Seasonal Turnover in Bat Skin Mycobiota: Contrasting Fungal Communities Between Hibernation and Reproduction in Greater Mouse-Eared Bats (Myotis myotis)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Microclimatic Parameter Measurement
2.3. Sampling Methods
2.4. Isolation of Fungi from Samples
2.5. Identification of Fungi
2.6. Data Analyses
3. Results
3.1. Biometric Features of Bats and Microclimatic Conditions in Habitats
3.2. Fungal Isolation and Identification
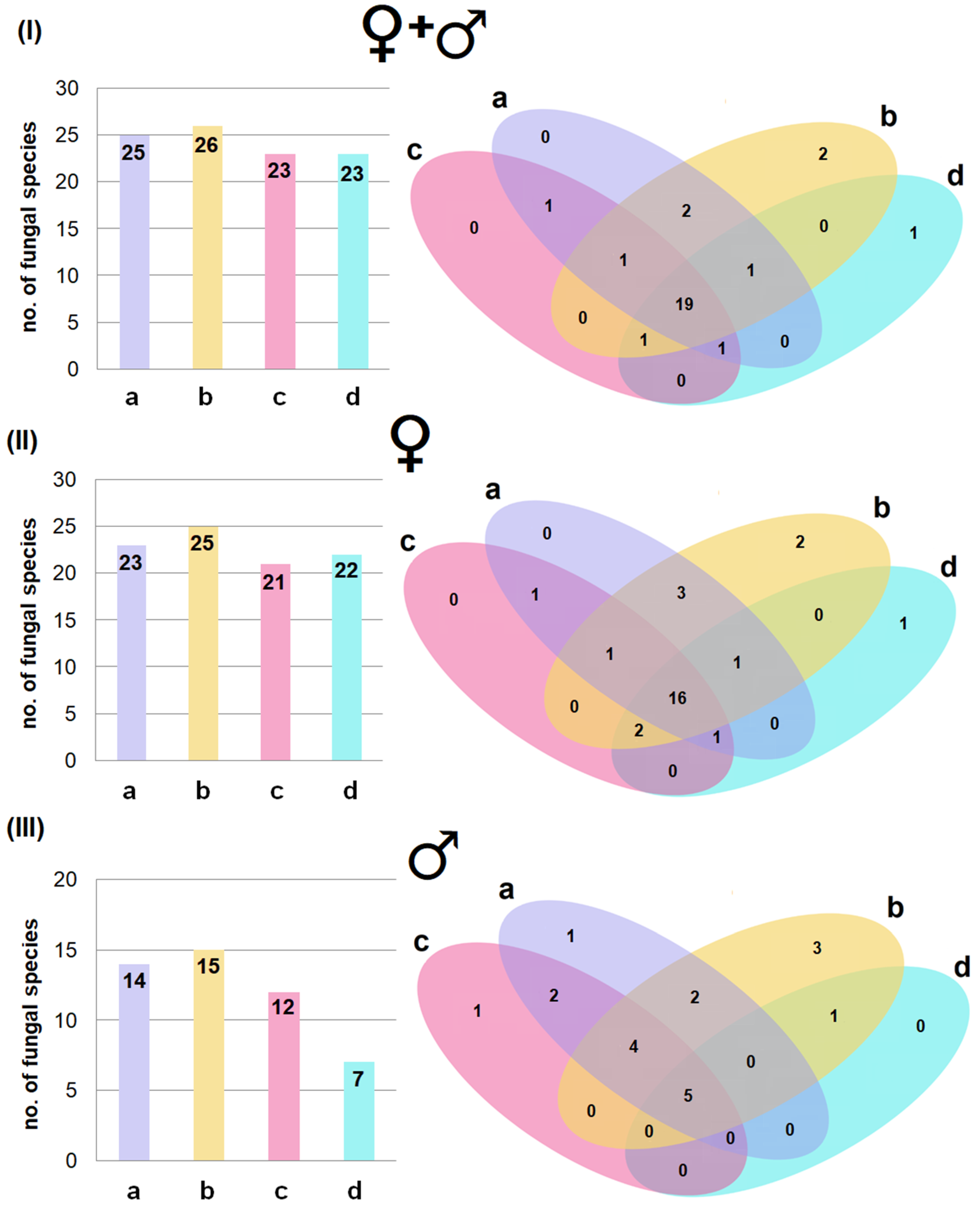
3.3. Fungal Diversity Across Body Regions and the Effect of Age and Sex
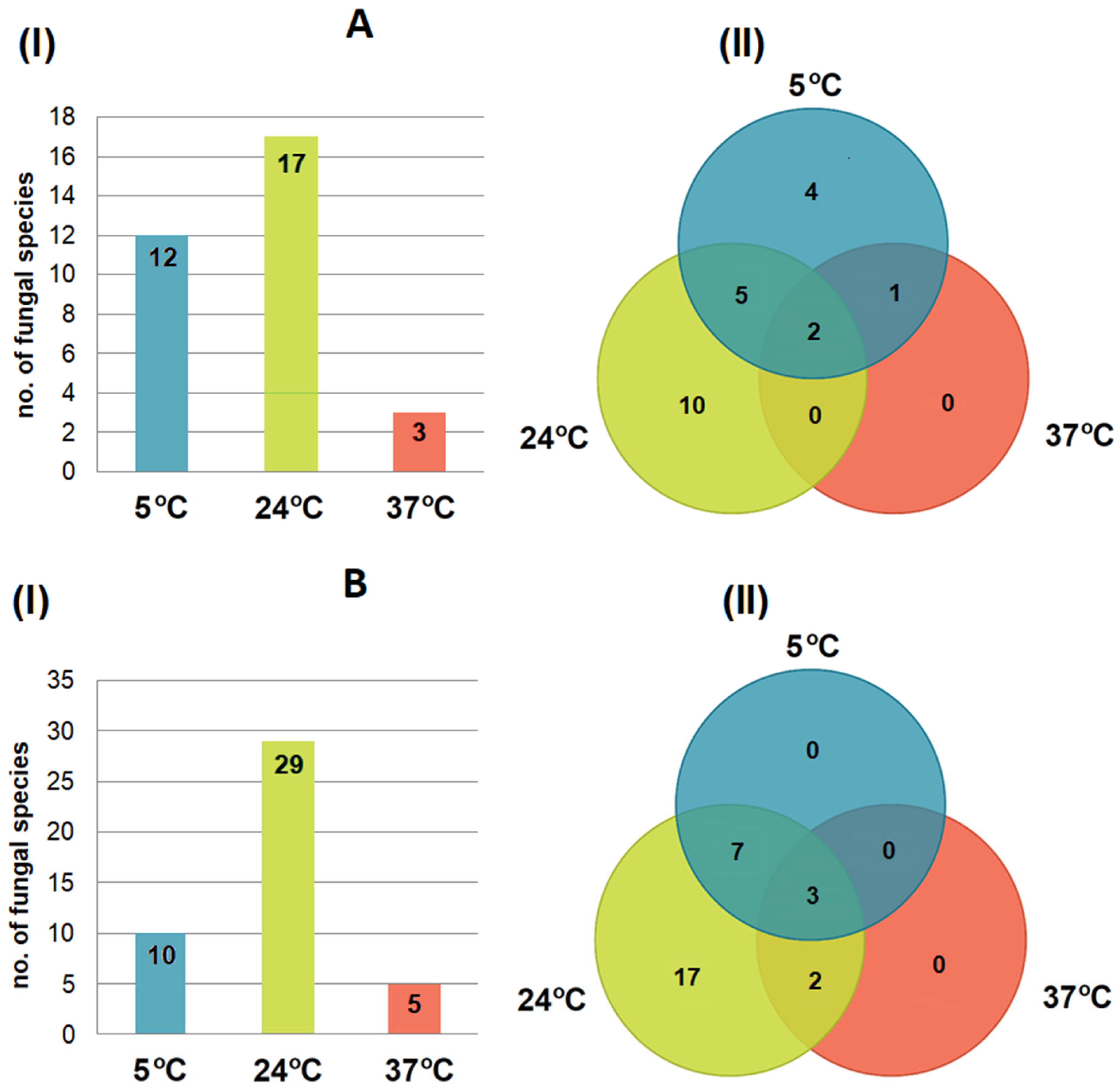
3.4. Effect of Incubation Temperature on Fungal Isolation
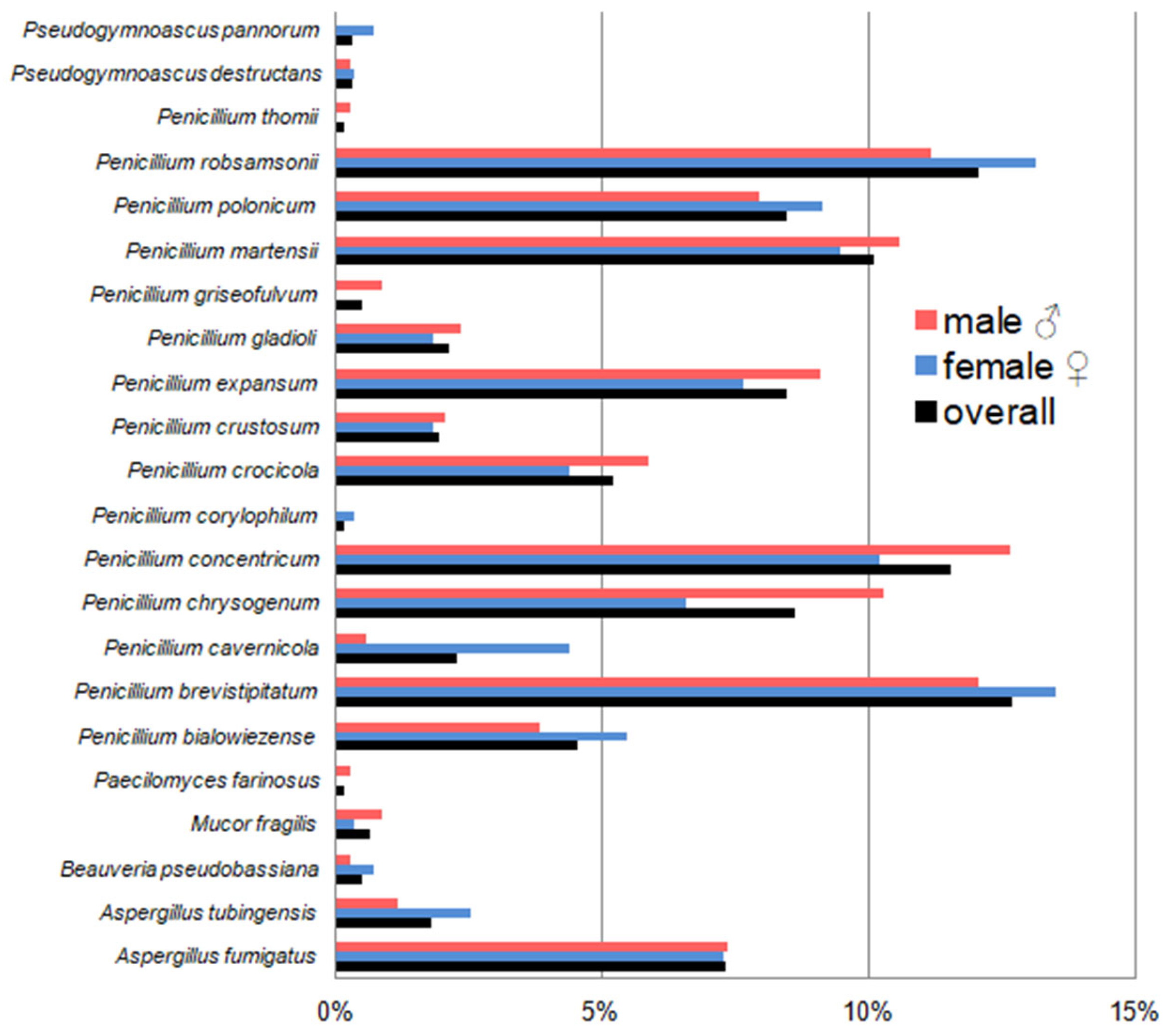
3.5. Dominant Fungal Species and the Effect of Biometric Features on Fungal Diversity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Study Sites | Bat No. | Sex | Forearm Length (mm) | Weight (g) | Age |
|---|---|---|---|---|---|
| Nietoperek bat reserve | 1 | male | 59.6 | 24.0 | adult |
| 2 | male | 59.3 | 25.0 | adult | |
| 3 | male | 60.5 | 26.5 | subadult | |
| 4 | female | 62.9 | 25.0 | subadult | |
| 5 | male | 60.7 | 23.0 | subadult | |
| 6 | male | 60.3 | 26.0 | adult | |
| 7 | male | 61.3 | 24.0 | subadult | |
| 8 | male | 59.1 | 25.0 | subadult | |
| 9 | male | 59.2 | 24.5 | adult | |
| 10 | male | 59.9 | 24.0 | adult | |
| 11 | female | 60.7 | 26.0 | adult | |
| 12 | male | 61.0 | 28.0 | adult | |
| 13 | female | 64.1 | 27.5 | adult | |
| 14 | male | 59.6 | 23.5 | subadult | |
| 15 | female | 62.7 | 22.0 | adult | |
| 16 | male | 59.4 | 24.5 | subadult | |
| 17 | female | 61.0 | 24.0 | adult | |
| 18 | female | 61.4 | 27.0 | subadult | |
| 19 | female | 63.2 | 23.0 | subadult | |
| 20 | male | 61.4 | 26.0 | adult | |
| 21 | female | 62.0 | 25.0 | adult | |
| 22 | male | 60.6 | 24.5 | adult | |
| 23 | female | 63.8 | 27.5 | adult | |
| 24 | female | 60.6 | 25.0 | adult | |
| 25 | male | 61.1 | 24.0 | adult | |
| 26 | male | 58.7 | 24.0 | adult | |
| 27 | female | 60.5 | 22.5 | subadult | |
| 28 | male | 60.5 | 23.0 | subadult | |
| 29 | female | 62.1 | 26.0 | adult | |
| 30 | female | 61.4 | 26.0 | subadult | |
| forester’s lodge in Lipy | 1 | female | 59.4 | 26.5 | subadult |
| 2 | female | 62.5 | 23.5 | subadult | |
| 3 | female | 61.1 | 21.0 | adult | |
| 4 | female | 60.8 | 22.5 | subadult | |
| 5 | female | 63.4 | 23.5 | subadult | |
| 6 | female | 63.5 | 24.5 | subadult | |
| 7 | male | 59.9 | 20.0 | subadult | |
| 8 | female | 63.7 | 23.0 | subadult | |
| 9 | female | 62.2 | 27.0 | subadult | |
| 10 | female | 64.8 | 25.5 | subadult | |
| 11 | female | 63.2 | 28.0 | subadult | |
| 12 | female | 61.8 | 23.5 | subadult | |
| 13 | female | 62.5 | 24.0 | subadult | |
| 14 | female | 63.0 | 25.0 | subadult | |
| 15 | female | 60.0 | 29.0 | subadult | |
| 16 | male | 58.1 | 24.5 | subadult | |
| 17 | female | 60.7 | 23.5 | subadult | |
| 18 | female | 61.6 | 24.5 | subadult | |
| 19 | male | 60.0 | 22.5 | subadult | |
| 20 | female | 62.6 | 25.0 | subadult | |
| 21 | female | 62.0 | 26.0 | subadult | |
| 22 | female | 62.2 | 25.5 | subadult | |
| 23 | female | 61.6 | 26.5 | subadult | |
| 24 | female | 62.4 | 24.5 | subadult | |
| 25 | female | 63.9 | 27.0 | subadult | |
| 26 | male | 56.8 | 22.5 | subadult | |
| 27 | female | 62.4 | 23.5 | adult | |
| 28 | male | 59.6 | 24.0 | subadult | |
| 29 | female | 61.9 | 24.5 | subadult | |
| 30 | female | 62.5 | 26.0 | subadult |
| Fungal Species | Bat Number and Swab Location | ||
|---|---|---|---|
| 5 °C | 24 °C | 37 °C | |
| Aspergillus fumigatus | 12 (a), 18 (a,b) | n.d. | 1 (c,d), 2 (a), 3 (b), 4 (b,d), 5 (a,b,c,d), 8 (a,c), 9 (d), 10 (c,d), 11 (b), 12 (a,b), 14 (a,b), 15 (a,d), 16 (a), 18 (a,c,d), 19 (b,c), 20 (c,d), 21 (d), 22 (c), 23 (b), 24 (a,d), 25 (d), 26 (b,c), 27 (c), 29 (b), 30 (a,c) |
| Aspergillus tubingensis | n.d. | 15 (a), 18 (c) | 1 (c), 5 (a), 8 (a), 16 (a), 18 (c,d), 27 (c), 29 (b), 30 (a) |
| Beauveria pseudobassiana | n.d. | 1 (a), 19 (a,b) | n.d. |
| Mucor fragilis | n.d. | 16 (b), 28 (a,b), 29 (a) | n.d. |
| Paecilomyces farinosus | n.d. | 12 (d) | n.d. |
| Penicillium bialowiezense | n.d. | 1 (a,d), 4 (a,c), 7 (b), 8 (c), 14 (b,d), 19 (b), 20 (b,d), 21 (a,b,c), 22 (a,b,d), 23 (c), 24 (a,b), 25 (b), 26 (d), 27 (a,b,c), 29 (d), 30 (a,c) | n.d. |
| Penicillium brevistipitatum | 2 (a,b), 6 (c,d), 10 (d), 11 (b), 15 (a), 19 (c), 20 (b), 30 (c) | 1 (a,b,c,d), 3 (a,b,d), 4 (a,b,c), 5 (a,b,d), 6 (b,d), 7 (a,b,d), 8 (d), 9 (b,d), 10 (d), 12 (b), 13 (a,b,c,d), 14 (b,d), 15 (a), 16 (a,b), 17 (a,b,c,d), 18 (a,b,c,d), 19 (a,b,c), 20 (d), 21 (a,b,c,d), 22 (a,d), 23 (a,b,d), 24 (c), 25 (a,c,d), 26 (b), 27 (b), 28 (a,b,c,d), 29 (b,d), 30 (b,c,d) | n.d. |
