Development and Validation of a New Scoring System (Total Leishmania Score) for Dogs with Leishmania infantum Infection Including Clinical and Laboratory Parameters
Abstract
1. Introduction
2. Materials and Methods
2.1. Development of the Scoring System for Dogs with L. infantum Infection
2.1.1. Clinical Parameters
2.1.2. Laboratory Parameters
2.2. Study Population
2.2.1. Enrolment
2.2.2. Study Appointments
2.2.3. Laboratory Methods
2.2.4. Therapeutical Management
2.3. Prospective Validation of the TLS
2.3.1. Pre-Testing
2.3.2. Evaluation of Content Validity
2.3.3. Evaluation of Reliability
2.3.4. Evaluation of Sensitivity to Change
2.4. Statistical Analysis
3. Results
3.1. Dogs
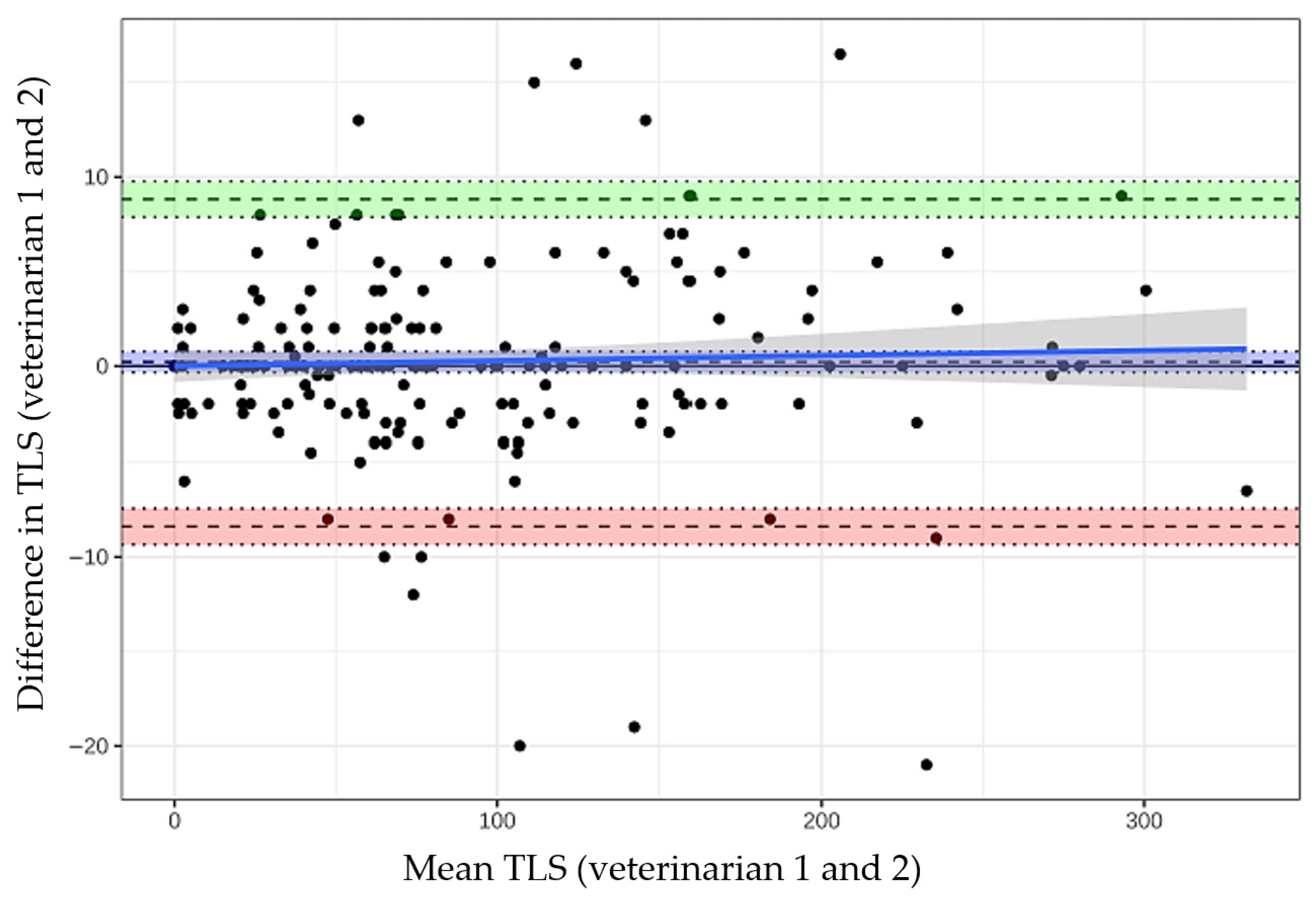
3.2. Scoring Results and Evaluation of Reliability
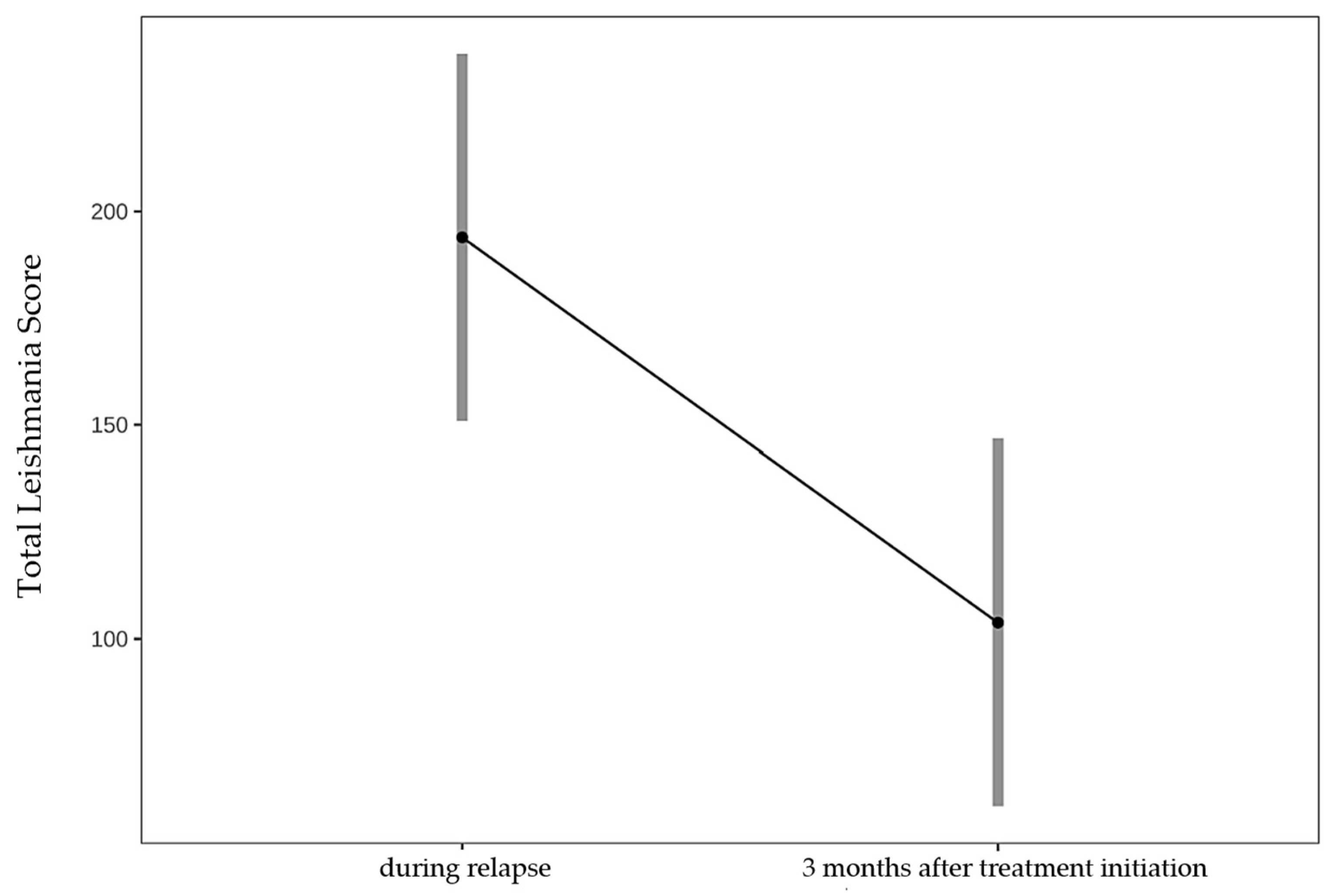
3.3. Evaluation of Sensitivity to Change
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maia, C.; Cardoso, L. Spread of Leishmania infantum in Europe with dog travelling. Vet. Parasitol. 2015, 213, 2–11. [Google Scholar] [CrossRef]
- Carvalho, B.M.; Maia, C.; Courtenay, O.; Llabres-Brustenga, A.; Lotto Batista, M.; Moirano, G.; van Daalen, K.R.; Semenza, J.C.; Lowe, R. A climatic suitability indicator to support Leishmania infantum surveillance in Europe: A modelling study. Lancet Reg. Health Eur. 2024, 43, 100971. [Google Scholar] [CrossRef]
- Solano-Gallego, L.; Miro, G.; Koutinas, A.; Cardoso, L.; Pennisi, M.G.; Ferrer, L.; Bourdeau, P.; Oliva, G.; Baneth, G.; The LeishVet Group. LeishVet guidelines for the practical management of canine leishmaniosis. Parasites Vectors 2011, 4, 86. [Google Scholar] [CrossRef]
- Paltrinieri, S.; Solano-Gallego, L.; Fondati, A.; Lubas, G.; Gradoni, L.; Castagnaro, M.; Crotti, A.; Maroli, M.; Oliva, G.; Roura, X.; et al. Guidelines for diagnosis and clinical classification of leishmaniasis in dogs. J. Am. Vet. Med. Assoc. 2010, 236, 1184–1191. [Google Scholar] [CrossRef]
- Garcia-Castro, A.; Egui, A.; Thomas, M.C.; Lopez, M.C. Humoral and Cellular Immune Response in Asymptomatic Dogs with Visceral Leishmaniasis: A Review. Vaccines 2022, 10, 947. [Google Scholar] [CrossRef]
- Ciaramella, P.; Oliva, G.; Luna, R.D.; Gradoni, L.; Ambrosio, R.; Cortese, L.; Scalone, A.; Persechino, A. A retrospective clinical study of canine leishmaniasis in 150 dogs naturally infected by Leishmania infantum. Vet. Rec. 1997, 141, 539–543. [Google Scholar] [CrossRef]
- Geisweid, K.; Mueller, R.; Sauter-Louis, C.; Hartmann, K. Prognostic analytes in dogs with Leishmania infantum infection living in a non-endemic area. Vet. Rec. 2012, 171, 399. [Google Scholar] [CrossRef] [PubMed]
- Roura, X.; Fondati, A.; Lubas, G.; Gradoni, L.; Maroli, M.; Oliva, G.; Paltrinieri, S.; Zatelli, A.; Zini, E. Prognosis and monitoring of leishmaniasis in dogs: A working group report. Vet. J. 2013, 198, 43–47. [Google Scholar] [CrossRef]
- Foglia Manzillo, V.; Di Muccio, T.; Cappiello, S.; Scalone, A.; Paparcone, R.; Fiorentino, E.; Gizzarelli, M.; Gramiccia, M.; Gradoni, L.; Oliva, G. Prospective study on the incidence and progression of clinical signs in naive dogs naturally infected by Leishmania infantum. PLoS Negl. Trop. Dis. 2013, 7, e2225. [Google Scholar] [CrossRef]
- Mancianti, F.; Gramiccia, M.; Gradoni, L.; Pieri, S. Studies on canine leishmaniasis control. 1. Evolution of infection of different clinical forms of canine leishmaniasis following antimonial treatment. Trans. R. Soc. Trop. Med. Hyg. 1988, 82, 566–567. [Google Scholar] [CrossRef]
