Robotic Liver Surgery for Alveolar Echinococcosis: A Single-Centre Experience
Abstract
1. Introduction
2. Results
2.1. Patients’ Baseline Characteristics
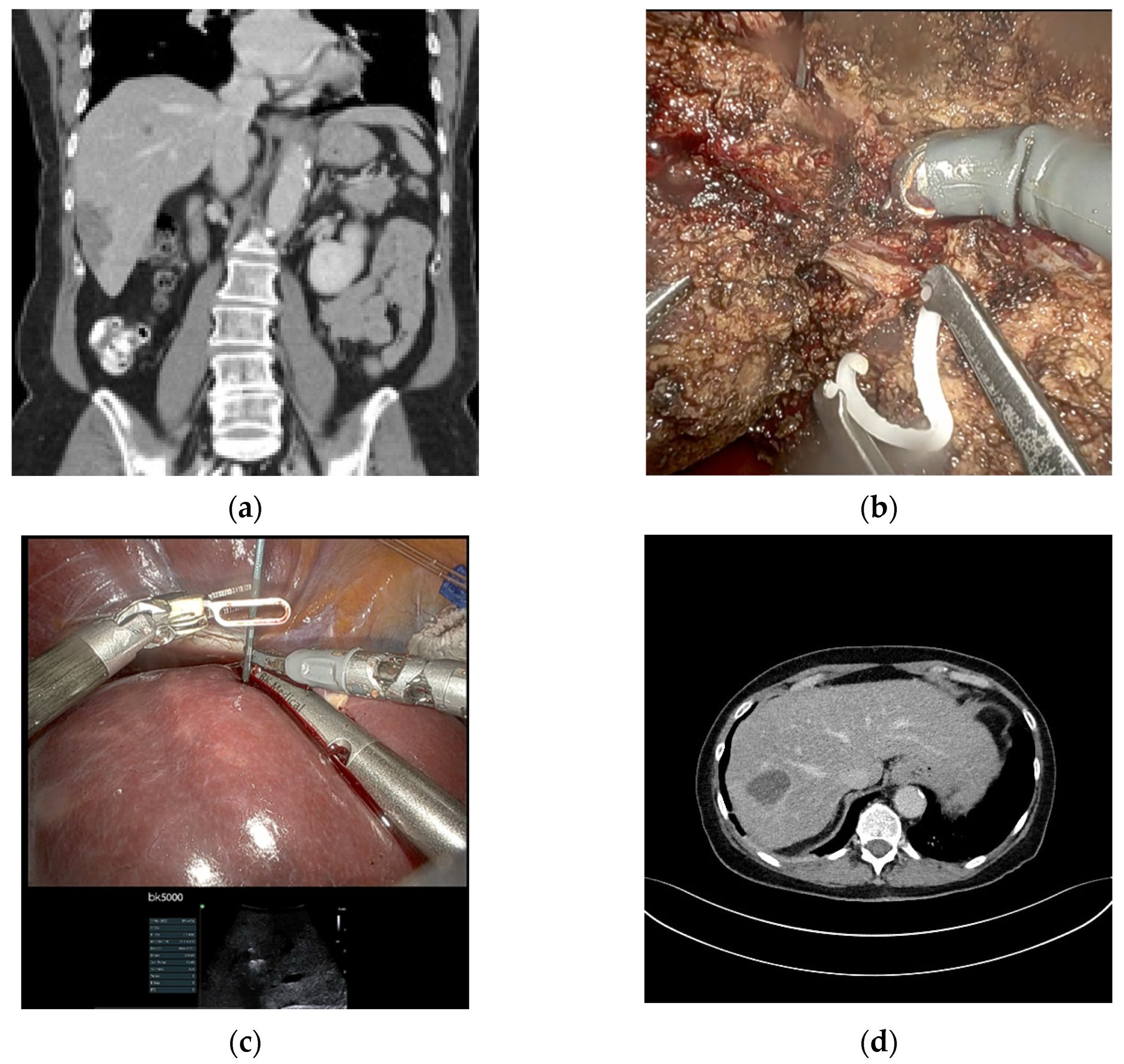
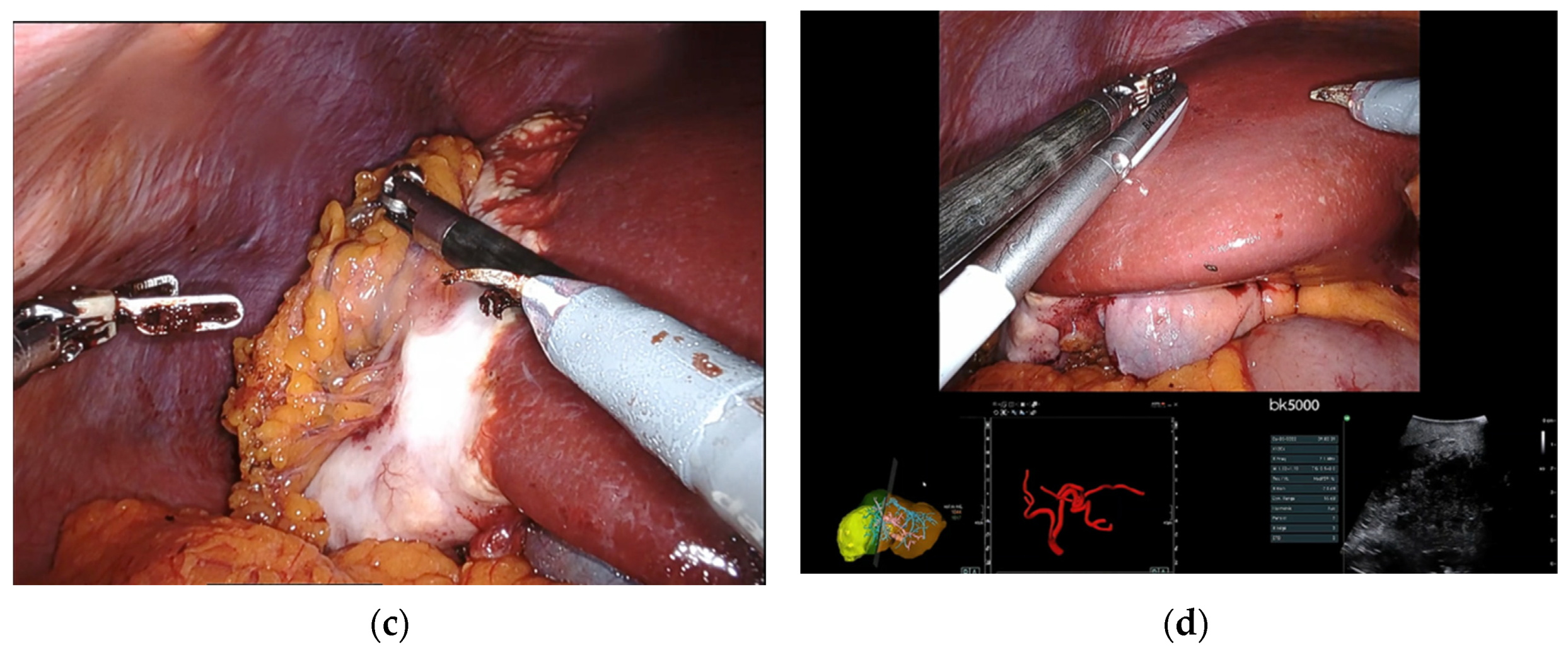
2.2. Surgical Details
2.3. Postoperative Course
3. Discussion
4. Materials and Methods
4.1. Patient Selection and Preoperative Management
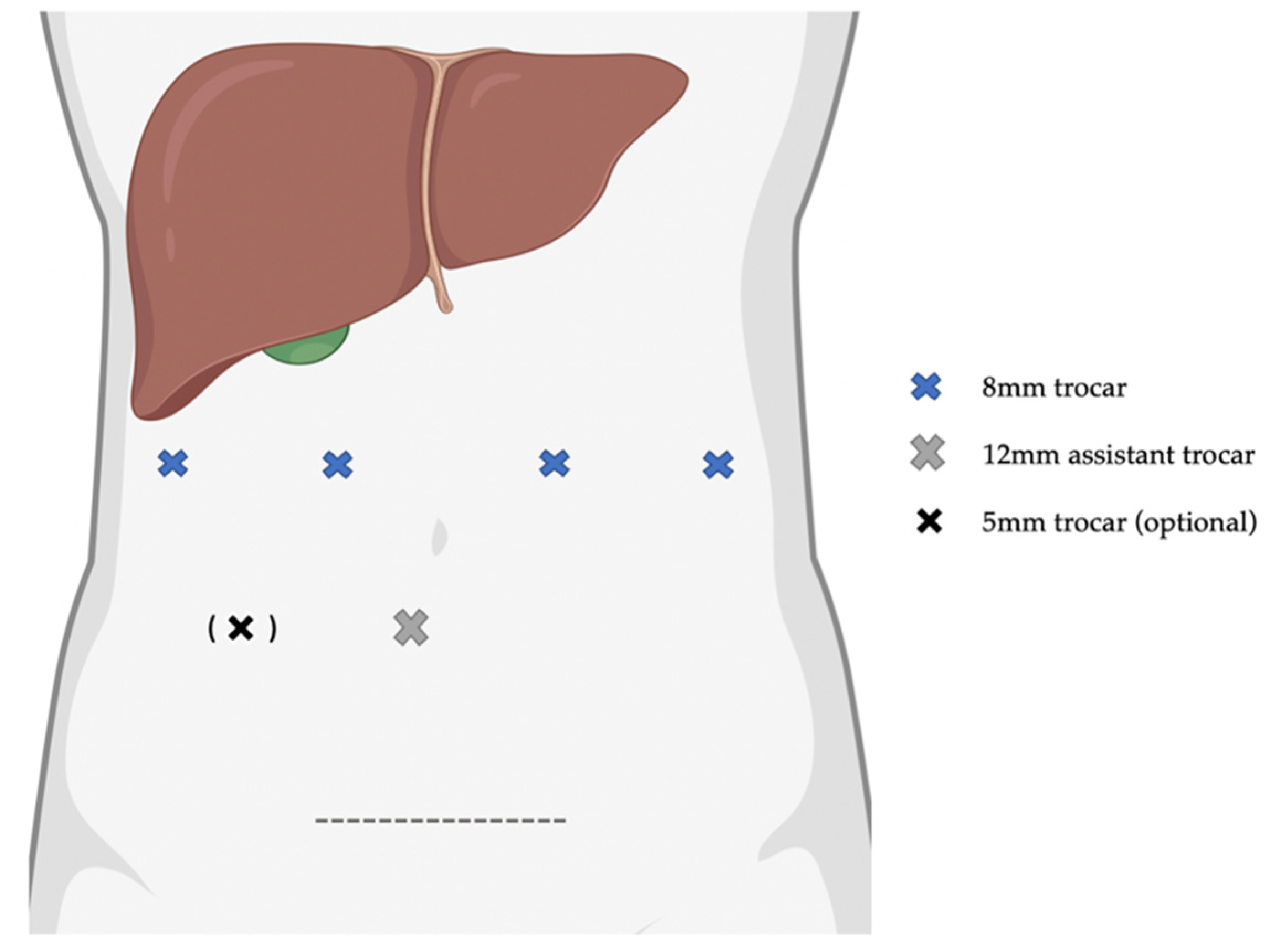
4.2. Surgical Procedure
4.3. Clinical Definitions
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wen, H.; Vuitton, L.; Tuxun, T.; Li, J.; Vuitton, D.A.; Zhang, W.; McManus, D.P. Echinococcosis: Advances in the 21st Century. Clin. Microbiol. Rev. 2019, 32, e00075-18. [Google Scholar] [CrossRef] [PubMed]
- Conraths, F.J.; Deplazes, P. Echinococcus multilocularis: Epidemiology, surveillance and state-of-the-art diagnostics from a veterinary public health perspective. Vet. Parasitol. 2015, 213, 149–161. [Google Scholar] [CrossRef]
- Dominique, A.V.; Wang, Q.; Zhou, H.X.; Francis, R.; Jenny, K.; Solange, B.H.; Wen, H.; Patrick, G. A historical view of alveolar echinococcosis, 160 years after the discovery of the first case in humans: Part 1. What have we learnt on the distribution of the disease and on its parasitic agent? Chin. Med. J.-Peking 2011, 124, 2943–2953. [Google Scholar]
- Jenkins, D.J. WHO/OIE manual on Echinococcosis in humans and animals: A public health problem of global concern. Int. J. Parasitol. 2001, 31, 1717–1718. [Google Scholar] [CrossRef]
- Graeter, T.; Bao, H.; Delabrousse, E.; Brumpt, E.; Shi, R.; Li, W.; Jiang, Y.; Schmidberger, J.; Kratzer, W.; Liu, W. Hepatic alveolar echinococcosis: Comparative computed tomography study between two Chinese and two European centres. Food Waterborne Parasitol. 2020, 19, e00082. [Google Scholar] [CrossRef]
- Eckert, J.; Deplazes, P. Biological, Epidemiological, and Clinical Aspects of Echinococcosis, a Zoonosis of Increasing Concern. Clin. Microbiol. Rev. 2004, 17, 107–135. [Google Scholar] [CrossRef]