| Penicillium cavernicola | n.d. | 4 (a,b), 5 (b), 6 (d), 13 (d), 17 (a,b,c,d), 18 (a,b,c,d), 30 (b) | n.d. |
| Penicillium chrysogenum | 1 (c), 2 (a,b,d), 6 (a), 7 (a,c,d), 9 (b), 11 (a,b), 12 (a), 14 (a), 15 (a,b), 19 (c,d), 20 (a,b,c), 27 (a,d), 30 (c) | 2 (a), 3 (a,d), 5 (a), 6 (a,d), 7 (a,b,c,d), 8 (a,b,c,d), 9 (a,b), 10 (d), 15 (b), 18 (a), 19 (a,c), 20 (a), 21 (b,c), 22 (d), 25 (d), 26 (a,d), 27 (a) | 5 (a), 19 (c), 30 (c) |
| Penicillium concentricum | 1 (d), 5 (d), 6 (b), 8 (c), 12 (b), 18 (b,c,d), 19 (a,b,c), 21 (b), 22 (c), 23 (b), 25 (a,c,d), 27 (c), 29 (a,c) | 1 (a,c,d), 2 (c), 3 (c,da), 4 (a,c), 5 (d), 7 (a,b), 8 (a,b,c,d), 9 (a,b,d), 10 (d), 11 (b), 13 (a), 14 (a,c), 15 (a,d), 16 (c,d), 17 (d), 18 (b), 19 (a,b,c), 20 (a,b), 21 (a,b,c), 22 (a,d), 24 (d), 26 (a,b,c,d), 28 (a,b,c,d), 29 (b), 30 (b) | n.d. |
| Penicillium corylophilum | n.d. | 18 (c) | n.d. |
| Penicillium crocicola | n.d. | 2 (a,d), 3 (a), 5 (d), 6 (c,d), 7 (c,d), 8 (d), 9 (b,d), 11 (b,c,d), 12 (b,c), 15 (b,c), 16 (b,c,d), 19 (a,c), 20 (c), 25 (c), 26 (a,d), 28 (b), 29 (a,b), 30 (c,d) | n.d. |
| Penicillium crustosum | 1 (c), 4 (a), 8 (c), 10 (c), 11 (c), 17 (a,c), 20 (a), 21 (b), 26 (b), 27 (b,c) | n.d. | n.d. |
| Penicillium expansum | 1 (a), 2 (a,b), 3 (a,b), 5 (c,d), 6 (c,d), 7 (a,c,d), 8 (c), 9 (b,c), 10 (c,d), 12 (b), 13 (a), 14 (d), 15 (a,d), 16 (a), 18 (a,b), 19 (c,d), 20 (b), 22 (a,c), 23 (b), 24 (d), 25 (b,d), 26 (a,b,c), 27 (b), 29 (a,b,d), 30 (b,c) | 6 (c), 24 (d) | 9 (d), 15 (d), 16 (a), 19 (b), 21 (d), 29 (b), 30 (a) |
| Penicillium gladioli | n.d. | 1 (b), 2 (a,b), 3 (b), 4 (b,c), 5 (b,c), 20 (a), 24 (d), 26 (b), 29 (a), 30 (b) | n.d. |
| Penicillium griseofulvum | 20 (c) | 2 (a,b) | n.d. |
| Penicillium martensii | 1 (c,d), 3 (a,b,c,d), 4 (a,b,c), 5 (a,b), 6 (b), 7 (a,b), 8 (a,b,d), 9 (a,d), 12 (b), 13 (a,b,d), 14 (a,c,d), 16 (a,b), 17 (a,d), 18 (c,d), 19 (a,b), 20 (a,d), 21 (a,b,d), 22 (a,b,d), 23 (a,b,d), 24 (a,b), 25 (a,b,c,d), 26 (b), 27 (a,b), 28 (a,b,c,d), 29 (b,d), 30 (b,d) | n.d. | n.d. |
| Penicillium polonicum | 1 (c), 2 (a), 3 (a,b,c,d), 4 (a,c), 5 (b), 7 (b), 8 (a,c,d), 9 (a), 12 (d), 13 (a), 14 (b,d), 16 (a), 17 (a,b,c,d), 18 (a,d), 19 (b), 20 (a,d), 21 (a,c,d), 22 (a,b,c), 23 (a,b,c), 24 (a,c), 26 (b), 27 (a,c), 28 (a,b,c,d), 29 (b,d), 30 (a,b,d) | 2 (a) | n.d. |
| Penicillium robsamsonii | 10 (d), 13 (c), 20 (c) | 1 (a,c), 3 (a,b,c,d), 4 (a,b,c), 5 (a,b,c,d), 6 (b,c), 7 (a,b,c,d), 9 (a,b,d), 10 (a), 11 (b), 13 (a,b,c,d), 14 (b,c,d), 17 (a,b,d), 19 (a,b,d), 20 (a,b,d), 21 (a,b,c,d), 22 (a,b,c), 23 (a,b,c,d), 24 (a,b,c), 25 (a,b), 26 (a,b), 27 (a,b,c,d), 28 (a,b,d), 29 (a,b,d), 30 (a,b,d) | n.d. |
| Penicillium thomii | 6 (a) | n.d. | |
| Pseudogymnoascus destructans | 11 (d), 12 (d) | n.d. | n.d. |
| Pseudogymnoascus pannorum | 27 (d), 30 (d) | n.d. | n.d. |
| Fungal Species | Bat Number and Swab Location | ||
|---|---|---|---|
| 5 °C | 24 °C | 37 °C | |
| Absidia virescens | n.d. | 3 (d), 9 (a,b,d), 10 (a), 12 (b), 15 (a), 17 (b,c), 26 (b,c), 28 (a,c), 30 (a,b) | n.d. |
| Alternaria alternata | n.d. | 2 (a,b,d), 10 (a,c), 24 (a,c) | 2 (d), 10 (a), 24 (c), 25 (a) |
| Apiospora arundinis | 12 (a), 13 (a,b), 15 (b,d), 17 (c), 18 (c), 20 (a), 25 (d), 27 (c,d), 28 (a,b) | 13 (a,b), 27 (d) | n.d. |
| Aspergillus fumigatus | n.d. | 1 (c), 2 (b), 3 (c), 6 (d), 7 (c), 8 (c), 10 (c,d), 11 (d), 17 (a) | 1 (a,b,c), 2 (a,d), 3 (b), 4 (a,b), 5 (a,b,c), 6 (a,b,d), 7 (a,b,d), 8 (c), 9 (c,d), 10 (c), 11 (a,c,d), 12 (a,d), 13 (a,b,c), 14 (a), 15 (d), 16 (a,b,d), 17 (a,b,c,d), 18 (a,c,d), 19 (c,d), 20 (a,b,d), 22 (a), 23 (b), 24 (a,c), 26 (a,d), 27 (c,d), 28 (c), 29 (a), 30 (c) |
| Aspergillus tubingensis | 13 (c,d) | 3 (c), 15 (a), 19 (c), 23 (d) | 7 (c), 9 (a) |
| Aureobasidium pullulans | 1 (a), 13 (b), 20 (d), 21 (a,b), 22 (a) | 21 (a,d) | n.d. |
| Botrytis cinerea | 19 (b) | 19 (b) | n.d. |
| Chaetomium angustispirale | n.d. | 1 (b,d), 2 (b,c), 17 (a,c), 18 (b), 20 (d), 23 (d), 25 (d), 28 (b) | |
| Cladosporium allicinum | n.d. | 20 (a,c) | n.d. |
| Cladosporium cladosporioides | 1 (b,d), 2 (a,d), 6 (a,d), 7 (b,c), 8 (c), 9 (a,b,d), 10 (a,d), 11 (d), 12 (a,b), 15 (a,b), 17 (a,b,d), 18 (b), 19 (b), 22 (a,d), 24 (a), 28 (a) | 1 (d), 2 (a,d), 6 (d), 7 (b,c), 9 (a,b), 10 (a,d), 12 (a,b), 15 (a,b), 17 (a,b), 22 (a,d) | n.d. |
| Fusarium sporotrichioides | n.d. | 9 (a,b,c,d), 19 (b,d) | n.d. |
| Mucor flavus | 10 (b) | 10 (a,b), 23 (a,b) | n.d. |
