First Molecular Identification of Babesia, Theileria, and Anaplasma in Goats from the Philippines
Abstract
1. Introduction
2. Results
2.1. Sample Composition and Background Information
2.2. Detection of Pathogens and Its Association with Host Parameters
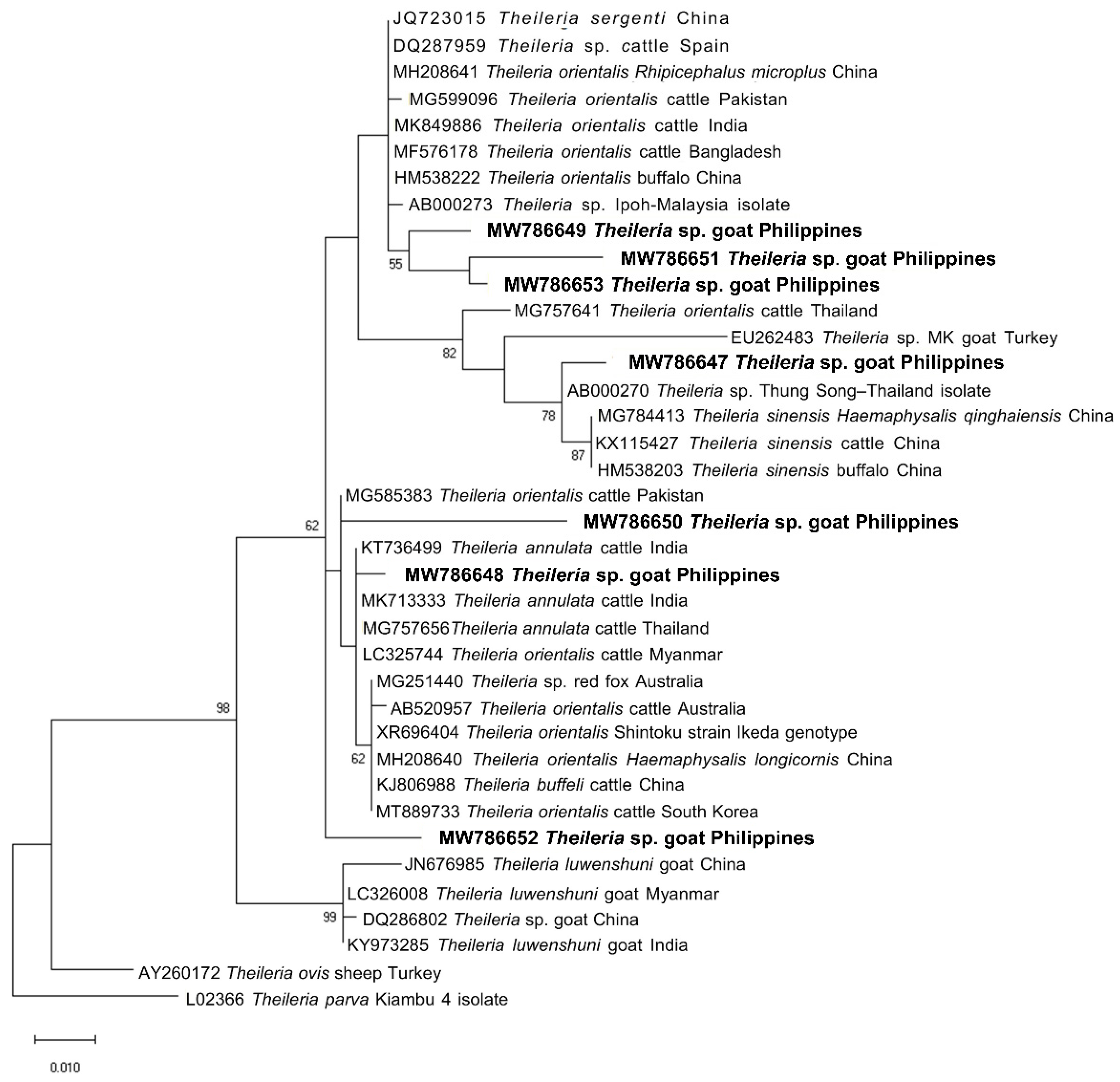
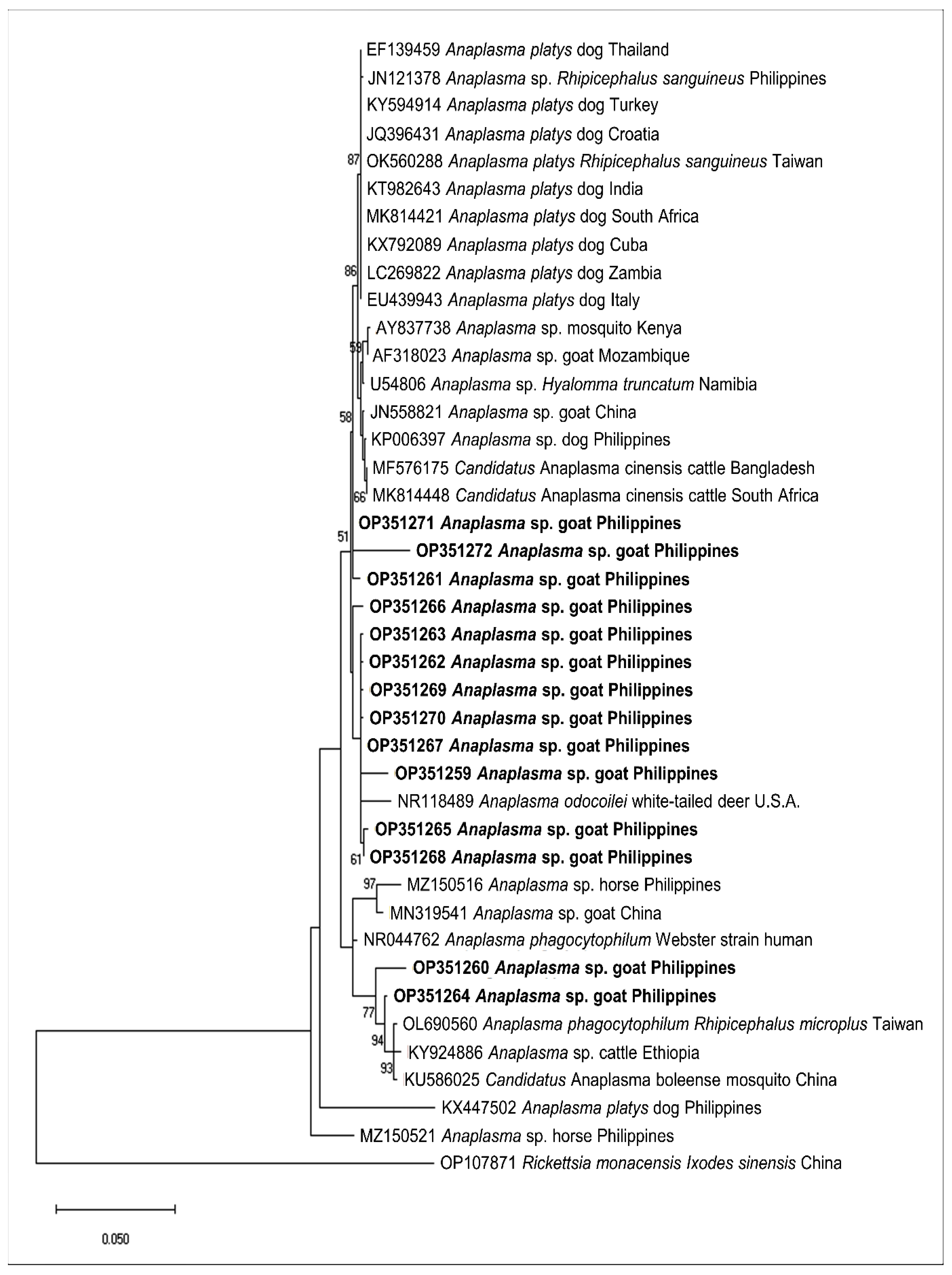
2.3. Sequencing and Phylogenetic Analysis of Representative Sequences
3. Discussion
4. Materials and Methods
4.1. Ethics Statements
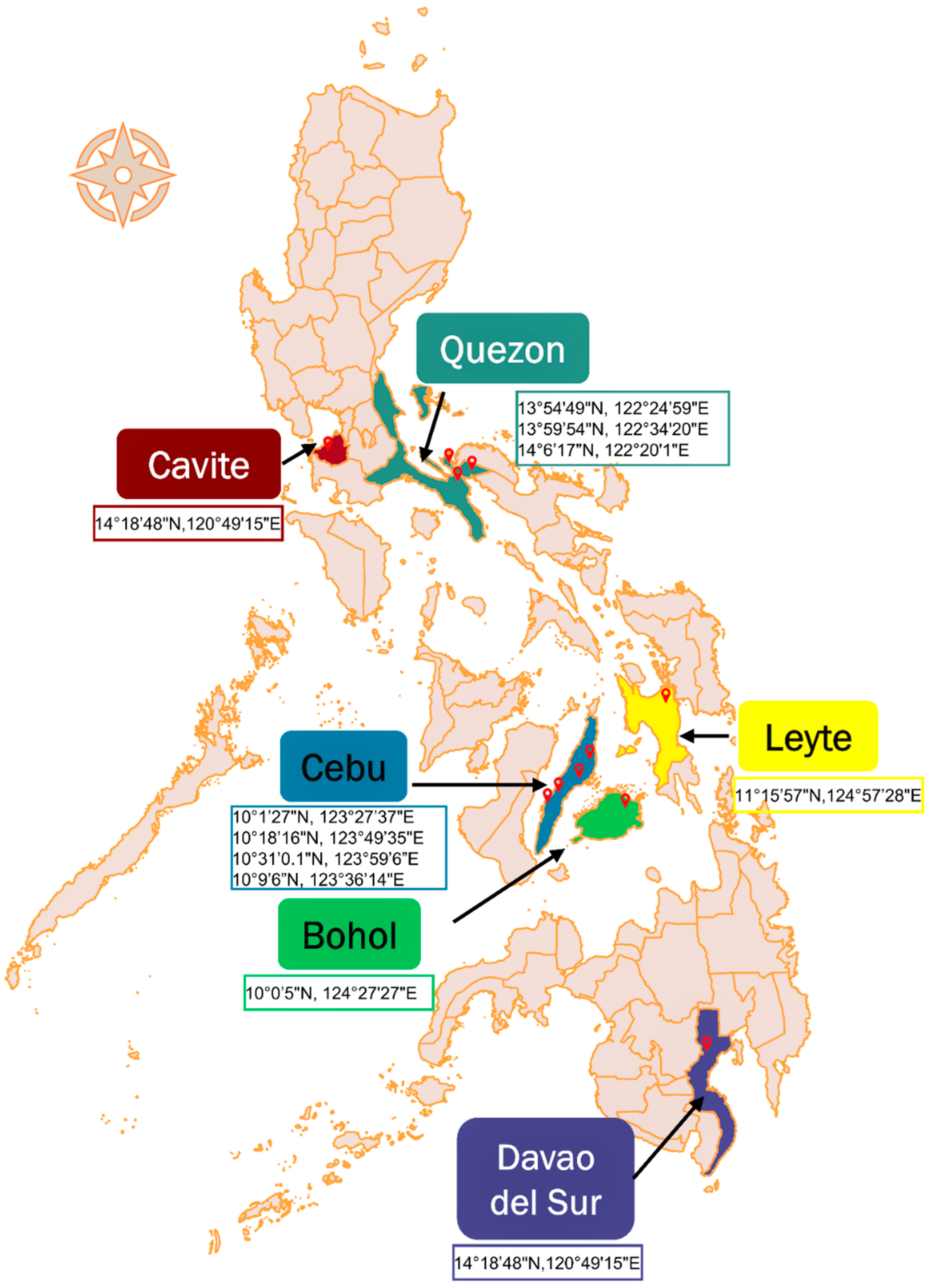
4.2. Sample Collection and Sampling Sites
4.3. Genomic DNA Isolation
4.4. PCR Assays for Pathogen Detection
4.5. Sequencing and Phylogenetic Analysis
4.6. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hurtado, O.J.B.; Giraldo-Ríos, C. Economic and health impact of the ticks in production animals. In Ticks and Tick-Borne Pathogens; Abubakar, M.K., Perera, P., Eds.; IntechOpen: London, UK, 2019; ISBN 978-1-78985-765-8. [Google Scholar]
- Bilgic, H.B.; Bakırcı, S.; Kose, O.; Unlu, A.H.; Hacılarlıoglu, S.; Eren, H.; Weir, W.; Karagenc, T. Prevalence of tick-borne haemoparasites in small ruminants in Turkey and diagnostic sensitivity of single-PCR and RLB. Parasites Vectors 2017, 10, 211. [Google Scholar] [CrossRef] [PubMed]
- Torina, A.; Caracappa, S. Tick-borne diseases in sheep and goats: Clinical and diagnostic aspects. Small Rumin. Res. 2012, 106, S6–S11. [Google Scholar] [CrossRef]
- Hashemi-Fesharki, R. Tick-borne diseases of sheep and goats and their related vectors in Iran. Parassitologia 1997, 39, 115–117. [Google Scholar]
- Smith, M.C.; Sherman, D.M. Goat Medicine; Wiley—Blackwell: Hoboken, NJ, USA, 2011; ISBN 978-1-119-94952-7. [Google Scholar]
- Hasherni-Fesharki, R.; Uilenberg, G. Babesia crassa n. sp. (Sporozoa, Babesiidae) of domestic sheep in Iran. Vet. Q. 1981, 3, 1–8. [Google Scholar] [CrossRef]
- Stuen, S. Haemoparasites—Challenging and wasting infections in small ruminants: A review. Animals 2020, 10, 2179. [Google Scholar] [CrossRef] [PubMed]
- Yeruham, I.; Hadani, A.; Galker, F. Some epizootiological and clinical aspects of ovine babesiosis caused by Babesia ovis—A review. Vet. Parasitol. 1998, 74, 153–163. [Google Scholar] [CrossRef]
- Liu, A.H.; Yin, H.; Guan, G.Q.; Schnittger, L.; Liu, Z.J.; Ma, M.L.; Dang, Z.S.; Liu, J.L.; Ren, Q.Y.; Bai, Q.; et al. At least two genetically distinct large Babesia species infective to sheep and goats in China. Vet. Parasitol. 2007, 147, 246–251. [Google Scholar] [CrossRef]
- Niu, Q.; Luo, J.; Guan, G.; Liu, Z.; Ma, M.; Liu, A.; Gao, J.; Ren, Q.; Li, Y.; Qiu, J.; et al. Differentiation of two ovine Babesia based on the ribosomal DNA internal transcribed spacer (ITS) sequences. Exp. Parasitol. 2009, 121, 64–68. [Google Scholar] [CrossRef]
- Ozubek, S.; Aktas, M. Molecular evidence of a new Babesia sp. in goats. Vet. Parasitol. 2017, 233, 1–8. [Google Scholar] [CrossRef]
