In Vitro Activity of Sertraline, an Antidepressant, Against Antibiotic-Susceptible and Antibiotic-Resistant Helicobacter pylori Strains
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Conditions
2.2. Disk-Diffusion Method
2.3. MIC/MBC Determination
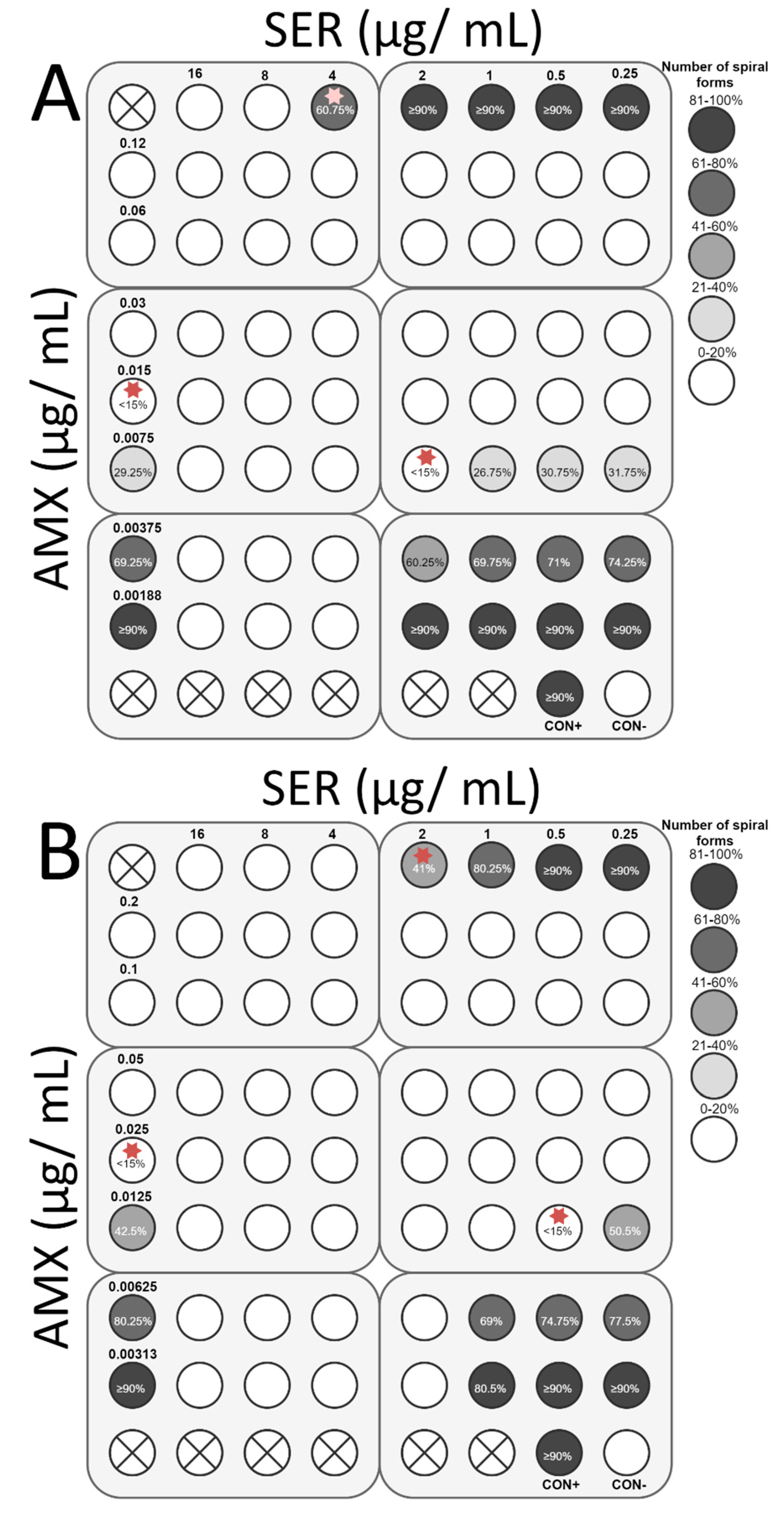
2.4. Checkerboard Assay
2.5. Time-Killing Assay
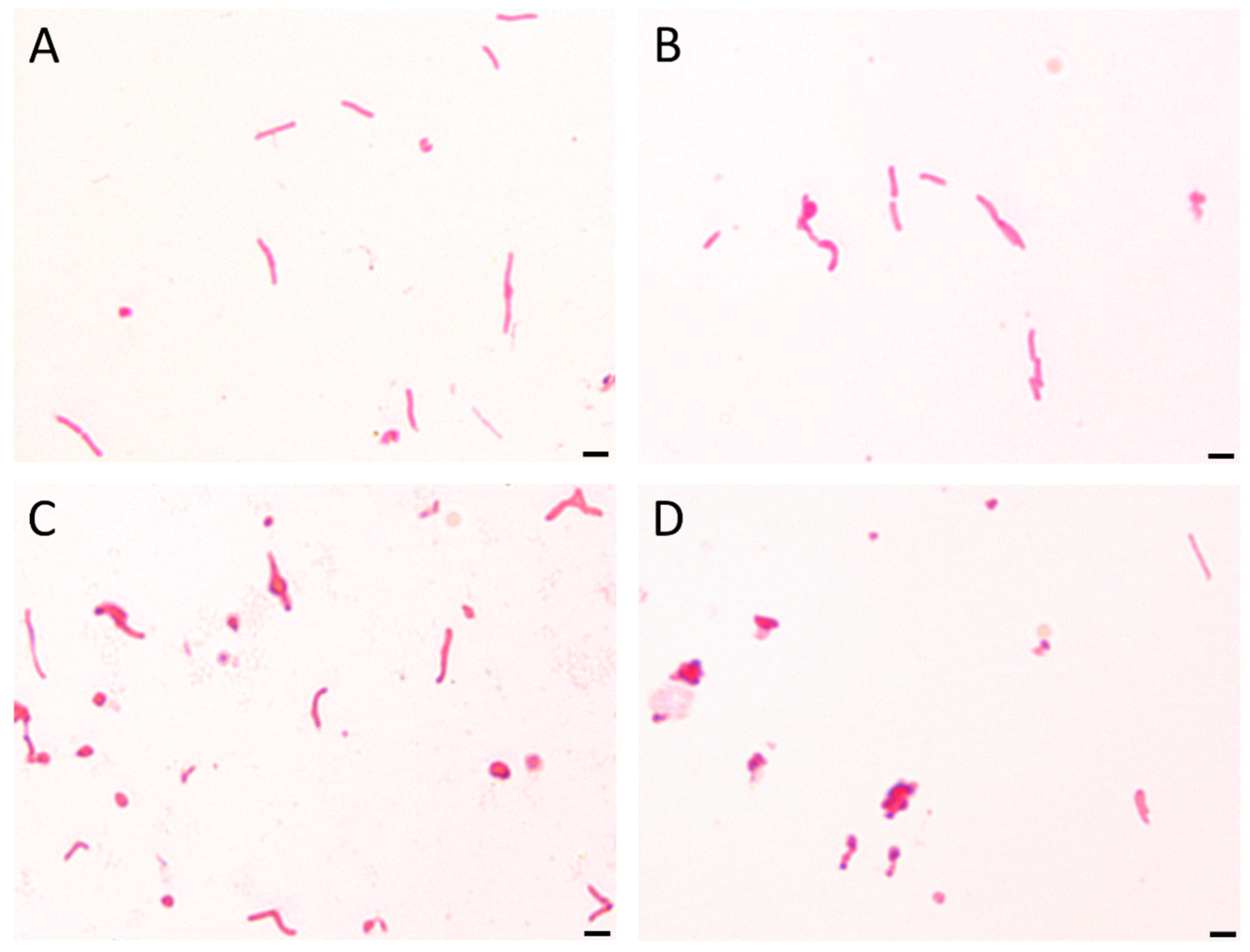
2.6. Light Microscopy
2.7. Fluorescence Microscopy
2.8. Scanning Electron Microscopy
2.9. Statistical Analysis
3. Results
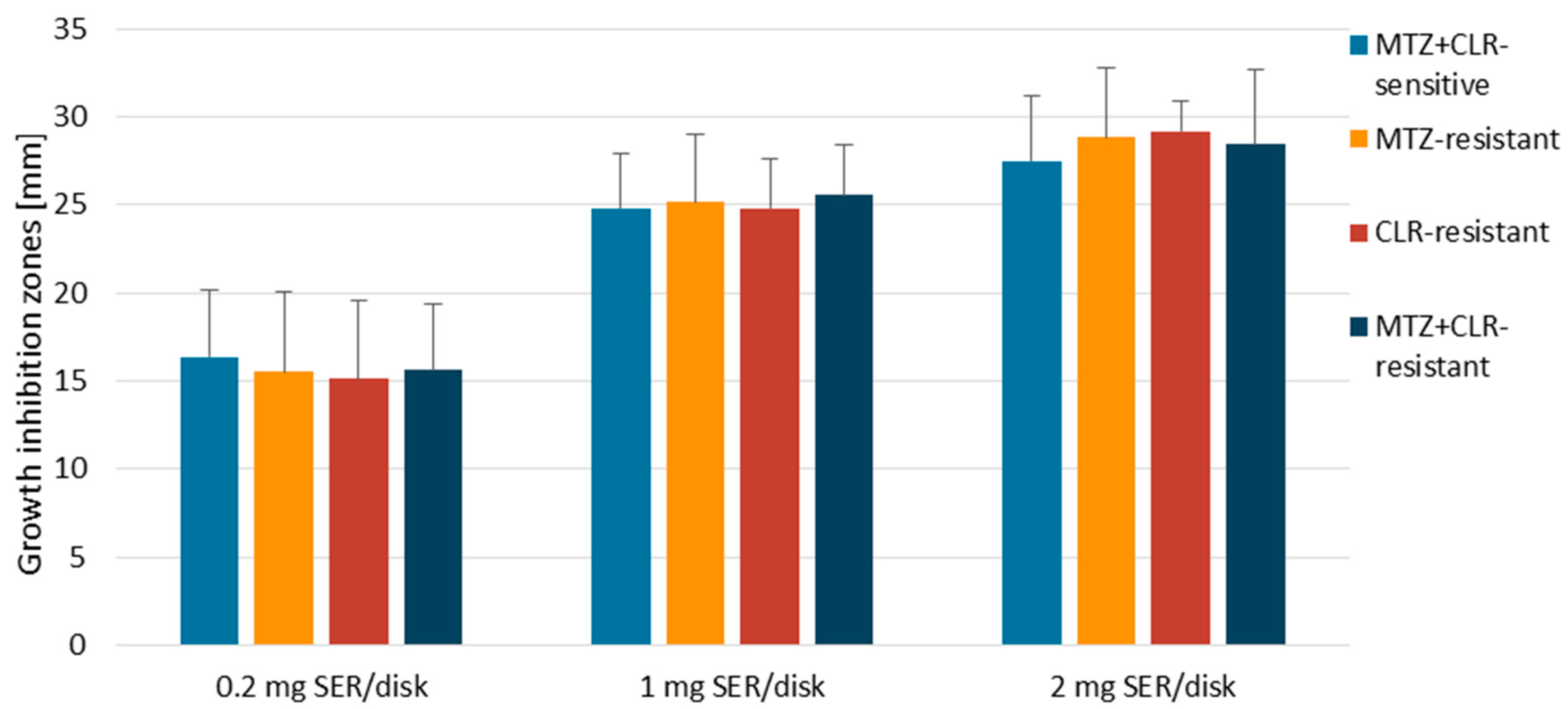
3.1. Disk-Diffusion Method
3.2. Determination of MICs and MBCs
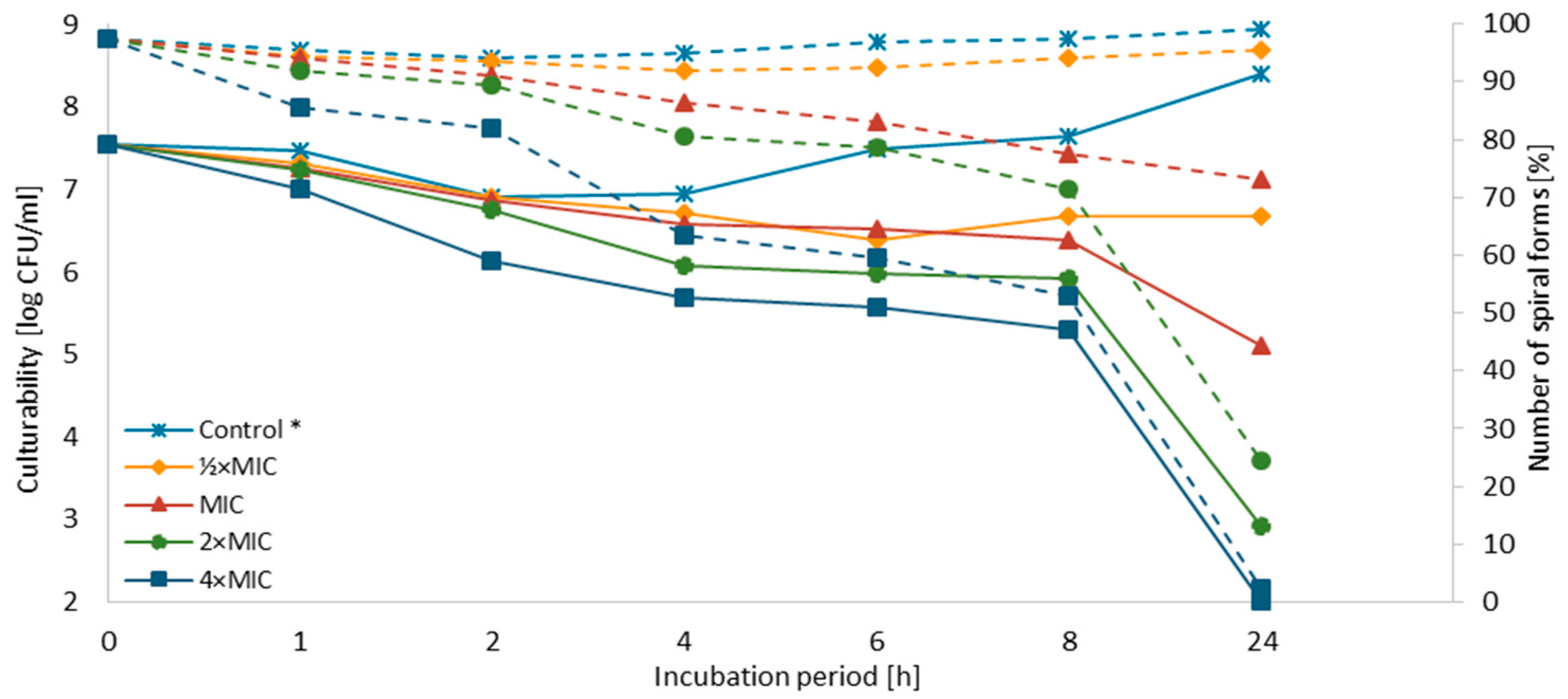
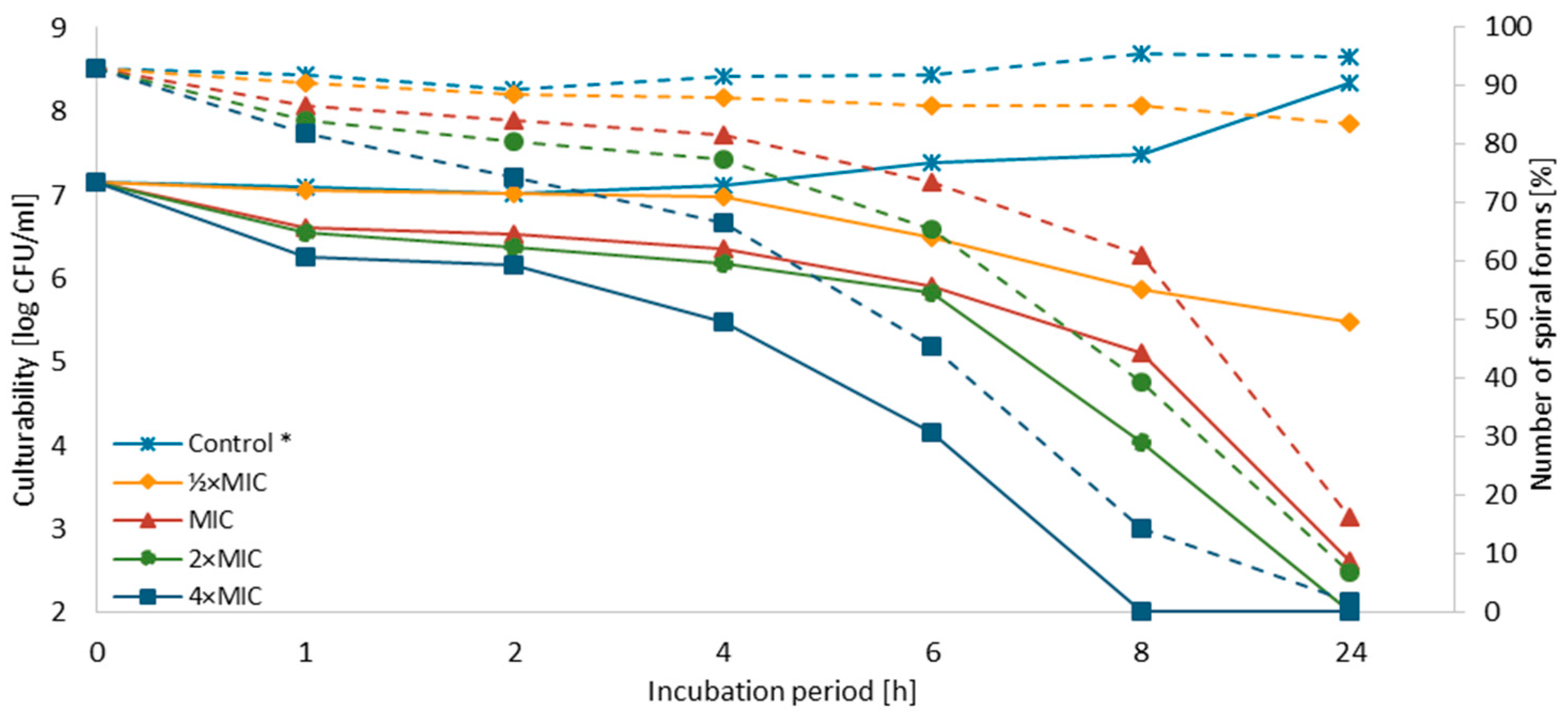
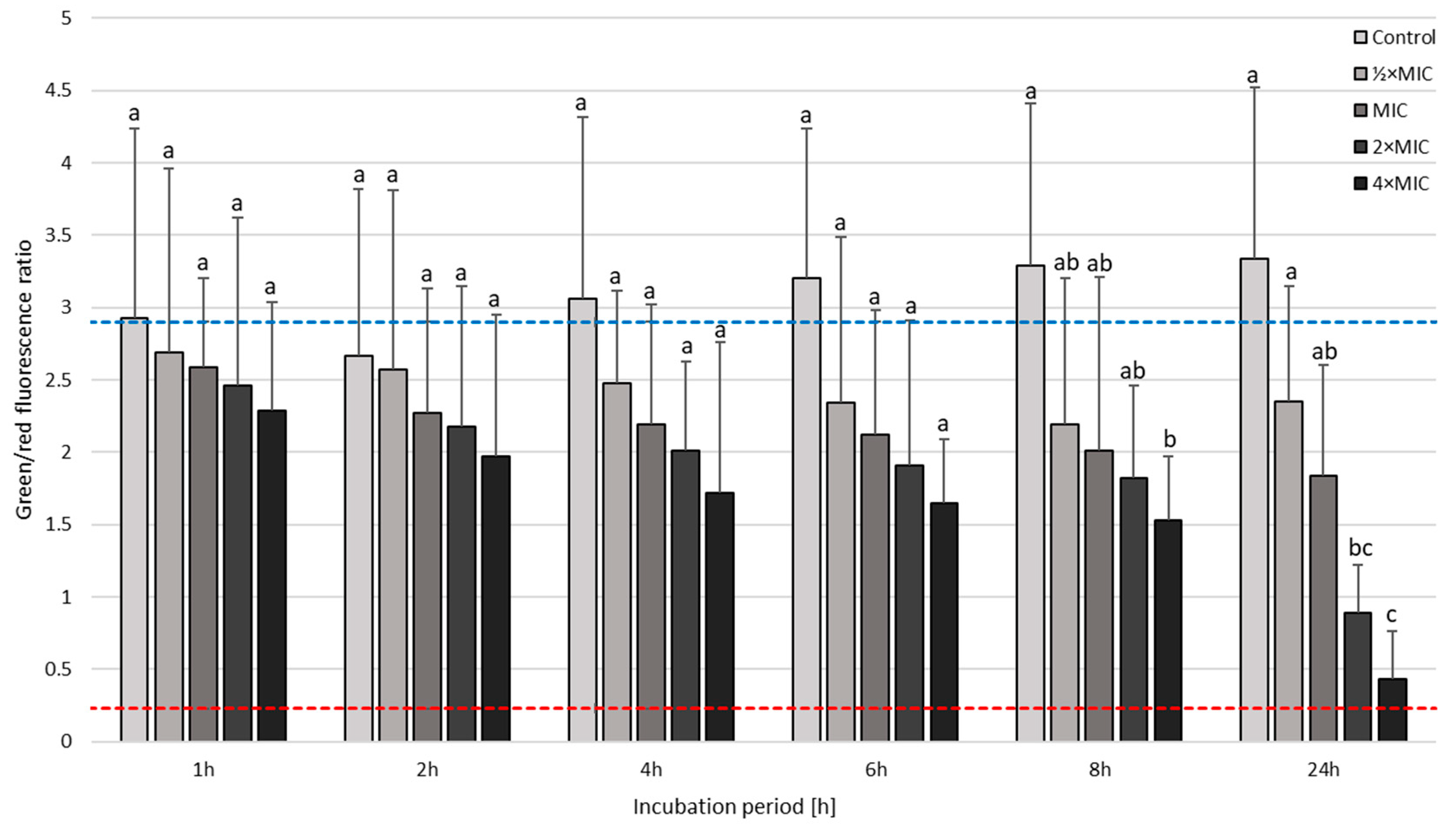
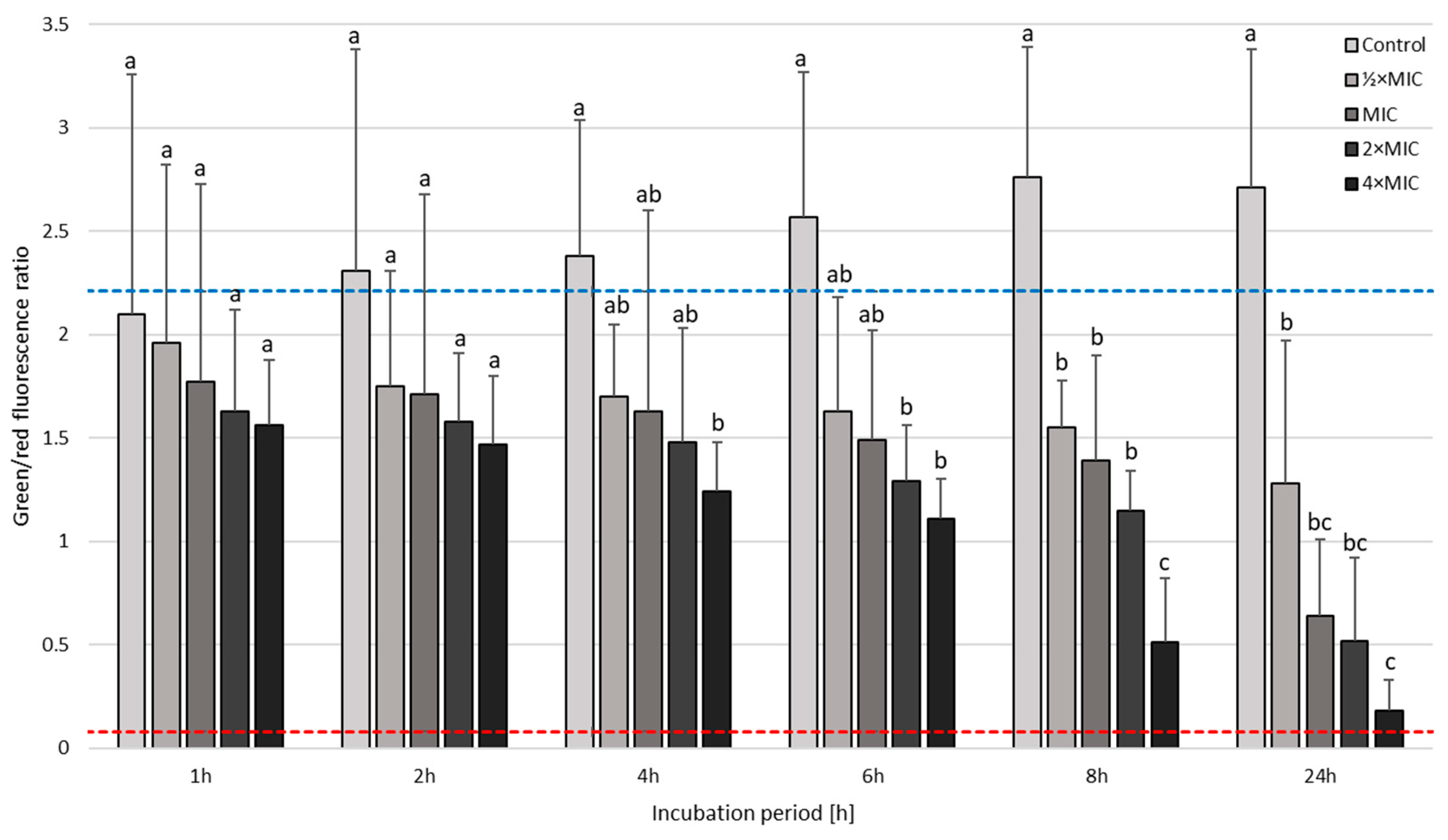
3.3. Time-Killing Assay
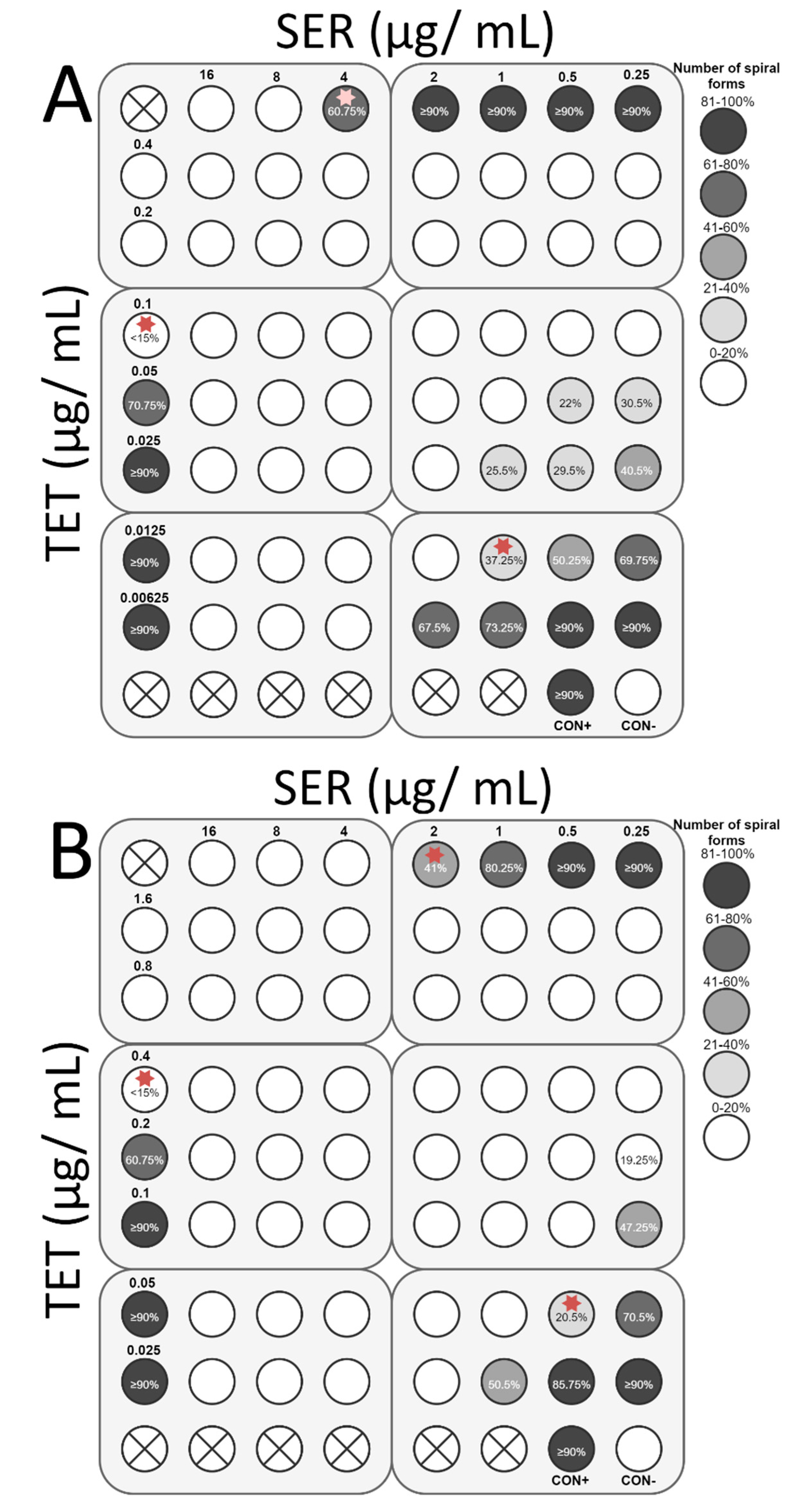
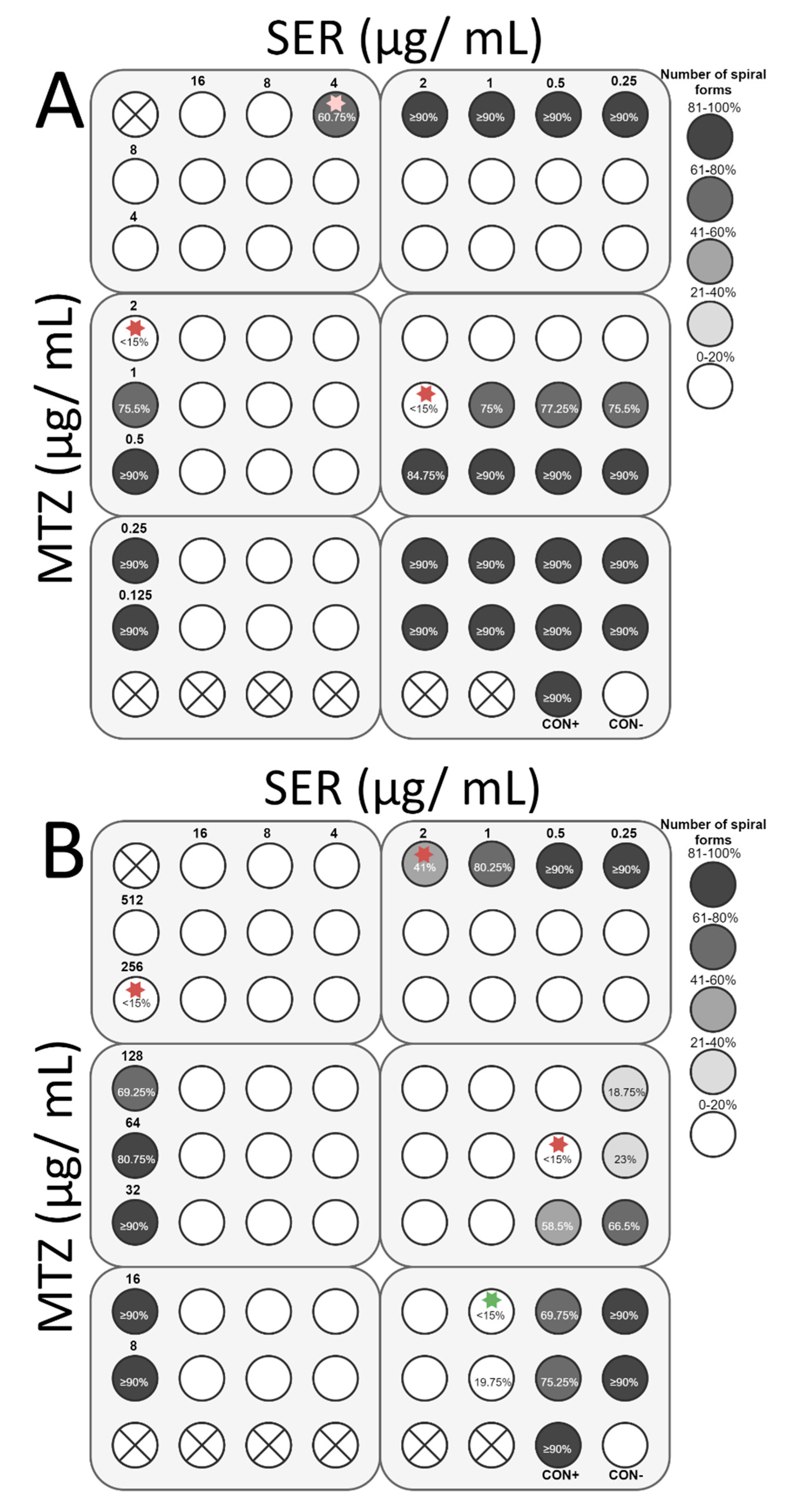
3.4. Checkerboard Assay
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Konturek, J.W. Discovery by Jaworski of Helicobacter pylori and Its Pathogenetic Role in Peptic Ulcer, Gastritis and Gastric Cancer. J. Physiol. Pharmacol. 2003, 54, 23–41. [Google Scholar] [PubMed]
- Lang, L. Barry Marshall 2005 Nobel Laureate in Medicine and Physiology. Gastroenterology 2005, 129, 1813–1814. [Google Scholar] [CrossRef] [PubMed]
- Dunne, C.; Dolan, B.; Clyne, M. Factors That Mediate Colonization of the Human Stomach by Helicobacter pylori. World J. Gastroenterol. 2014, 20, 5610–5624. [Google Scholar] [CrossRef] [PubMed]
- Coelho, L.G.V.; Marinho, J.R.; Genta, R.; Ribeiro, L.T.; Passos, M.D.C.F.; Zaterka, S.; Assumpção, P.P.; Barbosa, A.J.A.; Barbuti, R.; Braga, L.L.; et al. IVth Brazilian Consensus Conference on Helicobacter pylori Infection. Arq. Gastroenterol. 2018, 55, 97–121. [Google Scholar] [CrossRef] [PubMed]
