Comparing the Folds of Prions and Other Pathogenic Amyloids
Abstract
1. Introduction
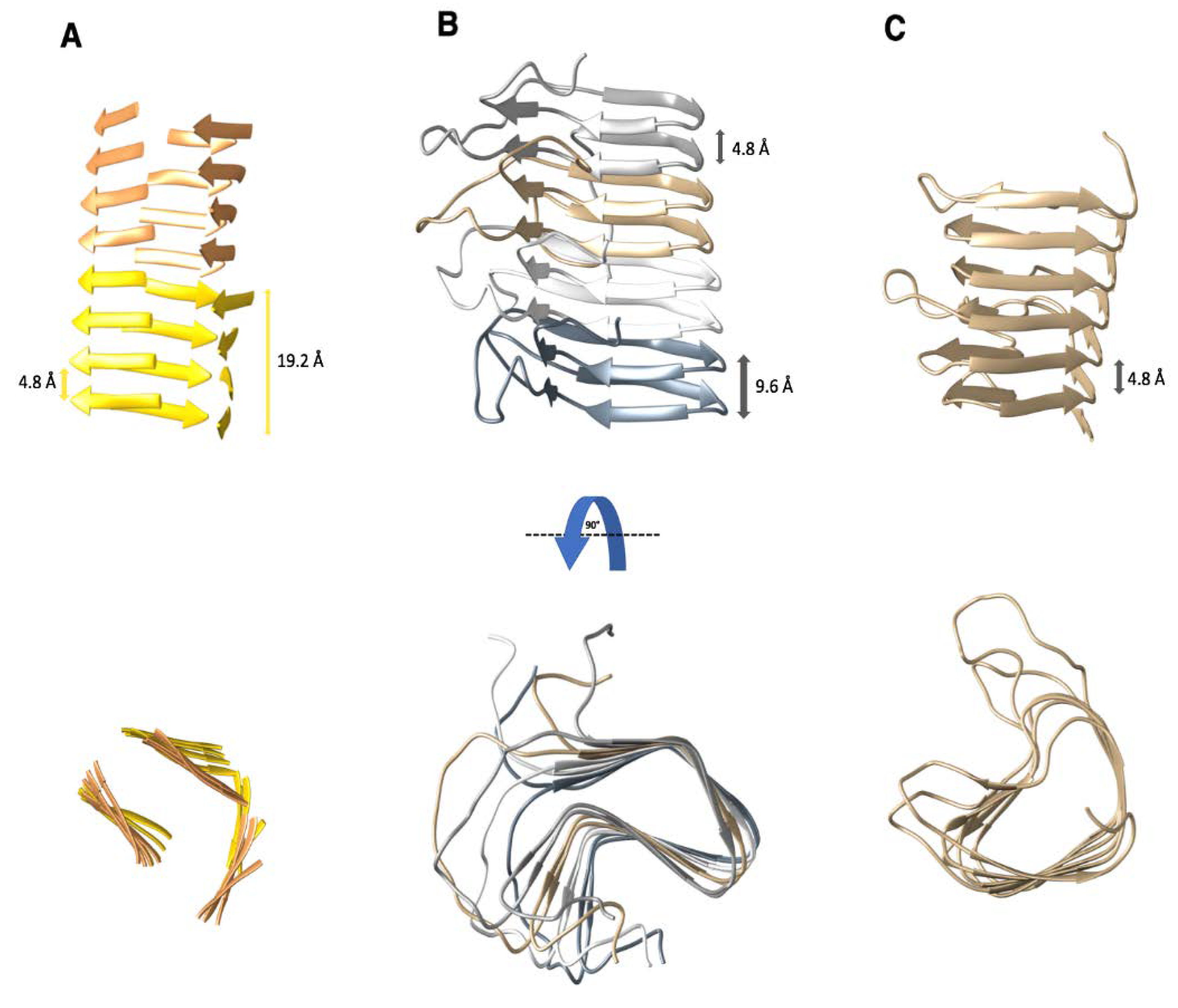
2. β-solenoid Amyloids: PrPSc and HET-s
2.1. The Structure of PrPSc
2.2. The Structure of the HET-s Prion Domain
3. The β-Solenoid Fold of Non-Pathogenic Proteins
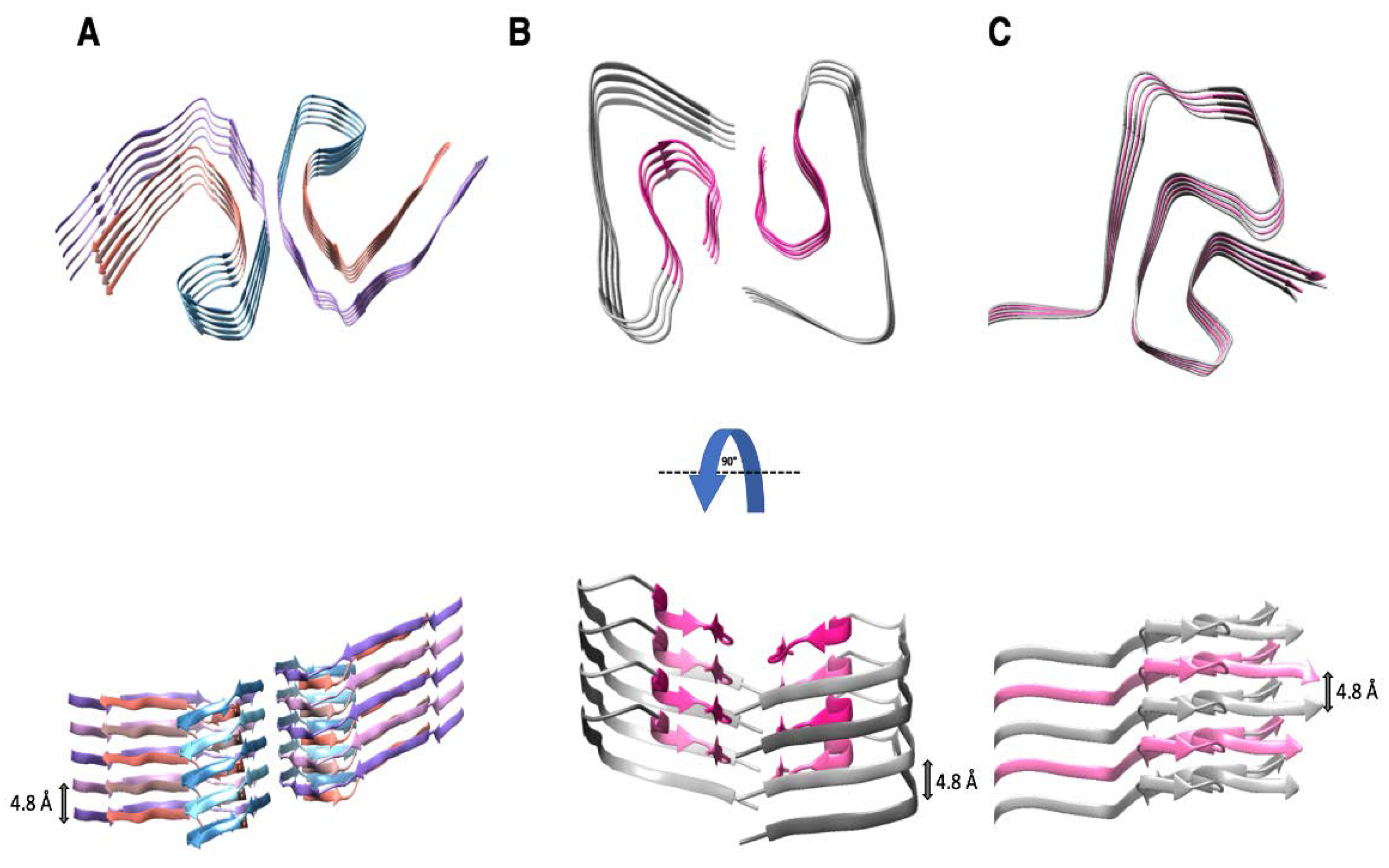
4. Amyloid Folds of Other Pathogenic Proteins
4.1. Short Amyloid Peptides
4.2. The Structure of PHF-Tau
4.3. The Structure of Aβ(1-42)
4.4. The Structure α-Synuclein
5. Similarities and Differences between Common Amyloid Folds
5.1. Similarities between PrPSc, HET-s, and β-solenoid Proteins
5.2. Comparisons of Aβ(1-42), α-Synuclein, PHF-tau and β-Solenoid Amyloids
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Gibbs, C.J.J.; Gajdusek, D.C.; Asher, D.M.; Alpers, M.P.; Beck, E.; Daniel, P.M.; Matthews, W.B. Creutzfeldt-Jakob Disease (Spongiform Encephalopathy): Transmission to Chimpanzee. Science 1968, 161, 388–389. [Google Scholar] [CrossRef] [PubMed]
- Moller, H.J.; Graeber, M.B. The case described by Alois Alzheimer in 1911. Eur. Arch. Psychiatry Clin. Neurosci. 1998, 248, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, J. An Essay on the Shaking Palsy; Whittingham & Rowland: London, UK, 1817. [Google Scholar]
- Eisenberg, D.; Jucker, M. The amyloid state of proteins in human diseases. Cell 2012, 148, 1188–1203. [Google Scholar] [CrossRef] [PubMed]
- Tatarnikova, O.G.; Orlov, M.A.; Bobkova, N.V. Beta-Amyloid and Tau-Protein: Structure, Interaction, and Prion-Like Properties. Biochemistry 2015, 80, 1800–1819. [Google Scholar] [CrossRef] [PubMed]
- Giles, K.; Olson, S.H.; Prusiner, S.B. Developing Therapeutics for PrP Prion Diseases. Cold Spring Harb. Perspect. Med. 2017, 7, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Wille, H.; Requena, J.R. The Structure of PrPSc Prions. Pathogens 2018, 7, 20. [Google Scholar] [CrossRef] [PubMed]
- Caughey, B.W.; Dong, A.; Bhat, K.S.; Ernst, D.; Hayes, S.F.; Caughey, W.S. Secondary structure analysis of the scrapie-associated protein PrP 27-30 in water by infrared spectroscopy. Biochemistry 1991, 30, 7672–7680. [Google Scholar] [CrossRef] [PubMed]
- Prusiner, S.B.; Bolton, D.C.; Groth, D.F.; Bowman, K.A.; Cochran, S.P.; McKinley, M.P. Further purification and characterization of scrapie prions. Biochemistry 1982, 21, 6942–6950. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Fernández, E.; Young, H.S.; Requena, J.R.; Wille, H. The Structure of Mammalian Prions and Their Aggregates. Int. Rev. Cell Mol. Biol. 2017, 329, 277–301. [Google Scholar] [PubMed]