- Oliva, G.; Scalone, A.; Foglia Manzillo, V.; Gramiccia, M.; Pagano, A.; Di Muccio, T.; Gradoni, L. Incidence and time course of Leishmania infantum infections examined by parasitological, serologic, and nested-PCR techniques in a cohort of naive dogs exposed to three consecutive transmission seasons. J. Clin. Microbiol. 2006, 44, 1318–1322. [Google Scholar] [CrossRef]
- Proverbio, D.; Spada, E.; Bagnagatti de Giorgi, G.; Perego, R.; Valena, E. Relationship between Leishmania IFAT Titer and Clinicopathological Manifestations (Clinical Score) in Dogs. BioMed Res. Int. 2014, 2014, 412808. [Google Scholar] [CrossRef] [PubMed]
- Silva, K.R.; Mendonca, V.R.; Silva, K.M.; Nascimento, L.F.; Mendes-Sousa, A.F.; Pinho, F.A.; Barral-Netto, M.; Barral, A.M.; Cruz, M.D. Scoring clinical signs can help diagnose canine visceral leishmaniasis in a highly endemic area in Brazil. Mem. Inst. Oswaldo Cruz. 2017, 112, 53–63. [Google Scholar] [CrossRef]
- Paradies, P.; Sasanelli, M.; de Caprariis, D.; Testini, G.; Traversa, D.; Lia, R.P.; Dantas-Torres, F.; Otranto, D. Clinical and laboratory monitoring of dogs naturally infected by Leishmania infantum. Vet. J. 2010, 186, 370–373. [Google Scholar] [CrossRef] [PubMed]
- Miro, G.; Oliva, G.; Cruz, I.; Canavate, C.; Mortarino, M.; Vischer, C.; Bianciardi, P. Multicentric, controlled clinical study to evaluate effectiveness and safety of miltefosine and allopurinol for canine leishmaniosis. Vet. Dermatol. 2009, 20, 397–404. [Google Scholar] [CrossRef]
- Manna, L.; Reale, S.; Vitale, F.; Gravino, A.E. Evidence for a relationship between Leishmania load and clinical manifestations. Res. Vet. Sci. 2009, 87, 76–78. [Google Scholar] [CrossRef]
- Meléndez-Lazo, A.; Ordeix, L.; Planellas, M.; Pastor, J.; Solano-Gallego, L. Clinicopathological findings in sick dogs naturally infected with Leishmania infantum: Comparison of five different clinical classification systems. Res. Vet. Sci. 2018, 117, 18–27. [Google Scholar] [CrossRef]
- Mokkink, L.B.; Terwee, C.B.; Patrick, D.L.; Alonso, J.; Stratford, P.W.; Knol, D.L.; Bouter, L.M.; de Vet, H.C. The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. J. Clin. Epidemiol. 2010, 63, 737–745. [Google Scholar] [CrossRef]
- Pereira, M.A.; Santos, R.; Oliveira, R.; Costa, L.; Prata, A.; Goncalves, V.; Roquette, M.; Vala, H.; Santos-Gomes, G. Prognostic Factors and Life Expectancy in Canine Leishmaniosis. Vet. Sci. 2020, 7, 128. [Google Scholar] [CrossRef] [PubMed]
- de Jong, M.K.; Rappoldt, A.; Broere, F.; Piek, C.J. Survival time and prognostic factors in canine leishmaniosis in a non-endemic country treated with a two-phase protocol including initial allopurinol monotherapy. Parasites Vectors 2023, 16, 163. [Google Scholar] [CrossRef]
- Plant, J.D.; Gortel, K.; Kovalik, M.; Polissar, N.L.; Neradilek, M.B. Development and validation of the Canine Atopic Dermatitis Lesion Index, a scale for the rapid scoring of lesion severity in canine atopic dermatitis. Vet. Dermatol. 2012, 23, 515-e103. [Google Scholar] [CrossRef] [PubMed]
- Members, W.N.A.G.T.F.; Freeman, L.; Becvarova, I.; Cave, N.; MacKay, C.; Nguyen, P.; Rama, B.; Takashima, G.; Tiffin, R.; Tsjimoto, H.; et al. WSAVA Nutritional Assessment Guidelines. J. Small Anim. Pract. 2011, 52, 385–396. [Google Scholar] [CrossRef]