- Friedhoff, K.T. Tick-borne diseases of sheep and goats caused by Babesia, Theileria or Anaplasma spp. Parassitologia 1997, 39, 99–109. [Google Scholar]
- Ahmed, J.S.; Luo, J.; Schnittger, L.; Seitzer, U.; Jongejan, F.; Yin, H. Phylogenetic position of small-ruminant infecting piroplasms. Ann. N. Y. Acad. Sci. 2006, 1081, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, J.; Yin, H.; Bakheit, M.; Liu, Z.; Mehlhorn, H.; Seitzer, U. Small ruminant theileriosis. In Progress in Parasitology; Mehlhorn, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 135–153. ISBN 978-3-642-21395-3. [Google Scholar]
- Woldehiwet, Z. The natural history of Anaplasma phagocytophilum. Vet. Parasitol. 2010, 167, 108–122. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Lu, C.; Yan, Y.; Shi, K.; Chen, Q.; Zhao, C.; Wang, R.; Zhang, L.; Jian, F.; Ning, C. The first detection of Anaplasma capra, an emerging zoonotic Anaplasma sp., in erythrocytes. Emerg. Microbes Infect. 2021, 10, 226–234. [Google Scholar] [CrossRef]
- Kocan, K.M.; de la Fuente, J.; Blouin, E.F.; Coetzee, J.F.; Ewing, S.A. The natural history of Anaplasma marginale. Vet. Parasitol. 2010, 167, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Pollott, G.; Wilson, R.T. Sheep and Goats for Diverse Products and Profits; FAO diversification booklet; FAO: Rome, Italy, 2009; ISBN 978-92-5-106137-4. [Google Scholar]
- Dar, W.; Faylon, P. Small ruminant development in the Philippines. In Sustainable Parasite Control in Small Ruminants; Watson Ferguson & Co.: Brisbane, Australia, 1996; pp. 75–81. [Google Scholar]
- The 2003 Goat Farming Committee. The Philippines Recommends for Goat Farming; Philippine Council for Agriculture, Forestry and Natural Resources Research and Development, Department of Science and Technology: Los Banos, Philippines, 2004; ISBN 978-971-20-0522-0. [Google Scholar]
- Philippine Statistics Authority. Goat Situation Report: January–December 2021; Philippine Statistics Authority: Quezon City, Philippines, 2022. Available online: https://psa.gov.ph/ (accessed on 19 July 2022).
- Terrili, C.E. Trends in Sheep and Goat Production over the Past 20 Years. In Small Ruminant Production in the Developing Countries; FAO Animal Production and Health Paper; Food and Agriculture Organization: Rome, Italy, 1986; pp. 1–11. [Google Scholar]
- Ybañez, A.P.; Arrabis, O.V.; Alvarez, D.J.M.; Galon, E.M.S.; Jayag, R.M.P.; Delan, E.S.; Ybañez, R.H.D.; Xuan, X. Evaluation on the presence of Anaplasma, Ehrlichia, and Babesia spp. in goats (Capra hircus) in Cebu, the Philippines. Vet. World 2019, 12, 774–777. [Google Scholar] [CrossRef] [PubMed]
- QGIS Development Team. QGIS Geographic Information System 2019. Open Source Geospatial Foundation Project. Available online: https://www.qgis.org (accessed on 22 July 2022).
- Iqbal, F.; Khattak, R.; Ozubek, S.; Khattak, M.; Rasul, A.; Aktas, M. Application of the reverse line blot assay for the molecular detection of Theileria and Babesia sp. in sheep and goat blood samples from Pakistan. Iran J. Parasitol. 2013, 8, 289–295. [Google Scholar] [PubMed]
