Systematic Review of Ticks and Tick-Borne Pathogens of Small Ruminants in Pakistan
Abstract
1. Introduction
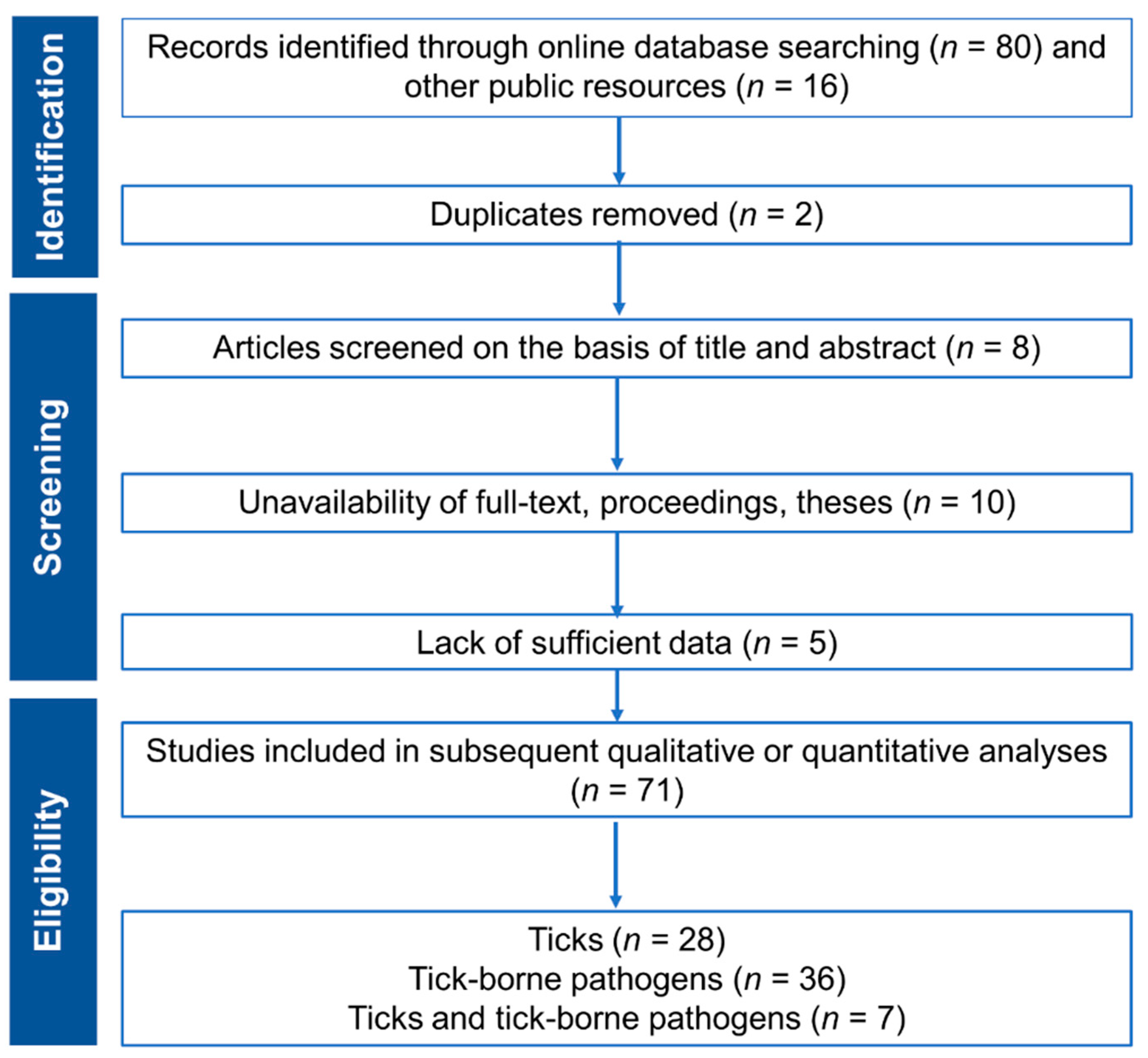
2. Methods
2.1. Review Protocol
2.2. Literature Search
2.3. Quality Assessment and Selection
2.4. Estimation of Prevalence
3. Results and Discussion
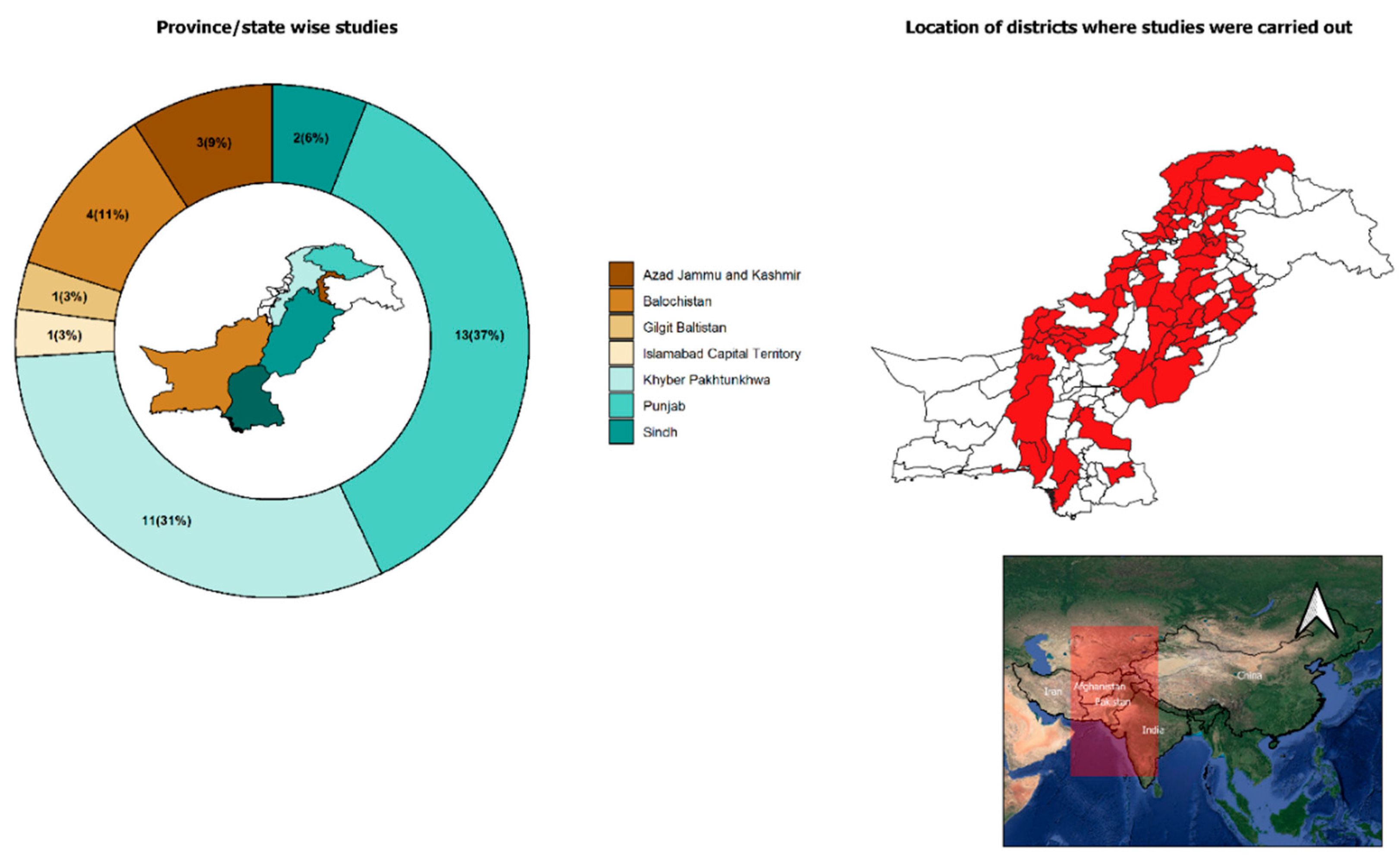
3.1. Studies on Ticks
3.1.1. Tick Species
3.1.2. Epidemiology of Ticks
3.1.3. Identification of Ticks
3.1.4. Control of Ticks in Small Ruminants
| State(s) | District(s) | Host(s) | Tick(s) | Method(s) of Identification | % Infested Animals (Proportion; 95% Confidence Interval) | Reference |
|---|---|---|---|---|---|---|
| Khyber Pakhtunkhwa | Charsadda, Karak, Mardan, Lower Kohistan, Peshawar | Sheep | Haemaphysalis longicornis, Hyalomma impeltatum | Morphological | 16.3 (13/80; 8.2–24.3) | [23] |
| Goats | Hae. montgomeryi, Hae. longicornis, Hy. impeltatum | 68.3 (82/120; 60.0–76.7) | ||||
| Peshawar | Sheep | Dermacentor variabilis, Ixodes ricinus, Rhipicephalus simus, Otobius megnini | Molecular | Not provided | [42] | |
| Goats | Rh. appendiculatus, Rh. microplus, Rh. simus | |||||
| Bannu | Sheep | Not provided | Not performed | 7.8 (39/500; 5.4–10.2) | [43] | |
| Goats | 10.2 (51/500; 7.5–12.9) | |||||
| Bajaur, Khyber, Mohmand, Orakzai, North and South Waziristan | Sheep, Goats | Hae. sulcata, Hae. punctata, Hy. anatolicum, Hy. detritum, Hy. excavatum, Hy. scupense, Rh. microplus, Rh. sanguineus | Morphological | Not Provided | [21] | |
| Karak | Sheep | Hy. marginatum, Rh. annulatus | Morphological | 26.7 (8/30; 10.8–42.5) | [44] | |
| Goats | Hae. bispinosa, Rh. microplus, Rh. sanguineus | 20.0 (9/45; 8.3–31.7) | ||||
| Dera Ismail Khan, Lakki Marwat | Sheep | Not provided | Not performed | 27.3 (9/33; 12.1–42.5) | [45] | |
| Goats | Not provided | 23.1 (34/147; 16.3-29.9) | ||||
| Peshawar | Sheep | Amblyomma, Boophilus, Haemaphysalis, Ixodes and Rhipicephalus species | Morphological | 66.7 (50/75; 56.0–77.3) | [46] | |
| Goats | 73.7 (70/95; 64.8–82.5) | |||||
| Bannu, Chitral, Dir, Mardan, Peshawar, Swat | Sheep | D. raskemensis, Hy. anatolicum, Hy. detritum, Rh. microplus, Rh. sanguineus | Morphological | Not provided | [47] | |
| Goats | Hae. montgomeryi, Hy. anatolicum, Hy. marginatum turanicum, Rh. haemaphysaloides, Rh. microplus, Rh. sanguineus | |||||
| Mansehra | Sheep | Rh. sanguineus | Morphological | Not provided | [48] | |
| Goats | ||||||
| Bajaur, Khyber, Mohmand, North Waziristan, Orakzai | Sheep | Hae. sulcata, Hy. anatolicum, Rh. microplus, Rh. turanicus | Morphological and molecular | Not provided | [22] | |
| Goats | Hae. punctata, Hae. sulcata, Hy. anatolicum, Rh. haemaphysaloides, Rh. microplus, Rh. turanicus | |||||
| Khyber Pakhtunkhwa and Gilgit-Baltistan | Astor, Diamer, Gilgit, Haripur, Kohistan, Mansehra, Shangala | Sheep | Hyalomma and Rhipicephalus spp. | Morphological | 81.5 (189/232; 76.5–86.5) | [49] |
| Goats | 72.1 (263/365; 67.5–76.7) | |||||
| Punjab | Attock, Bahawalpur, Bhakkar, Chakwal, Faisalabad, Gujranwala, Jhang, Khushab, Layyah, Muzaffargarh, Rajanpur, Rawalpindi | Sheep | Hy. anatolicum, Rh. appendiculatus, Rh. decolaratus, Rh. microplus, Rh. sanguineus | Morphological | 29.0 (812/2800; 27.3–30.7) | [25] |
| Goats | Hy. anatolicum, Hy. dromedarii, Hy. marginatum, Rh. appendiculatus, Rh. decolaratus, Rh. microplus, Rh. sanguineus | 36.1 (1012/2800; 34.4–37.9) | ||||
| Toba Tek Singh | Goats | Hy. anatolicum, Rh. microplus | Morphological | 6.7 (270/4020; 5.9–7.5) | [50] | |
| Sargodha | Goats | Hy. anatolicum and Amblyomma, Haemaphysalis, Ixodes, and Rhipicephalus spp. | Morphological | 86.5 (1038/1200; 84.6–88.4) | [51] | |
| Multan | Sheep | Hy. anatolicum, Hy. marginatum, Rh. sanguineus | Morphological | 68.0 (17/25; 49.7–86.3) | [52] | |
| Goats | 40.0 (8/20; 18.5–61.5) | |||||
| Multan | Sheep | Hae. punctata, Hy. anatolicum, Hy. excavatum | Morphological | 50.0 (100/200; 43.1–56.9) | [26] | |
| Goats | Hy. excavatum, Rh. microplus | 40.8 (102/250; 34.7–46.9) | ||||
| Attock, Bahawalpur, Gujranwala, Kasur, Khanewal, Multan, Okara, Rahim Yar Khan, Vehari | Sheep | Hy. anatolicum, Rh. microplus | Morphological and molecular | 11.1 (2/18; 1.3–34.7) | [19] | |
| Goats | Hy. anatolicum, Hy. dromedarii, Rh. microplus, Rh. turanicus | 60.0 (48/80; 49.3–70.7) | ||||
| Layyah, Muzaffargarh | Sheep | No ticks found | Morphological | 0.0 (0/1400; 0.0–0.2) | [24] | |
| Goats | Hy. anatolicum, Rh. sanguineus | 51.6 (723/1400; 49.0–54.3) | ||||
| Layyah, Muzaffargarh | Goats | Hy. anatolicum, Rh. sanguineus | Morphological | 60.1 (481/800; 56.7–63.5) | [53] | |
| Lahore | Sheep | Boophilus, Hyalomma and Rhipicephalus spp. | Morphological | Not provided | [27] | |
| Multan | Goats | Haemaphysalis and Rhipicephalus spp. | Morphological | 43.4 (201/463; 38.9–47.9) | [54] | |
| Faisalabad, Jhang, Toba Tek Singh | Sheep | D. marginatus, Hy. anatolicum, Hy. marginatum isaaci, Rh. annulatus, Rh. microplus, Rh. sanguineus | Morphological | 18.8 (846/4500; 17.7–19.9) | [55] | |
| Goats | Hy. aegyptium, Hy. anatolicum, Hy. marginatum isaaci, Rh. annulatus, Rh. microplus, Rh. sanguineus | 12.3 (553/4500; 11.3–13.2) | ||||
| Lahore, Sheikhupura | Sheep | Hae. burnati, Hy. anatolicum, Rh. annulatus, Rh. microplus, Rh. sanguineus | Morphological | Not provided | [56] | |
| Goats | ||||||
| Punjab and Islamabad Capital Territory | Livestock experimental stations located in Attock and Islamabad Capital Territory | Sheep | Haemaphysalis and Rhipicephalus spp. | Morphological | 43.4 (95/219; 36.8–49.9) | [57] |
| Goats | Amblyomma, Haemaphysalis, Ixodes and Rhipicephalus spp. | 41.5 (184/443; 36.9–46.1) | ||||
| Balochistan | Harnai | Sheep | Hy. anatolicum, Hy. dromedarii, Rh. annulatus, Rh. microplus | Morphological | 30.0 (12/40; 15.8–44.2) | [58] |
| Goats | Am. hebraeum, Hy. anatolicum, Hy. dromedarii, Rh. annulatus | 27.5 (11/40; 13.7–41.3) | ||||
| Mustang and Quetta | Goats | Hy. anatolicum, Hy. excavatum, Rh. appendiculatus, Rh. microplus | Morphological | Not provided | [59] | |
| Specimens collected from 26 districts (names not provided) | Sheep and Goats | Hae. flava, Hy. anatolicum | Morphological | Not provided | [60] | |