- Requena, J.R.; Wille, H. The Structure of the Infectious Prion Protein. Prion 2014, 8, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Horiuchi, M.; Karino, A.; Furuoka, H.; Ishiguro, N.; Kimura, K.; Shinagawa, M. Generation of Monoclonal Antibody That Distinguishes PrPSc from PrPC and Neutralizes Prion Infectivity. Virology 2009, 394, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Govaerts, C.; Wille, H.; Prusiner, S.B.; Cohen, F.E. Evidence for Assembly of Prions with Left-Handed-Helices into Trimers. Proc. Natl. Acad. Sci. USA 2004, 101, 8342–8347. [Google Scholar] [CrossRef] [PubMed]
- DeMarco, M.L.; Daggett, V. From Conversion to Aggregation: Protofibril Formation of the Prion Protein. Proc. Natl. Acad. Sci. USA 2004, 101, 2293–2298. [Google Scholar] [CrossRef] [PubMed]
- Cobb, N.J.; Sönnichsen, F.D.; Mchaourab, H.; Surewicz, W.K. Molecular Architecture of Human Prion Protein Amyloid: A Parallel, in-Register β-Structure. Proc. Natl. Acad. Sci. USA 2007, 104, 18946–18951. [Google Scholar] [CrossRef] [PubMed]
- Wille, H.; Bian, W.; McDonald, M.; Kendall, A.; Colby, D.W.; Bloch, L.; Ollesch, J.; Borovinskiy, A.L.; Cohen, F.E.; Prusiner, S.B.; et al. Natural and Synthetic Prion Structure from X-Ray Fiber Diffraction. Proc. Natl. Acad. Sci. USA 2009, 106, 16990–16995. [Google Scholar] [CrossRef] [PubMed]
- Wille, H.; Michelitsch, M.D.; Guenebaut, V.; Supattapone, S.; Serban, A.; Cohen, F.E.; Agard, D.A.; Prusiner, S.B. Structural Studies of the Scrapie Prion Protein by Electron Crystallography. Proc. Natl. Acad. Sci. USA 2002, 99, 3563–3568. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Fernández, E.; Alonso, J.; Pastrana, M.A.; Ramos, A.; Stitz, L.; Vidal, E.; Dynin, I.; Petsch, B.; Silva, C.J.; Requena, J.R. Structural Organization of Mammalian Prions as Probed by Limited Proteolysis. PLoS ONE 2012, 7, e50111. [Google Scholar] [CrossRef] [PubMed]
- Gremer, L.; Schölzel, D.; Schenk, C.; Reinartz, E.; Labahn, J.; Ravelli, R.B.G.; Tusche, M.; Lopez-Iglesias, C.; Hoyer, W.; Heise, H.; et al. Fibril Structure of Amyloid-β(1–42) by Cryoelectron Microscopy. Science 2017, 358, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, A.W.P.; Falcon, B.; He, S.; Murzin, A.G.; Murshudov, G.; Garringer, H.J.; Crowther, R.A.; Ghetti, B.; Goedert, M.; Scheres, S.H.W. Cryo-EM Structures of Tau Filaments from Alzheimer’s Disease. Nature 2017, 547, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Tuttle, M.D.; Comellas, G.; Nieuwkoop, A.J.; Covell, D.J.; Berthold, D.A.; Kloepper, K.D.; Courtney, J.M.; Kim, J.K.; Barclay, A.M.; Kendall, A.; et