Epidemiology of Malaria in Northern Madagascar as Exemplified by the Mampikony District
Abstract
1. Introduction
2. Materials and Methods
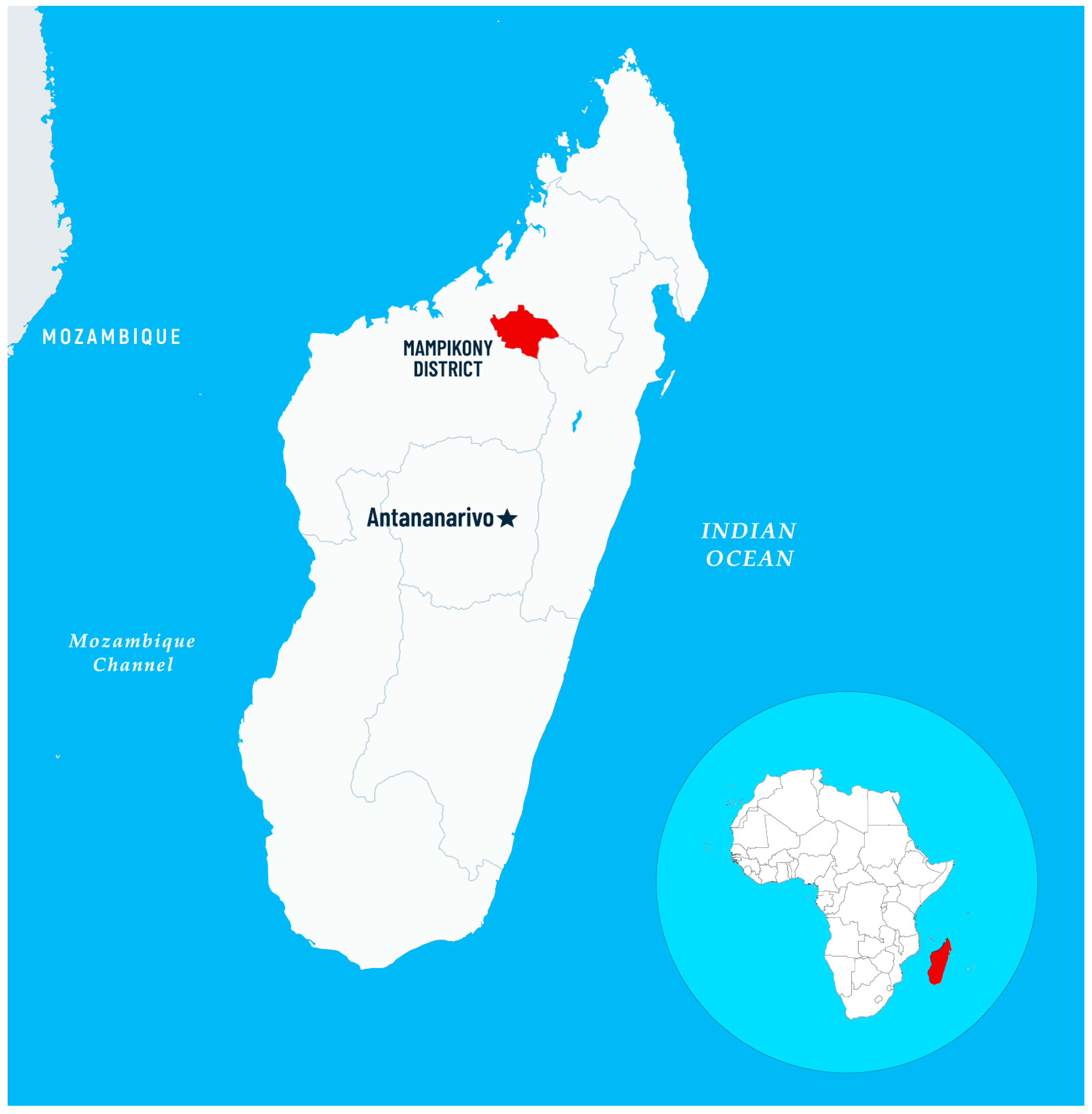
2.1. Study Group
2.2. Malaria Screening
2.2.1. Malaria Rapid Diagnostic Tests (mRDT)
2.2.2. Molecular Diagnostics RT-PCR
2.3. Statistical Analysis
2.4. Ethical Approval
3. Results
4. Discussion
5. Limitations of the Study
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Plewes, K.; Leopold, S.J.; Kingston, H.W.; Dondorp, A.M. Malaria. What’s New in the Management of Malaria? Infect. Dis. Clin. N. Am. 2019, 33, 39–60. [Google Scholar] [CrossRef] [PubMed]
- Douglas, R.G.; Amino, R.; Sinnis, P.; Frischknecht, F. Active migration and passive transport of malaria parasites. Trends Parasitol. 2015, 31, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Rakotoarison, N.; Raholijao, N.; Razafindramavo, L.M.; Rakotomavo, Z.A.P.H.; Rakotoarisoa, A.; Guillemot, J.S.; Randriamialisoa, Z.J.; Mafilaza, V.; Ramiandrisoa, V.A.M.P.; Rajaonarivony, R.; et al. Assessment of Risk, Vulnerability and Adaptation to Climate Change by the Health Sector in Madagascar. Int. J. Environ. Res. Public Health 2018, 15, 2643. [Google Scholar] [CrossRef] [PubMed]
- Barmania, S. Madagascar’s health challenges. Lancet 2015, 386, 729–730. [Google Scholar] [CrossRef] [PubMed]
- World Bank Group. February 2025. Available online: https://www.worldbank.org/en/country/madagascar/publication/madagascar-economic-update (accessed on 27 March 2025).
