Intra-Species and Inter-Species Differences in Cytokine Production by Porcine Antigen-Presenting Cells Stimulated by Mycoplasma hyopneumoniae, M. hyorhinis, and M. flocculare
Abstract
:1. Introduction
2. Results
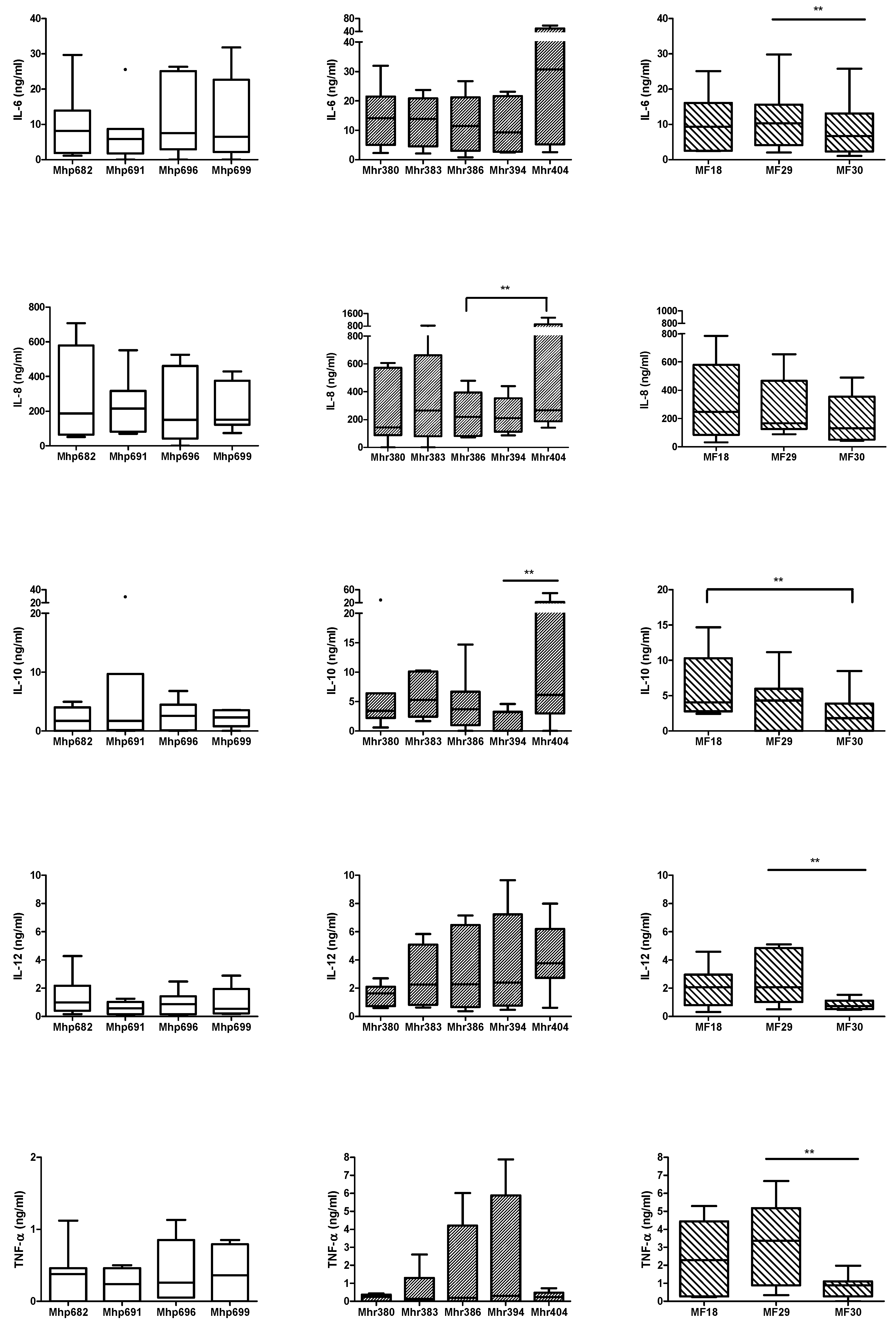
2.1. Comparison of Cytokine Production Levels Induced by M. hyopneumoniae, M. hyorhinis, or M. flocculare
2.2. Comparison of Cytokine Production Levels between M. hyopneumoniae, M. hyorhinis, and M. flocculare, Alone or in Association
2.2.1. Association between M. flocculare and M. hyopneumoniae Strains
2.2.2. Association between M. flocculare and M. hyorhinis Strains
2.2.3. Association between M. hyorhinis and M. hyopneumoniae Strains
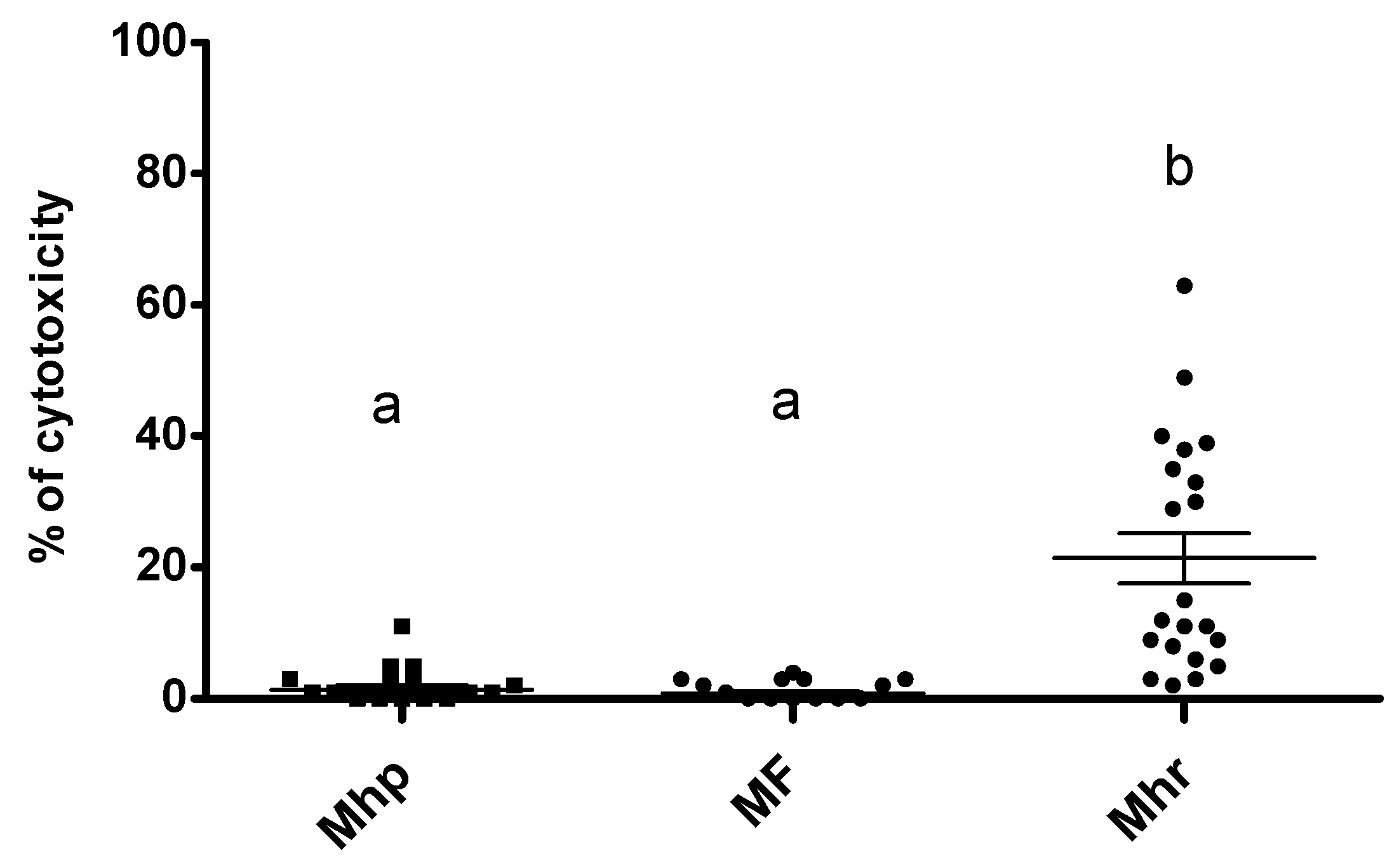
2.3. Viability of BM-DCs Stimulated by M. hyopneumoniae, M. hyorhinis, and M. flocculare Species
3. Discussion
4. Materials and Methods
4.1. Mycoplasma Strains and Growth Conditions
4.2. Isolation of Bone-Marrow Cells and Generation of Bone-Marrow-Derived Dendritic Cells (BM-DCs)
4.3. Stimulation of Immature BM-DCs by Mycoplasma Strains
4.4. Cytokine Quantification by Enzyme Linked Immunosorbent Assay (ELISA)
4.5. Cell Viability Assay
4.6. Statistical Analysis
4.6.1. Strain and Species Effects of M. hyopneumoniae, M. hyorhinis, or M. flocculare on Cytokine Production Levels
4.6.2. Mycoplasmal Co-Infection Effect on Cytokine Production Levels
4.6.3. Cytotoxicity effect of M. hyopneumoniae, M. hyorhinis, and M. flocculare on BM-DCs
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethical Approval
References
- Thacker, E.L.; Minion, C. Mycoplasmosis. In Diseases of Swine, 10th ed.; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G., Eds.; Wiley Blackwell: Hoboken, NJ, USA, 2012; pp. 779–808. [Google Scholar]
- Maes, D.; Segales, J.; Meyns, T.; Sibila, M.; Pieters, M.; Haesebrouck, F. Control of Mycoplasma hyopneumoniae infections in pigs. Vet. Microbiol. 2008, 126, 297–309. [Google Scholar] [CrossRef] [PubMed]
- Hillen, S.; von Berg, S.; Kohler, K.; Reinacher, M.; Willems, H.; Reiner, G. Occurrence and severity of lung lesions in slaughter pigs vaccinated against Mycoplasma hyopneumoniae with different strategies. Prev. Vet. Med. 2014, 113, 580–588. [Google Scholar] [CrossRef]