- Nunnari, G.; Pinzone, M.R.; Gruttadauria, S.; Celesia, B.M.; Madeddu, G.; Malaguarnera, G.; Pavone, P.; Cappellani, A.; Cacopardo, B. Hepatic echinococcosis: Clinical and therapeutic aspects. World J. Gastroenterol. 2012, 18, 1448–1458. [Google Scholar] [CrossRef]
- Vuitton, D.A.; McManus, D.P.; Rogan, M.T.; Romig, T.; Gottstein, B.; Naidich, A.; Tuxun, T.; Wen, H.; da Silva, A.M. International consensus on terminology to be used in the field of echinococcoses. Parasite 2020, 27, 41. [Google Scholar] [CrossRef]
- Grüner, B.; Schmidberger, J.; Drews, O.; Kratzer, W.; Gräter, T. Ulm for the IEWG. Imaging in alveolar echinococcosis (AE): Comparison of Echinococcus multilocularis classification for computed-tomography (EMUC-CT) and ultrasonography (EMUC-US). Radiol. Infect. Dis. 2017, 4, 70–77. [Google Scholar] [CrossRef]
- McManus, D.P.; Gray, D.J.; Zhang, W.; Yang, Y. Diagnosis, treatment, and management of echinococcosis. BMJ Br. Med. J. 2012, 344, e3866. [Google Scholar] [CrossRef]
- Kern, P.; Da Silva, A.M.; Akhan, O.; Müllhaupt, B.; Vizcaychipi, K.A.; Budke, C.; Vuitton, D.A. The Echinococcoses: Diagnosis, Clinical Management and Burden of Disease. Adv. Parasitol. 2017, 96, 259–369. [Google Scholar] [PubMed]
- Brunetti, E.; Kern, P.; Vuitton, D.A. WHO-IWGE WP for the. Expert consensus for the diagnosis and treatment of cystic and alveolar echinococcosis in humans. Acta Trop. 2010, 114, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ito, A. Review of “Echinococcus and Echinococcosis, Part A.” edited by R. C. Andrew Thompson, Alan J. Lymbery and Peter Deplazes. Parasite Vectors 2017, 10, 408. [Google Scholar] [CrossRef][Green Version]
- Botezatu, C.; Mastalier, B.; Patrascu, T. Hepatic hydatid cyst—Diagnose and treatment algorithm. J. Med. Life 2018, 11, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Higuita, N.I.A.; Brunetti, E.; McCloskey, C. Cystic Echinococcosis. J. Clin. Microbiol. 2015, 54, 518–523. [Google Scholar] [CrossRef]
