Severe West Nile Virus Neuroinvasive Disease: Clinical Characteristics, Short- and Long-Term Outcomes
Abstract
1. Introduction
2. Results
2.1. Laboratory Results
2.2. Virology Results
2.3. Antiviral Cytokine Concentrations in Serum and CSF
2.4. Neuroimaging and Electroencephalography Results
2.5. Outcomes of Patients with Severe WNV NID
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. West Nile Virus Detection and Phylogenetic Analysis
4.3. Quantification of Cytokines in Serum and CSF
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brown, J.A.; Factor, D.L.; Tkachenko, N.; Templeton, S.M.; Crall, N.D.; Pape, W.J.; Bauer, M.J.; Ambruso, D.R.; Dickey, W.C.; Marfin, A.A. West Nile viremic blood donors and risk factors for subsequent West Nile fever. Vector-Borne Zoonotic Dis. 2007, 7, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Nash, D.; Mostashari, F.; Fine, A.; Miller, J.; O’Leary, D.; Murray, K.; Huang, A.; Rosenberg, A.; Greenberg, A.; Sherman, M.; et al. The outbreak of West Nile virus infection in the New York City area in 1999. N. Engl. J. Med. 2001, 344, 1807–1814. [Google Scholar] [CrossRef]
- Orton, S.L.; Stramer, S.L.; Dodd, R.Y. Self-reported symptoms associated with West Nile virus infection in RNA-positive blood donors. Transfusion 2006, 46, 272–277. [Google Scholar] [CrossRef]
- Zou, S.; Foster, G.A.; Dodd, R.Y.; Petersen, L.R.; Stramer, S.L. West Nile fever characteristics among viremic persons identified through blood donor screening. J. Infect. Dis. 2010, 202, 1354–1361. [Google Scholar] [CrossRef]
- Weatherhead, J.E.; Miller, V.E.; Garcia, M.N.; Hasbun, R.; Salazar, L.; Dimachkie, M.M.; Murray, K.O. Long-term neurological outcomes in West Nile virus-infected patients: An observational study. Am. J. Trop. Med. Hyg. 2015, 92, 1006–1012. [Google Scholar] [CrossRef]
- Sejvar, J.J.; Leis, A.A.; Stokic, D.S.; Van Gerpen, J.A.; Marfin, A.A.; Webb, R.; Haddad, M.B.; Tierney, B.C.; Slavinski, S.A.; Polk, J.L.; et al. Acute flaccid paralysis and West Nile virus infection. Emerg. Infect. Dis. 2003, 9, 788–793. [Google Scholar] [CrossRef]
- Athar, P.; Hasbun, R.; Nolan, M.S.; Salazar, L.; Woods, S.P.; Sheikh, K.; Murray, K.O. Long-term neuromuscular outcomes of West Nile virus infection: A clinical and electromyographic evaluation of patients with a history of infection. Muscle Nerve 2018, 57, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Leis, A.A.; Stokic, D.S. Neuromuscular manifestations of human West Nile virus infection. Curr. Treat. Options Neurol. 2005, 7, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Popovic, N.; Milosevic, B.; Urosevic, A.; Poluga, J.; Popovic, N.; Stevanovic, G.; Milosevic, I.; Korac, M.; Mitrovic, N.; Lavadinovic, L.; et al. Clinical characteristics and functional outcome of patients with West Nile neuroinvasive disease in Serbia. J. Neurol. 2014, 261, 1104–1111. [Google Scholar] [CrossRef]
