Zika Virus Trafficking and Interactions in the Human Male Reproductive Tract
Abstract
1. Introduction
2. A Brief Historical Perspective of ZIKV Sexual Transmission
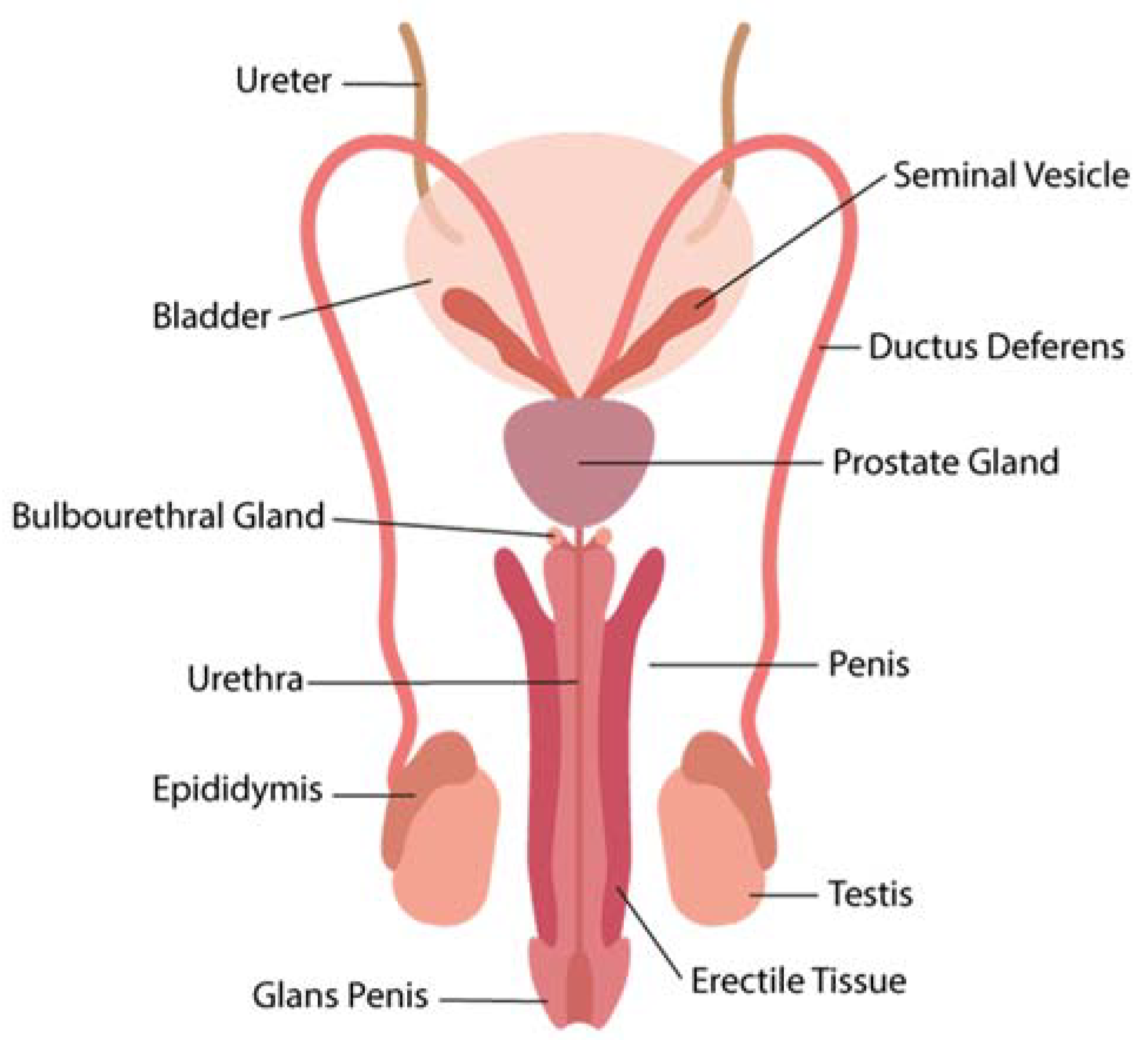
3. Interactions of ZIKV in the Male Reproductive Tract
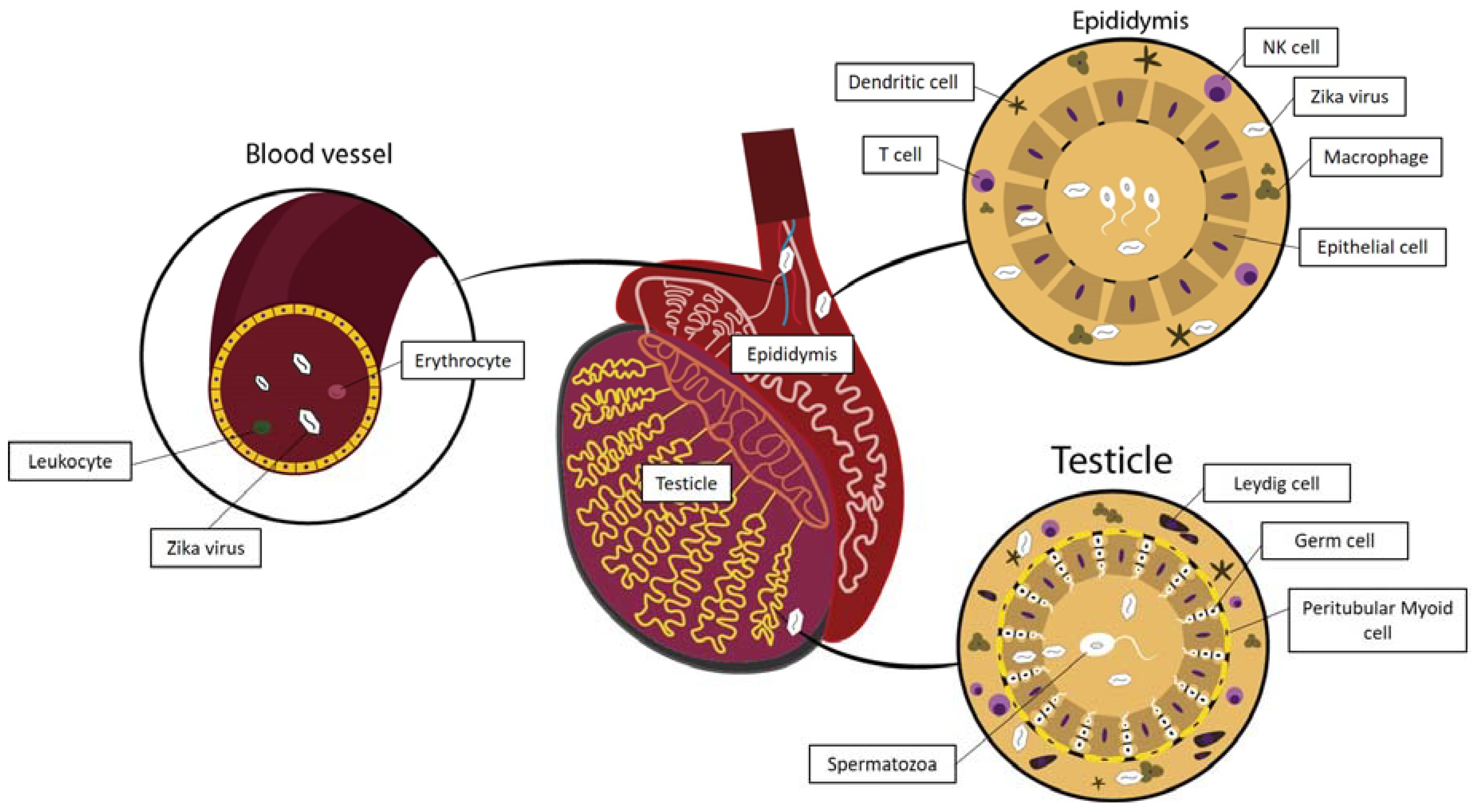
3.1. Zika Virus Interactions in the Testis
3.1.1. Zika Virus and the Hematotesticular Barrier
3.1.2. Zika Virus Interactions with Cells Present in the Testicular Interstitial Spaces
3.1.3. Zika Virus Interactions in the Peritubular Space and the Seminiferous Tubule Wall
3.2. Zika Virus in the Epididymis
3.3. Zika Virus in the Prostate Gland and in the Seminal Vesicle
3.4. Zika Virus in Semen
4. Conclusions
Conflicts of Interest
References
- Stekler, J.; Sycks, B.J.; Holte, S.; Maenza, J.; Stevens, C.E.; Dragavon, J.; Collier, A.C.; Coombs, R.W. HIV Dynamics in Seminal Plasma during Primary HIV Infection. AIDS Res. Hum. Retrovir. 2008, 24, 1269–1274. [Google Scholar] [CrossRef] [PubMed]
- Salam, A.P.; Horby, P.W. The breadth of viruses in human semen. Emerg. Infect. Dis. 2017, 23, 1922–1924. [Google Scholar] [CrossRef] [PubMed]
- Dejucq-Rainsford, N.; Jegou, B. Viruses in Semen and Male Genital Tissues—Consequences for the Reproductive System and Therapeutic Perspectives. Curr. Pharm. Des. 2004, 10, 557–575. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.; Walker, C.; Herrington, E.; Lewis, J.A.; McCormick, J.; Beasley, D.W.C.; Tesh, R.B.; Fisher-Hoch, S. Persistent Infection with West Nile Virus Years after Initial Infection. J. Infect. Dis. 2010, 201, 2–4. [Google Scholar] [CrossRef] [PubMed]
- Bandeira, A.C.; Campos, G.S.; Rocha, V.F.; Souza, B.S.; Soares, M.B.; Oliveira, A.A.; Abreu, Y.C.; Menezes, G.S.; Sardi, S. Prolonged shedding of Chikungunya virus in semen and urine: A new perspective for diagnosis and implications for transmission. IDCases 2016, 6, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Bandeira, A.C.; Campos, G.S.; Sardi, S.I.; Rocha, V.F.D.; Rocha, G.C.M. Neonatal encephalitis due to Chikungunya vertical transmission: First report in Brazil. IDCases 2016, 5, 57–59. [Google Scholar] [CrossRef] [PubMed]
- Dejucq, N.; Jégou, B. Viruses in the Mammalian Male Genital Tract and Their Effects on the Reproductive System. Microbiol. Mol. Biol. Rev. 2001, 65, 208–231. [Google Scholar] [CrossRef] [PubMed]
- Adefuye, A.O.; Adeola, H.A.; Sales, K.J.; Katz, A.A. Seminal Fluid-Mediated Inflammation in Physiology and Pathology of the Female Reproductive Tract. J. Immunol. Res. 2016, 2016, 9707252. [Google Scholar] [CrossRef] [PubMed]
- Mate, S.E.; Kugelman, J.R.; Nyenswah, T.G.; Ladner, J.T.; Wiley, M.R.; Cordier-Lassalle, T.; Christie, A.; Schroth, G.P.; Gross, S.M.; Davies-Wayne, G.J.; et al. Molecular Evidence of Sexual Transmission of Ebola Virus. N. Engl. J. Med. 2015, 373, 2448–2454. [Google Scholar] [CrossRef] [PubMed]
- Deen, G.F.; Broutet, N.; Xu, W.; Knust, B.; Sesay, F.R.; McDonald, S.L.R.; Ervin, E.; Marrinan, J.E.; Gaillard, P.; Habib, N.; et al. Ebola RNA Persistence in Semen of Ebola Virus Disease Survivors—Preliminary Report. N. Engl. J. Med. 2017, 377, 1428–1437. [Google Scholar] [CrossRef] [PubMed]
- Lowe, R.; Barcellos, C.; Brasil, P.; Cruz, O.G.; Honório, N.A.; Kuper, H.; Carvalho, M.S. The Zika Virus Epidemic in Brazil: From Discovery to Future Implications. Int. J. Environ. Res. Public Health 2018, 15, E96. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, L.R.C.; de Souza, A.M.; da Silva, L.R.C.; de Souza, A.M.; da Silva, L.R.C.; de Souza, A.M. Zika virus: What do we know about the viral structure, mechanisms of transmission, and neurological outcomes? Rev. Soc. Bras. Med. Trop. 2016, 49, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Rowland, A.; Washington, C.I.; Sheffield, J.S.; Pardo-Villamizar, C.A.; Segars, J.H. Zika virus infection in semen: A call to action and research. J. Assist. Reprod. Genet. 2016, 33, 435–437. [Google Scholar] [CrossRef] [PubMed]
- Tu-Xuan Nhan, D.M. Emergence of Zika Virus. Clin. Microbiol. Open Access 2015, 4, 222. [Google Scholar] [CrossRef]