- Budke, C.M.; Carabin, H.; Ndimubanzi, P.C.; Nguyen, H.; Rainwater, E.; Dickey, M.; Bhattarai, R.; Zeziulin, O.; Qian, M.B. A Systematic Review of the Literature on Cystic Echinococcosis Frequency Worldwide and Its Associated Clinical Manifestations. Am. J. Trop. Med. Hyg. 2013, 88, 1011–1027. [Google Scholar] [CrossRef]
- Torgerson, P.R.; Keller, K.; Magnotta, M.; Ragland, N. The Global Burden of Alveolar Echinococcosis. PLoS Negl. Trop. Dis. 2010, 4, e722. [Google Scholar] [CrossRef]
- Lötsch, F.; Budke, C.M.; Auer, H.; Kaczirek, K.; Waneck, F.; Lagler, H.; Ramharter, M. Evaluation of direct costs associated with alveolar and cystic echinococcosis in Austria. PLoS Negl. Trop. Dis. 2019, 13, e0007110. [Google Scholar] [CrossRef]
- Reuter, S.; Jensen, B.; Buttenschoen, K.; Kratzer, W.; Kern, P. Benzimidazoles in the treatment of alveolar echinococcosis: A comparative study and review of the literature. J. Antimicrob. Chemother. 2000, 46, 451–456. [Google Scholar] [CrossRef]
- Ricken, F.J.; Nell, J.; Gruener, B.; Schmidberger, J.; Kaltenbach, T.; Kratzer, W.; Hillenbrand, A.; Henne-Bruns, D.; Deplazes, P.; Moller, P.; et al. Albendazole increases the inflammatory response and the amount of Em2-positive small particles of Echinococcus multilocularis (spems) in human hepatic alveolar echinococcosis lesions. PLoS Negl. Trop. Dis. 2017, 11, e0005636. [Google Scholar] [CrossRef]
- Tamarozzi, F.; Horton, J.; Muhtarov, M.; Ramharter, M.; Siles-Lucas, M.; Gruener, B.; Vuitton, D.A.; Bresson-Hadni, S.; Manciulli, T.; Brunetti, E. A case for adoption of continuous albendazole treatment regimen for human echinococcal infections. PLoS Negl. Trop. Dis. 2020, 14, e0008566. [Google Scholar] [CrossRef] [PubMed]
- Graeter, T.; Shi, R.; Bao, H.H.; Kratzer, W.; Barth, T.F.; Hillenbrand, A.; Henne-Bruns, D.; Schmidberger, J.; Gruener, B.; Li, W.X. Follow-up in hepatic alveolar echinococcosis under benzimidazole therapy using computed tomography. Chin. Med. J.-Peking 2020, 133, 1507–1509. [Google Scholar] [CrossRef] [PubMed]
