/β, but not IFN-λ [30,39]. Infection with swine influenza virus (H3N2) lead to IFN-β, but neither IFN-α nor IFN-λ1 expression in porcine macrophages [40]. IFN-λ expression has been discovered in respiratory epithelial cells, keratinocytes, dendritic cells, hepatocytes, and primary neuronal cells [30]. Moreover, IFN-λ are the most common IFN produced by respiratory epithelium in response to dsRNA (poly(I:C), a TLR-3 agonist); agonists of other TLR do not induce production of IFN-α/β or IFN-λ in this cell type [38]. A potent IFN-λ response is observed upon infection of human respiratory epithelial cells with respiratory viruses, such as influenza or rhinovirus [30,38,39,41,42]. Additionally, swine influenza virus (H3N2) up-regulates IFN-λ1 in porcine epithelial cells as well as in precision-cut lung slices [40]. However, myeloid dendritic cells (mDC) and plasmacytoid dendritic cells (pDC) appear to be the major producers of IFN-λ [43]. It was shown that CpG DNA (a TLR-9 agonist) induces the expression of IFN-α/β and IFN-λ in pDC, while lipopolysaccharides (LPS) and poly(I:C) (TLR-4 and TLR-3 agonists, respectively) induce expression of IFN-β and IFN-λ1-3 in mDC (but not IFN-α) [29].
2.3. Molecular Mechanism of IFN-λ Induction
IFN-λ are induced by pathways and factors similar to those involved in the induction of IFN-α/β. Moreover, IFN-λ production is mediated by activation of the same PRRs as IFN-α/β [26]. For example, expression of IFN-λ is significantly mediated by activation of the RIG-I and MDA-5-dependent signaling pathways in respiratory and dendritic cells [5,44,45]. However, it was found that the production of IFN-λ1, but not type I IFN, can also be induced by the activation of certain DNA sensors (Ku70). Induction of IFN-λ1 synthesis, in this case, is mainly associated with activation of the IRF-1 and IRF-7 transcription factors [46].
It was shown that the transcription factors IRF-1, IRF-3, IRF-7, and NFκB can bind to the promoters of IFNL genes. The synergistic effect of these transcription factors allows for maximum induction efficiency [45]. However, it is worth noting that expression of IFN-λ2 and IFN-λ3, like IFN-α, is still predominantly regulated by IRF-7 and NFκB; expression of IFN-λ1, like IFN-β, requires the combined action of IRF-3, IRF-7, and NFκB [47]. In mice lacking IFN-λ1, IRF-3 is not involved in the induction of IFN-λ expression in response to metapneumovirus infection [5,44]. Additionally, the Med23 subunit of the eukaryotic multiprotein mediator, which interacts with transcription factors and RNA polymerase II, binds directly to IRF-7 and induces IFN-λ synthesis. However, Med23 is not able to enhance the IRF-7-mediated induction of IFN-β transcription. These data emphasize an additional selectivity of the IFN response [48]. A detailed review of type I and type III IFN induction mechanisms, and their differences, has been published [26].
Expression kinetics for IFN-λ depend on cell type and induction conditions. In PBMC and fibroblasts, it has been shown that peak IFN-λ3 expression occurs 24 h after infection with cytomegalovirus, while the IFN-λ1 peak is 6 h after infection [49]. When primary human hepatocytes are infected with HCV, increased IFN-λ4 mRNA levels can be detected 2–4h after infection. However, the expression subsides after 8 h, which may suggest either: absence of a positive-regulation feedback loop; or (conversely) induction of specific negative-feedback mechanisms [29]. There is limited information about IFN-λ negative regulation (reviewed in [50]). Stimulation by type III IFN leads to ISG expression that includes SOCS and IL-10 expression. Excessive SOCS-1 expression is associated with reduced STAT1 phosphorylation as well as reduced ISG expression [51]. Type III IFN activity may be inhibited in the presence of IL-10 [52]. Additionally, it should be noted that the level of IL10R2 subunit is modulated by ubiquitination leading to degradation of nonspecific subunits [53].
2.4. The IFN-λ Receptor (IFNλR)
IFN-λ actions are carried out through the heterodimeric IFNλR, consisting of the IFNλR1 and IL10R2 subunits [25,54]. The IL10R2 subunit is also part of the receptor complexes for IL-10, IL-22, and IL-26; it is expressed in cells of various tissues. Interestingly, the IFN-λ1 and IL-10-like cytokines bind with low affinity to the IL10R2 subunit itself. In turn, IFN-λ1 specifically binds to the IFNλR1 subunit in a 1:1 stoichiometric ratio [25]. An IFNλR has also been found in mice; the murine IFNλR amino acid sequence is approximately 67% similar to the human. It should be noted that both murine and human IFN-λ act non-specifically in terms of host species: murine IFN-λ can bind to human IFNλR [27]. On the other hand, murine and human IFN-λ exhibit some species specificity. For instance, murine IFN-λ3 is 51 times less active in human A549 cells than in mouse LKR10 cells. However, IFN-λ4 is more active in mouse cells [55].
Expression of the IFNλR1 subunit demonstrates restricted cellular distribution. For example, IFN-λ does not act on fibroblasts, splenocytes, macrophages, or (migrated, bone marrow-originating) endothelial cells, since IFNλR1 is not expressed in these cells, while IFN-α is able to activate all of them [27,30]. High IFNλR1 expression has been found in the lungs, intestines, liver, and upper epidermis [30]. Expression of IFNλR1 is mainly restricted to: epithelial cells [11], keratinocytes [56], differentiated dendritic cells (pDC and mDC) [57,58], and hepatocytes [59]. In monocytes and B cells, low levels of IFNλR1 expression are detected. Thus, these cells respond extremely weakly to IFN-λ [60]. As such, mucous membranes of the respiratory and gastrointestinal tracts are the primary tissue targets of IFN-λ [11]. This tissue specificity correlates with IFN-λ antiviral activity, which is seen mainly with viruses featuring high tropism for epithelial cells, like Orthomyxoviridae, Pneumoviridae, Coronaviridae, Picornaviridae, Herpesviridae, Flaviviridae, Reoviridae, Arenaviridae, Caliciviridae (Table 1. Viruses affected by IFN-lambda (Type III interferons)) [30]. In general, type III IFN control infection at mucosal barrier sites, while type I IFN are important for broad systemic infection control.

Table 1.
Viruses affected by IFN-lambda (Type III interferons).
2.5. The Effects of IFN-λ on Cells
Lambda IFN are secreted into the extracellular space and exert autocrine or paracrine effects by binding to cell surface receptors. Although IFN-α/β and IFN-λ actions are realized through different receptors, they lead to the activation of similar signaling pathways. Upon IFN-λ binding, receptor subunits dimerize leading to activation of JAK/STAT-dependent signaling pathways: activation of JAK1 and Tyk2 tyrosine kinases; phosphorylation of receptor subunits; recruitment and subsequent phosphorylation of STAT1 and STAT2 proteins, and to a lesser extent STAT3-STAT5; and formation of the ISGF3 transcription complex. Interestingly, STAT1 can also be activated by the actions of various cytokines, while STAT2 phosphorylation is caused by the specific action of type I or type III IFN [72]. The ISGF3 complex is also formed in response to the actions of type I IFN. Therefore, IFN-λ functions significantly overlap with type I IFN functions and cause the expression of similar ISG. In addition to activating JAK/STAT signaling cascades, IFN-λ also influences MAPK signaling pathways, including the Erk, Jnk, and p38 kinases [14,26,73,74].
There are some differences in the mechanisms activated by type I and type III IFN. In intact cells, IFN-λ activate STAT-dependent signaling pathways slightly more weakly than IFN-α, which is associated with higher basal type I IFN levels. Interestingly, the JAK2 kinase (necessary for phosphorylation of STAT1) is specifically activated in response to IFN-λ; this may underlie the differences in IFN-λ and IFN-α/β effects. Moreover, gene knockout of JAK2, or its inhibition by substances (such as AG490 or 1,2,3,4,5,6-hexabromocyclohexane), can specifically block IFN-λ-dependent signaling cascades without affecting type I IFN-depended signaling pathways [75].
2.6. Immuno-Modulatory Activity of IFN-λ
IFN-λ-mediated signaling also regulates the immune response. The presence of phosphorylated STAT-3, STAT-4, and STAT-5 forms indicates the existence of an additional level of complexity. Thus, there are several associations: STAT3 phosphorylation is also a signaling mechanism used by members of IL-10-like cytokines (IL-10, IL-19, IL-20); phosphorylated forms of STAT5 are often associated with IL-2, IL-3, and GM-CSF; and STAT4 is associated with a T helper type 1 (Th1)-mediated immune response. In general, an entire body of knowledge indicates the presence of additional immuno-modulatory activities of IFN-λ [52].
In early studies, it was shown that IFN-λ1 causes the secretion of IL-6, IL-8, and IL-10 in PBMC. Selective blocking of IL-10 with specific antibodies leads to a decrease in the required dose of IFN-λ1 for the secretion of IL-6. In turn, the addition of IL-10 reduced the effects of IFN-λ1. Therefore, the existence of a feedback mechanism can be assumed by which IFN-λ1 causes the secretion of IL-10, which inhibits the effects of the former. This mechanism may be associated with competition between IFN-λ1 and IL-10 for binding to the IL10R2 subunit [76]. The IL-22 receptor also contains the IL10R2 subunit. IL-22 acts synergistically with IFN-λ and causes activation of STAT1-dependent signaling pathways in the suppression of rotavirus infections [77]. Dendritic cells express IFNλR during differentiation from monocytes. The dendritic cells, maturing upon stimulation with IFN-λ, induce IL-2-dependent proliferation of a population of CD4+/CD25+/Foxp3+ regulatory T cells [74].
4. The Role of IFN-λ Specifically during Influenza Virus Infection
4.1. Influenza Virus Infection and Respiratory Airway Epithelium
Infiltration of influenza virus into a host leads to infection of type II epithelial cells lining the respiratory tract, and subsequent development of inflammatory processes. In mild cases, infection is limited to the upper respiratory tract (URT). In cases featuring severe pathologies, infection can penetrate into lung tissues [39,98,99,100]. The alveolar epithelium is responsible for gas exchange, and it is the main target of IAV and influenza-induced pneumonia [101]. Respiratory tract epithelia are known to be covered with mucus. Therefore, virions must pass through the mucus layer to reach the target cells [102]. Epithelial cell surfaces contain sialic acid receptors (SAR), which are sialyl oligosaccharides. The influenza virus hemagglutinin (HA) protein is able to bind to SAR, thereby permitting the virion to enter the epithelial cell through endocytosis. Accordingly, viral infiltration depends on SAR densities on respiratory epithelial cells and on interaction affinities with the various HA types. The interaction of HA with SAR is specific, and human influenza virus strains mainly interact with α-2,6-SAR, which are most prevalent in the URT. However, α-2,3-SAR sialyl oligosaccharides bind mainly to avian influenza virus strains, and they are most common in the lower respiratory tract. Thus, URT epithelial cells are the main targets of human influenza virus strains [103]. Through the use of traceable IAV (a modified A/Puerto Rico/8/34 (PR/08) strain with a green fluorescent protein within the viral genome NS segment), it was shown that IAV infection begins in the trachea and the main bronchi, and spreads to bronchiolar regions and, possibly, alveoli. Spread of influenza virus in the lungs depends on a wide variety of factors, including: virus origin (human, avian, etc.); HA specificity; accessibility of target cells and SAR; and even body temperature [104]. In early-stage infection with Balb/c mice, accumulation of viral NS protein has been observed in non-immune CD45− cells of the respiratory tract. Expression of IAV proteins was detected in hematopoietic cells only a day after infection [104]. Consequently, epithelial CD45−cells of the trachea and major bronchi are the primary targets of IAV in mice.