- Sugano, K.; Tack, J.; Kuipers, E.J.; Graham, D.Y.; El-Omar, E.M.; Miura, S.; Haruma, K.; Asaka, M.; Uemura, N.; Malfertheiner, P.; et al. Kyoto Global Consensus Report on Helicobacter pylori Gastritis. Gut 2015, 64, 1353–1367. [Google Scholar] [CrossRef]
- Malfertheiner, P.; Megraud, F.; O’Morain, C.A.; Gisbert, J.P.; Kuipers, E.J.; Axon, A.T.; Bazzoli, F.; Gasbarrini, A.; Atherton, J.; Graham, D.Y.; et al. Management of Helicobacter pylori Infection—The Maastricht V/Florence Consensus Report. Gut 2017, 66, 6–30. [Google Scholar] [CrossRef]
- Fallone, C.A.; Moss, S.F.; Malfertheiner, P. Reconciliation of Recent Helicobacter pylori Treatment Guidelines in a Time of Increasing Resistance to Antibiotics. Gastroenterology 2019, 157, 44–53. [Google Scholar] [CrossRef]
- Savoldi, A.; Carrara, E.; Graham, D.Y.; Conti, M.; Tacconelli, E. Prevalence of Antibiotic Resistance in Helicobacter pylori: A Systematic Review and Meta-analysis in World Health Organization Regions. Gastroenterology 2018, 155, 1372–1382. [Google Scholar] [CrossRef]
- Ayala, G.; Escobedo-Hinojosa, W.I.; de la Cruz-Herrera, C.F.; Romero, I. Exploring Alternative Treatments for Helicobacter pylori Infection. World J. Gastroenterol. 2014, 20, 1450–1469. [Google Scholar] [CrossRef]
- O’Connor, A.; Liou, J.; Gisbert, J.P.; O’Morain, C. Review: Treatment of Helicobacter pylori Infection 2019. Helicobacter 2019, 24, e12640. [Google Scholar] [CrossRef]
- Pellicano, R.; Zagari, R.M.; Zhang, S.; Saracco, G.M.; Moss, S.F. Pharmacological Considerations and Step-by-Step Proposal for the Treatment of Helicobacter pylori Infection in the Year 2018. Minerva Gastroenterol. Dietol. 2018, 64, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Bellido, J.L.; Munoz-Criado, S.; Garcìa-Rodrìguez, J.A. Antimicrobial Activity of Psychotropic Drugs: Selective Serotonin Reuptake Inhibitors. Int. J. Antimicrob. Agents 2000, 14, 177–180. [Google Scholar] [CrossRef]