| Mucor fragilis | n.d. | 2 (b), 3 (b), 4 (d), 5 (a), 6 (d), 7 (a,c), 23 (b), 29 (b,d) | n.d. |
| Mucor hiemalis | 16 (b,c,d), 24 (b,c) | 9 (a,b), 14 (b), 16 (b,c), 24 (d) | 26 (a,b), 29 (b) |
| Paecilomyces farinosus | n.d. | 13 (d), 22 (d) | n.d. |
| Penicillium aurantiogriseum | n.d. | 19 (a,b), 30 (a,b,c,d) | n.d. |
| Penicillium bialowiezense | n.d. | 1 (c,d), 2 (a,b,d), 4 (a), 6 (d), 7 (a), 10 (a), 12 (b), 17 (b,c), 28 (b) | n.d. |
| Penicillium chrysogenum | 1 (a,b), 3 (a), 4 (a,b,c), 5 (b,c,d), 6 (b,c,d), 7 (b,d), 8 (a,b,c,d), 10 (c,d), 11 (a,b), 14 (a,b,c,d), 15 (a,b,c), 16 (c,d), 18 (a,b,c,d), 19 (a), 20 (a,c), 21 (b), 22 (a,c,d), 23 (b), 24 (c,d), 25 (a), 26 (a,d,c), 27 (a,b,c), 28 (c), 29 (a,b,c,d), 30 (b) | 1 (a,c,d), 2 (a,c), 3 (b,c), 4 (a,b,c,d), 5 (b,c,d), 6 (a,b,c,d), 7 (a,b,d), 8 (a,b,d), 9 (c,d), 10 (d), 11 (a,b,c,d), 12 (c,d), 13 (a,b,d), 14 (a,b,d), 15 (a,d), 16 (c,d), 17 (b,c), 18 (b,c,d), 19 (a), 20 (c), 21 (a,c), 22 (b), 23 (c), 24 (c,d), 25 (a,b), 26 (a,d), 27 (a,b,c,d), 28 (a,b,c,d), 29 (b) | 3 (c), 4 (c), 5 (a), 6 (a,c,d), 8 (b), 16 (d), 17 (a,d) |
| Penicillium commune | n.d. | 12 (a,b), 21 (a), 23 (a) | n.d. |
| Penicillium crustosum | n.d. | 18 (b), 26 (a,b,c,d), 28 (b,d) | n.d. |
| Penicillium dipodomyicola | n.d. | 6 (b), 7 (b), 10 (d), 11 (c,d), 14 (c), 16 (a,c,d), 20 (b), 21 (b), 23 (b,c) | n.d. |
| Penicillium glabrum | n.d. | 2 (a,d), 4 (a), 8 (a), 9 (a,b), 10 (a,b,c), 16 (d), 17 (b), 18 (b), 19 (b), 20 (a,b), 22 (a), 24 (b), 25 (a), 26 (c), 27 (a), 28 (a), 30 (a) | n.d. |
| Penicillium griseofulvum | 1 (a,d), 3 (c), 7 (a), 9 (d), 18 (c,d), 20 (c), 23 (c,d), 26 (d) | 16 (a), 17 (d), 18 (a), 20 (b), 23 (b), 25 c), | n.d. |
| Penicillium hordei | n.d. | 29 (c), 30 (a,b,c,d) | n.d. |
| Penicillium thomii | n.d. | 30 (b) | n.d. |
| Penicillium virgatum | n.d. | 5 (a), 24 (a,d), 25 (b,c,d), 28 (a,b) | n.d. |
| Phoma herbarum | n.d. | 1 (b,c), 2 (d), 5 (a), 24 (a) | n.d. |
| Pseudogymnoascus pannorum | 6 (a), 7 (b), 8 (a), 13 (a), 20 (a), 26 (a,b) | 6 (a), 7 (c), 12 (b), 15 (a), 20 (a), 21 (a), 29 (c) | n.d. |
| Trichoderma paraviridescens | n.d. | 15 (b,c,d), 19 (b) | n.d. |
Appendix B. Supplementary Correlation Analyses
References
- Vilas, R.A. Ecological and economical impact of bats on ecosystem. Int. J. Life Sci. 2016, 4, 432–440. [Google Scholar]
- Camacho, J.; Heyde, A.; Bhullar, B.-A.S.; Haelewaters, D.; Simmons, N.B.; Abzhanov, A. Peramorphosis, anevolutionary developmental mechanism in neotropical bat skull diversity. Dev. Dynam. 2019, 248, 1129–1143. [Google Scholar] [CrossRef]
- Kunz, T.H.; Lumsden, L.F. Ecology of cavity and foliage roosting bats. In Bat Ecology; The University of Chicago Press: Chicago, IL, USA, 2003; pp. 3–87. [Google Scholar]
- Johnson, J.S.; Lacki, M.J. Effects of reproductive condition, roost microclimate, and weather patterns on summer torpor use by a vespertilionid bat. Ecol. Evol. 2014, 4, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Federici, L.; Masulli, M.; De Laurenzi, V.; Allocati, N. An overview of bats microbiota and its implication in transmissible diseases. Front. Microbiol. 2020, 20, 1012189. [Google Scholar] [CrossRef]
- Wang, D.; Wang, F.; Huang, Z.; Li, A.; Dai, W.; Leng, H.; Jin, L.; Li, Z.; Sun, K.; Feng, J. Structure and assembly process of skin fungal communities among bat species in Northern China. Front. Microbiol. 2024, 15, 1458258. [Google Scholar] [CrossRef]
- Borzęcka, J.; Suchodolski, J.; Cal-Smok, M.; Furmankiewicz, J.; Ogórek, R. Insight into the Skin Mycobiota of Myotis myotis: How Age, Sex, and Biometric Traits Correlate with Fungal Diversity. Animals 2025, 15, 3020. [Google Scholar] [CrossRef]
- Minnis, A.M.; Lindner, D.L. Phylogenetic evaluation of Geomyces and allies reveals no close relatives of Pseudogymnoascus destructans, comb. nov., in bat hibernacula of eastern North America. Fungal Biol. 2013, 117, 638–649. [Google Scholar] [CrossRef]
- Ogórek, R.; Kurczaba, K.; Cal, M.; Apoznański, G.; Kokurewicz, T. A culture-based ID of micromycetes on the wing membranes of Greater mouse-eared bats (Myotis myotis) from the “Nietoperek” site (Poland). Animals 2020, 10, 1337. [Google Scholar] [CrossRef]
- Borzęcka, J.; Piecuch, A.; Kokurewicz, T.; Lavoie, K.H.; Ogórek, R. Greater mouse-eared bats (Myotis myotis) hibernating in the Nietoperek bat reserve (Poland) as a vector of airborne culturable fungi. Biology 2021, 10, 593. [Google Scholar] [CrossRef]
- Ogórek, R.; Dyląg, M.; Kozak, B. Dark stains on rock surfaces in Driny Cave (Little Carpathian Mountains, Slovakia). Extremophiles 2016, 20, 641–652. [Google Scholar] [CrossRef]