- Ministere de L’Economie et des Finanses. Antananarivo. 2018. Available online: http://mef.gov.mg/page_personnalisee/index/news/122 (accessed on 23 March 2025).
- Anand, A.; Favero, R.; Dentinger, C.; Ralaivaomisa, A.; Ramamonjisoa, S.; Rabozakandraina, O.; Razafimandimby, E.; Razafindrakoto, J.; Wolf, K.; Steinhardt, L.; et al. Malaria case management and elimination readiness in health facilities of five districts of Madagascar in 2018. Malar. J. 2020, 19, 351. [Google Scholar] [CrossRef] [PubMed]
- Direction de Lutte Contre le Paludisme. Plan Stratégique National de Lutte Contre le Paludisme, 2018–2022. Elimination Progressive du Paludisme à Madagascar. 2018. Available online: https://www.scribd.com/document/589248203/PSN-2018-2022-avec-PSE (accessed on 23 March 2025).
- World Health Organization. World Malaria Report 2024; WHO: Geneva, Switzerland, 2024. [Google Scholar]
- Hanboonkunupakarn, B.; White, N.J. Advances and roadblocks in the treatment of malaria. Br. J. Clin. Pharmacol. 2022, 88, 374–382. [Google Scholar] [CrossRef] [PubMed]
- UNICEF. Protection Against Malaria: At the Heart of the Prevention Campaign. 15 February 2024. Available online: https://www.unicef.org/madagascar/en/stories/protection-against-malaria-heart-prevention-campaign (accessed on 23 March 2025).
- Atlas Visual de la Malaria, March 2025. Available online: https://atlasvisualdelamalaria.org/paises/malaria-madagascar (accessed on 23 March 2025).