- Jakobsen, J.C.; Katakam, K.K.; Schou, A.; Hellmuth, S.G.; Stallknecht, S.E.; Leth-Møller, K.; Iversen, M.; Banke, M.B.; Petersen, I.J.; Klingenberg, S.L.; et al. Selective Serotonin Reuptake Inhibitors Versus Placebo in Patients with Major Depressive Disorder. A Systematic Review with Meta-Analysis and Trial Sequential Analysis. BMC Psychiatry 2017, 17, 58. [Google Scholar]
- Valuck, R. Selective Serotonin Reuptake Inhibitors: A Class Review. Pharm. Ther. 2004, 29, 234–243. [Google Scholar]
- Cong, L.; Liao, Y.; Yang, S.; Yang, R. In Vitro Antifungal Activity of Sertraline and Synergistic Effects in Combination with Antifungal Drugs against Planktonic Forms and Biofilms of Clinical Trichosporon asahii Isolates. PLoS ONE 2016, 11, e0167903. [Google Scholar] [CrossRef]
- Gowri, M.; Jayashree, B.; Jeyakanthan, J.; Girija, E.K. Sertraline as a Promising Antifungal Agent: Inhibition of Growth and Biofilm of Candida auris with Special Focus on the Mechanism of Action In Vitro. J. Appl. Microbiol. 2019. [Google Scholar] [CrossRef]
- Zhai, B.; Wu, C.; Wang, L.; Sachs, M.S.; Lin, X. The Antidepressant Sertraline Provides a Promising Therapeutic Option for Neurotropic Cryptococcal Infections. Antimicrob. Agents Chemother. 2012, 56, 3758–3766. [Google Scholar] [CrossRef] [Green Version]
- Rossato, L.; Loreto, É.S.; Zanette, R.A.; Chassot, F.; Santurio, J.M.; Alves, S.H. In Vitro Synergistic Effects of Chlorpromazine and Sertraline in Combination with Amphotericin B against Cryptococcus neoformans var. grubii. Folia Microbiol. 2016, 61, 399–403. [Google Scholar] [CrossRef]
- Nayak, R.; Xu, J. Effects of Sertraline Hydrochloride and Fluconazole Combinations on Cryptococcus neoformans and Cryptococcus gattii. Mycology 2010, 1, 99–105. [Google Scholar] [CrossRef]
- Oliveira, A.S.; Martinez-de-Oliveira, J.; Palmeira-de-Oliveira, R.; Palmeira-de-Oliveira, A. Antifungal Activity of Antidepressant Sertraline against Candida Species In Vitro: A Potential Beneficial Association with Fluconazole. Med. J. Obstet. Gynecol. 2017, 5, 1095. [Google Scholar]