| Harnai, Kalat, Killa Abdullah, Khuzdar, Lasbela, Loralai, Pishin, Quetta, Sherani, Sibi, Ziarat, Zhob | Sheep | Hy. anatolicum, Hy. dromedarii, Hy. excavatum, Hy. marginatum, Hy. scupense, Rh. microplus, Rh. turanicus | Morphological and molecular | Not provided | [61] | |
| Goats | Hy. anatolicum, Hy. dromedarii, Hy. excavatum, Hy. marginatum | |||||
| Sindh | Khairpur, Larkana, Sehwan, Thatta, Umerkot | Sheep | Hae intermedia, Hae kutchensis, Hae. bispinosa, Hy. anatolicum, Hy. bravepunctata, Hy. detritum, Hy. dromedarii, Hy. hussaini, Hy. impeltatum, Hy. marginatum isaaci, Hy. marginatum turanicum, Rh. annulatus, Rh. haemaphysaloides, Rh. microplus, Rh. sanguineus, Rh. turanicus | Morphological | Not provided | [62] |
| Goats | ||||||
| Khairpur | Goats | Hy. anatolicum, Hy. dromedarii, Hy. impeltatum, Hy. marginatum isaaci, Rh. haemaphysaloides, Rh. turanicus | Morphological | Not provided | [63] | |
| Azad Jammu and Kashmir | Muzaffarabad | Sheep | Not provided | Not performed | 22.2 (2/9; 2.8–60.0) | [64] |
| Goats | 46.3 (19/41; 31.1–61.6) | |||||
| Poonch | Sheep | Haemaphysalis, Hyalomma and Otobius spp. | Morphological | 54.7 (82/150; 46.7–62.6) | [65] | |
| Goats | 48.3 (145/300; 42.7–54.0) | |||||
| Poonch | Sheep | Hy. anatolicum | Morphological | 54.7 (164/300; 49.0–60.3) | [66] | |
| Goats | 48.0 (288/600; 44.0–52.0) | |||||
| Azad Jammu and Kashmir, Balochistan, Gilgit Baltistan, Khyber Pakhtunkhwa, Punjab and Sindh | District information not provided | Sheep | Ar. persicus, Hae. bispinosa, Hae. cornupunctata, Hae kashmirensis, Hae. montomeryi, Hy. anatolicum, Hy. dromedarii, Hy. hussaini, Hy. isaaci, Hy. scupense, Or. tholozani, Rh. haemaphysaloides, Rh. microplus | Morphological | Not provided | [20] |
| Goats | Same as above except Argas (Ar.) persicus absent | |||||
| Azad Kashmir, Balochistan, Gilgit Baltistan, Khyber Pakhtunkhwa, Punjab and Sindh | Specimens were collected from 12 administrative divisions of West Pakistan and Azad Kashmir | Sheep | D. raskemensis, Hae. bispinosa, Hae. cornupunctata, Hae. kashmirensis, Hae. montgomeryi, Hae. sulcata, Hy. anatolicum, Hy. asiaticum, Hy. detritum, Hy. dromedarii, Hy. excavatum, Hy. kumari, Hy. marginatum isaaci, Hy. marginatum turanicum, Rh. annulatus, Rh. haemaphysaloides, Rh. microplus, Rh. sanguineus, Rh. turanicus | Morphological | Not provided | [41] |
| Goats | Same species as above except Hy. excavatum and Rh. annulatus absent |
| Ticks | Number of Species Reported | Selected References | |
|---|---|---|---|
| Hyalomma (Hy.) | Hy. anatolicum, Hy. asiaticum, Hy. bravepunctata, Hy. detritum, Hy. dromedarii, Hy. excavatum, Hy. hussaini, Hy. impeltatum, Hy. kumari, Hy. marginatum, Hy. marginatum isaaci, Hy. marginatum turanicum, Hy. scupense | 13 | [20,21,22,23,25,41,47,55] |
| Rhipicephalus (Rh.) | Rh. annulatus, Rh. appendiculatus, Rh. decolaratus, Rh. haemaphysaloides, Rh. microplus, Rh. sanguineus, Rh. simus, Rh. turanicus | 8 | [20,21,22,25,41,47,55] |
| Haemaphysalis (Hae.) | Hae. burnati, Hae. bispinosa, Hae. cornupunctata, Hae. flava, Hae. intermedia, Hae. kashmirensis, Hae. kutchensis, Hae. longicornis, Hae. montgomeryi, Hae. punctata, Hae. sulcata | 11 | [20,21,22,23,41,47] |
| Other ixodids and argasids | Amblyomma hebraeum, Dermacentor marginatus, D. variabilis, D. raskimensis, Ixodes ricinus, Ar. persicus, Otobius megnini, Ornithodoros tholozani | 8 | [20,41,47,55] |
| Tick-Borne Pathogens | |||
| Anaplasma (A.) | A. centrale, A. marginale, A. ovis | 3 | [22,86,87] |
| Babesia (B.) | B. ovis | 1 | [28,88] |
| Theileria (T.) | T. annulata, T. luwenshuni, T. ovis, T. lestoquardi, T. sp. MK, T. sp. OT1 | 6 | [27,89] |
| Rickettsia (R.) | Candidatus R. amblyommii, R. aeschlimannii, R. conorii, R. massiliae, R. slovaca, | 5 | [20,22] |
| Other pathogens | Coxiella burnetii, Crimean-Congo haemorrhagic fever virus | 2 | [32,34,35] |
| Study Type | Drug(s) Tested (Concentration/Dose/Method of Application) | Number of Animals per Group | Duration of Trial (Days) | Efficacy (%) | Reference | ||
|---|---|---|---|---|---|---|---|
| Sheep | Goats | Sheep | Goats | ||||
| Acaricidal efficacy against ticks | Diazinon (0.6% spray) | 20 | 20 | 56 | 89.5 | 92 | [48] |
| Coumaphos (0.1% spray) | 20 | 20 | 93.6 | 95 | |||
| Cypermethrin (2% spray) | 20 | 20 | 100 | 100 | |||
| Ivermectin (0.2 mg/kg, injection) | NS | 90 | 20 | NA | No | [53] | |
| Cypermethrin (5% spray) | 90 | Yes | |||||
| Drug efficacy against anaplasmosis | Oxytetracycline (1 mL/kg, injection) | NS | 10 | 30 * | NA | 30 | [98] |
| Imidocarb dipropionate (0.1/kg, injection) | 10 | 80 | |||||
| Diminazene aceturate (0.3 mL/kg, injection) | 10 | 60 | |||||
| Oxytetracycline (20 mg/kg, injection) | 4 | 4 | 10 | 100 | 100 | [87] | |
| Imidocarb dipropionate (3 mg/kg, injection) | 4 | 4 | 100 | 87.5 | |||
| Diminazene aceturate (3.5–7 mg/kg, injection) | 4 | 4 | 50 | 75 | |||
| Drug efficacy against babesiosis | Imidocarb dipropionate + oxytetracycline (2 mg/kg + 10 mg/kg, injection) | 10 | 10 | 10 | 100 | 100 | [99] |
| Imidocarb dipropionate (2 mg/kg, injection) | 10 | 10 | 80 | 80 | |||
| Diminazene aceturate + oxytetracycline (3.5 mg/kg + 10 mg/kg, injection) | 10 | 10 | 80 | 90 | |||
| Diminazene aceturate (3.5 mg/kg, injection) | 10 | 10 | 70 | 70 | |||
| Imidocarb dipropionate (2 mg/kg, injection) | 10 | NS | 10 | 100 | NA | [100] | |
| Diminazene aceturate (3.5 mg/kg, injection) | 10 | 80 | |||||
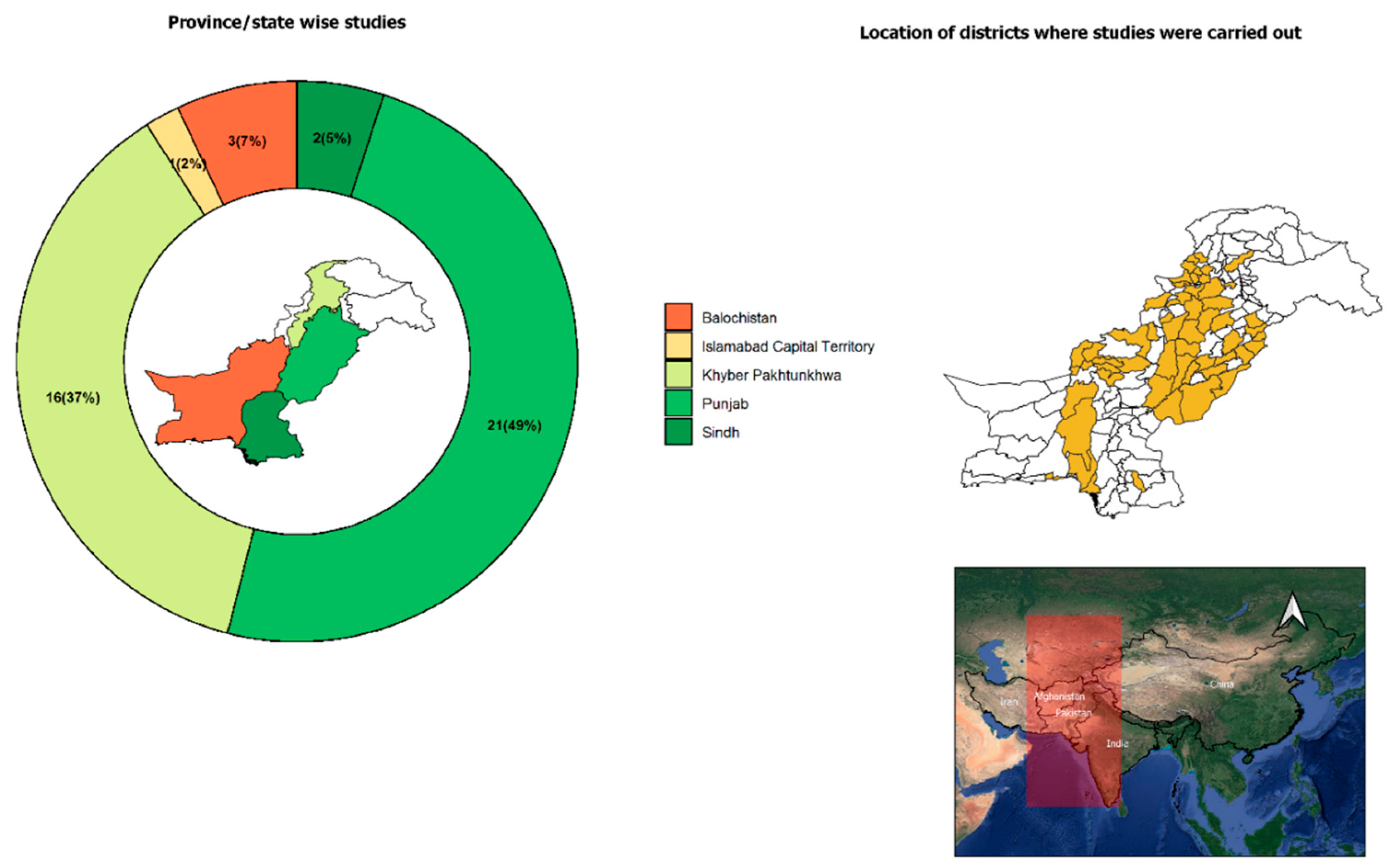
3.2. Tick-Borne Pathogens in Pakistani Small Ruminants and Their Ticks
3.2.1. Anaplasmosis
3.2.2. Babesiosis
3.2.3. Theileriosis
3.2.4. Other Tick-Borne Diseases (TBDs) of Small Ruminants
3.2.5. Diagnosis and Control of TBPs in Pakistan
| State | District(s) | Host(s)/Vector | Method(s) of Detection | Target Animal Population | Pathogen(s) Detected | % Test-Positive (Proportion; 95% CI) | Reference |
|---|---|---|---|---|---|---|---|
| Punjab | Lahore | Sheep | Morphological | Suspected of anaplasmosis | Anaplasma ovis | 55.3 (83/150; 47.4–63.3) | [87] |
| Goats | 30.7 (46/150; 23.3–38.0) | ||||||
| Mianwali | Sheep | Molecular | Healthy | Anaplasma species | 32.0 (24/75; 21.4–42.6) | [106] | |
| Goats | 25.3 (19/75; 15.5–35.2) | ||||||
| Sheep and goats | Morphological | 29.3 (44/150; 22.0–36.6) | |||||
| Attock, Bahawalpur, Gujranwala, Kasur, Khanewal, Multan, Okara, Rahim Yar Khan, Vehari | Ticks from sheep and goats | Molecular | Healthy | A. ovis, A. centrale, A. marginale, A. platys-like organism, Anaplasma sp. BL099-6 | 38.9 (21/54; 26.2–53.1) | [86] | |
