MRSA in Humans, Pets and Livestock in Portugal: Where We Came from and Where We Are Going
Abstract
1. Introduction
2. MRSA Clones
2.1. Human-Associated MRSA
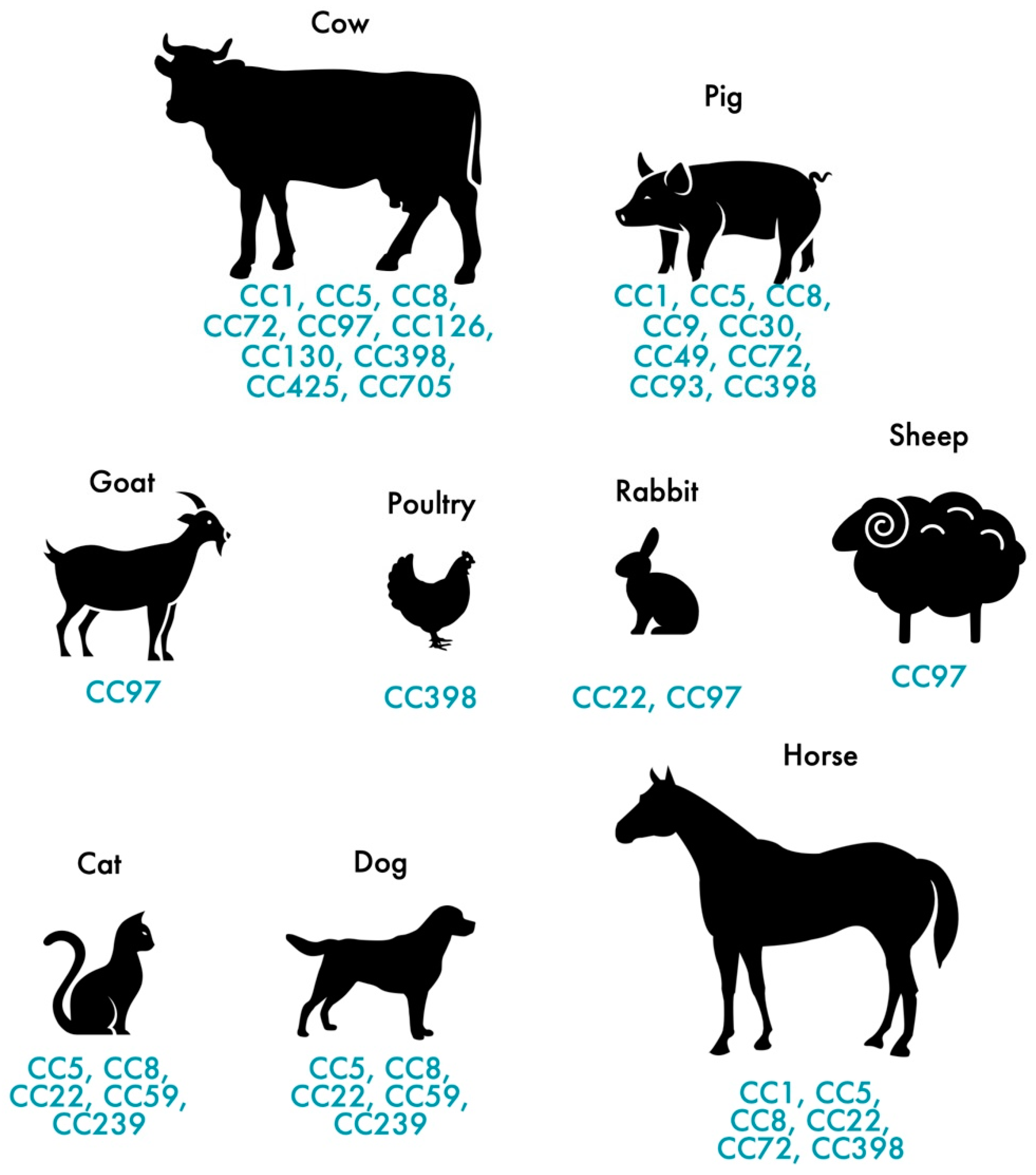
2.2. Animal-Associated MRSA
3. MRSA in Portugal
3.1. MRSA in Humans
3.2. MRSA in Pets and Livestock
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- González-García, S.; Hamdan-Partida, A.; Bustos-Hamdan, A.; Bustos-Martínez, J. Factors of Nasopharynx that Favor the Colonization and Persistence of Staphylococcus aureus. In Pharynx-Diagnosis and Treatment; IntechOpen: London, UK, 2021; ISBN 1789856094. [Google Scholar]
- European Centre for Disease Prevention and Control. Antimicrobial Resistance Surveillance in Europe 2022–2020 Data; WHO Regional Office for Europe: Copenhagen, Swenden, 2022. [Google Scholar]
- Sankar, N.; Haja Abdul Nazeer, M.J.; Moinuddin, K. Spectrum of surgical site infections with special reference to methicillin resistant Staphylococcus aureus (MRSA). Int. J. Surg. 2020, 4, 306–309. [Google Scholar]
- European Centre for Disease Prevention and Control. Healthcare-associated infections acquired in intensive care units. In ECDC: Annual Epidemiological Report for 2017; ECDC: Stockholm, Sweden, 2019. [Google Scholar]
- Saïd-Salim, B.; Mathema, B.; Kreiswirth, B.N. Community-acquired methicillin-resistant Staphylococcus aureus: An emerging pathogen. Infect. Control Hosp. Epidemiol. 2003, 24, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Otto, M. MRSA virulence and spread. Cell. Microbiol. 2012, 14, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xu, Z.; Yang, Z.; Sun, J.; Ma, L. Characterization of community-associated Staphylococcus aureus from skin and soft-tissue infections: A multicenter study in China. Emerg. Microbes Infect. 2016, 5, 1–11. [Google Scholar]
- Lee, A.S.; De Lencastre, H.; Garau, J.; Kluytmans, J.; Malhotra-kumar, S.; Peschel, A.; Harbarth, S. Methicillin-resistant Staphylococcus aureus. Nat. Publ. Gr. 2018, 4, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.A.; Sharma-Kuinkel, B.K.; Maskarinec, S.A.; Eichenberger, E.M.; Shah, P.P.; Carugati, M.; Holland, T.L.; Fowler, V.G. Methicillin-resistant Staphylococcus aureus: An overview of basic and clinical research. Nat. Rev. Microbiol. 2019, 17, 203–218. [Google Scholar] [CrossRef]
- Copin, R.; Sause, W.E.; Fulmer, Y.; Balasubramanian, D.; Dyzenhaus, S.; Ahmed, J.M.; Kumar, K.; Lees, J.; Stachel, A.; Fisher, J.C. Sequential evolution of virulence and resistance during clonal spread of community-acquired methicillin-resistant Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2019, 116, 1745–1754. [Google Scholar] [CrossRef]
- Aklilu, E.; Chia, H.Y. First mecC and mecA Positive Livestock-Associated Methicillin Resistant Staphylococcus aureus (mecC MRSA/LA-MRSA) from Dairy Cattle in Malaysia. Microorganisms 2020, 8, 147. [Google Scholar] [CrossRef]
- Sieber, R.N.; Larsen, A.R.; Urth, T.R.; Iversen, S.; Møller, C.H.; Skov, R.L.; Larsen, J.; Stegger, M. Genome investigations show host adaptation and transmission of LA-MRSA CC398 from pigs into Danish healthcare institutions. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Bernier-Lachance, J.; Arsenault, J.; Usongo, V.; Parent, É.; Labrie, J.; Jacques, M.; Malouin, F.; Archambault, M. Prevalence and characteristics of Livestock-Associated Methicillin-Resistant Staphylococcus aureus (LA-MRSA) isolated from chicken meat in the province of Quebec, Canada. PLoS ONE 2020, 15, e0227183. [Google Scholar] [CrossRef]
- Stone, M.J.; Swales, C.; Bond, S.E.; Muthayya, P.; Sarma, J.B. An outbreak of livestock-associated meticillin-resistant Staphylococcus aureus (LA-MRSA) clonal complex 398 in a regional burns centre. J. Hosp. Infect. 2022, 122, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.E.; Stegger, M.; Pedersen, K.; Sieber, R.N.; Larsen, J.; Larsen, G.; Lilje, B.; Chriél, M.; Andersen, P.S.; Larsen, A.R. Spread of LA-MRSA CC398 in Danish mink (Neovison vison) and mink farm workers. Vet. Microbiol. 2020, 245, 108705. [Google Scholar] [CrossRef] [PubMed]