- Petersen, E.; Wilson, M.E.; Touch, S.; McCloskey, B.; Mwaba, P.; Bates, M.; Dar, O.; Mattes, F.; Kidd, M.; Ippolito, G.; et al. Rapid Spread of Zika Virus in The Americas—Implications for Public Health Preparedness for Mass Gatherings at the 2016 Brazil Olympic Games. Int. J. Infect. Dis. 2016, 44, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Foy, B.D.; Kobylinski, K.C.; Foy, J.L.C.; Blitvich, B.J.; da Rosa, A.T.; Haddow, A.D.; Lanciotti, R.S.; Tesh, R.B. Probable Non-Vector-borne Transmission of Zika Virus, Colorado, USA. Emerg. Infect. Dis. 2011, 17, 880–882. [Google Scholar] [CrossRef] [PubMed]
- Brooks, R.B.; Carlos, M.P.; Myers, R.A.; White, W.G.; Bobo-Lenoci, T.; Aplan, D.; Blythe, D.; Feldman, K.A. Likely Sexual Transmission of Zika Virus from a Man with No Symptoms of Infection—Maryland. MMWR. Morb. Mortal. Wkly. Rep. 2016, 65, 915–916. [Google Scholar] [CrossRef] [PubMed]
- Baud, D.; Musso, D.; Vouga, M.; Alves, M.P.; Vulliemoz, N. Zika virus: A new threat to human reproduction. Am. J. Reprod. Immunol. 2017, 77, e12614. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, B.; Graham, V.; Miles, R.W.; Lewandowski, K.; Dowall, S.D.; Pullan, S.T.; Hewson, R. Complete Genome Sequence of Zika Virus Isolated from Semen. Genome Announc. 2016, 4, e01116-16. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Montalvá, A.; Pou, D.; Sulleiro, E.; Salvador, F.; Bocanegra, C.; Treviño, B.; Rando, A.; Serre, N.; Pumarola, T.; Almirante, B.; et al. Zika virus dynamics in body fluids and risk of sexual transmission in a non-endemic area. Trop. Med. Int. Health 2018, 23, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Zambrano, H.; Waggoner, J.; León, K.; Pinsky, B.; Vera, K.; Schettino, M.; Rivera, L.; Landivar, J.; Granda, M.; Lee, A.; et al. High incidence of Zika virus infection detected in plasma and cervical cytology specimens from pregnant women in Guayaquil, Ecuador. Am. J. Reprod. Immunol. 2017, 77. [Google Scholar] [CrossRef] [PubMed]
- Harrower, J.; Kiedrzynski, T.; Baker, S.; Upton, A.; Rahnama, F.; Sherwood, J.; Huang, Q.S.; Todd, A.; Pulford, D. Sexual Transmission of Zika Virus and Persistence in Semen, New Zealand, 2016. Emerg. Infect. Dis. 2016, 22, 1855–1857. [Google Scholar] [CrossRef] [PubMed]
- Turmel, J.M.; Abgueguen, P.; Hubert, B.; Vandamme, Y.M.; Maquart, M.; Le Guillou-Guillemette, H.; Leparc-Goffart, I. Late sexual transmission of Zika virus related to persistence in the semen. Lancet 2016, 387, 2501. [Google Scholar] [CrossRef]
- Couto-Lima, D.; Madec, Y.; Bersot, M.I.; Campos, S.S.; Motta, M.A.; Santos, F.B.D.; Vazeille, M.; Vasconcelos, P.F.D.C.; Lourenço-de-Oliveira, R.; Failloux, A.B. Potential risk of re-emergence of urban transmission of Yellow Fever virus in Brazil facilitated by competent Aedes populations. Sci. Rep. 2017, 7, 4848. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, C.M.; Di Paola, N.; Cunha, M.P.; Rodrigues-Jesus, M.J.; Araujo, D.B.; Silveira, V.B.; Leal, F.B.; Mesquita, F.S.; Botosso, V.F.; Zanotto, P.M.A.; et al. Yellow fever virus RNA in urine and semen of convalescent patient, Brazil. Emerg. Infect. Dis. 2018, 24, 176–178. [Google Scholar] [CrossRef] [PubMed]
- Venturi, G.; Zammarchi, L.; Fortuna, C.; Remoli, M.E.; Benedetti, E.; Fiorentini, C.; Trotta, M.; Rizzo, C.; Mantella, A.; Rezza, G.; et al. An autochthonous case of Zika due to possible sexual transmission, Florence, Italy, 2014. Euro Surveill. 2016, 21, 30148. [Google Scholar] [CrossRef] [PubMed]
- Musso, D.; Roche, C.; Robin, E.; Nhan, T.; Teissier, A.; Cao-Lormeau, V.M. Potential Sexual Transmission of Zika Virus. Emerg. Infect. Dis. 2015, 21, 2013–2015. [Google Scholar] [CrossRef] [PubMed]
- Mansuy, J.M.; Dutertre, M.; Mengelle, C.; Fourcade, C.; Marchou, B.; Delobel, P.; Izopet, J.; Martin-Blondel, G. Zika virus: High infectious viral load in semen, a new sexually transmitted pathogen? Lancet Infect. Dis. 2016, 16, 405. [Google Scholar] [CrossRef]
- Atkinson, B.; Hearn, P.; Afrough, B.; Lumley, S.; Carter, D.; Aarons, E.J.; Simpson, A.J.; Brooks, T.J.; Hewson, R. Detection of Zika Virus in Semen. Emerg. Infect. Dis. 2016, 22, 940. [Google Scholar] [CrossRef] [PubMed]
- Musso, D.; Richard, V.; Teissier, A.; Stone, M.; Lanteri, M.C.; Latoni, G.; Alsina, J.; Reik, R.; Busch, M.P. Detection of Zika virus RNA in semen of asymptomatic blood donors. Clin. Microbiol. Infect. 2017, 23, 1001.e1–1001.e3. [Google Scholar] [CrossRef] [PubMed]
