Detection of Schistosoma mekongi DNA in Human Stool and Intermediate Host Snail Neotricula aperta via Loop-Mediated Isothermal Amplification Assay in Lao PDR
Abstract
1. Introduction
2. Materials and Methods
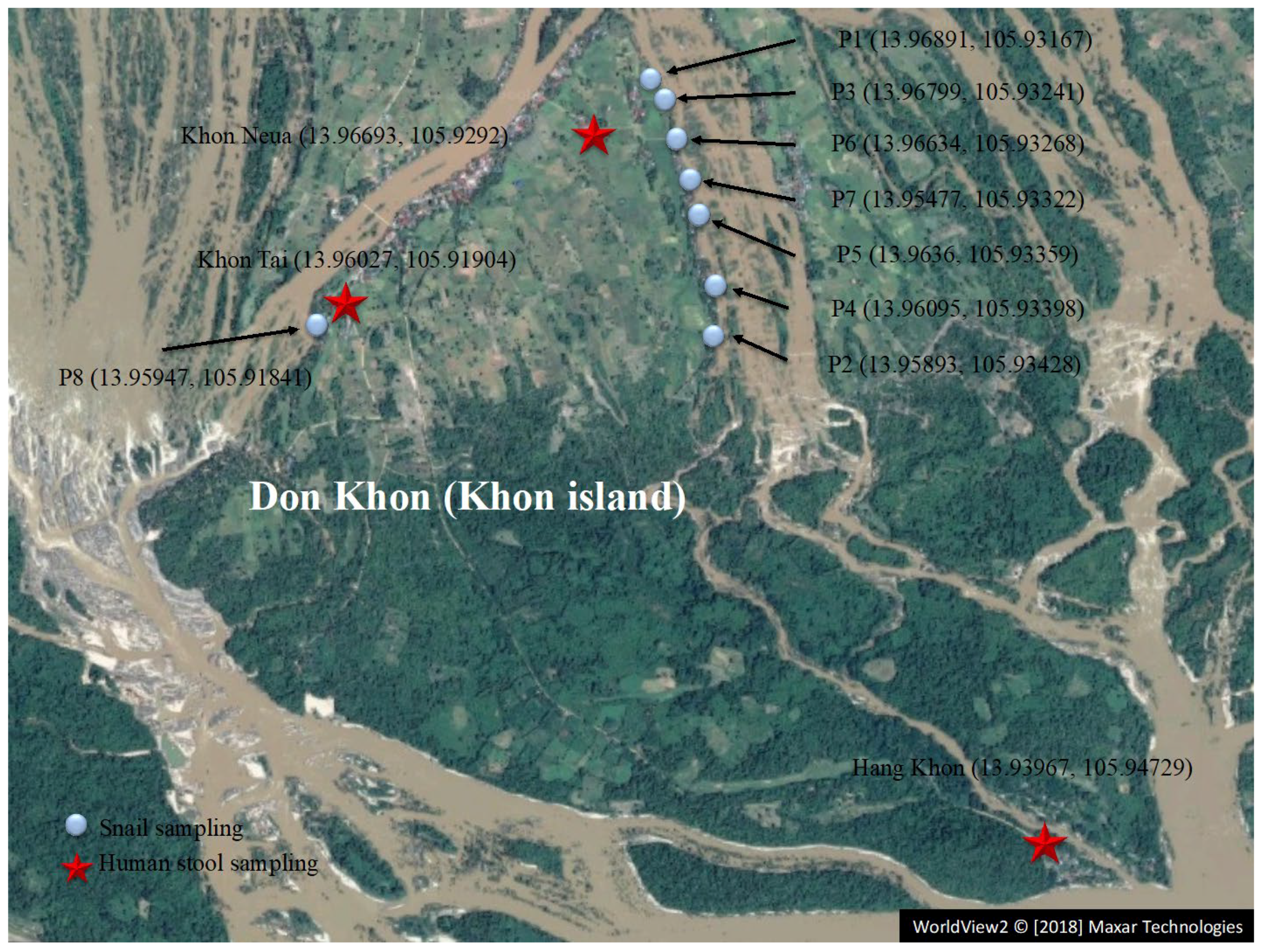
2.1. Study Area
2.2. Human Stool Sampling
2.3. Snail Sampling
2.4. Parasitological Tests Using the Kato–Katz Method
2.5. DNA Extraction
2.6. LAMP Assay
2.7. Heat Mapping
2.8. Statistical Analysis
3. Results
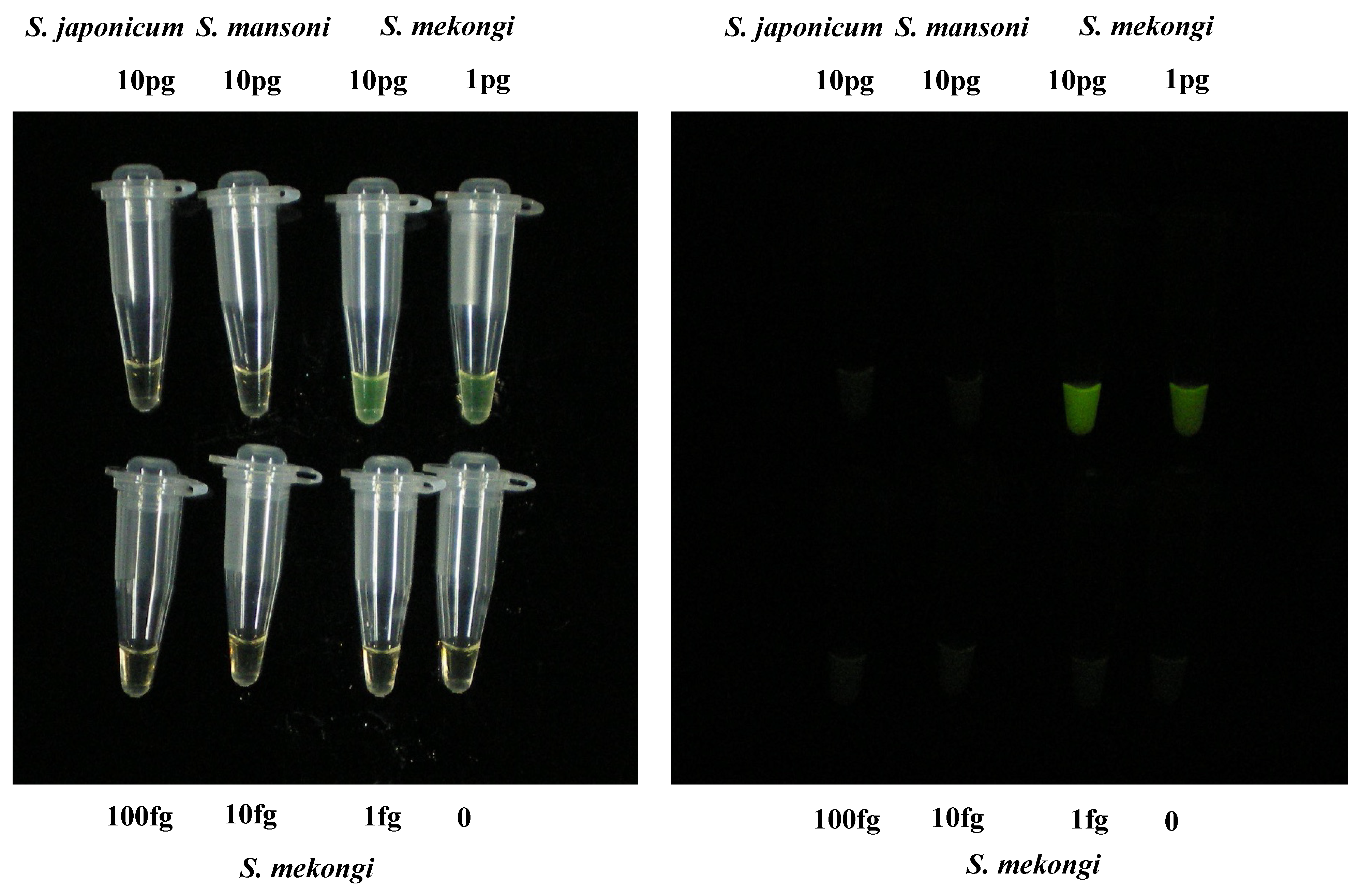
3.1. Sensitivity and Specificity of LAMP Assay Targeting S. mekongi
3.2. Comparison of LAMP Detection and the Kato–Katz Method in Stool Samples
3.3. Large-Scale Monitoring of Snail Samples via LAMP Detection
3.4. Heat Map of Infected Residents and Infected Snail Sampling Location and the Relationship between Infection and Lifestyle Habits
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Schistosomiasis and Soil-Transmitted Helminthiases: Numbers of People Treated in 2019–Schistosomiase et Géohelminthiases: Nombre de Personnes Traitées En 2019. Wkly. Epidemiol. Rec. = Relev. Épidémiologique HEBD. 2019, 95, 629–640. [Google Scholar]
