Potential Activity of 6-Pentyl-α-pyrone as an Antiviral for Bovine Coronavirus
Abstract
1. Introduction
2. Materials and Methods
2.1. Production, Isolation and Identification of 6PP
2.2. Cell Cultures and 6PP
2.3. Virus Production
2.4. Cytotoxicity Time-Course
2.5. Antiviral Activity
2.5.1. Viral Inhibition Assay
2.5.2. Viral Titer Inhibition Assay (Reed and Muench)
2.6. Examination of Cell Morphology and Cell Cytoskeleton
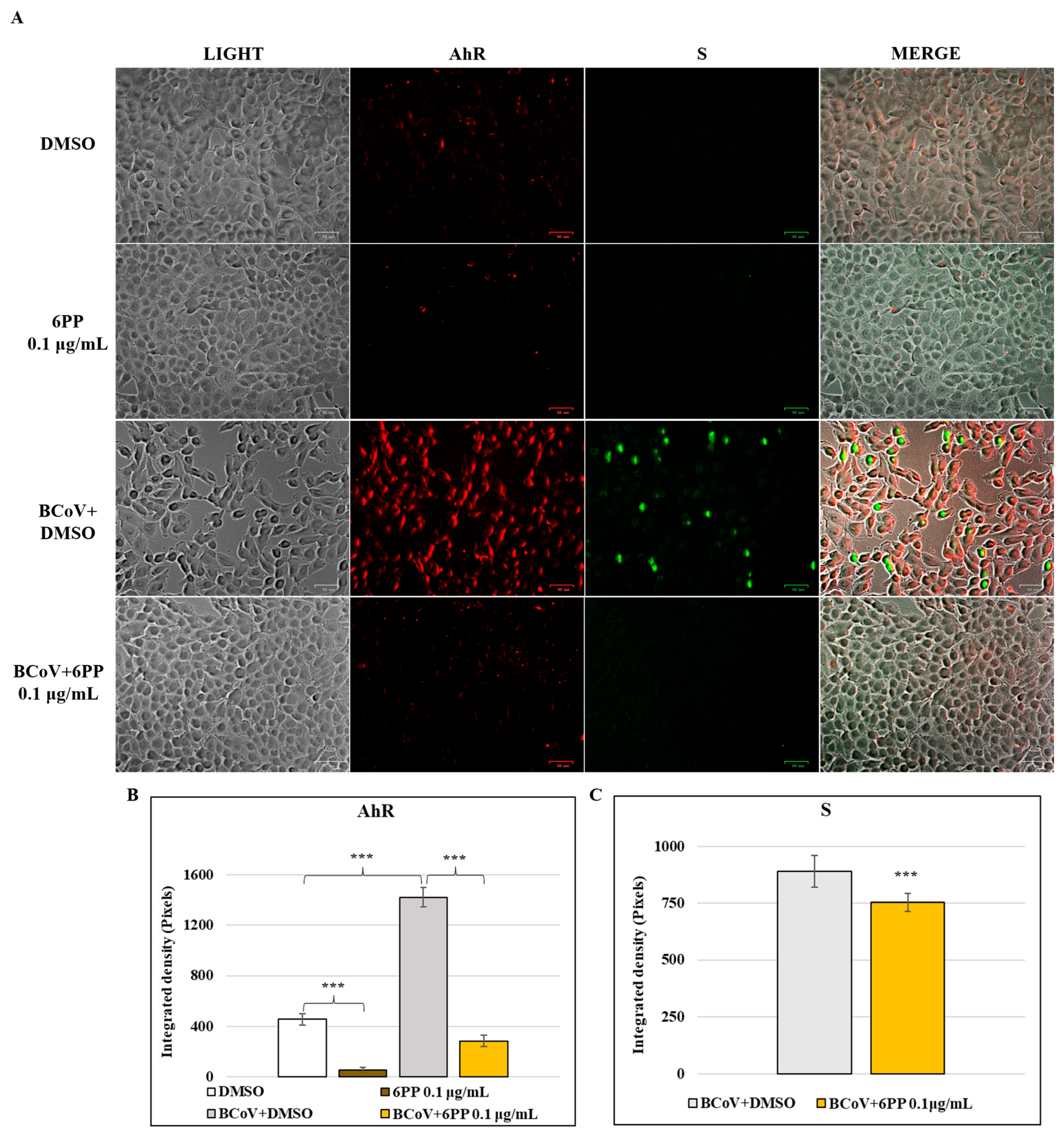
2.7. Immunofluorescence
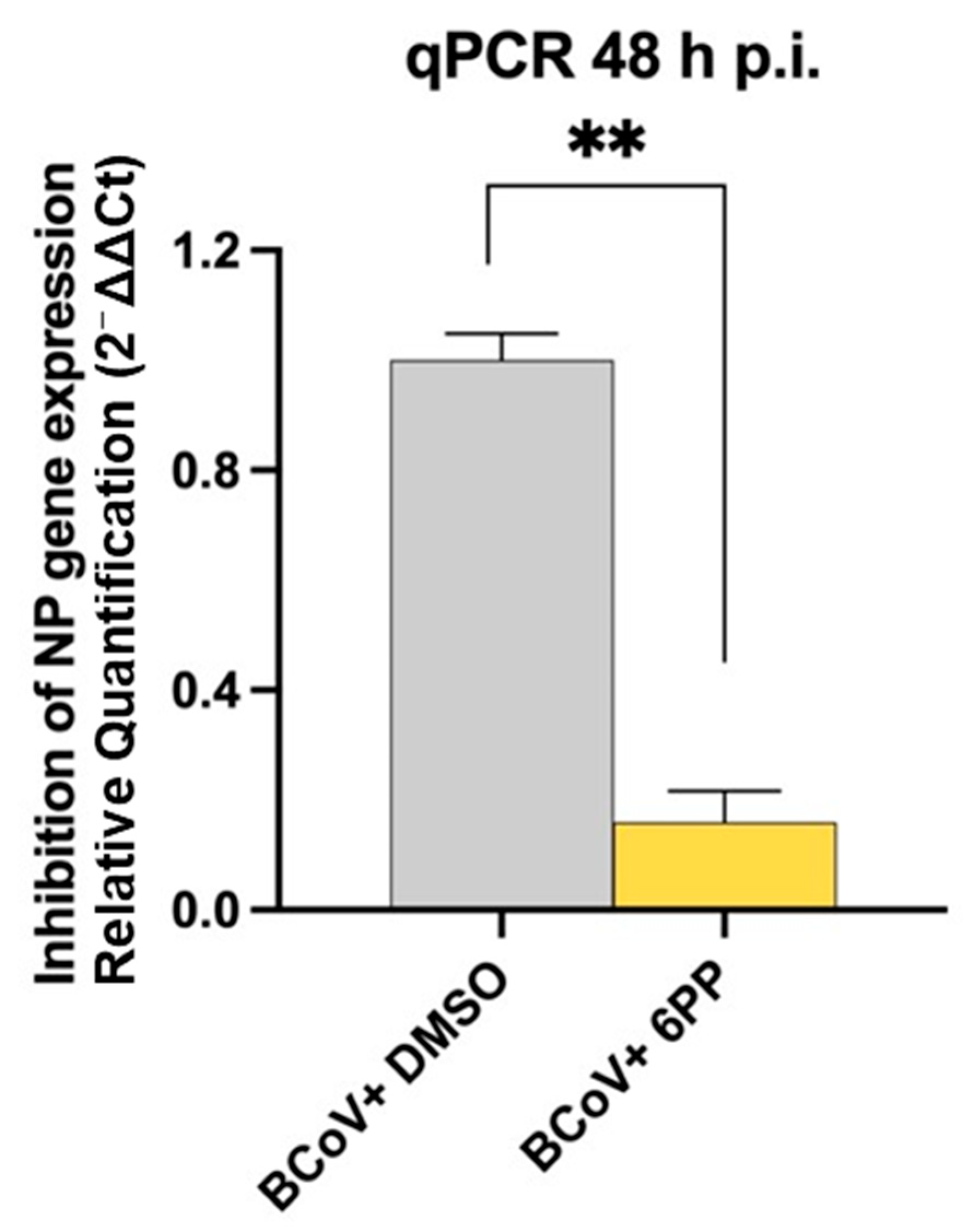
2.8. Real-Time PCR
2.9. LysoRed Staining
2.10. Three-Dimensional Structure Prediction and Molecular Docking Studies
2.11. Statistical Analysis
3. Results
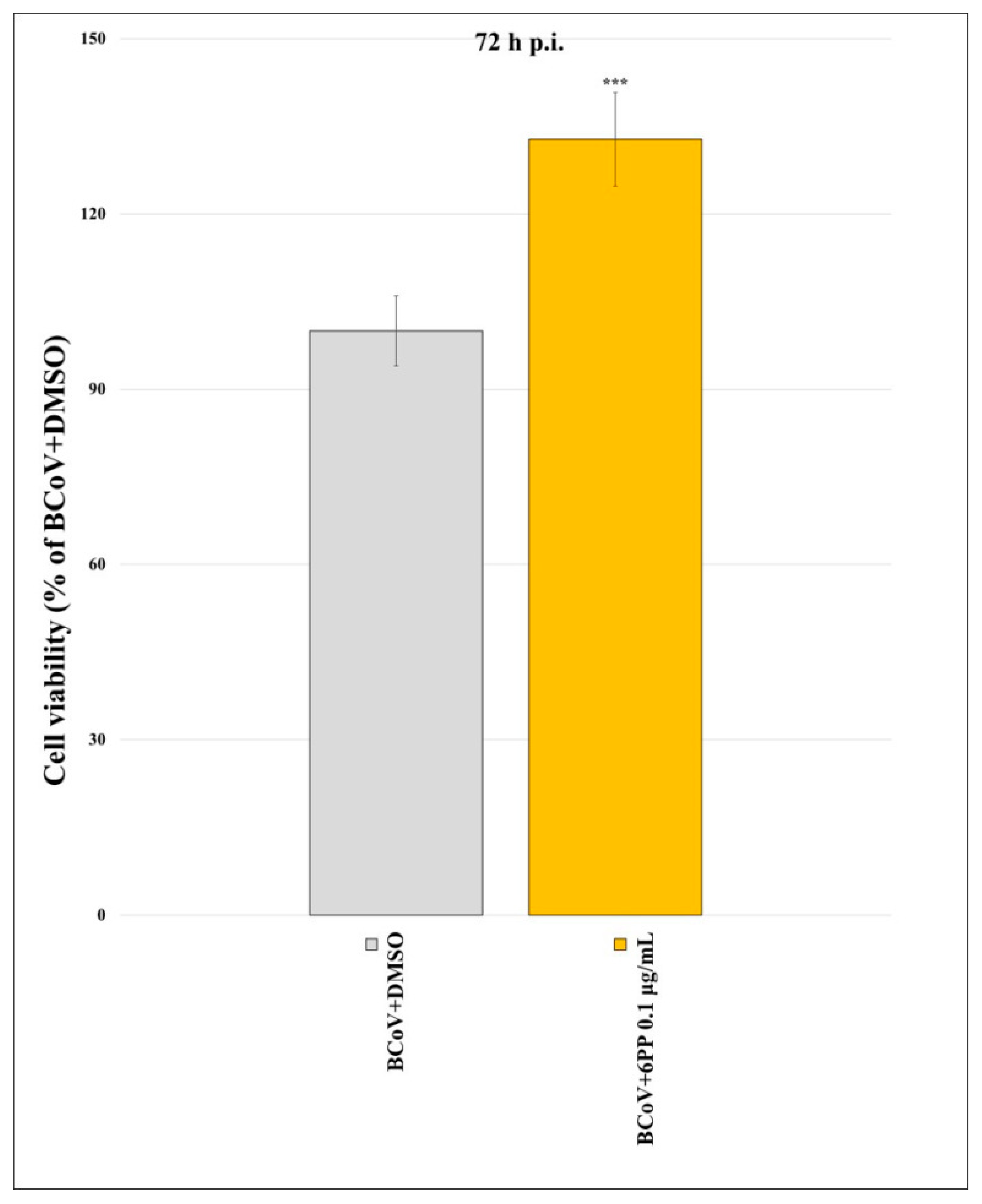
3.1. Determination of Cytotoxicity Time-Course
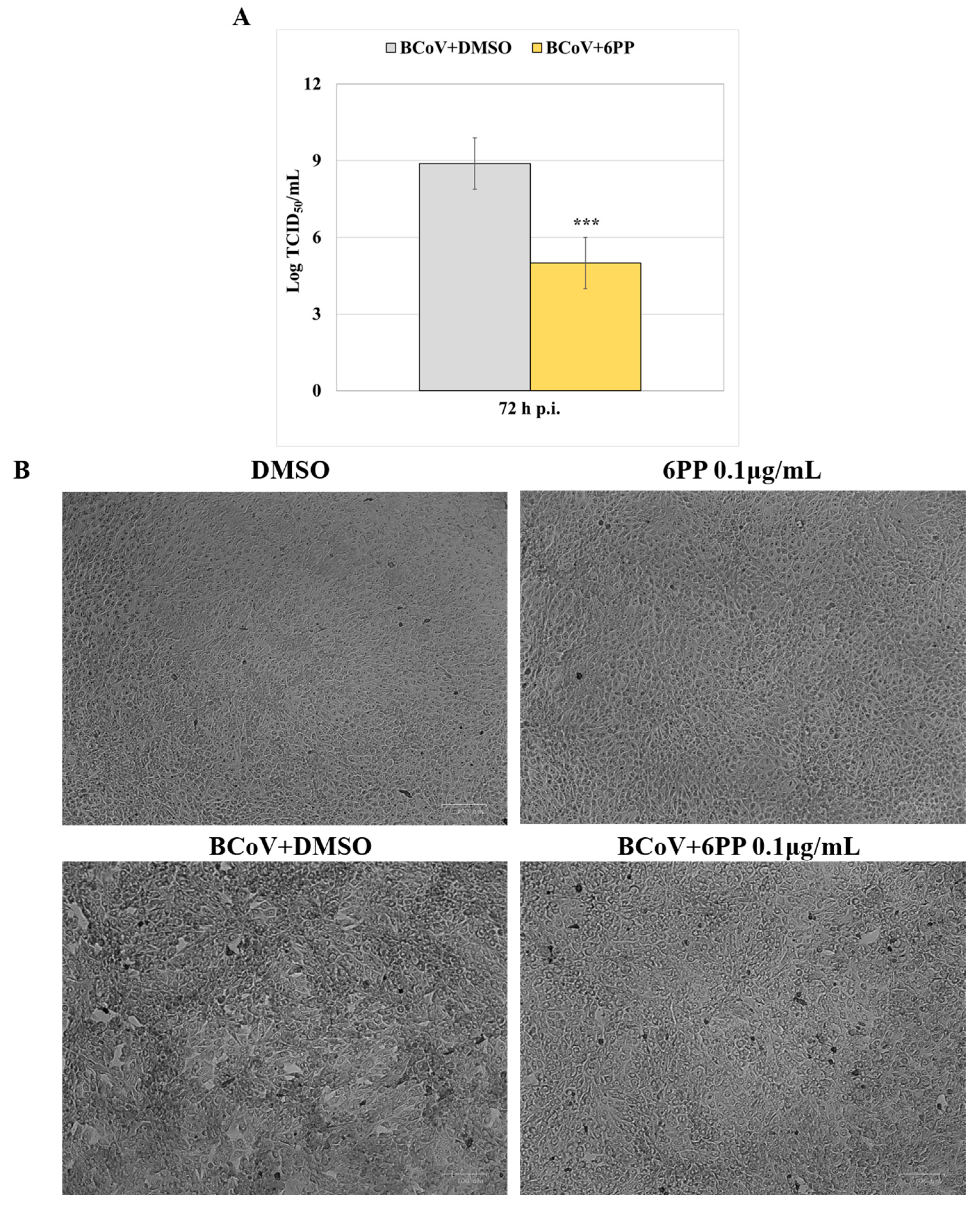
3.2. Antiviral Activity of 6PP
3.3. 6PP Reduces Signs of Cell Death Morphology During BCoV Infection in MDBK Cells
3.4. 6PP Causes a Reduction in the Expression of Cellular AhR Signaling During BCoV Infection
3.5. 6PP Deacidifies Lysosomes During BCoV Infection in MDBK Cells
3.6. Building the bAhR-6PP Complex by Molecular Docking
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Saif, L.J.; Jung, K. Comparative Pathogenesis of Bovine and Porcine Respiratory Coronaviruses in the Animal Host Species and SARS-CoV-2 in Humans. J. Clin. Microbiol. 2020, 58, e01355-20. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Know Your Treatment Options for COVID-19. Available online: https://www.fda.gov/consumers/consumer-updates/know-your-treatment-options-covid-19 (accessed on 7 October 2025).
- Rahmah, L.; Abarikwu, S.O.; Arero, A.G.; Essouma, M.; Jibril, A.T.; Fal, A.; Flisiak, R.; Makuku, R.; Marquez, L.; Mohamed, K.; et al. Oral antiviral treatments for COVID-19: Opportunities and challenges. Pharmacol. Rep. 2022, 74, 1255–1278. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yang, Q.; Ran, T.; Zhang, G.; Li, W.; Zhou, P.; Tang, J.; Dai, M.; Zhong, J.; Chen, H.; et al. Discovery of orally bioavailable SARS-CoV-2 papain-like protease inhibitor as a potential treatment for COVID-19. Nat. Commun. 2024, 15, 10169. [Google Scholar] [CrossRef] [PubMed]
- Barghash, R.F.; Gemmati, D.; Awad, A.M.; Elbakry, M.M.M.; Tisato, V.; Awad, K.; Singh, A.V. Navigating the COVID-19 Therapeutic Landscape: Unveiling Novel Perspectives on FDA-Approved Medications, Vaccination Targets, and Emerging Novel Strategies. Molecules 2024, 29, 5564. [Google Scholar] [CrossRef]