- Fan, E.; Needham, D.M.; Brunton, J.; Kern, R.Z.; Stewart, T.E. West Nile virus infection in the intensive care unit: A case series and literature review. Can. Respir. J. 2004, 11, 354–358. [Google Scholar] [CrossRef]
- Sejvar, J.J. Clinical manifestations and outcomes of West Nile virus infection. Viruses 2014, 6, 606–623. [Google Scholar] [CrossRef]
- Hawkes, M.A.; Carabenciov, I.D.; Wijdicks, E.F.M.; Rabinstein, A.A. Outcomes in patients with severe West Nile neuroinvasive disease. Crit. Care Med. 2018, 46, e955–e958. [Google Scholar] [CrossRef]
- Hawkes, M.A.; Carabenciov, I.D.; Wijdicks, E.F.M.; Rabinstein, A.A. Critical West Nile neuroinvasive disease. Neurocritical Care 2018, 29, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Pem-Novosel, I.; Vilibic-Cavlek, T.; Gjenero-Margan, I.; Pandak, N.; Peric, L.; Barbic, L.; Listes, E.; Cvitkovic, A.; Stevanovic, V.; Savini, G. First outbreak of West Nile virus neuroinvasive disease in humans, Croatia, 2012. Vector-Borne Zoonotic Dis. 2014, 14, 82–84. [Google Scholar] [CrossRef] [PubMed]
- Vilibic-Cavlek, T.; Kaic, B.; Barbic, L.; Pem-Novosel, I.; Slavic-Vrzic, V.; Lesnikar, V.; Kurecic-Filipovic, S.; Babic-Erceg, A.; Listes, E.; Stevanovic, V.; et al. First evidence of simultaneous occurrence of West Nile virus and Usutu virus neuroinvasive disease in humans in Croatia during the 2013 outbreak. Infection 2014, 42, 689–695. [Google Scholar] [CrossRef]
- Vilibic-Cavlek, T.; Savic, V.; Sabadi, D.; Peric, L.; Barbic, L.; Klobucar, A.; Miklausic, B.; Tabain, I.; Santini, M.; Vucelja, M.; et al. Prevalence and molecular epidemiology of West Nile and Usutu virus infections in Croatia in the ‘One health’ context, 2018. Transbound. Emerg. Dis. 2019, 66, 1946–1957. [Google Scholar] [CrossRef]
- Vilibic-Cavlek, T.; Barbic, L.; Mrzljak, A.; Brnic, D.; Klobucar, A.; Ilic, M.; Janev-Holcer, N.; Bogdanic, M.; Jemersic, L.; Stevanovic, V.; et al. Emerging and neglected viruses of zoonotic importance in Croatia. Pathogens 2021, 10, 73. [Google Scholar] [CrossRef]
- Konjevoda, S.; Dzelalija, B.; Canovic, S.; Pastar, Z.; Savic, V.; Tabain, I.; Barbic, L.; Peric, L.; Sabadi, D.; Stevanovic, V.; et al. West Nile virus retinitis in a patient with neuroinvasive disease. Rev. Soc. Bras. Med. Trop. 2019, 52, e20190065. [Google Scholar] [CrossRef]
- Vilibic-Cavlek, T.; Savic, V.; Klobucar, A.; Ferenc, T.; Ilic, M.; Bogdanic, M.; Tabain, I.; Stevanovic, V.; Santini, M.; Curman Posavec, M.; et al. Emerging trends in the West Nile virus epidemiology in Croatia in the ‘One Health’ Context, 2011–2020. Trop. Med. Infect. Dis. 2021, 6, 140. [Google Scholar] [CrossRef]
- Santini, M.; Zupetic, I.; Viskovic, K.; Krznaric, J.; Kutlesa, M.; Krajinovic, V.; Polak, V.L.; Savic, V.; Tabain, I.; Barbic, L.; et al. Cauda equina arachnoiditis—A rare manifestation of West Nile virus neuroinvasive disease: A case report. World J. Clin. Cases 2020, 8, 3797–3803. [Google Scholar] [CrossRef]