- Ohmae, H.; Sinuon, M.; Kirinoki, M.; Matsumoto, J.; Chigusa, Y.; Socheat, D.; Matsuda, H. Schistosomiasis Mekongi: From Discovery to Control. Parasitol. Int. 2004, 53, 135–142. [Google Scholar] [CrossRef]
- Muth, S.; Sayasone, S.; Odermatt-Biays, S.; Phompida, S.; Duong, S.; Odermatt, P. Schistosoma Mekongi in Cambodia and Lao People’s Democratic Republic. Adv. Parasitol. 2010, 72, 179–203. [Google Scholar] [CrossRef]
- Attwood, S.W.; Fatih, F.A.; Campbell, I.; Upatham, E.S. The Distribution of Mekong Schistosomiasis, Past and Future: Preliminary Indications from an Analysis of Genetic Variation in the Intermediate Host. Parasitol. Int. 2008, 57, 256–270. [Google Scholar] [CrossRef]
- Biays, S.; Stich, A.H.R.; Odermatt, P.; Long, C.; Yersin, C.; Man, C.; Saem, C.; Lormand, J.D. Foyer de Bilharziose a Schistosoma Mekongi Redecouvert Au Nord Du Cambodge: I. Perception Culturelle de La Maladie; Description et Suivi de 20 Cas Cliniques Graves. Trop. Med. Int. Health 1999, 4, 662–673. [Google Scholar] [CrossRef]
- Urbani, C.; Sinoun, M.; Socheat, D.; Pholsena, K.; Strandgaard, H.; Odermatt, P.; Hatz, C. Epidemiology and Control of Mekongi Schistosomiasis. Acta Trop. 2002, 82, 157–168. [Google Scholar] [CrossRef]
- Khieu, V.; Sayasone, S.; Muth, S.; Kirinoki, M.; Laymanivong, S.; Ohmae, H.; Huy, R.; Chanthapaseuth, T.; Yajima, A.; Phetsouvanh, R.; et al. Elimination of Schistosomiasis Mekongi from Endemic Areas in Cambodia and the Lao People’s Democratic Republic: Current Status and Plans. Trop. Med. Infect. Dis. 2019, 4, 30. [Google Scholar] [CrossRef]
- WHO. Expert Consultation to Accelerate Elimination of Asian Schistosomiasis, Shanghai, China, 22–23 May 2017: Meeting Report. Manila: WHO Regional Office for the Western Pacific. 2017. Available online: https://apps.who.int/iris/handle/10665/259630 (accessed on 10 October 2022).