- Schreiber, A.; Ludwig, S. Host-targeted antivirals against SARS-CoV-2 in clinical development—Prospect or disappointment? Antivir. Res. 2025, 235, 106101. [Google Scholar] [CrossRef]
- Gu, X.; Zheng, M.; Gao, Y.; Lin, S.; Zhang, X.; Chen, C.; Zhu, H.; Sun, W.; Zhang, Y. Overview of host-directed antiviral targets for future research and drug development. Acta Pharm. Sin. B 2025, 15, 1723–1751. [Google Scholar] [CrossRef]
- Grycova, A.; Vyhlidalova, B.; Dvorak, Z. The role of aryl hydrocarbon receptor in antiviral immunity: A focus on RNA viruses. Crit. Rev. Microbiol. 2025, 51, 1255–1269. [Google Scholar] [CrossRef]
- Barreira-Silva, P.; Lian, Y.; Kaufmann, S.H.E.; Moura-Alves, P. The role of the AHR in host-pathogen interactions. Nat. Rev. Immunol. 2025, 25, 178–194. [Google Scholar] [CrossRef]
- Hu, J.; Ding, Y.; Liu, W.; Liu, S. When AHR signaling pathways meet viral infections. Cell Commun. Signal. 2023, 21, 42. [Google Scholar] [CrossRef]
- Lawrence, B.P.; Vorderstrasse, B.A. New insights into the aryl hydrocarbon receptor as a modulator of host responses to infection. Semin. Immunopathol. 2013, 35, 615–626. [Google Scholar] [CrossRef]
- Healey, A.M.; Fenner, K.N.; O’Dell, C.T.; Lawrence, B.P. Aryl hydrocarbon receptor activation alters immune cell populations in the lung and bone marrow during coronavirus infection. Am. J. Physiol. Lung Cell. Mol. Physiol. 2024, 326, L313–L329. [Google Scholar] [CrossRef] [PubMed]
- Rothhammer, V.; Quintana, F.J. The aryl hydrocarbon receptor: An environmental sensor integrating immune responses in health and disease. Nat. Rev. Immunol. 2019, 19, 184–197. [Google Scholar] [CrossRef] [PubMed]
- Grunewald, M.E.; Shaban, M.G.; Mackin, S.R.; Fehr, A.R.; Perlman, S. Murine Coronavirus Infection Activates the Aryl Hydrocarbon Receptor in an Indoleamine 2,3-Dioxygenase-Independent Manner, Contributing to Cytokine Modulation and Proviral TCDD-Inducible-PARP Expression. J. Virol. 2020, 94, e01743-19. [Google Scholar] [CrossRef] [PubMed]
- Giovannoni, F.; Bosch, I.; Polonio, C.M.; Torti, M.F.; Wheeler, M.A.; Li, Z.; Romorini, L.; Rodriguez Varela, M.S.; Rothhammer, V.; Barroso, A.; et al. AHR is a Zika virus host factor and a candidate target for antiviral therapy. Nat. Neurosci. 2020, 23, 939–951. [Google Scholar] [CrossRef]