- Deckard, D.T.; Chung, W.M.; Brooks, J.T.; Smith, J.C.; Woldai, S.; Hennessey, M.; Kwit, N.; Mead, P. Male-to-Male Sexual Transmission of Zika Virus—Texas, January 2016. MMWR Morb. Mortal. Wkly. Rep. 2016, 65, 372–374. [Google Scholar] [CrossRef] [PubMed]
- Prisant, N.; Bujan, L.; Benichou, H.; Hayot, P.-H.; Pavili, L.; Lurel, S.; Herrmann, C.; Janky, E.; Joguet, G. Zika virus in the female genital tract. Lancet Infect. Dis. 2016, 16, 1000–1001. [Google Scholar] [CrossRef]
- Davidson, A.; Slavinski, S.; Komoto, K.; Rakeman, J.; Weiss, D. Suspected Female-to-Male Sexual Transmission of Zika Virus—New York City, 2016. MMWR. Morb. Mortal. Wkly. Rep. 2016, 65, 716–717. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, M.G.; Costa, M.d.N.; de Oliveira, W.K.; Nunes, M.L.; Rodrigues, L.C. The epidemic of Zika virus-related microcephaly in Brazil: Detection, control, etiology, and future scenarios. Am. J. Public Health 2016, 106, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.R.; Chen, T.-H.; Hancock, T.; Powers, A.M.; Kool, J.L.; Lanciotti, R.S.; Pretrick, M.; Marfel, M.; Holzbauer, S.; Dubray, C.; et al. Zika Virus Outbreak on Yap Island, Federated States of Micronesia. N. Engl. J. Med. 2009, 360, 2536–2543. [Google Scholar] [CrossRef] [PubMed]
- Visseaux, B.; Mortier, E.; Houhou-Fidouh, N.; Brichler, S.; Collin, G.; Larrouy, L.; Charpentier, C.; Descamps, D. Zika virus in the female genital tract. Lancet Infect. Dis. 2016, 16, 1220. [Google Scholar] [CrossRef]
- Murray, K.O.; Gorchakov, R.; Carlson, A.R.; Berry, R.; Lai, L.; Natrajan, M.; Garcia, M.N.; Correa, A.; Patel, S.M.; Aagaard, K.; et al. Prolonged Detection of Zika Virus in Vaginal Secretions and Whole Blood. Emerg. Infect. Dis. 2017, 23, 99–101. [Google Scholar] [CrossRef] [PubMed]
- Moreira, J.; Peixoto, T.M.; Siqueira, A.M.; Lamas, C.C. Sexually acquired Zika virus: A systematic review. Clin. Microbiol. Infect. 2017, 23, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Matheron, S.; D’Ortenzio, E.; Leparc-Goffart, I.; Hubert, B.; de Lamballerie, X.; Yazdanpanah, Y. Long-Lasting Persistence of Zika Virus in Semen. Clin. Infect. Dis. 2016, 63, ciw509. [Google Scholar] [CrossRef] [PubMed]
- Grischott, F.; Puhan, M.; Hatz, C.; Schlagenhauf, P. Non-vector-borne transmission of Zika virus: A systematic review. Travel Med. Infect. Dis. 2016, 14, 313–330. [Google Scholar] [CrossRef] [PubMed]
- Frank, C.; Cadar, D.; Schlaphof, A.; Neddersen, N.; Günther, S.; Schmidt-Chanasit, J.; Tappe, D. Sexual transmission of Zika virus in Germany, April 2016. Euro Surveill. 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Hills, S.L.; Russell, K.; Hennessey, M.; Williams, C.; Oster, A.M.; Fischer, M.; Mead, P. Transmission of Zika Virus Through Sexual Contact with Travelers to Areas of Ongoing Transmission—Continental United States, 2016. MMWR. Morb. Mortal. Wkly. Rep. 2016, 65, 215–216. [Google Scholar] [CrossRef] [PubMed]
- Katanami, Y.; Kutsuna, S.; Taniguchi, S.; Tajima, S.; Takaya, S.; Yamamoto, K.; Takeshita, N.; Hayakawa, K.; Kanagawa, S.; Kato, Y.; et al. Detection of Zika virus in a traveller from Vietnam to Japan. J. Travel Med. 2017, 24, 24–27. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Korhonen, E.M.; Huhtamo, E.; Smura, T.; Kallio-Kokko, H.; Raassina, M.; Vapalahti, O. Zika virus infection in a traveller returning from the Maldives, June 2015. Euro Surveill. 2016, 21, 30107. [Google Scholar] [CrossRef] [PubMed]
- Tappe, D.; Zammarchi, L.; Fortuna, C.; Remoli, M.E.; Günther, S.; Venturi, G.; Bartoloni, A.; Schmidt-Chanasit, J. Zika virus infection in a traveler returning to Europe from Brazil, March 2015. Euro Surveill. 2015, 20, 21153. [Google Scholar]