- Assaré, R.K.; Tra, M.B.I.; Ouattara, M.; Hürlimann, E.; Coulibaly, J.T.; N’Goran, E.K.; Utzinger, J. Sensitivity of the Point-of-Care Circulating Cathodic Antigen Urine Cassette Test for Diagnosis of Schistosoma Mansoni in Low-Endemicity Settings in Côte d’Ivoire. Am. J. Trop. Med. Hyg. 2018, 99, 1567–1572. [Google Scholar] [CrossRef]
- Sanpool, O.; Intapan, P.M.; Thanchomnang, T.; Sri-Aroon, P.; Lulitanond, V.; Sadaow, L.; Maleewong, W. Development of a Real-Time PCR Assay with Fluorophore-Labelled Hybridization Probes for Detection of Schistosoma Mekongi in Infected Snails and Rat Feces. Parasitology 2012, 139, 1266–1272. [Google Scholar] [CrossRef]
- Frickmann, H.; Loderstädt, U.; Nickel, B.; Poppert, S.; Odermatt, P.; Sayasone, S.; Van Esbroeck, M.; Micalessi, I.; Cnops, L.; Adisakwattana, P.; et al. Low Sensitivity of Real Time PCRs Targeting Retrotransposon Sequences for the Detection of Schistosoma Japonicum Complex DNA in Human Serum. Pathogens 2021, 10, 1067. [Google Scholar] [CrossRef]
- Kumagai, T.; Furushima-Shimogawara, R.; Ohmae, H.; Wang, T.P.; Lu, S.; Chen, R.; Wen, L.; Ohta, N. Detection of Early and Single Infections of Schistosoma Japonicum in the Intermediate Host Snail, Oncomelania Hupensis, by PCR and Loop-Mediated Isothermal Amplification (LAMP) Assay. Am. J. Trop. Med. Hyg. 2010, 83, 542–548. [Google Scholar] [CrossRef] [PubMed]
- Tong, Q.B.; Chen, R.; Zhang, Y.; Yang, G.J.; Kumagai, T.; Furushima-Shimogawara, R.; Lou, D.; Yang, K.; Wen, L.Y.; Lu, S.H.; et al. A New Surveillance and Response Tool: Risk Map of Infected Oncomelania Hupensis Detected by Loop-Mediated Isothermal Amplification (LAMP) from Pooled Samples. Acta Trop. 2015, 141, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Hamburger, J.; Abbasi, I.; Kariuki, C.; Wanjala, A.; Mzungu, E.; Mungai, P.; Muchiri, E.; King, C.H. Evaluation of Loop-Mediated Isothermal Amplification Suitable for Molecular Monitoring of Schistosome-Infected Snails in Field Laboratories. Am. J. Trop. Med. Hyg. 2013, 88, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Gandasegui, J.; Fernández-Soto, P.; Hernández-Goenaga, J.; López-Abán, J.; Vicente, B.; Muro, A. Biompha-LAMP: A New Rapid Loop-Mediated Isothermal Amplification Assay for Detecting Schistosoma Mansoni in Biomphalaria Glabrata Snail Host. PLoS Negl. Trop. Dis. 2016, 10, e0005225. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Soto, P.; Arahuetes, J.G.; Hernández, A.S.; Abán, J.L.; Santiago, B.V.; Muro, A. A Loop-Mediated Isothermal Amplification (LAMP) Assay for Early Detection of Schistosoma Mansoni in Stool Samples: A Diagnostic Approach in a Murine Model. PLoS Negl. Trop. Dis. 2014, 8, e3126. [Google Scholar] [CrossRef] [PubMed]
- Lodh, N.; Mikita, K.; Bosompem, K.M.; Anyan, W.K.; Quartey, J.K.; Otchere, J.; Shiff, C.J. Point of Care Diagnosis of Multiple Schistosome Parasites: Species-Specific DNA Detection in Urine by Loop-Mediated Isothermal Amplification (LAMP). Acta Trop. 2017, 173, 125–129. [Google Scholar] [CrossRef]
- Caldeira, R.L.; Jannotti-Passos, L.K.; Dos Santos Carvalho, O. Use of Molecular Methods for the Rapid Mass Detection of Schistosoma mansoni (Platyhelminthes: Trematoda) in Biomphalaria spp. (Gastropoda: Planorbidae). J. Trop. Med. 2017, 2017, 8628971. [Google Scholar] [CrossRef]
- Javier, G.; Fernández-Soto, P.; Muro, A.; Barbosa, C.S.; de Melo, F.L.; Loyo, R.; de Souza Gomes, E.C. A Field Survey Using LAMP Assay for Detection of Schistosoma Mansoni in a Low-Transmission Area of Schistosomiasis in Umbuzeiro, Brazil: Assessment in Human and Snail Samples. PLoS Negl. Trop. Dis. 2018, 12, e0006314. [Google Scholar] [CrossRef]