Type II alveolar cells synthesize and secrete pulmonary surfactant, chemokines, and cytokines; they are involved in the innate immune response of the lungs [39,101]. As such, infection with IAV (strain PR/08) induces secretion of IL-6, IL-8, MIP-1, RANTES, MCP-1, IL-10, and IFN-β, but neither IFN-α nor IFN-γ. IL-8 and MCP-1 are known to be the main stimulants for neutrophil and monocyte migration, respectively, in acute lung inflammation. However, it should be noted that IFN-λ1 is the main IFN induced by IAV infection in type II alveolar epithelial cells [39]. The pro-inflammatory cytokines and chemokines, together with PRR ligands, stimulate the production and activation of immune cells [103]. The host’s immune response varies greatly depending on the influenza virus strain. For instance, it was shown that infection with IAV H5N1 led to more pronounced induction of pro-inflammatory cytokines and chemokines (IP-10, IFN-β, IL-6, RANTES) in human primary epithelial cells, compared with infection with H1N1 [105]. The induction of pro-inflammatory cytokines and chemokines is correlated with viral strain pathogenicity. More pathogenic strains are able to induce higher levels of cytokines and chemokines [103].
IAV infection can activate pattern-recognition receptors (PRR) such as viral nucleic acid sensors. In one example, the “nucleic acid” is not coming only from the invading virus, but also from endogenous retroviruses (ERV) that are no longer repressed by the TRIM28 restriction element, because of an IAV-induced mechanism to suppress TRIM28 [106]. IAV causes the small ubiquitin-like modifier (SUMO) to modify TRIM28, thereby inactivating its restriction abilities and unleashing ERV RNA into the cell. Such RNA would ordinarily induce innate responses like IFN production were it not for the viral nucleic-acid binding protein NS1 that is able to soak up the excess RNA.
With influenza virus infection, a significant similarity in the functioning of type I and type III IFN has been noted [107,108]. While dendritic cells and alveolar macrophages mainly produce type I IFN, respiratory epithelial cells are the main producers of type III IFN. Induction of both types in virus-infected epithelial cells occurs in response to the same antigenic determinants and requires the activation of the same signaling pathways. Moreover, they induce almost identical sets of ISG [107,108]. IFNαR is expressed ubiquitously, while IFNλR1 subunit expression is observed mostly in epithelial cells of the respiratory and gastrointestinal tracts. During influenza virus infection, IFN-λ provides respiratory epithelial cells with an antiviral response and also limits direct activation of immune cells associated with the development of uncontrolled inflammatory processes in the lungs [6,39,108].
4.2. Knockout Mouse Models
To determine the involvement of a gene in the control of a viral infection, it is convenient to use models featuring a knocked out gene or group of genes. Thus, more than a decade ago, the involvement of IFN-λ-dependent signaling pathways in the suppression of influenza virus infection was shown. IFNLR1 gene knockout caused an increase in lung viral titers, but did not affect the survival curves of infected mice. Combined knockout of the IFNAR and IFNλR1 genes led to a significant increase in mortality, hypersensitivity (even to non-pathogenic strains lacking NS1), significant increases in viral titers, and blockage of Mx1 production. In addition, intranasal administration of recombinant IFN-λ2/3 protected IFNAR knockout animals from lethal doses of IAV (subtype H7N7) [109]. It has been also shown that mice with an IFNL2–IFNL3 double knockout are characterized by impaired control of viral replication in the lungs and exhibit a phenotype similar to that observed with IFNLR1 gene knockout [110]. The importance of IFN-λ-dependent signaling in the suppression of influenza virus infection has been shown in a number of other works [72,85,107,111] however IFN-λ was considered to be an auxiliary antiviral protective mechanism, acting in parallel with IFN-α/β [107]. Later, it was shown that IFN-λ, unlike IFN-α/β, does not cause the development of an uncontrolled pro-inflammatory response, or “cytokine storm” [108], which is one of the main causes of influenza virus infection pathology [103,112].
It has recently been demonstrated that the greatest differences between IFNλR1-defective mice and wild-type mice are observed during infection with low doses of influenza virus [110,111,113]. In particular, it was shown that IFNLR1 knockout was associated with a significant viral infiltration from the URT into the lungs and an increase in viral titer (lung, URT) upon infection with IAV SC35M (H7N7) and Udorn (H3N2). It should be noted that the development of an influenza virus infection depends on the dose of the injected viral inoculum. A high dose can lead to viral infiltration into the lungs (which is associated with a greater lethality of the model) and to the development of a “cytokine storm” hiding the protective effects of IFN-λ [113]. Correspondingly, a four-fold smaller inoculum dose permits selective infection of URT cells, thereby mimicking a natural course of the infection [113]. Mice with IFNAR1–IFNLR1 double knockout, as with IFNLR1 knockout alone, were characterized by increased viral loads at 3–5 days after infection, increased levels of pro-inflammatory immune cells in BALF (mainly neutrophils and macrophages), increased pro-inflammatory cytokine and chemokine levels (IFN-α, IL-6, CCL2, CCL3, CXCL1/keratinocyte chemoattractant (KC)), and lung tissue damage. At the same time, animals with single IFNΑR1 knockout did not differ from wild-type mice in resistance to viral challenge, and they were characterized by reduced production of pro-inflammatory, first-wave cytokines and chemokines. These data prove the importance of IFN-λ in the suppression of sub-lethal influenza virus infection [111]. In turn, with a lethal viral dose, knockout of IFNΑR1 becomes a key factor, while IFNλR1-deficient mice did not differ from the wild-type in viral load from the 3rd day after infection. With a lethal viral challenge, IFNLR1 knockout was associated with neutrophil infiltration, increased levels of IFN-α, CCL3, and CCL4, and impaired pulmonary function. These effects may be associated with either impaired control of viral replication or rather with IFN-λ immuno-modulatory activities [111].
Therefore, it can be concluded that IFN-λ and IFN-α/β are two complementary, non-redundant types of IFN for controlling influenza replication. The first type is implemented in the early stages of infection and is effective against sub-lethal doses of the virus, while IFN-α/β are activated at later stages and are effective against high doses of the virus; their action, however, is associated with the development of a systemic inflammatory process [111,113]. The mouse IFNAR–IFNLR1 double knockout model resembles the STAT1–STAT2 double knockout model, which emphasizes the unique role of type I and III IFN in inducing ISG expression and generating the innate antiviral response [72]. At the same time, IFN-λ levels in BALF and pDC of IAV (A/WSN/33 (H1N1)) infected mice exceed IFN-α/β by more than tenfold [72]. Such a significant expression of IFN-λ allows one to speculate that these IFN are the key components of the innate immune response against influenza virus infection.
4.3. Induction of IFN-λ in Influenza Virus Infection
The main mechanism of IFN-λ induction is influenza-induced PRR activation [107,114]. Notably, some research indicates that IFN-λ production is a response to the infiltration of live viruses specifically. Thermal inactivation of IAV (65 °C for 30 min.) decreased elicited IFN-λ production to the level of non-infected controls [73,107]. Such inactivation denatures HA and prevents host cell attachment [73], while inactivating viral polymerase [107]. Thus, IFN-λ expression is associated with infiltration of live viruses into host cells and replication of the viral genome [5,73,107]. It is known that IAV infection induces the expression of all IFN-λ subtypes [114]. It has recently been shown that IFN-λ1 is produced in most IAV-infected cells (strain PR08), while expression of the remaining type I and III IFN is limited to a relatively small group of cells [114]. Perhaps, a paracrine IFN-λ1 induction mechanism occurs that is based on intercellular communication [114]. At the same time, IFNAR1 gene knockout is associated with a decrease in basal expression of IFN-λ2 and IFN-λ3 [62,113], which can be explained by the absence of the IFN-β-dependent loop for positive regulation of IFN-λ expression. IFNLR1 knockout, in contrast, does not significantly affect IFN-α/β levels [62]. Therefore, it can be assumed that there is a positive—autocrine or paracrine—loop of IFN-λ induction that is mediated by IFN-β.
Influenza infiltration into host cells is related to activation of the RIG-I/MAVS signaling pathways. Infection of A549 cells or alveolar epithelial cells with IAV (strain PR08) promotes the dose-dependent expression of RIG-I and TLR-3, but not TLR-7 or NOD2. Activation of RIG-I and TLR-3 is associated with the expression of IFN-λ, as well as IFN-β and IP-10 [115]. DDX58–TLR3 double knockdown led to: a decrease in IRF-3 phosphorylation; almost complete blockage of IFN-λ1 and IFN-β production; and no significant changes in IFN-λ2 and IFN-λ3 expression [115]. Nevertheless, IFN-λ expression is known to be associated with RIG-I expression during influenza virus infection [40,73,116]. Using CIAP (calf intestine alkaline phosphatase), which is used to remove the triphosphate group from the 5′-end of viral RNA, a decrease in RIG-I-mediated signaling pathway activation was detected, leading to inhibition of IFN-λ expression [73]. Moreover, DDX58 silencing also markedly suppressed IFN-λ production in A549 cells [73]. Meanwhile, it was found that knockout of TLR7, its molecular adapter MYD88, or TICAM1, did not affect the production of IFN-λ2/3 in murine tracheal epithelial cells (MTEC) cells [107]. In addition, MAVS-deficient cells were not able to produce sufficient IFN-λ2 and IFN-λ3 in response to influenza virus infection [107]. Overall, these data emphasize the role of the RIG-I/MAVS signaling pathway in IFN-λ expression [107].
The expression of IFN-λ is mediated by the activation of several transcription factors. Knockout of IRF3 did not block the ability of MTEC to produce either IFN-λ2/3 or IFN-β [107]. It was also shown that IRF3–NFκB double knockdown inhibited poly(I:C)-mediated IFN-λ induction [117]. With IRF7 knockout, a significant decrease in type I and III IFN expression has been shown [107]. In response to IAV infection, however, IFN-λ expression was completely blocked only with a IRF3–IRF7 double knockout. MTEC cells with IFNAR1–IFNLR1 double knockout were able to produce both IFN-λ2/3 and IFN-β in amounts comparable to the wild-type. However, ISG expression (OASL-1, RSAD2, IFI3) was completely suppressed [107]. Therefore, it is reasonable to conclude that the synergistic action of several transcription factors (IRF-3, IRF-7, NFκB) is required for optimal IFN-λ induction in response to influenza virus infection [107].