- Agut, A.; Corzo, N.; Murciano, J.; Laredo, F.G.; Soler, M. Clinical and radiographic study of bone and joint lesions in 26 dogs with leishmaniasis. Vet. Rec. 2003, 153, 648–652. [Google Scholar] [CrossRef] [PubMed]
- International renal Interest Society (IRIS). Guidelines for Staging Chronic Kidney Disease (CKD); International Renal Interest Society (IRIS): Cambridge, UK, 2023. [Google Scholar]
- Werner, L.L.; Turnwald, G.H.; Willard, M.D. Immunologic and plasma protein disorders. In Small Animal Clinical Diagnosis by Laboratory Methods, 4th ed.; Willard, M.D., Tvedten, H., Eds.; Saunders: St. Louis, MO, USA, 2004; pp. 290–305. [Google Scholar]
- Baneth, G.; Koutinas, A.F.; Solano-Gallego, L.; Bourdeau, P.; Ferrer, L. Canine leishmaniosis—New concepts and insights on an expanding zoonosis: Part one. Trends Parasitol. 2008, 24, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Sarquis, J.; Sanz, C.R.; Raposo, L.M.; Montoya, A.; Checa, R.; Barrera, J.P.; Gomez-Velasco, C.; Sanchez, E.E.; Miro, G. Unveiling prognostic indicators in canine leishmaniosis: Two decades of evidence. Parasites Vectors 2025, 18, 467. [Google Scholar] [CrossRef] [PubMed]
- Maia, C.; Conceicao, C.; Pereira, A.; Rocha, R.; Ortuno, M.; Munoz, C.; Jumakanova, Z.; Perez-Cutillas, P.; Ozbel, Y.; Toz, S.; et al. The estimated distribution of autochthonous leishmaniasis by Leishmania infantum in Europe in 2005–2020. PLoS Negl. Trop. Dis. 2023, 17, e0011497. [Google Scholar] [CrossRef]
- Bourdeau, P.; Saridomichelakis, M.N.; Oliveira, A.; Oliva, G.; Kotnik, T.; Galvez, R.; Foglia Manzillo, V.; Koutinas, A.F.; Pereira da Fonseca, I.; Miro, G. Management of canine leishmaniosis in endemic SW European regions: A questionnaire-based multinational survey. Parasites Vectors 2014, 7, 110. [Google Scholar] [CrossRef]
- Kaempfle, M.; Hartmann, K.; Bergmann, M. Treatment of Leishmania infantum Infections in Dogs. Microorganisms 2025, 13, 1018. [Google Scholar] [CrossRef]
- Roura, X.; Cortadellas, O.; Day, M.J.; Benali, S.L.; Zatelli, A.; Canine Leishmaniosis Working, G. Canine leishmaniosis and kidney disease: Q&A for an overall management in clinical practice. J. Small Anim. Pract. 2021, 62, 3. [Google Scholar] [CrossRef]
- Torres, M.; Pastor, J.; Roura, X.; Tabar, M.D.; Espada, Y.; Font, A.; Balasch, J.; Planellas, M. Adverse urinary effects of allopurinol in dogs with leishmaniasis. J. Small Anim. Pract. 2016, 57, 299–304. [Google Scholar] [CrossRef]
- Oliveira, S.C.; Arenas, C.; Dominguez-Ruiz, M.; Prosper, E.; Dias, M.J.; Leal, R.O. Characterisation and evaluation of predisposing factors for the development of xanthinuria in dogs with leishmaniosis under allopurinol therapy. Parasites Vectors 2025, 18, 98. [Google Scholar] [CrossRef]
- Kaempfle, M.; Dorsch, R.; Zablotski, Y.; Hartmann, K.; Bergmann, M. Importance of Different Parameters for Monitoring Dogs with Leishmania infantum Infections in a Non-Endemic Country. Pathogens 2025, 14, 1282. [Google Scholar] [CrossRef] [PubMed]
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Solano-Gallego, L.; Koutinas, A.; Miro, G.; Cardoso, L.; Pennisi, M.G.; Ferrer, L.; Bourdeau, P.; Oliva, G.; Baneth, G. Directions for the diagnosis, clinical staging, treatment and prevention of canine leishmaniosis. Vet. Parasitol. 2009, 165, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Sarquis, J.; Raposo, L.M.; Sanz, C.R.; Montoya, A.; Barrera, J.P.; Checa, R.; Perez-Montero, B.; Rodriguez, M.L.F.; Miro, G. Relapses in canine leishmaniosis: Risk factors identified through mixed-effects logistic regression. Parasites Vectors 2024, 17, 357. [Google Scholar] [CrossRef] [PubMed]