- Shi, J.; Du, T.; Wang, J.; Tang, C.; Lei, M.; Yu, W.; Yang, Y.; Ma, Y.; Huang, P.; Chen, H.; et al. Aryl hydrocarbon receptor is a proviral host factor and a candidate pan-SARS-CoV-2 therapeutic target. Sci. Adv. 2023, 9, eadf0211. [Google Scholar] [CrossRef]
- Yousefi, M.; Lee, W.S.; Chan, W.O.Y.; He, W.; Mah, M.G.; Yong, C.L.; Deerain, J.M.; Wang, L.; Arcinas, C.; Yan, B.; et al. Betacoronaviruses SARS-CoV-2 and HCoV-OC43 infections in IGROV-1 cell line require aryl hydrocarbon receptor. Emerg. Microbes Infect. 2023, 12, 2256416. [Google Scholar] [CrossRef]
- Cerracchio, C.; Serra, F.; Amoroso, M.G.; Fiorito, F. Canine Coronavirus Activates Aryl Hydrocarbon Receptor during In Vitro Infection. Viruses 2022, 14, 2437. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, Y.; Cui, T.; Bao, H.; Gao, M.; Cheng, M.; Sun, Y.; Lu, Y.; Guan, J.; Zhang, D.; et al. AhR ligands from LGG metabolites promote piglet intestinal ILC3 activation and IL-22 secretion to inhibit PEDV infection. J. Virol. 2024, 98, e0103924. [Google Scholar] [CrossRef]
- Del Sorbo, L.; Acconcia, C.; Salvatore, M.M.; Fusco, G.; Vasinioti, V.; Lucente, M.S.; Zhu, L.; Pratelli, A.; Russo, L.; Andolfi, A.; et al. Insight into the Role of the Aryl Hydrocarbon Receptor in Bovine Coronavirus Infection by an Integrated Approach Combining In Vitro and In Silico Methods. Microorganisms 2025, 13, 579. [Google Scholar] [CrossRef]
- Del Sorbo, L.; Giugliano, R.; Cerracchio, C.; Iovane, V.; Salvatore, M.M.; Serra, F.; Amoroso, M.G.; Pellegrini, F.; Levante, M.; Capozza, P.; et al. In Vitro Evaluation of Aryl Hydrocarbon Receptor Involvement in Feline Coronavirus Infection. Viruses 2025, 17, 227. [Google Scholar] [CrossRef]
- Zhang, M.; Cai, Z.; An, H.; He, R.; Zhang, S.; Fang, S. Infectious Bronchitis Virus Activates the Aryl Hydrocarbon Receptor During In Vitro Infection. Vet. Sci. 2025, 12, 932. [Google Scholar] [CrossRef] [PubMed]
- Cerracchio, C.; Del Sorbo, L.; Serra, F.; Staropoli, A.; Amoroso, M.G.; Vinale, F.; Fiorito, F. Fungal metabolite 6-pentyl-alpha-pyrone reduces canine coronavirus infection. Heliyon 2024, 10, e28351. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Yao, L.; Chen, R.; He, J.; Lin, T.; Qiu, S.; Chen, G.; Chen, H.; Qiu, S.X. Pinostrobin from plants and propolis against human coronavirus HCoV-OC43 by modulating host AHR/CYP1A1 pathway and lipid metabolism. Antivir. Res. 2023, 212, 105570. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; The International Natural Product Sciences Taskforce; Supuran, C.T. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- Cerracchio, C.; Iovane, V.; Salvatore, M.M.; Amoroso, M.G.; Dakroub, H.; DellaGreca, M.; Nicoletti, R.; Andolfi, A.; Fiorito, F. Effectiveness of the Fungal Metabolite 3-O-Methylfunicone towards Canine Coronavirus in a Canine Fibrosarcoma Cell Line (A72). Antibiotics 2022, 11, 1594. [Google Scholar] [CrossRef]
