Molecular Epidemiology of Sapovirus in Children Living in the Northwest Amazon Region
Abstract
:1. Introduction
2. Results
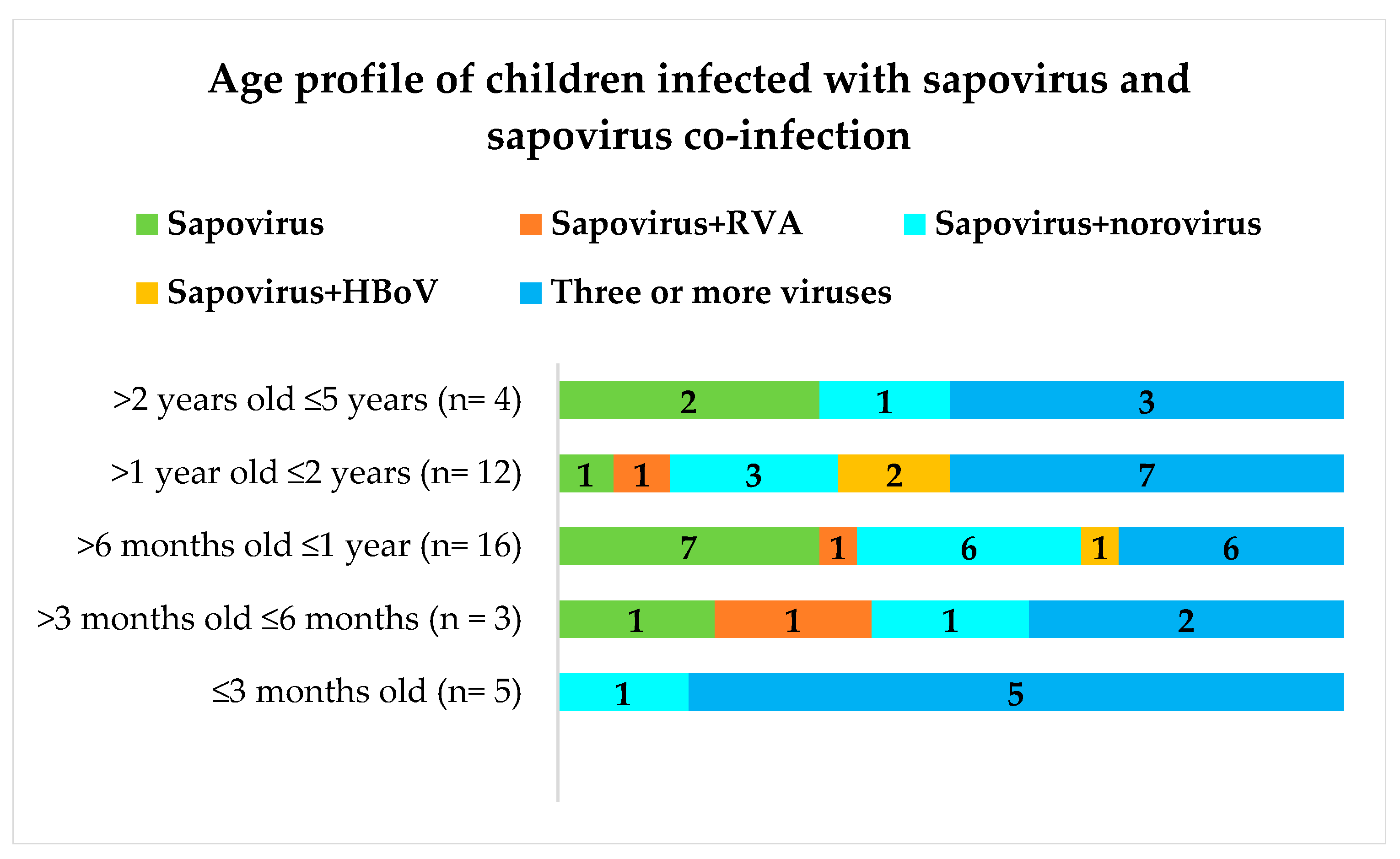
2.1. Genetically Diverse Sapoviruses Cause Acute Gastroenteritis in Children in the Amazon Region
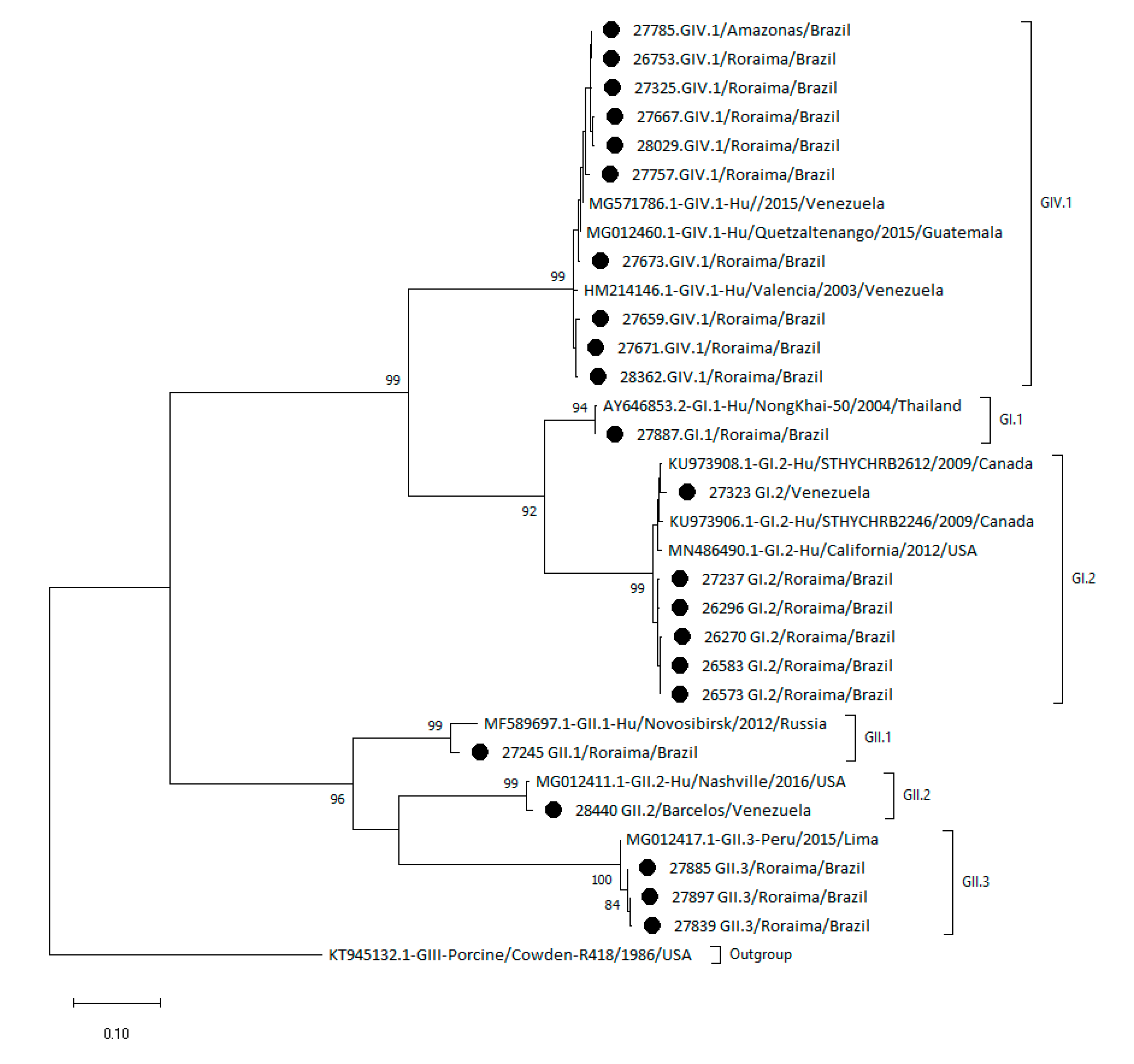
2.2. Phylogenetic Analysis Shows the Heterogeneity of Sapoviruses in the Amazon Region
2.3. No Association between Sapovirus Genotypes and Histo-Blood Group Susceptibility
3. Discussion
4. Materials and Methods
4.1. Information Regarding the Samples of This Study
4.2. Sapovirus Genotyping
4.3. Histo-Blood Group Antigen Phenotyping
4.4. Phylogenetic Tree and Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Becker-Dreps, S.; Bucardo, F.; Vinjé, J. Sapovirus: An important cause of acute gastroenteritis in children. Lancet Child Adolesc. Health 2019, 3, 758–759. [Google Scholar] [CrossRef]
- Desselberger, U. Caliciviridae Other Than Noroviruses. Viruses 2019, 11, 286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oka, T.; Wang, Q.; Katayama, K.; Saif, L.J. Comprehensive Review of Human Sapoviruses. Clin. Microbiol. Rev. 2015, 28, 32–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinjé, J.; Estes, M.K.; Esteves, P.; Green, K.Y.; Katayama, K.; Knowles, N.J.; L’Homme, Y.; Martella, V.; Vennema, H.; White, P.A. ICTV Report Consortium. ICTV Virus Taxonomy Profile: Caliciviridae. J. Gen. Virol. 2019, 100, 1469–1470. [Google Scholar] [CrossRef]
- Makhaola, K.; Moyo, S.; Kebaabetswe, L.P. Distribution and Genetic Variability of Sapoviruses in Africa. Viruses 2020, 12, 490. [Google Scholar] [CrossRef] [PubMed]
- Varela, M.F.; Rivadulla, E.; Lema, A.; Romalde, J.L. Human Sapovirus among Outpatients with Acute Gastroenteritis in Spain: A One-Year Study. Viruses 2019, 8, 144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diez-Valcarce, M.; Castro, C.J.; Marine, R.L.; Halasa, N.; Mayta, H.; Saito, M.; Tsaknaridis, L.; Pan, C.Y.; Bucardo, F.; Becker-Dreps, S.; et al. Genetic diversity of human sapovirus across the Americas. J. Clin. Virol. 2018, 104, 65–72. [Google Scholar] [CrossRef]