- Arambepola, R.; Keddie, S.H.; Collins, E.L.; Twohig, K.A.; Amratia, P.; Bertozzi-Villa, A.; Chestnutt, E.G.; Harris, J.; Millar, J.; Rozier, J.; et al. Spatiotemporal mapping of malaria prevalence in Madagascar using routine surveillance and health survey data. Sci. Rep. 2020, 10, 18129. [Google Scholar] [CrossRef] [PubMed]
- Howes, R.E.; Franchard, T.; Rakotomanga, T.A.; Ramiranirina, B.; Zikursh, M.; Cramer, E.Y.; Tisch, D.J.; Kang, S.Y.; Ramboarina, S.; Ratsimbasoa, A.; et al. Risk Factors for Malaria Infection in Central Madagascar: Insights from a Cross-Sectional Population Survey. Am. J. Trop. Med. Hyg. 2018, 9, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Bousema, T.; Okell, L.; Felger, I.; Drakeley, C. Asymptomatic malaria infections: Detectability, transmissibility and public health relevance. Nat. Rev. Microbiol. 2014, 12, 833–840. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. 2015. Available online: https://www.who.int/docs/default-source/documents/global-technical-strategy-for-malaria-2016-2030.pdf (accessed on 23 March 2025).
- Severe Malaria Observatory. 2024. Available online: https://www.severemalaria.org/countries/madagascar (accessed on 25 March 2025).
- SlidePlayer. 2025. Available online: https://slideplayer.fr/slide/5486905/ (accessed on 23 March 2025).
- Basu, S.; Sahi, P.K. Malaria: An Update. Indian J. Pediatr. 2017, 84, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Bento, A.I.; Nguyen, T.; Wing, C.; Lozano-Rojas, F.; Ahn, Y.; Simon, K. Plasmodium vivax clinical malaria is commonly observed in Duffy-negative Malagasy people. Proc. Natl. Acad. Sci. USA 2010, 107, 5967–5971. [Google Scholar] [CrossRef]
- Zimmerman, P.A. Plasmodium vivax Infection in Duffy-Negative People in Africa. Am. J. Trop. Med. Hyg. 2017, 97, 636–638. [Google Scholar] [CrossRef] [PubMed]
- African Leaders Malaria Alliance. Africa Malaria Progress Report 2023. Available online: https://alma2030.org/heads-of-state-and-government/african-union-malaria-progress-reports/2023-africa-malaria-progress-report/ (accessed on 27 March 2025).
- Centers fro Diseases Control and Prevention. 2024. Available online: https://www.cdc.gov/global-health/media/pdfs/2024/11/Madagascar-2024-Fact-Sheet.pdf (accessed on 28 March 2025).
- Arisco, N.J.; Rice, B.L.; Tantely, L.M.; Girod, R.; Emile, G.N.; Randriamady, H.J.; Castro, M.C.; Golden, C.D. Variation in Anopheles distribution and predictors of malaria infection risk across regions of Madagascar. Malar. J. 2020, 19, 348. [Google Scholar] [CrossRef] [PubMed]
- Rice, B.L.; Golden, C.D.; Randriamady, H.J.; Rakotomalala, A.A.N.A.; Vonona, M.A.; Anjaranirina, E.J.G.; Hazen, J.; Castro, M.C.; Metcalf, C.J.E.; Hartl, D.L. Fine-scale variation in malaria prevalence across ecological regions in Madagascar: A cross-sectional study. BMC Public Health 2021, 21, 1018. [Google Scholar] [CrossRef] [PubMed]
- BMJ Global Health. Using Routine Health Data to Evaluate the Impact of Indoor Residual Spraying on Malaria Transmission in Madagascar. 18 July 2023. Available online: https://gh.bmj.com/content/8/7/e010818 (accessed on 28 March 2025).