| Khyber Pakhtunkhwa | Charsadda, Mardan, Nowshera, Peshawar | Sheep | Morphological | Suspected of anaplasmosis | Anaplasma sp. | 29.6 (32/108; 21.0–38.2) | [107] |
| Goats | 1.7 (1/60; 0.0–8.9) | ||||||
| Karak | Sheep | Morphological | Suspected of anaplasmosis | A. marginale | 22.0 (55/250; 16.9–27.1) | [29] | |
| Goats | 17.2 (43/250; 12.5–21.9) | ||||||
| Sheep | Serological * | 36.8 (92/250; 30.8–42.8) | |||||
| Goats | 32.8 (82/250; 27.0–38.6) | ||||||
| Sheep | Molecular | 47.2 (118/250; 41.0–53.4) | |||||
| Goats | 34.8 (87/250; 28.9–40.7) | ||||||
| Peshawar | Sheep | Serological | Healthy | A. marginale | 24.5 (92/376; 20.1–28.8) | [108] | |
| Charsadda | Sheep | Serological | Healthy | Anaplasma sp. | 19.3 (58/300; 14.9–23.8) | [109] | |
| Goats | 25.0 (75/300; 20.1–29.9) | ||||||
| District information not provided | Goats | Morphological | Healthy | A. ovis | 9.6 (7/73; 2.8–16.3) | [110] | |
| Mardan | Sheep | Morphological | Healthy | Anaplasma sp. | 13.9 (25/180; 8.8–18.9) | [111] | |
| Goats | 8.3 (15/180; 4.3–12.4) | ||||||
| Sheep | Serological | 23.9 (43/180; 17.7–30.1) | |||||
| Goats | 20.6 (37/180; 14.7–26.5) | ||||||
| Bajaur, Khyber, Mohmand, North Waziristan, Orakzai | Ticks from sheep | Molecular | Healthy | A. centrale, A. marginale, A. ovis | 39.1 (9/23; 19.2–59.1) | [22] | |
| Ticks from goats | 35.5 (11/31; 18.6–52.3) | ||||||
| Peshawar | Sheep | Morphological and molecular | Suspected of anaplasmosis | Anaplasma sp. | 28.0 (28/100; 19.2–36.8) | [112] | |
| Sindh | Mirpur Khas | Goats | Morphological | Healthy | A. marginale | 13.3 (40/300; 9.5–17.2) | [98] |
| State(s) | District(s) | Host(s)/Ticks | Method(s) of Detection | Target Animal Population | Pathogen(s) Detected | % Test-Positive (Proportion; 95% CI) | Reference |
|---|---|---|---|---|---|---|---|
| Punjab | Lahore | Sheep | Morphological | Suspected of piroplasmosis | Theileria sp. | 22.0 (44/200; 16.3–27.7) | [27] |
| Molecular | Theileria ovis | 27.5 (55/200; 21.3–33.7) | |||||
| T. lestoquardi | 7.5 (15/200; 3.8–11.2) | ||||||
| Ticks from sheep | Not applicable | T. ovis | 65.9 (27/41; 51.3-80.4) | ||||
| T. lestoquardi | 66.7 (30/45; 52.9-80.4) | ||||||
| Bahawalnagar, Dera Ghazi Khan, Layyah, Multan, Muzaffargarh | Sheep | Molecular | Healthy | T. lestoquardi | 8.2 (4/49; 0.5-15.8) | [125] | |
| Goats | 0.0 (0/66; 0.0-5.4) | ||||||
| Lahore | Sheep | Morphological | Suspected of piroplasmosis | Babesia sp. | 23.5 (57/243; 18.1–28.8) | [99] | |
| Goats | 13.5 (51/377; 10.1–17.0) | ||||||
| Bahawalnagar, Dera Ghazi Khan, Khanewal, Layyah, Multan, Muzaffargarh, Vehari | Sheep | Molecular | Healthy | Babesia ovis | 50.0 (20/40; 34.5–65.5) | [118] | |
| Goats | 23.9 (16/67; 13.7–34.1) | ||||||
| Lahore | Sheep | Morphological | Suspected of piroplasmosis | Theileria sp. | 13.9 (38/273; 9.8–18.0) | [123] | |
| Goats | 8.2 (21/256; 4.8–11.6) | ||||||
| Sahiwal | Sheep | Morphological | Healthy | Babesia sp. | 9.7 (30/310; 6.4–13.0) | [100] | |
| Multan | Sheep and goats | Morphological | Healthy | Theileria sp. | 3.7 (11/300; 1.5–5.8) | [126] | |
| Sheep | Molecular | T. ovis | 15.3 (23/150; 9.6–21.1) | ||||
| T. lestoquardi | 10.7 (16/150; 5.7–15.6) | ||||||
| Goats | T. ovis | 5.3 (8/150; 2.5–10.6) | |||||
| T. lestoquardi | 4.0 (6/150; 1.6–8.9) | ||||||
| Multan | Sheep and goats | Morphological | Healthy | Theileria sp. | 12.4 (31/250; 8.3–16.5) | [127] | |
| Sheep | Molecular | 16.0 (25/156; 10.3–21.8) | |||||
| Goats | 69.1 (65/94; 59.8–78.5) | ||||||
| Attock, Bahawalpur, Gujranwala, Kasur, Khanewal, Multan, Okara, Rahim Yar Khan, Vehari | Ticks from sheep and goats | Molecular | Healthy | Babesia and Theileria spp. | Not provided | [86] | |
| Multan | Sheep and goats | Morphological | Healthy | Theileria sp. | 12.5 (25/200; 7.9–17.1) | [128] | |
| Molecular | 39.5 (79/200; 32.7–46.3) | ||||||
| Livestock Experimental Stations, Okara | Sheep | Morphological | Healthy | B. ovis | 29.0 (58/200; 22.7–35.3) | [28] | |
| T. ovis | 37.0 (74/200; 30.3–43.7) | ||||||
| Molecular | B. ovis | 55.0 (110/200; 48.1–61.9) | |||||
| T. ovis | 7.5 (15/200; 3.8–11.2) | ||||||
| Multan | Goats | Morphological | Healthy | Theileria | 5.4 (25/463; 3.3–7.5) | [54] | |
| Molecular | T. ovis, T. lestoquardi | 16.0 (74/463; 12.6–19.3) | |||||
| Okara | Sheep | Morphological | Healthy | Theileria sp. | 16.5 (66/400; 12.9–20.1) | [129] | |
| Punjab and Islamabad Capital Territory | Livestock Experimental Stations located at Attock and Islamabad Capital Territory | Sheep | Morphological | Healthy | Theileria sp. | 7.4 (7/95; 2.1–12.6) | [57] |
| Goats | 3.8 (7/184; 1.0–6.6) | ||||||
| Punjab and Khyber Pakhtunkhwa | Kohat, Multan | Sheep | Molecular | Healthy | Theileria sp. | 31.7 (26/82; 21.6–41.8) | [88] |
| Goats | 5.3 (6/114; 1.2–9.4) | ||||||
| Multan | Sheep and goats | 5.5 (7/128; 1.5–9.4) | |||||
| Kohat | 34.7 (25/72; 23.7–45.7) | ||||||
| Dera Ghazi Khan, Kohat, Layyah, Multan, Rahim Yar Khan | Sheep | Molecular | Healthy | T. ovis | 11.1 (11/99; 4.9–17.3) | [130] | |
| Goats | 0.9 (1/111; 0.0–4.9) | ||||||
| Sheep and goats | Morphological | 1.0 (2/210; 0.1–3.4) | |||||
| Dera Ghazi Khan, Layyah, Multan, Rahim Yar Khan | Molecular | 1.7 (2/118; 0.3–6.6) | |||||
| Kohat | 10.9 (10/92; 4.5–17.2) | ||||||
| Khyber Pakhtunkhwa | Charsadda, Mardan, Nowshera, Peshawar | Sheep | Morphological | Suspected of piroplasmosis | Theileria sp. | 15.7 (17/108; 8.9–22.6) | [107] |
| Babesia sp. | 14.8 (16/108; 8.1–21.5) | ||||||
| Goats | Theileria sp. | 0.0 (0/60; 0.0–7.5) | |||||
| Babesia sp. | 41.7 (25/60; 29.2–54.1) | ||||||
| Lower Dir | Sheep | Molecular | Healthy | T. annulata, T. luwenshuni, T. ovis, Theileria sp. MK, Theileria sp. OT1 | 72.5 (58/80; 62.7–82.3) | [89] | |
| Goats | 40.8 (49/120; 32.0–49.6) | ||||||
| Kohat, Peshawar | Sheep | Molecular | Healthy | T. lestoquardi | 4.5 (2/44; 0.6–15.5) | [131] | |
| Goats | 2.5 (3/121; 0.5–7.0) | ||||||
| Khyber, Peshawar | Sheep | Morphological | Healthy | Babesia sp. | 7.0 (21/300; 4.1–9.9) | [132] | |
| Goats | Theileria sp. | 6.0 (18/300; 3.3–8.7) | |||||
| Bannu, Dera Ismail Khan, Tank | Goats | Molecular | Healthy | Theileria ovis | 9.0 (54/600; 6.7–11.3) | [31] | |