- Sabadi, D.; Peric, L.; Savic, V.; Rubil, I.; Baraban, V.; Tabain, I.; Barbic, L.; Duvnjak, M.; Bogdanic, M.; Stevanovic, V.; et al. Fatal case of West Nile encephalitis associated with acute anteroseptal ST elevation myocardial infarction (STEMI): A case report. New Microbiol. 2020, 43, 51–53. [Google Scholar] [PubMed]
- Rizzoli, A.; Jimenez-Clavero, M.A.; Barzon, L.; Cordioli, P.; Figuerola, J.; Koraka, P.; Martina, B.; Moreno, A.; Nowotny, N.; Pardigon, N.; et al. The challenge of West Nile virus in Europe: Knowledge gaps and research priorities. Eurosurveillance 2015, 20, 21135. [Google Scholar] [CrossRef]
- Anastasiadou, A.; Kakoulidis, I.; Butel, D.; Kehagia, E.; Papa, A. Follow-up study of Greek patients with West Nile virus neuroinvasive disease. Int. J. Infect. Dis. 2013, 17, e494–e497. [Google Scholar] [CrossRef]
- Carson, P.J.; Konewko, P.; Wold, K.S.; Mariani, P.; Goli, S.; Bergloff, P.; Crosby, R.D. Long-term clinical and neuropsychological outcomes of West Nile virus infection. Clin. Infect. Dis. 2006, 43, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Klee, A.L.; Maidin, B.; Edwin, B.; Poshni, I.; Mostashari, F.; Fine, A.; Layton, M.; Nash, D. Long-term prognosis for clinical West Nile virus infection. Emerg. Infect. Dis. 2004, 10, 1405–1411. [Google Scholar] [CrossRef]
- Sejvar, J.J.; Haddad, M.B.; Tierney, B.C.; Campbell, G.L.; Marfin, A.A.; Van Gerpen, J.A.; Fleischauer, A.; Leis, A.A.; Stokic, D.S.; Petersen, L.R. Neurologic manifestations and outcome of West Nile virus infection. JAMA 2003, 290, 511–515. [Google Scholar] [CrossRef] [PubMed]
- Tsai, T.F.; Popovici, F.; Cernescu, C.; Campbell, G.L.; Nedelcu, N.I. West Nile encephalitis epidemic in southeastern Romania. Lancet 1998, 352, 767–771. [Google Scholar] [CrossRef]
- Bailly, L.; Fabre, R.; Courjon, J.; Carles, M.; Dellamonica, J.; Pradier, C. Obesity, diabetes, hypertension and severe outcomes among inpatients with coronavirus disease 2019: A nationwide study. Clin. Microbiol. Infect. 2021, 28, P114–P123. [Google Scholar] [CrossRef]
- Chaintoutis, S.C.; Papa, A.; Pervanidou, D.; Dovas, C.I. Evolutionary dynamics of lineage 2 West Nile virus in Europe, 2004–2018: Phylogeny, selection pressure and phylogeography. Mol. Phylogenetics Evol. 2019, 141, 106617. [Google Scholar] [CrossRef] [PubMed]
- Cotar, A.I.; Fălcuță, E.; Dinu, S.; Necula, A.; Bîrluțiu, V.; Ceianu, C.S.; Prioteasa, F.L. West Nile virus lineage 2 in Romania, 2015–2016: Co-circulation and strain replacement. Parasites Vectors 2018, 11, 562. [Google Scholar] [CrossRef]
- Fares-Gusmao, R.; Rocha, B.C.; Sippert, E.; Lanteri, M.C.; Anez, G.; Rios, M. Differential pattern of soluble immune markers in asymptomatic dengue, West Nile and Zika virus infections. Sci. Rep. 2019, 9, 17172. [Google Scholar] [CrossRef]