- Roy, B.G. Potential of small-molecule fungal metabolites in antiviral chemotherapy. Antivir. Chem. Chemother. 2017, 25, 20–52. [Google Scholar] [CrossRef]
- Deshmukh, S.K.; Agrawal, S.; Gupta, M.K.; Patidar, R.K.; Ranjan, N. Recent Advances in the Discovery of Antiviral Metabolites from Fungi. Curr. Pharm. Biotechnol. 2022, 23, 495–537. [Google Scholar] [CrossRef]
- Barreca, D.; Gattuso, G.; Bellocco, E.; Calderaro, A.; Trombetta, D.; Smeriglio, A.; Lagana, G.; Daglia, M.; Meneghini, S.; Nabavi, S.M. Flavanones: Citrus phytochemical with health-promoting properties. Biofactors 2017, 43, 495–506. [Google Scholar] [CrossRef]
- Linnakoski, R.; Reshamwala, D.; Veteli, P.; Cortina-Escribano, M.; Vanhanen, H.; Marjomaki, V. Antiviral Agents From Fungi: Diversity, Mechanisms and Potential Applications. Front. Microbiol. 2018, 9, 2325. [Google Scholar] [CrossRef]
- Vinale, F.; Sivasithamparam, K.; Ghisalberti, E.L.; Marra, R.; Barbetti, M.J.; Li, H.; Woo, S.L.; Lorito, M. A novel role for Trichoderma secondary metabolites in the interactions with plants. Physiol. Mol. Plant Pathol. 2008, 72, 80–86. [Google Scholar] [CrossRef]
- Mendoza-Mendoza, A.; Esquivel-Naranjo, E.U.; Soth, S.; Whelan, H.; Alizadeh, H.; Echaide-Aquino, J.F.; Kandula, D.; Hampton, J.G. Uncovering the multifaceted properties of 6-pentyl-alpha-pyrone for control of plant pathogens. Front. Plant Sci. 2024, 15, 1420068. [Google Scholar] [CrossRef]
- Kotb, A.M.E.; Abd El-Aziz, N.K.; Elariny, E.Y.T.; Yahya, R.; Alkhalifah, D.H.M.; Ahmed, R.M. Synergistic Antibacterial Potential of 6-Pentyl-alpha-pyrone Lactone and Zinc Oxide Nanoparticles against Multidrug-Resistant Enterobacterales Isolated from Urinary Tract Infections in Humans. Antibiotics 2022, 11, 440. [Google Scholar] [CrossRef] [PubMed]
- Vasinioti, V.I.; Odigie, A.E.; Lucente, M.S.; Del Sorbo, L.; Catella, C.; Casalino, E.; Salvatore, M.M.; Staropoli, A.; Vinale, F.; Tempesta, M.; et al. In Vitro Antiviral Activity of the Fungal Metabolite 6-Pentyl-alpha-Pyrone Against Bovine Coronavirus: A Translational Study to SARS-CoV-2. Vet. Sci. 2025, 12, 634. [Google Scholar] [CrossRef]