- Crespo-Piazuelo, D.; Lawlor, P.G. Livestock-associated methicillin-resistant Staphylococcus aureus (LA-MRSA) prevalence in humans in close contact with animals and measures to reduce on-farm colonisation. Ir. Vet. J. 2021, 74, 21. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-T.; Chen, E.-Z.; Yang, L.; Peng, C.; Wang, Q.; Xu, Z.; Chen, D.-Q. Emerging resistance mechanisms for 4 types of common anti-MRSA antibiotics in Staphylococcus aureus: A comprehensive review. Microb. Pathog. 2021, 156, 104915. [Google Scholar] [CrossRef] [PubMed]
- Basco, M.D.S.; Kothari, A.; McKinzie, P.B.; Revollo, J.R.; Agnihothram, S.; Azevedo, M.P.; Saccente, M.; Hart, M.E. Reduced vancomycin susceptibility and increased macrophage survival in Staphylococcus aureus strains sequentially isolated from a bacteraemic patient during a short course of antibiotic therapy. J. Med. Microbiol. 2019, 68, 848–859. [Google Scholar] [CrossRef]
- Lakhundi, S.; Zhang, K. Methicillin-Resistant Staphylococcus aureus: Molecular Characterization, Evolution, and Epidemiology. Clin. Microbiol. Rev. 2018, 31, e00020-18. [Google Scholar] [CrossRef]
- Chambers, H.F.; Deleo, F.R. Waves of resistance: Staphylococcus aureus in the antibiotic era. Nat. Rev. Microbiol. 2009, 7, 629–641. [Google Scholar] [CrossRef]
- Harmsen, D.; Claus, H.; Witte, W.; Rothgänger, J.; Claus, H.; Turnwald, D.; Vogel, U. Typing of Methicillin-Resistant Staphylococcus aureus in a University Hospital Setting by Using Novel Software for spa Repeat Determination and Database Management. J. Clin. Microbiol. 2003, 41, 5442–5448. [Google Scholar] [CrossRef]
- Uhlemann, A.; McAdam, P.R.; Sullivan, S.B.; Knox, J.R.; Khiabanian, H.; Rabadan, R.; Davies, P.R.; Fitzgerald, J.R.; Lowy, F.D. Evolutionary Dynamics of Pandemic Methicillin-Sensitive Staphylococcus aureus ST398 and Its International Spread via Routes of Human Migration. MBio 2022, 8, e01375-16. [Google Scholar] [CrossRef]
- Carrel, M.; Perencevich, E.N.; David, M.Z. USA300 Methicillin-Resistant Staphylococcus aureus, United States, 2000–2013. Emerg. Infect. Dis. 2015, 21, 1973–1980. [Google Scholar] [CrossRef]
- Dai, Y.; Liu, J.; Guo, W.; Meng, H.; Huang, Q.; He, L.; Gao, Q.; Lv, H.; Liu, Y.; Wang, Y.; et al. Decreasing methicillin-resistant Staphylococcus aureus (MRSA) infections is attributable to the disappearance of predominant MRSA ST239 clones, Shanghai, 2008–2017. Emerg. Microbes Infect. 2019, 8, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Rolain, J.-M.; Abat, C.; Brouqui, P.; Raoult, D. Worldwide decrease in methicillin-resistant Staphylococcus aureus: Do we understand something? Clin. Microbiol. Infect. 2015, 21, 515–517. [Google Scholar] [CrossRef] [PubMed]
- Monecke, S.; Coombs, G.; Shore, A.C.; Coleman, D.C.; Akpaka, P.; Borg, M.; Chow, H.; Ip, M.; Jatzwauk, L.; Jonas, D.; et al. A Field Guide to Pandemic, Epidemic and Sporadic Clones of Methicillin-Resistant Staphylococcus aureus. PLoS ONE 2011, 6, e17936. [Google Scholar] [CrossRef]
- Stefani, S.; Chung, D.R.; Lindsay, J.A.; Friedrich, A.W.; Kearns, A.M.; Westh, H.; MacKenzie, F.M. Meticillin-resistant Staphylococcus aureus (MRSA): Global epidemiology and harmonisation of typing methods. Int. J. Antimicrob. Agents 2012, 39, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Bal, A.M.; Coombs, G.W.; Holden, M.T.G.; Lindsay, J.A.; Nimmo, G.R.; Tattevin, P.; Skov, R.L. Genomic insights into the emergence and spread of international clones of healthcare-, community- and livestock-associated meticillin-resistant Staphylococcus aureus: Blurring of the traditional definitions. J. Glob. Antimicrob. Resist. 2016, 6, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; Almeida, F.; Carvalho, J.A.; Castro, A.P.; Ferreira, E.; Manageiro, V.; Tejedor-Junco, M.T.; Caniça, M.; Igrejas, G.; Poeta, P. Emergence of community-acquired methicillin-resistant Staphylococcus aureus EMRSA-15 clone as the predominant cause of diabetic foot ulcer infections in Portugal. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 179–186. [Google Scholar] [CrossRef]