- Kahraman, N.; Clauser, B.E.; Margolis, M.J. A comparison of alternative item weighting strategies on the data gathering component of a clinical skills performance assessment. Acad. Med. 2008, 83, S72–S75. [Google Scholar] [CrossRef]
- Freeman, L.M.; Michel, K.E.; Zanghi, B.M.; Vester Boler, B.M.; Fages, J. Evaluation of the use of muscle condition score and ultrasonographic measurements for assessment of muscle mass in dogs. Am. J. Vet. Res. 2019, 80, 595–600. [Google Scholar] [CrossRef]
- Freeman, L.M.; Michel, K.E.; Zanghi, B.M.; Vester Boler, B.M.; Fages, J. Usefulness of muscle condition score and ultrasonographic measurements for assessment of muscle mass in cats with cachexia and sarcopenia. Am. J. Vet. Res. 2020, 81, 254–259. [Google Scholar] [CrossRef]
- Cugmas, B.; Olivry, T. Evaluation of skin erythema severity by dermatoscopy in dogs with atopic dermatitis. Vet. Dermatol. 2021, 32, 183-e146. [Google Scholar] [CrossRef]
- Khantavee, N.; Chanthick, C.; Sookrung, N.; Prapasarakul, N. Antibody levels to Malassezia pachydermatis and Staphylococcus pseudintermedius in atopic dogs and their relationship with lesion scores. Vet. Dermatol. 2020, 31, 111–115. [Google Scholar] [CrossRef] [PubMed]
- German, A.L.; Fleming, K.; Kaye, P.; Davies, S.; Goldin, R.; Hubscher, S.G.; Tiniakos, D.; McGregor, A.; Wyatt, J.I. Can reference images improve interobserver agreement in reporting liver fibrosis? J. Clin. Pathol. 2018, 71, 368–371. [Google Scholar] [CrossRef] [PubMed]
- Pantaleon, L. Why measuring outcomes is important in health care. J. Vet. Intern. Med. 2019, 33, 356–362. [Google Scholar] [CrossRef]
- Koutinas, A.F.; Polizopoulou, Z.S.; Saridomichelakis, M.N.; Argyriadis, D.; Fytianou, A.; Plevraki, K.G. Clinical considerations on canine visceral leishmaniasis in Greece: A retrospective study of 158 cases (1989–1996). J. Am. Anim. Hosp. Assoc. 1999, 35, 376–383. [Google Scholar] [CrossRef]
- Mylonakis, M.E.; Saridomichelakis, M.N.; Lazaridis, V.; Leontides, L.S.; Kostoulas, P.; Koutinas, A.F. A retrospective study of 61 cases of spontaneous canine epistaxis (1998 to 2001). J. Small Anim. Pract. 2008, 49, 191–196. [Google Scholar] [CrossRef]
- Koutinas, A.F.; Koutinas, C.K. Pathologic mechanisms underlying the clinical findings in canine leishmaniasis due to Leishmania infantum/chagasi. Vet. Pathol. 2014, 51, 527–538. [Google Scholar] [CrossRef]
- Saridomichelakis, M.N.; Baneth, G.; Colombo, S.; Dantas-Torres, F.; Ferrer, L.; Fondati, A.; Miro, G.; Ordeix, L.; Otranto, D.; Noli, C. World Association for Veterinary Dermatology Consensus Statement for Diagnosis, and Evidence-Based Clinical Practice Guidelines for Treatment and Prevention of Canine Leishmaniosis. Vet. Dermatol. 2025, 36, 723–787. [Google Scholar] [CrossRef]
- Vamvakidis, C.D.; Koutinas, A.F.; Kanakoudis, G.; Georgiadis, G.; Saridomichelakis, M. Masticatory and skeletal muscle myositis in canine leishmaniasis (Leishmania infantum). Vet. Rec. 2000, 146, 698–703. [Google Scholar] [CrossRef]