- Mouchet, J.; Blanchy, S.; Rakotonjanabelo, A.; Ranaivoson, G.; Rajaonarivelo, E.; Laventure, S. Stratification épidémiologique du paludisme à Madagascar. Arch. Inst. Pasteur Madagascar. 1993, 60, 50–59. [Google Scholar] [PubMed]
- Iyaloo, D.P.; Zohdy, S.; Carney, R.M.; Mosawa, V.R.; Elahee, K.B.; Munglee, N.; Latchooman, N.; Puryag, S.; Bheecarry, A.; Bhoobun, H.; et al. A regional One Health approach to the risk of invasion by Anopheles stephensi in Mauritius. PLoS Neglected Trop. Dis. 2024, 18, e0011827. [Google Scholar] [CrossRef] [PubMed]
- Nepomichene, T.N.; Tata, E.; Boyer, S. Malaria case in Madagascar, probable implication of a new vector, Anopheles coustani. Malar. J. 2015, 14, 475. [Google Scholar] [CrossRef] [PubMed]
- Institut Pasteur de Madagascar. 2025. Available online: https://www.pasteur.mg/projet-rise-missions-survano-paludisme-madagascar/ (accessed on 28 March 2025).
| Cycles | Phase | Temperature [°C] | Time [min] |
|---|---|---|---|
| 1 | Polymerase activation | 95 | 14:30 |
| 50 | Denaturation | 97 | 00:30 |
| Annealing (data collection) | 60 | 01:00 | |
| 1 | Hold | 32 | 05:00 |
| 2023–2024 | 2023 | 2024 | ||||
|---|---|---|---|---|---|---|
| Total | 782 | 484 | 298 | |||
| N | % | N | % | N | % | |
| Gender | ||||||
| Female | 431 | 55.1 | 277 | 57.2 | 154 | 51.7 |
| Male | 351 | 44.9 | 207 | 42.8 | 144 | 48.3 |
| Age | ||||||
| <5 | 182 | 23.3 | 76 | 15.7 | 106 | 35.6 |
| 5–14 | 481 | 61.5 | 323 | 66.7 | 158 | 53.0 |
| >15 | 119 | 15.2 | 85 | 17.6 | 34 | 11.4 |
| Medical Data | ||||||
| Symptoms | 54 | 6.9 | 19 | 3.9 | 35 | 11.7 |
| Antimalarial drugs | 6 | 0.8 | 3 | 0.6 | 3 | 1.0 |
| Variables | mRDT | RT-PCR | p-Value | |||
|---|---|---|---|---|---|---|
| Pf | Pf + Pan | Total | P. falciparum | |||
| N (%) [95% Cl] | N (%) [95% Cl] | N (%) [95% Cl] | N (%) [95% Cl] | |||
| Total | 484 (100%) | 8 (1.7) [0.5; 2.8] | 2 (0.4) [0.0; 1.0] | 10 (2.1) [0.8; 3.3] | 27 (5.6) [3.5; 7.6] | |
| Gender | 0.326 1 | |||||
| Female | 277(57.2%) | 4 (1.4) [0.0; 2.8] | 1 (0.4) [0.0; 1.1] | 5 (1.8) [0.2; 3.4] | 13 (14.3) [2.2; 7.2] | |
| Male | 207 (42.8%) | 4 (1.9) [0.1; 3.8] | 1 (0.5) [0.0; 1.4] | 5 (2.4) [0.3; 4.5] | 14 (6.8) [3.3; 10.2] | |
| Age | 0.022 1 | |||||
| <5 | 76 (15.7%) | 0 (0.0) [0.0; 0.0] | 0 (0.0) [0.0; 0.0] | 0 (0.0) [0.0; 0.0] | 4 (5.3) [0.1; 10.3] | |
| 6–14 | 323 (66.7%) | 6 (1.9) [0.4; 3.3] | 0 (0.0) [0.0; 0.0] | 6 (1.9) [0.4; 3.3] | 13 (4.0) [1.9; 6.2] | |
| 15–60 | 85 (17.6%) | 2 (2.4) [0.0; 5.6] | 2 (2.4) [0.0; 5.6] | 4 (4.8) [0.2; 9.2] | 10 (11.8) [4.9; 18.6] | |
