HIV Replication in Humanized IL-3/GM-CSF-Transgenic NOG Mice
Abstract
1. Introduction
2. Results and Discussion
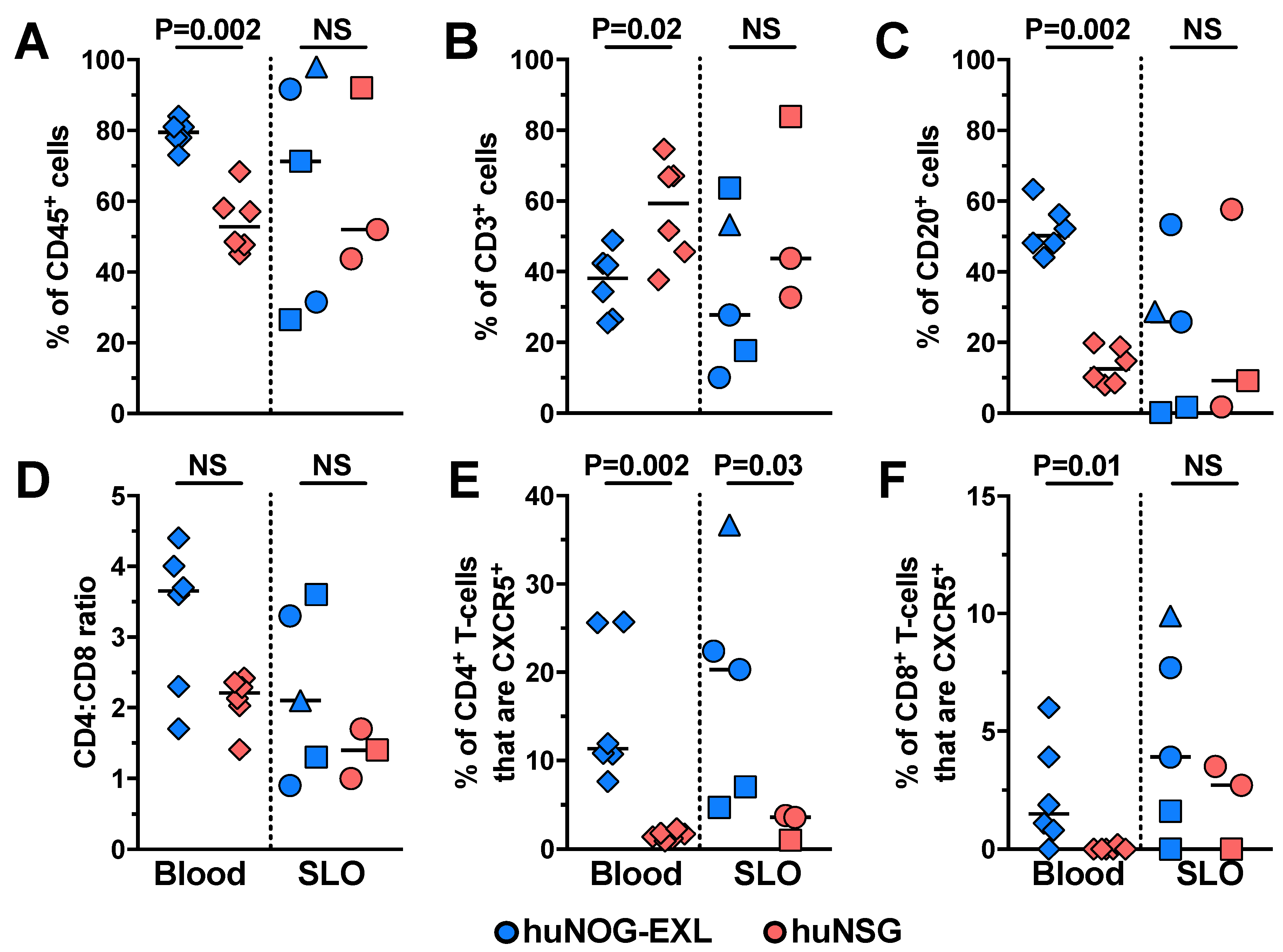
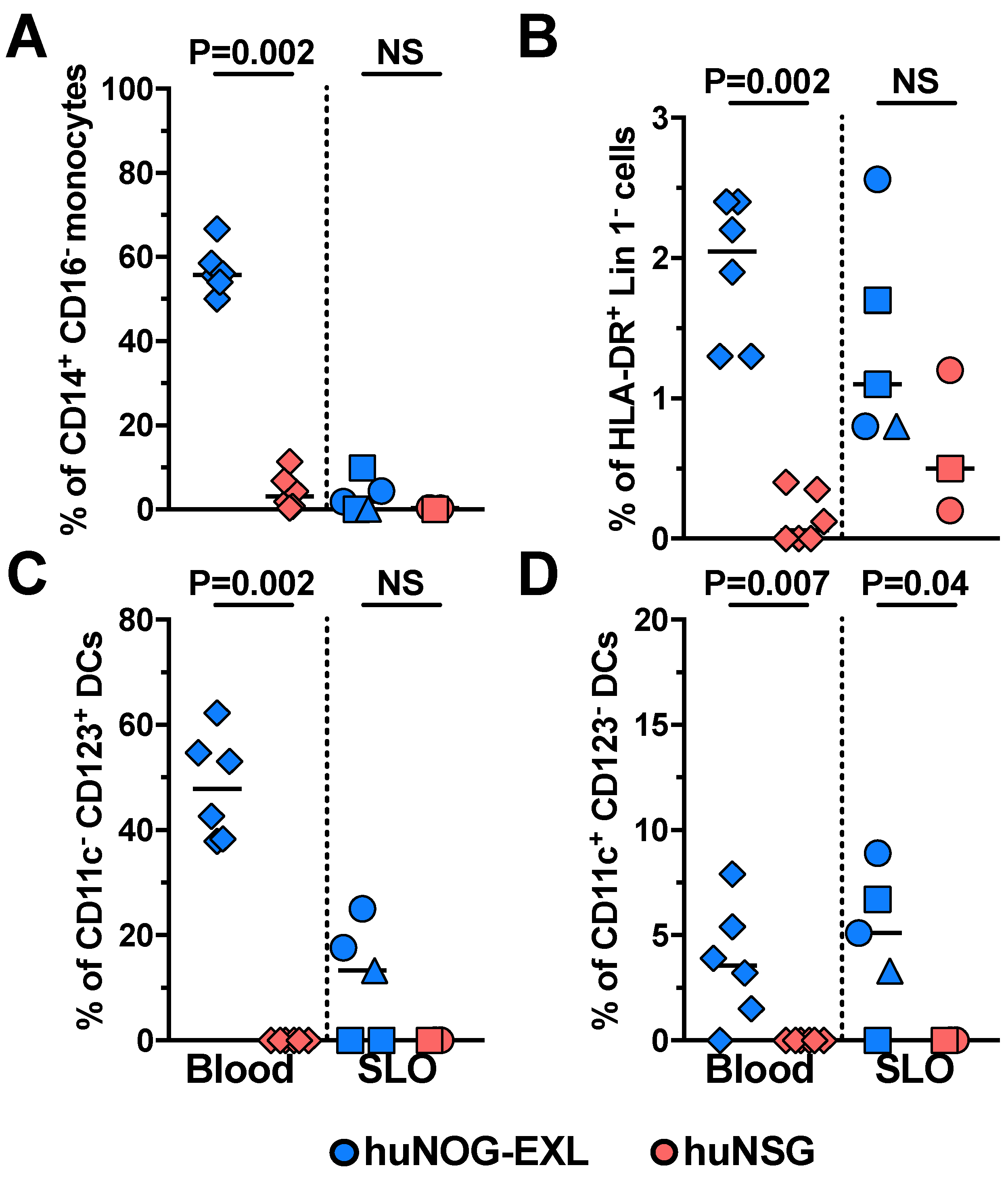
2.1. Characterizing Lymphoid and Myeloid Populations in huNOG-EXL Mice
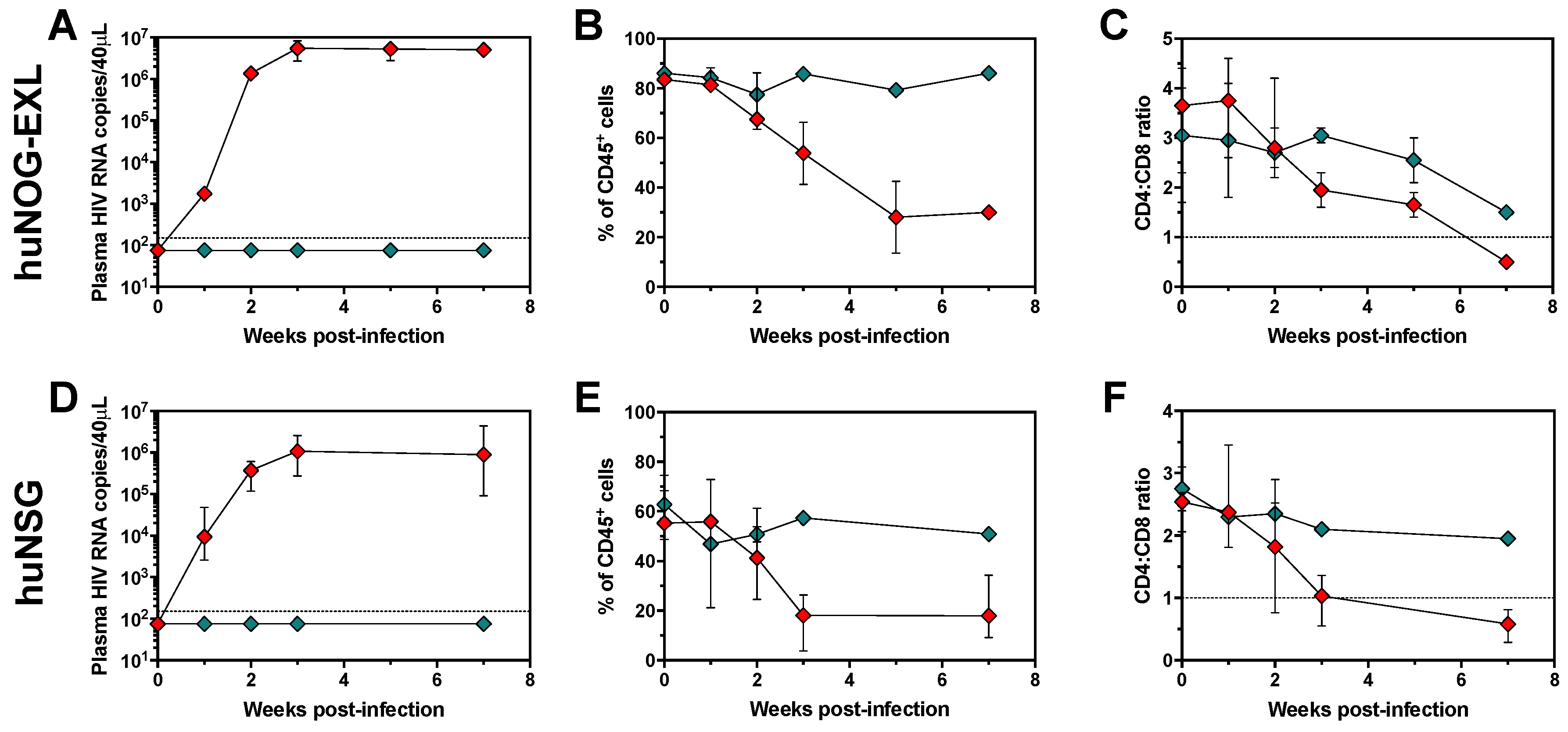
2.2. HIV Replication in huNOG-EXL Mice
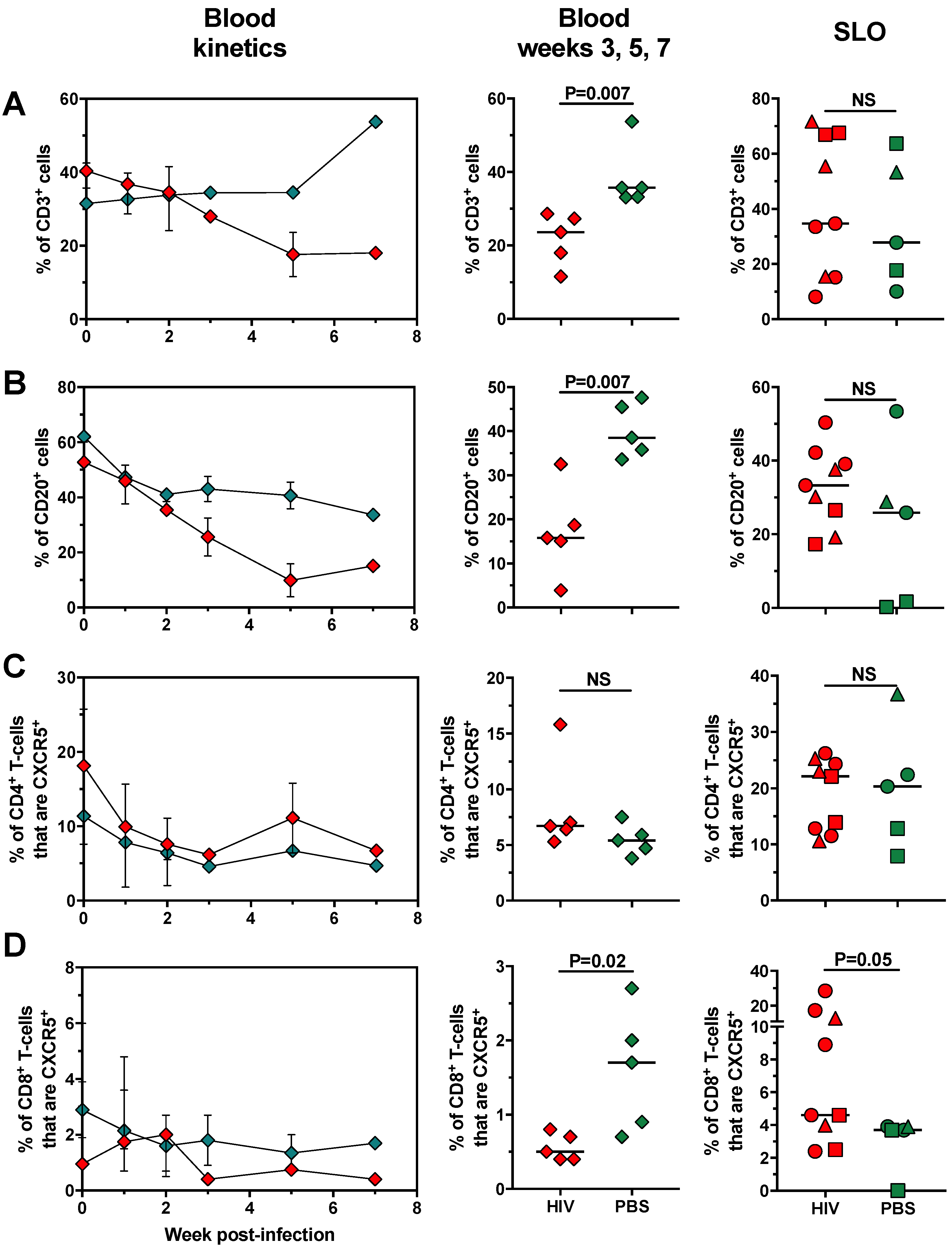
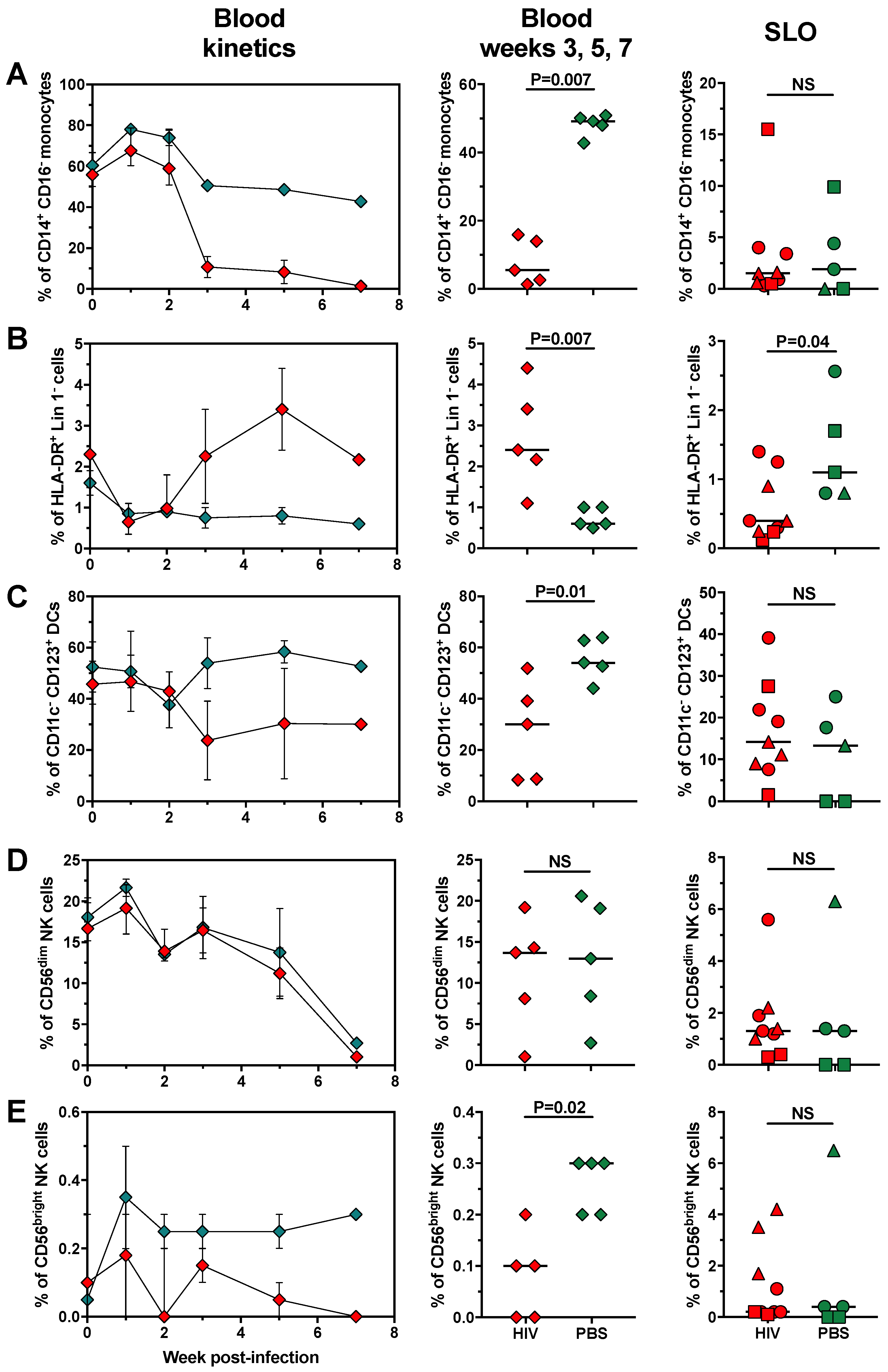
2.3. Changes in Cell Populations along HIV Infection Time in huNOG-EXL Mice
2.4. The Changes in Lymphoid and Myeloid Cells are Associated with the Level of HIV Replication in huNOG-EXL Mice
3. Conclusions
4. Materials and Methods
4.1. Ethics Statement
4.2. Generation and HIV Infection of Humanized Mice
4.3. Flow Cytometry
4.4. Determination of Viral Load
4.5. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Malim, M.H.; Bieniasz, P.D. HIV restriction factors and mechanisms of evasion. Cold Spring Harb. Perspect. Med. 2012, 2, a006940. [Google Scholar] [CrossRef] [PubMed]
- Masse-Ranson, G.; Mouquet, H.; Di Santo, J.P. Humanized mouse models to study pathophysiology and treatment of HIV infection. Curr. Opin. HIV AIDS 2018, 13, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Estes, J.D.; Wong, S.W.; Brenchley, J.M. Nonhuman primate models of human viral infections. Nat. Rev. Immunol. 2018, 18, 390–404. [Google Scholar] [CrossRef] [PubMed]
- Victor Garcia, J. Humanized mice for HIV and AIDS research. Curr. Opin. Virol. 2016, 19, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Billerbeck, E.; Barry, W.T.; Mu, K.; Dorner, M.; Rice, C.M.; Ploss, A. Development of human CD4+FoxP3+ regulatory T cells in human stem cell factor-, granulocyte-macrophage colony-stimulating factor-, and interleukin-3-expressing NOD-SCID IL2R??null humanized mice. Blood 2011, 117, 3076–3086. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Hiramatsu, H.; Kobayashi, K.; Suzue, K.; Kawahata, M.; Hioki, K.; Ueyama, Y.; Koyanagi, Y.; Sugamura, K.; Tsuji, K.; et al. NOD/SCID/γcnull mouse: An excellent recipient mouse model for engraftment of human cells. Blood 2002, 100, 3175–3182. [Google Scholar] [CrossRef] [PubMed]
- Ito, R.; Takahashi, T.; Katano, I.; Kawai, K.; Kamisako, T.; Ogura, T.; Ida-Tanaka, M.; Suemizu, H.; Nunomura, S.; Ra, C.; et al. Establishment of a human allergy model using human IL-3/GM-CSF-transgenic NOG mice. J. Immunol. 2013, 191, 2890–2899. [Google Scholar] [CrossRef] [PubMed]