Induction of IFN-λ can also be caused by the close cooperation of respiratory epithelial cells with bacterial microflora of the URT. Viral infections can induce an ideal environment for a bacterial superinfection through different mechanisms such as the destruction of the epithelial barrier, the over-expression of the receptors involved in the bacterial adhesion to the cells, and the alteration of the host immune response [118]. On the other hand, certain commensal bacterial exhibit antiviral effects. Staphylococcus epidermidis is the most common commensal bacteria of the healthy nasal mucosa [119]. Under stimulation with Staphylococcus epidermidis, normal human nasal epithelial cells are capable of TRL-2-independent IFN-λ production and expression of antiviral ISG (CXCL10, IFIT1, MX1, OAS1), which facilitate the suppression of IAV replication. However, IFN-α,-β, or -γ production is not observed with such stimulation. Silencing of IFNLR1 with short hairpin RNA attenuates the antiviral effect of commensal bacteria, which indicates the specificity of IFN-λ-dependent signaling pathways. Moreover, Staphylococcus epidermidis has been shown to prevent the spread of IAV to the lungs by stimulating IFN-λ production and suppressing viral replication in the nasal mucosa. Intranasal administration of commensal bacteria promotes the survival of mice. It seems to be important that administration of Staphylococcus epidermidis in that research was not associated with bacterial infiltration into the lungs, and IFN-λ levels in BALF were not increased. Thus, Staphylococcus epidermidis is part of the first line of defense against influenza virus infection, and its activity modulates IFN-λ production in the nasal mucosa [119].
4.4. The Antiviral Effect of IFN-λ in Influenza Virus Infection
It has been shown that type I IFN and IFN-λ can independently induce antiviral ISG [21,61,107,108,111,120]. In the absence of both mechanisms of antiviral protection, a loss of ability to induce ISG is observed, which indicates a lack of additional mechanisms for the activation of these genes [107]. The antiviral effect of IFN-λ against various influenza virus subtypes has been shown. In type II alveolar epithelial cells infected with IAV (PR/08), IFN-λ1 induced antiviral ISG (MX1, OAS, ISG56) in a dose-dependent manner. Moreover, IFN-λ1 reduced viral release and suppressed influenza-induced secretion of chemokines (MIP-1β, RANTES, MCP-1 and IL-8). IFN-λ1 also attenuates IFN-β expression [39]. One day after infection of MTEC with IAV (PR/08), expression of genes associated with activation of IRF and IFN signaling was noted. A significant increase in IFN-λ2/3 and a slight increase in type I IFN (IFN-β, IFN-α4, IFN-α5) levels was shown [107].
Antiviral IFN-λ activity was more pronounced with an avian IAV strain (A/chicken/Germany/27) than with the human PR/08 strain [121]. In A549 cells, IFN-λ1,2,3 reduced avian IAV (H7N7) titer and showed the ability to induce IFN-β expression. Interestingly, IFN-λ1,2,3 decreased the expression of RIG-I; type I IFN, on the contrary, increased. Avian H7N7 did not affect the RIG-1 level [122]. In IBV-infected A549 cells, there was a significant increase in the expression of IFN-λ and canonical antiviral ISG (MX1, OAS, IFITM1), but not IFN-α/β. Peak IFN-λ expression occurred 8 h after infection. IFN-λ, in a dose-dependent manner, inhibited IBV replication [61].
Compared with IFN-α and IFN-ω, IFN-λ more effectively reduces viral load, which may be due to differences in ISG induction kinetics [121]. It was shown that the level of IFN-λ-induced STAT1 phosphorylation was significantly reduced in IAV-infected A549 cells, and already 15 h after infection, it was indistinguishable from those of intact cells. Therefore, IAV is able to inhibit JAK/STAT-dependent signaling pathways. One of the possible inhibitors is the SOCS-1 protein [21], whose peak expression is observed approximately 10 h after infection. SOCS-1 can be induced by a cytokine-independent mechanism [73]. It has also been shown that infection of human tracheobronchial epithelial cells with IAV (subtypes H5N1 and H3N2) is associated with a significant increase in the expression of SOCS-1 and SOCS-3. This may be one of the mechanisms influenza exploits to avoid the host innate immune response [123]. Additionally, an inverse correlation between the expression of IFN and viral NS1 is seen in A549 cells, which may serve as indirect evidence of IFN response inhibition through NS1 protein [114]. Interestingly, pre-treatment of respiratory epithelial cells with IL-17A led to a decrease in poly (I:C)-mediated induction of IFN-λ, both at the mRNA and protein levels. Additionally, IL-17A is able to inhibit IAV (H3N2) induced IFN-λ production. Investigation of IL-17A’s inhibitory effects showed that it has a prolonged action and reduces IFN-λ levels, even two days after administration into the medium. Moreover, the addition of IL-17A inhibited STAT1 phosphorylation for up to 24 hrs after treatment. Thus, suppression of JAK/STAT-dependent signaling pathways may be one of the mechanisms by which IL-17A’s inhibitory effects are realized. IL-17A activity is mediated through the SOCS-1 and SOCS-3 proteins [117]. These data are interesting examples of the relationship between IFN-λ and other cytokine immuno-modulators.
4.5. Immuno-Modulatory Effects
IFN-λ is known to possess immuno-modulatory activities during infection with respiratory viruses, mainly rhinovirus and respiratory syncytial virus [29,42,93]. There are nuances specific to the influenza virus. It has been shown that IFN-λ3 polymorphism rs8099917 may be associated with influenza vaccination effectiveness. The presence of minor alleles at this polymorphism is associated with a decrease in IFN-λ3 level, which influences the Th1/Th2 balance. In response to IAV stimulation, PBMC of patients with the minor alleles were characterized by decreased Th1-cytokines and increased Th2-cytokines; HLA-DR expression in B lymphocytes was also increased. Moreover, recombinant IFN-λ3 in PBMC led to: increased IFN-γ and IL-6; inhibited expression of IL-4, IL-5, and IL-13; and reduced IAV-inducible proliferation of B cells and IgG production [29]. However, other researchers have shown that B cells are capable of expressing IFNλR1 and, conversely, IFN-λ increases TLR-7-mediated activation of B cells, causing immunoglobulin production [124]. Moreover, it was shown that murine IFN-λ2 is able to activate virus-specific CD8+ T cells and induce thymic stromal lymphopoietin (TSLP), an adaptive immune response regulator, in respiratory epithelial M cells [125]. Interestingly, the induction of TSLP in M cells is specific to IFN-λ, but not type I IFN. TSLP activates antigen presentation by CD103+ migratory dendritic cells and biases the immune response towards IgG1 and IgA production. The use of IFN-λ2 as an adjuvant is able to enhance the protective effect of a vaccine against IAV (PR08 and A/Udorn/72) [125]. To date, some contradictions regarding IFN-λ’s effect on the humoral immune response against influenza remain unresolved [29,125]. A possible cause may be differences in the functioning of the adaptive immune response in humans and mice. Additional research is required to resolve these contradictions.
The relationship between IFN-λ and the development of a specific, CD8+ T cell response has been shown with heterosubtypic IAV challenge. Neutralizing antibodies produced by B cells are known to protect against only one IAV subtype, but are not effective against infection with other subtypes. In contrast, the CD8+ T cell response is efficient, regardless of IAV subtype. Knockout of the IFNLR1 gene led to an increased susceptibility to H1N1 (strain PR/08) in mice that had previously been infected with the IAV-X31 reassortant strain (with HA and NA from A/HK/1/68 (H3N2)). It was noted: a decreased production of IFN-γ and TNF-α on day 9 after infection with the PR/08 strain, and a reduction in the number of IAV-specific CD4+ and CD8+ T cells in the lung lymph nodes of IFNLR1-defective mice, 35 days post-challenge. Defects in the CD8+ T cell response are associated with impaired cellular memory and antigen priming. It has been shown that IFN-λ-dependent signaling is required in pulmonary dendritic cells (CD103+ dendritic cells and CD8α+ dendritic cell sub-populations) for maturation, migration to lymph nodes, upregulation of costimulant molecules, and antigen presentation in acute influenza virus infection. On the 4th day after infection, when an accumulation of pulmonary dendritic cells in the lymph nodes was observed (necessary for the CD8+ T-cell response), it was noted that IFNLR1 knockout leads to upregulation of the LOC101055769, RGS9BP, TMEM246, WRD31, and IL10 genes. Therefore, in dendritic cells, IFN-λ is required to inhibit IL-10 production, the immuno-modulatory effect of which is associated with impaired CD8+ T cell priming [126]. In addition, IFN-λ enhanced IDO1 activity, and IFNL knockdown decreased IDO1 expression during influenza virus infection [127].
It has also been shown that overexpression of IFN-λ3 led to an increase in the number of natural killer cells in the spleen, liver, and lungs. IFN-λ3 promoted the proliferation and maturation of these immune cells and contributed to the suppression of influenza (PR/08) infection in mice. It was shown that the IFN-λ3′s effect on natural killer cells is mediated by activation of macrophages which, in turn, influence natural killer cells [128]. In neutrophils, high levels of the IFNλR1 and IL10R2 subunits are observed, which makes these cells, along with epithelial cells, the main targets of IFN-λ. Neutrophil activation through IFN-λ is neither associated with significant infiltration of these immune cells into the lungs nor a significant increase in the level of pro-inflammatory cytokines. Gene expression data (RNA-seq) revealed that IFN-λ and IFN-α affect neutrophils, including induced expression of an extensive set of ISG (ISG15, ISG20, OAS1, OASL1-2, IFI47, IFIT1-3, RSAD2) and PRR (DDX58, IFIH1, TLR3, TLR7). IFN-α, however, was additionally characterized by stimulation of pro-inflammatory cytokine and chemoattractant production (TNF, IL1b, IL6, CCL2, CCL4, CCL5, CXCL9-11). Therefore, it is possible that one of the functions of respiratory-epithelium-produced IFN-λ is the protection of neutrophils that infiltrate the lung during influenza virus infection [111]. Another study noted no significant neutrophil infiltration into the lungs [108]. It is also important to note that, in PBMC, IFN-λ does not significantly increase the expression of certain ISG (IRF7, RSAD2, OAS1), nor does it lead to the secretion of high amounts of certain pro-inflammatory cytokines or chemokines (IL-6, MCP-1, IP-10); this contributes to the localization of the inflammatory process [108].