- Diez-Valcarce, M.; Lopez, M.R.; Lopez, B.; Morales, O.; Sagastume, M.; Cadena, L.; Kaydos-Daniels, S.; Jarquin, C.; McCracken, J.P.; Bryan, J.P. Prevalence and genetic diversity of viral gastroenteritis viroses in children younger than 5 years of age Guatemala, 2014–2015. J. Clin. Virol. 2019, 114, 6–11. [Google Scholar] [CrossRef]
- Murray, T.Y.; Nadan, S.; Page, N.A.; Taylor, M.B. Diverse sapovirus genotypes identified in children hospitalised with gastroenteritis in selected regions of South Africa. J. Clin. Virol. 2016, 76, 24–29. [Google Scholar] [CrossRef] [Green Version]
- Pang, X.L.; Lee, B.E.; Tyrrell, G.J.; Preiksaitis, J.K. Epidemiology and genotype analysis of sapovirus associated with gastroenteritis outbreaks in Alberta, Canada: 2004–2007. J. Infect. Dis. 2009, 199, 547–551. [Google Scholar] [CrossRef] [Green Version]
- Grant, L.R.; O’Brien, K.L.; Weatherholtz, R.C.; Reid, R.; Goklish, N.; Ham, M.S.; Parashar, U.; Vinjé, J. Norovirus and Sapovirus Epidemiology and Strain Characteristics among Navajo and Apache Infants. PLoS ONE 2017, 12, e0169491. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, G.J.; Mayta, H.; Pajuelo, M.J.; Neira, K.; Xiaofang, L.; Cabrera, L.; Ballard, S.B.; Crabtree, J.E.; Kelleher, D.; Cama, V.; et al. Epidemiology of Sapovirus infections in a Birth Cohort in Peru. Clin. Infect. Dis. 2018, 66, 1858–1863. [Google Scholar] [CrossRef] [Green Version]
- González, G.G.; Liprandi, F.; Ludert, J.E. Molecular epidemiology of enteric viruses in children with sporadic gastroenteritis in Valencia, Venezuela. J. Med. Virol. 2011, 83, 1972–1982. [Google Scholar] [CrossRef]
- Farkas, T.; Zhong, W.M.; Jing, Y.; Huang, P.W.; Espinosa, S.M.; Martinez, N.; Morrow, A.L.; Ruiz-Palacios, G.M.; Pickering, L.K.; Jiang, X. Genetic diversity among sapovirus. Arch. Virol. 2004, 149, 1309–1323. [Google Scholar] [CrossRef]