| T. lestoquardi | 5.3 (32/600; 3.5–7.1) | ||||||
| Bannu, Dera Ismail Khan, Tank | Sheep | Morphological | Healthy | Theileria sp. | 20 (120/600; 16.8–23.2) | [133] | |
| Mansehra | Ticks from sheep | Morphological | Not applicable | B. ovis | 1.5 (3/200; 0.3–4.3) | [48] | |
| Ticks from goats | 1.0 (2/201; 0.1–3.5) | ||||||
| Bannu, Dera Ismail Khan, Tank | Sheep | Molecular | Healthy | T. ovis | 13.0 (78/600; 10.3–15.7) | [30] | |
| T. lestoquardi | 9.0 (54/600; 6.7–11.3) | ||||||
| Bajaur, Khyber, Mohmand, North Waziristan, Orakzai | Ticks from sheep | Molecular | Healthy | Theileria sp. | 30.4 (7/23; 11.6–49.2) | [22] | |
| Ticks from goats | 35.5 (11/31; 18.6–52.3) | ||||||
| Ticks from sheep | B. ovis | 0.0 (0/23; 0.0–17.8) | |||||
| Ticks from goats | 0.0 (0/31; 0.0–13.7) | ||||||
| Balochistan | Loralai, Quetta | Sheep | Morphological and molecular | Healthy | T. ovis | 5.5 (120/2200; 4.5–6.4) | [134] |
| T. lestoquardi | 15.4 (338/2200; 13.9–16.9) | ||||||
| Goats | T. ovis | 6.7 (45/670; 4.8–8.6) | |||||
| T. lestoquardi | 3.0 (20/670; 1.7–4.3) |
| State | District(s) | Host(s) | Method(s) of Detection | Target Animal Population | Pathogen(s) Detected | % Test-Positive (Proportion; 95% CI) | Reference |
|---|---|---|---|---|---|---|---|
| Punjab | Bahawalpur, Bhakkar, Khanewal, Khushab, Layyah, Okara, Rajanpur | Sheep | Serological (ELISA) | Healthy | Coxiella burnetti (antibodies) | 15.6 (78/500; 12.4–18.8) | [34] |
| Goats | 15.0 (75/500; 11.9–18.1) | ||||||
| Ticks from sheep | Molecular (qPCR) | Coxiella burnetti (DNA) | 31.0 (9/29; 14.2–47.9) | ||||
| Ticks from goats | 7.7 (2/26; −2.6–17.9) | ||||||
| Layyah, Muzaffargarh | Sheep | Serological (ELISA) | Healthy | C. burnetti (antibodies) | 33.2 (90/271; 27.6–38.8) | [33] | |
| Goats | 28.4 (77/271; 23.0–33.8) | ||||||
| Attock, Chakwal, DG Khan, Faisalabad, Gujranwala, Lahore, Sahiwal, Sargodha, Sheikhupur | Sheep | Serological (ELISA) | Healthy | C. burnetti (antibodies) | 17.9% (33/184; 12.4–23.5) | [136] | |
| Goat | 16.4% (46/280; 12.1–20.8) | ||||||
| Khyber Pakhtunkhwa | Bajaur, Khyber, Mohmand, North Waziristan Orakzai | Ticks from sheep | Molecular (qPCR) | Not applicable | Rickettsia (DNA) | 73.9 (71/23; 56.0–91.9) | [22] |
| Ehrlichia (DNA) | 8.7 (2/23; 1.1–22.0) | ||||||
| Francisella-like (DNA) | 30.4 (7/23; 11.6–49.2) | ||||||
| Coxiella-like (DNA) | 8.7 (2/23; 1.1–22.0) | ||||||
| Ticks from goats | Rickettsia (DNA) | 83.9 (26/31; 70.9–96.8) | |||||
| Ehrlichia (DNA) | 3.2 (1/31; 0.0–16.7) | ||||||
| Francisella-like (DNA) | 16.1 (5/31; 3.2–29.1) | ||||||
| Coxiella-like (DNA) | 6.5 (2/31; 1.1–22.8) | ||||||
| Balochistan | Harnai, Kalat, Killa Abdullah, Khuzdar, Lasbela, Loralai, Pishin, Quetta, Sherani, Sibi, Ziarat, Zhob | Ticks from sheep and goats | Molecular (qPCR) | Not applicable | Crimean-Congo haemorrhagic fever Virus (DNA) | 3.8 (20/525; 2.2–5.4) | [61] |
| Harnai, Kalat, Killa Abdullah, Khuzdar, Lasbela, Loralai, Pishin, Quetta, Sherani, Sibi, Ziarat, Zhob | Sheep | Serological (ELISA and IFA) and molecular | Healthy | CCHF virus (antibodies) | 18.6 (149/800; 15.9–21.3) | [35] | |
| Goats | 4.6 (37/800; 3.2–6.1) | ||||||
| Sindh | Karachi | Sheep | Serological (CFT) | Not applicable | C. burnetti (antibodies) | 18.3 (11/60; 8.5–28.1) | [135] |
| Goats | 4.6 (3/65; 1.0–12.9) | ||||||
| Punjab, Sindh, Khyber Pakhtunkhwa, and Balochistan | District information not provided | Sheep | Serological (ELISA) | Healthy | CCHF virus (antigen) | 32.5 (138/424; 28.1–37.0) | [32] |
| Goats | 18.9 (83/440; 15.2–22.5) | ||||||
| Azad Jammu and Kashmir, Balochistan, Gilgit Baltistan, Khyber Pakhtunkhwa, Punjab and Sindh | District information not provided | Ticks from sheep and goats | Next-Generation Sequencing | Not applicable | Several bacterial species (DNA) | Not provided | [20] |
4. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- FAO; IFAD; UNICEF; WFP; WHO. The State of Food Security and Nutrition in the World. Safeguarding Against Economic Slowdowns and Downturns; FAO: Rome, Italy, 2019; Available online: http://www.fao.org/3/ca5162en/ca5162en.pdf (accessed on 15 September 2020).
- Upton, M. The Role of Livestock in Economic Development and Poverty Reduction; PPLPI Working Papers 23783, Food and Agriculture Organization of the United Nations, Pro-Poor Livestock Policy Initiative; FAO: Rome, Italy, 2004. [Google Scholar] [CrossRef]
- Hatab, A.A.; Cavinato, M.E.R.; Lagerkvist, C.J. Urbanization, livestock systems and food security in developing countries: A systematic review of the literature. Food Secur. 2019, 11, 279–299. [Google Scholar] [CrossRef]
- Akinmoladun, O.F.; Muchenje, V.; Fon, F.N. Small ruminants: Farmers’ hope in a world threatened by water scarcity. Animals 2019, 9, 456. [Google Scholar] [CrossRef]
- Devendra, C. Small ruminants: Imperatives for productivity enhancement improved livelihoods and rural growth-a review. Asian Australas. J. Anim. Sci. 2001, 14, 1483–1496. [Google Scholar] [CrossRef]
- FAO. Shaping the future of livestock sustainably, responsibly, efficiently. In Proceedings of the 10th Global Forum for Food and Agriculture (GFFA), Berlin, Germany, 18–20 January 2018; Available online: http://www.fao.org/3/I8384EN/i8384en.pdf (accessed on 15 September 2020).
- Devendra, C. Small ruminants in Asia. Contribution to food security, poverty alleviation and opportunities for productivity enhancement. In Proceedings of the International Workshop on Small Ruminant Production and Development in South East Asia; MEKARN: Nong Lam, Ho Chi Minh, Vietnam, 2005; pp. 19–32. [Google Scholar]
- Government of Pakistan. Pakistan Economic Survey 2019-2020; Ministry of Finance: Islamabad, Pakistan, 2020; pp. 17–41. Available online: http://www.finance.gov.pk/survey_1920.html (accessed on 15 September 2020).
- Khan, M.S.; Khan, M.; Mahmood, S. Genetic resources and diversity in Pakistani goats. Int. J. Agric. Biol. 2008, 10, 227–231. [Google Scholar]
- Khan, M.S.; Khan, M.; Ahmad, S.; Mahmood, S. Genetic resources and diversity in Pakistani sheep. Int. J. Agric. Biol. 2007, 6, 941–944. [Google Scholar]
- Agricultural Census Organization. Pakistan Livestock Census; Government of Pakistan: Islamabad, Pakistan, 2006. Available online: http://www.pbs.gov.pk/content/pakistan-livestock-census-2006 (accessed on 15 September 2020).