- Nasreen; Khan, A.; Niaz, S.; Hassan shah, M.; Khan, A.; Ahmed, H.; Khattak, I.; Zeb, J.; Naeem, H.; Hassan, M.A.; et al. Molecular detection of small ruminant piroplasmosis and first report of Theileria luwenshuni (Apicomplexa: Theileridae) in small ruminants of Pakistan. Exp. Parasitol. 2020, 212, 107872. [Google Scholar] [CrossRef]
- Ozubek, S.; Aktas, M. Molecular and parasitological survey of ovine piroplasmosis, including the first report of Theileria annulata (Apicomplexa: Theileridae) in sheep and goats from Turkey. J. Med. Entomol. 2017, 54, 212–220. [Google Scholar] [CrossRef]
- Wang, H.; Yang, J.; Mukhtar, M.U.; Liu, Z.; Zhang, M.; Wang, X. Molecular detection and identification of tick-borne bacteria and protozoans in goats and wild siberian roe deer (Capreolus pygargus) from Heilongjiang province, northeastern China. Parasites Vectors 2019, 12, 296. [Google Scholar] [CrossRef]
- Yang, L.; Wang, J.-H.; Upadhyay, A.; Zhao, J.-G.; Huang, L.-Y.; Liao, C.-H.; Han, Q. Identification of Theileria spp. and investigation of hematological profiles of their infections in goats in Hainan island, China. Parasite 2022, 29, 13. [Google Scholar] [CrossRef]
- Gebrekidan, H.; Hailu, A.; Kassahun, A.; Rohoušová, I.; Maia, C.; Talmi-Frank, D.; Warburg, A.; Baneth, G. Theileria infection in domestic ruminants in northern Ethiopia. Vet. Parasitol. 2014, 200, 31–38. [Google Scholar] [CrossRef] [PubMed]
- M’ghirbi, Y.; Ros-García, A.; Iribar, P.; Rhaim, A.; Hurtado, A.; Bouattour, A. A molecular study of tick-borne haemoprotozoan parasites (Theileria and Babesia) in small ruminants in northern Tunisia. Vet. Parasitol. 2013, 198, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Chatanga, E.; Kainga, H.; Maganga, E.; Hayashida, K.; Katakura, K.; Sugimoto, C.; Nonaka, N.; Nakao, R. Molecular identification and genetic characterization of tick-borne pathogens in sheep and goats at two farms in the central and southern regions of Malawi. Ticks Tick-Borne Dis. 2021, 12, 101629. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Faruque, M.d.R.; Rahman, M.d.M.; Chowdhury, M.Y.E. Epidemiology and molecular detection of Anaplasma spp. in goats from Chattogram district, Bangladesh. Vet. Med. Sci. 2022, 8, 1240–1249. [Google Scholar] [CrossRef] [PubMed]
- Aung, A.; Kaewlamun, W.; Narapakdeesakul, D.; Poofery, J.; Kaewthamasorn, M. Molecular detection and characterization of tick-borne parasites in goats and ticks from Thailand. Ticks Tick-Borne Dis. 2022, 13, 101938. [Google Scholar] [CrossRef]
- Niaz, S.; Rahman, Z.U.; Ali, I.; Cossío-Bayúgar, R.; Amaro-Estrada, I.; Alanazi, A.D.; Khattak, I.; Zeb, J.; Nasreen, N.; Khan, A. Molecular prevalence, characterization and associated risk factors of Anaplasma spp. and Theileria spp. in small ruminants in northern Pakistan. Parasite 2021, 28, 3. [Google Scholar] [CrossRef]