al. Solid-State NMR Structure of a Pathogenic Fibril of Full-Length Human α-Synuclein. Nat. Struct. Mol. Biol. 2016, 23, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Wälti, M.A.; Ravotti, F.; Arai, H.; Glabe, C.G.; Wall, J.S.4; Böckmann, A.; Güntert, P.; Meier, B.H.; Riek, R. Atomic-resolution structure of a disease-relevant Aβ(1–42) amyloid fibril. Proc. Natl. Acad. Sci. USA 2016, 113, E4976–E4984. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Ferreira, R.; Nicholas, M.I.; Mona, T.D.; Ringler, P.; Lauer, M.E.; Riek, R.; Britschgi, M.; Stahlberg, H. Cryo-EM structure of alpha-synuclein fibrils. bioRxiv 2018, 276436. [Google Scholar] [CrossRef]
- Pan, K.M.; Baldwin, M.; Nguyen, J.; Gasset, M.; Serban, A.; Groth, D.; Mehlhorn, I.; Huang, Z.; Fletterick, R.J.; Cohen, F.E. Conversion of α-Helices into β-Sheets Features in the Formation of the Scrapie Prion Proteins. Proc. Natl. Acad. Sci. USA 1993, 90, 10962–10966. [Google Scholar] [CrossRef] [PubMed]
- Wan, W.; Wille, H.; Stöhr, J.; Kendall, A.; Bian, W.; McDonald, M.; Tiggelaar, S.; Watts, J.C.; Prusiner, S.B.; Stubbs, G. Structural Studies of Truncated Forms of the Prion Protein PrP. Biophys. J. 2015, 108, 1548–1554. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Fernández, E.; Vos, M.R.; Afanasyev, P.; Cebey, L.; Sevillano, A.M.; Vidal, E.; Rosa, I.; Renault, L.; Ramos, A.; Peters, P.J.; et al. The Structural Architecture of an Infectious Mammalian Prion Using Electron Cryomicroscopy. PLoS Pathog. 2016, 12, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Smirnovas, V.; Baron, G.S.; Offerdahl, D.K.; Raymond, G.J.; Caughey, B.; Surewicz, W.K. Structural Organization of Brain-Derived Mammalian Prions Examined by Hydrogen-Deuterium Exchange. Nat. Struct. Mol. Biol. 2011, 18, 504–506. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.J.; Vázquez-Fernández, E.; Onisko, B.; Requena, J.R. Proteinase K and the Structure of PrPSc: The Good, the Bad and the Ugly. Virus Res. 2015, 207, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Tsemekhman, K.; Goldschmidt, L.; Eisenberg, D.S.; Baker, D. Cooperative Hydrogen Bonding in Amyloid Formation. Protein Sci. 2007, 16, 761–764. [Google Scholar] [CrossRef] [PubMed]
- Kobe, B.; Kajava, A.V. When Protein Folding Is Simplified to Protein Coiling: The Continuum of Solenoid Protein Structures. Trends Biochem. Sci. 2000, 25, 509–515. [Google Scholar] [CrossRef]
- Wasmer, C.; Lange, A.; Melckebeke, H.V.; Siemer, A.B.; Riek, R.; Meier, B.H. Amyloid Fibrils of the HET-s(218–289) Prion Form a B Solenoid with a Triangular Hydrophobic Core. Science 2008, 319, 1523–1527. [Google Scholar] [CrossRef] [PubMed]