- Ghaffar, A.; Anwar, M.; Khan, M. Socio-economic importance, production systems, research and development of small ruminants in Pakistan. In Proceedings of the ACIAR, Bogor, Indonesia, 22–25 April 1996; pp. 21–26. [Google Scholar]
- Ribeiro, D.M.; Salama, A.; Vitor, A.C.; Arguello, A.; Moncau, C.T.; Santos, E.M.; Caja, G.; de Oliveira, J.S.; Balieiro, J.C.; Hernández-Castellano, L.E. The application of omics in ruminant production: A review in the tropical and sub-tropical animal production context. J. Proteom. 2020, 227, 103905. [Google Scholar] [CrossRef]
- Jabbar, A.; Abbas, T.; Sandhu, Z.D.; Saddiqi, H.A.; Qamar, M.F.; Gasser, R.B. Tick-borne diseases of bovines in Pakistan: Major scope for future research and improved control. Parasit. Vectors 2015, 8, 283. [Google Scholar] [CrossRef] [PubMed]
- Uilenberg, G. Veterinary Significance of Ticks and Tick-Borne Diseases. In Tick Vector Biology: Medical and Veterinary Aspects; Springer: Berlin/Heidelberg, Germany, 1992; pp. 23–33. [Google Scholar]
- De la Fuente, J.; Estrada-Pena, A.; Venzal, J.M.; Kocan, K.M.; Sonenshine, D.E. Overview: Ticks as vectors of pathogens that cause disease in humans and animals. Front. Biosci. 2008, 13, 6938–6946. [Google Scholar] [CrossRef] [PubMed]
- Uilenberg, G. General review of tick-borne diseases of sheep and goats world-wide. Parassitologia 1997, 39, 161–165. [Google Scholar] [PubMed]
- Ahmed, J.S.; Luo, J.; Schnittger, L.; Seitzer, U.; Jongejan, F.; Yin, H. Phylogenetic position of small-ruminant infecting piroplasms. Ann. N. Y. Acad. Sci. 2006, 1081, 498–504. [Google Scholar] [CrossRef]
- Rehman, A.; Nijhof, A.M.; Sauter-Louis, C.; Schauer, B.; Staubach, C.; Conraths, F.J. Distribution of ticks infesting ruminants and risk factors associated with high tick prevalence in livestock farms in the semi-arid and arid agro-ecological zones of Pakistan. Parasit. Vectors 2017, 10, 190. [Google Scholar] [CrossRef]
- Karim, S.; Budachetri, K.; Mukherjee, N.; Williams, J.; Kausar, A.; Hassan, M.J.; Adamson, S.; Dowd, S.E.; Apanskevich, D.; Arijo, A.; et al. A study of ticks and tick-borne livestock pathogens in Pakistan. PLoS Negl. Trop. Dis. 2017, 11, e0005681. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Nasreen, N.; Niaz, S.; Sajjad Ali Shah, S.; Mitchell, R.D., III; Ayaz, S.; Naeem, H.; Khan, L.; De León, A.P. Tick burden and tick species prevalence in small ruminants of different agencies of the Federally Administered Tribal Areas (FATA), Pakistan. Int. J. Acarol. 2019, 45, 374–380. [Google Scholar] [CrossRef]
- Ghafar, A.; Khan, A.; Cabezas-Cruz, A.; Gauci, C.G.; Niaz, S.; Ayaz, S.; Mateos-Hernández, L.; Galon, C.; Nasreen, N.; Moutailler, S. An assessment of the molecular diversity of ticks and tick-borne microorganisms of small ruminants in Pakistan. Microorganisms 2020, 8, 1428. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Khan, M.A.; Zahid, H.; Yaseen, P.M.; Khan, M.Q.; Nawab, J.; Rehman, Z.U.; Ateeq, M.; Khan, S.; Ibrahim, M. Seasonal dynamics, record of ticks infesting humans, wild and domestic animals and molecular phylogeny of Rhipicephalus microplus in Khyber Pakhtunkhwa Pakistan. Front. Physiol. 2019, 10. [Google Scholar] [CrossRef]
- Sajid, M.S.; Iqbal, Z.; Khan, M.N.; Muhammad, G. Point prevalence of hard ticks (ixodids) infesting domestic ruminants of lower Punjab, Pakistan. Int. J. Agric. Biol. 2008, 10, 349–351. [Google Scholar]
- Batool, M.; Nasir, S.; Rafique, A.; Yousaf, I.; Yousaf, M. Prevalence of tick infestation in farm animals from Punjab, Pakistan. Pak. J. Zool. 2019, 39, 406–410. [Google Scholar]
- Ramzan, M.; Naeem-Ullah, U.; Saba, S.; Iqbal, N.; Saeed, S. Prevalence and identification of tick species (Ixodidae) on domestic animals in district Multan, Punjab Pakistan. Int. J. Acarol. 2020, 46, 83–87. [Google Scholar] [CrossRef]
- Durrani, A.Z.; Younus, M.; Kamal, N.; Mehmood, N.; Shakoori, A.R. Prevalence of ovine Theileria species in district Lahore, Pakistan. Pak. J. Zool. 2011, 43, 57–60. [Google Scholar]
- Shahzad, W.; Haider, N.; Mansur-ud-Din, A.; Munir, R.; Saghar, M.S.; Mushtaq, M.H.; Ahmad, N.; Akbar, G.; Mehmood, F. Prevalence and molecular diagnosis of Babesia ovis and Theileria ovis in Lohi sheep at livestock experiment station (LES), Bahadurnagar, Okara, Pakistan. Iran. J. Parasitol. 2013, 8, 570–578. [Google Scholar]
- Hussain, M.; Junaid, A.; Gul, R.; Jamal, M.A.; Ahmed, I.; Talpur, M.Z.; Rahim, K.; Fatima, M.; Munir, S. First detection on prevalence of Anaplasma marginale in sheep and goat in Karak District, Pakistan. Asian Pac. J. Trop. Dis. 2017, 7, 531–535. [Google Scholar] [CrossRef]
- Ullah, N.; Durrani, A.Z.; Ullah, S.; Ullah, S.; Shah, M.K.; Khan, A.Z.; Khan, M.S.; Khan, N.U.; Khan, M.A. A study on potential factors and physiological biomarkers associated with the occurrence of ovine theileriosis. Small Rumin. Res. 2018, 168, 32–38. [Google Scholar] [CrossRef]
- Ullah, N.; Durrani, A.Z.; Avais, M.; Ahmad, N.; Ullah, S.; Ullah, S.; Khan, M.A.; Khan, N.U. A first report on prevalence of caprine theileriosis and its association with host biomarkers in Southern Khyber Pakhtunkhwa, Pakistan. Small Rumin. Res. 2018, 159, 56–61. [Google Scholar] [CrossRef]
- Zohaib, A.; Saqib, M.; Athar, M.A.; Hussain, M.H.; Sial, A.-U.-R.; Tayyab, M.H.; Batool, M.; Sadia, H.; Taj, Z.; Tahir, U. Crimean-Congo hemorrhagic fever virus in humans and livestock, Pakistan, 2015–2017. Emerg. Infect. Dis. 2020, 26, 773–777. [Google Scholar] [CrossRef]
- Zahid, M.U.; Hussain, M.H.; Saqib, M.; Neubauer, H.; Abbas, G.; Khan, I.; Mansoor, M.K.; Asi, M.N.; Ahmad, T.; Muhammad, G. Seroprevalence of Q fever (Coxiellosis) in small ruminants of two districts in Punjab, Pakistan. Vector Borne Zoonotic Dis. 2016, 16, 449–454. [Google Scholar] [CrossRef]
- Ullah, Q.; El-Adawy, H.; Jamil, T.; Jamil, H.; Qureshi, Z.I.; Saqib, M.; Ullah, S.; Shah, M.K.; Khan, A.Z.; Zubair, M. Serological and molecular investigation of Coxiella burnetii in small ruminants and ticks in Punjab, Pakistan. Int. J. Environ. Res. Public Health 2019, 16, 4271. [Google Scholar] [CrossRef]
- Kasi, K.K.; Sas, M.A.; Sauter-Louis, C.; von Arnim, F.; Gethmann, J.M.; Schulz, A.; Wernike, K.; Groschup, M.H.; Conraths, F.J. Epidemiological investigations of Crimean-Congo haemorrhagic fever virus infection in sheep and goats in Balochistan, Pakistan. Ticks Tick Borne Dis. 2020, 11, 101324. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Bush, A.O.; Lafferty, K.D.; Lotz, J.M.; Shostak, A.W. Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol. 1997, 83, 575–583. [Google Scholar] [CrossRef]
- Dorai-Raj, S. binom: Binomial Confidence Intervals for Several Parameterizations. R Package Version 1.1.1. Available online: http://cran.r-project.org/package=binom (accessed on 15 September 2020).
- Clopper, C.; Pearson, E.S. The use of confidence or fiducial limits illustrated in the case of the binomial. Biometrika 1934, 26. [Google Scholar] [CrossRef]
- Koch, H.G. Suitability of white-tailed deer, cattle, and goats as hosts for the lone star tick, Amblyomma americanum (Acari: Ixodidae). J. Kans. Entomol. Soc. 1988, 61, 251–257. [Google Scholar]
- McCarthy, V.C. Ixodid ticks (Acarina, Ixodidae) of West Pakistan. Ph.D. Thesis, University of Maryland, College Park, MD, USA, 1967. [Google Scholar]
- Aziz, S.; Shah, S.F.; Amin, F.; Khan, M.A.; Ahmad, M. Taxonomic study of arthropod pests of livestock in district Peshawar, Khyber Pakhtunkhwa. Pak. J. Life Soc. Sci. 2018, 16, 85–96. [Google Scholar]
- Khan, N.U.; Shafee, M.; Khan, J.A.; Ashraf, K.; Khan, R.; Khan, A.; Khan, M.A. Sero-epidemiological study of ecto-and endo-parasites and their hematological effects in small ruminants reared under pastoral system in district Bannu, Pakistan. Pure Appl. Bio. 2015, 4, 50–56. [Google Scholar] [CrossRef]
- Khatoon, N.; Noureen, S.; Khan, Z.; Gul, S.U.; Ur, H. Domestic animals ectoparasite fauna of district Karak, KP, Pakistan. Int. J. Biosci. 2018, 13, 384–388. [Google Scholar]
- Perveen, F.; Naqvi, S.; Yasmin, N.; Bibi, N. Ixodid ticks infestation in livestock and their traditional control in NWFP, Pakistan. Pakistan J. Zool. 2010, 42, 43–54. [Google Scholar]
- Shah, A.; Shah, S.; Rafi, M.; Shah, M.; Mitra, M. Identification of the prevalent ticks (Ixodid) in goats and sheep in Peshawar, Pakistan. J. Entomol. Zool. Stud. 2015, 3, 11–14. [Google Scholar]
- Siddiqi, M.; Jan, A. Ixodid ticks (ixodidae) of NWFP (Pakistan). Pak. J. Zool. 1986, 6, 124–126. [Google Scholar]
- Khan, M. Taxonomical Study of Ticks of Genus Rhipicephalus and Their Relation to the Incidence of Haemoparasites and Comparative Efficacy of Different Acaricides on Ticks in Sheep and Goats in Kaghan Valley. Master’s Thesis, College of Veterinary Sciences, Lahore, University of Agriculture, Faisalabad, Pakistan, 1993. [Google Scholar]
- Sajid, M.S.; Iqbal, Z.; Shamim, A.; Siddique, R.M.; Hassan, M.J.U.; Rizwan, H.M. Distribution and abundance of ticks infesting livestock population along Karakorum highway from Mansehra to Gilgit, Pakistan. J. Hell. Vet. Med. Soc. 2017, 68, 51–58. [Google Scholar] [CrossRef]
- Iqbal, A.; Siddique, F.; Mahmood, M.S.; Shamim, A.; Zafar, T.; Rasheed, I.; Saleem, I.; Ahmad, W. Prevalence and impacts of ectoparasitic fauna infesting goats (Capra hircus) of district Toba Tek Singh, Punjab, Pakistan. Glob. Vet. 2014, 12, 158–164. [Google Scholar]
- Mustafa, I.; Shabbir, R.M.K.; Subhani, M.; Ahmad, I.; Aleem, R.; Jamil, S.; Muqaddas, H.; Shabbir, R.G.; Ghani, A.; Mahmood, T. Seasonal activity of tick infestation in goats and buffalo of Punjab province (district Sargodha), Pakistan. Kafkas Univ. Vet. Fak. Derg 2014, 20, 655–662. [Google Scholar]