- Treviño-Rangel, R.D.J.; Villanueva-Lozano, H.; Méndez-Galomo, K.S.; Solís-Villegas, E.M.; Becerril-García, M.A.; Montoya, A.M.; Robledo-Leal, E.R.; González, G.M. In Vivo Evaluation of the Antifungal Activity of Sertraline against Aspergillus fumigatus. J. Antimicrob. Chemother. 2019, 74, 663–666. [Google Scholar] [CrossRef] [PubMed]
- Treviño-Rangel, R.D.J.; Villanueva-Lozano, H.; Hernández-Rodríguez, P.; Martínez-Reséndez, M.F.; García-Juárez, J.; Rodríguez-Rocha, H.; González, G.M. Activity of Sertraline against Cryptococcus neoformans: In Vitro and In Vivo Assays. Med. Mycol. 2016, 54, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Villanueva-Lozano, H.; Treviño-Rangel, R.D.J.; Téllez-Marroquín, R.; Bonifaz, A.; Rojas, O.C.; Hernández-Rodríguez, P.A.; González, G.M. In Vitro Inhibitory Activity of Sertraline against Clinical Isolates of Sporothrix schenckii. Rev. Iberoam. Micol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S.; Martinez-de-Oliveira, J.; Donders, G.G.G.; Palmeira-de-Oliveira, R.; Palmeira-de-Oliveira, A. Anti-Candida Activity of Antidepressants Sertraline and Fluoxetine: Effect upon Pre-formed Biofilms. Med. Microbiol. Immunol. 2018, 207, 195–200. [Google Scholar] [CrossRef]
- Costa Silva, R.A.; da Silva, C.R.; de Andrade Neto, J.B.; da Silva, A.R.; Campos, R.S.; Sampaio, L.S.; do Nascimento, F.B.S.A.; da Silva Gaspar, B.; da Cruz Fonseca, S.G.; Josino, M.A.A.; et al. In Vitro Anti-Candida Activity of Selective Serotonin Reuptake Inhibitors against Fluconazole-Resistant Strains and Their Activity against Biofilm-Forming Isolates. Microb. Pathog. 2017, 107, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Palit, P.; Ali, N. Oral Therapy with Sertraline, a Selective Serotonin Reuptake Inhibitor, Shows Activity against Leishmania donovani. J. Antimicrob. Chemother. 2008, 61, 1120–1124. [Google Scholar] [CrossRef]