- Lindholz, C.G.; Favero, V.; de Marco Verissimo, C.; Candido, R.R.F.; de Souza, R.P.; dos Santos, R.R.; Morassutti, A.L.; Bittencourt, H.R.; Jones, M.K.; St. Pierre, T.G.; et al. Study of Diagnostic Accuracy of Helmintex, Kato-Katz, and POC-CCA Methods for Diagnosing Intestinal Schistosomiasis in Candeal, a Low Intensity Transmission Area in Northeastern Brazil. PLoS Negl. Trop. Dis. 2018, 12, e0006274. [Google Scholar] [CrossRef]
- He, P.; Gordon, C.A.; Williams, G.M.; Li, Y.; Wang, Y.; Hu, J.; Gray, D.J.; Ross, A.G.; Harn, D.; McManus, D.P. Real-Time PCR Diagnosis of Schistosoma Japonicum in Low Transmission Areas of China. Infect. Dis. Poverty 2018, 7, 8. [Google Scholar] [CrossRef] [PubMed]
- Gandasegui, J.; Fernández-Soto, P.; Carranza-Rodríguez, C.; Pérez-Arellano, J.L.; Vicente, B.; López-Abán, J.; Muro, A. The Rapid-Heat LAMPellet Method: A Potential Diagnostic Method for Human Urogenital Schistosomiasis. PLoS Negl. Trop. Dis. 2015, 9, 1–23. [Google Scholar] [CrossRef]
- Kubota, T.; Shige, S.; Hashizume, H.; Aonashi, K.; Takahashi, N.; Seto, S.; Hirose, M.; Takayabu, Y.N.; Ushio, T.; Nakagawa, K.; et al. Global Precipitation Map Using Satellite-Borne Microwave Radiometers by the GSMaP Project: Production and Validation. IEEE Trans. Geosci. Remote Sens. 2007, 45, 2259–2275. [Google Scholar] [CrossRef]
- Wan, Z. New Refinements and Validation of the Collection-6 MODIS Land-Surface Temperature/Emissivity Product. Remote Sens. Environ. 2014, 140, 36–45. [Google Scholar] [CrossRef]
- Id, J.T.; Turner, H.C.; Truscott, J.E.; Werkman, M.; Phillips, E.; Id, R.A.; Medley, G.F.; King, C.H.; Anderson, R.M. The Design of Schistosomiasis Monitoring and Evaluation Programmes: The Importance of Collecting Adult Data to Inform Treatment Strategies for Schistosoma Mansoni. PLoS Negl. Trop. Dis. 2018, 12, e0006717. [Google Scholar] [CrossRef]
- Toor, J.; Alsallaq, R.; Truscott, J.E.; Turner, H.C.; Werkman, M.; Gurarie, D.; King, C.H.; Anderson, R.M. Are We on Our Way to Achieving the 2020 Goals for Schistosomiasis Morbidity Control Using Current World Health Organization Guidelines? Clin. Infect. Dis. 2018, 66, S245–S252. [Google Scholar] [CrossRef]
- De Ruiter, C.M.; Van Der Veer, C.; Leeflang, M.M.G.; Deborggraeve, S.; Lucas, C.; Adams, E.R. Molecular Tools for Diagnosis of Visceral Leishmaniasis: Systematic Review and Meta-Analysis of Diagnostic Test Accuracy. J. Clin. Microbiol. 2014, 52, 3147–3155. [Google Scholar] [CrossRef]
- Oriero, E.C.; Jacobs, J.; van Geertruyden, J.P.; Nwakanma, D.; D’alessandro, U. Molecular-Based Isothermal Tests for Field Diagnosis of Malaria and Their Potential Contribution to Malaria Elimination. J. Antimicrob. Chemother. 2015, 70, 2–13. [Google Scholar] [CrossRef]
- Rahman, S.M.M.; Song, H.B.; Jin, Y.; Oh, J.K.; Lim, M.K.; Hong, S.T.; Choi, M.H. Application of a Loop-Mediated Isothermal Amplification (LAMP) Assay Targeting Cox1 Gene for the Detection of Clonorchis Sinensis in Human Fecal Samples. PLoS Negl. Trop. Dis. 2017, 11, e0005995. [Google Scholar] [CrossRef]
- Wong, Y.P.; Othman, S.; Lau, Y.L.; Radu, S.; Chee, H.Y. Loop-Mediated Isothermal Amplification (LAMP): A Versatile Technique for Detection of Micro-Organisms. J. Appl. Microbiol. 2018, 124, 626–643. [Google Scholar] [CrossRef]
- Chadsuthi, S.; Chalvet-monfray, K.; Wiratsudakul, A.; Suwancharoen, D. A Remotely Sensed Flooding Indicator Associated with Cattle and Buffalo Leptospirosis Cases in Thailand 2011–2013. BMC Infect. Dis. 2018, 18, 602. [Google Scholar] [CrossRef]
- Kitikoon, V.; Schneider, C.R.; Sornmani, S.; Harinasuta, C.; Lanza, G.R. Mekong Schistosomiasis: 2. Evidence of the Natural Transmission of Schistosoma Japonicum, Mekong Strain, at Khong Island, Laos. Southeast Asian J Trop. Med. Public Health 1973, 4, 350–358. [Google Scholar] [PubMed]
- Attwood SW Schistosomiasis in the Mekong Region: Epidemiology and Phylogeography. Adv. Parasitol. 2001, 50, 87–152.