- Yoder, M.D.; Jurnak, F. Protein motifs. 3. The parallel beta helix and other coiled folds. FASEB J. 1995, 9, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Coustou, V.; Deleu, C.; Saupe, S.; Begueret, J. The Protein Product of the Het-S Heterokaryon Incompatibility Gene of the Fungus Podospora Anserina Behaves as a Prion Analog. Proc. Natl. Acad. Sci. USA 1997, 94, 9773–9778. [Google Scholar] [CrossRef] [PubMed]
- Wickner, R.B. A New Prion Controls Fungal Cell Fusion Incompatibility. Proc. Natl. Acad. Sci. USA 1997, 94, 10012–10014. [Google Scholar] [CrossRef] [PubMed]
- Dos Reis, S.; Coulary-Salin, B.; Forge, V.; Lascu, I.; Bégueret, J.; Saupe, S.J. The HET-S Prion Protein of the Filamentous Fungus Podospora Anserina Aggregates in Vitro into Amyloid-like Fibrils. J. Biol. Chem. 2002, 277, 5703–5706. [Google Scholar] [CrossRef] [PubMed]
- Melckebeke, V.; Wasmer, C.; Lange, A.; Ab, E.; Loquet, A.; Böckmann, A.; Meier, B.H. Atomic-Resolution Three-Dimensional Structure of HET-S(218–289) Amyloid Fibrils by Solid-State NMR. J. Am. Chem. Soc. 2010, 132, 13765–13775. [Google Scholar] [CrossRef] [PubMed]
- Ritter, C.; Maddelein, M.-L.; Siemer, A.B.; Lührs, T.; Ernst, M.; Meier, B.H.; Saupe, S.J.; Riek, R. Correlation of Structural Elements and Infectivity of the HET-S Prion. Nature 2005, 435, 844–848. [Google Scholar] [CrossRef] [PubMed]
- Balguerie, A.; Dos Reis, S.; Ritter, C.; Chaignepain, S.; Coulary-Salin, B.; Forge, V.; Bathany, K.; Lascu, I.; Schmitter, J.M.; Riek, R.; et al. Domain Organization and Structure-Function Relationship of the HET-S Prion Protein of Podospora Anserina. EMBO J. 2003, 22, 2071–2081. [Google Scholar] [CrossRef] [PubMed]
- Greenwald, J.; Buhtz, C.; Ritter, C.; Kwiatkowski, W.; Choe, S.; Maddelein, M.-L.; Ness, F.; Cescau, S.; Soragni, A.; Leitz, D.; et al. The Mechanism of Prion Inhibition by HET-S. Mol. Cell 2010, 38, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Liebman, S.W.; Chernoff, Y.O. Prions in Yeast. Genetics 2012, 191, 1041–1072. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, N.; Baxa, U.; Steven, A.C. Structural Dependence of HET-S Amyloid Fibril Infectivity Assessed by Cryoelectron Microscopy. Proc. Natl. Acad. Sci. USA 2011, 108, 3252–3257. [Google Scholar] [CrossRef] [PubMed]
- Wan, W.; Wille, H.; Stöhr, J.; Baxa, U.; Prusiner, S.B.; Stubbs, G. Degradation of fungal prion HET-s(218–289) induces formation of a generic amyloid fold. Biophys. J. 2012, 102, 2339–2344. [Google Scholar] [CrossRef] [PubMed]