- Citil Dogan, A.; Wayne, S.; Bauer, S.; Ogunyemi, D.; Kulkharni, S.K.; Maulik, D.; Carpenter, C.F.; Bahado-Singh, R.O. The Zika virus and pregnancy: Evidence, management, and prevention. J. Matern. Neonatal Med. 2017, 30, 386–396. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Russell, K.; Oliver, S.E.; Lewis, L.; Barfield, W.D.; Cragan, J.; Meaney-Delman, D.; Staples, J.E.; Fischer, M.; Peacock, G.; Oduyebo, T.; et al. Update: Interim Guidance for the Evaluation and Management of Infants with Possible Congenital Zika Virus Infection—United States, August 2016. MMWR Morb. Mortal. Wkly. Rep. 2016, 65, 870–878. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Wang, T.; Han, D. Structural, cellular and molecular aspects of immune privilege in the testis. Front. Immunol. 2012, 3, 152. [Google Scholar] [CrossRef] [PubMed]
- Loveland, K.L.; Klein, B.; Pueschl, D.; Indumathy, S.; Bergmann, M.; Loveland, B.E.; Hedger, M.P.; Schuppe, H.-C. Cytokines in male fertility and reproductive pathologies: Immunoregulation and beyond. Front. Endocrinol. (Lausanne) 2017, 8, 307. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Deng, T.; Han, D. Testicular immunoregulation and spermatogenesis. Semin. Cell Dev. Biol. 2016, 59, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.V.; Kafka, J.K.; Ferreira, V.H.; Roth, K.; Kaushic, C. Innate and adaptive immune responses in male and female reproductive tracts in homeostasis and following HIV infection. Cell. Mol. Immunol. 2014, 11, 410–427. [Google Scholar] [CrossRef] [PubMed]
- Jenabian, M.A.; Costiniuk, C.T.; Mehraj, V.; Ghazawi, F.M.; Fromentin, R.; Brousseau, J.; Brassard, P.; Bélanger, M.; Ancuta, P.; Bendayan, R.; et al. Immune tolerance properties of the testicular tissue as a viral sanctuary site in ART-treated HIV-infected adults. AIDS 2016, 30, 2777–2786. [Google Scholar] [CrossRef] [PubMed]
- Kalkeri, R.; Murthy, K.K. Zika virus reservoirs: Implications for transmission, future outbreaks, drug and vaccine development. F1000Research 2017, 6, 1850. [Google Scholar] [CrossRef] [PubMed]
- Barzon, L.; Pacenti, M.; Franchin, E.; Lavezzo, E.; Trevisan, M.; Sgarabotto, D.; Palù, G. Infection dynamics in a traveller with persistent shedding of Zika virus RNA in semen for six months after returning from Haiti to Italy, January 2016. Euro Surveill. 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Joguet, G.; Mansuy, J.-M.; Matusali, G.; Hamdi, S.; Walschaerts, M.; Pavili, L.; Guyomard, S.; Prisant, N.; Lamarre, P.; Dejucq-Rainsford, N.; et al. Effect of acute Zika virus infection on sperm and virus clearance in body fluids: A prospective observational study. Lancet Infect. Dis. 2017, 17, 1200–1208. [Google Scholar] [CrossRef]
- Barzon, L.; Lavezzo, E.; Palù, G. Zika virus infection in semen: Effect on human reproduction. Lancet Infect. Dis. 2017, 17, 1107–1109. [Google Scholar] [CrossRef]
- Paz-Bailey, G.; Rosenberg, E.S.; Doyle, K.; Munoz-Jordan, J.; Santiago, G.A.; Klein, L.; Perez-Padilla, J.; Medina, F.A.; Waterman, S.H.; Gubern, C.G.; et al. Persistence of Zika Virus in Body Fluids—Preliminary Report. N. Engl. J. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Lustig, Y.; Mendelson, E.; Paran, N.; Melamed, S.; Schwartz, E. Detection of Zika virus RNA in whole blood of imported Zika virus disease cases up to 2 months after symptom onset. Eurosurveillance 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Mansuy, J.M.; Mengelle, C.; Pasquier, C.; Chapuy-Regaud, S.; Delobel, P.; Martin-Blondel, G.; Izopet, J. Zika Virus Infection and Prolonged Viremia in Whole-Blood Specimens. Emerg. Infect. Dis. 2017, 23, 863–865. [Google Scholar] [CrossRef] [PubMed]
- Froeschl, G.; Huber, K.; von Sonnenburg, F.; Nothdurft, H.-D.; Bretzel, G.; Hoelscher, M.; Zoeller, L.; Trottmann, M.; Pan-Montojo, F.; Dobler, G.; et al. Long-term kinetics of Zika virus RNA and antibodies in body fluids of a vasectomized traveller returning from Martinique: A case report. BMC Infect. Dis. 2017, 17, 55. [Google Scholar] [CrossRef] [PubMed]
- Rios, M.; Daniel, S.; Chancey, C.; Hewlett, I.K.; Stramer, S.L. West Nile Virus Adheres to Human Red Blood Cells in Whole Blood. Clin. Infect. Dis. 2007, 45, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Lanteri, M.C.; Lee, T.H.; Wen, L.; Kaidarova, Z.; Bravo, M.D.; Kiely, N.E.; Kamel, H.T.; Tobler, L.H.; Norris, P.J.; Busch, M.P. West Nile virus nucleic acid persistence in whole blood months after clearance in plasma: Implication for transfusion and transplantation safety. Transfusion 2014, 54, 3232–3241. [Google Scholar] [CrossRef] [PubMed]