- Graeter, T.; Ehing, F.; Oeztuerk, S.; Mason, R.A.; Haenle, M.M.; Kratzer, W.; Seufferlein, T.; Gruener, B. Hepatobiliary complications of alveolar echinococcosis: A long-term follow-up study. World J. Gastroenterol. 2015, 21, 4925–4932. [Google Scholar] [CrossRef] [PubMed]
- Gloor, S.; Candinas, D.; Beldi, G.; Lachenmayer, A. Laparoscopic resection of hepatic alveolar echinococcosis: A single-center experience. PLoS Negl. Trop. Dis. 2022, 16, e0010708. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Ran, B.; Aji, T.; Shalayiadang, P.; Jiang, T.; Shao, Y.; Wen, H. Laparoscopic hepatectomy for the treatment of hepatic alveolar echinococcosis. Parasite 2021, 28, 5. [Google Scholar] [CrossRef]
- Heinrich, S.; Tripke, V.; Huber, T.; Siegel, E.; Dennebaum, M.; Staib, L.; Wörns, M.A.; Oberholzer, K.; Mittler, J.; Lang, H. Results of multimodal treatment of hepatic echinococcosis. Der Chir. Z. Fur Alle Geb. Der Oper. Medizen 2020, 91, 943–954. [Google Scholar] [CrossRef] [PubMed]
- Joliat, G.R.; Melloul, E.; Petermann, D.; Demartines, N.; Gillet, M.; Uldry, E.; Halkic, N. Outcomes After Liver Resection for Hepatic Alveolar Echinococcosis: A Single-Center Cohort Study. World J. Surg. 2015, 39, 2529–2534. [Google Scholar] [CrossRef]
- Buttenschoen, K.; Carli Buttenschoen, D.; Gruener, B.; Kern, P.; Beger, H.G.; Henne-Bruns, D.; Reuter, S. Long-term experience on surgical treatment of alveolar echinococcosis. Langenbeck’s Arch. Surg. 2008, 394, 689–698. [Google Scholar] [CrossRef]
- Hillenbrand, A.; Gruener, B.; Kratzer, W.; Kern, P.; Graeter, T.; Barth, T.F.; Buttenschoen, K.; Henne-Bruns, D. Impact of Safe Distance on Long-Term Outcome After Surgical Therapy of Alveolar Echinococcosis. World J. Surg. 2017, 41, 1012–1018. [Google Scholar] [CrossRef]