- McGlacken, G.P.; Fairlamb, I.J. 2-Pyrone natural products and mimetics: Isolation, characterisation and biological activity. Nat. Prod. Rep. 2005, 22, 369–385. [Google Scholar] [CrossRef] [PubMed]
- Fiorito, F.; Cerracchio, C.; Salvatore, M.M.; Serra, F.; Pucciarelli, A.; Amoroso, M.G.; Nicoletti, R.; Andolfi, A. Antiviral Property of the Fungal Metabolite 3-O-Methylfunicone in Bovine Herpesvirus 1 Infection. Microorganisms 2022, 10, 188. [Google Scholar] [CrossRef]
- Del Sorbo, L.; Salvatore, M.M.; Acconcia, C.; Giugliano, R.; Fusco, G.; Galdiero, M.; Vasinioti, V.I.; Lucente, M.S.; Capozza, P.; Pratelli, A.; et al. Structural Features and In Vitro Antiviral Activities of Fungal Metabolites Sphaeropsidins A and B Against Bovine Coronavirus. Int. J. Mol. Sci. 2025, 26, 7045. [Google Scholar] [CrossRef]
- Ghosh, S.; Dellibovi-Ragheb, T.A.; Kerviel, A.; Pak, E.; Qiu, Q.; Fisher, M.; Takvorian, P.M.; Bleck, C.; Hsu, V.W.; Fehr, A.R.; et al. beta-Coronaviruses Use Lysosomes for Egress Instead of the Biosynthetic Secretory Pathway. Cell 2020, 183, 1520–1535.e14. [Google Scholar] [CrossRef]
- Cerracchio, C.; Salvatore, M.M.; Del Sorbo, L.; Serra, F.; Amoroso, M.G.; DellaGreca, M.; Nicoletti, R.; Andolfi, A.; Fiorito, F. In Vitro Evaluation of Antiviral Activities of Funicone-like Compounds Vermistatin and Penisimplicissin against Canine Coronavirus Infection. Antibiotics 2023, 12, 1319. [Google Scholar] [CrossRef]
- Payne, H.R.; Storz, J.; Henk, W.G. Initial events in bovine coronavirus infection: Analysis through immunogold probes and lysosomotropic inhibitors. Arch. Virol. 1990, 114, 175–189. [Google Scholar] [CrossRef]
- Dea, S.; Roy, R.S.; Begin, M.E. Bovine coronavirus isolation and cultivation in continuous cell lines. Am. J. Vet. Res. 1980, 41, 30–38. [Google Scholar] [CrossRef]
- Saif, L.J.; Heckert, R.A.; Miller, K.L.; Tarek, M.M. Cell culture propagation of bovine coronavirus. J. Tissue Cult. Methods 1988, 11, 139–145. [Google Scholar] [CrossRef]
- Mohamad, S.A.; Zahran, E.M.; Abdel Fadeel, M.R.; Albohy, A.; Safwat, M.A. New Acaciin-Loaded Self-Assembled Nanofibers as M(Pro) Inhibitors Against BCV as a Surrogate Model for SARS-CoV-2. Int. J. Nanomed. 2021, 16, 1789–1804. [Google Scholar] [CrossRef]