- Shultz, L.D.; Lyons, B.L.; Burzenski, L.M.; Gott, B.; Chen, X.; Chaleff, S.; Kotb, M.; Gillies, S.D.; King, M.; Mangada, J.; et al. Human lymphoid and myeloid cell development in NOD/LtSz-scid IL2R gamma null mice engrafted with mobilized human hemopoietic stem cells. J. Immunol. 2005, 174, 6477–6489. [Google Scholar] [CrossRef]
- Shultz, L.D.; Ishikawa, F.; Greiner, D.L. Humanized mice in translational biomedical research. Nat. Rev. Immunol. 2007, 7, 118. [Google Scholar] [CrossRef]
- Medina-Moreno, S.; Dowling, T.C.; Zapata, J.C.; Le, N.M.; Sausville, E.; Bryant, J.; Redfield, R.R.; Heredia, A. Targeting of CDK9 with indirubin 3’-monoxime safely and durably reduces HIV viremia in chronically infected humanized mice. PLoS ONE 2017, 12, e0183425. [Google Scholar] [CrossRef]
- Heredia, A.; Le, N.; Gartenhaus, R.B.; Sausville, E.; Medina-Moreno, S.; Zapata, J.C.; Davis, C.; Gallo, R.C.; Redfield, R.R. Targeting of mTOR catalytic site inhibits multiple steps of the HIV-1 lifecycle and suppresses HIV-1 viremia in humanized mice. Proc. Natl. Acad. Sci. USA 2015, 112, 9412–9417. [Google Scholar] [CrossRef] [PubMed]
- Araínga, M.; Su, H.; Poluektova, L.Y.; Gorantla, S.; Gendelman, H.E. HIV-1 cellular and tissue replication patterns in infected humanized mice. Sci. Rep. 2016, 6, 23513. [Google Scholar] [CrossRef] [PubMed]
- Satheesan, S.; Li, H.; Burnett, J.C.; Takahashi, M.; Li, S.; Wu, S.X.; Synold, T.W.; Rossi, J.J.; Zhou, J. HIV replication and latency in a humanized NSG mouse model during suppressive oral combinational ART. J. Virol. 2018, 92, e02118-17. [Google Scholar] [CrossRef]
- Rochat, M.-A.; Schlaepfer, E.; Kuster, S.P.; Li, D.; Audige, A.; Ivic, S.; Fahrny, A.; Speck, R.F. Monitoring HIV DNA and cellular activation markers in HIV-infected humanized mice under cART. Virol. J. 2018, 15, 191. [Google Scholar] [CrossRef]
- Flórez-Álvarez, L.; Hernandez, J.C.; Zapata, W. NK Cells in HIV-1 Infection: From Basic Science to Vaccine Strategies. Front. Immunol. 2018, 9, 2290. [Google Scholar] [CrossRef] [PubMed]
- Ziegler-Heitbrock, L. Reprint of: Monocyte subsets in man and other species. Cell. Immunol. 2014, 289, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Orsini, G.; Legitimo, A.; Failli, A.; Massei, F.; Biver, P.; Consolini, R. Enumeration of human peripheral blood dendritic cells throughout the life. Int. Immunol. 2012, 24, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.P.; Patterson, S.; English, N.; Davies, D.; Knight, S.C.; Reid, C.D.L. Human peripheral blood contains two distinct lineages of dendritic cells. Eur. J. Immunol. 1999, 29, 2769–2778. [Google Scholar] [CrossRef]