- Stupar, M.; Savković, Ž.; Pećić, M.; Jerinkić, D.; Jakovljević, O.; Popović, S. Some Like It Rock ‘N’ Cold: Speleomycology of Ravništarka Cave (Serbia). J. Fungi 2025, 11, 706. [Google Scholar] [CrossRef] [PubMed]
- Poulson, T.L.; White, W.B. The cave environment. Science 1969, 165, 971–981. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.F.; Karunarathna, S.C.; Tibpromma, S.; Chethana, K.W.T.; Hyde, K.D.; Elgorban, A.M.; Suwannarach, N.; Kumla, J.; Mortimer, P.E.; Hughes, A.C. Understanding the role of bats as fungal vectors in the environment. IMA Fungus 2024, 15, 28. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Directive 92/43/EEC—The Conservation of Natural Habitats and of Wild Fauna and Flora in the Habitats Directive. 1992. Available online: http://ec.europa.eu/environment/nature/legislation/habitatsdirective/index_en.htm (accessed on 18 December 2024).
- Rogowska, K.; Kokurewicz, T. The longest migrations of three bat species to the “Nietoperek” bat reserve (Western Poland). Berichte Naturforschenden Ges. Oberlausitz Suppl. Band 2007, 15, 53–60. [Google Scholar]
- Kokurewicz, T.; Apoznański, G.; Gyselings, R.; Kirkpatrick, L.; De Bruyn, L.; Haddow, J.; Glover, A.; Schofield, H.; Schmidt, C.; Bongers, F.; et al. 45 years of bat study and conservation in Nietoperek bat reserve (Western Poland). Nyctalus 2019, 19, 252–269. [Google Scholar]
- Hutterer, R.; Ivanova, T.; Meyer-Cords, C.; Rodrigues, L. Bat Migrations in Europe: A Review of Banding Data and Literature; Biologische Vielfalt in Deutschland; Federal Agency for Nature Conservation: Bonn, Germany, 2005; pp. 11–109. [Google Scholar]
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. PLoS Biol. 2020, 18, e3000410. [Google Scholar] [CrossRef]
- Dietz, C.; von Helversen, O. Illustrated Identification Key to the Bats of Europe. Tech. Rep. 2004, 1, 6–72. [Google Scholar]
- Brunet-Rossinni, A.K.; Wilkinson, G.S. Methods for age estimation and the study of senescence in bats. In Ecological and Behavioral Methods for the Study of Bats; Kunz, T.H., Parsons, S., Eds.; Johns Hopkins University Press: Baltimore, MD, USA, 2009. [Google Scholar]
- Vanderwolf, K.J.; McAlpine, D.F.; Malloch, D.; Forbes, G.J. Ectomycota associated with hibernating bats in eastern Canada caves prior to the emergence of white-nose syndrome. Northeast. Nat. 2013, 20, 115–130. [Google Scholar]
- Spychała, K.; Kłosińska, K.; Salwińska, W.; Ogórek, R. Diversity of soil-borne fungi isolated from places frequently visited by people in the city of Wrocław (Poland). Appl. Sci. 2024, 14, 2782. [Google Scholar] [CrossRef]
- Zalar, P.; Gostincar, C.; de Hoog, G.S.; Ursic, V.; Sudhadham, M.; Gunde-Cimerman, N. Redefinition of Aureobasidium pullulans and its varieties. Stud. Mycol. 2008, 61, 21–38. [Google Scholar] [CrossRef]
- Bensch, K.; Braun, U.; Groenewald, J.Z.; Crous, P.W. The genus Cladosporium. Stud. Mycol. 2012, 72, 1–401. [Google Scholar] [CrossRef] [PubMed]
- Woudenberg, J.H.; Groenewald, J.Z.; Binder, M.; Crous, P.W. Alternaria redefined. Stud. Mycol. 2013, 75, 171–212. [Google Scholar] [CrossRef] [PubMed]
- Visagie, C.M.; Hirooka, Y.; Tanney, J.B.; Whitfield, E.; Mwange, K.; Meijer, M.; Amend, A.S.; Seifert, K.A.; Samson, R.A. Aspergillus, Penicillium, and Talaromyces isolated from house dust samples collected around the world. Stud. Mycol. 2014, 78, 63–139. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Zheng, R.Y. New taxa of Ambomucor (Mucorales, Mucoromycotina) from China. Mycotaxon 2015, 30, 165–171. [Google Scholar] [CrossRef]
- Goh, J.; Nam, B.; Lee, J.S.; Mun, H.Y.; Oh, Y.; Lee, H.B.; Chung, N.; Choi, Y.J. First report of six Trichoderma species isolated from freshwater environment in Korea. Korean J. Mycol. 2018, 46, 213–225. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, D.X.; Duan, D.E.; Wang, Y.B.; Yu, H. Morphology, molecular characterization, and virulence of Beauveria pseudobassiana isolated from different hosts. J. Invertebr. Pathol. 2020, 172, 107333. [Google Scholar] [CrossRef]
- Crous, P.W.; Lombard, L.; Sandoval-Denis, M.; Seifert, K.A.; Schroers, H.J.; Chaverri, P.; Gené, J.; Guarro, J.; Hirooka, Y.; Bensch, K.; et al. Fusarium: More than a node or a foot-shaped basal cell. Stud. Mycol. 2021, 98, 100116. [Google Scholar] [CrossRef]
- Liao, C.; Senanayake, I.C.; Dong, W.; Thilini Chethana, K.W.; Tangtrakulwanich, K.; Zhang, Y.; Doilom, M. Taxonomic and phylogenetic updates on Apiospora: Introducing four new species from Wurfbainia villosa and Grasses in China. J. Fungi 2023, 9, 1087. [Google Scholar] [CrossRef]