- Newman, R.A.; Chase, C.C.L.; Matos, J.R.; Abdelsalam, K.; Buterbaugh, R.; Van Holland, S.; Abdelaal, H.; Woolum, A.; Jagannadha Sastry, K. Efficacy of oleandrin and PBI-05204 against bovine viruses of importance to commercial cattle health. Antivir. Chem. Chemother. 2022, 30, 20402066221103960. [Google Scholar] [CrossRef] [PubMed]
- Ramon, A.C.; Perez, G.V.; Caballero, E.; Rosales, M.; Aguilar, D.; Vazquez-Blomquist, D.; Ramos, Y.; Rodriguez-Ulloa, A.; Falcon, V.; Rodriguez-Molto, M.P.; et al. Targeting of Protein Kinase CK2 Elicits Antiviral Activity on Bovine Coronavirus Infection. Viruses 2022, 14, 552. [Google Scholar] [CrossRef] [PubMed]
- Staropoli, A.; Iacomino, G.; De Cicco, P.; Woo, S.L.; Di Costanzo, L.; Vinale, F. Induced secondary metabolites of the beneficial fungus Trichoderma harzianum M10 through OSMAC approach. Chem. Biol. Technol. Agric. 2023, 10, 28. [Google Scholar] [CrossRef]
- Abolhassani, A.; Riazi, G.H.; Azizi, E.; Amanpour, S.; Muhammadnejad, S.; Haddadi, M.; Zekri, A.; Shirkoohi, R. FGF10: Type III Epithelial Mesenchymal Transition and Invasion in Breast Cancer Cell Lines. J. Cancer 2014, 5, 537–547. [Google Scholar] [CrossRef]
- Kadhim, Z.A.; Sulaiman, G.M.; Al-Shammari, A.M.; Khan, R.A.; Al Rugaie, O.; Mohammed, H.A. Oncolytic Newcastle Disease Virus Co-Delivered with Modified PLGA Nanoparticles Encapsulating Temozolomide against Glioblastoma Cells: Developing an Effective Treatment Strategy. Molecules 2022, 27, 5757. [Google Scholar] [CrossRef]
- Bank, H.L. Assessment of islet cell viability using fluorescent dyes. Diabetologia 1987, 30, 812–816. [Google Scholar] [CrossRef]
- Leite, M.; Quinta-Costa, M.; Leite, P.S.; Guimaraes, J.E. Critical evaluation of techniques to detect and measure cell death—Study in a model of UV radiation of the leukaemic cell line HL60. Anal. Cell. Pathol. 1999, 19, 139–151. [Google Scholar] [CrossRef]
- Kroemer, G.; Levine, B. Autophagic cell death: The story of a misnomer. Nat. Rev. Mol. Cell Biol. 2008, 9, 1004–1010. [Google Scholar] [CrossRef]
- Zakeri, Z.; Lockshin, R.A. Cell death: History and future. In Advances in Experimental Medicine and Biology; Springer: Dordrecht, The Netherlands, 2008; Volume 615, pp. 1–11. [Google Scholar] [CrossRef]
- Wulf, E.; Deboben, A.; Bautz, F.A.; Faulstich, H.; Wieland, T. Fluorescent phallotoxin, a tool for the visualization of cellular actin. Proc. Natl. Acad. Sci. USA 1979, 76, 4498–4502. [Google Scholar] [CrossRef] [PubMed]