- Lustig, Y.; Mannasse, B.; Koren, R.; Katz-Likvornik, S.; Hindiyeh, M.; Mandelboim, M.; Dovrat, S.; Sofer, D.; Mendelson, E. Superiority of west nile virus RNA detection in whole blood for diagnosis of acute infection. J. Clin. Microbiol. 2016, 54, 2294–2297. [Google Scholar] [CrossRef] [PubMed]
- Klungthong, C.; Gibbons, R.V.; Thaisomboonsuk, B.; Nisalak, A.; Kalayanarooj, S.; Thirawuth, V.; Nutkumhang, N.; Mammen, M.P., Jr.; Jarman, R.G. Dengue Virus Detection Using Whole Blood for Reverse Transcriptase PCR and Virus Isolation. J. Clin. Microbiol. 2007, 45, 2480–2485. [Google Scholar] [CrossRef] [PubMed]
- Musso, D.; Nhan, T.; Robin, E.; Roche, C.; Bierlaire, D.; Zisou, K.; Shan Yan, A.; Cao-Lormeau, V.M.; Broult, J. Potential for Zika virus transmission through blood transfusion demonstrated during an outbreak in French Polynesia, November 2013 to February 2014. Euro Surveill. 2014, 19, 20761. [Google Scholar] [CrossRef] [PubMed]
- Barjas-Castro, M.L.; Angerami, R.N.; Cunha, M.S.; Suzuki, A.; Nogueira, J.S.; Rocco, I.M.; Maeda, A.Y.; Vasami, F.G.; Katz, G.; Boin, I.F. Probable transfusion-transmitted Zika virus in Brazil. Transfusion 2016, 56, 1684–1688. [Google Scholar] [CrossRef] [PubMed]
- De Noronha, L.; Zanluca, C.; Azevedo, M.L.V.; Luz, K.G.; Santos, C.N.D.d. Zika virus damages the human placental barrier and presents marked fetal neurotropism. Mem. Inst. Oswaldo Cruz 2016, 111, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Tabata, T.; Petitt, M.; Puerta-Guardo, H.; Michlmayr, D.; Wang, C.; Fang-Hoover, J.; Harris, E.; Pereira, L. Zika Virus Targets Different Primary Human Placental Cells, Suggesting Two Routes for Vertical Transmission. Cell Host Microbe 2016, 20, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; DeLalio, L.J.; Isakson, B.E.; Wang, T.T. AXL-Mediated Productive Infection of Human Endothelial Cells by Zika VirusNovelty and Significance. Circ. Res. 2016, 119, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Mladinich, M.C.; Schwedes, J.; Mackow, E.R. Zika Virus Persistently Infects and Is Basolaterally Released from Primary Human Brain Microvascular Endothelial Cells. mBio 2017, 8, e00952-17. [Google Scholar] [CrossRef] [PubMed]
- Modhiran, N.; Watterson, D.; Muller, D.A.; Panetta, A.K.; Sester, D.P.; Liu, L.; Hume, D.A.; Stacey, K.J.; Young, P.R. Dengue virus NS1 protein activates cells via Toll-like receptor 4 and disrupts endothelial cell monolayer integrity. Sci. Transl. Med. 2015, 7, 304ra142. [Google Scholar] [CrossRef] [PubMed]
- Dalrymple, N.A.; Mackow, E.R. Virus interactions with endothelial cell receptors: Implications for viral pathogenesis. Curr. Opin. Virol. 2014, 7, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Wang, R.; Gao, F.; Li, M.; Liu, J.; Wang, J.; Hong, W.; Zhao, L.; Wen, Y.; Yin, C.; et al. Delineating antibody recognition against Zika virus during natural infection. JCI Insight 2017, 2, 93042. [Google Scholar] [CrossRef] [PubMed]
- Hastings, A.K.; Yockey, L.J.; Jagger, B.W.; Hwang, J.; Uraki, R.; Gaitsch, H.F.; Parnell, L.A.; Cao, B.; Mysorekar, I.U.; Rothlin, C.V.; et al. TAM Receptors Are Not Required for Zika Virus Infection in Mice. Cell Rep. 2017, 19, 558–568. [Google Scholar] [CrossRef] [PubMed]
- Nowakowski, T.J.; Pollen, A.A.; di Lullo, E.; Sandoval-Espinosa, C.; Bershteyn, M.; Kriegstein, A.R. Expression Analysis Highlights AXL as a Candidate Zika Virus Entry Receptor in Neural Stem Cells. Cell Stem Cell 2016, 18, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Hamel, R.; Dejarnacb, O.; Wichita, S.; Ekchariyawata, P.; Neyretc, A.; Luplertlopd, N.; Perera-Lecoina, M.; Surasombatpattanae, P.; Talignania, L.; Thomas, F.; et al. Biology of Zika Virus Infection in Human Skin Cells. J. Virol. 2015, 89, 8880–8896. [Google Scholar] [CrossRef] [PubMed]
- Valadão, A.L.C.; Aguiar, R.S.; de Arruda, L.B. Interplay between inflammation and cellular stress triggered by Flaviviridae viruses. Front. Microbiol. 2016, 7, 1233. [Google Scholar] [CrossRef] [PubMed]
- Barr, K.L.; Anderson, B.D.; Prakoso, D.; Long, M.T. Working with Zika and Usutu Viruses In Vitro. PLoS Negl. Trop. Dis. 2016, 10, e0004931. [Google Scholar] [CrossRef] [PubMed]