- Kawamura, N.; Kamiyama, T.; Sato, N.; Nakanishi, K.; Yokoo, H.; Kamachi, H.; Tahara, M.; Yamaga, S.; Matsushita, M.; Todo, S. Long-Term Results of Hepatectomy for Patients with Alveolar Echinococcosis: A Single-Center Experience. J. Am. Coll. Surg. 2011, 212, 804–812. [Google Scholar] [CrossRef]
- Zhao, Z.M.; Yin, Z.Z.; Meng, Y.; Jiang, N.; Ma, Z.G.; Pan, L.C.; Tan, X.L.; Chen, X.; Liu, R. Successful robotic radical resection of hepatic echinococcosis located in posterosuperior liver segments. World J. Gastroenterol. 2020, 26, 2831–2838. [Google Scholar] [CrossRef] [PubMed]
- Golriz, M.; Flossmann, V.; Ramouz, A.; Majlesara, A.; Kulu, Y.; Stojkovic, M.; Mehrabi, A. Case Report: Successful DaVinci-Assisted Major Liver Resection for Alveolar Echinococcosis. Front. Surg. 2021, 8, 639304. [Google Scholar] [CrossRef] [PubMed]
- D’Hondt, M.; Devooght, A.; Willems, E.; Wicherts, D.; De Meyere, C.; Parmentier, I.; Provoost, A.; Pottel, H.; Verslype, C. Transition from laparoscopic to robotic liver surgery: Clinical outcomes, learning curve effect, and cost-effectiveness. J. Robot. Surg. 2022, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Melstrom, L.G.; Warner, S.G.; Woo, Y.; Sun, V.; Lee, B.; Singh, G.; Fong, Y. Selecting incision-dominant cases for robotic liver resection: Towards outpatient hepatectomy with rapid recovery. Hepatobiliary Surg. Nutr. 2017, 7, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Bijlstra, O.D.; Broersen, A.; Oosterveer, T.T.; Faber, R.A.; Achterberg, F.B.; Hurks, R.; Burgmans, M.C.; Dijkstra, J.; Mieog, J.S.; Vahrmeijer, A.L.; et al. Integration of Three-Dimensional Liver Models in a Multimodal Image-Guided Robotic Liver Surgery Cockpit. Life 2022, 12, 667. [Google Scholar] [CrossRef]
- Zhu, P.; Liao, W.; Ding, Z.Y.; Luo, H.C.; Zhang, B.H.; Zhang, W.G.; Zhang, W.; Zhang, Z.G.; Zhang, B.X.; Chen, X.P. Intraoperative ultrasonography of robot-assisted laparoscopic hepatectomy: Initial experiences from 110 consecutive cases. Surg. Endosc. 2018, 32, 4071–4077. [Google Scholar] [CrossRef]