- Förster, R.; Mattis, A.E.; Kremmer, E.; Wolf, E.; Brem, G.; Lipp, M. A putative chemokine receptor, BLR1, directs B cell migration to defined lymphoid organs and specific anatomic compartments of the spleen. Cell 1996, 87, 1037–1047. [Google Scholar] [CrossRef]
- Ansel, K.M.; Ngo, V.N.; Hyman, P.L.; Luther, S.A.; Förster, R.; Sedgwlck, J.D.; Browning, J.L.; Upp, M.; Cyster, J.G. A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature 2000, 406, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Fehniger, T.A.; Cooper, M.A.; Nuovo, G.J.; Cella, M.; Facchetti, F.; Colonna, M.; Caligiuri, M.A. CD56bright natural killer cells are present in human lymph nodes and are activated by T cell-derived IL-2: A potential new link between adaptive and innate immunity. Blood 2003, 101, 3052–3057. [Google Scholar] [CrossRef]
- Lanier, L.L.; Le, A.M.; Civin, C.I.; Loken, M.R.; Phillips, J.H. The relationship of CD16 (Leu-11) and Leu-19 (NKH-1) antigen expression on human peripheral blood NK cells and cytotoxic T lymphocytes. J. Immunol. 1986, 136, 4480–4486. [Google Scholar]
- Poli, A.; Michel, T.; Thérésine, M.; Andrès, E.; Hentges, F.; Zimmer, J. CD56brightnatural killer (NK) cells: An important NK cell subset. Immunology 2009, 126, 458–465. [Google Scholar] [CrossRef]
- Lopez-Vergès, S.; Milush, J.M.; Pandey, S.; York, V.A.; Arakawa-Hoyt, J.; Pircher, H.; Norris, P.J.; Nixon, D.F.; Lanier, L.L. CD57 defines a functionally distinct population of mature NK cells in the human CD56dimCD16+NK-cell subset. Blood 2010, 116, 3865–3874. [Google Scholar] [CrossRef]
- Mattei, F.; Schiavoni, G.; Belardelli, F.; Tough, D.F. IL-15 Is Expressed by Dendritic Cells in Response to Type I IFN, Double-Stranded RNA, or Lipopolysaccharide and Promotes Dendritic Cell Activation. J. Immunol. 2001, 167, 1179–1187. [Google Scholar] [CrossRef]
- Anguille, S.; Van Acker, H.H.; Van Den Bergh, J.; Willemen, Y.; Goossens, H.; Van Tendeloo, V.F.; Smits, E.L.; Berneman, Z.N.; Lion, E. Interleukin-15 dendritic cells harness NK cell cytotoxic effector function in a contact- and IL-15-dependent manner. PLoS ONE 2015, 10, e0123340. [Google Scholar] [CrossRef]
- Gasteiger, G.; Fan, X.; Dikiy, S.; Lee, S.Y.; Rudensky, A.Y. Tissue residency of innate lymphoid cells in lymphoid and nonlymphoid organs. Science 2015, 350, 981–985. [Google Scholar] [CrossRef]
- Ginhoux, F.; Jung, S. Monocytes and macrophages: Developmental pathways and tissue homeostasis. Nat. Rev. Immunol. 2014, 14, 392. [Google Scholar] [CrossRef]