- Grimes, J.E.; Croll, D.; Harrison, W.E.; Utzinger, J.; Freeman, M.C.; Templeton, M.R. The Roles of Water, Sanitation and Hygiene in Reducing Schistosomiasis: A Review. Parasites Vectors 2015, 8, 156. [Google Scholar] [CrossRef] [PubMed]
- Atalabi, T.E.; Adoh, S.D.; Eze, K.M. The Current Epidemiological Status of Urogenital Schistosomiasis among Primary School Pupils in Katsina State, Nigeria: An Imperative for a Scale up of Water and Sanitation Initiative and Mass Administration of Medicines with Praziquantel. PLoS Negl. Trop. Dis. 2018, 12, e0006636. [Google Scholar] [CrossRef]
- Abdulkareem, B.O.; Habeeb, K.O.; Kazeem, A.; Adam, A.O.; Samuel, U.U. Urogenital Schistosomiasis among Schoolchildren and the Associated Risk Factors in Selected Rural Communities of Kwara State, Nigeria. J. Trop. Med. 2018, 2018, 6913918. [Google Scholar] [CrossRef]
- Campbell, S.J.; Savage, G.B.; Gray, D.J.; Atkinson, J.A.M.; Soares Magalhães, R.J.; Nery, S.V.; McCarthy, J.S.; Velleman, Y.; Wicken, J.H.; Traub, R.J.; et al. Water, Sanitation, and Hygiene (WASH): A Critical Component for Sustainable Soil-Transmitted Helminth and Schistosomiasis Control. PLoS Negl. Trop. Dis. 2014, 8, e2651. [Google Scholar] [CrossRef]
- Grimes, J.E.T.; Templeton, M.R. School Water, Sanitation, and Hygiene to Reduce the Transmission of Schistosomes and Soil-Transmitted Helminths. Trends Parasitol. 2016, 32, 661–664. [Google Scholar] [CrossRef]
- Campbell, S.J.; Biritwum, N.K.; Woods, G.; Velleman, Y.; Fleming, F.; Stothard, J.R. Tailoring Water, Sanitation, and Hygiene (WASH) Targets for Soil-Transmitted Helminthiasis and Schistosomiasis Control. Trends Parasitol. 2018, 34, 53–63. [Google Scholar] [CrossRef]
- Madon, S.; Malecela, M.N.; Mashoto, K.; Donohue, R.; Mubyazi, G.; Michael, E. The Role of Community Participation for Sustainable Integrated Neglected Tropical Diseases and Water, Sanitation and Hygiene Intervention Programs: A Pilot Project in Tanzania. Soc. Sci. Med. 2018, 202, 28–37. [Google Scholar] [CrossRef]
- Abou-Zeid, A.H.A.; Abkar, T.A.; Mohamed, R.O. Schistosomiasis and Soil-Transmitted Helminths among an Adult Population in a War Affected Area, Southern Kordofan State, Sudan. Parasites Vectors 2012, 5, 133. [Google Scholar] [CrossRef]
- Nyati-Jokomo, Z.; Chimbari, M.J. Risk Factors for Schistosomiasis Transmission among School Children in Gwanda District, Zimbabwe. Acta Trop. 2017, 175, 84–90. [Google Scholar] [CrossRef] [PubMed]

| LAMP Method | Kato–Katz Method | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Schitosoma mekongi | S. mekongi | Opisthorchis viverrini | Hookworms | Trichuris trichiura | Taenia sp | |||||||