- Garcia, M.N.; Hause, A.M.; Walker, C.M.; Orange, J.S.; Hasbun, R.; Murray, K.O. Evaluation of prolonged fatigue post-West Nile virus infection and association of fatigue with elevated antiviral and proinflammatory cytokines. Viral Immunol. 2014, 27, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, K.W.; Lee, J.J.; Foster, G.A.; Krysztof, D.; Stramer, S.L.; Lim, J.K. Sex differences in cytokine production following West Nile virus infection: Implications for symptom manifestation. Pathog. Dis. 2019, 77, ftz016. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, K.W.; Sachs, D.; Bardina, S.V.; Michlmayr, D.; Rodriguez, C.A.; Sum, J.; Foster, G.A.; Krysztof, D.; Stramer, S.L.; Lim, J.K. Differences in early cytokine production are associated with development of a greater number of symptoms following West Nile virus infection. J. Infect. Dis. 2016, 214, 634–643. [Google Scholar] [CrossRef]
- Qian, F.; Thakar, J.; Yuan, X.; Nolan, M.; Murray, K.O.; Lee, W.T.; Wong, S.J.; Meng, H.; Fikrig, E.; Kleinstein, S.H.; et al. Immune markers associated with host susceptibility to infection with West Nile virus. Viral Immunol. 2014, 27, 39–47. [Google Scholar] [CrossRef]
- Zidovec-Lepej, S.; Vilibic-Cavlek, T.; Barbic, L.; Ilic, M.; Savic, V.; Tabain, I.; Ferenc, T.; Grgic, I.; Gorenec, L.; Bogdanic, M.; et al. Antiviral cytokine response in neuroinvasive and non-neuroinvasive West Nile virus infection. Viruses 2021, 13, 342. [Google Scholar] [CrossRef] [PubMed]
- Rothaug, M.; Becker-Pauly, C.; Rose-John, S. The role of interleukin-6 signaling in nervous tissue. Biochim. Biophys. Acta 2016, 1863, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Zak, I.T.; Altinok, D.; Merline, J.R.; Chander, S.; Kish, K.K. West Nile virus infection. Am. J. Roentgenol. 2005, 184, 957–961. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.; Mach, J.; Kataria, S.; Tandon, M.; Lakhani, D.A.; Sriwastava, S. West Nile virus encephalitis: A report of two cases and review of neuroradiological features. Radiol. Case Rep. 2020, 15, 2422–2426. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Factsheet about West Nile Virus Infection. Available online: https://www.ecdc.europa.eu/en/west-nile-fever/facts (accessed on 1 November 2021).
- Ali, M.; Safriel, Y.; Sohi, J.; Llave, A.; Weathers, S. West Nile virus infection: MR imaging findings in the nervous system. Am. J. Neuroradiol. 2005, 26, 289–297. [Google Scholar]
- DeQuesada, I.; Saindane, A. West Nile virus presenting as flaccid paralysis: Case report and literature review. Radiol. Case Rep. 2012, 7, 705. [Google Scholar] [CrossRef][Green Version]
- Hwang, J.; Ryu, H.S.; Kim, H.; Lee, S.A. The first reported case of West Nile encephalitis in Korea. J. Korean Med. Sci. 2015, 30, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Jeha, L.E.; Sila, C.A.; Lederman, R.J.; Prayson, R.A.; Isada, C.M.; Gordon, S.M. West Nile virus infection: A new acute paralytic illness. Neurology 2003, 61, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Petropoulou, K.A.; Gordon, S.M.; Prayson, R.A.; Ruggierri, P.M. West Nile virus meningoencephalitis: MR imaging findings. Am. J. Neuroradiol. 2005, 26, 1986–1995. [Google Scholar]