- Herbst, B.; Köhler, G.; Mackensen, A.; Veelken, H.; Lindemann, A. GM-CSF promotes differentiation of a precursor cell of monocytes and langerhans-type dendritic cells from CD34+ haemopoietic progenitor cells. Br. J. Haematol. 1998, 101, 231–241. [Google Scholar] [CrossRef]
- Kingston, D.; Schmid, M.A.; Onai, N.; Obata-Onai, A.; Baumjohann, D.; Manz, M.G. The concerted action of GM-CSF and Flt3-ligand on in vivo dendritic cell homeostasis. Blood 2009, 114, 835–843. [Google Scholar] [CrossRef]
- MacDonald, K.P.A.; Munster, D.J.; Clark, G.J.; Dzionek, A.; Schmitz, J.; Hart, D.N.J. Characterization of human blood dendritic cell subsets. Blood 2002, 100, 4512–4520. [Google Scholar] [CrossRef] [PubMed]
- Grouard, G.; Rissoan, M.C.; Filgueira, L.; Durand, I.; Banchereau, J.; Liu, Y.J. The enigmatic plasmacytoid T cells develop into dendritic cells with interleukin (IL)-3 and CD40-ligand. J. Exp. Med. 1997, 185, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Cella, M.; Jarrossay, D.; Faccheth, F.; Alebardi, O.; Nakajima, H.; Lanzavecchia, A.; Colonna, M. Plasmacytoid monocytes migrate to inflamed lymph nodes and produce large amounts of type I interferon. Nat. Med. 1999, 5, 919. [Google Scholar] [CrossRef]
- Mckenna, H.J.; Stocking, K.L.; Miller, R.E.; Brasel, K.; De Smedt, T.; Maraskovsky, E.; Maliszewski, C.R.; Lynch, D.H.; Smith, J.; Pulendran, B.; et al. Mice lacking flt3 ligand have deficient hematopoiesis affecting hematopoietic progenitor cells, dendritic cells, and natural killer cells. Blood 2000, 95, 3489–3497. [Google Scholar] [PubMed]
- Masten, B.J.; Olson, G.K.; Kusewitt, D.F.; Lipscomb, M.F. Flt3 Ligand Preferentially Increases the Number of Functionally Active Myeloid Dendritic Cells in the Lungs of Mice. J. Immunol. 2004, 172, 4077–4083. [Google Scholar] [CrossRef]
- Waskow, C.; Liu, K.; Darrasse-Jèze, G.; Guermonprez, P.; Ginhoux, F.; Merad, M.; Shengelia, T.; Yao, K.; Nussenzweig, M. The receptor tyrosine kinase Flt3 is required for dendritic cell development in peripheral lymphoid tissues. Nat. Immunol. 2008, 9, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Collin, M.; Mcgovern, N.; Haniffa, M. Human dendritic cell subsets. Immunology 2013, 140, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Nie, C.; Sato, K.; Misawa, N.; Kitayama, H.; Fujino, H.; Hiramatsu, H.; Heike, T.; Nakahata, T.; Tanaka, Y.; Ito, M.; et al. Selective infection of CD4+ effector memory T lymphocytes leads to preferential depletion of memory T lymphocytes in R5 HIV-1-infected humanized NOD/SCID/IL-2Rγnull mice. Virology 2009, 394, 64–72. [Google Scholar] [CrossRef]
