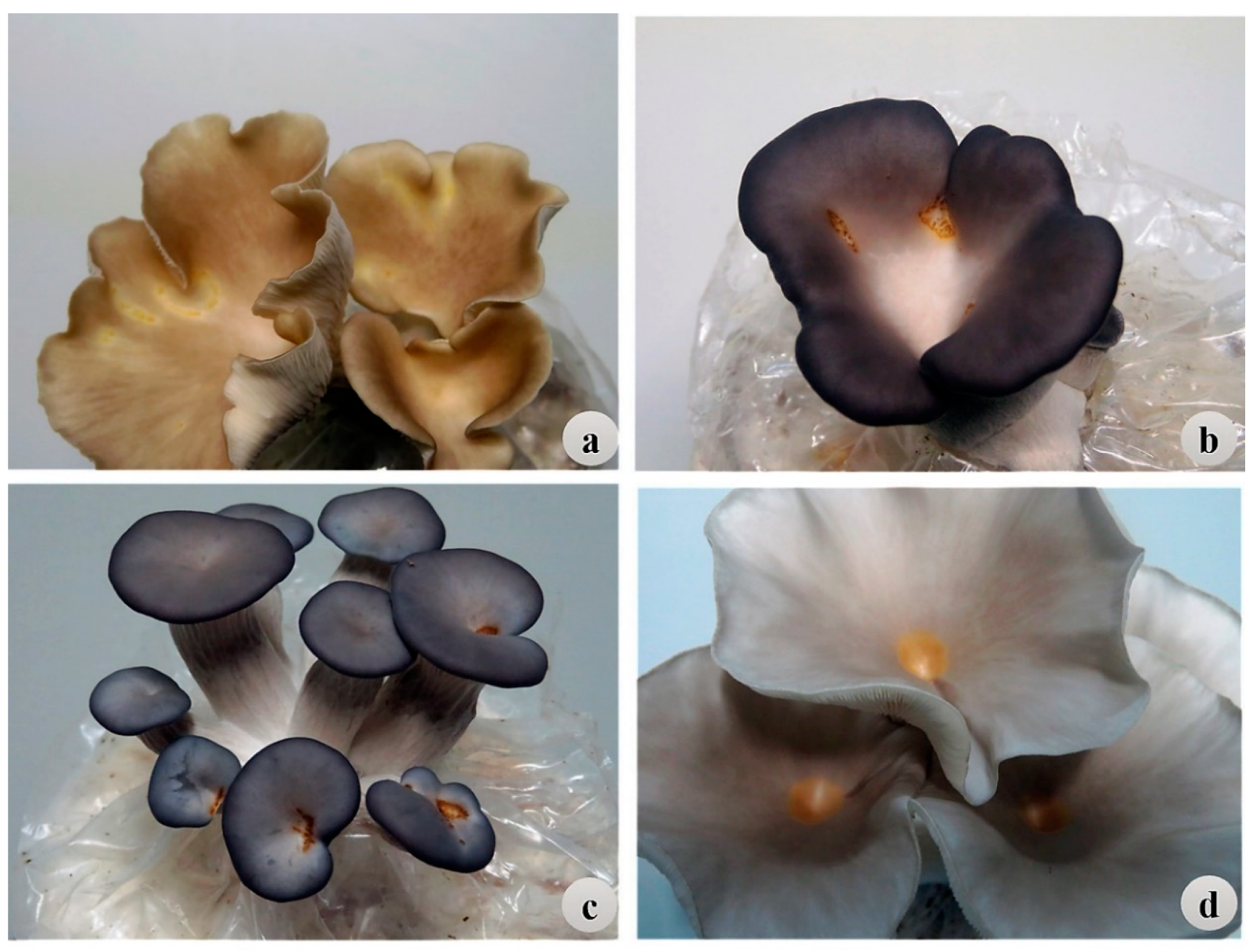
1. Introduction
The
Pleurotus genus consists of approximately 50 recognized species, including more than 10 important species that are highly nutritious and among the most commercially cultivated edible fungi in the world [
1,
2]. In China,
Pleurotus ostreatus (oyster mushroom),
Pleurotus pulmonarius (phoenix mushroom), and
Pleurotus cf.
floridanus (Florida oyster mushroom) are among the most commonly cultivated species with considerable economic value. Their production exceeds 500 million tons annually for both domestic consumption and export purposes [
3].
However, one of the important diseases that threatens
Pleurotus mushroom production is brown blotch disease (BBD), caused by
Pseudomonas tolaasii. The bacterium can also infect other economically important mushrooms, such as
Agaricus bisporus, Lentinus edodes, and
Flammulina velutipes [
4,
5,
6,
7].