- Lima, M.L.; Abengózar, M.A.; Nácher-Vázquez, M.; Martínez-Alcázar, M.P.; Barbas, C.; Tempone, A.G.; López-Gonzálvez, Á.; Rivas, L. Molecular Basis of the Leishmanicidal Activity of the Antidepressant Sertraline as a Drug Repurposing Candidate. Antimicrob. Agents Chemother. 2018, 62, e01928. [Google Scholar] [CrossRef]
- Weeks, J.C.; Roberts, W.M.; Leasure, C.; Suzuki, B.M.; Robinson, K.J.; Currey, H.; Wangchuk, P.; Eichenberger, R.M.; Saxton, A.D.; Bird, T.D.; et al. Sertraline, Paroxetine, and Chlorpromazine Are Rapidly Acting Anthelmintic Drugs Capable of Clinical Repurposing. Sci. Rep. 2018, 8, 975. [Google Scholar] [CrossRef]
- Ferreira, D.D.; Mesquita, J.T.; da Costa Silva, T.A.; Romanelli, M.M.; da Gama Jaen Batista, D.; da Silva, C.F.; da Gama, A.N.S.; Neves, B.J.; Melo-Filho, C.C.; Correia Soeiro, M.D.N.; et al. Efficacy of Sertraline against Trypanosoma cruzi: An In Vitro and In Silico Study. J. Venom. Anim. Toxins Incl. Trop. Dis. 2018, 24, 30. [Google Scholar] [CrossRef]
- Kalaycı, S.; Demirci, S.; Sahin, F. Antimicrobial Properties of Various Psychotropic Drugs against Broad Range Microorganisms. Curr. Psychopharmacol. 2014, 3, 195–202. [Google Scholar] [CrossRef]
- Muñoz-Criado, S.; Muñoz-Bellido, J.L.; Alonso-Manzanares, M.A.; Gutiérrez-Zufiaurre, M.N.; García-Rodríguez, J.A. Psychotropic Drugs Inhibit Swarming in Proteus spp. and Related Genera. Clin. Microbiol. Infect. 1998, 4, 447–449. [Google Scholar] [CrossRef]
- Samanta, A.; Chattopadhyay, D.; Sinha, C.; Dulal Jana, A.; Ghosh, S.; Mandal, A.; Banerjee, A.; Hendricks, O.B.; Christensen, J.; Elisabeth Kristiansen, J. Evaluation of In Vivo and In Vitro Antimicrobial Activities of a Selective Serotonin Reuptake Inhibitor Sertraline Hydrochloride. Anti-Infect. Agents 2012, 10, 95–104. [Google Scholar] [CrossRef]