- Xu, J.J.; Campbell, G.; Alsaffar, H.; Brandt, M.G.; Doyle, P.C.; Glicksman, J.T.; Fung, K. Lymphadenopathy: Defining a palpable lymph node. Head Neck 2015, 37, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Verhoeven, R.L.J.; Leoncini, F.; Slotman, J.; de Korte, C.; Trisolini, R.; van der Heijden, E.; Group, E.p.S. Accuracy and Reproducibility of Endoscopic Ultrasound B-Mode Features for Observer-Based Lymph Nodal Malignancy Prediction. Respiration 2021, 100, 1088–1096. [Google Scholar] [CrossRef]
- Burns, G.O.; Scrivani, P.V.; Thompson, M.S.; Erb, H.N. Relation between age, body weight, and medial retropharyngeal lymph node size in apparently healthy dogs. Vet. Radiol. Ultrasound 2008, 49, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Lima, W.G.; Michalick, M.S.; de Melo, M.N.; Luiz Tafuri, W.; Luiz Tafuri, W. Canine visceral leishmaniasis: A histopathological study of lymph nodes. Acta Trop. 2004, 92, 43–53. [Google Scholar] [CrossRef]
- Ling, G.V.; Ruby, A.L.; Harrold, D.R.; Johnson, D.L. Xanthine-containing urinary calculi in dogs given allopurinol. J. Am. Vet. Med. Assoc. 1991, 198, 1935–1940. [Google Scholar] [CrossRef]
- Buckley, C.M.; Hawthorne, A.; Colyer, A.; Stevenson, A.E. Effect of dietary water intake on urinary output, specific gravity and relative supersaturation for calcium oxalate and struvite in the cat. Br. J. Nutr. 2011, 106, S128–S130. [Google Scholar] [CrossRef]
- Carreira, L.M.; Monteiro, P.; Azevedo, P. Total Proteins, β- and γ-globulins as efficacy therapy response indicators in dogs infected with Leishmania infantum—A Review. J. Vet. Healthc. 2017, 1, 1–11. [Google Scholar] [CrossRef][Green Version]
- de Carvalho, C.A.; Hiramoto, R.M.; Meireles, L.R.; de Andrade, H.F., Jr. Understanding hypergammaglobulinemia in experimental or natural visceral leishmaniasis. Parasite Immunol. 2024, 46, e13021. [Google Scholar] [CrossRef] [PubMed]
- Silva-O’Hare, J.; de Oliveira, I.S.; Klevorn, T.; Almeida, V.A.; Oliveira, G.G.; Atta, A.M.; de Freitas, L.A.; Dos-Santos, W.L. Disruption of Splenic Lymphoid Tissue and Plasmacytosis in Canine Visceral Leishmaniasis: Changes in Homing and Survival of Plasma Cells. PLoS ONE 2016, 11, e0156733. [Google Scholar] [CrossRef]
- Ceron, J.J.; Pardo-Marin, L.; Caldin, M.; Furlanello, T.; Solano-Gallego, L.; Tecles, F.; Bernal, L.; Baneth, G.; Martinez-Subiela, S. Use of acute phase proteins for the clinical assessment and management of canine leishmaniosis: General recommendations. BMC Vet. Res. 2018, 14, 196. [Google Scholar] [CrossRef]
- Rotondi, M.A.; Donner, A. A confidence interval approach to sample size estimation for interobserver agreement studies with multiple raters and outcomes. J. Clin. Epidemiol. 2012, 65, 778–784. [Google Scholar] [CrossRef] [PubMed]




| Parameter | 0 | 1 | 2 | 3 | Score (0–3) 1 | Weighting Factor 2 | Adjusted Score 3 | |
|---|---|---|---|---|---|---|---|---|
| clinical parameters | ||||||||
| general condition | normal | mildly reduced | moderately reduced | severely reduced | 20 | |||
| appetite | normal | mildly reduced | moderately reduced | anorexia | 20 | |||
| lymph node enlargement | right mandibular ln. | not present | mild | moderate | severe | 1 | ||
| left mandibular ln. | not present | mild | moderate | severe | 1 | |||
| right cervical ln. | not present | mild | moderate | severe | 1 | |||
| left cervical ln. | not present | mild | moderate | severe | 1 | |||
| right inguinal ln. | not present | mild | moderate | severe | 1 | |||
| left inguinal ln. | not present | mild | moderate | severe | 1 | |||
| right popliteal ln. | not present | mild | moderate | severe | 1 | |||