- Watanabe, S.; Terashima, K.; Ohta, S.; Horibata, S.; Yajima, M.; Shiozawa, Y.; Zahidunnabi Dewan, M.; Yu, Z.; Ito, M.; Morio, T.; et al. Hematopoietic stem cell-engrafted NOD/SCID/IL2Rγnullmice develop human lymphoid systems and induce long-lasting HIV-1 infection with specific humoral immune responses. Blood 2007, 109, 212–218. [Google Scholar] [CrossRef]
- Sato, K.; Nie, C.; Misawa, N.; Tanaka, Y.; Ito, M.; Koyanagi, Y. Dynamics of memory and naïve CD8+ T lymphocytes in humanized NOD/SCID/IL-2Rγnull mice infected with CCR5-tropic HIV-1. Vaccine 2010, 28 (Suppl. 2), B32–B37. [Google Scholar] [CrossRef]
- Février, M.; Dorgham, K.; Rebollo, A. CD4+T cell depletion in human immunodeficiency virus (HIV) infection: Role of apoptosis. Viruses 2011, 3, 586–612. [Google Scholar] [CrossRef] [PubMed]
- Cummins, N.W.; Badley, A.D. Mechanisms of HIV-associated lymphocyte apoptosis: 2010. Cell Death Dis. 2010, 1, e99. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chen, J.J.; Gelman, B.B.; Konig, R.; Cloyd, M.W. A novel mechanism of CD4 lymphocyte depletion involves effects of HIV on resting lymphocytes: Induction of lymph node homing and apoptosis upon secondary signaling through homing receptors. J. Immunol. 1999, 162, 268–276. [Google Scholar] [PubMed]
- Chen, J.J.-Y.; Huang, J.C.; Shirtliff, M.; Briscoe, E.; Ali, S.; Cesani, F.; Paar, D.; Cloyd, M.W. CD4 lymphocytes in the blood of HIV + individuals migrate rapidly to lymph nodes and bone marrow: Support for homing theory of CD4 cell depletion. J. Leukoc. Biol. 2002, 72, 271–278. [Google Scholar] [PubMed]
- Tedla, N.; Palladinetti, P.; Kelly, M.; Kumar, R.K.; DiGirolamo, N.; Chattophadhay, U.; Cooke, B.; Truskett, P.; Dwyer, J.; Wakefield, D.; et al. Chemokines and T lymphocyte recruitment to lymph nodes in HIV infection. Am. J. Pathol. 1996, 148, 1367. [Google Scholar] [PubMed]
- Cagigi, A.; Mowafi, F.; Phuong Dang, L.V.; Tenner-Racz, K.; Atlas, A.; Grutzmeier, S.; Racz, P.; Chiodi, F.; Nilsson, A. Altered expression of the receptor-ligand pair CXCR5/CXCL13 in B cells during chronic HIV-1 infection. Blood 2008, 112, 4401–4410. [Google Scholar] [CrossRef] [PubMed]
- Moir, S.; Malaspina, A.; Pickeral, O.K.; Donoghue, E.T.; Vasquez, J.; Miller, N.J.; Krishnan, S.R.; Planta, M.A.; Turney, J.F.; Justement, J.S.; et al. Decreased survival of B cells of HIV-viremic patients mediated by altered expression of receptors of the TNF superfamily. J. Exp. Med. 2004, 200, 587–599. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.; Moir, S.; Malaspina, A.; Howell, M.L.; Wang, W.; DiPoto, A.C.; O’Shea, M.A.; Roby, G.A.; Kwan, R.; Mican, J.M.; Chun, T.-W.; et al. Two overrepresented B cell populations in HIV-infected individuals undergo apoptosis by different mechanisms. Proc. Natl. Acad. Sci. USA 2006, 103, 19436–19441. [Google Scholar] [CrossRef] [PubMed]
