Curvularia spicifera in Non-Invasive Fungal Rhinosinusitis: Case Reports and Diagnostic Insights
Abstract
1. Introduction
2. Case Presentation
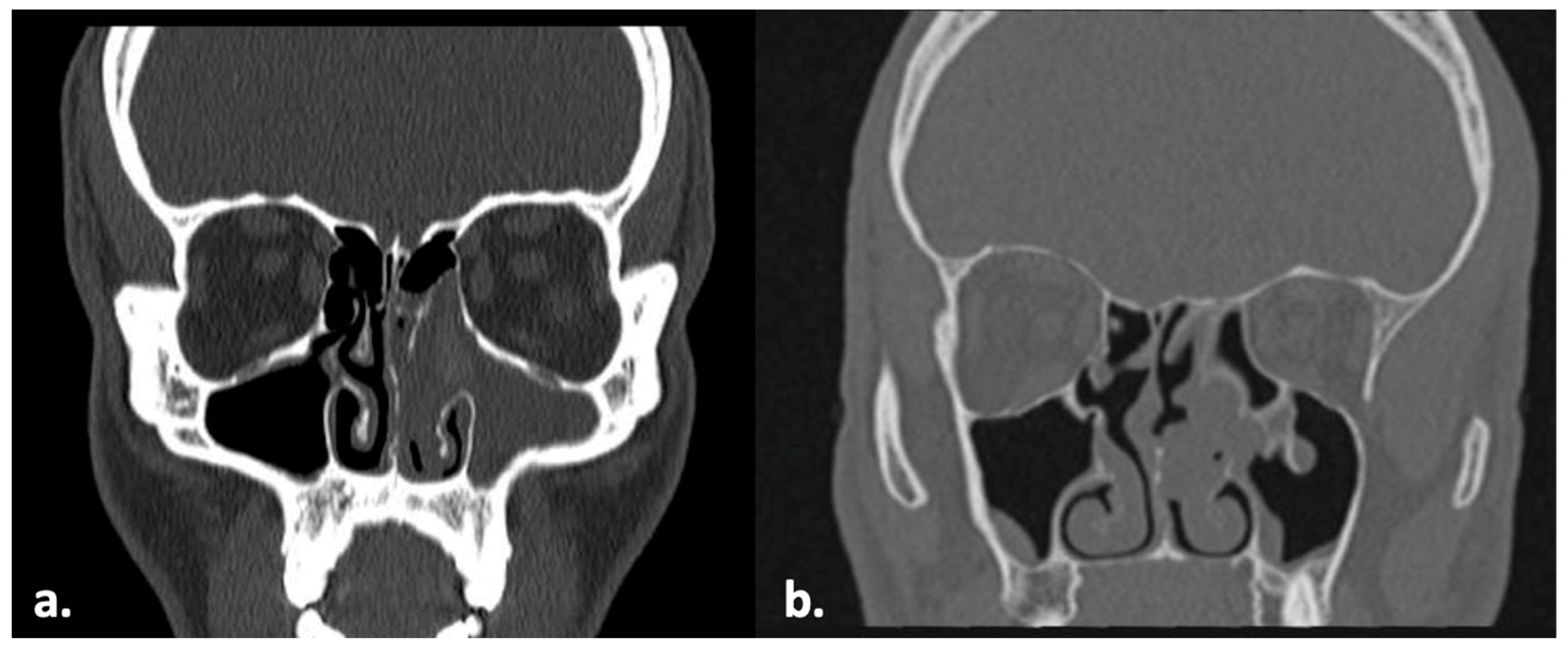
2.1. Case 1
2.2. Case 2
3. Materials and Methods
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jain, N.; Lodha, R.; Kabra, S.K. Upper respiratory tract infections. Indian J. Pediatr. 2001, 68, 1135–1138. [Google Scholar] [CrossRef] [PubMed]
- Schubert, M.S. Fungal rhinosinusitis: Diagnosis and therapy. Curr. Allergy Asthma Rep. 2001, 1, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Schubert, M.S. Allergic fungal sinusitis: Pathophysiology, diagnosis and management. Med. Mycol. 2009, 47, S324–S330. [Google Scholar] [CrossRef] [PubMed]
- Buzina, W.; Braun, H.; Schimpl, K.; Stammberger, H. Bipolaris spicifera causes fungus balls of the sinuses and triggers polypoid chronic rhinosinusitis in an immunocompetent patient. J. Clin. Microbiol. 2003, 41, 4885–4887. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, K.; Kawabata, T.; Wakayama, M.; Oharaseki, T.; Yokouchi, Y.; Takahashi, K.; Naoe, S.; Ogoshi, T.; Iwabuchi, S.; Shibuya, K.; et al. A case of allergic fungal sinusitis caused by Bipolaris spicifera. Nihon Ishinkin Gakkai Zasshi 2004, 45, 239–245. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.D.; Lee, S.B.; Taylor, J.W. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Schleimer, R.P. Immunopathogenesis of Chronic Rhinosinusitis and Nasal Polyposis. Annu. Rev. Pathol. 2017, 12, 331–357. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, K.; Oharaseki, T.; Yokouchi, Y.; Kawabata, T.; Wakayama, M.; Ogoshi, T.; Iwabuchi, S.; Shibuya, K.; Nishimura, K.; Takahashi, K. Allergic fungal sinusitis caused by Bipolaris spicifera and Schizophyllum commune. Med. Mycol. 2007, 45, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Gourley, D.S.; Whisman, B.A.; Jorgensen, N.L.; Martin, M.E.; Reid, M.J. Allergic Bipolaris sinusitis: Clinical and immunopathologic characteristics. J. Allergy Clin. Immunol. 1990, 85, 583–591. [Google Scholar] [CrossRef]
- Klapper, S.R.; Lee, A.G.; Patrinely, J.R.; Stewart, M.; Alford, E.L. Orbital involvement in allergic fungal sinusitis. Ophthalmology 1997, 104, 2094–2100. [Google Scholar] [CrossRef]
- Ambrosetti, D.; Hofman, V.; Castillo, L.; Gari-Toussaint, M.; Hofman, P. An expansive paranasal sinus tumour-like lesion caused by Bipolaris spicifera in an immunocompetent patient. Histopathology 2006, 49, 660–662. [Google Scholar] [CrossRef]
- Coop, C.A.; England, R.W. Allergic fungal sinusitis presenting with proptosis and diplopia: A review of ophthalmologic complications and treatment. Allergy Asthma Proc. 2006, 27, 72–76. [Google Scholar] [PubMed]
- McGinnis, M.R.; Campbell, G.; Gourley, W.K.; Lucia, H.L. Phaeohyphomycosis caused by Bipolaris spicifera: An informative case. Eur. J. Epidemiol. 1992, 8, 383–386. [Google Scholar] [CrossRef] [PubMed]