- Ayaz, M.; Subhan, F.; Ahmed, J.; Khan, A.-U.; Ullah, F.; Ullah, I.; Ali, G.; Syed, N.-I.-H.; Hussain, S. Sertraline Enhances the Activity of Antimicrobial Agents against Pathogens of Clinical Relevance. J. Biol. Res. 2015, 22, 4. [Google Scholar] [CrossRef] [PubMed]
- Coban, A.Y.; Tanriverdi Cayci, Y.; Keleş Uludağ, S.; Durupinar, B. Investigation of Antibacterial Activity of Sertralin. Mikrobiyol. Bul. 2009, 43, 651–656. [Google Scholar] [PubMed]
- Muthu, D.; Gowri, M.; Suresh Kumar, G.; Kattimani, V.S.; Girija, E.K. Repurposing of Antidepression Drug Sertraline for Antimicrobial Activity against Staphylococcus aureus: A Potential Approach for the Treatment of Osteomyelitis. New J. Chem. 2019, 43, 5315–5324. [Google Scholar] [CrossRef]
- Li, L.; Kromann, S.; Olsen, J.E.; Svenningsen, S.W.; Olsen, R.H. Insight into Synergetic Mechanisms of Tetracycline and the Selective Serotonin Reuptake Inhibitor, Sertraline, in a Tetracycline-Resistant Strain of Escherichia coli. J. Antibiot. 2017, 70, 944–953. [Google Scholar] [CrossRef]
- Bohnert, J.A.; Szymaniak-Vits, M.; Schuster, S.; Kern, W.V. Efflux Inhibition by Selective Serotonin Reuptake Inhibitors in Escherichia coli. J. Antimicrob. Chemother. 2011, 66, 2057–2060. [Google Scholar] [CrossRef]
- Krzyżek, P.; Franiczek, R.; Krzyżanowska, B.; Łaczmański, Ł.; Migdał, P.; Gościniak, G. In Vitro Activity of 3-Bromopyruvate, an Anti-Cancer Compound, against Antibiotic-Susceptible and Antibiotic-Resistant Helicobacter pylori Strains. Cancers 2019, 11, 229. [Google Scholar] [CrossRef]
- EUCAST The European Committee on Antimicrobial Susceptibility Testing. Breakpoint Tables for Interpretation of MICs and Zone Diameters, Version 5.0; The European Committee on Antimicrobial Susceptibility Testing, 2015. Available online: http://www.eucast.org/clinical_breakpoints/ (accessed on 15 May 2019).