- Ramzan, M.; Naeem-Ullah, U.; Abbas, H.; Adnan, M.; Rasheed, Z.; Khan, S. Diversity of hard ticks in goats and sheep in Multan, Punjab, Pakistan. Agric. Biol. Res. 2019, 35, 7–9. [Google Scholar]
- Sajid, M.S.; Iqbal, Z.; Khan, M.N.; Muhammad, G.; Needham, G.; Khan, M.K. Prevalence, associated determinants, and in vivo chemotherapeutic control of hard ticks (Acari: Ixodidae) infesting domestic goats (Capra hircus) of lower Punjab, Pakistan. Parasitol. Res. 2011, 108, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Riaz, M.; Nazir, M.M.; Tasawar, Z.; Ahmed, A.N.; Ayaz, M.M.; Akram, Q.; Lindsay, D.S. Molecular epidemiology and prevalence of Theileria lestoquardi and Theileria ovis infection in goats infested with tick vectors from Multan, Pakistan. J. Med. Entomol. 2019, 56, 844–848. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Hayar, C.; Iqbal, Z.; Hayat, B. Prevalence of ticks on livestock in Faisalabad (Pakistan). Pak. J. Zool. 1993, 13, 182–184. [Google Scholar]
- Durrani, H. A study on the Taxonomy and Bionomics of Genus Haemaphysalis in Domestic Animals. Master’s Thesis, College of Veterinary Sciences, Lahore, University of Agriculture, Faisalabad, Pakistan, 1992. [Google Scholar]
- Irshad, N.; Qayyum, M.; Hussain, M.; Khan, M. Prevalence of tick infestation and theileriosis in sheep and goats. Pak. J. Zool. 2010, 30, 178–180. [Google Scholar]
- Bibi, S.; Rafique, N.; Karim, A.; Taj, M.K.; Iqbal, K.; Bibi, A.; Shafiq, M.; Ghafoor, G.; Ghafoor, A.; Ijaz, A. Prevalence and taxonomic identification of hard ticks (Ixodidea) found in livestock of Harnai district, Balochistan, Pakistan. Pure Appl. Biol. 2020, 9, 2330–2338. [Google Scholar] [CrossRef]
- Haneef, M.; Kakar, A.; Naseem, M.; Kurd, A.; Rafiq, N.; Kakar, B.; Uddin, S. Incidence of ectoparasite in chiltan wild goat (Artiodactyla: Caprinae) native of Hazarganji chiltan national park (HCNP), Balochistan, Pakistan. Pure Appl. Biol. 2019, 8, 389–396. [Google Scholar] [CrossRef]
- Iqbal, A.; Nawaz, M. Taxonomic studies of Haemaphysalis flava (Neumann), its seasonal prevalence and role in parasitic diseases of sheep/goat in Balochistan. Pak. Entomol. 2007, 29, 1–4. [Google Scholar]
- Kasi, K.K.; von Arnim, F.; Schulz, A.; Rehman, A.; Chudhary, A.; Oneeb, M.; Sas, M.A.; Jamil, T.; Maksimov, P.; Sauter-Louis, C. Crimean-Congo haemorrhagic fever virus in ticks collected from livestock in Balochistan, Pakistan. Transbound. Emerg. Dis. 2020, 67, 1543–1552. [Google Scholar] [CrossRef]
- Hussain, S.I.; Kumar, G.A. The incidence of ticks (Ixodoidea: Ixodidae) infesting sheep and goat in Sind Province Pakistan. Pak. J. Zool. 1985, 17, 89–97. [Google Scholar]
- Hussain, S.; Gyanchandani, A. Prevalence of Ixodid ticks of Goats at Khairpur Mir’s and its adjoining areas. Pakistan J. Zool. 1983, 15, 51–55. [Google Scholar]
- Sayyad, B.; Mughal, S.H.; Iqbal, M.N.; Ashraf, A.; Muhammad, A. Prevalence of ectoparasites of ruminants in Muzaffarabad district, Azad Jammu and Kashmir. PSM Vet. Res. 2016, 1, 22–25. [Google Scholar]
- Sultana, N.; Awan, M.S.; Shamim, A.; Iqbal, A.; Ali, U.; Minhas, R.A.; Majid, M.M.; Bangash, N. Prevalence of ticks infesting selected domestic livestock population of Azad Jammu and Kashmir. Sch. Adv. Anim. Vet. Res. 2015, 2, 98–106. [Google Scholar]
- Sultana, N.; Shamim, A.; Awan, M.; Ali, U.; Hassan, M.; Siddique, R. First pilot study on the prevalence of tick infestation in livestock of Tehsil Hajira, Rawalakot, Azad Kashmir. Adv. Anim. Vet. Sci. 2015, 3, 430–434. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Ayllón, N.; De La Fuente, J. Impact of climate trends on tick-borne pathogen transmission. Front. Physiol. 2012, 3, 64. [Google Scholar] [CrossRef]
- Randolph, S. Tick ecology: Processes and patterns behind the epidemiological risk posed by ixodid ticks as vectors. Parasitology 2004, 129, S37–S65. [Google Scholar] [CrossRef]
- Descamps, S. Winter temperature affects the prevalence of ticks in an Arctic seabird. PLoS ONE 2013, 8, e65374. [Google Scholar] [CrossRef]
- Regasa, T.D.; KebedeTsegay, A.; Waktole, H. Prevalence of major ectoparasites of calves and associated risk factors in and around Bishoftu town. Afr. J. Agric. Res. 2015, 10, 1127–1135. [Google Scholar]
- Silatsa, B.A.; Simo, G.; Githaka, N.; Mwaura, S.; Kamga, R.M.; Oumarou, F.; Keambou, C.; Bishop, R.P.; Djikeng, A.; Kuiate, J.-R. A comprehensive survey of the prevalence and spatial distribution of ticks infesting cattle in different agro-ecological zones of Cameroon. Parasit. Vectors 2019, 12, 489. [Google Scholar] [CrossRef]
- Ghafar, A.; Gasser, R.B.; Rashid, I.; Ghafoor, A.; Jabbar, A. Exploring the prevalence and diversity of bovine ticks in five agro-ecological zones of Pakistan using phenetic and genetic tools. Ticks Tick Borne Dis. 2020, 11, 101472. [Google Scholar] [CrossRef]
- Diarra, A.Z.; Almeras, L.; Laroche, M.; Berenger, J.-M.; Koné, A.K.; Bocoum, Z.; Dabo, A.; Doumbo, O.; Raoult, D.; Parola, P. Molecular and MALDI-TOF identification of ticks and tick-associated bacteria in Mali. PLoS Negl. Trop. Dis. 2017, 11, e0005762. [Google Scholar] [CrossRef] [PubMed]
- Caporale, D.A.; Rich, S.M.; Spielman, A.; Telford, S.R.; Kocher, T.D. Discriminating between Ixodes ticks by means of mitochondrial DNA sequences. Mol. Phylogenet. Evol. 1995, 4, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Guglielmone, A.A.; Nava, S. Names for Ixodidae (Acari: Ixodoidea): Valid, synonyms, incertae sedis, nomina dubia, nomina nuda, lapsus, incorrect and suppressed names–with notes on confusions and misidentifications. Zootaxa 2014, 3767, 1–256. [Google Scholar] [CrossRef] [PubMed]
- Jinbo, U.; Kato, T.; Ito, M. Current progress in DNA barcoding and future implications for entomology. Entomol. Sci. 2011, 14, 107–124. [Google Scholar] [CrossRef]
- Walker, A.; Bouattour, A.; Camicas, J.; Estrada-Pena, A.; Horak, I.; Latif, A. Ticks of Domestic Animals in Africa. A Guide to Identification of Species; Bioscience Reports: London, UK, 2003. [Google Scholar]
- Lu, X.; Lin, X.D.; Wang, J.B.; Qin, X.C.; Tian, J.H.; Guo, W.P.; Fan, F.N.; Shao, R.; Xu, J.; Zhang, Y.Z. Molecular survey of hard ticks in endemic areas of tick-borne diseases in China. Ticks Tick Borne Dis. 2013, 4, 288–296. [Google Scholar] [CrossRef]
- Barker, S.C. Distinguishing species and populations of Rhipicephaline ticks with ITS 2 ribosomal RNA. J. Parasitol. 1998, 84, 887–892. [Google Scholar] [CrossRef]
- Zahler, M.; Filippova, N.A.; Morel, P.C.; Gothe, R.; Rinder, H. Relationships between species of the Rhipicephalus sanguineus group: A molecular approach. J. Parasitol. 1997, 83, 302–306. [Google Scholar] [CrossRef]
- Yao, H.; Song, J.; Liu, C.; Luo, K.; Han, J.; Li, Y.; Pang, X.; Xu, H.; Zhu, Y.; Xiao, P. Use of ITS2 region as the universal DNA barcode for plants and animals. PLoS ONE 2010, 5, e13102. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Wu, S.; Zhang, Y.; Chen, Y.; Feng, C.; Yuan, X.; Jia, G.; Deng, J.; Wang, C.; Wang, Q. Assessment of four DNA fragments (COI, 16S rDNA, ITS2, 12S rDNA) for species identification of the Ixodida (Acari: Ixodida). Parasit. Vectors 2014, 7, 93. [Google Scholar] [CrossRef]
- Karger, A.; Bettin, B.; Gethmann, J.M.; Klaus, C. Whole animal matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) mass spectrometry of ticks–Are spectra of Ixodes ricinus nymphs influenced by environmental, spatial, and temporal factors? PLoS ONE 2019, 14, e0210590. [Google Scholar] [CrossRef]
- Iqbal, A.; Usman, M.; Abubakar, M. Mini Review: Current tick control strategies in Pakistan are possible environmental risks. Iraqi J. Vet. Sci. 2017, 31, e81–e86. [Google Scholar] [CrossRef]
- Graf, J.-F.; Gogolewski, R.; Leach-Bing, N.; Sabatini, G.; Molento, M.; Bordin, E.; Arantes, G. Tick control: An industry point of view. Parasitology 2004, 129, S427–S442. [Google Scholar] [CrossRef] [PubMed]
- Rehman, A.; Conraths, F.J.; Sauter-Louis, C.; Krücken, J.; Nijhof, A.M. Epidemiology of tick-borne pathogens in the semi-arid and arid agro ecological zones of Punjab province, Pakistan. Transbound. Emerg. Dis. 2019, 66, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Ijaz, M.; Durrani, A.Z.; Ali, M.M.; Mehmood, K.; Sabir, A.J. Prevalence and chemotherapy of anaplasmosis in clinically affected small ruminants in the river Ravi region, Lahore. Pak. J. Zool. 2014, 46, 876–880. [Google Scholar]
- Iqbal, F.; Khattak, R.; Ozubek, S.; Khattak, M.; Rasul, A.; Aktas, M. Application of the reverse line blot assay for the molecular detection of Theileria and Babesia sp. in sheep and goat blood samples from Pakistan. Iran. J. Parasitol. 2013, 8, 289–295. [Google Scholar]
- Khan, A.; Niaz, S.; Hassan Shah, M.; Khan, A.; Ahmed, H.; Khattak, I.; Zeb, J.; Naeem, H.; Hassan, M.A.; Ulucesme, M.C.; et al. Molecular detection of small ruminant piroplasmosis and first report of Theileria luwenshuni (Apicomplexa: Theileridae) in small ruminants of Pakistan. Exp. Parasitol. 2020, 212, 107872. [Google Scholar] [CrossRef]
- Butt, M.H.; Ahmad, A.; Misbah, S.; Mallhi, T.H.; Khan, Y.H. Crimean-Congo hemorrhagic fever and Eid-Ul-Adha: A potential threat during the COVID-19 pandemic (Letter to the Editor). J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Ul Ain, Q.; Saeed, A.; Larik, E.; Ghafoor, T.; Khosa, Z.; Ali, A.; Naveed, A.; Malik, T. Evaluation of event based surveillance system of Crimean Congo haemorrhagic fever in Balochistan Pakistan, 2017. Glob. Biosecur. 2019, 1. [Google Scholar] [CrossRef]