- Platts-Mills, J.A.; Rogawski, E.T.; Kabir, F.; Lertsethtakarn, P.; Siguas, M.; Khan, S.S.; Praharaj, I.; Murei, A.; Nshama, R.; Mujaga, B.; et al. The MAL-ED Network Investigators. Use of quantitative molecular diagnostic methods to assess the aetiology, burden, and clinical characteristics of diarrhoea in children in low-resource settings: A reanalysis of the MAL-ED cohort study. Lancet Glob. Health 2018, 6, 1309–1318. [Google Scholar] [CrossRef] [Green Version]
- Colston, J.; Olortegui, M.P.; Zaitchik, B.; Yori, P.P.; Kang, G.; Ahmed, T.; Bessong, P.; Mduma, E.; Bhutta, Z.; Shrestha, P.S.; et al. Pathogen-Specific Impacts of the 2011–2012 La Niña-Associated Floods on Enteric Infections in the MAL-ED Peru Cohort: A Comparative Interrupted Time Series Analysis. Int. J. Environ. Res. Public Health 2020, 12, 487. [Google Scholar] [CrossRef] [Green Version]
- McQuade, E.T.R.; Liu, J.; Kang, G.; Kosek, M.N.; Lima, A.A.M.; Bessong, P.O.; Samie, A.; Haque, R.; Mduma, E.R.; Shrestha, S.; et al. Protection From Natural Immunity Against Enteric Infections and Etiology-Specific Diarrhea in a Longitudinal Birth Cohort. J. Infect. Dis. 2020, 27, 1858–1868. [Google Scholar] [CrossRef]
- das Neves Costa, L.C.P.; Siqueira, J.A.M.; Portal, T.M.; Junior, E.C.S.; Linhares, A.C.; Gabbay, Y.B.; Resque, H.R. Detection and genotyping of human adenovirus and sapovirus in children with acute gastroenteritis in Belém, Pará, between 1990 and 1992: First detection of GI.7 and GV.2 sapoviruses in Brazil. Rev. Soc. Bras. Med. Trop. 2017, 50, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Portal, T.M.; Reymão, T.K.A.; Quinderé Neto, G.A.; Fiuza, M.K.D.C.; Teixeira, D.M.; Lima, I.C.G.; Sousa, E.C., Jr.; Bandeira, R.D.S.; De Deus, D.R.; Justino, M.C.A.; et al. Detection and genotyping of enteric viruses in hospitalized children with acute gastroenteritis in Belém, Brazil: Occurrence of adenovirus viremia by species F, types 40/41. J. Med. Virol. 2019, 91, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Reymão, T.K.; Hernandez, J.D.; Costa, S.T.; Sousa, M.S.; Oliveira, D.S.; Silva, L.D.; Bandeira, R.D.; Lima, I.C.; Soares, L.D.; Mascarenhas, J.D.; et al. Sapoviruses in children with acute gastroenteritis from Manaus, Amazon region, Brazil, 2010–2011. Rev. Inst. Med. Trop. Sao Paulo. 2016, 58, 81. [Google Scholar] [CrossRef] [Green Version]