4.6. Use of Recombinant IFN-λ: A Two-Faced Janus
A significant increase in IFN-λ expression has been shown in response to IAV infection in vivo [62,73]. Murine models are extensively used to investigate the specific effects of type I and III IFN. In vivo studies have shown that IFN-α/β treatment yielded contradictory results [129]. This is associated with the pro-inflammatory effects of type I IFN: recruitment of innate immune cells to the site of infection (mainly pDC and monocytes); increases in pro-inflammatory cytokines and chemokines in BALF (IL-6, IP-10, MCP-1, MIP-1α); and increases in the frequency of respiratory epithelial cell apoptosis. However, it has been shown that treatment with either IFN-α or IFN-λ is able to completely suppress influenza virus infection in the murine lung [108]. As such, the intranasal administration of recombinant IFN-λ2 has a protective effect against IAV (PR/08). It is notable that there was no increase in pro-inflammatory cytokine levels, and the action of IFN-λ2 protected pulmonary epithelium from virus-induced apoptosis. As part of an analysis of genes induced by IFN-α or IFN-λ, it was shown that a cluster of genes exists, whose expression is specifically induced by IFN-α and which is associated with immune cell recruitment, hypercytokinemia, and hyperchemokinemia [108,111]. This contributes to the development of a “cytokine storm” and inflammatory processes in lung tissue [103,112]. Therefore, IFN-α/β is a powerful immuno-modulatory mechanism [107], and the addition of exogenous IFN-α/β can lead to detrimental outcomes, whereas IFN-λ lacks at least some of the adverse effects [108,111].
It has been shown that viral load is the key factor determining the features of the innate immune response [111]. Administration of PEGylated IFN-λ2 in a sub-lethal model of influenza virus infection suppressed viral replication and decreased the number of pro-inflammatory immune cells in BALF. It also reduced a number of other factors: pro-inflammatory cytokines and chemokines (TNF, IFN-γ, CCL3, CCL4, CXCL9, IP-10); as well as type I IFN. It has also been shown that both IFN-α and IFN-λ reduced IAV (subtype H3N2) titer in the lung [113]. However, IFN-λ is able to inhibit viral replication in the URT to a much greater extent than IFN-α, and IFN-λ reduces the risk of transmission of the virus from one animal to another. Moreover, the antiviral effects of IFN-α against IAV in the URT were not apparent three days after intranasal administration. The effects of the IFN-λ, however, were still present, and MxA expression reached its peak only a day after administration [113], which correlates with IFN-λ3 expression kinetics [29]. These data cumulatively indicate the unique value of IFN-λ in antiviral protection precisely in UTR tissues [113].
In addition to inhibiting influenza infection, IFN-λ may reduce the risk of some complications. The presence of allergic rhinitis may be associated with increased susceptibility to influenza infection. One of the explanations is the disturbance of IFN-λ-dependent signaling pathways. Thus, in patients with allergic rhinitis, the IFN-λ expression in nasal mucosal cells is attenuated, and recombinant IFN-λ reduces viral load in allergic rhinitis nasal epithelial cells. Using an allergic rhinitis model, IFN-λ has been shown to reduce viral load in the nasal mucosa of IAV-infected mice [130].
The aforementioned supports the use of IFN-λ in the treatment of influenza infection. However, questions about the possible risks of such an approach have appeared in recent years. The risks are related to the anti-proliferative properties of IFN-λ. The proliferation of type II respiratory epithelial cells is observed 6–8 days after infection, which correlates with the time frame of influenza elimination and weight gain. It has been shown that in this time period IFN-λ, but not IFN-α/β, is present in BALF. IFN-λ inhibits the proliferation and differentiation of pulmonary epithelium. Moreover, single knockout of the IFNAR1 or IFNLR1 genes improved cell proliferation compared to wild type [62], while not significantly disrupting suppression of viral replication by innate immunity due to the parallel effects of IFN-α/β and IFN-λ [107,109]. IFN-β and IFN-λ have the highest antiproliferative effects, which is associated with: impaired cell cycle promotion, and apoptosis. These effects, however, were observed only in actively dividing cell cultures [62]. The antiproliferative effects of IFN-β and IFN-λ are associated with upregulation of the p53 tumor suppressor protein, with IFN-λ inducing a more significant increase in p53. The anti-proliferative effect of IFN-λ can lead to increased susceptibility to bacterial pathogens, such as Streptococcus pneumonia [62].
It is worth noting that the period of maximum susceptibility to secondary bacterial lung infections occurs on the 6–7th day after influenza infection. IFNLR1 knockout mice were characterized by: increased expression of IL-22 and Ngal; lower URT bacterial load; and less susceptibility to bacterial pneumonia [131]. Some research presents data that IFN-λ administration leads to complications in cases of mixed infection [132]. In that study, IFN-λ administration was carried out on the fifth day after infection with IAV (strain PR/08); one day after administration, mice were infected with Staphylococcus aureus or Streptococcus pneumoniae. Despite an increase in URT bacterial load, it was found that IFN-λ did not inhibit the expression of IL-17, IL-22, Ngal, or pulmonary antimicrobial peptides. Perhaps the deleterious effect of IFN-λ in mixed infections is based on a decrease in neutrophil mobility and, consequently, a decrease in the number of neutrophils recruited to lung tissue. Attenuated levels of neutrophil chemo-attractants (CXCL1/keratinocyte chemoattractant (KC), granulocyte colony-stimulating factor (G-CSF), IL-1α) in lung tissue homogenates may be a convincing indication of this assumption. It has also been shown that IFN-λ inhibits bacterial phagocytosis by pulmonary neutrophils. Interestingly, an increase in bacterial load and a decrease in the number of neutrophils in BALF was detected only with mixed infection, but not with Staphylococcus aureus infection alone. It can be assumed that IFN-λ, acting in combination with IAV-induced cytokines, can inhibit the activity of neutrophils [132]. Therefore, although a decrease in neutrophil recruitment to the lungs has a protective effect during influenza infection, it should be taken into account that neutrophils play a critical role in the suppression of secondary bacterial infections caused by Staphylococcus aureus or Streptococcus pneumoniae [133].
5. Conclusions
Lambda IFN are important mediators of the innate immune response and, along with type I IFN, form the primary line of defense against viral infections [108]. In contrast to both IFNAR subunits, expression of IFNλR1 is limited to a relatively narrow group of cells primarily mucosal epithelial cells [30]. This ensures the development of an IFN-λ-dependent antiviral response in respiratory epithelium which is not accompanied by direct activation of immune cells [108]. In contrast, IFN-α/β action is associated with the development of uncontrolled inflammatory processes in the lungs and occasionally, “cytokine storm” [112]. Therefore, IFN-λ can be considered a promising therapeutic agent against respiratory infections [26,30]. For example, some researchers tested IFN-λ as a therapeutic agent against SARS-CoV-2 [70,90]. On the other hand, IFN-λ has an ambiguous role in the suppression of influenza infection: it has a protective effect in the early stages of the illness [108,111,113], but has a negative influence on epithelial tissue recovery and increases the risk of bacterial secondary infections [62,132]. Therefore, IFN-λ can serve as a therapeutic agent only in the early stages of the influenza-induced inflammatory process, and regulation of IFN-λ expression may be considered a key mechanism for influenza therapy.
Funding
This research work was supported by the Russian state assignment for fundamental research0784-2020-0023 (Andrey V Vasin), the Academic Excellence Project 5-100 proposed by Peter the Great St. Petersburg Polytechnic University (Andrey V Vasin). The financial support was divided in the following way: research work on article sections “Lambda interferons play a distinct anti-viral role in collaboration with other interferons” and “Antiviral Effects” was supported by the Russian state assignment for fundamental research 0784-2020-0023, research work on article section “Lambda interferons during influenza infection” was supported by the Academic Excellence Project 5-100 proposed by Peter the Great St. Petersburg Polytechnic University.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| BALF | bronchoalveolar fluid |
| HA | hemagglutinin |
| HBV | hepatitis B virus |