- Atif, M.; Saqib, A.; Ikram, R.; Sarwar, M.R.; Scahill, S. The reasons why Pakistan might be at high risk of Crimean Congo haemorrhagic fever epidemic; a scoping review of the literature. Virol. J. 2017, 14, 63. [Google Scholar] [CrossRef]
- Abbas, R.Z.; Zaman, M.A.; Colwell, D.D.; Gilleard, J.; Iqbal, Z. Acaricide resistance in cattle ticks and approaches to its management: The state of play. Vet. Parasitol. 2014, 203, 6–20. [Google Scholar] [CrossRef]
- Singh, N.K.; Gelot, I.S.; Singh, V.; Rath, S.S. Detection of amitraz resistance in Rhipicephalus (Boophilus) microplus from North Gujarat, India. J. Parasit. Dis. 2015, 39, 49–52. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Thomas, D.B.; Klafke, G.; Busch, J.D.; Olafson, P.U.; Miller, R.A.; Mosqueda, J.; Stone, N.E.; Scoles, G.; Wagner, D.M.; Perez-De-Leon, A. Tracking the increase of acaricide resistance in an invasive population of cattle fever tcks (Acari: Ixodidae) and implementation of real-time PCR assays to rapidly genotype resistance mutations. Ann. Entomol. Soc. Am. 2020, 112, 298–309. [Google Scholar] [CrossRef]
- Torina, A.; Moreno-Cid, J.A.; Blanda, V.; de Mera, I.G.F.; de la Lastra, J.M.P.; Scimeca, S.; Blanda, M.; Scariano, M.E.; Briganò, S.; Disclafani, R. Control of tick infestations and pathogen prevalence in cattle and sheep farms vaccinated with the recombinant Subolesin-Major Surface Protein 1a chimeric antigen. Parasit. Vectors 2014, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, D.O.; Corona-González, B.; Rodríguez-Mallón, A.; Rodríguez Gonzalez, I.; Alfonso, P.; Noda Ramos, A.A.; Díaz-Sánchez, A.A.; González Navarrete, M.; Rodríguez Fernández, R.; Méndez Mellor, L.; et al. Ticks and Tick-Borne Diseases in Cuba, Half a Century of Scientific Research. Pathogens 2020, 9, 616. [Google Scholar] [CrossRef] [PubMed]
- Memon, M.I.; Leghari, R.A.; Kumbhar, H.K.; Korejo, N.A.; Parveen, S.; Ram, S. Study on the prevalence of anaplasmosis and efficacy of different antiprotozoal drugs in goats at district Mirpurkhas, Sindh–Pakistan. Pure Appl. Biol. 2019, 8, 1025–1032. [Google Scholar] [CrossRef]
- Ijaz, M.; Rehman, A.; Ali, M.; Umair, M.; Khalid, S.; Mehmood, K.; Hanif, A. Clinico-epidemiology and therapeutical trials on babesiosis in sheep and goats in Lahore, Pakistan. J. Anim. Plant Sci. 2013, 23, 666–669. [Google Scholar]
- Rashid, A.; Khan, J.; Khan, M.; Rasheed, K.; Maqbool, A.; Iqbal, J. Prevalence and chemotherapy of babesiosis among Lohi sheep in the Livestock Experiment Station, Qadirabad, Pakistan, and environs. J. Venom. Anim. Toxins Incl. Trop. Dis. 2010, 16, 587–591. [Google Scholar] [CrossRef]
- Dumler, J.S.; Barbet, A.F.; Bekker, C.; Dasch, G.A.; Palmer, G.H.; Ray, S.C.; Rikihisa, Y.; Rurangirwa, F.R. Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: Unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and’HGE agent’as subjective synonyms of Ehrlichia phagocytophila. Int. J. Syst. Evol. Microbiol. 2001, 51, 2145–2165. [Google Scholar]
- De la Fuente, J.; Atkinson, M.W.; Naranjo, V.; de Mera, I.G.F.; Mangold, A.J.; Keating, K.A.; Kocan, K.M. Sequence analysis of the msp4 gene of Anaplasma ovis strains. Vet. Microbiol. 2007, 119, 375–381. [Google Scholar] [CrossRef]
- Friedhoff, K. Tick-borne diseases of sheep and goats caused by Babesia, Theileria or Anaplasma spp. Parassitologia 1997, 39, 99–109. [Google Scholar]
- Yin, H.; Luo, J. Ticks of small ruminants in China. Parasitol. Res. 2007, 101, 187–189. [Google Scholar] [CrossRef] [PubMed]
- Alessandra, T.; Santo, C. Tick-borne diseases in sheep and goats: Clinical and diagnostic aspects. Small Rumin. Res. 2012, 106, S6–S11. [Google Scholar] [CrossRef]
- Ghaffar, A.; Ijaz, M.; Ali, A.; Farooqi, S.H.; Rehman, A.; Ali, M.M.; Zafar, M.Z.; Naeem, M.A. First report on molecular characterization of anaplasmosis in small ruminants in Pakistan. J. Parasitol. 2020, 106, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Anwar, K.; Din, A. Epidemiology of tick borne haemoprotozoan infection in ruminants in district Peshawar, and periphery, Khyber Pakhtunkhwa,(Pakistan). Am. Sci. Res. J. Eng. Technol. Sci. 2017, 35, 191–200. [Google Scholar]
- Kashif, M.; Ahmad, M. Geographical seroprevalence of Anaplasma marginale infection (anaplasmosis) by ELISA in Ovis aries, in district Peshawar, Pakistan. J. Zool. Stud. 2014, 1, 15–18. [Google Scholar]
- Khan, A.; Mitchell, R.D., III; Niaz, S.; Ayaz, S.; Khattak, I.; Naeem, H.; de León, A.A.P.; Zaman, M.A. Seroprevalence of Anaplasma spp. among sheep and goats in Charsadda District, Pakistan. Small Rumin. Res. 2019, 176, 5–10. [Google Scholar] [CrossRef]
- Talat, R.; Khanum, T.; Hayat, A. Studies on mammalian haematozoan parasites of NWFP Pakistan. Pak. J. Biol. Sci. 2005, 8, 726–729. [Google Scholar]
- Nasreen, K.S.; Khan, A.; Niaz, S.; Akhtar, N. Serodiagnosis and haematological effect of anaplasmosis in goats and sheep of district Mardan, Khyber Pakhtunkhwa, Pakistan. World J. Zool. 2016, 11, 67–80. [Google Scholar]
- Khan, A.; Khan, M.R.; Tufail, M.; Saleem, G.; Masood, S. Pathogenesis of anaplasmosis in Bulkhi sheep and their confirmation with polymerase chain reaction. Sch. Adv. Anim. Vet. Res. 2015, 2, 107–114. [Google Scholar]
- Renneker, S.; Abdo, J.; Salih, D.; Karagenç, T.; Bilgiç, H.; Torina, A.; Oliva, A.; Campos, J.; Kullmann, B.; Ahmed, J. Can Anaplasma ovis in small ruminants be neglected any longer? Transbound. Emerg. Dis. 2013, 60, 105–112. [Google Scholar] [CrossRef]
- Ghafar, A.; McGill, D.; Stevenson, M.A.; Badar, M.; Kumbher, A.; Warriach, H.M.; Gasser, R.B.; Jabbar, A. A participatory investigation of bovine health and production issues in Pakistan. Front. Vet. Sci. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Chochlakis, D.; Ioannou, I.; Tselentis, Y.; Psaroulaki, A. Human anaplasmosis and Anaplasma ovis variant. Emerg. Infect. Dis. 2010, 16, 1031–1032. [Google Scholar] [CrossRef] [PubMed]
- Homer, M.J.; Aguilar-Delfin, I.; Telford, S.R.; Krause, P.J.; Persing, D.H. Babesiosis. Clin. Microbiol. Rev. 2000, 13, 451–469. [Google Scholar] [CrossRef] [PubMed]
- Uilenberg, G.; Rombach, M.; Perié, N.; Zwart, D. Blood parasites of sheep in the Netherlands. II. Babesia motasi (Sporozoa, Babesiidae). Vet. Q. 1980, 2, 3–14. [Google Scholar]
- Iqbal, F.; Fatima, M.; Shahnawaz, S.; Naeem, M.; Shaikh, R.; Ali, M.; Shaikh, A.; Aktas, M.; Ali, M. A study on the determination of risk factors associated with babesiosis and prevalence of Babesia sp., by PCR amplification, in small ruminants from Southern Punjab (Pakistan). Parasite 2011, 18, 229–234. [Google Scholar] [CrossRef]
- Ghafar, A.; Cabezas-Cruz, A.; Galon, C.; Obregon, D.; Gasser, R.B.; Moutailler, S.; Jabbar, A. Bovine ticks harbour a diverse array of microorganisms in Pakistan. Parasit. Vectors 2020, 13, 1. [Google Scholar] [CrossRef]
- Yeruham, I.; Handani, A.; Galker, F.; Rosen, S.; Schlien, J. A field study of haemoparasites in two flocks of sheep in Israel. Isr J. Vet. Med. 1992, 47, 107–111. [Google Scholar]
- Ahmed, J.; Yin, H.; Bakheit, M.; Liu, Z.; Mehlhorn, H.; Seitzer, U. Small ruminant theileriosis. In Progress in Parasitology; Mehlhorn, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 135–153. [Google Scholar]
- Schnittger, L.; Yin, H.; Gubbels, M.J.; Beyer, D.; Niemann, S.; Jongejan, F.; Ahmed, J.S. Phylogeny of sheep and goat Theileria and Babesia parasites. Parasitol. Res. 2003, 91, 398–406. [Google Scholar]
- Naz, S.; Maqbool, A.; Ahmed, S.; Ashraf, K.; Ahmed, N.; Saeed, K.; Latif, M.; Iqbal, J.; Ali, Z.; Shafi, K. Prevalence of theileriosis in small ruminants in Lahore-Pakistan. J. Vet. Ani. Sci. 2012, 2, 16–20. [Google Scholar]
- Yaghfoori, S.; Razmi, G.R.; Mohri, M.; Razavizadeh, A.R.T.; Movassaghi, A.R. An experimental ovine Theileriosis: The effect of Theileria lestoquardi infection on cardiovascular system in sheep. Acta Trop. 2016, 161, 55–61. [Google Scholar] [CrossRef]
- Fatima, M.; Saeed, S.; Shaikh, R.S.; Ali, M.; Iqbal, F. A study on molecular detection of Theileria lestoquardi by PCR amplification in apparently healthy small ruminants from five districts of Southern Punjab. Pak. J. Zool. 2015, 47, 441–446. [Google Scholar]
- Riaz, M.; Tasawar, Z. Identification of Theileria species (Theileria ovis and Theileria lestoquardi) by PCR in apparently healthy small ruminants in and around Multan, Southern Punjab, Pakistan. J. Anim. Plant Sci. 2017, 27, 809–818. [Google Scholar]
- Riaz, M.; Tasawar, Z.; Ullah, M.Z. A study on molecular prevalence, intensity and associated risk factors for ovine and caprine theileriosis from Southern Punjab, Pakistan. Pak. J. Life Soc. Sci. 2017, 15, 150–157. [Google Scholar]
- Riaz, M.; Tasawar, Z. A study on molecular surveillance of Theileria spp. infection and its impact on hematological and biochemical changes in naturally infected small ruminants at Multan, Pakistan. Pure Appl. Biol. 2017, 6, 1427–1435. [Google Scholar] [CrossRef]
- Zia-ur-Rehman; Khan, M.S.; Avais, M.; Aleem, M.; Shabbir, M.Z.; Khan, J.A. Prevalence of theileriosis in sheep in Okara district, Pakistan. Pakistan J. Zool. 2010, 42, 639–643. [Google Scholar]
- Durrani, S.; Khan, Z.; Khattak, R.M.; Andleeb, M.; Ali, M.; Hameed, H.; Taqddas, A.; Faryal, M.; Kiran, S.; Anwar, H. A comparison of the presence of Theileria ovis by PCR amplification of their SSU rRNA gene in small ruminants from two provinces of Pakistan. Asian Pac. J. Trop. Dis. 2012, 2, 43–47. [Google Scholar] [CrossRef]
- Saeed, S.; Jahangir, M.; Fatima, M.; Shaikh, R.; Khattak, R.; Ali, M.; Iqbal, F. PCR based detection of Theileria lestoquardi in apparently healthy sheep and goats from two districts in Khyber Pukhtoon Khwa (Pakistan). Trop. Biomed. 2015, 32, 225–232. [Google Scholar]