- Wan, W.; Stubbs, G. Fungal Prion HET-S as a Model for Structural Complexity and Self-Propagation in Prions. Proc. Natl. Acad. Sci. USA 2014, 111, 5201–5206. [Google Scholar] [CrossRef] [PubMed]
- Kajava, A.V.; Steven, A.C. β-Rolls, β-Helices, and Other β-Solenoid Proteins. Adv. Protein Chem. 2006, 73, 55–96. [Google Scholar] [PubMed]
- Kajava, A.V.; Steven, A.C. The Turn of the Screw: Variations of the Abundant β-Solenoid Motif in Passenger Domains of Type V Secretory Proteins. J. Struct. Biol. 2006, 155, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Kajava, A.V.; Baxa, U.; Steven, A.C. Beta Arcades: Recurring Motifs in Naturally Occurring and Disease-Related Amyloid Fibrils. FASEB J. 2010, 24, 1311–1319. [Google Scholar] [CrossRef] [PubMed]
- Hennetin, J.; Jullian, B.; Steven, A.C.; Kajava, A.V. Standard Conformations of β-Arches in β-Solenoid Proteins. J. Mol. Biol. 2006, 358, 1094–1105. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, J.; Pickersgill, R. The Architecture of Parallel β-Helices and Related Folds. Prog. Biophys. Mol. Biol. 2001, 77, 111–175. [Google Scholar] [CrossRef]
- Yoder, M.D.; Lietzke, S.E.; Jurnak, F. Unusual Structural Features in the Parallel β-Helix in Pectate Lyases. Structure 1993, 1, 241–251. [Google Scholar] [CrossRef]
- Henrissat, B.; Heffron, S.E.; Yoder, M.D.; Lietzke, S.E.; Jurnak, F. Functional Implications of Structure-Based Sequence Alignment of Proteins in the Extracellular Pectate Lyase Superfamily. Plant Physiol. 1995, 107, 963–976. [Google Scholar] [CrossRef] [PubMed]
- Bryan, A.W.; Starner-Kreinbrink, J.L.; Hosur, R.; Clark, P.L.; Berger, B. Structure-Based Prediction Reveals Capping Motifs That Inhibit β-Helix Aggregation. Proc. Natl. Acad. Sci. USA 2011, 108, 11099–11104. [Google Scholar] [CrossRef] [PubMed]
- Kondo, H.; Hanada, Y.; Sugimoto, H.; Hoshino, T.; Garnham, C.P.; Davies, P.L.; Tsuda, S. Ice-Binding Site of Snow Mold Fungus Antifreeze Protein Deviates from Structural Regularity and High Conservation. Proc. Natl. Acad. Sci. USA 2012, 109, 9360–9365. [Google Scholar] [CrossRef] [PubMed]
- Nelson, R.; Sawaya, M.R.; Balbirnie, M.; Madsen, A.Ø.; Riekel, C.; Grothe, R.; Eisenberg, D. Structure of the Cross-β Spine of Amyloid-like Fibrils. Nature 2005, 435, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Sawaya, M.R.; Sambashivan, S.; Nelson, R.; Ivanova, M.I.; Sievers, S.A.; Apostol, M.I.; Thompson, M.J.; Balbirnie, M.; Wiltzius, J.J.W.; McFarlane, H.T.; et al. Atomic Structures of Amyloid Cross-β Spines Reveal Varied