- Santini, M.; Vilibic-Cavlek, T.; Barsic, B.; Barbic, L.; Savic, V.; Stevanovic, V.; Listes, E.; Di Gennaro, A.; Savini, G. First cases of human Usutu virus neuroinvasive infection in Croatia, August–September 2013: Clinical and laboratory features. J. Neurovirol. 2015, 21, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Schwaiger, M.; Cassinotti, P. Development of a quantitative real-time RT-PCR assay with internal control for the laboratory detection of tick borne encephalitis virus (TBEV) RNA. J. Clin. Virol. 2003, 27, 136–145. [Google Scholar] [CrossRef]
- Nikolay, B.; Weidmann, M.; Dupressoir, A.; Faye, O.; Boye, C.S.; Diallo, M.; Sall, A.A. Development of a Usutu virus specific real-time reverse transcription PCR assay based on sequenced strains from Africa and Europe. J. Virol. Methods 2014, 197, 51–54. [Google Scholar] [CrossRef]
- Weidmann, M.; Sanchez-Seco, M.P.; Sall, A.A.; Ly, P.O.; Thiongane, Y.; Lo, M.M.; Schley, H.; Hufert, F.T. Rapid detection of important human pathogenic Phleboviruses. J. Clin. Virol. 2008, 41, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Cao, Y.X.; He, X.X.; Fu, S.H.; Lyu, Z.; He, Y.; Gao, X.Y.; Liang, G.D.; Wang, H.Y. Real-time RT-PCR Assay for the detection of Tahyna Virus. Biomed. Environ. Sci. 2015, 28, 374–377. [Google Scholar] [CrossRef]
- Matsuno, K.; Weisend, C.; da Rosa, A.P.T.; Anzick, S.L.; Dahlstrom, E.; Porcella, S.F.; Dorward, D.W.; Yu, X.J.; Tesh, R.B.; Ebihara, H. Characterization of the Bhanja serogroup viruses (Bunyaviridae): A novel species of the genus Phlebovirus and its relationship with other emerging tick-borne phleboviruses. J. Virol. 2013, 87, 3719–3728. [Google Scholar] [CrossRef]
- Tang, Y.; Hapip, C.A.; Liu, B.; Fang, C.T. Highly sensitive TaqMan RT-PCR assay for detection and quantification of both lineages of West Nile virus RNA. J. Clin. Virol. 2006, 36, 177–182. [Google Scholar] [CrossRef]
- Weissenbock, H.; Kolodziejek, J.; Url, A.; Lussy, H.; Rebel-Bauder, B.; Nowotny, N. Emergence of Usutu virus, an African mosquito-borne flavivirus of the Japanese encephalitis virus group, central Europe. Emerg. Infect. Dis. 2002, 8, 652–656. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Vilibic-Cavlek, T.; Kristofic, B.; Savic, V.; Kolaric, B.; Barbic, L.; Tabain, I.; Peric, L.; Sabadi, D.; Miklausic, B.; Potocnik-Hunjadi, T.; et al. Diagnostic significance of immunoglobulin G avidity in symptomatic and asymptomatic West Nile virus infection. Rev. Soc. Bras. Med. Trop. 2018, 51, 591–595. [Google Scholar] [CrossRef]
- Di Gennaro, A.; Lorusso, A.; Casaccia, C.; Conte, A.; Monaco, F.; Savini, G. Serum neutralization assay can efficiently replace plaque reduction neutralization test for detection and quantitation of West Nile virus antibodies in human and animal serum samples. Clin. Vaccine Immunol. 2014, 21, 1460–1462. [Google Scholar] [CrossRef] [PubMed]
- LEGENDplex™ HU Th Cytokine Panel (12-plex) w/ FP V02, Th Cytokine. Available online: https://www.biolegend.com/en-us/products/legendplex-hu-th-cytokine-panel-12-plex-wfp-v02-19471 (accessed on 19 September 2021).