- White, T.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M., Gelfand, D., Sninsky, J., White, T., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Glass, N.L.; Donaldson, G.C. Development of Primer Sets Designed for Use with the PCR to Amplify Conserved Genes from Filamentous Ascomycetes. Appl. Environ. Microbiol. 1995, 61, 1323–1330. [Google Scholar] [CrossRef]
- Zhang, T.; We, X.-L.; Zhang, Y.Q.; Liu, H.-Y.; Yu, L.-Y. Diversity and distribution of lichen-associated fungi in the Ny-Ålesund region (Svalbard, High Arctic) as revealed by 454 pyrosequencing. Sci. Rep. 2015, 5, 14850. [Google Scholar] [CrossRef]
- Grisnik, M.; Walker, D.M. Bat cutaneous microbial assemblage functional redundancy across a host-mediated disturbance. Microb. Ecol. 2024, 87, 161. [Google Scholar] [CrossRef] [PubMed]
- Vanderwolf, K.J.; McAlpine, D.F.; Forbes, G.J.; Malloch, D. Bat populations and cave microclimate prior to and at the outbreak of white-nose syndrome in New Brunswick. Can. Field-Nat. 2012, 126, 125–134. [Google Scholar] [CrossRef]
- Vanderwolf, K.J.; Malloch, D.; McAlpine, D.F. Fungi on white-nose infected bats (Myotis spp.) in Eastern Canada show no decline in diversity associated with Pseudogymnoascus destructans (Ascomycota: Pseudeurotiaceae). Int. J. Speleol. 2016, 45, 43–50. [Google Scholar] [CrossRef]
- Furtado, B.G.; Savi-Bortolotto, G.D.; Soto, T.S.; Rocha, L.O.; Supi, K.P.; Angioletto, E.; Carvalho, F. Molecular characterization of fungal species in bats from Brazil’s Atlantic forest. Braz. J. Biol. 2025, 85, e293859. [Google Scholar] [CrossRef]
- Jurado, V.; Porca, E.; Cuezva, S.; Fernandez-Cortes, A.; Sánchez-Moral, S.; Sáiz-Jiménez, C. Fungal outbreak in a show cave. Sci. Total Environ. 2010, 408, 3632–3638. [Google Scholar] [CrossRef]
- Raudabaugh, D.B.; Rivera, N.A.; Anchor, G.C.; Bach, E.; Miller, A.N.; Mateus-Pinilla, N.E. Preliminary Study of Cave Sample Storage Conditions on Fungal Community Diversity. Diversity 2021, 13, 188. [Google Scholar] [CrossRef]
- Stupar, M.; Savković, Ž.; Popović, S.; Simić, G.S.; Grbić, M.L. Speleomycology of air in Stopića cave (Serbia). Microb. Ecol. 2023, 86, 2021–2031. [Google Scholar] [CrossRef]
- Li, J.; Li, L.; Jiang, H.; Yuan, L.; Zhang, L.; Ma, J.E.; Zhang, X.; Cheng, M.; Chen, J. Fecal bacteriome and mycobiome in bats with diverse diets in South China. Curr. Microbiol. 2018, 75, 1352–1361. [Google Scholar] [CrossRef]
- Thomma, B.P.H.J. Alternaria spp.: From general saprophyte to specific parasite. Mol. Plant Pathol. 2003, 4, 225–236. [Google Scholar] [CrossRef]
- Deb, D.; Khan, A.; Dey, N. Phoma diseases: Epidemiology and control. Plant Pathol. 2020, 69, 1203–1217. [Google Scholar] [CrossRef]
- Dean, R.; Van Kan, J.A.L.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Rudd, J.J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 2012, 13, 414–430. [Google Scholar] [CrossRef]
- Kerth, G. Causes and consequences of sociality in bats. Bioscience 2008, 58, 737–746. [Google Scholar] [CrossRef]
- Lorch, J.M.; Meteyer, C.U.; Behr, M.J.; Boyles, J.G.; Cryan, P.M.; Hicks, A.C.; Ballmann, A.E.; Coleman, J.T.H.; Redell, D.N.; Reeder, D.M.; et al. Experimental infection of bats with Geomyces destructans causes white-nose syndrome. Nature 2011, 480, 376–378. [Google Scholar] [CrossRef] [PubMed]
- Poli, A.; Zanellati, A.; Piano, E.; Biagioli, F.; Coleine, C.; Nicolosi, G.; Selbmann, L.; Isaia, M.; Prigione, V.; Varese, G.C. Cultivable fungal diversity in two karstic caves in Italy: Under-investigated habitats as source of putative novel taxa. Sci. Rep. 2024, 14, 4164. [Google Scholar] [CrossRef] [PubMed]
- Pannkuk, E.L.; Blair, H.B.; Fischer, A.E.; Gerdes, C.L.; Gilmore, D.F.; Savary, B.J.; Risch, T.S. Triacylglyceride composition and fatty acyl saturation profile of a psychrophilic and psychrotolerant fungal species grown at different temperatures. Fungal Biol. 2014, 118, 792–799. [Google Scholar] [CrossRef]
- Hassan, N.; Rafiq, M.; Haya, M.; Shah, A.A.; Hasan, F. Psychrophilic and psychrotrophic fungi: A comprehensive review. Rev. Environ. Sci. Biotechnol. 2016, 15, 147–172. [Google Scholar] [CrossRef]
- Fernandes, E.K.; Rangel, D.E.; Moraes, A.M.; Bittencourt, V.R.; Roberts, D.W. Cold activity of Beauveria and Metarhizium, and thermotolerance of Beauveria. J Invertebr. Pathol. 2008, 98, 69–78. [Google Scholar] [CrossRef]
- Boratyński, J.; Rusiński, M.; Kokurewicz, T.; Bereszyński, A.; Wojciechowski, M.S. Clustering behavior in wintering greater mouse-eared bats Myotis myotis—The effect of micro-environmental conditions. Acta Chiropterologica 2012, 4, 417–424. [Google Scholar] [CrossRef]