- Delvecchio, R.; Higa, L.M.; Pezzuto, P.; Valadão, A.L.; Garcez, P.P.; Monteiro, F.L.; Loiola, E.C.; Dias, A.A.; Silva, F.J.; Aliota, M.T.; et al. Chloroquine, an endocytosis blocking agent, inhibits zika virus infection in different cell models. Viruses 2016, 8, E322. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhu, W.; Xue, S.; Han, D. Testicular defense systems: Immune privilege and innate immunity. Cell. Mol. Immunol. 2014, 11, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Shi, L.; Wang, Q.; Cheng, L.; Zhao, X.; Chen, Q.; Jiang, Q.; Feng, M.; Li, Q.; Han, D. Mumps virus-induced innate immune responses in mouse Sertoli and Leydig cells. Sci. Rep. 2016, 6, 19507. [Google Scholar] [CrossRef] [PubMed]
- Govero, J.; Esakky, P.; Scheaffer, S.M.; Fernandez, E.; Drury, A.; Platt, D.J.; Gorman, M.J.; Richner, J.M.; Caine, E.A.; Salazar, V.; et al. Zika virus infection damages the testes in mice. Nature 2016, 540, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Bhushan, S.; Schuppe, H.C.; Fijak, M.; Meinhardt, A. Testicular infection: Microorganisms, clinical implications and host-pathogen interaction. J. Reprod. Immunol. 2009, 83, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Siemann, D.N.; Strange, D.P.; Maharaj, P.N.; Shi, P.-Y.; Verma, S. Zika Virus Infects Human Sertoli Cells and Modulates the Integrity of the In Vitro Blood-Testis Barrier Model. J. Virol. 2017, 91, e00623-17. [Google Scholar] [CrossRef] [PubMed]
- Frumence, E.; Roche, M.; Krejbich-Trotot, P.; El-Kalamouni, C.; Nativel, B.; Rondeau, P.; Missé, D.; Gadea, G.; Viranaicken, W.; Desprès, P. The South Pacific epidemic strain of Zika virus replicates efficiently in human epithelial A549 cells leading to IFN-β production and apoptosis induction. Virology 2016, 493, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Simoni, M.K.; Jurado, K.A.; Abrahams, V.M.; Fikrig, E.; Guller, S. Zika virus infection of Hofbauer cells. Am. J. Reprod. Immunol. 2017, 77, e12613. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.R.; Quicke, K.M.; Maddur, M.S.; O’Neal, J.T.; McDonald, C.E.; Fedorova, N.B.; Puri, V.; Shabman, R.; Pulendran, B.; Suthar, M.S. Zika Virus Antagonizes Type I Interferon Responses during Infection of Human Dendritic Cells. PLOS Pathog. 2017, 13, e1006164. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, D.G. The relationship of interacting immunological components in dengue pathogenesis. Virol. J. 2009, 6, 211. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Zhu, B.; Fu, Z.F.; Chen, H.; Cao, S. Immune evasion strategies of flaviviruses. Vaccine 2013, 31, 461–471. [Google Scholar] [CrossRef] [PubMed]
- Suthar, M.S.; Brassil, M.M.; Blahnik, G.; McMillan, A.; Ramos, H.J.; Proll, S.C.; Belisle, S.E.; Katze, M.G.; Gale, M., Jr. A Systems Biology Approach Reveals that Tissue Tropism to West Nile Virus Is Regulated by Antiviral Genes and Innate Immune Cellular Processes. PLoS Pathog. 2013, 9, e1003168. [Google Scholar] [CrossRef] [PubMed]
- Grant, A.; Ponia, S.S.; Tripathi, S.; Balasubramaniam, V.; Miorin, L.; Sourisseau, M.; Schwarz, M.C.; Sánchez-Seco, M.P.; Evans, M.J.; Best, S.M.; et al. Zika Virus Targets Human STAT2 to Inhibit Type I Interferon Signaling. Cell Host Microbe 2016, 19, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Li, S.; Ma, S.; Jia, L.; Zhang, F.; Zhang, Y.; Zhang, J.; Wong, G.; Zhang, S.; Lu, X.; et al. Zika Virus Causes Testis Damage and Leads to Male Infertility in Mice. Cell 2017, 168, 542. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.F.-W.; Yip, C.C.; Tsang, J.O.; Tee, K.M.; Cai, J.P.; Chik, K.K.; Zhu, Z.; Chan, C.C.; Choi, G.K.; Sridhar, S.; et al. Differential cell line susceptibility to the emerging Zika virus: Implications for disease pathogenesis, non-vector-borne human transmission and animal reservoirs. Emerg. Microbes Infect. 2016, 5, e93. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Wang, F.; Shi, L.; Zhao, X.; Gong, M.; Liu, W.; Song, C.; Li, Q.; Chen, Y.; Wu, H.; Han, D. C-X-C motif chemokine ligand 10 produced by mouse Sertoli cells in response to mumps virus infection induces male germ cell apoptosis. Nat. Publ. Gr. 2017, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.E.; Braun, R.E. Germ Cell Migration Across Sertoli Cell Tight Junctions. Science 2012, 338, 798–802. [Google Scholar] [CrossRef] [PubMed]