Steric Zippers. Nature 2007, 447, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Riek, R.; Eisenberg, D.S. The Activities of Amyloids from a Structural Perspective. Nature 2016, 539, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Wiltzius, J.J.W.; Landau, M.; Nelson, R.; Sawaya, M.R.; Apostol, M.I.; Goldschmidt, L.; Soriaga, A.B.; Cascio, D.; Rajashankar, K.; Eisenberg, D. Molecular Mechanisms for Protein-Encoded Inheritance. Nat. Struct. Mol. Biol. 2009, 16, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Goedert, M.; Spillantini, M.G.; Jakes, R.; Rutherford, D.; Crowther, R.A. Multiple Isoforms of Human Microtubule-Associated Protein Tau: Sequences and Localization in Neurofibrillary Tangles of Alzheimer’s disease. Neuron 1989, 3, 519–526. [Google Scholar] [CrossRef]
- Mandelkow, E.M.; Mandelkow, E. Tau in Alzheimer’s Disease. Trends Cell Biol. 1998, 8, 425–427. [Google Scholar] [CrossRef]
- Crowther, R.A. Straight and Paired Helical Filaments in Alzheimer Disease Have a Common Structural Unit. Proc. Natl. Acad. Sci. USA 1991, 88, 2288–2292. [Google Scholar] [CrossRef] [PubMed]
- Šimić, G.; Babić Leko, M.; Wray, S.; Harrington, C.; Delalle, I.; Jovanov-Milošević, N.; Bažadona, D.; Buée, L.; de Silva, R.; Di Giovanni, G.; et al. Tau Protein Hyperphosphorylation and Aggregation in Alzheimer’s Disease and Other Tauopathies, and Possible Neuroprotective Strategies. Biomolecules 2016, 6, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Kirschner, D.A.; Abraham, C.; Selkoe, D.J. X-ray diffraction from intraneuronal paired helical filaments and extraneuronal amyloid fibers in Alzheimer disease indicates cross-beta conformation. Proc. Natl. Acad. Sci. USA 1986, 83, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Schweers, O.; Schönbrunn-Hanebeck, E.; Marx, A.; Mandelkow, E. Structural Studies of Tau-Protein and Alzheimer Paired Helical Filaments Show No Evidence for Beta-Structure. J. Biol. Chem. 1994, 269, 24290–24297. [Google Scholar] [PubMed]
- von Bergen, M.; Barghorn, S.; Biernat, J.; Mandelkow, E.M.; Mandelkow, E. Tau aggregation is driven by a transition from random coil to beta sheet structure. Biochim. Biophys. Acta 2005, 1739, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Margittai, M.; Langen, R. Template-Assisted Filament Growth by Parallel Stacking of Tau. Proc. Natl. Acad. Sci. USA 2004, 101, 10278–10283. [Google Scholar] [CrossRef] [PubMed]