P. tolaasii can infect all the stages of
Pleurotus spp. fruiting body [
8] by secreting tolaasin, a core toxin used to cause BBD on the mushroom [
9]. Tolaasin causes lysis of hyphal cell membranes and induces an increase in the amount of active tyrosinase, leading to melanin synthesis at the site of infection [
10]. The characteristic symptoms of BBD include initially small, separated pale brown discolorations, which then spread all over the pileus surface. When BBD is severe, blotches become darker and sunken, ultimately rendering the pileus unappealing for human consumption [
11]. These symptoms lead to reduced quality [
12,
13], which can cause a significant economic loss of up to 25% [
14,
15].
Over the past years, strategies to control BBD and interrupt pathogen spread have included the use of chemicals (antibiotics, hydrogen peroxide, and chlorinated compounds), management of host environment, and biocontrol agents (plant extracts and antagonistic microbes) [
16,
17,
18,
19,
20,
21,
22]; however, none of these strategies appear to be fully efficient in managing the disease. There is even an increased concern about the effects of these chemicals on consumers. Further, the ability of
P. tolaasii to tolerate adverse environmental conditions through phenotypic plasticity makes BBD control difficult [
23,
24,
25], thereby resulting in huge crop losses during severe epidemics.
At present, the use of resistant strains is the most efficient and long-term method for BBD control [
26,
27,
28], since it not only reduces the loss of the mushroom quality, but also the cost associated with measures undertaken to control the disease. Therefore, resistance sources in
Pleurotus germplasm are needed for the sustainable management of BBD. Several studies have described partial resistance to BBD in
A. bisporus strains [
26,
27,
29,
30]. However, only one has reported partial resistance of
P. ostreatus strains to
P.
tolaasii [
31].
The
Pleurotus genus contains many species with genetically diverse strains in the wild, and many are also maintained by various mycological germplasm repositories [
32,
33]. Additionally, interspecific hybridization can occur between species [
34]. Therefore, developing BBD-resistant strains may rely on characterization and incorporation of resistance genes contributed from wild strains or closely related species. Despite the large collection of
Pleurotus species in many germplasm repositories, little is known about their resistance to BBD. Thus, searching for novel sources of resistance to BBD is of high priority.
In screening for disease resistance in mushrooms, several studies have recommended the artificial inoculation of the pathogen and assessment of the host response under a controlled environment. Oliver et al. [
27] described the use of the direct inoculation of the pathogen on the mushroom pileus/cap (IMP) method to screen for BBD resistance in
A.
bisporus. However, Zhang et al. [
31] could not clearly distinguish between the host responses from
P.
ostreatus and proposed the inoculation of the pathogen on the mushroom spawned substrate (IMSS) as the effective method for successful evaluation of BBD resistance in
P.
ostreatus. The objectives of this study were to (1) evaluate the efficacy of the IMP screening method on
Pleurotus species; (2) determine the influence of inoculum dose, temperature and disease assessment time on the resistance test; and (3) identify possible resistance sources from a collection of 97
Pleurotus strains.
3. Discussion
Pleurotus species are among the important cultivated mushrooms that are susceptible to BBD. To help minimize economic losses in commercial production of
Pleurotus mushroom, the use of resistant strains is considered the most effective and desirable solution for BBD management. Among
Pleurotus species, only
P.
ostreatus strains have been evaluated for BBD resistance [
31]. Therefore, it is imperative to find and develop resistant strains of other
Pleurotus species to control BBD. In this study, we screened 97 strains from three major cultivated
Pleurotus species (
P.
pulmonarius,
P. cf.
floridanus, and
P.
ostreatus) to identify germplasm resistant to BBD using the IMP and IMSS methods.
We first found the IMP screening method could be used to assess the response of the three species in the
Pleurotus genus to the pathogen
P.
tolaasii, which is consistent with previous studies that used this method on other kinds of mushrooms [
27,
29,
37,
38,
39]. However, Zhang et al. [
31] reported that it was difficult using the IMP method to distinguish between the host response or symptoms in
P.
ostreatus. This might be due to the selected disease scale and the length of the disease incubation period used for symptom evaluation. The main advantage of the IMP method is the application of a uniform inoculum directly on the host pileus for resistance evaluation. The high inoculum concentration (1 × 10
8 CFU/mL) applied on the pileus was a complete resistance detection assay [
40], which minimized the chance of disease escape to make immunity detection easy. Also, the IMP depicted all sources of inoculum that may land on the mushroom pileus, especially secondary infections that occur during the cultivation of the mushroom in farms, transportation, storage, and marketing [
12].
The three
Pleurotus species had a different incubating temperature for fruiting. However, it was not surprising to find that temperature had no significant influence on BBD severity. This was consistent with Wong and Preece [
41], who noted changes in temperature and relative humidity during cultivation had no effects on BBD severity in
A. bisporus. Thus, it was possible to compare and select resistant strains under the two different incubation temperatures.