- Redondo, E.; Masot, A.J.; Fernandez, A.; Gazquez, A. Histopathological and immunohistochemical findings in the lungs of pigs infected experimentally with Mycoplasma hyopneumoniae. J. Comp. Pathol. 2009, 140, 260–270. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, H.; Quesada, O.; Assuncao, P.; Castro, A.; Rodriguez, F. Cytokine expression in porcine lungs experimentally infected with Mycoplasma hyopneumoniae. Vet. Immunol. Immunopathol. 2006, 109, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.; Kwon, D.; Jung, K.; Ha, Y.; Lee, Y.H.; Kim, O.; Park, H.K.; Kim, S.H.; Hwang, K.K.; Chae, C. Expression of inflammatory cytokines in pigs experimentally infected with Mycoplasma hyopneumoniae. J. Comp. Pathol. 2006, 134, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, F.; Ramirez, G.A.; Sarradell, J.; Andrada, M.; Lorenzo, H. Immunohistochemical labelling of cytokines in lung lesions of pigs naturally infected with Mycoplasma hyopneumoniae. J. Comp. Pathol. 2004, 130, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Asai, T.; Okada, M.; Ono, M.; Irisawa, T.; Mori, Y.; Yokomizo, Y.; Sato, S. Increased levels of tumor necrosis factor and interleukin 1 in bronchoalveolar lavage fuids from pigs infected with Mycoplasma hyopneumoniae. Vet. Immunol. Immunopathol. 1993, 38, 253–260. [Google Scholar] [CrossRef]
- Hwang, M.H.; Damte, D.; Lee, J.S.; Gebru, E.; Chang, Z.Q.; Cheng, H.; Jung, B.Y.; Rhee, M.H.; Park, S.C. Mycoplasma hyopneumoniae induces pro-inflammatory cytokine and nitric oxide production through NFkappaB and MAPK pathways in RAW264.7 cells. Vet. Res. Commun. 2011, 35, 21–34. [Google Scholar] [CrossRef]
- Thanawongnuwech, R.; Young, T.; Thacker, B.; Thacker, E. Differential production of proinfammatory cytokines: In vitro PRRSV and Mycoplasma hyopneumoniae co-infection model. Vet. Immunol. Immunopathol. 2001, 79, 115–127. [Google Scholar] [CrossRef]
- DeVries, M.E.; Ran, L.; Kelvin, D.J. On the edge: The physiological and pathophysiological role of chemokines during inflammatory and immunological responses. Immunology 1999, 11, 95–104. [Google Scholar] [CrossRef]
- Jones, H.P.; Tabor, L.; Sun, X.; Woolard, M.D.; Simecka, J.W. Depletion of CD8+ T Cells Exacerbates CD4+ Th Cell-Associated Inflammatory Lesions During Murine Mycoplasma Respiratory Disease. J. Immunol. 2002, 168, 3493–3501. [Google Scholar] [CrossRef]
- Woolard, M.D.; Hodge, L.M.; Jones, H.P.; Schoeb, T.R.; Simecka, J.W. The Upper and Lower Respiratory Tracts Differ in Their Requirement of IFN- g and IL-4 in Controlling Respiratory Mycoplasma Infection and Disease. J. Immunol. 2004, 172, 6875–6883. [Google Scholar] [CrossRef] [PubMed]