- Miranda, E.A.; Han, S.-W.; Cho, Y.-K.; Choi, K.-S.; Chae, J.-S. Co-infection with Anaplasma species and novel genetic variants detected in cattle and goats in the Republic of Korea. Pathogens 2021, 10, 28. [Google Scholar] [CrossRef]
- Yang, X.; Fu, M.; Yu, Z.; Wang, J.; Song, J.; Zhao, G. Molecular characterization of Anaplasma spp. among dairy, cashmere, and meat goats in Shaanxi Province, northwestern China. Animals 2022, 12, 1566. [Google Scholar] [CrossRef]
- Genchi, C.; Manfredi, M.T. Tick species infesting ruminants in Italy: Ecological and bio-climatic factors affecting the different regional distribution. Parassitologia 1999, 41 (Suppl. S1), 41–45. [Google Scholar]
- Zhou, M.; Cao, S.; Sevinc, F.; Sevinc, M.; Ceylan, O.; Ekici, S.; Jirapattharasate, C.; Moumouni, P.F.A.; Liu, M.; Wang, G.; et al. Molecular detection and genetic characterization of Babesia, Theileria and Anaplasma amongst apparently healthy sheep and goats in the central region of Turkey. Ticks Tick-borne Dis. 2017, 8, 246–252. [Google Scholar] [CrossRef]
- Rjeibi, M.R.; Gharbi, M.; Mhadhbi, M.; Mabrouk, W.; Ayari, B.; Nasfi, I.; Jedidi, M.; Sassi, L.; Rekik, M.; Darghouth, M.A. Prevalence of piroplasms in small ruminants in north-west Tunisia and the first genetic characterisation of Babesia ovis in Africa. Parasite 2014, 21, 23. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.d.F.; Rudra, P.G.; Singha, S.; Das, T.; Gebrekidan, H.; Uddin, M.B.; Chowdhury, M.Y.E. Molecular epidemiology and characterization of Theileria in goats. Protist 2021, 172, 125804. [Google Scholar] [CrossRef] [PubMed]
- Said, M.B.; Belkahia, H.; Alberti, A.; Zobba, R.; Bousrih, M.; Yahiaoui, M.; Daaloul-Jedidi, M.; Mamlouk, A.; Gharbi, M.; Messadi, L. Molecular survey of Anaplasma species in small ruminants reveals the presence of novel strains closely related to A. phagocytophilum in Tunisia. Vector-Borne Zoonotic Dis. 2015, 15, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Schwalback, M.; Greyling, J.; David, M. The efficacy of a 10% aqueous neem (Azadirachta indica) seed extract for tick control in Small East African and Toggenburg female goat kids in Tanzania. S. Afr. J. Anim. Sci. 2003, 33, 83–88. [Google Scholar] [CrossRef]
- Al-Fahdi, A.; Alqamashoui, B.; Al-Hamidhi, S.; Kose, O.; Tageldin, M.H.; Bobade, P.; Johnson, E.H.; Hussain, A.-R.; Karagenc, T.; Tait, A.; et al. Molecular surveillance of Theileria parasites of livestock in Oman. Ticks Tick-borne Dis. 2017, 8, 741–748. [Google Scholar] [CrossRef]
- Prado, I.C.B.; Capuno, L.X.B.; Collera, P.D.L.P.; Cabralda, A.P.D.; De Ramos, K.A.S.; Bernardo, J.M.G.; Divina, B.P.; Masatani, T.; Tanaka, T.; Galay, R.L. Molecular detection and characterization of Babesia and Theileria in cattle and water buffaloes from southern Luzon, Philippines. Microorganisms 2022, 10, 678. [Google Scholar] [CrossRef]