- Meachem, S.; Nieschlag, E.; Simoni, M. Inhibin B in male reproduction: Pathophysiology and clinical relevance. Eur. J. Endocrinol. 2001, 145, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S.K.; Dang, J.; Qin, Y.; Lichinchi, G.; Bansal, V.; Rana, T.M. Zika virus infection reprograms global transcription of host cells to allow sustained infection. Emerg. Microbes Infect. 2017, 6, e24. [Google Scholar] [CrossRef] [PubMed]
- Mansuy, J.M.; Suberbielle, E.; Chapuy-Regaud, S.; Mengelle, C.; Bujan, L.; Marchou, B.; Delobel, P.; Gonzalez-Dunia, D.; Malnou, C.E.; Izopet, J.; et al. Zika virus in semen and spermatozoa. Lancet Infect. Dis. 2016, 16, 1106–1107. [Google Scholar] [CrossRef]
- Griffin, B.D.; Muthumani, K.; Warner, B.M.; Majer, A.; Hagan, M.; Audet, J.; Stein, D.R.; Ranadheera, C.; Racine, T.; De La Vega, M.-A.; et al. DNA vaccination protects mice against Zika virus-induced damage to the testes. Nat. Commun. 2017, 8, 15743. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.-G.; Wang, P.; Zheng, W.; Zhang, Q.; Huang, W.; Jin, F.; Cai, Z. Characterisation of dendritic cell subsets in chronically inflamed human epididymis. Andrologia 2016, 48, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Duan, Y.-G. The role of dendritic cells in male reproductive tract. Am. J. Reprod. Immunol. 2016, 76, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.F.-W.; Yip, C.C.; Tee, K.M.; Zhu, Z.; Tsang, J.O.; Chik, K.K.; Tsang, T.G.; Chan, C.C.; Poon, V.K.; Sridhar, S.; et al. Improved detection of Zika virus RNA in human and animal specimens by a novel, highly sensitive and specific real-time RT-PCR assay targeting the 5′-untranslated region of Zika virus. Trop. Med. Int. Health 2017, 22, 594–603. [Google Scholar] [CrossRef] [PubMed]
- Osuna, C.E.; Lim, S.Y.; Deleage, C.; Griffin, B.D.; Stein, D.; Schroeder, L.T.; Omange, R.; Best, K.; Luo, M.; Hraber, P.T.; et al. Zika viral dynamics and shedding in rhesus and cynomolgus macaques. Nat. Med. 2016, 22, 1448–1455. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-R.; Liu, Y.-X. Focuses on impact of zika virus infection on male reproductive tract. Natl. Sci. Rev. 2017, 4, 157. [Google Scholar] [CrossRef][Green Version]
- Arsuaga, M.; Bujalance, S.G.; Díaz-Menéndez, M.; Vázquez, A.; Arribas, J.R. Probable sexual transmission of Zika virus from a vasectomised man. Lancet Infect. Dis. 2016, 16, 1107. [Google Scholar] [CrossRef]
- Hirsch, A.J.; Smith, J.L.; Haese, N.N.; Broeckel, R.M.; Parkins, C.J.; Kreklywich, C.; DeFilippis, V.R.; Denton, M.; Smith, P.P.; Messer, W.B.; et al. Zika Virus infection of rhesus macaques leads to viral persistence in multiple tissues. PLoS Pathog. 2017, 13, e1006219. [Google Scholar] [CrossRef] [PubMed]
- Heang, V.; Yasuda, C.Y.; Sovann, L.; Haddow, A.D.; da Rosa, A.P.T.; Tesh, R.B.; Kasper, M.R. Zika virus infection, Cambodia, 2010. Emerg. Infect. Dis. 2012, 18, 349–351. [Google Scholar] [CrossRef] [PubMed]
- Estofolete, C.F.; Terzian, A.C.; Parreira, R.; Esteves, A.; Hardman, L.; Greque, G.V.; Rahal, P.; Nogueira, M.L. Clinical and Laboratory Profile of Zika Virus Infection in Dengue Suspected Patients: A case series. J. Clin. Virol. 2016, 81, 25–30. [Google Scholar] [CrossRef] [PubMed]
- D’Ortenzio, E.; Matheron, S.; Yazdanpanah, Y.; de Lamballerie, X.; Hubert, B.; Piorkowski, G.; Maquart, M.; Descamps, D.; Damond, F.; Leparc-Goffart, I. Evidence of Sexual Transmission of Zika Virus. N. Engl. J. Med. 2016, 374, 2195–2198. [Google Scholar] [CrossRef] [PubMed]
- Souto, I.O.; Alejo-Cancho, I.; Brustenga, J.G.; Mestres, A.P.; Gutiérrez, J.M.; Yoldi, M.J.M. Persistence of Zika virus in semen 93 days after the onset of symptoms. Enferm. Infecc. Microbiol. Clin. 2018, 36, 21–23. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.R.; Martínez, N.; Moros, Z. Microhematospermia in acute Zika virus infection. Int. J. Infect. Dis. 2016, 51, 127. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gaskell, K.M.; Houlihan, C.; Nastouli, E.; Checkley, A.M. Persistent zika virus detection in semen in a traveler returning to the United Kingdom from Brazil, 2016. Emerg. Infect. Dis. 2017, 23, 137–139. [Google Scholar] [CrossRef] [PubMed]