- Sarradell, J.; Andrada, M.; Ramirez, A.S.; Fernandez, A.; Gomez-Villamandos, J.C.; Jover, A.; Lorenzo, H.; Herraez, P.; Rodriguez, F. A Morphologic and Immunohistochemical Study of the Bronchus-associated Lymphoid Tissue of Pigs Naturally Infected with Mycoplasma hyopneumoniae. Vet. Pathol. 2003, 40, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Hu, W.; Wei, Y.; Feng, Z.; Yang, Q. Effects of Mycoplasma hyopneumoniae on porcine nasal cavity dendritic cells. Vet. Microbiol. 2017, 198, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Jones, H.P.; Dobbs, N.; Bodhankar, S.; Simecka, J.W. Dendritic cells are the major antigen presenting cells in inflammatory lesions of murine Mycoplasma respiratory disease. PLoS ONE 2013, 8, e55984. [Google Scholar] [CrossRef]
- Zhang, H.; Lunney, J.K.; Baker, R.B.; Opriessnig, T. Cytokine and chemokine mRNA expression profiles in tracheobronchial lymph nodes from pigs singularly infected or coinfected with porcine circovirus type 2 (PCV2) and Mycoplasma hyopneumoniae (MHYO). Vet. Immunol. Immunopathol. 2011, 140, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Fourour, S.; Fablet, C.; Tocqueville, V.; Dorenlor, V.; Eono, F.; Eveno, E.; Kempf, I.; Marois-Crehan, C. A new multiplex real-time TaqMan((R)) PCR for quantification of Mycoplasma hyopneumoniae, M. hyorhinis and M. flocculare: Exploratory epidemiological investigations to research mycoplasmal association in enzootic pneumonia-like lesions in slaughtered pigs. J. Appl. Microbiol. 2018, 125, 345–355. [Google Scholar] [CrossRef]
- Madec, F.; Kobisch, M. Bilan Lesionnel des Poumons de Porcs Charcutiers à l’abattoir; Journées de la Recherche Porcine: Paris, France, 1982; pp. 405–412. [Google Scholar]
- Shen, Y.; Hu, W.; Wei, Y.; Feng, Z.; Yang, Q. The immune mechanism of Mycoplasma hyopneumoniae 168 vaccine strain through dendritic cells. BMC Vet. Res. 2017, 13, 285. [Google Scholar] [CrossRef]
- Martelet, L.; Lacouture, S.; Goyette-Desjardins, G.; Beauchamp, G.; Surprenant, C.; Gottschalk, M.; Segura, M. Porcine Dendritic Cells as an In Vitro Model to Assess the Immunological Behaviour of Streptococcus suis Subunit Vaccine Formulations and the Polarizing Effect of Adjuvants. Pathogens 2017, 6, 13. [Google Scholar] [CrossRef]
- Lecours, M.P.; Segura, M.; Lachance, C.; Mussa, T.; Surprenant, C.; Montoya, M.; Gottschalk, M. Characterization of porcine dendritic cell response to Streptococcus suis. Vet. Res. 2011, 42, 72. [Google Scholar] [CrossRef]