| Variables | mRDT | RT-PCR | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Pf | Pf + Pan | Total | P. falciparum | P. vivax | P. falciparum + P. vivax | Total | |||
| N (%) [95% Cl] | N (%) [95% Cl] | N (%) [95% Cl] | N (%) [95% Cl] | N (%) [95% Cl] | N (%) [95% Cl] | N (%) [95% Cl] | |||
| Total | 298 (100%) | 10 (3.4) [1.3; 5.4] | 15 (5.0) [2.6; 7.5] | 25 (8.4) [5.2; 11.5] | 34 (11.4) [7.8; 15.1] | 2 (0.7) [0.0; 1.6] | 1 (0.3) [0.0; 1.0] | 37 (12.4) [8.7; 16.2] | |
| Gender | 0.039 1 | ||||||||
| Female | 154 (51.7%) | 8 (5.2) [1.7; 8.7] | 8 (5.2) [1.7; 8.7] | 16 (10.4) [5.6; 15.2] | 22 (14.3) [8.8; 19.8] | 2 (1.3) [0.0; 3.1] | 1 (0.7) [0.0; 1.9] | 25 (16.3) [10.4; 22.1] | |
| Male | 144 (48.3%) | 2 (1.4) [0.0; 3.3] | 7 (4.9) [1.3; 8.4] | 9 (6.3) [2.3; 10.2] | 12 (8.3) [3.8; 12.8] | 0 (0.0) [0.0; 0.0] | 0 (0.0) [0.0; 0.0] | 12 (8.3) [3.8; 12.8] | |
| Age | 0.910 1 | ||||||||
| <5 | 106 (35.6%) | 0 (0.0) [0.0; 0.0] | 5 (4.7) [0.7; 8.8] | 5 (4.7) [0.7; 8.8] | 12 (11.3) [5.3; 17.4] | 1 (0.9) [0.0; 2.8] | 0 (0.0) [0.0; 0.0] | 13 (12.4) [6.0; 18.5] | |
| 6–14 | 158 (53.0%) | 8 (5.1) [1.6; 8.5] | 8 (5.1) [1.6; 8.5] | 16 (10.2) [5.4; 14.8] | 17 (10.8) [5.9; 15.6] | 1 (0.6) [0.0; 1.9] | 1 (0.6) [0.0; 1.9] | 19 (12.0) [6.9; 17.1] | |
| 15–81 | 34 (11.4%) | 2 (5.9) [0.0; 13.8] | 2 (5.9) [0.0; 13.8] | 4 (11.8) [0.9; 22.6] | 5 (14.7) [2.8; 22.6] | 0 (0.0) [0.0; 0.0] | 0 (0.0) [0.0; 0.0] | 5 (14.7) [2.8; 22.6] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kołodziej, D.; Wilczyńska, W.; Kasprowicz, D.; Marchelek-Myśliwiec, M.; Korzeniewski, K. Epidemiology of Malaria in Northern Madagascar as Exemplified by the Mampikony District. Pathogens 2025, 14, 848. https://doi.org/10.3390/pathogens14090848
Kołodziej D, Wilczyńska W, Kasprowicz D, Marchelek-Myśliwiec M, Korzeniewski K. Epidemiology of Malaria in Northern Madagascar as Exemplified by the Mampikony District. Pathogens. 2025; 14(9):848. https://doi.org/10.3390/pathogens14090848
Chicago/Turabian StyleKołodziej, Daria, Wanesa Wilczyńska, Daniel Kasprowicz, Małgorzata Marchelek-Myśliwiec, and Krzysztof Korzeniewski. 2025. "Epidemiology of Malaria in Northern Madagascar as Exemplified by the Mampikony District" Pathogens 14, no. 9: 848. https://doi.org/10.3390/pathogens14090848
APA StyleKołodziej, D., Wilczyńska, W., Kasprowicz, D., Marchelek-Myśliwiec, M., & Korzeniewski, K. (2025). Epidemiology of Malaria in Northern Madagascar as Exemplified by the Mampikony District. Pathogens, 14(9), 848. https://doi.org/10.3390/pathogens14090848