- Strasberg, S.M.; Belghiti, J.; Clavien, P.A.; Gadzijev, E.; Garden, J.O.; Lau, W.Y.; Makuuchi, M.; Strong, R.W. The Brisbane 2000 Terminology of Liver Anatomy and Resections. Hpb 2000, 2, 333–339. [Google Scholar] [CrossRef]
- Dindo, D.; Demartines, N.; Clavien, P.A. Classification of Surgical Complications: A New Proposal With Evaluation in a Cohort of 6336 Patients and Results of a Survey. Ann. Surg. 2004, 240, 205–213. [Google Scholar] [CrossRef]
- Koch, M.; Garden, O.J.; Padbury, R.; Rahbari, N.N.; Adam, R.; Capussotti, L.; Fan, S.T.; Yokoyama, Y.; Crawford, M.; Makuuchi, M.; et al. Bile leakage after hepatobiliary and pancreatic surgery: A definition and grading of severity by the International Study Group of Liver Surgery. Surgery 2011, 149, 680–688. [Google Scholar] [CrossRef]
- Rahbari, N.N.; Garden, O.J.; Padbury, R.; Brooke-Smith, M.; Crawford, M.; Adam, R.; Koch, M.; Makuuchi, M.; Dematteo, R.P.; Christophi, C.; et al. Posthepatectomy liver failure: A definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery 2011, 149, 713–724. [Google Scholar] [CrossRef]
- Rahbari, N.N.; Garden, O.J.; Padbury, R.; Maddern, G.; Koch, M.; Hugh, T.J.; Fan, S.T.; Nimura, Y.; Figueras, J.; Vauthey, J.N.; et al. Post-hepatectomy haemorrhage: A definition and grading by the International Study Group of Liver Surgery (ISGLS). Hpb 2011, 13, 528–535. [Google Scholar] [CrossRef] [PubMed]
- Mangram, A.J.; Horan, T.C.; Pearson, M.L.; Silver, L.C.; Jarvis, W.R.; Hospital Infection Control Practices Advisory Committee. Guideline for Prevention of Surgical Site Infection, 1999. Infect. Control Hosp. Epidemiol. 1999, 20, 247–280. [Google Scholar] [CrossRef] [PubMed]
- Bassi, C.; Marchegiani, G.; Dervenis, C.; Sarr, M.; Hilal, M.A.; Adham, M.; Allen, P.; Andersson, R.; Asbun, H.J.; Besselink, M.G.; et al. The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 Years After. Surgery 2017, 161, 584–591. [Google Scholar] [CrossRef] [PubMed]


| N = 16 | |
|---|---|