- Zahn, A. Reproductive success, colony size and roost temperature in attic-dwelling bat Myotis myotis. J. Zool. 1999, 247, 275–280. [Google Scholar] [CrossRef]
- Pusz, W.; Król, M.; Zwijacz-Kozica, T. Airborne fungi as indicators of ecosystem disturbance: An example from selected Tatra Mountains caves (Poland). Aerobiologia 2018, 34, 111–118. [Google Scholar] [CrossRef]
- Jurado, V.; Del Rosal, Y.; Liñan, C.; Martin-Pozas, T.; Gonzalez-Pimentel, J.L.; Saiz-Jimenez, C. Diversity and seasonal dynamics of airborne fungi in nerja cave, Spain. Appl. Sci. 2021, 11, 6236. [Google Scholar] [CrossRef]
- Samson, R.A.; Houbraken, J.; Thrane, U.; Frisvad, J.C.; Andersen, B. Food and Indoor Fungi; CBS Laboratory Manual Series 2; CBS-KNAW Fungal Biodiversity Centre: Utrecht, The Netherlands, 2010; p. 390. [Google Scholar]
- Gocheva, Y.G.; Tosi, S.; Krumova, E.T.; Slokoska, L.S.; Miteva, J.G.; Vassilev, S.V.; Angelova, M.B. Temperature-downshift induction of antioxidant enzyme responses in fungi isolated from Antarctica. Extremophiles 2009, 13, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Miteva-Staleva, J.; Krumova, E.T.; Vassilev, S.V.; Angelova, M.B. Cold-stress response during the stationary-growth phase of Antarctic and temperate-climate Penicillium strains. Microbiology 2017, 163, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Nováková, A. Microscopic fungi isolated from the Domica Cave system (Slovak Karst National Park, Slovakia). A review. Int. J. Speleol. 2009, 38, 71–82. [Google Scholar] [CrossRef]
- Pitt, I.J. The current role of Aspergillus and Penicillium in human and animal health. J. Med. Vet. Mycol. 1994, 32, 17–32. [Google Scholar]
- Kwon-Chung, K.J.; Sugui, J.A. Aspergillus fumigatus—What makes the species a ubiquitous human fungal pathogen? PLoS Pathog. 2013, 9, e1003743. [Google Scholar] [CrossRef]
- Marshall, W. Aerial transport of keratinaceous substrate and distribution of the fungus Geomyces pannorum in Antarctic Soils. Microb. Ecol. 1998, 36, 212–219. [Google Scholar] [CrossRef]
- Lueschow, S.R.; Johnson, L.J.A.N.; Williams, T.F.; McCleery, R.; Porras-Alfaro, A. Opportunistic Pseudogymnoascus and Geomyces species isolated from humans and bats. Sydowia 2019, 71, 25–33. [Google Scholar] [CrossRef]
- Gardiner, J.D.; Dimitriadis, G.; Codd, J.R.; Nudds, R.L. A potential role for bat tail membranes in flight control. PLoS ONE 2011, 6, e18214. [Google Scholar] [CrossRef]
- Ogórek, R.; Dyląg, M.; Kozak, B.; Višňovská, Z.; Tančinová, D.; Lejman, A. Fungi isolated and quantified from bat guano and air in Harmanecká and Driny Caves (Slovakia). J. Cave Karst. Stud. 2016, 78, 41–49. [Google Scholar] [CrossRef]
- Voyron, S.; Lazzari, A. First mycological investigation on Italian bats. Hystrix 2011, 22, 189–197. [Google Scholar] [CrossRef]
- Becker, P.; Van Den Eynde, C.; Baert, F.; Elizabet, D.; De Pauw, R.; Normand, A.; Piarroux, R.; Stubbe, D. Remarkable fungal biodiversity on northern Belgium bats and hibernacula. Mycologia 2023, 115, 484–498. [Google Scholar] [CrossRef] [PubMed]
- Holz, P.H.; Lumsden, L.F.; Marenda, M.S.; Browning, G.F.; Hufschmid, J. Two subspecies of bent-winged bats (Miniopterus orianae bassanii and oceanensis) in southern Australia have diverse fungal skin flora but not Pseudogymnoascus destructans. PLoS ONE 2018, 13, e0204282. [Google Scholar] [CrossRef]
- Grbić, M.L.; Dimkić, I.; Savković, Ž.; Stupar, M.; Knežević, A.; Jelikić, A.; Unković, N. Mycobiome Diversity of the Cave Church of St. Peter and Paul in Serbia—Risk assessment implication for the conservation of rare cavern habitat housing a peculiar fresco painting. J. Fungi 2022, 8, 1263. [Google Scholar] [CrossRef]
- Cunha, A.O.B.; Bezerra, J.D.P.; Oliveira, T.G.L.; Barbier, E.; Bernard, E.; Machado, A.R.; Souza-Motta, C.M. Living in the dark: Bat caves as hotspots of fungal diversity. PLoS ONE 2020, 15, e0243494. [Google Scholar] [CrossRef]
- Sanchez-Moral, S.; Jurado, V.; Fernandez-Cortes, A.; Cuezva, S.; Martin-Pozas, T.; Gonzalez-Pimentel, J.L.; Ontañon, R.; Saiz-Jimenez, C. Environment-driven control of fungi in subterranean ecosystems: The case of La Garma Cave (Northern Spain). Int. Microbiol. 2021, 24, 573–591. [Google Scholar] [CrossRef]
- Jacobs, A.; Msimang, D.; Venter, E. First survey of the fungi from the Bakwena Cave in South Africa suggests low human disturbance. J. Cave Karst. Stud. 2017, 79, 89–94. [Google Scholar] [CrossRef]
- Šimonovičová, A.; Pangallo, D.; Chovanová, K.; Lehotská, B. Geomyces destructans associated with bat disease WNS detected in Slovakia. Biologia 2011, 66, 562–564. [Google Scholar] [CrossRef][Green Version]