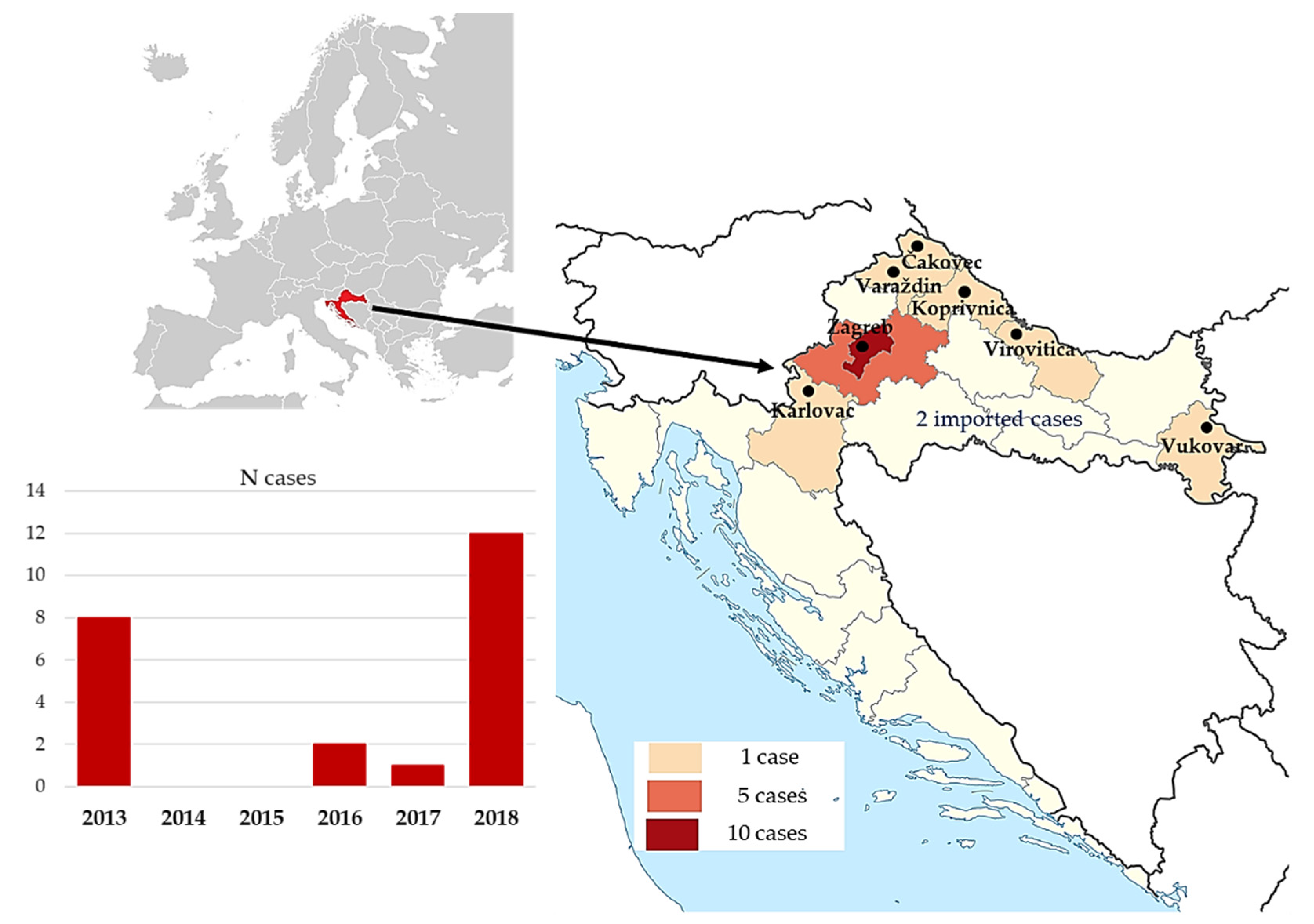



| Characteristic. | n = 23 (% or Range) |
|---|---|
| Demographics | |
| Age, median (range) years | 72 (33–84) |
| Male gender, n (%) | 18 (78.3) |
| Area of residence | |
| Urban, n (%) | 16 (69.6) |
| Suburban/rural, n (%) | 5 (21.7) |
| Imported infections, n (%) | 2 (8.7) |
| Patients with comorbidities | 22 (95.6) |
| Arterial hypertension, n (%) | 19 (82.6) |
| Diabetes mellitus, n (%) | 9 (39.1) |
| Coronary artery disease, n (%) | 4 (17.8) |
| Kidney transplant, n (%) | 3 (13.0) |
| Date of admittance (range) | 21 July–13 September |
| TBE/YF/JE vaccination | 0 (0) |
| Clinical presentation | |
| Meningitis, n (%) | 0 (0) |
| Encephalitis, n (%) | 13 (56.5) |
| Encephalitis + AFP, n (%) | 10 (43.5) |
| Isolated AFP, n (%) | 0 (0) |
| Signs and symptoms at presentation | |
| Fever, n (%) | 23 (100) |
| Fever (°C), mean ± SD | 39.3 ± 0.6 |
| Consciousness impairment, n (%) | 18 (78.3%) |
| GCS, median (range) | 10 (3–15) |
| Headache, n (%) | 10 (43.5) |
| Tremor, n (%) | 6 (26.1) |
| Diarrhea, n (%) | 4 (17.4) |
| Days to hospitalization, median (range) | 4 (1–13) |
| Mechanical ventilation, n (%) | 12 (52.2) |
| Duration of mechanical ventilation, median days (range) | 12 (5–73) |
| CSF Results | ||
|---|---|---|
| Patients with elevated CSF cell count, n (%) | 23 (100%) | Reference values |
| Cells/mm3, median (range) | 197 (12–1520) | 0–5 |
| Patients with CSF mononuclear predominance, n (%) | 17 (73.9%) | |
| Mononuclear cells, % median (range) | 60 (25–98) | Mononuclear 100% |
| Proteins (g/L) mean ± SD | 1.1 ± 0.5 | 0.17–0.37 g/L |
| Glucose (mmol/L) mean ± SD | 5.0 ± 2.1 | 2.5–3.3 mmol/L |
| Bacterial cultures and PCR | Negative (100%) | |
| Hematological and biochemistry results (average ± standard deviation) | ||
| CRP (mg/L) | 44.2 ± 45.9 | <5.0 mg/L |
| White blood cell count (×109/L) | 11.17 ± 4.00 | 4.0–10.0 (×109/L) |
| Red blood cells count (×1012/L) | 4.45 ± 0.08 | 4.4–5.8 (×1012/L) |
| Hemoglobin (g/L) | 133.26 ± 19.73 | 120–180 g/L |