- Ochirkhuu, N.; Konnai, S.; Mingala, C.N.; Okagawa, T.; Villanueva, M.; Pilapil, F.M.I.R.; Murata, S.; Ohashi, K. Molecular Epidemiological survey and genetic analysis of vector-borne infections of cattle in Luzon island, the Philippines. Vet. Parasitol. 2015, 212, 161–167. [Google Scholar] [CrossRef]
- Belotindos, L.; Lazaro, J.; Villanueva, M.; Mingala, C. Molecular detection and characterization of Theileria species in the Philippines. Acta Parasitol. 2014, 59, 448–453. [Google Scholar] [CrossRef]
- Brown, C.G.D.; Ilhan, T.; Kirvar, E.; Thomas, M.; Wilkie, G.; Leemans, I.; Hooshmand-Rad, P. Theileria lestoquardi and T. annulata in cattle, sheep, and goats: In vitro and in vivo Studies. Ann. N. Y. Acad. Sci. 1998, 849, 44–51. [Google Scholar] [CrossRef]
- Lawrence, K.E.; Gedye, K.; Hickson, R.; Wang, B.; Carvalho, L.; Zhao, Y.; Pomroy, W.E. The role of sheep (Ovis aries) in maintaining Theileria orientalis Ikeda type infection. Vet. Parasitol. 2021, 291, 109391. [Google Scholar] [CrossRef]
- Khukhuu, A.; Lan, D.T.B.; Long, P.T.; Ueno, A.; Li, Y.; Luo, Y.; de Macedo, A.C.C.; Matsumoto, K.; Inokuma, H.; Kawazu, S.-I.; et al. Molecular epidemiological survey of Theileria orientalis in Thua Thien Hue province, Vietnam. J. Vet. Med. Sci. 2011, 73, 701–705. [Google Scholar] [CrossRef] [PubMed]
- Chansiri, K.; Sarataphan, N. Molecular phylogenetic study of Theileria sp. (Thung Song) based on the thymidylate synthetase gene. Parasitol. Res. 2002, 88, S33–S35. [Google Scholar] [CrossRef] [PubMed]
- Chansiri, K.; Kawazu, S.; Kamio, T.; Terada, Y.; Fujisaki, K.; Philippe, H.; Sarataphan, N. Molecular phylogenetic studies on Theileria parasites based on small subunit ribosomal RNA gene sequences. Vet. Parasitol. 1999, 83, 99–105. [Google Scholar] [CrossRef]
- Kaewhom, P.; Thitasarn, W. The prevalence of Theileria spp. of goat in Watthana Nakhon District, Sa Kaeo province. J. Mahanakorn Vet. Med. 2017, 12, 57–66. [Google Scholar]
- Tu, H.L.C.; Nugraheni, Y.R.; Tiawsirisup, S.; Saiwichai, T.; Thiptara, A.; Kaewthamasorn, M. Development of a novel multiplex PCR assay for the detection and differentiation of Plasmodium caprae from Theileria luwenshuni and Babesia spp. in goats. Acta Trop. 2021, 220, 105957. [Google Scholar] [CrossRef] [PubMed]
- Udonsom, R.; Mahittikorn, A.; Jirapattharasate, C. Molecular detection and genetic diversity of tick-borne pathogens in goats from the southern part of Thailand. Pathogens 2022, 11, 477. [Google Scholar] [CrossRef]
- Bawm, S.; Kakisaka, K.; Thu, M.J.; Chel, H.M.; Oo, Y.M.N.; Soe, N.C.; Win, S.Y.; Htun, L.L.; Win, M.M.; Suzuki, H.; et al. First molecular detection of Theileria luwenshuni from goats in Myanmar. Parasitol. Res. 2018, 117, 3361–3364. [Google Scholar] [CrossRef]
- Sivakumar, T.; Lan, D.T.B.; Long, P.T.; Yoshinari, T.; Tattiyapong, M.; Guswanto, A.; Okubo, K.; Igarashi, I.; Inoue, N.; Xuan, X.; et al. PCR detection and genetic diversity of bovine hemoprotozoan parasites in Vietnam. J. Vet. Med. Sci. 2013, 75, 1455–1462. [Google Scholar] [CrossRef]