- Krzyżek, P.; Biernat, M.M.; Gościniak, G. Intensive Formation of Coccoid Forms as a Feature Strongly Associated with Highly Pathogenic Helicobacter pylori Strains. Folia Microbiol. 2019, 64, 273–281. [Google Scholar] [CrossRef]
- Cellini, L.; Di Bartolomeo, S.; Di Campli, E.; Genovese, S.; Locatelli, M.; Di Giulio, M. In Vitro Activity of Aloe vera Inner Gel against Helicobacter pylori Strains. Lett. Appl. Microbiol. 2014, 59, 43–48. [Google Scholar] [CrossRef]
- Hirschl, A.M.; Apfalter, P.; Makristathis, A.; Rotter, M.L.; Wimmer, M. In Vitro Activities of Linezolid Alone and in Combination with Amoxicillin, Clarithromycin, and Metronidazole against Helicobacter pylori. Antimicrob. Agents Chemother. 2000, 44, 1977–1979. [Google Scholar] [CrossRef] [PubMed]
- Nostro, A.; Cellini, L.; Bartolomeo, S.D.; Cannatelli, M.A.; Campli, E.D.; Procopio, F.; Grande, R.; Marzio, L.; Alonzo, V. Effects of Combining Extracts (from Propolis or Zingiber officinale) with Clarithromycin on Helicobacter pylori. Phyther. Res. 2006, 20, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Abu-Qatouseh, L.; Abu-Sini, M.; Mayyas, A.; Al-Hiari, Y.; Darwish, R.; Aburjai, T. Synthesis of New Nitrofluoroquinolone Derivatives with Novel Anti-Microbial Properties against Metronidazole Resistant H. pylori. Molecules 2017, 22, 71. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.C.; Jiang, X. Activities of Muscadine Grape Skin and Polyphenolic Constituents against Helicobacter pylori. J. Appl. Microbiol. 2013, 114, 982–991. [Google Scholar] [CrossRef]
- Rhein, J.; Morawski, B.M.; Hullsiek, K.H.; Nabeta, H.W.; Kiggundu, R.; Tugume, L.; Musubire, A.; Akampurira, A.; Smith, K.D.; Alhadab, A.; et al. Efficacy of Adjunctive Sertraline for the Treatment of HIV-Associated Cryptococcal Meningitis: An Open-Label Dose-Ranging Study. Lancet. Infect. Dis. 2016, 16, 809–818. [Google Scholar] [CrossRef]
- Levison, M.E. Pharmacodynamics of Antimicrobial Drugs. Infect. Dis. Clin. North Am. 2004, 18, 451–465. [Google Scholar] [CrossRef]
- Tharmalingam, N.; Port, J.; Castillo, D.; Mylonakis, E. Repurposing the Anthelmintic Drug Niclosamide to Combat Helicobacter pylori. Sci. Rep. 2018, 8, 3701. [Google Scholar] [CrossRef]
- Nilius, M.; Ströhle, A.; Bode, G.; Malfertheiner, P. Coccoid Like Forms (CLF) of Helicobacter pylori. Enzyme Activity and Antigenicity. Zentralbl. Bakteriol. 1993, 280, 272. [Google Scholar] [CrossRef]
- Bode, G.; Mauch, F.; Malfertheiner, P. The Coccoid Forms of Helicobacter pylori. Criteria for Their Viability. Epidemiol. Infect. 1993, 111, 483–490. [Google Scholar] [CrossRef]
- Berry, V.; Jennings, K.; Woodnutt, G. Bactericidal and Morphological Effects of Amoxicillin on Helicobacter pylori. Antimicrob. Agents Chemother. 1995, 39, 1861. [Google Scholar] [CrossRef]
- Fagoonee, S.; Pellicano, R. Helicobacter pylori: Molecular Basis for Colonization and Survival in Gastric Environment and Resistance to Antibiotics. A Short Review. Infect. Dis. 2019, 51, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Horii, T.; Mase, K.; Suzuki, Y.; Kimura, T.; Ohta, M.; Maekawa, M.; Kanno, T.; Kobayashi, M. Antibacterial Activities of Beta-Lactamase Inhibitors Associated with Morphological Changes of Cell Wall in Helicobacter pylori. Helicobacter 2002, 7, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Cole, S.P.; Harwood, J.; Lee, R.; She, R.; Guiney, D.G. Characterization of Monospecies Biofilm Formation by Helicobacter pylori. J. Bacteriol. 2004, 186, 3124–3132. [Google Scholar] [CrossRef] [PubMed]
- Loke, M.F.; Ng, C.G.; Vilashni, Y.; Lim, J.; Ho, B. Understanding the Dimorphic Lifestyles of Human Gastric Pathogen Helicobacter pylori Using the SWATH-Based Proteomics Approach. Sci. Rep. 2016, 6, 26784. [Google Scholar] [CrossRef] [PubMed]
- Attaran, B.; Falsafi, T.; Ghorbanmehr, N. Effect of Biofilm Formation by Clinical Isolates of Helicobacter pylori on the Efflux-Mediated Resistance to Commonly Used Antibiotics. World J. Gastroenterol. 2017, 23, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, F.; Starosta, A.L.; Arenz, S.; Sohmen, D.; Dönhöfer, A.; Wilson, D.N. Tetracycline Antibiotics and Resistance Mechanisms. Biol. Chem. 2014, 395, 559–575. [Google Scholar] [CrossRef] [PubMed]
- Davidson, R.J. In Vitro Activity and Pharmacodynamic/Pharmacokinetic Parameters of Clarithromycin and Azithromycin: Why They Matter in the Treatment of Respiratory Tract Infections. Infect. Drug Resist. 2019, 12, 585–596. [Google Scholar] [CrossRef]
- Salvo, F.; De Sarro, A.; Caputi, A.P.; Polimeni, G. Amoxicillin and Amoxicillin Plus Clavulanate: A Safety Review. Expert Opin. Drug Saf. 2009, 8, 118. [Google Scholar] [CrossRef]
- Dingsdag, S.A.; Hunter, N. Metronidazole: An Update on Metabolism, Structure-Cytotoxicity and Resistance Mechanisms. J. Antimicrob. Chemother. 2018, 73, 265–279. [Google Scholar] [CrossRef]
- Heller, I.; Leitner, S.; Dierich, M.P.; Lass-Flörl, C. Serotonin (5-HT) Enhances the Activity of Amphotericin B against Aspergillus fumigatus In Vitro. Int. J. Antimicrob. Agents 2004, 24, 401–404. [Google Scholar] [CrossRef]
- Tsugawa, H.; Suzuki, H.; Satoh, K.; Hirata, K.; Matsuzaki, J.; Saito, Y.; Suematsu, M.; Hibi, T. Two Amino Acids Mutation of Ferric Uptake Regulator Determines Helicobacter pylori Resistance to Metronidazole. Antioxid. Redox Signal. 2011, 14, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Olekhnovich, I.N.; Vitko, S.; Valliere, M.; Hoffmana, P.S. Response to Metronidazole and Oxidative Stress is Mediated through Homeostatic Regulator HsrA (hp1043) in Helicobacter pylori. J. Bacteriol. 2014, 196, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Tsugawa, H.; Mori, H.; Matsuzaki, J.; Masaoka, T.; Hirayama, T.; Nagasawa, H.; Sakakibara, Y.; Suematsu, M.; Suzuki, H. Nordihydroguaiaretic Acid Disrupts the Antioxidant Ability of Helicobacter pylori through the Repression of SodB Activity In Vitro. Biomed Res. Int. 2015, 2015, 734548. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Kim, N.; Kwon, Y.H.; Nam, R.H.; Kim, J.M.; Park, J.Y.; Lee, Y.S.; Lee, D.H. rdxA, frxA, and Efflux Pump in Metronidazole-Resistant Helicobacter pylori: Their Relation to Clinical Outcomes. J. Gastroenterol. Hepatol. 2018, 33, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Tsugawa, H.; Suzuki, H.; Muraoka, H.; Ikeda, F.; Hirata, K.; Matsuzaki, J.; Saito, Y.; Hibi, T. Enhanced Bacterial Efflux System is the First Step to the Development of Metronidazole Resistance in Helicobacter pylori. Biochem. Biophys. Res. Commun. 2011, 404, 660. [Google Scholar] [CrossRef]