| Platelets (×109/L) | 155.5 ± 58.35 | 100–400 (×109/L) |
| AST (U/L) | 37.13 ± 20.90 | 11–38 U/L |
| ALT (U/L) | 34.9 ± 22.5 | 12–48 U/L |
| GGT (U/L) | 73.9 ± 84.4 | 11–55 U/L |
| Cytokine | Serum (N = 15) | CSF (N = 11) | ||
|---|---|---|---|---|
| N Positive (%) | 95% CI | N Positive (%) | 95% CI | |
| IL-5 | 4 (26.6) | 7.8–55.1 | 1 (9.1) | 0.2–41.3 |
| IL-13 | 1 (6.6) | 0.2–31.9 | 1 (9.1) | 0.2–41.3 |
| IL-2 | 1 (6.6) | 0.2–31.9 | 0 (0) | 0–28.5 * |
| IL-6 | 13 (86.6) | 59.5–98.3 | 11 (100) | 71.5–100 * |
| IL-9 | 3 (20.0) | 4.3–48.1 | 3 (27.2) | 6.0–60.9 |
| IL-10 | 3 (20.0) | 4.3–48.1 | 3 (27.2) | 6.0–60.9 |
| IFN-γ | 10 (66.6) | 38.4–88.2 | 9 (81.8) | 48.2–97.7 |
| TNF-α | 4 (26.6) | 7.8–55.1 | 0 (0) | 0–28.5 * |
| IL-17A | 4 (26.6) | 7.8–55.1 | 0 (0) | 0–28.5 * |
| IL-17F | 2 (13.3) | 1.6–40.5 | 0 (0) | 0–28.5 * |
| IL-4 | 1 (6.6) | 0.2–31.9 | 0 (0) | 0–28.5 * |
| IL-21 | 2 (13.3) | 1.6–40.5 | 0 (0) | 0–28.5 * |
| IL-22 | 3 (20.0) | 4.3–48.1 | 4 (36.3) | 10.9–69.2 |
| Outcomes at Discharge | |
|---|---|
| Lethal outcome in ICU, n (%) | 2 (8.6) |
| Lethal outcome in hospital, n (%) | 0 (0) |
| ICU length of stay, median days (range) | 19 (5–73) |
| Hospital length of stay, median days (range) | 34 (7–97) |
| Outcomes at follow-up | |
| Available at follow-up, n (%) | 17 (73.9%) |
| Months to follow-up for survivors, median (range) | 9 (6–69) |
| Deceased, n (%) | 7 (30.4%) |
| Improved, n (%) | 10 (43.5%) |
| Aggravated, n (%) | 1 (4.3%) |
| Unaltered, n (%) | 1 (4.3%) |
| Lost to follow-up, n (%) | 4 (17.5%) |
| European Union clinical and laboratory criteria for WNV [40]. Laboratory diagnosis was confirmed by detecting WNV RNA in CSF (n = 3), WNV IgM in CSF (n = 5), WNV IgM/IgG in serum (n = 15), and confirmed by a Virus Neutralization Test (VNT), and 1 of the following: |
| Clinical evidence of meningitis (one or more of the following) |
| Positive meningeal signs |
| Pleocytosis in cerebrospinal fluid |
| Brain CT or MR finding consistent with inflammation |
| Clinical evidence of encephalitis (1 or more of the following) |