- García-Bujalance, S.; Gutiérrez-Arroyo, A.; De la Calle, F.; Díaz-Menéndez, M.; Arribas, J.R.; García-Rodríguez, J.; Arsuaga, M. Persistence and infectivity of Zika virus in semen after returning from endemic areas: Report of 5 cases. J. Clin. Virol. 2017, 96, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Nicastri, E.; Castilletti, C.; Liuzzi, G.; Iannetta, M.; Capobianchi, M.R.; Ippolito, G. Persistent detection of Zika virus RNA in semen for six months after symptom onset in a traveller returning from Haiti to Italy, February 2016. Euro Surveill. 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Huits, R.M.; De Smet, B.; Ariën, K.K.; Van Esbroeck, M.; De Jong, B.C.; Bottieau, E.; Cnops, L. Kinetics of Zika virus persistence in semen. Bull. World Health Organ. 2016. [Google Scholar] [CrossRef]
- Tyler, K.L.; Roos, K.L. The Expanding Spectrum of Zika Virus Infections of the Nervous System. JAMA Neurol. 2017, 74, 1169–1171. [Google Scholar] [CrossRef] [PubMed]
- Biava, M.; Caglioti, C.; Castilletti, C.; Bordi, L.; Carletti, F.; Colavita, F.; Quartu, S.; Nicastri, E.; Iannetta, M.; Vairo, F.; et al. Persistence of ZIKV-RNA in the cellular fraction of semen is accompanied by a surrogate-marker of viral replication. Diagnostic implications for sexual transmission. New Microbiol. 2017, 41, 30–33. [Google Scholar] [PubMed]
- Araujo, A.Q.C.; Silva, M.T.T.; Araujo, A.P.Q.C. Zika virus-associated neurological disorders: A review. Brain 2016, 139, 2122–2130. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Y.; Wang, Z.; Zhen, Z.D.; Feng, K.H.; Guo, J.; Gao, N.; Fan, D.Y.; Han, D.S.; Wang, P.G.; An, J. Axl is not an indispensable factor for zika virus infection in mice. J. Gen. Virol. 2017, 98, 2061–2068. [Google Scholar] [CrossRef] [PubMed]
- Salam, A.P.; Horby, P. Isolation of viable Zika virus from spermatozoa. Lancet. Infect. Dis. 2018, 18, 144. [Google Scholar] [CrossRef]
- Yockey, L.J.; Varela, L.; Rakib, T.; Khoury-Hanold, W.; Fink, S.L.; Stutz, B.; Szigeti-Buck, K.; Van den Pol, A.; Lindenbach, B.D.; Horvath, T.L.; et al. Vaginal Exposure to Zika Virus during Pregnancy Leads to Fetal Brain Infection. Cell 2016, 166, 1247–1256. [Google Scholar] [CrossRef] [PubMed]
- Brazilian Ministry of Health. Health Surveillance Department, Integrated monitoring of growth and developmental alterations in relation to Zika virus and other infectious ethiologies up to 52 epidemiological week of 2017. Epidemiol. Bull. 2018, 49, 1–10. [Google Scholar]
- Doncel, G.F.; Anderson, S.; Zalenskaya, I. Role of Semen in Modulating the Female Genital Tract Microenvironment—Implications for HIV Transmission. Am. J. Reprod. Immunol. 2014, 71, 564–574. [Google Scholar] [CrossRef] [PubMed]
| Date | Advances | Reference |
|---|---|---|
| May 2011 | First report of ZIKV probable sexual transmission | [16] |
| May 2014 | Two cases of probable sexual transmission reported | [26] |
| February 2015 | Infectious ZIKV particles detected in semen samples | [27] |
| March 2016 | High viral load reported in semen sample | [28] |
| May 2016 | Zika virus presence in semen confirmed | [29] |
| April 2016 | Zika RNA found in semen of asymptomatic blood donors | [30] |
| April 2016 | First report of male-to-male sexual transmission | [31] |
| July 2016 | First report of Zika RNA in cervical mucus | [32] |
| July 2016 | First report of suspected female-to-male sexual transmission | [33] |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Silva, L.R.C. Zika Virus Trafficking and Interactions in the Human Male Reproductive Tract. Pathogens 2018, 7, 51. https://doi.org/10.3390/pathogens7020051
Da Silva LRC. Zika Virus Trafficking and Interactions in the Human Male Reproductive Tract. Pathogens. 2018; 7(2):51. https://doi.org/10.3390/pathogens7020051
Chicago/Turabian StyleDa Silva, Lucia Regina Cangussu. 2018. "Zika Virus Trafficking and Interactions in the Human Male Reproductive Tract" Pathogens 7, no. 2: 51. https://doi.org/10.3390/pathogens7020051
APA StyleDa Silva, L. R. C. (2018). Zika Virus Trafficking and Interactions in the Human Male Reproductive Tract. Pathogens, 7(2), 51. https://doi.org/10.3390/pathogens7020051