- Visagie, C.M.; Yilmaz, N.; Vanderwolf, K.; Renaud, J.B.; Sumarah, M.W.; Houbraken, J.; Assebgui, R.; Seifert, K.A.; Malloch, D. Penicillium diversity in Canadian bat caves, including a new species, P. speluncae. Fungal Syst. Evol. 2020, 5, 1–15. [Google Scholar] [CrossRef]
- Swezey, C.S.; Garrity, C.P. Geographical and geological data from caves and mines infected with white-nose syndrome (WNS) before September 2009 in the eastern United States. J. Cave Karst. Stud. 2011, 73, 125–157. [Google Scholar] [CrossRef][Green Version]
- Urbina, J.; Chestnut, T.; Schwalm, D.; Allen, J.; Levi, T. Experimental evaluation of genomic DNA degradation rates for the pathogen Pseudogymnoascus destructans (Pd) in bat guano. PeerJ 2020, 8, e8141. [Google Scholar] [CrossRef] [PubMed]
- Urbina, J.; Chestnut, T.; Allen, J.M.; Levi, T. Pseudogymnoascus destructans growth in wood, soil and guano substrates. Sci. Rep. 2021, 11, 763. [Google Scholar] [CrossRef] [PubMed]
- Ogórek, R.; Speruda, M.; Borzęcka, J.; Piecuch, A.; Cal, M. First speleomycological study on the occurrence of psychrophilic and psychrotolerant aeromycota in the Brestovská cave (Western Tatras Mts., Slovakia) and first reports for some species at underground sites. Biology 2021, 10, 497. [Google Scholar] [CrossRef] [PubMed]
- Ogórek, R.; Suchodolski, J.; Piecuch, A.; Przywara, K.; Višňovská, Z. Keratinophilic and keratinolytic fungi in cave ecosystems: A culture-based study of Brestovská cave and Demänovská L’adová and Slobody caves (Slovakia). Appl. Sci. 2022, 12, 1455. [Google Scholar] [CrossRef]
- Ogórek, R.; Borzęcka, J.; Spychała, K.; Piecuch, A.; Suchodolski, J. Soil and sediments in natural underground ecosystems as a source of culturable micromycetes: A case study of the Brestovská cave (Western Tatras, Slovakia). Appl. Sci. 2024, 14, 3517. [Google Scholar] [CrossRef]
- Ogórek, R.; Spychała, K.; Cal, M.; Lejman, A.; Suchodolski, J. Speleomycological and chemical assessment of sediments in Demänovská Slobody cave (Slovakia). Int. Biodeterior. Biodegrad. 2024, 192, 105828. [Google Scholar] [CrossRef]
- Wasti, I.G.; Seelan, J.S.S. The fungal diversity of Madai Cave, Sabah, Malaysia. Asian Sch. Netw. 2019, 264–274. [Google Scholar]
- Kokurewicz, T.; Ogórek, R.; Pusz, W.; Matkowski, K. Bats Increase the Number of Cultivable Airborne Fungi in the “Nietoperek” Bat Reserve in Western Poland. Microb. Ecol. 2016, 72, 36–48. [Google Scholar] [CrossRef]
- Latgé, J.-P.; Chamilos, G. Aspergillus fumigatus and aspergillosis in 2019. Clin. Microbiol. Rev. 2020, 33, e00140-18. [Google Scholar] [CrossRef]
- Campbell, L.J.; Walsh, D.P.; Blehert, D.S.; Lorch, J.M. Log-term survival of Pseudogymnoascus destructans at elevated temperatures. J. Wildl. Dis. 2020, 56, 278–287. [Google Scholar] [CrossRef]
- Suchodolski, J.; Parol, M.; Cal-Smok, M.; Piecuch, A.; Ogórek, R. First report of increased amphotericin B resistance in Coniochaeta polymorpha isolates from Rangifer tarandus platyrhynchus droppings in Spitsbergen. Fungal Biol. 2025, 129, 101567. [Google Scholar] [CrossRef]



| Isolate Number | Identified Fungi | GenBank Accession No. | The Sequence Length (bp) | Identity with Sequence from GenBank | ||
|---|---|---|---|---|---|---|
| Query Cover (%) | Identity (%) | Accession | ||||
| UWR_744 | Aspergillus fumigatus | PX393922 * | 522 * | 100% * | 100% * | MN588001.1 * |
| UWR_745 | Aspergillus tubingensis | PX393923 */PX502280 ** | 403 */388 ** | 100% */100% ** | 99.50% */99.23% ** | HQ262499.1 */KU711869.1 ** |
| UWR_746 | Beauveria pseudobassiana | PX393924 | 390 | 100% | 100% | OR544477.1 |
| UWR_747 | Mucor fragilis | PX393925 | 412 | 100% | 100% | PV801950.1 |
| UWR_748 | Paecilomyces farinosus (syn. Cordyceps farinosa) | PX393926 | 448 | 100% | 100% | AF368793.1 |
| UWR_749 | Penicillium bialowiezense | PX393927/PX502281 | 459/381 | 100%/100% | 100/99.48% | OK094894.1/PP524992.1 |
| UWR_750 | Penicillium brevistipitatum | PX393928 | 357 | 100% | 100% | MW534763.1 |
| UWR_751 | Penicillium cavernicola | PX393929/PX502282 | 386/370 | 100%/100% | 99.22%/100% | NR_163684.1/KJ834439.1 |
| UWR_752 | Penicillium chrysogenum | PX393930 | 416 | 100% | 99.76% | MK690561.1 |
| UWR_753 | Penicillium concentricum | PX393931/PX502283 | 483/388 | 100%/100% | 100%/98.20% | PV871543.1/OR217443.1 |
| UWR_754 | Penicillium corylophilum | PX393932/PX502284 | 503/381 | 100%/100% | 100%/99.48% | JN986758.1/MK450958.1 |
| UWR_755 | Penicillium crocicola | PX393933/PX502285 | 459/361 | 100%/100% | 100%/100% | JX869556.1/KU516393.1 |