| Altered level of consciousness lasting at least 24 h |
| Focal neurological deficit |
| Seizure |
| Brain CT or MR finding consistent with brain inflammation |
| Abnormal electroencephalography |
| Acute-onset asymmetric limb weakness progressing over 48 h, and 2 or more of the following: |
| Hyporeflexia or areflexia of affected limbs |
| No pain or paresthesia of affected limbs |
| Pleocytosis ≥ 5 cells/mm3 , and elevated protein in cerebrospinal fluid |
| Spinal cord MR finding showing inflammation in anterior horns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santini, M.; Haberle, S.; Židovec-Lepej, S.; Savić, V.; Kusulja, M.; Papić, N.; Višković, K.; Župetić, I.; Savini, G.; Barbić, L.; et al. Severe West Nile Virus Neuroinvasive Disease: Clinical Characteristics, Short- and Long-Term Outcomes. Pathogens 2022, 11, 52. https://doi.org/10.3390/pathogens11010052
Santini M, Haberle S, Židovec-Lepej S, Savić V, Kusulja M, Papić N, Višković K, Župetić I, Savini G, Barbić L, et al. Severe West Nile Virus Neuroinvasive Disease: Clinical Characteristics, Short- and Long-Term Outcomes. Pathogens. 2022; 11(1):52. https://doi.org/10.3390/pathogens11010052
Chicago/Turabian StyleSantini, Marija, Sara Haberle, Snježana Židovec-Lepej, Vladimir Savić, Marija Kusulja, Neven Papić, Klaudija Višković, Ivana Župetić, Giovanni Savini, Ljubo Barbić, and et al. 2022. "Severe West Nile Virus Neuroinvasive Disease: Clinical Characteristics, Short- and Long-Term Outcomes" Pathogens 11, no. 1: 52. https://doi.org/10.3390/pathogens11010052
APA StyleSantini, M., Haberle, S., Židovec-Lepej, S., Savić, V., Kusulja, M., Papić, N., Višković, K., Župetić, I., Savini, G., Barbić, L., Tabain, I., Kutleša, M., Krajinović, V., Potočnik-Hunjadi, T., Dvorski, E., Butigan, T., Kolaric-Sviben, G., Stevanović, V., Gorenec, L., ... Vilibić-Čavlek, T. (2022). Severe West Nile Virus Neuroinvasive Disease: Clinical Characteristics, Short- and Long-Term Outcomes. Pathogens, 11(1), 52. https://doi.org/10.3390/pathogens11010052