| Median age in years (range) | 55.5 (23–73) |
| Median BMI in kg/m2 (range) | 25.8 (20.2–36.8) |
| Gender | |
| Female | 12 (75%) |
| Male | 4 (25%) |
| ASA * score | |
| I | 1 (6.2%) |
| II | 8 (50%) |
| III | 7 (43.8%) |
| WHO classification (PNM †) | |
| P1N0M0 | 4 (25%) |
| P2N0M0 | 5 (31.3%) |
| P2N1M0 | 2 (12.5%) |
| P3N0M0 | 4 (25%) |
| P4N0M0 | 1 (6.2%) |
| Distribution of affected liver lobes | |
| Left | 5 (31.2%) |
| Right | 5 (31.2%) |
| Both | 6 (37.5%) |
| Distribution of affected liver segments | |
| II | 1(6.2%) |
| II, III | 2 (12.5%) |
| II, III, V | 1 (6.2%) |
| II, III, V, VIII | 1 (6.2%) |
| II, V | 1 (6.2%) |
| II, III, IVa, IVb | 1 (6.2%) |
| II, III, VIII | 1 (6.2%) |
| III, IVb | 1 (6.2%) |
| IVb, V, VIII | 1 (6.2%) |
| V, VI, VII, VIII | 1 (6.2%) |
| VI | 1 (6.2%) |
| VI, VII | 2 (12.5%) |
| II, III, VI und IV | 1 (6.2%) |
| VI, VIII | 1 (6.2%) |
| Number of Lesions | |
| 1 | 10 (62.5%) |
| 2 | 4 (25.0%) |
| 3 | 1 (6.2%) |
| 5 | 1 (6.2%) |
| Median size of the lesions in cm (range) | 5.3 (2.0–12.0) |
| Median time period of preoperative albendazole therapy in years (range) | 1.78 (0.1–9.0) |
| N = 16 | |
|---|---|
| Extent of resection | |
| Major (>3 segments) | 4 (25%) |
| Minor (≤3 segments) | 12 (75%) |
| Type of resection | |
| Anatomic | 14 (87.5%) |
| Non-anatomic | 2 (12.5%) |
| Multivisceral resection * | 2 (12.5%) |
| Previous hepatic surgery | 1 (6.2%) |
| Surgical procedure | |
| Right hemihepatectomy | 2 (12.5%) |
| Extended right hemihepatectomy (ALPPS) | 1 (6.2%) |
| Left hemihepatectomy | 1 (6.2%) |
| Left lateral sectionectomy | 3 (18.8%) |
| Anatomic segmentectomy † | 7 (43.8%) |
| Atypical resection ‡ | 2 (12.5%) |
| R-status | |
| R0 | 12 (75%) |
| R1 (<1 mm) | 4 (25%) |
| Median duration of surgery in minutes (range) § | 251 (160–395) |
| Median estimated blood loss in millilitre (range) ∥ | 300 (100–1500) |
| Perioperative blood transfusions | 2 (12.5%) |
| N = 16 | |
|---|---|
| 90-day mortality | 0 (0%) |