- Altamura, G.; Power, K.; Martano, M.; Degli Uberti, B.; Galiero, G.; De Luca, G.; Maiolino, P.; Borzacchiello, G. Felis catus papillomavirus type-2 E6 binds to E6AP, promotes E6AP/p53 binding and enhances p53 proteasomal degradation. Sci. Rep. 2018, 8, 17529. [Google Scholar] [CrossRef] [PubMed]
- Pratelli, A.; Cirone, F.; Capozza, P.; Trotta, A.; Corrente, M.; Balestrieri, A.; Buonavoglia, C. Bovine respiratory disease in beef calves supported long transport stress: An epidemiological study and strategies for control and prevention. Res. Vet. Sci. 2021, 135, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Alkharashi, N.A.O.; Periasamy, V.S.; Athinarayanan, J.; Alshatwi, A.A. Sulforaphane alleviates cadmium-induced toxicity in human mesenchymal stem cells through POR and TNFSF10 genes expression. Biomed. Pharmacother. 2019, 115, 108896. [Google Scholar] [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Zidek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Laskowski, R.A.; Rullmannn, J.A.; MacArthur, M.W.; Kaptein, R.; Thornton, J.M. AQUA and PROCHECK-NMR: Programs for checking the quality of protein structures solved by NMR. J. Biomol. NMR 1996, 8, 477–486. [Google Scholar] [CrossRef]
- Delano, W.L. The PyMOL Molecular Graphics System; Delano Scientific: San Carlos, CA, USA, 2002. [Google Scholar]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Jensen, M.H.; Morris, E.J.; Weitz, D.A. Mechanics and dynamics of reconstituted cytoskeletal systems. Biochim. Biophys. Acta 2015, 1853, 3038–3042. [Google Scholar] [CrossRef]
- Radtke, K.; Dohner, K.; Sodeik, B. Viral interactions with the cytoskeleton: A hitchhiker’s guide to the cell. Cell. Microbiol. 2006, 8, 387–400. [Google Scholar] [CrossRef]
- Swain, J.; Merida, P.; Rubio, K.; Bracquemond, D.; Neyret, A.; Aguilar-Ordonez, I.; Gunther, S.; Barreto, G.; Muriaux, D. F-actin nanostructures rearrangements and regulation are essential for SARS-CoV-2 particle production in host pulmonary cells. iScience 2023, 26, 107384. [Google Scholar] [CrossRef]
- Yang, T.; Feng, Y.L.; Chen, L.; Vaziri, N.D.; Zhao, Y.Y. Dietary natural flavonoids treating cancer by targeting aryl hydrocarbon receptor. Crit. Rev. Toxicol. 2019, 49, 445–460. [Google Scholar] [CrossRef]
- Yamada, T.; Horimoto, H.; Kameyama, T.; Hayakawa, S.; Yamato, H.; Dazai, M.; Takada, A.; Kida, H.; Bott, D.; Zhou, A.C.; et al. Constitutive aryl hydrocarbon receptor signaling constrains type I interferon-mediated antiviral innate defense. Nat. Immunol. 2016, 17, 687–694. [Google Scholar] [CrossRef]