- Nordgren, J.; Svensson, L. Genetic Susceptibility to Human Norovirus Infection: An Update. Viruses 2019, 11, 226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matussek, A.; Dienus, O.; Djeneba, O.; Simpore, J.; Nitiema, L.; Nordgren, J. Molecular characterization and genetic susceptibility of sapovirus in children with diarrhea in Burkina Faso. Infect. Genet. Evol. 2015, 32, 396–400. [Google Scholar] [CrossRef]
- Bucardo, F.; Carlsson, B.; Nordgren, J.; Larson, G.; Blandon, P.; Vilchez, S.; Svensson, L. Susceptibility of Children to Sapovirus Infections, Nicaragua, 2005–2006. Emerg. Infect. Dis. 2012, 18, 1875–1878. [Google Scholar] [CrossRef] [PubMed]
- Shirato-Horikoshi, H.; Ogawa, S.; Takeda, W.N.; Hansman, G.S. Binding activity of norovirus and sapovirus to histo-blood group antigens. Arch. Virol. 2007, 152, 457–461. [Google Scholar] [CrossRef]
- Olivares, A.I.O.; Leitão, G.A.L.; Pimenta, Y.C.; Cantelli, C.P.; Fumian, T.M.; Fialho, A.M.; da Silva e Mouta, S., Jr.; Delgado, I.F.; Nordgren, J.; Svensson, L.; et al. Epidemiology of enteric virus infections in children living in the Amazon region. Int. J. Infect. Dis. 2021, 108, 494–502. [Google Scholar] [CrossRef]
- Yan, H.; Yagyu, F.; Okitsu, S.; Nishio, O.; Ushijima, H. Detection of norovirus (GI, GII), Sapovirus and astrovirus in fecal samples using reverse transcription single-round multiplex PCR. J. Virol. Methods 2003, 114, 37–44. [Google Scholar] [CrossRef]
- Okada, M.; Yamashita, Y.; Oseto, M.; Shinozaki, K. The detection of human sapoviruses with universal and genogroup-specific primers. Arch. Virol. 2006, 151, 2503–2509. [Google Scholar] [CrossRef]
- Oka, T.; Yamamoto, S.P.; Iritani, N.; Sato, S.; Tatsumi, C.; Mita, T.; Yahiro, S.; Shibata, S.; Wu, F.T.; Takagi, H. Polymerase chain reaction primer set for the detection of genetically diverse human sapovirus. Arch. Virol. 2020, 165, 2335–2340. [Google Scholar] [CrossRef]