- Silva, V.; Hermenegildo, S.; Ferreira, C.; Manaia, C.M.; Capita, R.; Alonso-Calleja, C.; Carvalho, I.; Pereira, J.E.; Maltez, L.; Capelo, J.L. Genetic Characterization of Methicillin-Resistant Staphylococcus aureus Isolates from Human Bloodstream Infections: Detection of MLSB Resistance. Antibiotics 2020, 9, 375. [Google Scholar] [CrossRef]
- Oliveira, D.; Santos-Sanches, I.; Mato, R.; Tamayo, M.; Ribeiro, G.; Costa, D.; De Lencastre, H. Virtually all methicillin-resistant Staphylococcus aureus (MRSA) infections in the largest Portuguese teaching hospital are caused by two internationally spread multiresistant strains: The ‘Iberian’and the ‘Brazilian’clones of MRSA. Clin. Microbiol. Infect. 1998, 4, 373–384. [Google Scholar] [CrossRef]
- De Sousa, M.A.; de Lencastre, H. Bridges from hospitals to the laboratory: Genetic portraits of methicillin-resistant Staphylococcus aureus clones. FEMS Immunol. Med. Microbiol. 2004, 40, 101–111. [Google Scholar] [CrossRef]
- Pardos de la Gandara, M.; Curry, M.; Berger, J.; Burstein, D.; Della-Latta, P.; Kopetz, V.; Quale, J.; Spitzer, E.; Tan, R.; Urban, C.; et al. MRSA Causing Infections in Hospitals in Greater Metropolitan New York: Major Shift in the Dominant Clonal Type between 1996 and 2014. PLoS ONE 2016, 11, e0156924. [Google Scholar] [CrossRef]
- Moore, P.C.L.; Lindsay, J.A. Molecular characterisation of the dominant UK methicillin-resistant Staphylococcus aureus strains, EMRSA-15 and EMRSA-16. J. Med. Microbiol. 2002, 51, 516–521. [Google Scholar] [CrossRef] [PubMed]
- Van, T.T.H.; Yidana, Z.; Smooker, P.M.; Coloe, P.J. Antibiotic use in food animals worldwide, with a focus on Africa: Pluses and minuses. J. Glob. Antimicrob. Resist. 2020, 20, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Köck, R.; Ballhausen, B.; Bischoff, M.; Cuny, C.; Eckmanns, T.; Fetsch, A.; Harmsen, D.; Goerge, T.; Oberheitmann, B.; Schwarz, S.; et al. The impact of zoonotic MRSA colonization and infection in Germany. Berl. Munch. Tierarztl. Wochenschr. 2014, 127, 384–398. [Google Scholar] [PubMed]
- Silva, V.; Vieira-Pinto, M.; Saraiva, C.; Manageiro, V.; Reis, L.; Ferreira, E.; Caniça, M.; Capelo, J.L.; Igrejas, G.; Poeta, P. Prevalence and Characteristics of Multidrug-Resistant Livestock-Associated Methicillin-Resistant Staphylococcus aureus (LA-MRSA) CC398 Isolated from Quails (Coturnix Coturnix Japonica) Slaughtered for Human Consumption. Animals 2021, 11, 2038. [Google Scholar] [CrossRef] [PubMed]
- Armand-Lefevre, L.; Ruimy, R.; Andremont, A. Clonal comparison of Staphylococcus aureus isolates from healthy pig farmers, human controls, and pigs. Emerg. Infect. Dis. 2005, 11, 711. [Google Scholar] [CrossRef] [PubMed]
- Voss, A.; Loeffen, F.; Bakker, J.; Klaassen, C.; Wulf, M. Methicillin-resistant Staphylococcus aureus in Pig Farming. Emerg. Infect. Dis. 2005, 11, 1965–1966. [Google Scholar] [CrossRef]
- Hansen, J.E.; Ronco, T.; Stegger, M.; Sieber, R.N.; Fertner, M.E.; Martin, H.L.; Farre, M.; Toft, N.; Larsen, A.R.; Pedersen, K. LA-MRSA CC398 in dairy cattle and veal calf farms indicates spillover from pig production. Front. Microbiol. 2019, 10, 2733. [Google Scholar] [CrossRef]
- Schmitt, S.; Stephan, R.; Huebschke, E.; Schaefle, D.; Merz, A.; Johler, S. DNA microarray-based characterization and antimicrobial resistance phenotypes of clinical MRSA strains from animal hosts. J. Vet. Sci. 2020, 21, e54. [Google Scholar] [CrossRef]
- Chen, C.-J.; Lauderdale, T.-L.Y.; Lu, C.-T.; Chuang, Y.-Y.; Yang, C.-C.; Wu, T.-S.; Lee, C.-Y.; Lu, M.-C.; Ko, W.-C.; Huang, Y.-C. Clinical and molecular features of MDR livestock-associated MRSA ST9 with staphylococcal cassette chromosome mec XII in humans. J. Antimicrob. Chemother. 2018, 73, 33–40. [Google Scholar] [CrossRef]
- McCarthy, A.J.; Lindsay, J.A.; Loeffler, A. Are all meticillin-resistant Staphylococcus aureus (MRSA) equal in all hosts? Epidemiological and genetic comparison between animal and human MRSA. Vet. Dermatol. 2012, 23, 267-e54. [Google Scholar]
- Shore, A.C.; Deasy, E.C.; Slickers, P.; Brennan, G.; O’Connell, B.; Monecke, S.; Ehricht, R.; Coleman, D.C. Detection of staphylococcal cassette chromosome mec type XI carrying highly divergent mecA, mecI, mecR1, blaZ, and ccr genes in human clinical isolates of clonal complex 130 methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2011, 55, 3765–3773. [Google Scholar] [CrossRef] [PubMed]