- Perreau, M.; Savoye, A.-L.; De Crignis, E.; Corpataux, J.-M.; Cubas, R.; Haddad, E.K.; De Leval, L.; Graziosi, C.; Pantaleo, G. Follicular helper T cells serve as the major CD4 T cell compartment for HIV-1 infection, replication, and production. J. Exp. Med. 2013, 210, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Pallikkuth, S.; Sharkey, M.; Babic, D.Z.; Gupta, S.; Stone, G.W.; Fischl, M.A.; Stevenson, M.; Pahwa, S. Peripheral T Follicular Helper Cells Are the Major HIV Reservoir within Central Memory CD4 T Cells in Peripheral Blood from Chronically HIV-Infected Individuals on Combination Antiretroviral Therapy. J. Virol. 2016, 90, 2718–2728. [Google Scholar] [CrossRef] [PubMed]
- Boswell, K.L.; Paris, R.; Boritz, E.; Ambrozak, D.; Yamamoto, T.; Darko, S.; Wloka, K.; Wheatley, A.; Narpala, S.; McDermott, A.; et al. Loss of Circulating CD4 T Cells with B Cell Helper Function during Chronic HIV Infection. PLoS Pathog. 2014, 10, e1003853. [Google Scholar] [CrossRef] [PubMed]
- Muema, D.M.; Macharia, G.N.; Olusola, B.A.; Hassan, A.S.; Fegan, G.W.; Berkley, J.A.; Urban, B.C.; Nduati, E.W. Proportions of circulating follicular helper T cells are reduced and correlate with memory B cells in HIV-infected children. PLoS ONE 2017, 12, e0175570. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, M.; van Lunzen, J.; Soghoian, D.Z.; Kuhl, B.D.; Ranasinghe, S.; Kranias, G.; Flanders, M.D.; Cutler, S.; Yudanin, N.; Muller, M.I.; et al. Expansion of HIV-specific T follicular helper cells in chronic HIV infection. J. Clin. Investig. 2012, 122, 3271–3280. [Google Scholar] [CrossRef] [PubMed]
- Perdomo-Celis, F.; Taborda, N.A.; Rugeles, M.T. Circulating CXCR5-expressing CD8+T-Cells are Major Producers of IL-21 and Associate with Limited HIV Replication. J. Acquir. Immune Defic. Syndr. 2018, 78, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Petrovas, C.; Ferrando-Martinez, S.; Gerner, M.Y.; Casazza, J.P.; Pegu, A.; Deleage, C.; Cooper, A.; Hataye, J.; Andrews, S.; Ambrozak, D.; et al. H I V Follicular CD8 T cells accumulate in HIV infection and can kill infected cells in vitro via bispecific antibodies. Sci. Transl. Med. 2017, 9, 1–14. [Google Scholar] [CrossRef]
- Chen, P.; Su, B.; Zhang, T.; Zhu, X.; Xia, W.; Fu, Y.; Zhao, G.; Xia, H.; Dai, L.; Sun, L.; et al. Perturbations of monocyte subsets and their association with T helper cell differentiation in acute and chronic HIV-1-infected patients. Front. Immunol. 2017, 8, 272. [Google Scholar] [CrossRef]