- Sivakumar, T.; Tuvshintulga, B.; Kothalawala, H.; Silva, S.S.P.; Lan, D.T.B.; Long, P.T.; Ybañez, A.P.; Ybañez, R.H.D.; Benitez, D.F.; Tayebwa, D.S.; et al. Host range and geographical distribution of Babesia sp. Mymensingh. Transbound. Emerg. Dis. 2020, 67, 2233–2239. [Google Scholar] [CrossRef]
- Tate, C.M.; Howerth, E.W.; Mead, D.G.; Dugan, V.G.; Luttrell, M.P.; Sahora, A.I.; Munderloh, U.G.; Davidson, W.R.; Yabsley, M.J. Anaplasma odocoilei sp. nov. (Family Anaplasmataceae) from white-tailed deer (Odocoileus virginianus). Ticks Tick-Borne Dis. 2013, 4, 110–119. [Google Scholar] [CrossRef]
- Yabsley, M.J.; Davidson, W.R.; Stallknecht, D.E.; Varela, A.S.; Swift, P.K.; Devos, J.C.; Dubay, S.A. Evidence of tick-borne organisms in mule deer (Odocoileus hemionus) from the western United States. Vector-Borne Zoonotic Dis. 2005, 5, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Ojeda-Chi, M.M.; Rodriguez-Vivas, R.I.; Esteve-Gasent, M.D.; Pérez de León, A.; Modarelli, J.J.; Villegas-Perez, S. Molecular detection of rickettsial tick-borne agents in white-tailed deer (Odocoileus virginianus yucatanensis), Mazama deer (Mazama temama), and the ticks they host in Yucatan, Mexico. Ticks Tick-Borne Dis. 2019, 10, 365–370. [Google Scholar] [CrossRef]
- Orozco, M.M.; Argibay, H.D.; Minatel, L.; Guillemi, E.C.; Berra, Y.; Schapira, A.; Di Nucci, D.; Marcos, A.; Lois, F.; Falzone, M.; et al. A participatory surveillance of marsh deer (Blastocerus dichotomus) morbidity and mortality in Argentina: First results. BMC Vet. Res. 2020, 16, 321. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Li, J.; Wang, Y.-W.; Jiang, B.-G.; Liu, H.-B.; Wei, R.; Jiang, R.-R.; Cui, X.-M.; Li, L.-F.; Yuan, T.-T.; et al. Anaplasma platys-like infection in goats, Beijing, China. Vector-Borne Zoonotic Dis. 2020, 20, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Galon, E.M.; Macalanda, A.M.; Garcia, M.M.; Ibasco, C.J.; Garvida, A.; Ji, S.; Zafar, I.; Hasegawa, Y.; Liu, M.; Ybañez, R.H.; et al. Molecular identification of selected tick-borne protozoan and bacterial pathogens in thoroughbred racehorses in Cavite, Philippines. Pathogens 2021, 10, 1318. [Google Scholar] [CrossRef]
- Sivakumar, T.; Hayashida, K.; Sugimoto, C.; Yokoyama, N. Evolution and genetic diversity of Theileria. Infect. Genet. Evol. 2014, 27, 250–263. [Google Scholar] [CrossRef]
- Swann, P.H.; Claveria, F. Rhipicephalus (Boophilus) microplus ticks (Family Ixodidae) in goats raised in a small private farm in San Jose del Monte, Bulacan, central Luzon, Philippines. Philipp. J. Sci. 2017, 146, 493–496. [Google Scholar]
- Hirunkanokpun, S.; Ahantarig, A.; Baimai, V.; Pramual, P.; Rakthong, P.; Trinachartvanit, W. Spotted fever group Rickettsia, Anaplasma and Coxiella-like endosymbiont in Haemaphysalis ticks from mammals in Thailand. Vet. Res. Commun. 2022. [Google Scholar] [CrossRef]
- Lim, F.S.; Khoo, J.J.; Akhavanrezeai, M.; Loong, S.K.; Khor, C.S.; AbuBakar, S. Detection of Theileria luwenshuni (Piroplasmida: Theileriidae) from ticks infesting goats in Peninsular Malaysia. Syst. Appl. Acarol. 2019, 24, 1971–1974. [Google Scholar] [CrossRef]