- Bent, J.P., 3rd; Kuhn, F.A. Diagnosis of allergic fungal sinusitis. Otolaryngol. Head. Neck Surg. 1994, 111, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Muthu, V.; Sehgal, I.S.; Dhooria, S.; Prasad, K.T.; Aggarwal, A.N. Allergic Bronchopulmonary Aspergillosis. Clin. Chest Med. 2022, 43, 99–125. [Google Scholar] [CrossRef] [PubMed]
- Schubert, M.S.; Hutcheson, P.S.; Graff, R.J.; Santiago, L.; Slavin, R.G. HLA-DQB1 *03 in allergic fungal sinusitis and other chronic hypertrophic rhinosinusitis disorders. J. Allergy Clin. Immunol. 2004, 114, 1376–1383. [Google Scholar] [CrossRef] [PubMed]


| Reference | Age | Clinical Manifestation | Infectious Agent Identified | Immunity Profile | Source of Infection | Identification Method | Antifungal Treatment | Outcome |
|---|---|---|---|---|---|---|---|---|
| Buzina et al., 2003 [4] | 19 | Restricted nasal breathing for 1 year, total nasal obstruction for more than 4 months, massive polyposis, fungus balls in sinuses | Bipolaris spicifera | Immunocompetent | Likely environmental exposure to Bipolaris spores, common in hot, dry climates (e.g., Kuwait) | Fungal cultures, morphological identification on Sabouraud agar, GMS staining, PCR and sequencing of the internal transcribed spacer region of the ribosomal gene cluster. | Systemic itraconazole (100 mg twice daily for 6 weeks, then 100 mg daily for 8 weeks), systemic steroids (betamethasone), topical steroids (budesonide), saline nasal douches | Improvement post-surgery with no further fungal invasion, observed mucosal healing after antifungal and steroid treatment. |
| Taguchi et al., 2004 [5] | 70 | Diplopia, bilateral nasal obstruction, nasal discharge | Bipolaris spicifera | Immunocompetent | Not specified | Squash cytology of the contents of the paranasal sinuses, microscopic examination of fungal hyphae, microbiological identification. | - | Allergic fungal sinusitis. |
| Taguchi et al., 2007 [8] | 70 | Bilateral nasal obstruction, purulent nasal discharge, double vision | Bipolaris spicifera | Immunocompetent | Likely environmental exposure (fungus in sinuses) | Squash cytology (Papanicolaou and Grocott stains), culture on Sheep Blood and Czapek–Dox agar. | - | Allergic fungal sinusitis. |
| Gourley DS et al., 1990 [9] | Patient 1: 16 Patient 2: 20 Patient 3: 40 | Patient 1: pansinusitis, nasal polyps, frontal pain, nasal obstruction and bilateral tecanthus. Patient 2: pansinusitis, nasal polyps, headaches, periorbital swelling, and medial orbital wall bone erosion. Patient 3: intermittent nasal congestion and headaches, as well as recurrent nasal polyps. | Bipolaris spicifera | Immunocompetent patients | Aeroallergens, probably in the environment where the patients lived (Southern United States) | The identification process was conducted through fungal culture and microscopic observation of conidia and conidiophores. A histological examination was also conducted, which includes the presence of Charcot–Leyden crystals and fungal hybrids. | - | The surgical intervention involved the excision of allergic mucin, with no evidence of tissue or bone invasion. Postoperative recovery was positive in all cases. |