- Goret, J.; Beven, L.; Faustin, B.; Contin-Bordes, C.; Le Roy, C.; Claverol, S.; Renaudin, H.; Bebear, C.; Pereyre, S. Interaction of Mycoplasma hominis PG21 with Human Dendritic Cells: Interleukin-23-Inducing mycoplasmal Lipoproteins and Inflammasome Activation of the Cell. J. Bacteriol. 2017, 199, e00213-17. [Google Scholar] [CrossRef] [PubMed]
- Rottem, S. Interaction of mycoplasmas With Host Cells. Physiol. Rev. 2003, 83, 417–432. [Google Scholar] [CrossRef]
- Calcutt, M.J.; Foecking, M.F.; Heidari, M.B.; McIntosh, M.A. Complete Genome Sequence of Mycoplasma flocculare Strain Ms42T (ATCC 27399T). Genome Announc. 2015, 3, e00124-15. [Google Scholar] [CrossRef] [PubMed]
- Woolley, L.K.; Fell, S.; Gonsalves, J.R.; Walker, M.J.; Djordjevic, S.P.; Jenkins, C.; Eamens, G.J. Evaluation of clinical, histological and immunological changes and qPCR detection of Mycoplasma hyopneumoniae in tissues during the early stages of mycoplasmal pneumonia in pigs after experimental challenge with two field isolates. Vet. Microbiol. 2012, 161, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Hooper, W.C.; Phillips, D.J.; Talkington, D.F. Cytokines in Mycoplasma pneumoniae infections. Cytokine Growth Factor Rev. 2004, 15, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Michiels, A.; Arsenakis, I.; Boyen, F.; Krejci, R.; Haesebrouck, F.; Maes, D. Efficacy of one dose vaccination against experimental infection with two Mycoplasma hyopneumoniae strains. BMC Vet. Res. 2017, 13, 274. [Google Scholar] [CrossRef]
- Moser, M.; Murphy, K.M. Dendritic cell regulation of TH1-TH2 development. Nat. Immunol. 2000, 1, 199–205. [Google Scholar] [CrossRef]
- Thanawongnuwech, R.; Thacker, B.; Halbur, P.; Thacker, E.L. Increased production of proinflammatory cytokines following infection with porcine reproductive and respiratory syndrome virus and Mycoplasma hyopneumoniae. Clin. Diagn. Lab. Immunol. 2004, 11, 901–908. [Google Scholar] [CrossRef]
- Bai, F.; Ni, B.; Liu, M.; Feng, Z.; Xiong, Q.; Xiao, S.; Shao, G. Mycoplasma hyopneumoniae-derived lipid-associated membrane proteins induce apoptosis in porcine alveolar macrophage via increasing nitric oxide production, oxidative stress, and caspase-3 activation. Vet. Immunol. Immunopathol. 2013, 155, 155–161. [Google Scholar] [CrossRef]