- Moraes, M.T.B.; Olivares, A.I.O.; Fialho, A.M.; Malta, F.C.; Mouta Junior, S.S.; Bispo, R.S.; Velloso, A.J.; Leitão, G.A.L.; Cantelli, C.P.; Nordgren, J.; et al. Phenotyping of Lewis and secretor HBGA from saliva and detection of new FUT2 gene SNPs from young children from the Amazon presenting acute gastroenteritis and respiratory infection. Infect. Genet. Evol. 2019, 70, 61–70. [Google Scholar] [CrossRef]
- Szumilas, M. Explaining odds ratios as conditional risk ratios. J. Clin. Epidemiol. 2018, 97, 122–125. [Google Scholar]
- Li, W.; Dong, S.; Xu, J.; Zhou, X.; Han, J.; Xie, Z.; Gong, Q.; Peng, H.; Zhou, C.; Lin, M. Viral metagenomics reveals sapoviruses of different genogroups in stool samples from children with acute gastroenteritis in Jiangsu, China. Arch. Virol. 2020, 165, 955–958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hebbelstrup, B.J.; Jokelainen, P.; de Nielsen, A.C.Y.; Franck, K.T.; Holm, D.R.; Schønning, K.; Peterson, A.M.; Frogfelt, K.A. Children Attending Day Care Centers are a Year-round Reservoir of Gastrointestinal Viruses. Sci. Rep. 2019, 9, 3286. [Google Scholar] [CrossRef]
- Oka, T.; Mori, K.; Iritane, N.; Harada, S.; Ueki, Y.; Lizuka, S.; Mise, K.; Murakami, K.; Wakita, T.; Katayama, K. Human sapovirus classification based on complete capsid nucleotide sequences. Arch. Virol. 2011, 157, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Cilli, A.; Luchs, A.; Leal, E.; Gill, D.; Milagres, F.A.P.; Komninakis, S.V.; Brustulin, R.; Teles, M.D.A.R.; Lobato, M.C.A.B.S.; Chagas, R.T.D.; et al. Human sapovirus GI.2 and GI.3 from children with acute gastroenteritis in northern Brazil. Mem. Inst. Oswaldo Cruz. 2019, 114, e180574. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| PCR Amplification Strategy | Primer (Forward-F, Reverse-R, Round-1 1, Round-2 1, Multiplex) | Primer Sequence 5′–3′; (VP1 Nucleotide Position) | Annealing Temperature/Number of Cycles | Round Number | Reference |
|---|---|---|---|---|---|
| Initial | SLV5317-F | CTCGCCACCTACRAWGCBTGGTT | 50 °C/40 | 1 | [26] |
| SLV5749-R | CGGRCYTCAAAVSTACCBCCCCA | ||||
| Second | 1 1 SV-F13-F | GAYYWGGCYCTCGCYACCTAC | 2 | [27] | |