| Village | Positive | Negative | Positive | Negative | Positive | Negative | Positive | Negative | Positive | Negative | Positive | Negative |
| Khon Neua | 4 (4.2%) | 92 | 0 | 96 | 48 (50.0%) | 48 | 22 (22.9%) | 74 | 1 (1.0%) | 95 | 2 (2.1%) | 94 |
| Khon Tai | 3 (3.4%) | 86 | 1 (1.1%) | 88 | 59 (66.3%) | 30 | 20 (22.5%) | 69 | 0 | 89 | 5 (5.6%) | 84 |
| Hang Khon | 1 (1.1%) | 86 | 0 | 87 | 45 (51.7%) | 42 | 20 (23.0%) | 67 | 3 (3.4%) | 84 | 0 | 87 |
| Total | 8 (2.9%) | 264 | 1 (0.4%) | 271 | 152 (55.9%) | 120 | 62 (22.8%) | 210 | 4 (1.5%) | 268 | 7 (2.6%) | 265 |
| Year | 2016 | 2017 | 2018 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Spot | Total Number of Examined Snails | Number of Positive Groups/Number of Total Groups | Infection Rate (95%CI) (%) | Total Number of Examined Snails | Number of Positive Groups/Number of Total Groups | Infection Rate (95%CI) (%) | Total Number of Examined Snails | Number of Positive Groups /Number of Total Groups | Infection Rate (95%CI) (%) | ||
| Group Size of Snails Crushed Together | Group size of Snails Crushed Together | Group Size of Snails Crushed Together | |||||||||
| 176 or 86 | 200 | 50 | 200 | 50 | |||||||
| P1 | 200 | - | 0/1 | 0 (0~1.49) | - | - | - | - | - | - | - |
| P2 | 1400 | - | 3/7 | 0.28 (0.07~0.74) | 1000 | 1/20 | - | 0.10 (0.005~0.56) a | 1000 | 0/20 | 0 (0~0.36) |
| P2 | - | - | - | - | 1000 | - | 1/5 | 0.11 (0.005~0.53) b | - | - | - |
| P3 | 176 | 0/1 | - | 0 (0~1.69) | - | - | - | - | - | - | - |
| P4 | 400 | - | 2/2 | not calculated—(0.13~) c | 600 | 1/12 | - | 0.17 (0.008~0.92) | 1000 | 0/20 | 0 (0~0.36) |
| P5 | 86 | 1/1 | - | not calculated—(0.06~) c | 700 | 0/14 | - | 0 (0~0.54) | 1000 | 0/20 | 0 (0~0.36) |
| P6 | 600 | - | 0/3 | 0 (0~0.50) | 400 | 0/8 | - | 0 (0~0.90) | - | - | - |
| P7 | - | - | - | - | 1050 | 0/21 | - | 0 (0~0.35) | - | - | - |
| P8 | - | - | - | - | 1150 | 0/23 | - | 0 (0~0.31) | - | - | - |
| Total | 2862 | 5900 | 3000 | ||||||||
| c: 95%CI lower bound | a: estimated value by using a group size of 50 b: estimated value by using a group size of 200 | ||||||||||
| Variables | Total | Negative | Positive | p-Value |
|---|---|---|---|---|
| (n = 275) | (n = 267) | (n = 8) | ||
| Gender 1 | □ | □ | □ | □ |
| Female | 164 | 160 | 4 | 0.414 |
| Male | 111 | 107 | 4 | □ |
| Age 1 | □ | □ | □ | □ |
| Child (≤18) | 53 | 53 | 1 | 0.508 |
| Adult | 220 | 212 | 7 | □ |
| Educational status 1 | □ | □ | □ | □ |
| No grade completed | 42 | 40 | 2 | 0.849 |
| Primary school | 135 | 131 | 4 | □ |
| Secondary school | 96 | 94 | 2 | □ |
| Higher | 2 | 2 | 0 | □ |
| Occupation 1,2 | □ | □ | □ | □ |