| left popliteal ln. | not present | mild | moderate | severe | 1 | |||
| any other ln. | not present | mild | moderate | severe | 2 | |||
| skin ulcers * | head & pinna | not present | mild | moderate | severe | 2.5 | ||
| forelimb | not present | mild | moderate | severe | 2.5 | |||
| hindlimb | not present | mild | moderate | severe | 2.5 | |||
| torso | not present | mild | moderate | severe | 2.5 | |||
| skin scaling/exfoliation/ seborrheic dermatitis/ skin papules/nodules * | head & pinna | not present | mild | moderate | severe | 2.5 | ||
| forelimb | not present | mild | moderate | severe | 2.5 | |||
| hindlimb | not present | mild | moderate | severe | 2.5 | |||
| torso | not present | mild | moderate | severe | 2.5 | |||
| conjunctivitis/ blepharitis/keratitis | right eye | not present | mild | moderate | severe | 4 | ||
| left eye | not present | mild | moderate | severe | 4 | |||
| uveitis | right eye | not present | mild | moderate | severe | 6 | ||
| left eye | not present | mild | moderate | severe | 6 | |||
| arthritis | forelimb right | not present | joint swelling | pain on palpation/ROM | joint swelling + pain | 2.5 | ||
| forelimb left | not present | joint swelling | pain on palpation/ROM | joint swelling + pain | 2.5 | |||
| hindlimb right | not present | joint swelling | pain on palpation/ROM | joint swelling + pain | 2.5 | |||
| hindlimb left | not present | joint swelling | pain on palpation/ROM | joint swelling + pain | 2.5 | |||
| muscle atrophy ** | head | not present | mild | moderate | severe | 2 | ||
| forelimb right | not present | mild | moderate | severe | 2 | |||
| forelimb left | not present | mild | moderate | severe | 2 | |||
| hindlimb right | not present | mild | moderate | severe | 2 | |||
| hindlimb left | not present | mild | moderate | severe | 2 | |||
| epistaxis | not present | sporadic | frequent | persistent | 10 | |||
| laboratory parameters | ||||||||
| complete blood count | anaemia (HCT) | not present (ref: 37.3–61.7%) | mild (<37.3–30%) | moderate (<30–20%) | severe (<20%) | 15 | ||
| thrombocytopenia (PLT) | not present (ref: 148–484 × 109/L) | mild (<148–110 × 109/L) | moderate (<110–60 × 109/L) | severe (<60 × 109/L) | 15 | |||
| neutropenia (NEU) | not present (ref: 2.95–11.64 × 109/L) | mild (<2.95–1.5 × 109/L) | moderate (<1.5–1.0 × 109/L) | severe (<1.0 × 109/L) | 15 | |||
| lymphopenia (LYM) | not present (ref: 1.05–5.1 × 109/L) | mild (<1.05–0.8 × 109/L) | moderate (<0.8–0.5 × 109/L) | severe (<0.5 × 109/L) | 15 | |||
| serum biochemistry | renal azotaemia (creatinine) | not present (ref: 44–125 µmol/L) | mild (>125–250 µmol/L) | moderate (>250–440 µmol/L) | severe (>440 µmol/L) | 40 | ||
| hyperglobulinaemia (Glob) | not present (ref: 2.4–4.3 g/dL) | mild (>4.3–5 g/dL) | moderate (>5–6 g/dL) | severe (>6 g/dL) | 20 | |||
| others | proteinuria (UPC) | not present (0–0.5) | mild (0.6–2) | moderate (2.1–4) | severe (>4) | 40 | ||
| antibody level (ELISA) | normal (0–12 TU) | mildly elevated (12.1–24.9 TU) | moderately elevated (25–40 TU) | severely elevated (>40 TU) | 20 | |||
| Total Leishmania Score = sum of all adjusted scores (range: 0—900) | ||||||||
| Parameter | 0 | 1 | 2 | 3 | ||
|---|---|---|---|---|---|---|
| clinical parameters | general condition | 474 (97.1) | 14 (2.9) | 0 (0) | 0 (0) | |
| appetite | 486 (99.6) | 2 (0.4) | 0 (0) | 0 (0) | ||
| lymph node enlargement | right mandibular ln. | 392 (80.4) | 70 (14.3) | 24 (4.9) | 2 (0.4) | |