- Baxa, U. Structural Basis of Infectious and Non-Infectious Amyloids. Curr. Alzheimer Res. 2008, 5, 308–318. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lührs, T.; Ritter, C.; Adrian, M.; Riek-Loher, D.; Bohrmann, B.; Döbeli, H.; Schubert, D.; Riek, R. 3D structure of Alzheimer’s amyloid-beta(1–42) fibrils. Proc. Natl. Acad. Sci. USA 2005, 102, 17342–17347. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Rohou, A.; Lasker, K.; Yadav, J.K.; Schiene-Fischer, C.; Fändrich, M.; Grigorieff, N. Peptide Dimer Structure in an Aβ(1–42) Fibril Visualized with Cryo-EM. Proc. Natl. Acad. Sci. USA 2015, 112, 11858–11863. [Google Scholar] [CrossRef] [PubMed]
- Riek, R. The Three-Dimensional Structures of Amyloids. Cold Spring Harb. Perspect. Biol. 2017, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Goldsbury, C.S.; Wirtz, S.; Müller, S.A.; Sunderji, S.; Wicki, P.; Aebi, U.; Frey, P. Studies on the in Vitro Assembly of Aβ1-40: Implications for the Search for a Beta Fibril Formation Inhibitors. J. Struct. Biol. 2000, 130, 217–231. [Google Scholar] [CrossRef] [PubMed]
- Sachse, C.; Xu, C.; Wieligmann, K.; Diekmann, S.; Grigorieff, N.; Fändrich, M. Quaternary Structure of a Mature Amyloid Fibril from Alzheimer’s Aβ(1–40) Peptide. J. Mol. Biol. 2006, 362, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.A.; Ivanova, M.I.; Sawaya, M.R.; Cascio, D.; Reyes, F.E.; Shi, D.; Sangwan, S.; Guenther, E.L.; Johnson, L.M.; Zhang, M.; et al. Structure of the Toxic Core of α-Synuclein from Invisible Crystals. Nature 2015, 525, 486–490. [Google Scholar] [CrossRef] [PubMed]
- Emamzadeh, F.N. Alpha-Synuclein Structure, Functions, and Interactions. J. Res. Med. Sci. 2016, 21, 29. [Google Scholar] [CrossRef] [PubMed]
- Spillantini, M.G.; Schmidt, M.L.; Lee, V.M.-Y.; Trojanowski, J.Q.; Jakes, R.; Goedert, M. Alpha-Synuclein in Lewy Bodies. Nature 1997, 388, 839–840. [Google Scholar] [CrossRef] [PubMed]
- Roeters, S.J.; Iyer, A.; Pletikapić, G.; Kogan, V.; Subramaniam, V.; Woutersen, S. Evidence for Intramolecular Antiparallel Beta-Sheet Structure in Alpha-Synuclein Fibrils from a Combination of Two-Dimensional Infrared Spectroscopy and Atomic Force Microscopy. Sci. Rep. 2017, 7, 41051. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, E.G.; Thornton, J.M. The Greek Key Motif: Extraction, Classification and Analysis. Protein Eng. 1993, 6, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Giasson, B.I.; Murray, I.V.J.; Trojanowski, J.Q.; Lee, V.M.Y. A Hydrophobic Stretch of 12 Amino Acid Residues in the Middle of α-Synuclein Is Essential for Filament Assembly. J. Biol. Chem. 2001, 276, 2380–2386. [Google Scholar] [CrossRef] [PubMed]