| Agriculture | 179 | 173 | 6 | 0.667 |
| Housewife | 20 | 19 | 1 | □ |
| Other | 18 | 18 | 0 | □ |
| Village 1 | □ | □ | □ | □ |
| Khon Neua | 96 | 92 | 4 | 0.442 |
| Khon Tai | 89 | 86 | 3 | □ |
| Hang Khon | 90 | 89 | 1 | □ |
| Number of people in the household (mean SD) 3 | 6.1 (2.3) | 6.3 (2.2) | 6.0 (2.7) | 0.753 |
| Family use of Mekong River 1 | □ | □ | □ | □ |
| No | 32 | 29 | 3 | 0.054 |
| Yes | 243 | 238 | 5 | □ |
| Well 1 | □ | □ | □ | □ |
| No | 252 | 247 | 5 | 0.022 * |
| Yes | 23 | 20 | 3 | □ |
| Rainwater storage 1 | □ | □ | □ | □ |
| No | 268 | 260 | 8 | 0.811 |
| Yes | 7 | 7 | 0 | □ |
| Purchase of bottled water 1 | □ | □ | □ | □ |
| No | 213 | 205 | 8 | 0.126 |
| Yes | 62 | 62 | 0 | □ |
| Latrine1 | □ | □ | □ | □ |
| No | 72 | 67 | 5 | 0.031 * |
| Yes | 203 | 200 | 3 | □ |
| Enter Mekong River 1 | □ | □ | □ | □ |
| No | 48 | 45 | 3 | 0.147 |
| Yes | 227 | 222 | 5 | □ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumagai, T.; Matsumoto-Takahashi, E.L.A.; Ishikawa, H.; Keomalaphet, S.; Khattignavong, P.; Soundala, P.; Hongvanthong, B.; Oyoshi, K.; Sasaki, Y.; Mizukami, Y.; et al. Detection of Schistosoma mekongi DNA in Human Stool and Intermediate Host Snail Neotricula aperta via Loop-Mediated Isothermal Amplification Assay in Lao PDR. Pathogens 2022, 11, 1413. https://doi.org/10.3390/pathogens11121413
Kumagai T, Matsumoto-Takahashi ELA, Ishikawa H, Keomalaphet S, Khattignavong P, Soundala P, Hongvanthong B, Oyoshi K, Sasaki Y, Mizukami Y, et al. Detection of Schistosoma mekongi DNA in Human Stool and Intermediate Host Snail Neotricula aperta via Loop-Mediated Isothermal Amplification Assay in Lao PDR. Pathogens. 2022; 11(12):1413. https://doi.org/10.3390/pathogens11121413
Chicago/Turabian StyleKumagai, Takashi, Emilie Louise Akiko Matsumoto-Takahashi, Hirofumi Ishikawa, Sengdeuane Keomalaphet, Phonepadith Khattignavong, Pheovaly Soundala, Bouasy Hongvanthong, Kei Oyoshi, Yoshinobu Sasaki, Yousei Mizukami, and et al. 2022. "Detection of Schistosoma mekongi DNA in Human Stool and Intermediate Host Snail Neotricula aperta via Loop-Mediated Isothermal Amplification Assay in Lao PDR" Pathogens 11, no. 12: 1413. https://doi.org/10.3390/pathogens11121413
APA StyleKumagai, T., Matsumoto-Takahashi, E. L. A., Ishikawa, H., Keomalaphet, S., Khattignavong, P., Soundala, P., Hongvanthong, B., Oyoshi, K., Sasaki, Y., Mizukami, Y., Kano, S., Brey, P. T., & Iwagami, M. (2022). Detection of Schistosoma mekongi DNA in Human Stool and Intermediate Host Snail Neotricula aperta via Loop-Mediated Isothermal Amplification Assay in Lao PDR. Pathogens, 11(12), 1413. https://doi.org/10.3390/pathogens11121413