- Kerschner, H.; Harrison, E.M.; Hartl, R.; Holmes, M.A.; Apfalter, P. First report of mecC MRSA in human samples from Austria: Molecular characteristics and clinical data. New Microbes New Infect. 2015, 3, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Lindgren, A.-K.; Gustafsson, E.; Petersson, A.C.; Melander, E. Methicillin-resistant Staphylococcus aureus with mecC: A description of 45 human cases in southern Sweden. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Haag, A.F.; Fitzgerald, J.R.; Penadés, J.R. Staphylococcus aureus in Animals. Microbiol. Spectr. 2019, 7, 7-3. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, Y.; Koide, S.; Maeyama, Y.; Tamai, K.; Hayashi, W.; Tanaka, H.; Iimura, M.; Suzuki, M.; Nagano, Y.; Arakawa, Y.; et al. Predominance of methicillin-resistant Staphylococcus aureus SCCmec type II-CC5 and SCCmec type IV-CC1/CC8 among companion animal clinical isolates in Japan: Findings from phylogenetic comparison with human clinical isolates. J. Glob. Antimicrob. Resist. 2020, 20, 253–259. [Google Scholar] [CrossRef]
- Matuszewska, M.; Murray, G.G.R.; Harrison, E.M.; Holmes, M.A.; Weinert, L.A. The Evolutionary Genomics of Host Specificity in Staphylococcus aureus. Trends Microbiol. 2020, 28, 465–477. [Google Scholar] [CrossRef]
- Kaspar, U.; von Lützau, K.; Schlattmann, A.; Rösler, U.; Köck, R.; Becker, K. Zoonotic multidrug-resistant microorganisms among non-hospitalized horses from Germany. One Health 2019, 7, 100091. [Google Scholar] [CrossRef]
- Amorim, M.L.; Aires de Sousa, M.; Sanches, I.S.; Sa-Leao, R.; Cabeda, J.M.; Amorim, J.M.; De Lencastre, H. Clonal and antibiotic resistance profiles of methicillin-resistant Staphylococcus aureus (MRSA) from a Portuguese hospital over time. Microb. Drug Resist. 2002, 8, 301–309. [Google Scholar] [CrossRef]
- Amorim, M.L.; Faria, N.A.; Oliveira, D.C.; Vasconcelos, C.; Cabeda, J.C.; Mendes, A.C.; Calado, E.; Castro, A.P.; Ramos, M.H.; Amorim, J.M. Changes in the clonal nature and antibiotic resistance profiles of methicillin-resistant Staphylococcus aureus isolates associated with spread of the EMRSA-15 clone in a tertiary care Portuguese hospital. J. Clin. Microbiol. 2007, 45, 2881–2888. [Google Scholar] [CrossRef]
- Amorim, M.L.; Vasconcelos, C.; Oliveira, D.C.; Azevedo, A.; Calado, E.; Faria, N.A.; Pereira, M.; Castro, A.P.; Moreira, A.; Aires, E. Epidemiology of methicillin-resistant Staphylococcus aureus (MRSA) nasal colonization among patients and healthcare workers in a Portuguese hospital: A pre-intervention study toward the control of MRSA. Microb. Drug Resist. 2009, 15, 19–26. [Google Scholar] [CrossRef]
- Espadinha, D.; Faria, N.A.; Miragaia, M.; Lito, L.M.; Melo-Cristino, J.; de Lencastre, H.; Network, M.S. Extensive Dissemination of Methicillin-Resistant Staphylococcus aureus (MRSA) between the Hospital and the Community in a Country with a High Prevalence of Nosocomial MRSA. PLoS ONE 2013, 8, e59960. [Google Scholar] [CrossRef] [PubMed]
- Faria, N.A.; Miragaia, M.; de Lencastre, H.; Multi Laboratory Project Collaborators. Massive Dissemination of Methicillin Resistant Staphylococcus aureus in Bloodstream Infections in a High MRSA Prevalence Country: Establishment and Diversification of EMRSA-15. Microb. Drug Resist. 2013, 19, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Knight, G.M.; Budd, E.L.; Lindsay, J.A. Large mobile genetic elements carrying resistance genes that do not confer a fitness burden in healthcare-associated meticillin-resistant Staphylococcus aureus. Microbiology 2013, 159, 1661–1672. [Google Scholar] [CrossRef]
- Tavares, A.; Miragaia, M.; Rolo, J.; Coelho, C.; de Lencastre, H.; CA-MRSA/MSSA Working Group. High prevalence of hospital-associated methicillin-resistant Staphylococcus aureus in the community in Portugal: Evidence for the blurring of community–hospital boundaries. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 1269–1283. [Google Scholar] [CrossRef] [PubMed]