- Neil, S.; Martin, F.; Ikeda, Y.; Collins, M. Postentry Restriction to Human Immunodeficiency Virus-Based Vector Transduction in Human Monocytes. J. Virol. 2001, 75, 5448–5456. [Google Scholar] [CrossRef] [PubMed]
- Arfi, V.; Riviere, L.; Jarrosson-Wuilleme, L.; Goujon, C.; Rigal, D.; Darlix, J.-L.; Cimarelli, A. Characterization of the Early Steps of Infection of Primary Blood Monocytes by Human Immunodeficiency Virus Type 1. J. Virol. 2008, 82, 6557–6565. [Google Scholar] [CrossRef] [PubMed]
- Shive, C.L.; Jiang, W.; Anthony, D.D.; Lederman, M.M. Soluble CD14 is a nonspecific marker of monocyte activation. AIDS 2015, 29, 1263–1265. [Google Scholar] [CrossRef] [PubMed]
- Sonza, S.; Mutimer, H.P.; Oelrichs, R.; Jardine, D.; Harvey, K.; Dunne, A.; Purcell, D.F.; Birch, C.; Crowe, S.M. Monocytes harbour replication-competent, non-latent HIV-1 in patients on highly active antiretroviral therapy. AIDS 2001, 15, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, J.; Coutlée, F.; Tremblay, C.; Routy, J.; Poudrier, J.; Roger, M. HIV Infection Affects Blood Myeloid Dendritic Cells after Successful Therapy and despite Nonprogressing Clinical Disease. J. Infect. Dis. 2009, 199, 1007–1018. [Google Scholar] [CrossRef] [PubMed]
- Pacanowski, J.; Kahi, S.; Baillet, M.; Lebon, P.; Deveau, C.; Goujard, C.; Meyer, L.; Oksenhendler, E.; Sinet, M.; Hosmalin, A. Reduced blood CD123+(lymphoid) and CD11c+(myeloid) dendritic cell numbers in primary HIV-1 infection. Blood 2001, 98, 3016–3021. [Google Scholar] [CrossRef] [PubMed]
- Iannetta, M.; Savinelli, S.; Rossi, R.; Mascia, C.; Marocco, R.; Vita, S.; Zuccalà, P.; Zingaropoli, M.A.; Mengoni, F.; Massetti, A.P.; et al. Myeloid and lymphoid activation markers in AIDS and non-AIDS presenters. Immunobiology 2018, S0171-298, 30214–30216. [Google Scholar] [CrossRef] [PubMed]
- Barron, M.A.; Blyveis, N.; Palmer, B.E.; MaWhinney, S.; Wilson, C.C. Influence of plasma viremia on defects in number and immunophenotype of blood dendritic cell subsets in human immunodeficiency virus 1-infected individuals. J. Infect. Dis. 2003, 18, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Pinzon-Charry, A.; Ho, C.S.K.; Laherty, R.; Maxwell, T.; Walker, D.; Gardiner, R.A.; O’Connor, L.; Pyke, C.; Schmidt, C.; Furnival, C.; et al. A Population of HLA-DR+ Immature Cells Accumulates in the Blood Dendritic Cell Compartment of Patients with Different Types of Cancer. Neoplasia 2005, 7, 1112–1122. [Google Scholar] [CrossRef]