| HCV | hepatitis C virus |
| HSV | herpes simplex virus |
| IAV | influenza A virus |
| IBV | influenza B virus |
| IFN | interferon(s) |
| IFNαR | type I (α/β) interferon receptor |
| IFNαR1 | interferon-α/β receptor subunit 1 |
| ISG | interferon stimulated gene(s) |
| mDC | myeloid dendritic cell(s) |
| MTEC | murine tracheal epithelial cell(s) |
| pDC | plasmacytoid dendritic cell(s) |
| RLR | RIG-I-like receptor(s) |
| PRR | pattern recognition receptor(s) |
| SAR | sialic acid receptor(s) |
| SARS-CoV | severe acute respiratory syndrome coronavirus |
| TLR | Toll-like receptor(s) |
| URT | upper respiratory tract |
References
- Thompson, M.R.; Kaminski, J.J.; Kurt-Jones, E.A.; Fitzgerald, K.A. Pattern Recognition Receptors and the Innate Immune Response to Viral Infection. Viruses 2011, 3, 920–940. [Google Scholar] [CrossRef] [PubMed]
- Kaisho, T.; Akira, S. Toll-like receptor function and signaling. J. Allergy Clin. Immunol. 2006, 117, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Randall, R.E.; Goodbourn, S. Interferons and viruses: An interplay between induction, signalling, antiviral responses and virus countermeasures. J. Gen. Virol. 2008, 89, 1–47. [Google Scholar] [CrossRef] [PubMed]
- Hamming, O.J.; Terczyńska-Dyla, E.; Vieyres, G.; Dijkman, R.; Jørgensen, S.E.; Akhtar, H.; Siupka, P.; Pietschmann, T.; Thiel, V.; Hartmann, R. Interferon lambda 4 signals via the IFNλ receptor to regulate antiviral activity against HCV and coronaviruses. EMBO J. 2013, 32, 3055–3065. [Google Scholar] [CrossRef] [PubMed]
- Baños-Lara, M.D.R.; Harvey, L.; Mendoza, A.; Simms, D.; Chouljenko, V.N.; Wakamatsu, N.; Kousoulas, K.G.; Guerrero-Plata, A. Impact and Regulation of Lambda Interferon Response in Human Metapneumovirus Infection. J. Virol. 2015, 89, 730–742. [Google Scholar] [CrossRef] [PubMed]
- Killip, M.J.; Fodor, E.; Randall, R.E. Influenza virus activation of the interferon system. Virus Res. 2015, 209, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Syedbasha, M.; Egli, A. Interferon Lambda: Modulating immunity in infectious diseases. Front. Immunol. 2017, 8, 119. [Google Scholar] [CrossRef]
- Capobianchi, M.R.; Uleri, E.; Caglioti, C.; Dolei, A. Type I IFN family members: Similarity, differences and interaction. Cytokine Growth Factor Rev. 2015, 26, 103–111. [Google Scholar] [CrossRef]
- François-Newton, V.; de Freitas Almeida, G.M.; Payelle-Brogard, B.; Monneron, D.; Pichard-Garcia, L.; Piehler, J.; Pellegrini, S.; Uzé, G. USP18-based negative feedback control is induced by type I and type III interferons and specifically inactivates interferon α response. PLoS ONE 2011, 6, e22200. [Google Scholar] [CrossRef]
- Jaitin, D.A.; Roisman, L.C.; Jaks, E.; Gavutis, M.; Piehler, J.; Van Der Heyden, J.; Uze, G.; Schreiber, G. Inquiring into the Differential Action of Interferons (IFNs): An IFN-2 Mutant with Enhanced Affinity to IFNAR1 Is Functionally Similar to IFN. Mol. Cell. Biol. 2006, 26, 1888–1897. [Google Scholar] [CrossRef]
- Sommereyns, C.; Paul, S.; Staeheli, P.; Michiels, T. IFN-lambda (IFN-λ) is expressed in a tissue-dependent fashion and primarily acts on epithelial cells in vivo. PLoS Pathog. 2008, 4, e1000017. [Google Scholar] [CrossRef] [PubMed]
- Platanias, L.C. Mechanisms of type-I- and type-II-interferon-mediated signalling. Nat. Rev. Immunol. 2005, 5, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.Y.; Ma, Y.; Wang, J.; Fan, Y.; Feng, S.; Lu, Q.; Yu, Q.; Sui, D.; Rothbart, M.K.; Fan, M.; et al. Short-term meditation training improves attention and self-regulation. Proc. Natl. Acad. Sci. USA 2007, 104, 17152–17156. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Hamming, O.J.; Ank, N.; Paludan, S.R.; Nielsen, A.L.; Hartmann, R. Type III Interferon (IFN) Induces a Type I IFN-Like Response in a Restricted Subset of Cells through Signaling Pathways Involving both the Jak-STAT Pathway and the Mitogen-Activated Protein Kinases. J. Virol. 2007, 81, 7749–7758. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.C.; Tavares, L.P.; Dias, A.C.F.; Kehdy, F.; Alvarado-Arnez, L.E.; Queiroz-Junior, C.M.; Galvão, I.; Lima, B.H.; Matos, A.R.; Gonçalves, A.P.F.; et al. Phosphatidyl inositol 3 kinase-gamma balances antiviral and inflammatory responses during influenza A H1N1 infection: From murine model to genetic association in patients. Front. Immunol. 2018, 9, 975. [Google Scholar] [CrossRef]
- Katsoulidis, E.; Li, Y.; Mears, H.; Platanias, L.C. The p38 mitogen-activated protein kinase pathway in interferon signal transduction. J. Int. Cytokine Res. 2005, 25, 749–756. [Google Scholar] [CrossRef]
- Jewell, N.A.; Vaghefi, N.; Mertz, S.E.; Akter, P.; Peebles, R.S.; Bakaletz, L.O.; Durbin, R.K.; Flaño, E.; Durbin, J.E. Differential Type I Interferon Induction by Respiratory Syncytial Virus and Influenza A Virus In Vivo. J. Virol. 2007, 81, 9790–9800. [Google Scholar] [CrossRef]
- Honda, K.; Ohba, Y.; Yanai, H.; Hegishi, H.; Mizutani, T.; Takaoka, A.; Taya, C.; Taniguchi, T. Spatiotemporal regulation of MyD88-IRF-7 signalling for robust type-I interferon induction. Nature 2005, 434, 1035–1040. [Google Scholar] [CrossRef]
- Stetson, D.B.; Medzhitov, R. Type I Interferons in Host Defense. Immunity 2006, 25, 373–381. [Google Scholar] [CrossRef]
- Sarasin-Filipowicz, M.; Wang, X.; Yan, M.; Duong, F.H.T.; Poli, V.; Hilton, D.J.; Zhang, D.E.; Heim, M.H. Alpha Interferon Induces Long-Lasting Refractoriness of JAK-STAT Signaling in the Mouse Liver through Induction of USP18/UBP43. Mol. Cell. Biol. 2009, 29, 4841–4851. [Google Scholar] [CrossRef]
- Delgado-Ortega, M.; Marc, D.; Dupont, J.; Trapp, S.; Berri, M.; Meurens, F. SOCS proteins in infectious diseases of mammals. Vet. Immunol. Immunopathol. 2013, 151, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Malakhov, M.P.; Malakhova, O.A.; Il Kim, K.; Ritchie, K.J.; Zhang, D.E. UBP43 (USP18) specifically removes ISG15 from conjugated proteins. J. Biol. Chem. 2002, 277, 9976–9981. [Google Scholar] [CrossRef] [PubMed]
- Malakhova, O.A.; Kim, K., II; Luo, J.K.; Zou, W.; Kumar, K.G.S.; Fuchs, S.Y.; Shuai, K.; Zhang, D.E. UBP43 is a novel regulator of interferon signaling independent of its ISG15 isopeptidase activity. EMBO J. 2006, 25, 2358–2367. [Google Scholar] [CrossRef] [PubMed]
- Dellgren, C.; Gad, H.; Hamming, O.; Melchjorsen, J.; Hartmann, R. Human interferon-λ3 is a potent member of the type III interferon family. Genes Immun. 2009, 10, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Miknis, Z.; Magracheva, E.; Li, W.; Zdanov, A.; Kotenko, S.V.; Wlodawer, A. Crystal structure of human interferon-λ1 in complex with its high-affinity receptor interferon-λR1. J. Mol. Biol. 2010, 404, 650–664. [Google Scholar] [CrossRef]
- Lazear, H.M.; Nice, T.J.; Diamond, M.S. Interferon-λ: Immune Functions at Barrier Surfaces and Beyond. Immunity 2015, 43, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Lasfar, A.; Lewis-Antes, A.; Smirnov, S.V.; Anantha, S.; Abushahba, W.; Tian, B.; Reuhl, K.; Dickensheets, H.; Sheikh, F.; Donnelly, R.P.; et al. Characterization of the mouse IFN-λ ligand-receptor system: IFN-λs exhibit antitumor activity against B16 melanoma. Cancer Res. 2006, 66, 4468–4477. [Google Scholar] [CrossRef]
- Prokunina-Olsson, L.; Muchmore, B.; Tang, W.; Pfeiffer, R.M.; Park, H.; Dickensheets, H.; Hergott, D.; Porter-Gill, P.; Mumy, A.; Kohaar, I.; et al. A variant upstream of IFNL3 (IL28B) creating a new interferon gene IFNL4 is associated with impaired clearance of hepatitis C virus. Nat. Genet. 2013, 45, 164–171. [Google Scholar] [CrossRef]
- Egli, A.; Santer, D.M.; O’Shea, D.; Tyrrell, D.L.; Houghton, M. The impact of the interferon-lambda family on the innate and adaptive immune response to viral infections. Emerg. Microbes Infect. 2014, 3, e51. [Google Scholar] [CrossRef]
- Hermant, P.; Michiels, T. Interferon-λ in the context of viral infections: Production, response and therapeutic implications. J. Innate Immun. 2014, 6, 563–574. [Google Scholar] [CrossRef]
- Pott, J.; Mahlakõiv, T.; Mordstein, M.; Duerr, C.U.; Michiels, T.; Stockinger, S.; Staeheli, P.; Hornef, M.W. IFN-λ determines the intestinal epithelial antiviral host defense. Proc. Natl. Acad. Sci. USA 2011, 108, 7944–7949. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.; Nie, P.; Secombes, C.J.; Zou, J. Intron-Containing Type I and Type III IFN Coexist in Amphibians: Refuting the Concept That a Retroposition Event Gave Rise to Type I IFNs. J. Immunol. 2010, 184, 5038–5046. [Google Scholar] [CrossRef] [PubMed]
- Hamming, O.J.; Lutfalla, G.; Levraud, J.P.; Hartmann, R. Crystal Structure of Zebrafish Interferons I and II Reveals Conservation of Type I Interferon Structure in Vertebrates. J. Virol. 2011, 85, 8181–8187. [Google Scholar] [CrossRef] [PubMed]
- Reuter, A.; Soubies, S.; Hartle, S.; Schusser, B.; Kaspers, B.; Staeheli, P.; Rubbenstroth, D.; Garcia-Sastre, A. Antiviral Activity of Lambda Interferon in Chickens. J. Virol. 2014, 88, 2835–2843. [Google Scholar] [CrossRef]