| 1 1 SV-R13-R | GGTGANAYNCCATTKTCCAT | ||||
| 2 1 SV-F22-F | SMWAWTAGTGTTTGARATG | ||||
| 2 1 SV-R2-R | GWGGGRTCAACMCCWGGTGG | ||||
| Third | 1 1 SV-F14-F | GAACAAGCTGTGGCATGCTAC | 2 | [27] | |
| 1 1 SV-R14-R | GGTGAGMMYCCATTCTCCAT | ||||
| 2 1 SV-F22-F | SMWAWTAGTGTTTGARATG | ||||
| 2 1 SV-R2-R | GWGGGRTCAACMCCWGGTGG | ||||
| Fourth | Multiplex-SV-F13-F | --- | 1 | [27] | |
| SV-F14-F | --- | ||||
| SV-G1-R-R | CCCBGGTGGKAYGACAGAAG | ||||
| SV-G2-R-R | CCANCCAGCAAACATNGCRCT | ||||
| SV-G4-R-R | GCGTAGCAGATCCCAGATAA | ||||
| SV-G5-R-R | TTGGAGGWTGTTGCTCCTGTG | ||||
| Fifth | SaVPanF-F | CAGTTCWACTGGSTNAAGGC (5047-5066) | 1 | This study | |
| SaVPanR-R | GCATCAACRAANGCGTGNGG (5816-5835) | ||||
| Sixth | HuSaV-5159F-F | TAGTGTTTGARATGGARGG | 53 °C/50 | 1 | [28] |
| HuSaV-5498R-R | CCCCANCCNGCVHACAT | ||||
| Seventh | Multiplex-SaV 1245Rfwd-F | TAGTGTTTGARATGGAGGG | |||
| SV-G1-R-R | --- | ||||
| SV-G2-R-R | --- | ||||
| SV-G4-R-R | --- | ||||
| SV-G5-R-R | --- | ||||
| Eighth | HuSaV-F1-F | GGCHCTYGCCACCTAYAA YG | |||
| HuSaV-5498R-R | --- |
| Genotype (n) | Child Age 1 (n) | Clinical Aspects 2 | HBGA Status 3 |
|---|---|---|---|
| GIV.1 (10) | >3 months ≤6 months (3) | Mucus in feces, fever, abdominal pain, dehydration | Lea+Leb−, Se−; Lea+Leb+, Se+ |
| >6 months ≤1 year (2) | Mucus in feces, fever, abdominal pain; mucus in feces, fever, vomit | Lea−Leb−, Se−; Lea−Leb+, Se+ | |
| >1 year ≤2 years (3) | Mucus in feces, fever, vomit, abdominal pain | Lea−Leb+, Se+; Lea−Leb−, Se+; Lea−Leb−, Se− | |
| >2 years ≤5 years (2) | Mucus and blood in feces, vomit, abdominal pain; fever, abdominal pain | Lea+Leb+, Se+; Lea−Leb+, Se+ | |
| GI.2 (7) | >6 months ≤1 year (3) | Coryza, couch; mucus in feces, vomit, abdominal pain; mucus in feces, fever, vomit, abdominal pain | Lea+Leb+, Se+; Lea−Leb+, Se+ |
| >1 year ≤2 years (4) | Mucus in feces, fever, vomit, abdominal pain; mucus in feces, fever, abdominal pain; fever, couch | Lea−Leb+, Se+; Lea−Leb−, Se+ | |
| GII.3 (5) | ≤3 months (1) | Fever, abdominal pain | Lea+Leb+, Se+ |