- Conceição, T.; Aires-de-Sousa, M.; Pona, N.; Brito, M.J.; Barradas, C.; Coelho, R.; Sardinha, T.; Sancho, L.; De Sousa, G.; do Céu Machado, M. High prevalence of ST121 in community-associated methicillin-susceptible Staphylococcus aureus lineages responsible for skin and soft tissue infections in Portuguese children. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 293–297. [Google Scholar] [CrossRef][Green Version]
- Mottola, C.; Semedo-Lemsaddek, T.; Mendes, J.J.; Melo-Cristino, J.; Tavares, L.; Cavaco-Silva, P.; Oliveira, M. Molecular typing, virulence traits and antimicrobial resistance of diabetic foot staphylococci. J. Biomed. Sci. 2016, 23, 33. [Google Scholar] [CrossRef]
- McManus, B.A.; Daly, B.; Polyzois, I.; Wilson, P.; Brennan, G.I.; Fleming, T.E.; Grealy, L.D.; Healy, M.-L.; Coleman, D.C. Comparative Microbiological and Whole-Genome Analysis of Staphylococcus aureus Populations in the Oro-Nasal Cavities, Skin and Diabetic Foot Ulcers of Patients With Type 2 Diabetes Reveals a Possible Oro-Nasal Reservoir for Ulcer Infection. Front. Microbiol. 2020, 11, 748. [Google Scholar] [CrossRef]
- Cabete, J.; Martins de Carvalho, F.; Moniz, L.; Pinto, M.; Neves, J. Microbiological profile and antibiotic susceptibility patterns of organisms isolated from diabetic foot ulcers in a Portuguese hospital. Rev. Port. Cir. Cardiotorac. Vasc. 2011, 18, 53–60. [Google Scholar]
- Mendes, J.J.; Marques-Costa, A.; Vilela, C.; Neves, J.; Candeias, N.; Cavaco-Silva, P.; Melo-Cristino, J. Clinical and bacteriological survey of diabetic foot infections in Lisbon. Diabetes Res. Clin. Pract. 2012, 95, 153–161. [Google Scholar] [CrossRef]
- Neves, J.M.; Duarte, B.; Pinto, M.; Formiga, A.; Neves, J. Diabetic foot infection: Causative pathogens and empiric antibiotherapy considerations—the experience of a tertiary center. Int. J. Low. Extrem. Wounds 2019, 18, 122–128. [Google Scholar] [CrossRef]
- Silva, V.; Almeida, F.; Silva, A.; Correia, S.; Carvalho, J.A.; Castro, A.P.; Ferreira, E.; Manageiro, V.; Caniça, M.; Igrejas, G.; et al. First report of linezolid-resistant cfr-positive methicillin-resistant Staphylococcus aureus in humans in Portugal. J. Glob. Antimicrob. Resist. 2019, 17, 323–325. [Google Scholar] [CrossRef] [PubMed]
- Melo-Cristino, J.; Resina, C.; Manuel, V.; Lito, L.; Ramirez, M. First case of infection with vancomycin-resistant Staphylococcus aureus in Europe. Lancet 2013, 382, 205. [Google Scholar] [CrossRef]
- Finks, J.; Wells, E.; Dyke, T.L.; Husain, N.; Plizga, L.; Heddurshetti, R.; Wilkins, M.; Rudrik, J.; Hageman, J.; Patel, J. Vancomycin-resistant Staphylococcus aureus, Michigan, USA, 2007. Emerg. Infect. Dis. 2009, 15, 943. [Google Scholar] [CrossRef]
- Sá-Leão, R.; Santos Sanches, I.; Couto, I.; Alves, C.R.; de Lencastre, H. Low prevalence of methicillin-resistant strains among Staphylococcus aureus colonizing young and healthy members of the community in Portugal. Microb. Drug Resist. 2001, 7, 237–245. [Google Scholar] [CrossRef]
- David, M.Z. The Importance of Staphylococcus aureus Genotypes in Outcomes and Complications of Bacteremia. Clin. Infect. Dis. 2019, 69, 1878–1880. [Google Scholar] [CrossRef]
- Almeida, S.T.; Nunes, S.; Paulo, A.C.S.; Faria, N.A.; de Lencastre, H.; Sá-Leão, R. Prevalence, risk factors, and epidemiology of methicillin-resistant Staphylococcus aureus carried by adults over 60 years of age. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; Alfarela, C.; Caniça, M.; Manageiro, V.; Nóvoa, M.; Leiva, B.; Kress, M.; Capelo, J.L.; Poeta, P.; Igrejas, G. A One Health Approach Molecular Analysis of Staphylococcus aureus Reveals Distinct Lineages in Isolates from Miranda Donkeys (Equus asinus) and Their Handlers. Antibiotics 2022, 11, 374. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; Correia, S.; Rocha, J.; Manaia, C.; Silva, A.; García-Díez, J.; Pereira, J.E.; Semedo-Lemsaddek, T.; Igrejas, G.; Poeta, P. Antimicrobial resistance and clonal lineages of Staphylococcus aureus from cattle, their handlers, and their surroundings: A cross-sectional study in the One Health perspective. Microorganisms 2022, 10, 941. [Google Scholar] [CrossRef]