- van Amsterdam, K.; Bart, A.; van der Ende, A. A Helicobacter pylori TolC Efflux Pump Confers Resistance to Metronidazole. Antimicrob. Agents Chemother. 2005, 49, 1477–1482. [Google Scholar] [CrossRef]
- Mehrabadi, J.F.; Sirous, M.; Daryani, N.E.; Eshraghi, S.; Akbari, B.; Shirazi, M.H. Assessing the Role of the RND Efflux Pump in Metronidazole Resistance of Helicobacter pylori by RT-PCR Assay. J. Infect. Dev. Ctries. 2011, 5, 88–93. [Google Scholar] [CrossRef]
- Anoushiravani, M.; Falsafi, T.; Niknam, V. Proton Motive Force-Dependent Efflux of Tetracycline in Clinical Isolates of Helicobacter pylori. J. Med. Microbiol. 2009, 58, 1313. [Google Scholar] [CrossRef]
- Hirata, K.; Suzuki, H.; Nishizawa, T.; Tsugawa, H.; Muraoka, H.; Saito, Y.; Matsuzaki, J.; Hibi, T. Contribution of Efflux Pumps to Clarithromycin Resistance in Helicobacter pylori. J. Gastroenterol. Hepatol. 2010, 25, 79. [Google Scholar] [CrossRef]
- Iwamoto, A.; Tanahashi, T.; Okada, R.; Yoshida, Y.; Kikuchi, K.; Keida, Y.; Murakami, Y.; Yang, L.; Yamamoto, K.; Nishiumi, S.; et al. Whole-Genome Sequencing of Clarithromycin Resistant Helicobacter pylori Characterizes Unidentified Variants of Multidrug Resistant Efflux Pump Genes. Gut Pathog. 2014, 6, 27. [Google Scholar] [CrossRef]
- Kutschke, A.; De Jonge, B.L.M. Compound Efflux in Helicobacter pylori. Antimicrob. Agents Chemother. 2005, 49, 3009–3010. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, Z.Q.; Zheng, P.Y.; Tang, F.A.; Yang, P.C. Influence of Efflux Pump Inhibitors on the Multidrug Resistance of Helicobacter pylori. World J. Gastroenterol. 2010, 16, 1279–1284. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhao, D.; Liu, Z.; Liu, F. Repurposing Psychiatric Drugs as Anti-Cancer Agents. Cancer Lett. 2018, 419, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Mandrioli, R.; Mercolini, L.; Raggi, M.A. Evaluation of the Pharmacokinetics, Safety and Clinical Efficacy of Sertraline Used to Treat Social Anxiety. Expert Opin. Drug Metab. Toxicol. 2013, 9, 1495–1505. [Google Scholar] [CrossRef] [PubMed]
- Topiwala, A.; Chouliaras, L.; Ebmeier, K.P. Prescribing Selective Serotonin Reuptake Inhibitors in Older Age. Maturitas 2014, 77, 123. [Google Scholar] [CrossRef]
- Uppala, P.; Shaik, S.; Anusha Valluru, S.; Nalluri, B.N. Development of Enteric Coated Sustained Release Matrix Tablets of Sertraline Hydrochloride. J. Appl. Pharm. Sci. 2015, 5, 64. [Google Scholar] [CrossRef]
- Johnson, B.M.; Chang, P.T.L. Sertraline Hydrochloride. In Analytical Profiles of Drug Substances and Excipients; Brittain, H.G., Ed.; Academic Press, Inc.: San Diego, CA, USA, 1996; Volume 24, pp. 443–486. [Google Scholar]
- Lewis, R.J.; Angier, M.K.; Williamson, K.S.; Johnson, R.D. Analysis of Sertraline in Postmortem Fluids and Tissues in 11 Aviation Accident Victims. J. Anal. Toxicol. 2013, 37, 216. [Google Scholar] [CrossRef]
- Tremaine, L.M.; Welch, W.M.; Ronfeld, R.A. Metabolism and Disposition of the 5-Hydroxytryptamine Uptake Blocker Sertraline in the Rat and Dog. Drug Metab. Dispos. 1989, 17, 542–550. [Google Scholar]
- Goddard, A.F.; Jessa, M.J.; Barrett, D.A.; Shaw, P.N.; Idström, J.P.; Cederberg, C.; Spiller, R.C. Effect of Omeprazole on the Distribution of Metronidazole, Amoxicillin, and Clarithromycin in Human Gastric Juice. Gastroenterology 1996, 111, 367. [Google Scholar] [CrossRef]
- Sherwood, P.V.; Wibawa, J.I.D.; Atherton, J.C.; Jordan, N.; Jenkins, D.; Barrett, D.A.; Shaw, P.N.; Spiller, R.C. Impact of Acid Secretion, Gastritis, and Mucus Thickness on Gastric Transfer of Antibiotics in Rats. Gut 2002, 51, 490–495. [Google Scholar] [CrossRef]


| Strains | Antibiotic Resistance * | Activity of SER | |||
|---|---|---|---|---|---|
| MET | CLR | MIC ** | MBC ** | MBC/MIC Ratio | |
| J99 | S | S | 4 | 4 | 1 |
| Tx30a | S | S | 4 | 8 | 2 |
| 6237 | S | S | 4 | 4 | 1 |
| 7471 | S | S | 8 | 8 | 1 |
| 7189 | S | R | 4 | 8 | 2 |
| 7556 | S | R | 2 | 4 | 2 |
| 7388 | R | S | 4 | 4 | 1 |
| 7394 | R | S | 4 | 8 | 2 |
| 7143 | R | R | 2 | 8 | 4 |
| 7649 | R | R | 4 | 4 | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krzyżek, P.; Franiczek, R.; Krzyżanowska, B.; Łaczmański, Ł.; Migdał, P.; Gościniak, G. In Vitro Activity of Sertraline, an Antidepressant, Against Antibiotic-Susceptible and Antibiotic-Resistant Helicobacter pylori Strains. Pathogens 2019, 8, 228. https://doi.org/10.3390/pathogens8040228
Krzyżek P, Franiczek R, Krzyżanowska B, Łaczmański Ł, Migdał P, Gościniak G. In Vitro Activity of Sertraline, an Antidepressant, Against Antibiotic-Susceptible and Antibiotic-Resistant Helicobacter pylori Strains. Pathogens. 2019; 8(4):228. https://doi.org/10.3390/pathogens8040228
Chicago/Turabian StyleKrzyżek, Paweł, Roman Franiczek, Barbara Krzyżanowska, Łukasz Łaczmański, Paweł Migdał, and Grażyna Gościniak. 2019. "In Vitro Activity of Sertraline, an Antidepressant, Against Antibiotic-Susceptible and Antibiotic-Resistant Helicobacter pylori Strains" Pathogens 8, no. 4: 228. https://doi.org/10.3390/pathogens8040228
APA StyleKrzyżek, P., Franiczek, R., Krzyżanowska, B., Łaczmański, Ł., Migdał, P., & Gościniak, G. (2019). In Vitro Activity of Sertraline, an Antidepressant, Against Antibiotic-Susceptible and Antibiotic-Resistant Helicobacter pylori Strains. Pathogens, 8(4), 228. https://doi.org/10.3390/pathogens8040228