- Friis, N.F. Some recommendations concerning primary isolation of Mycoplasma suipneumoniae and Mycoplasma flocculare a survey. Nord. Vet. 1975, 27, 337–339. [Google Scholar]

| Mycoplasma Strains | Herds | Gross Pneumonia-Like Lesion Score | |
|---|---|---|---|
| Lung Score where the Strain Was Isolated (/28) | Average Score of 15 Lungs from the Herd (/28) | ||
| Mycoplasma hyopneumoniae 682 (Mhp682) | a | 17 | 10.8 |
| Mycoplasma hyopneumoniae 691 (Mhp691) | b | 0 | 10.4 |
| Mycoplasma hyopneumoniae 696 (Mhp696) | c | 15 | 7.3 |
| Mycoplasma hyopneumoniae 699 (Mhp699) | d | 11 | 10.9 |
| Mycoplasma hyorhinis 380 (Mhr380) | a | 17 | 10.8 |
| Mycoplasma hyorhinis 383 (Mhr383) | b | 5 | 10.4 |
| Mycoplasma hyorhinis 386 (Mhr386) | d | 12 | 10.9 |
| Mycoplasma hyorhinis 394 (Mhr394) | f | 2 | 1.4 |
| Mycoplasma hyorhinis 404 (Mhr404) | e | 19 | 8.9 |
| Mycoplasma flocculare 18 (MF18) | e | 6 | 8.9 |
| Mycoplasma flocculare 29 (MF29) | c | 8 | 7.3 |
| Mycoplasma flocculare 30 (MF30) | f | 0 | 1.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fourour, S.; Marois-Créhan, C.; Martelet, L.; Fablet, C.; Kempf, I.; Gottschalk, M.; Segura, M. Intra-Species and Inter-Species Differences in Cytokine Production by Porcine Antigen-Presenting Cells Stimulated by Mycoplasma hyopneumoniae, M. hyorhinis, and M. flocculare. Pathogens 2019, 8, 34. https://doi.org/10.3390/pathogens8010034
Fourour S, Marois-Créhan C, Martelet L, Fablet C, Kempf I, Gottschalk M, Segura M. Intra-Species and Inter-Species Differences in Cytokine Production by Porcine Antigen-Presenting Cells Stimulated by Mycoplasma hyopneumoniae, M. hyorhinis, and M. flocculare. Pathogens. 2019; 8(1):34. https://doi.org/10.3390/pathogens8010034
Chicago/Turabian StyleFourour, Sarah, Corinne Marois-Créhan, Léa Martelet, Christelle Fablet, Isabelle Kempf, Marcelo Gottschalk, and Mariela Segura. 2019. "Intra-Species and Inter-Species Differences in Cytokine Production by Porcine Antigen-Presenting Cells Stimulated by Mycoplasma hyopneumoniae, M. hyorhinis, and M. flocculare" Pathogens 8, no. 1: 34. https://doi.org/10.3390/pathogens8010034
APA StyleFourour, S., Marois-Créhan, C., Martelet, L., Fablet, C., Kempf, I., Gottschalk, M., & Segura, M. (2019). Intra-Species and Inter-Species Differences in Cytokine Production by Porcine Antigen-Presenting Cells Stimulated by Mycoplasma hyopneumoniae, M. hyorhinis, and M. flocculare. Pathogens, 8(1), 34. https://doi.org/10.3390/pathogens8010034