- Hong, M.A.; Schwerk, J.; Lim, C.; Kell, A.; Jarret, A.; Pangallo, J.; Loo, Y.; Liu, S.; Hagedorn, C.H.; Gale, M.; et al. Interferon lambda 4 expression is suppressed by the host during viral infection. J. Exp. Med. 2016, 213, 2539–2552. [Google Scholar] [CrossRef]
- Terczyńska-Dyla, E.; Bibert, S.; Duong, F.H.T.; Krol, I.; Jørgensen, S.; Collinet, E.; Kutalik, Z.; Aubert, V.; Cerny, A.; Kaiser, L.; et al. Reduced IFNλ4 activity is associated with improved HCV clearance and reduced expression of interferon-stimulated genes. Nat. Commun. 2014, 5, 5699. [Google Scholar] [CrossRef]
- Bamford, C.G.G.; Aranday-Cortes, E.; Filipe, I.C.; Sukumar, S.; Mair, D.; da Silva Filipe, A.; Mendoza, J.L.; Garcia, K.C.; Fan, S.; Tishkoff, S.A.; et al. A polymorphic residue that attenuates the antiviral potential of interferon lambda 4 in hominid lineages. PLoS Pathog. 2018, 14, e1007307. [Google Scholar] [CrossRef]
- Ioannidis, I.; Ye, F.; McNally, B.; Willette, M.; Flano, E. Toll-Like Receptor Expression and Induction of Type I and Type III Interferons in Primary Airway Epithelial Cells. J. Virol. 2013, 87, 3261–3270. [Google Scholar] [CrossRef]
- Wang, J.; Oberley-Deegan, R.; Wang, S.; Nikrad, M.; Funk, C.J.; Hartshorn, K.L.; Mason, R.J. Differentiated Human Alveolar Type II Cells Secrete Antiviral IL-29 (IFN-λ1) in Response to Influenza A Infection. J. Immunol. 2009, 182, 1296–1304. [Google Scholar] [CrossRef]
- Delgado-Ortega, M.; Melo, S.; Punyadarsaniya, D.; Ramé, C.; Olivier, M.; Soubieux, D.; Marc, D.; Simon, G.; Herrler, G.; Berri, M.; et al. Innate immune response to a H3N2 subtype swine influenza virus in newborn porcine trachea cells, alveolar macrophages, and precision-cut lung slices. Vet. Res. 2014, 45. [Google Scholar] [CrossRef]
- Contoli, M.; Message, S.D.; Laza-Stanca, V.; Edwards, M.R.; Wark, P.A.B.; Bartlett, N.W.; Kebadze, T.; Mallia, P.; Stanciu, L.A.; Parker, H.L.; et al. Role of deficient type III interferon-λ production in asthma exacerbations. Nat. Med. 2006, 12, 1023–1026. [Google Scholar] [CrossRef] [PubMed]
- Koltsida, O.; Hausding, M.; Stavropoulos, A.; Koch, S.; Tzelepis, G.; Übel, C.; Kotenko, S.V.; Sideras, P.; Lehr, H.A.; Tepe, M.; et al. IL-28A (IFN-λ2) modulates lung DC function to promote Th1 immune skewing and suppress allergic airway disease. EMBO Mol. Med. 2011, 3, 348–361. [Google Scholar] [CrossRef] [PubMed]
- Hillyer, P.; Mane, V.P.; Schramm, L.M.; Puig, M.; Verthelyi, D.; Chen, A.; Zhao, Z.; Navarro, M.B.; Kirschman, K.D.; Bykadi, S.; et al. Expression profiles of human interferon-alpha and interferon-lambda subtypes are ligand- and cell-dependent. Immunol. Cell Biol. 2012, 90, 774–783. [Google Scholar] [CrossRef] [PubMed]
- Banos-Lara, M.D.R.; Ghosh, A.; Guerrero-Plata, A. Critical Role of MDA5 in the Interferon Response Induced by Human Metapneumovirus Infection in Dendritic Cells and In Vivo. J. Virol. 2013, 87, 1242–1251. [Google Scholar] [CrossRef] [PubMed]
- Onoguchi, K.; Yoneyama, M.; Takemura, A.; Akira, S.; Taniguchi, T.; Namiki, H.; Fujita, T. Viral infections activate types I and III interferon genes through a common mechanism. J. Biol. Chem. 2007, 282, 7576–7581. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Brann, T.W.; Zhou, M.; Yang, J.; Oguariri, R.M.; Lidie, K.B.; Imamichi, H.; Huang, D.W.; Lempicki, R.A.; Baseler, M.W.; et al. Cutting Edge: Ku70 Is a Novel Cytosolic DNA Sensor That Induces Type III Rather Than Type I IFN. J. Immunol. 2011, 186, 4541–4545. [Google Scholar] [CrossRef] [PubMed]
- Österlund, P.I.; Pietilä, T.E.; Veckman, V.; Kotenko, S.V.; Julkunen, I. IFN Regulatory Factor Family Members Differentially Regulate the Expression of Type III IFN (IFN-λ) Genes. J. Immunol. 2007, 179, 3434–3442. [Google Scholar] [CrossRef]
- Griffiths, S.J.; Koegl, M.; Boutell, C.; Zenner, H.L.; Crump, C.M.; Pica, F.; Gonzalez, O.; Friedel, C.C.; Barry, G.; Martin, K.; et al. A Systematic Analysis of Host Factors Reveals a Med23-Interferon-λ Regulatory Axis against Herpes Simplex Virus Type 1 Replication. PLoS Pathog. 2013, 9, e1003514. [Google Scholar] [CrossRef]
- Egli, A.; Levin, A.; Santer, D.M.; Joyce, M.; O’Shea, D.; Thomas, B.S.; Lisboa, L.F.; Barakat, K.; Bhat, R.; Fischer, K.P.; et al. Immunomodulatory function of interleukin 28B during primary infection with cytomegalovirus. J. Infect. Dis. 2014, 210, 717–727. [Google Scholar] [CrossRef]
- Stanifer, M.L.; Pervolaraki, K.; Boulant, S. Differential regulation of type I and type III interferon signaling. Int. J. Mol. Sci. 2019, 20, 1445. [Google Scholar] [CrossRef]
- Liu, B.; Chen, S.; Guan, Y.; Chen, L. Type III interferon induces distinct SOCS1 expression pattern that contributes to delayed but prolonged activation of Jak/STAT signaling pathway: Implications for treatment non-response in HCV patients. PLoS ONE 2015, 10, e0133800. [Google Scholar] [CrossRef] [PubMed]
- Jordan, W.; Eskdale, J.; Srinivas, S.; Pekarek, V.; Kelner, D.; Rodia, M.; Gallagher, G. Human interferon lambda-1 (IFN-λ 1/IL-29) modulates the Th1/Th2 response. Genes Immun. 2007, 8, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Wolk, K.; Witte, E.; Wallace, E.; Döcke, W.D.; Kunz, S.; Asadullah, K.; Volk, H.D.; Sterry, W.; Sabat, R. IL-22 regulates the expression of genes responsible for antimicrobial defense, cellular differentiation, and mobility in keratinocytes: A potential role in psoriasis. Eur. J. Immunol. 2006, 36, 1309–1323. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, J.; Schneider, W.M.; Hoffmann, H.; Vercauteren, K.; Jude, K.M.; Xiong, A.; Moraga, I.; Horton, T.M.; Glenn, J.S.; de Jong, Y.P.; et al. The IFN-λ-IFN-λR1-IL-10Rβ complex reveals structural features underlying type III IFN functional plasticity. Immunity 2017, 46, 379–392. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, S.; Wavreil, F.; Schepens, B.; Gad, H.H.; Hartmann, R.; Rocha-Pereira, J.; Neyts, J.; Saelens, X.; Michiels, T. Species specificity of type III interferon activity and development of a sensitive luciferase-based bioassay for quantitation of mouse interferon-λ. J. Int. Cytokine Res. 2018, 38, 469–479. [Google Scholar] [CrossRef]
- Zahn, S.; Rehkämper, C.; Kümmerer, B.M.; Ferring-Schmidt, S.; Bieber, T.; Tüting, T.; Wenzel, J. Evidence for a pathophysiological role of keratinocyte-derived type III interferon (IFNλ) in cutaneous lupus erythematosus. J. Investig. Dermatol. 2011, 131, 133–140. [Google Scholar] [CrossRef]
- Yin, Z.; Dai, J.; Deng, J.; Sheikh, F.; Natalia, M.; Shih, T.; Lewis-Antes, A.; Amrute, S.B.; Garrigues, U.; Doyle, S.; et al. Type III IFNs Are Produced by and Stimulate Human Plasmacytoid Dendritic Cells. J. Immunol. 2012, 189, 2735–2745. [Google Scholar] [CrossRef]
- Zhang, S.; Kodys, K.; Li, K.; Szabo, G. Human type 2 myeloid dendritic cells produce interferon-λ and amplify interferon-α in response to hepatitis C virus infection. Gastroenterology 2013, 144, 414–425. [Google Scholar] [CrossRef]
- Dickensheets, H.; Sheikh, F.; Park, O.; Gao, B.; Donnelly, R.P. Interferon-lambda (IFN-) induces signal transduction and gene expression in human hepatocytes, but not in lymphocytes or monocytes. J. Leukoc. Biol. 2013, 93, 377–385. [Google Scholar] [CrossRef]
- Mennechet, F.J.D.; Uzé, G. Interferon-λ-treated dendritic cells specifically induce proliferation of FOXP3-expressing suppressor T cells. Blood 2006, 107, 4417–4423. [Google Scholar] [CrossRef]
- Sun, Y.; Jiang, J.; Tien, P.; Liu, W.; Li, J. IFN-λ: A new spotlight in innate immunity against influenza virus infection. Protein Cell 2018, 9, 832–837. [Google Scholar] [CrossRef] [PubMed]
- Major, J.; Crotta, S.; Llorian, M.; McCabe, T.M.; Gad, H.H.; Priestnall, S.L.; Hartmann, R.; Wack, A. Type I and III interferons disrupt lung epithelial repair during recovery from viral infection. Science 2020, 369, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, M.; Yanagi, Y.; Ohno, S. Both type I and type III interferons are required to restrict measles virus growth in lung epithelial cells. Arch. Virol. 2019, 164, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Lukacikova, L.; Oveckova, I.; Betakova, T.; Laposova, K.; Polcicova, K.; Pastorekova, S.; Pastorek, J.; Tomaskova, J. Antiviral Effect of Interferon Lambda Against Lymphocytic Choriomeningitis Virus. J. Int. Cytokine Res. 2015, 35, 540–553. [Google Scholar] [CrossRef]
- Ge, D.; Fellay, J.; Thompson, A.J.; Simon, J.S.; Shianna, K.V.; Urban, T.J.; Heinzen, E.L.; Qiu, P.; Bertelsen, A.H.; Muir, A.J.; et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature 2009, 461, 399–401. [Google Scholar] [CrossRef]
- Hsu, Y.L.; Wang, M.Y.; Ho, L.J.; Lai, J.H. Dengue virus infection induces interferon-lambda1 to facilitate cell migration. Sci. Rep. 2016, 6, 24530. [Google Scholar] [CrossRef]
- Palma-Ocampo, H.K.; Flores-Alonso, J.C.; Vallejo-Ruiz, V.; Reyes-Leyva, J.; Flores-Mendoza, L.; Herrera-Camacho, I.; Rosas-Murrieta, N.H.; Santos-López, G. Interferon lambda inhibits dengue virus replication in epithelial cells. Virol. J. 2015, 12, 150. [Google Scholar] [CrossRef]