- Donaghy, H.; Gazzard, B.; Gotch, F.; Patterson, S. Dysfunction and infection of freshly isolated blood myeloid and plasmacytoid dendritic cells in patients infected with HIV-1. Blood 2003, 101, 4505–4511. [Google Scholar] [CrossRef] [PubMed]
- Tarazona, R.; Casado, J.G.; Delarosa, O.; Torre-Cisneros, J.; Villanueva, J.L.; Sanchez, B.; Galiani, M.D.; Gonzalez, R.; Solana, R.; Pena, J. Selective depletion of CD56dimNK cell subsets and maintenance of CD56brightNK cells in treatment-naive HIV-1-seropositive individuals. J. Clin. Immunol. 2002, 22, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.S.; Ahmad, F.; Eberhard, J.M.; Bhatnagar, N.; Bollmann, B.A.; Keudel, P.; Ballmaier, M.; Zielinska-Skowronek, M.; Schmidt, R.E.; Meyer-Olson, D. Loss of CCR7 Expression on CD56bright NK Cells Is Associated with a CD56dimCD16+ NK Cell-Like Phenotype and Correlates with HIV Viral Load. PLoS ONE 2012, 7, e44820. [Google Scholar] [CrossRef]
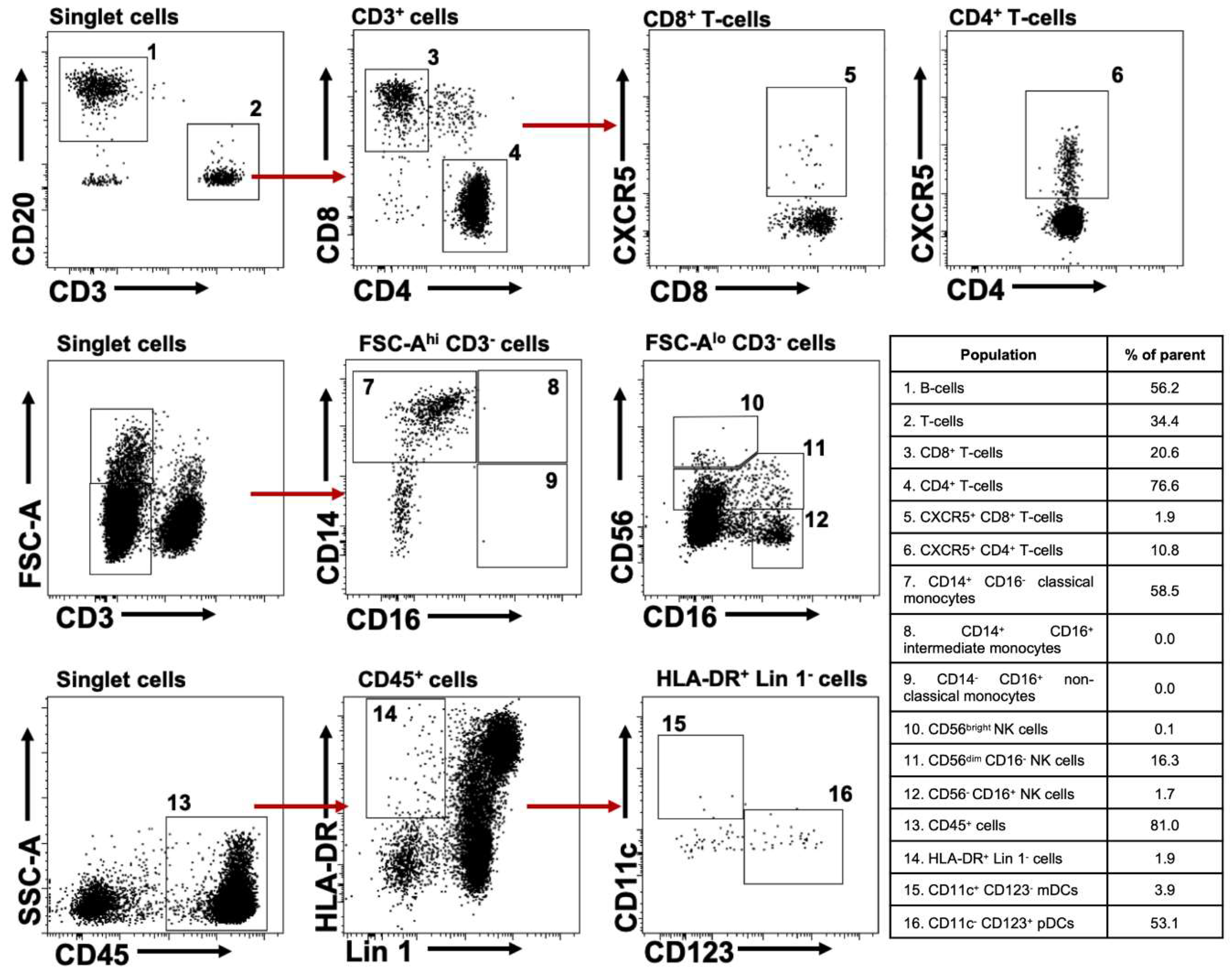

| Correlations Plasma Viral Load vs. Cell Populations | Spearman Test | ||
|---|---|---|---|
| Compartment 1 | Population | rho | p Value |
| Blood | CD45+ cells | −0.89 | 0.0005 |
| Blood | CD3+ T-cells | −0.3 | 0.1 |
| Blood | CD20+ B-cells | −0.85 | 0.02 |
| Blood | CD4:CD8 ratio | −0.66 | 0.02 |
| Spleen | CXCR5+ CD8+ T-cells | 0.92 | 0.02 |
| Blood | HLA-DR+ Lin 1− cells | 0.91 | 0.0004 |
| Axillary lymph node | HLA-DR+ Lin 1− cells | −0.73 | 0.07 |
| Blood | CD14+ CD16− classical monocytes | −0.88 | 0.001 |
| Blood | CD11c− CD123+ pDCs | −0.87 | 0.002 |
| Blood | CD56bright NK cells | −0.72 | 0.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perdomo-Celis, F.; Medina-Moreno, S.; Davis, H.; Bryant, J.; Zapata, J.C. HIV Replication in Humanized IL-3/GM-CSF-Transgenic NOG Mice. Pathogens 2019, 8, 33. https://doi.org/10.3390/pathogens8010033
Perdomo-Celis F, Medina-Moreno S, Davis H, Bryant J, Zapata JC. HIV Replication in Humanized IL-3/GM-CSF-Transgenic NOG Mice. Pathogens. 2019; 8(1):33. https://doi.org/10.3390/pathogens8010033
Chicago/Turabian StylePerdomo-Celis, Federico, Sandra Medina-Moreno, Harry Davis, Joseph Bryant, and Juan C. Zapata. 2019. "HIV Replication in Humanized IL-3/GM-CSF-Transgenic NOG Mice" Pathogens 8, no. 1: 33. https://doi.org/10.3390/pathogens8010033
APA StylePerdomo-Celis, F., Medina-Moreno, S., Davis, H., Bryant, J., & Zapata, J. C. (2019). HIV Replication in Humanized IL-3/GM-CSF-Transgenic NOG Mice. Pathogens, 8(1), 33. https://doi.org/10.3390/pathogens8010033