- Silva, V.; Caniça, M.; Manageiro, V.; Vieira-Pinto, M.; Pereira, J.E.; Maltez, L.; Poeta, P.; Igrejas, G. Staphylococcus aureus from hunters and hunting dogs: Molecular epidemiological evidence supporting human-to-dog and dog-to-dog transmission. Pathogens 2022, 11, 548. [Google Scholar] [CrossRef]
- Silva, V.; Capelo, J.L.; Igrejas, G.; Poeta, P. Molecular Epidemiology of Staphylococcus aureus Lineages in Wild Animals in Europe: A Review. Antibiotics 2020, 9, 122. [Google Scholar] [CrossRef]
- Silva, V.; Monteiro, A.; Porto, M.; Sampaio, A.; Maltez, L.; Pereira, J.E.; Aonofriesei, F.; Capelo, J.L.; Igrejas, G.; Poeta, P. Molecular Diversity of Methicillin-Resistant and -Susceptible Staphylococcus aureus Detected in Animals: A Focus on Aquatic Animals. Diversity 2021, 13, 417. [Google Scholar] [CrossRef]
- Graveland, H.; Duim, B.; van Duijkeren, E.; Heederik, D.; Wagenaar, J.A. Livestock-associated methicillin-resistant Staphylococcus aureus in animals and humans. Int. J. Med. Microbiol. 2011, 301, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Cienfuegos-Gallet, A.V.; Cunningham, M.H.; Jin, Y.; Wang, B.; Kreiswirth, B.N.; Chen, L. Molecular Evolution and Adaptation of Livestock-Associated Methicillin-Resistant Staphylococcus aureus (LA-MRSA) Sequence Type 9. Msystems 2021, 6, e00492-21. [Google Scholar] [CrossRef] [PubMed]
- Ballhausen, B.; Jung, P.; Kriegeskorte, A.; Makgotlho, P.E.; Ruffing, U.; von Müller, L.; Köck, R.; Peters, G.; Herrmann, M.; Ziebuhr, W. LA-MRSA CC398 differ from classical community acquired-MRSA and hospital acquired-MRSA lineages: Functional analysis of infection and colonization processes. Int. J. Med. Microbiol. 2014, 304, 777–786. [Google Scholar] [CrossRef]
- Gajdács, M.; Albericio, F. Antibiotic Resistance: From the Bench to Patients. Antibiotics 2019, 8, 129. [Google Scholar] [CrossRef]
- Masud, A.A.; Rousham, E.K.; Islam, M.A.; Alam, M.-U.; Rahman, M.; Mamun, A.A.; Sarker, S.; Asaduzzaman, M.; Unicomb, L. Drivers of antibiotic use in poultry production in Bangladesh: Dependencies and dynamics of a patron-client relationship. Front. Vet. Sci. 2020, 7, 78. [Google Scholar] [CrossRef]
- Bennani, H.; Mateus, A.; Mays, N.; Eastmure, E.; Stärk, K.D.C.; Häsler, B. Overview of evidence of antimicrobial use and antimicrobial resistance in the food chain. Antibiotics 2020, 9, 49. [Google Scholar] [CrossRef]
- Mascaro, V.; Leonetti, M.; Nobile, C.G.A.; Barbadoro, P.; Ponzio, E.; Recanatini, C.; Prospero, E.; Pavia, M.; Group, C.W. Prevalence of livestock-associated methicillin-resistant Staphylococcus aureus (LA-MRSA) among farm and slaughterhouse workers in Italy. J. Occup. Environ. Med. 2018, 60, e416–e425. [Google Scholar] [CrossRef]
- Chen, C.; Wu, F. Livestock-associated methicillin-resistant Staphylococcus aureus (LA-MRSA) colonisation and infection among livestock workers and veterinarians: A systematic review and meta-analysis. Occup. Environ. Med. 2021, 78, 530–540. [Google Scholar] [CrossRef]
- Schmithausen, R.M.; Schulze-Geisthoevel, S.V.; Heinemann, C.; Bierbaum, G.; Exner, M.; Petersen, B.; Steinhoff-Wagner, J. Reservoirs and Transmission Pathways of Resistant Indicator Bacteria in the Biotope Pig Stable and along the Food Chain: A Review from a One Health Perspective. Sustainability 2018, 10, 3967. [Google Scholar] [CrossRef]
- Ma, F.; Xu, S.; Tang, Z.; Li, Z.; Zhang, L. Use of antimicrobials in food animals and impact of transmission of antimicrobial resistance on humans. Biosaf. Health 2021, 3, 32–38. [Google Scholar] [CrossRef]
- Van der Mee-Marquet, N.; Corvaglia, A.-R.; Valentin, A.-S.; Hernandez, D.; Bertrand, X.; Girard, M.; Kluytmans, J.; Donnio, P.-Y.; Quentin, R.; François, P. Analysis of prophages harbored by the human-adapted subpopulation of Staphylococcus aureus CC398. Infect. Genet. Evol. 2013, 18, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Cuny, C.; Abdelbary, M.; Layer, F.; Werner, G.; Witte, W. Prevalence of the immune evasion gene cluster in Staphylococcus aureus CC398. Vet. Microbiol. 2015, 177, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Stegger, M.; Liu, C.M.; Larsen, J.; Soldanova, K.; Aziz, M.; Contente-Cuomo, T.; Petersen, A.; Vandendriessche, S.; Jiménez, J.N.; Mammina, C.; et al. Rapid Differentiation between Livestock-Associated and Livestock-Independent Staphylococcus aureus CC398 Clades. PLoS ONE 2013, 8, e79645. [Google Scholar] [CrossRef] [PubMed]