Based on the IMP method, all of the 97
Pleurotus strains were susceptible to
P.
tolaasii infection, but the degrees of susceptibility varied among the strains. For example, among the
P. pulmonarius strains, F65, F365, and F18 had significantly different means for disease severity, ranging from 1.07–3.30 (
Table 2). Oliver et al. [
27] also obtained a wide variation in response to BBD among
Agaricus strains. The variations in susceptibility among the strains may be attributed to differences in genotype [
33], fruiting body nutrient composition, texture, and quality [
10,
42]. Murata [
43] noticed the activity of tolaasin, the core virulence factor, was triggered by the host and the signal strength may be influenced by the chemical composition of the mushroom fruiting body. Estrada et al. [
37] also found differences in blotch symptom severity between two strains of
P. eryngii, which contained varied inherent levels of copper that is a cofactor for tyrosinase in melanin synthesis or the browning of the diseased portions on the pileus [
44,
45].
Compared to the IMP method, the IMSS method revealed a marked difference in resistance levels among the 97 strains. For example, among the
P. cf.
floridanus strains, P282 was highly susceptible, while P287 was highly resistant. Moreover, a higher level of resistance to BBD was observed for some strains as their fruiting bodies were completely symptomless. This contrasts with Zhang et al. [
31], who observed partial resistance in 37 strains of
P. ostreatus using the IMSS method. The study showed that besides the host pileus interacting with
P. tolaasii, host vegetative mycelia interaction with
P. tolaasii could prevent pathogen spread onto the pileus surface during the initial stages of fruiting body development. The vegetative mycelia of
Pleurotus spp. are known to degrade living bacteria [
46], and the antagonistic interactions between the host mycelia and the pathogen may reduce
P. tolaasii population in the spawned substrate, which leads to less pathogen load reaching the mushroom pileus. The
Pleurotus strains showed variable levels of
P. tolaasii (CFU) in the substrate after the first flush harvest. Thus, the resistance of the strains may be related to
P. tolaasii population in the spawn substrate. In contrast, Zhang et al. [
31] reported no relationship between mushroom resistance and
P. tolaasii population in the substrate.
Further, a possible resistance strategy obtained with the IMSS method is the escape of the host’s fruiting body from the minimum inoculum dosage required for successful infection. We found that three strains during the preliminary evaluation of the IMP method could carry ≈2.6 × 10
5 cells of
P. tolaasii/cap without showing symptoms on their fruiting bodies. Similar observations had been made on
Agaricus mushrooms [
27,
47].
The classification of the strains into the susceptible and resistant groups was influenced by the methods (IMSS or IMP) used for assessment. For example, some strains (F324, P334, and P346) were completely symptomless using the IMSS; however, they showed symptoms in the IMP method. Additionally, there was a weak or no linear relationship for the responses of strain between the two screening methods. The discrepancy between the methods may be due to differences in the disease scales used for the study. Further, because the specific disease resistance mechanisms at the different mushroom growth stages (vegetative mycelia and fruiting) are poorly understood, it is possible that the IMP and IMSS screening methods exhibit distinct disease resistance mechanisms. The tendency of hosts to advance defenses throughout their developmental stages has been reported in numerous plant pathological systems and are considered paramount in disease management [
48,
49,
50]. Considering the sources of pathogen inoculum and dissemination in mushroom farms, as well as the host developmental stage at which infection occurs, to identify resistance to BBD in a collection, the selection of genotypes was based on those with lowered DS in both screening methods, using their overall disease index (DI). The results suggest that the data from IMP and IMSS screening methods should be combined to enhance the selection process when evaluating resistance to BBD, because they reflect the primary and secondary routes of infection by
P.
tolaasii and demonstrate the accurate total resistance in the mushroom.
Our study identified P. pulmonarius and P. cf. floridanus strains as sources of resistance to P. tolaasii. On average, P. pulmonarius exhibited higher levels of resistance to P. tolaasii than P. cf. floridanus and P. ostreatus. Sixteen strains identified herein as partially resistant (both HR and MR) have the potential to reduce mushroom production losses due to BBD under both primary and secondary infections. Five strains, F122, F324, F344, and F9 of P. pulmonarius and P287 of P. cf. floridanus, ranked highly resistant. Compared to the other two species, none of the P. ostreatus strains included in the study were classified as partially resistant based on the DI. However, the large variance in susceptibility attributed to strains in the study indicated that individual strains rather than species are the most needed for breeding against P. tolaasii in further study. The Pleurotus collection originated from five provinces in China and two other countries (Pakistan and USA). However, there were no obvious trends for the association of BBD resistance to biogeography. Also, there was a range of resistance within the collection for both wild and cultivated strains. For example, most of the P. pulmonarius strains that were resistant to P. tolaasii were wild-type strains. In order to exploit the five highly resistant Pleurotus strains, exhaustive histological and molecular work is needed to elucidate the precise resistance mechanisms present in the Pleurotus collection. Further analyses on the nutritional composition and structural characteristics of the resistant strains are required in order to introgress the resistance to other popular strains by breeding.