| Klapper et al., 1997 [10] | Case 1: 26 | Mass in the left medial canthal area, epiphora, proptosis, reduced retropulsion | Bipolaris spicifera | Immunocompetent | Not specified | Culture on BHI agar with blood, penicillin, streptomycin; histology with Fontana–Masson stain. | No systemic antifungal therapy; surgery only | Complete resolution of symptoms after first surgery, recurrence after 2 months (on the contralateral side), resolved with second surgery. |
| D Ambrosetti et al. (2006) [11] | 50 | Paranasal sinus tumour-like lesion with orbital involvement, chronic nasal obstruction | Bipolaris spicifera | Immunocompetent | Probable fungal inhalation | Histology exam and fungal culture-positive. | No antifungal treatment after surgery | No recurrence after 2 years. |
| Coop CA, England RW, 2006 [12] | 23 | Right-sided proptosis, diplopia, nasal obstruction | Bipolaris spicifera, Aspergillus fumigatus | Immunocompetent | Chronic sinusitis with nasal polyposis | Skin testing (positive), silver stain on surgical specimen. | No specific antifungal treatment; treated with surgery and systemic corticosteroids | Improvement in proptosis and diplopia after surgery. |
| McGinnis et al. (1992) [13] | 26 | Brain abscess and allergic sinusitis | Bipolaris spicifera | Immunocompetent | Sinus infection (hematogenous spread) | Histological brain and sinus tissue exam with hematoxylin and eosin stain. | Amphotericin B + Ketoconazole | Survival. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ferraro, N.; Iskandar, E.; Pitrolo, A.M.G.; Ramus, M.; Pagella, F.; Introini, S.; Baldanti, F.; Cavanna, C. Curvularia spicifera in Non-Invasive Fungal Rhinosinusitis: Case Reports and Diagnostic Insights. Pathogens 2026, 15, 523. https://doi.org/10.3390/pathogens15050523
Ferraro N, Iskandar E, Pitrolo AMG, Ramus M, Pagella F, Introini S, Baldanti F, Cavanna C. Curvularia spicifera in Non-Invasive Fungal Rhinosinusitis: Case Reports and Diagnostic Insights. Pathogens. 2026; 15(5):523. https://doi.org/10.3390/pathogens15050523
Chicago/Turabian StyleFerraro, Nicola, Elizabeth Iskandar, Antonino Maria Guglielmo Pitrolo, Marina Ramus, Fabio Pagella, Sveva Introini, Fausto Baldanti, and Caterina Cavanna. 2026. "Curvularia spicifera in Non-Invasive Fungal Rhinosinusitis: Case Reports and Diagnostic Insights" Pathogens 15, no. 5: 523. https://doi.org/10.3390/pathogens15050523
APA StyleFerraro, N., Iskandar, E., Pitrolo, A. M. G., Ramus, M., Pagella, F., Introini, S., Baldanti, F., & Cavanna, C. (2026). Curvularia spicifera in Non-Invasive Fungal Rhinosinusitis: Case Reports and Diagnostic Insights. Pathogens, 15(5), 523. https://doi.org/10.3390/pathogens15050523