| >6 months ≤1 year (3) | Fever, vomit, cough; fever, abdominal pain, cough; mucus in feces, fever, abdominal pain | Lea+Leb+, Se+ | |
| >2 years ≤5 years (1) | Mucus in feces, fever, vomit, abdominal pain | Lea−Leb+, Se+ | |
| GI.1 (3) | >6 months ≤1 year (2) | Fever, abdominal pain, cough; mucus in feces, fever, abdominal pain | Lea+Leb−, Se−; Lea+Leb−, Se+ |
| >2 years ≤5 years (1) | Mucus and blood in feces, fever, vomit, abdominal pain | Lea+Leb−, Se− | |
| GII.1 (1) | >6 months ≤1 year | Mucus in feces, fever, abdominal pain | Lea+Leb+, Se+ |
| GII.2 (1) | >1 year ≤2 years | Mucus in feces, fever, vomit, abdominal pain | Lea+Leb+, Se+ |
| GII.5 (1) | ≤3 months | Not available | Lea−Leb+, Se+ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moraes, M.T.B.d.; Leitão, G.A.A.; Olivares, A.I.O.; Xavier, M.d.P.T.P.; Bispo, R.d.S.; Sharma, S.; Leite, J.P.G.; Svensson, L.; Nordgren, J. Molecular Epidemiology of Sapovirus in Children Living in the Northwest Amazon Region. Pathogens 2021, 10, 965. https://doi.org/10.3390/pathogens10080965
Moraes MTBd, Leitão GAA, Olivares AIO, Xavier MdPTP, Bispo RdS, Sharma S, Leite JPG, Svensson L, Nordgren J. Molecular Epidemiology of Sapovirus in Children Living in the Northwest Amazon Region. Pathogens. 2021; 10(8):965. https://doi.org/10.3390/pathogens10080965
Chicago/Turabian StyleMoraes, Marcia Terezinha Baroni de, Gabriel Azevedo Alves Leitão, Alberto Ignácio Olivares Olivares, Maria da Penha Trindade Pinheiro Xavier, Romanul de Souza Bispo, Sumit Sharma, José Paulo Gagliardi Leite, Lennart Svensson, and Johan Nordgren. 2021. "Molecular Epidemiology of Sapovirus in Children Living in the Northwest Amazon Region" Pathogens 10, no. 8: 965. https://doi.org/10.3390/pathogens10080965
APA StyleMoraes, M. T. B. d., Leitão, G. A. A., Olivares, A. I. O., Xavier, M. d. P. T. P., Bispo, R. d. S., Sharma, S., Leite, J. P. G., Svensson, L., & Nordgren, J. (2021). Molecular Epidemiology of Sapovirus in Children Living in the Northwest Amazon Region. Pathogens, 10(8), 965. https://doi.org/10.3390/pathogens10080965