- Ybañez, A.P.; Perez, Z.O.; Gabotero, S.R.; Yandug, R.T.; Kotaro, M.; Inokuma, H. First molecular detection of Ehrlichia canis and Anaplasma platys in ticks from dogs in Cebu, Philippines. Ticks Tick-Borne Dis. 2012, 3, 288–293. [Google Scholar] [CrossRef]
- Galay, R.L.; Manalo, A.A.L.; Dolores, S.L.D.; Aguilar, I.P.M.; Sandalo, K.A.C.; Cruz, K.B.; Divina, B.P.; Andoh, M.; Masatani, T.; Tanaka, T. Molecular detection of tick-borne pathogens in canine population and Rhipicephalus sanguineus (sensu lato) ticks from southern Metro Manila and Laguna, Philippines. Parasites Vectors 2018, 11, 643. [Google Scholar] [CrossRef] [PubMed]
- Dantas-Torres, F. Biology and ecology of the brown dog tick, Rhipicephalus sanguineus. Parasites Vectors 2010, 3, 26. [Google Scholar] [CrossRef]
- Gubbels, J.M.; de Vos, A.P.; van der Weide, M.; Viseras, J.; Schouls, L.M.; de Vries, E.; Jongejan, F. Simultaneous detection of bovine Theileria and Babesia species by reverse line blot hybridization. J. Clin. Microbiol. 1999, 37, 1782–1789. [Google Scholar] [CrossRef] [PubMed]
- Georges, K.; Loria, G.R.; Riili, S.; Greco, A.; Caracappa, S.; Jongejan, F.; Sparagano, O. Detection of haemoparasites in cattle by reverse line blot hybridisation with a note on the distribution of ticks in Sicily. Vet. Parasitol. 2001, 99, 273–286. [Google Scholar] [CrossRef]
- Barlough, J.E.; Madigan, J.E.; DeRock, E.; Bigornia, L. Nested polymerase chain reaction for detection of Ehrlichia equi genomic DNA in horses and ticks (Ixodes pacificus). Vet. Parasitol. 1996, 63, 319–329. [Google Scholar] [CrossRef]
- Aktaş, M.; Altay, K.; Dumanlı, N. Development of a polymerase chain reaction method for diagnosis of Babesia ovis infection in sheep and goats. Vet. Parasitol. 2005, 133, 277–281. [Google Scholar] [CrossRef]
- Tumwebaze, M.A.; Byamukama, B.; Tayebwa, D.S.; Byaruhanga, J.; Angwe, M.K.; Galon, E.M.; Liu, M.; Lee, S.-H.; Ringo, A.E.; Adjou Moumouni, P.F.; et al. First molecular detection of Babesia ovis, Theileria spp., Anaplasma spp., and Ehrlichia ruminantium in goats from western Uganda. Pathogens 2020, 9, 895. [Google Scholar] [CrossRef]
- Madeira, F.; Park, Y.M.; Lee, J.; Buso, N.; Gur, T.; Madhusoodanan, N.; Basutkar, P.; Tivey, A.R.N.; Potter, S.C.; Finn, R.D.; et al. The EMBL-EBI search and sequence analysis tools APIs in 2019. Nucleic Acids Res. 2019, 47, W636–W641. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]

| Variable | N | Babesia/Theileria spp. | Anaplasma spp. | ||
|---|---|---|---|---|---|
| No. of Positives (%) | p Value | No. of Positives (%) | p Value | ||
| Age-group | |||||
| Young (<1 year) | 159 | 114 (71.70) | 0.051 | 77 (48.43) | 0.001 ** |
| Adult (≥1 year) | 237 | 191 (80.59) | 76 (32.07) | ||
| Sex | |||||
| Male | 52 | 32 (61.54) | 0.007 ** | 25 (48.08) | 0.169 |