- Nice, T.J.; Baldridge, M.T.; McCune, B.T.; Norman, J.M.; Lazear, H.M.; Artyomov, M.; Diamond, M.S.; Virgin, H.W. Interferon-λ cures persistent murine norovirus infection in the absence of adaptive immunity. Science 2015, 347, 269–273. [Google Scholar] [CrossRef]
- Lind, K.; Svedin, E.; Utorova, R.; Stone, V.M.; Flodström-Tullberg, M. Type III interferons are expressed by Coxsackievirus-infected human primary hepatocytes and regulate hepatocyte permissiveness to infection. Clin. Exp. Immunol. 2014, 177, 687–695. [Google Scholar] [CrossRef]
- Dinnon, K.H.; Leist, S.R.; Schäfer, A.; Edwards, C.E.; Martinez, D.R.; Montgomery, S.A.; West, A.; Yount, B.L.; Hou, Y.J.; Adams, L.E.; et al. A mouse-adapted SARS-CoV-2 model for the evaluation of COVID-19 medical countermeasures. bioRxiv Prepr. Serv. Biol. 2020. [Google Scholar] [CrossRef]
- Doyle, S.E.; Schreckhise, H.; Khuu-Duong, K.; Henderson, K.; Rosler, R.; Storey, H.; Yao, L.; Liu, H.; Barahmand-Pour, F.; Sivakumar, P.; et al. Interleukin-29 uses a type 1 interferon-like program to promote antiviral responses in human hepatocytes. Hepatology 2006, 44, 896–906. [Google Scholar] [CrossRef] [PubMed]
- Jewell, N.A.; Cline, T.; Mertz, S.E.; Smirnov, S.V.; Flaño, E.; Schindler, C.; Grieves, J.L.; Durbin, R.K.; Kotenko, S.V.; Durbin, J.E. Lambda Interferon Is the Predominant Interferon Induced by Influenza A Virus Infection In Vivo. J. Virol. 2010, 84, 11515–11522. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Wang, S.; Chen, Q.; Chen, Y.; Chi, X.; Zhang, L.; Huang, S.; Gao, G.F.; Chen, J.L. Suppression of Interferon Lambda Signaling by SOCS-1 Results in Their Excessive Production during Influenza Virus Infection. PLoS Pathog. 2014, 10, e1003845. [Google Scholar] [CrossRef] [PubMed]
- Koch, S.; Finotto, S. Role of Interferon-λ in allergic asthma. J. Innate Immun. 2015, 7, 224–230. [Google Scholar] [CrossRef]
- Odendall, C.; Dixit, E.; Stavru, F.; Bierne, H.; Franz, K.M.; Durbin, A.F.; Boulant, S.; Gehrke, L.; Cossart, P.; Kagan, J.C. Diverse intracellular pathogens activate type III interferon expression from peroxisomes. Nat. Immunol. 2014, 15, 717. [Google Scholar] [CrossRef]
- Jordan, W.; Eskdale, J.; Boniotto, M.; Rodia, M.; Kellner, D.; Gallagher, G. Modulation of the human cytokine response by interferon lambda-1 (IFN-λ 1/IL-29). Genes Immun. 2007, 8, 13–20. [Google Scholar] [CrossRef]
- Hernández, P.P.; Mahlakõiv, T.; Yang, I.; Schwierzeck, V.; Nguyen, N.; Guendel, F.; Gronke, K.; Ryffel, B.; Hölscher, C.; Dumoutier, L.; et al. Interferon-γ and interleukin 22 act synergistically for the induction of interferon-stimulated genes and control of rotavirus infection. Nat. Immunol. 2015, 16, 698–707. [Google Scholar] [CrossRef]
- Bierne, H.; Travier, L.; Mahlakõiv, T.; Tailleux, L.; Subtil, A.; Lebreton, A.; Paliwal, A.; Gicquel, B.; Staeheli, P.; Lecuit, M.; et al. Activation of type III interferon genes by pathogenic bacteria in infected epithelial cells and mouse placenta. PLoS ONE 2012, 7, e39080. [Google Scholar] [CrossRef]
- Cohen, T.S.; Prince, A.S. Activation of inflammasome signaling mediates pathology of acute P. aeruginosa pneumonia. J. Clin. Investig. 2013, 123, 1630–1637. [Google Scholar] [CrossRef]
- Zhou, L.; Li, J.L.; Zhou, Y.; Liu, J.B.; Zhuang, K.; Gao, J.F.; Liu, S.; Sang, M.; Wu, J.G.; Ho, W.Z. Induction of interferon-λ contributes to TLR3 and RIG-I activation-mediated inhibition of herpes simplex virus type 2 replication in human cervical epithelial cells. Mol. Hum. Reprod. 2015, 21, 917–929. [Google Scholar] [CrossRef]
- Diegelmann, J.; Beigel, F.; Zitzmann, K.; Kaul, A.; Göke, B.; Auernhammer, C.J.; Bartenschlager, R.; Diepolder, H.M.; Brand, S. Comparative analysis of the lambda-interferons IL-28A and IL-29 regarding their transcriptome and their antiviral properties against hepatitis C virus. PLoS ONE. 2010, 5, e15200. [Google Scholar] [CrossRef] [PubMed]
- Marcello, T.; Grakoui, A.; Barba-Spaeth, G.; Machlin, E.S.; Kotenko, S.V.; Macdonald, M.R.; Rice, C.M. Interferons α and λ Inhibit Hepatitis C Virus Replication with Distinct Signal Transduction and Gene Regulation Kinetics. Gastroenterology 2006, 131, 1887–1898. [Google Scholar] [CrossRef] [PubMed]
- Langhans, B.; Kupfer, B.; Braunschweiger, I.; Arndt, S.; Schulte, W.; Nischalke, H.D.; Nattermann, J.; Oldenburg, J.; Sauerbruch, T.; Spengler, U. Interferon-lambda serum levels in hepatitis C. J. Hepatol. 2011, 54, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Pan, Y.; Wang, M.; Wang, D.; Li, W.; Jiang, T.; Zhang, P.; Chi, X.; Jiang, Y.; Gao, Y.; et al. IL28B genetic variation is associated with spontaneous clearance of hepatitis C virus, treatment response, serum IL-28B levels in Chinese population. PLoS ONE 2012, 7, e37054. [Google Scholar] [CrossRef]
- Mordstein, M.; Neugebauer, E.; Ditt, V.; Jessen, B.; Rieger, T.; Falcone, V.; Sorgeloos, F.; Ehl, S.; Mayer, D.; Kochs, G.; et al. Lambda Interferon Renders Epithelial Cells of the Respiratory and Gastrointestinal Tracts Resistant to Viral Infections. J. Virol. 2010, 84, 5670–5677. [Google Scholar] [CrossRef]
- Chan, R.W.Y.; Chan, M.C.W.; Agnihothram, S.; Chan, L.L.Y.; Kuok, D.I.T.; Fong, J.H.M.; Guan, Y.; Poon, L.L.M.; Baric, R.S.; Nicholls, J.M.; et al. Tropism of and Innate Immune Responses to the Novel Human Betacoronavirus Lineage C Virus in Human Ex Vivo Respiratory Organ Cultures. J. Virol. 2013, 87, 6604–6614. [Google Scholar] [CrossRef]
- Zhou, J.; Chu, H.; Li, C.; Wong, B.H.Y.; Cheng, Z.S.; Poon, V.K.M.; Sun, T.; Lau, C.C.Y.; Wong, K.K.Y.; Chan, J.Y.W.; et al. Active replication of middle east respiratory syndrome coronavirus and aberrant induction of inflammatory cytokines and chemokines in human macrophages: Implications for pathogenesis. J. Infect. Dis. 2014, 209, 1331–1342. [Google Scholar] [CrossRef]
- Qian, Z.; Travanty, E.A.; Oko, L.; Edeen, K.; Berglund, A.; Wang, J.; Ito, Y.; Holmes, K.V.; Mason, R.J. Innate Immune Response of Human Alveolar Type II Cells Infected with Severe Acute Respiratory Syndrome-Coronavirus. Am. J. Respir. Cell Mol. Biol. 2013, 48, 742–748. [Google Scholar] [CrossRef]
- Chu, H.; Chan, J.F.W.; Wang, Y.; Yuen, T.T.T.; Chai, Y.; Hou, Y.; Shuai, H.; Yang, D.; Hu, B.; Huang, X.; et al. Comparative replication and immune activation profiles of SARS-CoV-2 and SARS-CoV in human lungs: An ex vivo study with implications for the pathogenesis of COVID-19. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- O’Brien, T.R.; Thomas, D.L.; Jackson, S.S.; Prokunina-Olsson, L.; Donnelly, R.P.; Hartmann, R. Weak Induction of Interferon Expression by SARS-CoV-2 Supports Clinical Trials of Interferon Lambda to Treat Early COVID-19. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Felgenhauer, U.; Schoen, A.; Gad, H.H.; Hartmann, R.; Schaubmar, A.R.; Failing, K.; Drosten, C.; Weber, F. Inhibition of SARS-CoV-2 by type I and type III interferons. J. Biol. Chem. 2020. [Google Scholar] [CrossRef] [PubMed]
- Stanifer, M.L.; Kee, C.; Cortese, M.; Zumaran, C.M.; Triana, S.; Mukenhirn, M.; Kraeusslich, H.G.; Alexandrov, T.; Bartenschlager, R.; Boulant, S. Critical Role of Type III Interferon in Controlling SARS-CoV-2 Infection in Human Intestinal Epithelial Cells. Cell Rep. 2020, 32, 107863. [Google Scholar] [CrossRef] [PubMed]
- Villenave, R.; Broadbent, L.; Douglas, I.; Lyons, J.D.; Coyle, P.V.; Teng, M.N.; Tripp, R.A.; Heaney, L.G.; Shields, M.D.; Power, U.F. Induction and Antagonism of Antiviral Responses in Respiratory Syncytial Virus-Infected Pediatric Airway Epithelium. J. Virol. 2015, 89, 12309–12318. [Google Scholar] [CrossRef] [PubMed]
- Essaidi-Laziosi, M.; Geiser, J.; Huang, S.; Constant, S.; Kaiser, L.; Tapparel, C. Interferon-Dependent and Respiratory Virus-Specific Interference in Dual Infections of Airway Epithelia. Sci. Rep. 2020, 10, 10246. [Google Scholar] [CrossRef]
- Srinivas, S.; Dai, J.; Eskdale, J.; Gallagher, G.E.; Megjugorac, N.J.; Gallagher, G. Interferon-λ1 (interleukin-29) preferentially down-regulates interleukin-13 over other T helper type 2 cytokine responses in vitro. Immunology 2008, 125, 492–502. [Google Scholar] [CrossRef]
- Li, Y.; Gao, Q.; Yuan, X.; Zhou, M.; Peng, X.; Liu, X.; Zheng, X.; Xu, D.; Li, M. Adenovirus expressing IFN-λ1 (IL-29) attenuates allergic airway inflammation and airway hyperreactivity in experimental asthma. Int. Immunopharmacol. 2014, 21, 156–162. [Google Scholar] [CrossRef]
- Yu, R.Y.; Dai, J.; Megjugorac, N.J.; Gallagher, G.E.; Yu, R.Y.L.; Gallagher, G. IFN-1 (IL-29) inhibits GATA3 expression and suppresses Th2 responses in human naive and memory T cells. Blood J. Am. Soc. Hematol. 2009. [Google Scholar] [CrossRef]
- Wareing, M.D.; Shea, A.L.; Inglis, C.A.; Dias, P.B.; Sarawar, S.R. CXCR2 is required for neutrophil recruitment to the lung during influenza virus infection, but is not essential for viral clearance. Viral Immunol. 2007, 20, 369–378. [Google Scholar] [CrossRef]