| UWR_756 | Penicillium crustosum | PX393934 | 413 | 100% | 100% | PV935558.1 |
| UWR_757 | Penicillium expansum | PX393935/PX502286 | 480/372 | 100%/100% | 100%/98.92% | MN587988.1/MT387277.1 |
| UWR_758 | Penicillium gladioli | PX393936 | 404 | 100% | 100% | PV688793.1 |
| UWR_759 | Penicillium griseofulvum | PX393937/PX502287 | 504/373 | 100%/100% | 100%/99.73% | KR135143.1/MW080344.1 |
| UWR_760 | Penicillium martensii | PX393938 | 451 | 100% | 100% | MH865218.1 |
| UWR_761 | Penicillium polonicum | PX393939 | 378 | 100% | 100% | OM892855.1 |
| UWR_762 | Penicillium robsamsonii | PX393940 | 374 | 100% | 100% | NR_144866.1 |
| UWR_763 | Penicillium thomii | PX393941 | 503 | 100% | 100% | OM415949.1 |
| UWR_764 | Pseudogymnoascus destructans | PX393942 | 421 | 100% | 100% | MT015949.1 |
| UWR_765 | Pseudogymnoascus pannorum | PX393943 | 394 | 100% | 100% | MT072091.1 |
| UWR_766 | Absidia virescens | PX393944 | 484 | 100% | 100% | MZ354150.1 |
| UWR_767 | Alternaria alternata | PX393945 | 474 | 100% | 100% | PP781340.1 |
| UWR_768 | Apiospora arundinis | PX393946 | 429 | 100% | 99.53% | KX778673.1 |
| UWR_769 | Aspergillus fumigatus | PX393947/PX502288 | 415/483 | 100%/100% | 100%/100% | OL589185.1/MN637746.1 |
| UWR_770 | Aspergillus tubingensis | PX393948 | 502 | 100% | 100% | PP718796.1 |
| UWR_771 | Aureobasidium pullulans | PX393949 | 366 | 100% | 100% | PQ849090.1 |
| UWR_772 | Botrytis cinerea | PX393950 | 367 | 100% | 99.18% | OQ625843.1 |
| UWR_773 | Chaetomium angustispirale | PX393951 | 368 | 100% | 99.73% | JN209862.1 |
| UWR_774 | Cladosporium allicinum | PX393952 | 358 | 100% | 99.44% | OK445637.1 |
| UWR_775 | Cladosporium cladosporioides | PX393953 | 481 | 100% | 100% | KM816685.1 |
| UWR_777 | Fusarium sporotrichioides | PX393955 | 374 | 100% | 100% | PQ340452.1 |
| UWR_778 | Mucor flavus | PX393956 | 400 | 99% | 99.75% | NR_103633.1 |
| UWR_779 | Mucor fragilis | PX393957 | 469 | 100% | 100% | PP956644.1 |
| UWR_780 | Mucor hiemalis | PX393958 | 457 | 100% | 100% | MN817788.1 |
| UWR_776 | Paecilomyces farinosus (syn. Cordyceps farinose) | PX393954 | 488 | 100% | 100% | PQ678803.1 |
| UWR_781 | Penicillium aurantiogriseum | PX393959 | 498 | 100% | 100% | MZ157166.1 |
| UWR_782 | Penicillium bialowiezense | PX393960 | 529 | 100% | 100% | OK510276.1 |
| UWR_783 | Penicillium chrysogenum | PX393961/PX502289 | 425/391 | 100%/100% | 100%/96.93% | PQ329214.1/KP329944.1 |
| UWR_784 | Penicillium commune | PX393962 | 372 | 100% | 100% | KF990135.1 |
| UWR_785 | Penicillium crustosum | PX393963 | 506 | 100% | 100% | KY558627.1 |
| UWR_786 | Penicillium dipodomyicola | PX393964 | 468 | 100% | 100% | DQ339570.1 |
| UWR_787 | Penicillium glabrum | PX393965 | 388 | 100% | 100% | MK761053.1 |
| UWR_788 | Penicillium griseofulvum | PX393966 | 503 | 100% | 100% | PV240451.1 |
| UWR_789 | Penicillium hordei | PX393967 | 420 | 100% | 100% | OR513086.1 |
| UWR_790 | Penicillium thomii | PX393968 | 499 | 100% | 100% | MZ423029.1 |
| UWR_791 | Penicillium virgatum | PX393969 | 389 | 100% | 100% | KF578441.1 |
| UWR_792 | Phoma herbarum | PX393970 | 455 | 100% | 100% | KP794136.1 |
| UWR_793 | Pseudogymnoascus pannorum | PX393971 | 465 | 100% | 100% | MW113278.1 |
| UWR_794 | Trichoderma paraviridescens | PX393972 | 507 | 100% | 100% | KJ728696.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ogórek, R.; Suchodolski, J.; Borzęcka, J.; Kokurewicz, T. Seasonal Turnover in Bat Skin Mycobiota: Contrasting Fungal Communities Between Hibernation and Reproduction in Greater Mouse-Eared Bats (Myotis myotis). Pathogens 2026, 15, 83. https://doi.org/10.3390/pathogens15010083
Ogórek R, Suchodolski J, Borzęcka J, Kokurewicz T. Seasonal Turnover in Bat Skin Mycobiota: Contrasting Fungal Communities Between Hibernation and Reproduction in Greater Mouse-Eared Bats (Myotis myotis). Pathogens. 2026; 15(1):83. https://doi.org/10.3390/pathogens15010083
Chicago/Turabian StyleOgórek, Rafał, Jakub Suchodolski, Justyna Borzęcka, and Tomasz Kokurewicz. 2026. "Seasonal Turnover in Bat Skin Mycobiota: Contrasting Fungal Communities Between Hibernation and Reproduction in Greater Mouse-Eared Bats (Myotis myotis)" Pathogens 15, no. 1: 83. https://doi.org/10.3390/pathogens15010083
APA StyleOgórek, R., Suchodolski, J., Borzęcka, J., & Kokurewicz, T. (2026). Seasonal Turnover in Bat Skin Mycobiota: Contrasting Fungal Communities Between Hibernation and Reproduction in Greater Mouse-Eared Bats (Myotis myotis). Pathogens, 15(1), 83. https://doi.org/10.3390/pathogens15010083