- Shah, S.; Khan, M.; Rahman, H. Epidemiological and hematological investigations of tick-borne diseases in small ruminants in Peshawar and Khyber agency. Pakistan J. Adv. Parasitol. 2017, 4, 15–22. [Google Scholar]
- Ullah, N.; Durrani, A.Z.; Avais, M.; Nisar, A.; Ullah, S.; Khan, M.S.; Mehmood, K.; Khan, M.A.; Haq, I. Prevalence, risk factors and host biomarkers of ovine theileriosis. Pak. J. Zool. 2018, 50, 1211–1216. [Google Scholar] [CrossRef]
- Khan, M.A.; Khan, M.; Ahmad, I.; Khan, M.; Anjum, A.; Durrani, A.; Hameed, K.; Kakar, I.; Wajid, A.; Ramazan, M. Risk factors assessment and molecular characterization of Theileria in small ruminants of Balochistan. J. Anim. Plant Sci. 2017, 27, 1190–1196. [Google Scholar]
- Ahmed, I.P. A serological investigation of Q fever in Pakistan. J. Pak. Med. Assoc. 1987, 37, 126–129. [Google Scholar] [PubMed]
- Shabbir, M.Z.; Akram, S.; Hassan, Z.; Hanif, K.; Rabbani, M.; Muhammad, J. Evidence of Coxiella burnetii in Punjab province, Pakistan. Acta Trop. 2016, 163, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Georgiev, M.; Afonso, A.; Neubauer, H.; Needham, H.; Thiery, R.; Rodolakis, A.; Roest, H.; Stärk, K.; Stegeman, J.; Vellema, P. Q fever in humans and farm animals in four European countries, 1982 to 2010. Euro Surveill. 2013, 18, 20407. [Google Scholar] [PubMed]
- Garrison, A.R.; Smith, D.R.; Golden, J.W. Animal models for Crimean-Congo hemorrhagic fever human disease. Viruses 2019, 11, 590. [Google Scholar] [CrossRef] [PubMed]
- Marcondes, C.B. Arthropod Borne Diseases; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Lew-Tabor, A.E. Anaplasmosis. In The Merck Veterinary Manual; Merck & Co., Inc.: Whitehouse Station, NJ, USA, 2016. [Google Scholar]
- Lempereur, L.; Beck, R.; Fonseca, I.; Marques, C.; Duarte, A.; Santos, M.; Zúquete, S.; Gomes, J.; Walder, G.; Domingos, A. Guidelines for the detection of Babesia and Theileria parasites. Vector Borne Zoonotic Dis. 2017, 17, 51–65. [Google Scholar] [CrossRef]
- Shabana, I.; Alhadlag, N.; Zaraket, H. Diagnostic tools of caprine and ovine anaplasmosis: A direct comparative study. BMC Vet. Res. 2018, 14, 165. [Google Scholar] [CrossRef]
- Snodgrass, D.; Ramachandran, S. A complement fixation test for tick-borne fever of sheep. Br. Vet. J. 1971, 127, xliv–xlvi. [Google Scholar] [CrossRef]
- Igarashi, I.; Parrodi, F. Bovine Babesiosis. In OIE Terrestrial Manual; World Organization for Animal Health: Paris, France, 2014. [Google Scholar]
- Mans, B.J.; Pienaar, R.; Latif, A.A. A review of Theileria diagnostics and epidemiology. Int. J. Parasitol. Parasites Wildl. 2015, 4, 104–118. [Google Scholar] [CrossRef]
- Wang, J.; Liu, A.; Zhang, S.; Gao, S.; Rashid, M.; Li, Y.; Liu, J.; Ma, Q.; Li, Z.; Liu, Z. High resolution melting analysis of the 18S rRNA gene for the rapid diagnosis of bovine babesiosis. Parasit. Vectors 2019, 12, 523. [Google Scholar] [CrossRef]
- Criado-Fornelio, A.; Buling, A.; Asenzo, G.; Benitez, D.; Florin-Christensen, M.; Gonzalez-Oliva, A.; Henriques, G.; Silva, M.; Alongi, A.; Agnone, A. Development of fluorogenic probe-based PCR assays for the detection and quantification of bovine piroplasmids. Vet. Parasitol. 2009, 162, 200–206. [Google Scholar] [CrossRef]
- Michelet, L.; Delannoy, S.; Devillers, E.; Umhang, G.; Aspan, A.; Juremalm, M.; Chirico, J.; van der Wal, F.J.; Sprong, H.; Boye Pihl, T.P. High-throughput screening of tick-borne pathogens in Europe. Front. Cell. Infect. Microbiol. 2014, 4, 103. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, J.M.; Nickerson, D.A.; Reiner, A.P. Massively parallel sequencing: The new frontier of hematologic genomics. Blood 2013, 122, 3268–3275. [Google Scholar] [CrossRef] [PubMed]
- Schnittger, L.; Yin, H.; Qi, B.; Gubbels, M.J.; Beyer, D.; Niemann, S.; Jongejan, F.; Ahmed, J.S. Simultaneous detection and differentiation of Theileria and Babesia parasites infecting small ruminants by reverse line blotting. Parasitol. Res. 2004, 92, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Afzal, M. Improving veterinary service in Pakistan. Pak. Vet. J. 2009, 29, 206–210. [Google Scholar]
- Mosqueda, J.; Olvera-Ramirez, A.; Aguilar-Tipacamu, G.J.; Canto, G. Current advances in detection and treatment of babesiosis. Curr. Med. Chem. 2012, 19, 1504–1518. [Google Scholar] [CrossRef]
- Notomi, T.; Okayama, H.; Masubuchi, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000, 28, e63. [Google Scholar] [CrossRef]
- Mori, Y.; Kanda, H.; Notomi, T. Loop-mediated isothermal amplification (LAMP): Recent progress in research and development. J. Infect. Chemother. 2013, 19, 404–411. [Google Scholar] [CrossRef]
- Yang, J.; Guan, G.; Niu, Q.; Liu, Z.; Li, Y.; Liu, J.; Ma, M.; Ren, Q.; Liu, A.; Luo, J. Development and application of a Loop-mediated Isothermal Amplification Assay for rapid detection of Borrelia burgdorferi sl in ticks. Transbound. Emerg. Dis. 2013, 60, 238–244. [Google Scholar] [CrossRef]
- Njiru, Z.K. Loop-mediated isothermal amplification technology: Towards point of care diagnostics. PLoS Negl. Trop. Dis. 2012, 6, e1572. [Google Scholar] [CrossRef]
- Reboud, J.; Xu, G.; Garrett, A.; Adriko, M.; Yang, Z.; Tukahebwa, E.M.; Rowell, C.; Cooper, J.M. Paper-based microfluidics for DNA diagnostics of malaria in low resource underserved rural communities. Proc. Natl. Acad. Sci. USA 2019, 116, 4834–4842. [Google Scholar] [CrossRef]
- Ganguli, A.; Ornob, A.; Yu, H.; Damhorst, G.; Chen, W.; Sun, F.; Bhuiya, A.; Cunningham, B.; Bashir, R. Hands-free smartphone-based diagnostics for simultaneous detection of Zika, Chikungunya, and Dengue at point-of-care. Biomed. Microdevices 2017, 19, 73. [Google Scholar] [CrossRef] [PubMed]
- Ong, D.S.; Poljak, M. Smartphones as mobile microbiological laboratories. Clin. Microbiol. Infect. 2020, 26, 421–424. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, N.; Mukhtar, M.U.; Yang, J.; Sajid, M.S.; Niu, Q.; Guan, G.; Liu, Z.; Yin, H. First molecular evidence of Anaplasma bovis and Anaplasma phagocytophilum in bovine from central Punjab, Pakistan. Pathogens 2019, 8, 155. [Google Scholar] [CrossRef] [PubMed]
- Zeb, J.; Shams, S.; Din, I.U.; Ayaz, S.; Khan, A.; Nasreen, N.; Khan, H.; Khan, M.A.; Senbill, H. Molecular epidemiology and associated risk factors of Anaplasma marginale and Theileria annulata in cattle from North-western Pakistan. Vet. Parasitol. 2020, 279, 109044. [Google Scholar] [CrossRef]
- Hassan, M.A.; Liu, J.; Sajid, M.S.; Mahmood, A.; Zhao, S.; Abbas, Q.; Guan, G.; Yin, H.; Luo, J. Molecular detection of Theileria annulata in cattle from different regions of Punjab, Pakistan, by using recombinase polymerase amplification and polymerase chain reaction. J. Parasitol. 2018, 104, 196–201. [Google Scholar] [CrossRef]
- Nyangiwe, N.; Horak, I.G. Goats as alternative hosts of cattle ticks. Onderstepoort J. Vet. Res. 2007, 74, 1–7. [Google Scholar] [CrossRef]
- Jongejan, F.; Berger, L.; Busser, S.; Deetman, I.; Jochems, M.; Leenders, T.; De Sitter, B.; Van der Steen, F.; Wentzel, J.; Stoltsz, H. Amblyomma hebraeum is the predominant tick species on goats in the Mnisi community area of Mpumalanga province South Africa and is co-infected with Ehrlichia ruminantium and Rickettsia africae. Parasit. Vectors 2020, 13, 172. [Google Scholar] [CrossRef]
- Ogden, N.H.; Lindsay, L.R. Effects of climate and climate change on vectors and vector-borne diseases: Ticks are different. Trends Parasitol. 2016, 32, 646–656. [Google Scholar] [CrossRef]
- Ostfeld, R.S.; Brunner, J.L. Climate change and Ixodes tick-borne diseases of humans. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20140051. [Google Scholar] [CrossRef]
- Gray, J.S.; Dautel, H.; Estrada-Pena, A.; Kahl, O.; Lindgren, E. Effects of climate change on ticks and tick-borne diseases in europe. Interdiscip. Perspect. Infect. Dis. 2009, 2009, 593232. [Google Scholar] [CrossRef]
- Ullah, W.; Nafees, M.; Khurshid, M.; Nihei, T. Assessing farmers’ perspectives on climate change for effective farm-level adaptation measures in Khyber. Environ. Monit. Assess. 2019, 191, 547. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Butt, A.R.; Uzma, F.; Ahmed, R.; Irshad, S.; Rehman, A.; Yousaf, B. A comprehensive review of climate change impacts, adaptation, and mitigation on environmental and natural calamities in Pakistan. Environ. Monit. Assess. 2020, 192, 48. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.A.; Jabbar, A. “Smart diagnosis” of parasitic diseases by use of smartphones. J. Clin. Microbiol. 2018, 56, e01469-17. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghafar, A.; Abbas, T.; Rehman, A.; Sandhu, Z.-u.-D.; Cabezas-Cruz, A.; Jabbar, A. Systematic Review of Ticks and Tick-Borne Pathogens of Small Ruminants in Pakistan. Pathogens 2020, 9, 937. https://doi.org/10.3390/pathogens9110937
Ghafar A, Abbas T, Rehman A, Sandhu Z-u-D, Cabezas-Cruz A, Jabbar A. Systematic Review of Ticks and Tick-Borne Pathogens of Small Ruminants in Pakistan. Pathogens. 2020; 9(11):937. https://doi.org/10.3390/pathogens9110937
Chicago/Turabian StyleGhafar, Abdul, Tariq Abbas, Abdul Rehman, Zia-ud-Din Sandhu, Alejandro Cabezas-Cruz, and Abdul Jabbar. 2020. "Systematic Review of Ticks and Tick-Borne Pathogens of Small Ruminants in Pakistan" Pathogens 9, no. 11: 937. https://doi.org/10.3390/pathogens9110937
APA StyleGhafar, A., Abbas, T., Rehman, A., Sandhu, Z.-u.-D., Cabezas-Cruz, A., & Jabbar, A. (2020). Systematic Review of Ticks and Tick-Borne Pathogens of Small Ruminants in Pakistan. Pathogens, 9(11), 937. https://doi.org/10.3390/pathogens9110937