| left mandibular ln. | 398 (81.6) | 68 (13.9) | 22 (4.5) | 0 (0) | ||
| right cervical ln. | 452 (92.7) | 28 (5.7) | 8 (1.6) | 0 (0) | ||
| left cervical ln. | 452 (92.7) | 26 (5.3) | 10 (2.1) | 0 (0) | ||
| right inguinal ln. | 484 (99.2) | 4 (0.8) | 0 (0) | 0 (0) | ||
| left inguinal ln. | 484 (99.2) | 4 (0.8) | 0 (0) | 0 (0) | ||
| right popliteal ln. | 316 (64.8) | 132 (27.0) | 38 (7.8) | 2 (0.4) | ||
| left popliteal ln. | 316 (64.8) | 140 (28.7) | 28 (5.7) | 4 (0.8) | ||
| any other ln. | 472 (96.8) | 8 (1.6) | 6 (1.2) | 2 (0.4) | ||
| skin ulcers * | head & pinna | 450 (92.3) | 22 (4.5) | 8 (1.6) | 8 (1.6) | |
| forelimb | 476 (97.6) | 2 (0.4) | 6 (1.2) | 4 (0.8) | ||
| hindlimb | 478 (98.0) | 10 (2.0) | 0 (0) | 0 (0) | ||
| torso | 484 (99.2) | 4 (0.8) | 0 (0) | 0 (0) | ||
| skin scaling/exfoliation/ seborrheic dermatitis/ skin papules/nodules * | head & pinna | 374 (76.7) | 108 (22.1) | 4 (0.8) | 2 (0.4) | |
| forelimb | 458 (93.9) | 30 (6.1) | 0 (0) | 0 (0) | ||
| hindlimb | 456 (93.4) | 32 (6.6) | 0 (0) | 0 (0) | ||
| Torso | 430 (88.1) | 54 (11.1) | 2 (0.4) | 2 (0.4) | ||
| conjunctivitis/ blepharitis/keratitis | right eye | 445 (91.2) | 37 (7.6) | 6 (1.2) | 0 (0) | |
| left eye | 446 (91.4) | 32 (6.6) | 10 (2.0) | 0 (0) | ||
| uveitis | right eye | 474 (97.1) | 0 (0) | 0 (0) | 14 (2.9) | |
| left eye | 472 (96.7) | 0 (0) | 2 (0.4) | 14 (2.9) | ||
| arthritis | forelimb right | 464 (95.2) | 10 (2.0) | 10 (2.0) | 4 (0.8) | |
| forelimb left | 464 (95.1) | 14 (2.9) | 8 (1.6) | 2 (0.4) | ||
| hindlimb right | 464 (95.1) | 12 (2.5) | 10 (2.0) | 2 (0.4) | ||
| hindlimb left | 462 (94.8) | 10 (2.0) | 10 (2.0) | 6 (1.2) | ||
| muscle atrophy ** | head | 464 (95.1) | 22 (4.5) | 2 (0.4) | 0 (0) | |
| forelimb right | 466 (95.5) | 18 (3.7) | 4 (0.8) | 0 (0) | ||
| forelimb left | 472 (96.7) | 14 (2.9) | 2 (0.4) | 0 (0) | ||
| hindlimb right | 456 (93.5) | 24 (4.9) | 6 (1.2) | 2 (0.4) | ||
| hindlimb left | 454 (93.1) | 26 (5.3) | 6 (1.2) | 2 (0.4) | ||
| epistaxis | 486 (99.6) | 2 (0.4) | 0 (0) | 0 (0) | ||
| laboratory parameters | complete blood count | anaemia (HCT) | 392 (80.3) | 78 (16.0) | 18 (3.7) | 0 (0) |
| thrombocytopenia (PLT) | 464 (95.1) | 16 (3.3) | 6 (1.2) | 2 (0.4) | ||
| neutropenia (NEU) | 408 (83.6) | 80 (16.4) | 0 (0) | 0 (0) | ||
| lymphopenia (LYM) | 440 (90.2) | 26 (5.3) | 20 (4.1) | 2 (0.4) | ||
| serum biochemistry | renal azotaemia (creatinine) | 454 (93.1) | 26 (5.3) | 6 (1.2) | 2 (0.4) | |
| hyperglobulinaemia (Glob) | 422 (86.5) | 34 (7.0) | 18 (3.7) | 14 (8.2) | ||
| others | proteinuria (UPC) | 366 (75.0) | 64 (13.1) | 18 (3.7) | 40 (8.2) | |
| antibody level (ELISA) | 96 (19.7) | 114 (23.4) | 92 (18.9) | 186 (38.0) |
| Parameter | κ | Accuracy | 95% CI | McNemar’s p Value | ||
|---|---|---|---|---|---|---|
| clinical parameters | general condition | 1.00 | 1.00 | (0.99–1.00) | - | |
| appetite | 1.00 | 1.00 | (0.99–1.00) | - | ||
| lymph node enlargement | right mandibular ln. | 0.46 | 0.83 | (0.78–0.88) | - | |
| left mandibular ln. | 0.38 | 0.82 | (0.76–0.86) | - | ||
| right cervical ln. | 0.46 | 0.91 | (0.86–0.94) | 0.06 | ||
| left cervical ln. | 0.51 | 0.92 | (0.87–0.95) | 0.02 | ||
| right inguinal ln. | 0.21 | 0.97 | (0.94–0.99) | 0.13 | ||
| left inguinal ln. | 0.16 | 0.96 | (0.93–0.98) | 0.03 | ||
| right popliteal ln. | 0.54 | 0.78 | (0.72–0.83) | - | ||
| left popliteal ln. | 0.54 | 0.78 | (0.72–0.83) | - | ||