- Argudín, M.A.; Deplano, A.; Vandendriessche, S.; Dodémont, M.; Nonhoff, C.; Denis, O.; Roisin, S. CC398 Staphylococcus aureus subpopulations in Belgian patients. Eur. J. Clin. Microbiol. Infect. Dis. 2018, 37, 911–916. [Google Scholar] [CrossRef]
- Ji, X.; Krüger, H.; Feßler, A.T.; Liu, J.; Zeng, Z.; Wang, Y.; Wu, C.; Schwarz, S. A novel SCCmec type V variant in porcine MRSA ST398 from China. J. Antimicrob. Chemother. 2020, 75, 484–486. [Google Scholar] [CrossRef]
- Pomba, C.; Hasman, H.; Cavaco, L.M.; da Fonseca, J.D.; Aarestrup, F.M. First description of meticillin-resistant Staphylococcus aureus (MRSA) CC30 and CC398 from swine in Portugal. Int. J. Antimicrob. Agents 2009, 34, 193–194. [Google Scholar] [CrossRef]
- Conceição, T.; De Lencastre, H.; Aires-De-Sousa, M. Frequent isolation of methicillin resistant Staphylococcus aureus (MRSA) ST398 among healthy pigs in Portugal. PLoS ONE 2017, 12, e0175340. [Google Scholar] [CrossRef]
- Couto, N.; Belas, A.; Centeno, M.; Van Duijkeren, E.; Pomba, C. First description of fexA-positive meticillin-resistant Staphylococcus aureus ST398 from calves in Portugal. J. Glob. Antimicrob. Resist. 2014, 2, 342–343. [Google Scholar] [CrossRef]
- Kehrenberg, C.; Schwarz, S. Florfenicol-chloramphenicol exporter gene fexA is part of the novel transposon Tn 558. Antimicrob. Agents Chemother. 2005, 49, 813–815. [Google Scholar] [CrossRef]
- Conceição, T.; de Lencastre, H.; Aires-de-Sousa, M. Healthy Bovines as Reservoirs of Major Pathogenic Lineages of Staphylococcus aureus in Portugal. Microb. Drug Resist. 2017, 23, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Correia, S.; Silva, V.; García-Díez, J.; Teixeira, P.; Pimenta, K.; Pereira, J.E.; Oliveira, S.; Rocha, J.; Manaia, C.M.; Igrejas, G.; et al. One Health Approach Reveals the Absence of Methicillin-Resistant Staphylococcus aureus in Autochthonous Cattle and Their Environments. Front. Microbiol. 2019, 10, 2735. [Google Scholar] [CrossRef] [PubMed]
- Correia, S.; Silva, V.; García-Díez, J.; Teixeira, P.; Pimenta, K.; Tejedor-Junco, M.T.; Oliveira, S.; Igrejas, G.; Poeta, P. Absence of Methicillin-Resistant Staphylococcus aureus (MRSA) in Cattle from Portugal: A One Health Approach. Infect. Drug Resist. 2019, 12, 3421–3423. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Grúa, E.; Pérez-Fuentes, S.; Muñoz-Silvestre, A.; Viana, D.; Fernández-Ros, A.B.; Sanz-Tejero, C.; Corpa, J.M.; Selva, L. Characterization of Livestock-Associated Methicillin-Resistant Staphylococcus aureus Isolates Obtained From Commercial Rabbitries Located in the Iberian Peninsula. Front. Microbiol. 2018, 9, 1812. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; de Sousa, T.; Gómez, P.; Sabença, C.; Vieira-Pinto, M.; Capita, R.; Alonso-Calleja, C.; Torres, C.; Capelo, J.L.; Igrejas, G.; et al. Livestock-Associated Methicillin-Resistant Staphylococcus aureus (MRSA) in Purulent Subcutaneous Lesions of Farm Rabbits. Foods 2020, 9, 439. [Google Scholar] [CrossRef]
- Vincze, S.; Stamm, I.; Kopp, P.A.; Hermes, J.; Adlhoch, C.; Semmler, T.; Wieler, L.H.; Lübke-Becker, A.; Walther, B. Alarming Proportions of Methicillin-Resistant Staphylococcus aureus (MRSA) in Wound Samples from Companion Animals, Germany 2010–2012. PLoS ONE 2014, 9, e85656. [Google Scholar] [CrossRef]
- Petinaki, E.; Spiliopoulou, I. Methicillin-resistant Staphylococcus aureus colonization and infection risks from companion animals: Current perspectives. Veter. Med. Res. Rep. 2015, 6, 373–382. [Google Scholar]
- Couto, N.; Monchique, C.; Belas, A.; Marques, C.; Gama, L.T.; Pomba, C. Trends and molecular mechanisms of antimicrobial resistance in clinical staphylococci isolated from companion animals over a 16 year period. J. Antimicrob. Chemother. 2016, 71, 1479–1487. [Google Scholar] [CrossRef]