| 90-day morbidity | |
| Clavien–Dindo I–II | 3 (18.8%) |
| Clavien–Dindo III–V | 1 (6.2%) |
| Median comprehensive complication index (range) | 0 (0–33.5) |
| Surgical site infection (SSI) | |
| Superficial incisional SSI | 0 (0%) |
| Deep incisional SSI | 0 (0%) |
| Organ/space SSI | 1 (6.2%) |
| Postoperative bile leakage | 0 (0%) |
| Posthepatectomy haemorrhage | 0 (0%) |
| Posthepatectomy liver failure | |
| Grade A | 0 (0%) |
| Grade B | 1 (6.2%) |
| Grade C | 0 (0%) |
| Pleural effusion | 1 (6.2%) |
| Postoperative pancreatic fistula | |
| Biochemical leakage (Grade A) * | 1 (6.2%) |
| Grade B | 0 (0%) |
| Grade C | 0 (0%) |
| Reintervention † | 1 (6.2%) |
| Reoperation | 0 (0%) |
| Rehospitalization ‡ | 1 (6.2%) |
| Median length of postoperative hospital stay in days (range) | 7 (4–30) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Steinkraus, K.C.; Jötten, L.; Traub, B.; Zaimi, M.; Denzinger, M.; Michalski, C.W.; Kornmann, M.; Hüttner, F.J. Robotic Liver Surgery for Alveolar Echinococcosis: A Single-Centre Experience. Pathogens 2022, 11, 1276. https://doi.org/10.3390/pathogens11111276
Steinkraus KC, Jötten L, Traub B, Zaimi M, Denzinger M, Michalski CW, Kornmann M, Hüttner FJ. Robotic Liver Surgery for Alveolar Echinococcosis: A Single-Centre Experience. Pathogens. 2022; 11(11):1276. https://doi.org/10.3390/pathogens11111276
Chicago/Turabian StyleSteinkraus, Kira C., Laila Jötten, Benno Traub, Marin Zaimi, Maximilian Denzinger, Christoph W. Michalski, Marko Kornmann, and Felix J. Hüttner. 2022. "Robotic Liver Surgery for Alveolar Echinococcosis: A Single-Centre Experience" Pathogens 11, no. 11: 1276. https://doi.org/10.3390/pathogens11111276
APA StyleSteinkraus, K. C., Jötten, L., Traub, B., Zaimi, M., Denzinger, M., Michalski, C. W., Kornmann, M., & Hüttner, F. J. (2022). Robotic Liver Surgery for Alveolar Echinococcosis: A Single-Centre Experience. Pathogens, 11(11), 1276. https://doi.org/10.3390/pathogens11111276