- Del Sorbo, L.; Cerracchio, C.; Serra, F.; Canzanella, S.; Giugliano, R.; Lambiase, S.; Aranguiz, N.P.; Esposito, M.; Amoroso, M.G.; Fusco, G.; et al. Canine coronavirus infection is intensified by 2,3,7,8-tetrachlorodibenzo-p-dioxin. Arch. Toxicol. 2025, 99, 2211–2223. [Google Scholar] [CrossRef]
- Guarnieri, T. Hypothesis: Emerging Roles for Aryl Hydrocarbon Receptor in Orchestrating CoV-2-Related Inflammation. Cells 2022, 11, 648. [Google Scholar] [CrossRef]
- Pereira, G.; Leao, A.; Erustes, A.G.; Morais, I.B.M.; Vrechi, T.A.M.; Zamarioli, L.D.S.; Pereira, C.A.S.; Marchioro, L.O.; Sperandio, L.P.; Lins, I.V.F.; et al. Pharmacological Modulators of Autophagy as a Potential Strategy for the Treatment of COVID-19. Int. J. Mol. Sci. 2021, 22, 4067. [Google Scholar] [CrossRef]
- Sun, H.; Liu, Y.; Yang, X.; Qin, W.; Lu, X.; Wang, Y.; Tong, W.; Yu, H.; Zheng, H.; Tong, G.; et al. Bovine coronavirus Nsp14 protein promotes viral replication by degrading TRAF3 to inhibit interferon production. Vet. Microbiol. 2025, 310, 110708. [Google Scholar] [CrossRef]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Del Sorbo, L.; Giugliano, R.; Acconcia, C.; Salvatore, M.M.; Staropoli, A.; Vasinioti, V.I.; Lucente, M.S.; Capozza, P.; Vinale, F.; Pratelli, A.; et al. Potential Activity of 6-Pentyl-α-pyrone as an Antiviral for Bovine Coronavirus. Pathogens 2026, 15, 332. https://doi.org/10.3390/pathogens15030332
Del Sorbo L, Giugliano R, Acconcia C, Salvatore MM, Staropoli A, Vasinioti VI, Lucente MS, Capozza P, Vinale F, Pratelli A, et al. Potential Activity of 6-Pentyl-α-pyrone as an Antiviral for Bovine Coronavirus. Pathogens. 2026; 15(3):332. https://doi.org/10.3390/pathogens15030332
Chicago/Turabian StyleDel Sorbo, Luca, Rosa Giugliano, Clementina Acconcia, Maria Michela Salvatore, Alessia Staropoli, Violetta Iris Vasinioti, Maria Stella Lucente, Paolo Capozza, Francesco Vinale, Annamaria Pratelli, and et al. 2026. "Potential Activity of 6-Pentyl-α-pyrone as an Antiviral for Bovine Coronavirus" Pathogens 15, no. 3: 332. https://doi.org/10.3390/pathogens15030332
APA StyleDel Sorbo, L., Giugliano, R., Acconcia, C., Salvatore, M. M., Staropoli, A., Vasinioti, V. I., Lucente, M. S., Capozza, P., Vinale, F., Pratelli, A., Russo, L., Iacovino, R., & Fiorito, F. (2026). Potential Activity of 6-Pentyl-α-pyrone as an Antiviral for Bovine Coronavirus. Pathogens, 15(3), 332. https://doi.org/10.3390/pathogens15030332