- Tzotzos, S.; Doig, A.J. Amyloidogenic Sequences in Native Protein Structures. Protein Sci. 2010, 19, 327–348. [Google Scholar] [CrossRef] [PubMed]
- Zweckstetter, M.; Requena, J.R.; Wille, H. Elucidating the structure of an infectious protein. PLoS Pathog. 2017, 13, e1006229. [Google Scholar] [CrossRef] [PubMed]
- Peralta, M.D.R.; Karsai, A.; Ngo, A.; Sierra, C.; Fong, K.T.; Hayre, N.R.; Mirzaee, N.; Ravikumar, K.M.; Kluber, A.J.; Chen, X.; et al. Engineering Amyloid Fibrils from β-Solenoid Proteins for Biomaterials Applications. ACS Nano 2015, 9, 449–463. [Google Scholar] [CrossRef] [PubMed]
- Helmus, J.J.; Surewicz, K.; Surewicz, W.K.; Jaroniec, C.P. Conformational flexibility of Y145Stop human prion protein amyloid fibrils probed by solid-state nuclear magnetic resonance spectroscopy. J. Am. Chem. Soc. 2010, 132, 2393–2403. [Google Scholar] [CrossRef] [PubMed]
- Helmus, J.J.; Surewicz, K.; Apostol, M.I.; Surewicz, W.K.; Jaroniec, C.P. Intermolecular alignment in Y145Stop human prion protein amyloid fibrils probed by solid-state NMR spectroscopy. J. Am. Chem. Soc. 2011, 133, 13934–13937. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Protein/Protein Aggregate | β-Solenoid | Parallel in-Register | Steric Zippers | Salt Bridges 2 | Hydro-Phobic Core | Symmetry of Proto-Filaments | |||
|---|---|---|---|---|---|---|---|---|---|
| Homo-Steric 1 | Hetero-Steric 2 | Intra-Molecular | Inter-Molecular | ||||||
| Pathogenic proteins | PHF-tau | (partial) | + | 1 | 6 | 5 | 2 | + | 21 screw |
| Aβ(1-42) | − | + | 1 | 2 | 0 | 2 | + | 21 screw | |
| α-synuclein | − | + | 1 | 2 | 2 | 2 | + | 21 screw | |
| PrPSc | + | − | 0 | 0 | unknown | unknown | + | unknown | |
| Non-pathogenic proteins | HET-s | + | − | 0 | 0 | 3 | 0 | + | N. A. |
| Pectate lyase C | + | − | 0 | 0 | 15 | N. A. | + | N. A. | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores-Fernández, J.M.; Rathod, V.; Wille, H. Comparing the Folds of Prions and Other Pathogenic Amyloids. Pathogens 2018, 7, 50. https://doi.org/10.3390/pathogens7020050
Flores-Fernández JM, Rathod V, Wille H. Comparing the Folds of Prions and Other Pathogenic Amyloids. Pathogens. 2018; 7(2):50. https://doi.org/10.3390/pathogens7020050
Chicago/Turabian StyleFlores-Fernández, José Miguel, Vineet Rathod, and Holger Wille. 2018. "Comparing the Folds of Prions and Other Pathogenic Amyloids" Pathogens 7, no. 2: 50. https://doi.org/10.3390/pathogens7020050
APA StyleFlores-Fernández, J. M., Rathod, V., & Wille, H. (2018). Comparing the Folds of Prions and Other Pathogenic Amyloids. Pathogens, 7(2), 50. https://doi.org/10.3390/pathogens7020050