- Suzuki, Y.; Ito, T.; Suzuki, T.; Holland, R.E.; Chambers, T.M.; Kiso, M.; Ishida, H.; Kawaoka, Y. Sialic Acid Species as a Determinant of the Host Range of Influenza A Viruses. J. Virol. 2000, 74, 11825–11831. [Google Scholar] [CrossRef]
- Tate, M.D.; Pickett, D.L.; van Rooijen, N.; Brooks, A.G.; Reading, P.C. Critical Role of Airway Macrophages in Modulating Disease Severity during Influenza Virus Infection of Mice. J. Virol. 2010, 84, 7569–7580. [Google Scholar] [CrossRef]
- Mason, R.J. Biology of alveolar type II cells. Respirology 2006, 11, S12–S15. [Google Scholar] [CrossRef]
- Iwasaki, A.; Pillai, P.S. Innate immunity to influenza virus infection. Nat. Rev. Immunol. 2014, 14, 315–328. [Google Scholar] [CrossRef]
- Cole, S.L.; Ho, L.P. Contribution of innate immune cells to pathogenesis of severe influenza virus infection. Clin. Sci. 2017, 131, 269–283. [Google Scholar] [CrossRef]
- Manicassamy, B.; Manicassamy, S.; Belicha-Villanueva, A.; Pisanelli, G.; Pulendran, B.; García-Sastre, A. Analysis of in vivo dynamics of influenza virus infection in mice using a GFP reporter virus. Proc. Natl. Acad. Sci. USA 2010, 107, 11531–11536. [Google Scholar] [CrossRef]
- Chan, M.C.W.; Cheung, C.Y.; Chui, W.H.; Tsao, G.S.W.; Nicholls, J.M.; Chan, Y.O.; Chan, R.W.Y.; Long, H.T.; Poon, L.L.M.; Guan, Y.; et al. Proinflammatory cytokine responses induced by influenza A (H5N1) viruses in primary human alveolar and bronchial epithelial cells. Respir. Res. 2005, 6, 135. [Google Scholar] [CrossRef]
- Schmidt, N.; Domingues, P.; Golebiowski, F.; Patzina, C.; Tatham, M.H.; Hay, R.T.; Hale, B.G. An influenza virus-triggered SUMO switch orchestrates co-opted endogenous retroviruses to stimulate host antiviral immunity. Proc. Natl. Acad. Sci. USA 2019, 116, 17399–17408. [Google Scholar] [CrossRef]
- Crotta, S.; Davidson, S.; Mahlakoiv, T.; Desmet, C.J.; Buckwalter, M.R.; Albert, M.L.; Staeheli, P.; Wack, A. Type I and Type III Interferons Drive Redundant Amplification Loops to Induce a Transcriptional Signature in Influenza-Infected Airway Epithelia. PLoS Pathog. 2013, 9, e1003773. [Google Scholar] [CrossRef]
- Davidson, S.; McCabe, T.M.; Crotta, S.; Gad, H.H.; Hessel, E.M.; Beinke, S.; Hartmann, R.; Wack, A. IFN λ is a potent anti-influenza therapeutic without the inflammatory side effects of IFN α treatment. EMBO Mol. Med. 2016, 8, 1099–1112. [Google Scholar] [CrossRef]
- Mordstein, M.; Kochs, G.; Dumoutier, L.; Renauld, J.C.; Paludan, S.R.; Klucher, K.; Staeheli, P. Interferon-λ contributes to innate immunity of mice against influenza A virus but not against hepatotropic viruses. PLoS Pathog. 2008, 4, e1000151. [Google Scholar] [CrossRef]
- Peterson, S.T.; Kennedy, E.A.; Brigleb, P.H.; Taylor, G.M.; Urbanek, K.; Bricker, T.L.; Lee, S.; Shin, H.; Dermody, T.S.; Boon, A.C.M.; et al. Disruption of Type III Interferon (IFN) Genes Ifnl2 and Ifnl3 Recapitulates Loss of the Type III IFN Receptor in the Mucosal Antiviral Response. J. Virol. 2019, 93. [Google Scholar] [CrossRef]
- Galani, I.E.; Triantafyllia, V.; Eleminiadou, E.E.; Koltsida, O.; Stavropoulos, A.; Manioudaki, M.; Thanos, D.; Doyle, S.E.; Kotenko, S.V.; Thanopoulou, K.; et al. Interferon-λ Mediates Non-redundant Front-Line Antiviral Protection against Influenza Virus Infection without Compromising Host Fitness. Immunity 2017, 46, 875–890. [Google Scholar] [CrossRef]
- Guo, X.; Zhi, J.; Thomas, P.G. New fronts emerge in the influenza cytokine storm. Semin. Immunopathol. 2017, 39, 541–550. [Google Scholar] [CrossRef]
- Klinkhammer, J.; Schnepf, D.; Ye, L.; Schwaderlapp, M.; Gad, H.H.; Hartmann, R.; Garcin, D.; Mahlakõiv, T.; Staeheli, P. IFN-λ prevents influenza virus spread from the upper airways to the lungs and limits virus transmission. Elife 2018, 7, e33354. [Google Scholar] [CrossRef]
- Ramos, I.; Smith, G.; Ruf-Zamojski, F.; Martínez-Romero, C.; Fribourg, M.; Carbajal, E.A.; Hartmann, B.M.; Nair, V.D.; Marjanovic, N.; Monteagudo, P.L.; et al. Innate Immune Response to Influenza Virus at Single-Cell Resolution in Human Epithelial Cells Revealed Paracrine Induction of Interferon Lambda 1. J. Virol. 2019, 93, e00559-19. [Google Scholar] [CrossRef]
- Wu, W.; Zhang, W.; Duggan, E.S.; Booth, J.L.; Zou, M.H.; Metcalf, J.P. RIG-I and TLR3 are both required for maximum interferon induction by influenza virus in human lung alveolar epithelial cells. Virology 2015, 482, 181–188. [Google Scholar] [CrossRef]
- Švancarová, P.; Svetlíková, D.; Betáková, T. Induction of interferon lambda in influenza a virus infected cells treated with shRNAs against M1 transcript. Acta Virol. 2015, 59, 148. [Google Scholar] [CrossRef][Green Version]
- Niwa, M.; Fujisawa, T.; Mori, K.; Yamanaka, K.; Yasui, H.; Suzuki, Y.; Karayama, M.; Hozumi, H.; Furuhashi, K.; Enomoto, N.; et al. IL-17A Attenuates IFN-λ Expression by Inducing Suppressor of Cytokine Signaling Expression in Airway Epithelium. J. Immunol. 2018, 201, 2392–2402. [Google Scholar] [CrossRef]
- Saade, G.; Deblanc, C.; Bougon, J.; Marois-Créhan, C.; Fablet, C.; Auray, G.; Belloc, C.; Leblanc-Maridor, M.; Gagnon, C.A.; Zhu, J.; et al. Coinfections and their molecular consequences in the porcine respiratory tract. Vet. Res. 2020, 51, 80. [Google Scholar] [CrossRef]
- Kim, H.J.; Jo, A.; Jeon, Y.J.; An, S.; Lee, K.M.; Yoon, S.S.; Choi, J.Y. Nasal commensal Staphylococcus epidermidis enhances interferon-λ-dependent immunity against influenza virus. Microbiome 2019, 7, 80. [Google Scholar] [CrossRef]
- Knosp, C.A.; Johnston, J.A. Regulation of CD4+ T-cell polarization by suppressor of cytokine signalling proteins. Immunology 2012, 135, 101–111. [Google Scholar] [CrossRef]
- Skorvanova, L.; Svancarova, P.; Svetlikova, D.; Betakova, T. Protective efficacy of IFN-ω AND IFN-λs against influenza viruses in induced A549 cells. Acta Virol. 2015, 59, 4137. [Google Scholar] [CrossRef]
- Lachová, V.; Škorvanová, L.; Svetlíková, D.; Turianová, L.; Kostrábová, A.; Betáková, T. Comparison of transcriptional profiles of interferons, CXCL10 and RIG-1 in influenza infected A549 cells stimulated wiThexogenous interferons. Acta Virol. 2017, 61, 183. [Google Scholar] [CrossRef][Green Version]
- Cao, Y.; Huang, Y.; Xu, K.; Liu, Y.; Li, X.; Xu, Y.; Zhong, W.; Hao, P. Differential responses of innate immunity triggered by different subtypes of influenza a viruses in human and avian hosts. BMC Med. Genom. 2017, 10, 70. [Google Scholar] [CrossRef]
- de Groen, R.A.; Groothuismink, Z.M.A.; Liu, B.S.; Boonstra, A. IFN- is able to augment TLR-mediated activation and subsequent function of primary human B cells. J. Leukoc. Biol. 2015, 98, 623–630. [Google Scholar] [CrossRef]
- Ye, L.; Schnepf, D.; Becker, J.; Ebert, K.; Tanriver, Y.; Bernasconi, V.; Gad, H.H.; Hartmann, R.; Lycke, N.; Staeheli, P. Interferon-λ enhances adaptive mucosal immunity by boosting release of thymic stromal lymphopoietin. Nat. Immunol. 2019, 20, 593–601. [Google Scholar] [CrossRef]
- Hemann, E.A.; Green, R.; Turnbull, J.B.; Langlois, R.A.; Savan, R.; Gale, M. Interferon-λ modulates dendritic cells to facilitate T cell immunity during infection with influenza A virus. Nat. Immunol. 2019, 20, 1035–1045. [Google Scholar] [CrossRef]
- Fox, J.M.; Crabtree, J.M.; Sage, L.K.; Tompkins, S.M.; Tripp, R.A. Interferon Lambda Upregulates IDO1 Expression in Respiratory Epithelial Cells after Influenza Virus Infection. J. Int. Cytokine Res. 2015, 35, 554–562. [Google Scholar] [CrossRef]
- Wang, Y.; Li, T.; Chen, Y.; Wei, H.; Sun, R.; Tian, Z. Involvement of NK Cells in IL-28B–Mediated Immunity against Influenza Virus Infection. J. Immunol. 2017, 199, 1012–1020. [Google Scholar] [CrossRef]
- Davidson, S.; Crotta, S.; McCabe, T.M.; Wack, A. Pathogenic potential of interferon αβ in acute influenza infection. Nat. Commun. 2014, 5, 3864. [Google Scholar] [CrossRef]
- Jeon, Y.J.; Lim, J.H.; An, S.; Jo, A.; Han, D.H.; Won, T.B.; Kim, D.Y.; Rhee, C.S.; Kim, H.J. Type III interferons are critical host factors that determine susceptibility to Influenza A viral infection in allergic nasal mucosa. Clin. Exp. Allergy 2018, 48, 253–265. [Google Scholar] [CrossRef]
- Planet, P.J.; Parker, D.; Cohen, T.S.; Smith, H.; Leon, J.D.; Ryan, C.; Hammer, T.J.; Fierer, N.; Chen, E.I.; Prince, A.S. Lambda interferon restructures the nasal microbiome and increases susceptibility to Staphylococcus aureus superinfection. MBio 2016, 7. [Google Scholar] [CrossRef]
- Rich, H.E.; McCourt, C.C.; Zheng, W.Q.; McHugh, K.J.; Robinson, K.M.; Wang, J.; Alcorna, J.F. Interferon lambda inhibits bacterial uptake during influenza superinfection. Infect. Immun. 2019, 87. [Google Scholar] [CrossRef]
- Robertson, C.M.; Perrone, E.E.; McConnell, K.W.; Dunne, W.M.; Boody, B.; Brahmbhatt, T.; Diacovo, M.J.; Van Rooijen, N.; Hogue, L.A.; Cannon, C.L.; et al. Neutrophil Depletion Causes a Fatal Defect in Murine Pulmonary Staphylococcus aureus clearance. J. Surg. Res. 2008, 150, 278–285. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).