| any other ln. | 0.90 | 0.99 | (0.97–1.00) | - | ||
| skin ulcers | head & pinna | 0.86 | 0.98 | (0.95–0.99) | - | |
| forelimb | 1.00 | 1.00 | (0.99–1.00) | - | ||
| hindlimb | 0.44 | 0.98 | (0.95–0.99) | - | ||
| torso | 0.67 | 1.00 | (0.98–1.00) | 1.00 | ||
| skin scaling/exfoliation/ seborrheic dermatitis/ skin papules/nodules | head & pinna | 0.77 | 0.92 | (0.87–0.95) | - | |
| forelimb | 0.53 | 0.95 | (0.92–0.98) | - | ||
| hindlimb | 0.61 | 0.95 | (0.92–0.98) | - | ||
| torso | 0.48 | 0.90 | (0.86–0.94) | - | ||
| conjunctivitis/ blepharitis/keratitis | right eye | 0.70 | 0.95 | (0.92–0.98) | 0.54 | |
| left eye | 0.73 | 0.96 | (0.92–0.98) | 0.55 | ||
| uveitis | right eye | 0.93 | 1.00 | (0.98–1.00) | 1.00 | |
| left eye | 0.82 | 0.99 | (0.97–1.00) | - | ||
| arthritis | forelimb right | 0.68 | 0.98 | (0.95–0.99) | - | |
| forelimb left | 0.85 | 0.99 | (0.97–1.00) | - | ||
| hindlimb right | 0.58 | 0.97 | (0.94–0.99) | - | ||
| hindlimb left | 0.54 | 0.96 | (0.93–0.98) | - | ||
| muscle atrophy | head | 0.90 | 0.99 | (0.97–1.00) | - | |
| forelimb right | 0.51 | 0.96 | (0.93–0.98) | - | ||
| forelimb left | 0.64 | 0.98 | (0.95–0.99) | - | ||
| hindlimb right | 0.68 | 0.96 | (0.93–0.98) | - | ||
| hindlimb left | 0.64 | 0.95 | (0.92–0.98) | - | ||
| epistaxis | 0.00 | 1.00 | (0.98–1.00) | 1.00 | ||
| laboratory parameters | complete blood count | anaemia (HCT) | 1.00 | 1.00 | (0.99–1.00) | - |
| thrombocytopenia (PLT) | 1.00 | 1.00 | (0.99–1.00) | - | ||
| neutropenia (NEU) | 1.00 | 1.00 | (0.99–1.00) | - | ||
| lymphopenia (LYM) | 1.00 | 1.00 | (0.99–1.00) | - | ||
| serum biochemistry | renal azotaemia (creatinine) | 1.00 | 1.00 | (0.99–1.00) | - | |
| hyperglobulinaemia (Glob) | 1.00 | 1.00 | (0.99–1.00) | - | ||
| others | proteinuria (UPC) | 1.00 | 1.00 | (0.99–1.00) | - | |
| antibody level (ELISA) | 1.00 | 1.00 | (0.99–1.00) | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Voelk, J.C.; Kaempfle, M.; Dorsch, R.; Geisen, V.; Mueller, R.S.; Lauer, S.K.; Zablotski, Y.; Hartmann, K.; Bergmann, M. Development and Validation of a New Scoring System (Total Leishmania Score) for Dogs with Leishmania infantum Infection Including Clinical and Laboratory Parameters. Pathogens 2026, 15, 517. https://doi.org/10.3390/pathogens15050517
Voelk JC, Kaempfle M, Dorsch R, Geisen V, Mueller RS, Lauer SK, Zablotski Y, Hartmann K, Bergmann M. Development and Validation of a New Scoring System (Total Leishmania Score) for Dogs with Leishmania infantum Infection Including Clinical and Laboratory Parameters. Pathogens. 2026; 15(5):517. https://doi.org/10.3390/pathogens15050517
Chicago/Turabian StyleVoelk, Julia C., Melanie Kaempfle, Roswitha Dorsch, Vera Geisen, Ralf S. Mueller, Susanne K. Lauer, Yury Zablotski, Katrin Hartmann, and Michèle Bergmann. 2026. "Development and Validation of a New Scoring System (Total Leishmania Score) for Dogs with Leishmania infantum Infection Including Clinical and Laboratory Parameters" Pathogens 15, no. 5: 517. https://doi.org/10.3390/pathogens15050517
APA StyleVoelk, J. C., Kaempfle, M., Dorsch, R., Geisen, V., Mueller, R. S., Lauer, S. K., Zablotski, Y., Hartmann, K., & Bergmann, M. (2026). Development and Validation of a New Scoring System (Total Leishmania Score) for Dogs with Leishmania infantum Infection Including Clinical and Laboratory Parameters. Pathogens, 15(5), 517. https://doi.org/10.3390/pathogens15050517