- Harrison, E.M.; Weinert, L.A.; Holden, M.T.G.; Welch, J.J.; Wilson, K.; Morgan, F.J.E.; Harris, S.R.; Loeffler, A.; Boag, A.K.; Peacock, S.J. A shared population of epidemic methicillin-resistant Staphylococcus aureus 15 circulates in humans and companion animals. MBio 2014, 5, e00985-13. [Google Scholar] [CrossRef]
- Coelho, C.; Torres, C.; Radhouani, H.; Pinto, L.; Lozano, C.; Gómez-Sanz, E.; Zaragaza, M.; Igrejas, G.; Poeta, P. Molecular Detection and Characterization of Methicillin-Resistant Staphylococcus aureus (MRSA) Isolates from Dogs in Portugal. Microb. Drug Resist. 2011, 17, 333–337. [Google Scholar] [CrossRef]
- Cuny, C.; Strommenger, B.; Witte, W.; Stanek, C. Clusters of Infections in Horses with MRSA ST1, ST254, and ST398 in a Veterinary Hospital. Microb. Drug Resist. 2008, 14, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Couto, N.; Tilley, P.; Simões, J.; Luis, J.P.S.; Pomba, C. First report of methicillin-resistant Staphylococcus aureus ST5 and ST398 from purebred Lusitano horses. J. Equine Vet. Sci. 2012, 32, 300–304. [Google Scholar] [CrossRef]
- Albert, E.; Biksi, I.; Német, Z.; Csuka, E.; Kelemen, B.; Morvay, F.; Bakos, Z.; Bodó, G.; Tóth, B.; Collaud, A. Outbreaks of a methicillin-resistant Staphylococcus aureus clone ST398-t011 in a Hungarian equine clinic: Emergence of rifampicin and chloramphenicol resistance after treatment with these antibiotics. Microb. Drug Resist. 2019, 25, 1219–1226. [Google Scholar] [CrossRef] [PubMed]

| Clonal Complex (CC) | Sequence Type (ST) | Spa-Type | SCCmec Type | Clone | Geographic Distribution |
|---|---|---|---|---|---|
| CC1 | ST6 | t304 and variants | IVa | Middle East | The Middle East and Europe |
| CC5 | ST5 | t001, t002, t003, t010, t045, t053, t062, t105, t178, t179, t187, t214, t311, t319, t389, t443 | II | New York/Japan, USA100 | United States, Japan, Europe, Australia and South Korea |
| t001, t002, t003, t010, t045, t053, t062, t105, t178, t179, t187, t214, t311, t319, t389, t443 | IV | Pediatric/USA800 | South America and Europe | ||
| CC8 | ST239 | t030, t037, t234, t387, t388 | III | Brazilian/Hungarian | Europe, South America, Asia and Africa |
| ST247 | t008, t051, t052, t054, t200 | I | Iberian/EMRSA-5 | Europe and the United States | |
| ST250 | t008, t009, t194 | I | Archaic | Worldwide | |
| CC22 | ST22 | t005, t022, t032, t223, t309, t310, t417, t420 | IV | EMRSA-15 | Europe, Australia and Canada |
| CC30 | ST36 | t018, t253, t418, t419 | II | EMRSA-16, USA200 | Europe, North America, and Australia |
| ST30 | t012, t019, t1143, t300 | IV | USA1100/South West Pacific | America, Australia and the Western Pacific | |
| CC45 | ST45 | t004, t015, t026, t031, t038, t050, t065, t204, t230, t390 | IV | Berlin, USA600 | Europe and the United States |
| ST80 | ST80 | t044, t203, t131, t1028, t1200 | IV | European | Europe, North Africa and the Middle East |
| ST93 | t3949 t202 t15361 t4699 t17089 t16949 t17272 | IV | Queensland | Australia |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, V.; Monteiro, A.; Pereira, J.E.; Maltez, L.; Igrejas, G.; Poeta, P. MRSA in Humans, Pets and Livestock in Portugal: Where We Came from and Where We Are Going. Pathogens 2022, 11, 1110. https://doi.org/10.3390/pathogens11101110
Silva V, Monteiro A, Pereira JE, Maltez L, Igrejas G, Poeta P. MRSA in Humans, Pets and Livestock in Portugal: Where We Came from and Where We Are Going. Pathogens. 2022; 11(10):1110. https://doi.org/10.3390/pathogens11101110
Chicago/Turabian StyleSilva, Vanessa, Andreia Monteiro, José Eduardo Pereira, Luís Maltez, Gilberto Igrejas, and Patrícia Poeta. 2022. "MRSA in Humans, Pets and Livestock in Portugal: Where We Came from and Where We Are Going" Pathogens 11, no. 10: 1110. https://doi.org/10.3390/pathogens11101110
APA StyleSilva, V., Monteiro, A., Pereira, J. E., Maltez, L., Igrejas, G., & Poeta, P. (2022). MRSA in Humans, Pets and Livestock in Portugal: Where We Came from and Where We Are Going. Pathogens, 11(10), 1110. https://doi.org/10.3390/pathogens11101110