| Female | 344 | 273 (79.36) | 128 (37.21) | ||
| Location | |||||
| Davao del Sur | 199 | 183 (91.96) | <0.001 *** | 82 (41.21) | <0.001 *** |
| Cebu | 74 | 35 (47.30) | 37 (50.00) | ||
| Bohol | 35 | 16 (45.71) | 10 (28.57) | ||
| Quezon | 20 | 8 (40.00) | 16 (80.00) | ||
| Leyte | 26 | 24 (92.31) | 2 (7.69) | ||
| Cavite | 42 | 39 (92.86 | 6 (14.29) | ||
| Breed # | |||||
| Purebred (Anglo-Nubian or Boer) | 222 | 179 (80.63) | 0.027 * | 101 (45.50) | 0.026 * |
| Crossbred or upgrades | 87 | 58 (66.67) | 25 (28.74) | ||
| Philippine native | 61 | 44 (72.13) | 25 (40.98) | ||
| Total | 396 | 305 (77.02) | 153 (38.64) | ||
| Pathogen | Target Gene | Primer Sequence | Annealing Temperature (°C) | Target Length (bp) | Reference |
|---|---|---|---|---|---|
| Piroplasma (Babesia/Theileria) | 18S rRNA (V4 hypervariable region) | F1: 5’—GAGGTAGTGACAAGAAATAACAATA—3’ | 50 | ~460–520 | [72] |
| R1: 5’—TCTTCGATCCCCTAACTTTC—3’ | |||||
| F2: 5’—GACACAGGGAGGTAGTGACAAG—3’ | 60 | ~390–420 | [73] | ||
| R2: 5’—CTAAGAATTTCACCTCTGACAGT—3’ | |||||
| Babesia ovis | Small subunit 18S rRNA | F: 5’—TGGGCAGGACCTTGGTTCTTCT—3’ | 62 | ~549 | [75] |
| R: 5’—CCGCGTAGCGCCGGCTAAATA—3’ | |||||
| Anaplasma spp. (A. phagocytophilum) | 16S rRNA | F1: 5’—TCCTGGCTCAGAACGAACGCTGGCGGC—3’ | 50 | ~1433 | [74] |
| R1: 5’—AGTCACTGACCCAACCTTAAATGGCTG—3’ | |||||
| F2: 5’—GTCGAACGGATTATTCTTTATAGCTTGC—3’ | 50 | ~925 | |||
| R2: 5’—CCCTTCCGTTAAGAAGGATCTAATCTCC—3’ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galon, E.M.; Ybañez, R.H.; Macalanda, A.M.; Estabillo, G.R.; Montano, M.T.R.; Veedor, M.D.; Garvida, A.; Fabon, R.J.; Callanta, M.R.; Labutong, K.J.; et al. First Molecular Identification of Babesia, Theileria, and Anaplasma in Goats from the Philippines. Pathogens 2022, 11, 1109. https://doi.org/10.3390/pathogens11101109
Galon EM, Ybañez RH, Macalanda AM, Estabillo GR, Montano MTR, Veedor MD, Garvida A, Fabon RJ, Callanta MR, Labutong KJ, et al. First Molecular Identification of Babesia, Theileria, and Anaplasma in Goats from the Philippines. Pathogens. 2022; 11(10):1109. https://doi.org/10.3390/pathogens11101109
Chicago/Turabian StyleGalon, Eloiza May, Rochelle Haidee Ybañez, Adrian Miki Macalanda, Giemelene Rose Estabillo, Margaret Therese Rose Montano, Marielle Danise Veedor, Anatolio Garvida, Ralph Joselle Fabon, Mary Ruth Callanta, Kim Joseph Labutong, and et al. 2022. "First Molecular Identification of Babesia, Theileria, and Anaplasma in Goats from the Philippines" Pathogens 11, no. 10: 1109. https://doi.org/10.3390/pathogens11101109
APA StyleGalon, E. M., Ybañez, R. H., Macalanda, A. M., Estabillo, G. R., Montano, M. T. R., Veedor, M. D., Garvida, A., Fabon, R. J., Callanta, M. R., Labutong, K. J., Tumwebaze, M. A., Byamukama, B., Ji, S., Zafar, I., Ybañez, A., & Xuan, X. (2022). First Molecular Identification of Babesia, Theileria, and Anaplasma in Goats from the Philippines. Pathogens, 11(10), 1109. https://doi.org/10.3390/pathogens11101109








