No Evidence Known Viruses Play a Role in the Pathogenesis of Onchocerciasis-Associated Epilepsy. An Explorative Metagenomic Case-Control Study
Abstract
1. Introduction
2. Results
2.1. Characteristics of the Cases and Controls
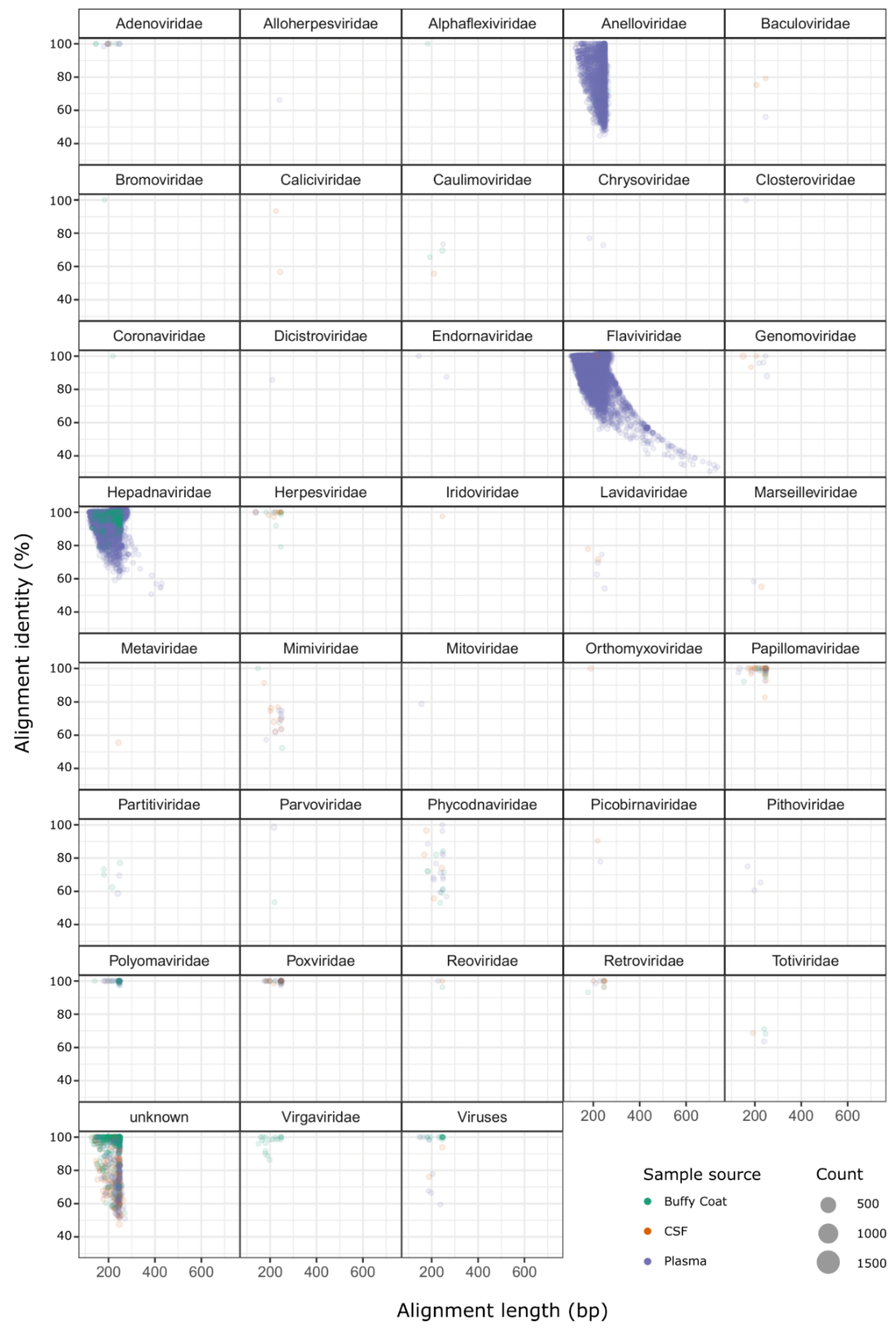
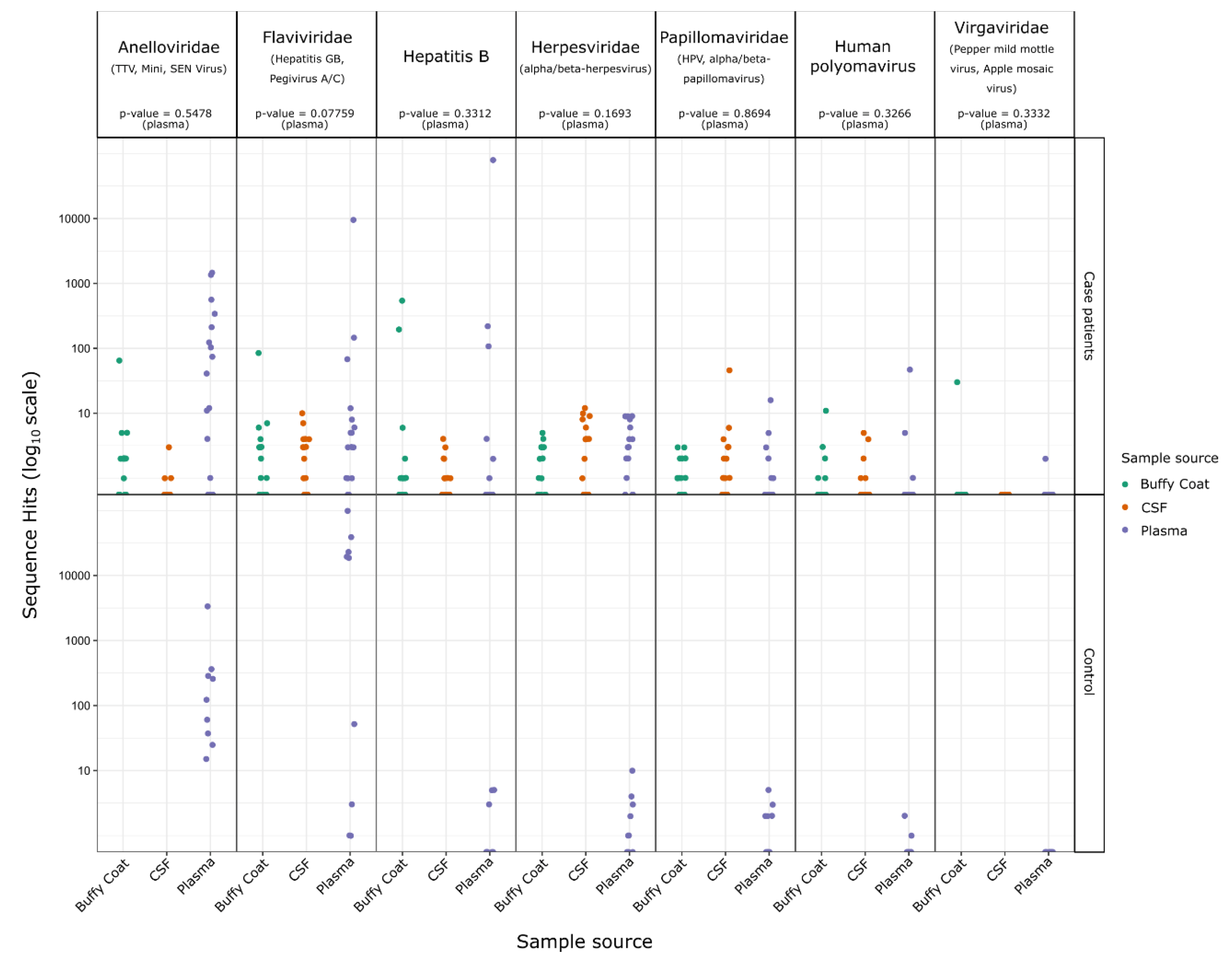
2.2. Identification of Sequences from Known Viruses
3. Discussion
4. Materials and Methods
4.1. Study Design and Procedures
- Does the person have major difficulties expressing themselves?
- Does the person get lost in the village?
- Does the person understand questions?
4.2. Testing for Onchocerciasis
4.3. Metagenomic Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Colebunders, R.; Fodjo, J.N.S.; Hopkins, A.; Hotterbeekx, A.; Lakwo, T.L.; Kalinga, A.; Logora, M.Y.; Basáñez, M.-G. From river blindness to river epilepsy: Implications for onchocerciasis elimination programmes. PLoS Negl. Trop. Dis. 2019, 13, e0007407. [Google Scholar] [CrossRef]
- Colebunders, R.N.A.; Menon, S.; Newton, C.R.; Hotterbeekx, A.; Preux, P.M.; Hopkins, A.; Vaillant, M.; SieweFodjo, J.N. Onchocerca volvulus and epilepsy: A comprehensive review using the Bradford Hill criteria for causation. PLoS Neglect. Trop. Dis. 2021, 15, e0008965. [Google Scholar] [CrossRef]
- Chesnais, C.B.N.A.; Zoung-Bissek, A.C.; Tatah, G.Y.; Nana-Djeunga, H.C.; Kamgno, J.; Colebunders, R.; Boussinesq, M. First evidence by a cohort study in Cameroon that onchocerciasis does induce epilepsy. In Proceedings of the 1st International Workshop on Onchocer-Ciasis Associated Epilepsy, Antwerp, Belgium, 12–14 October 2017. Abstract 15. [Google Scholar]
- Chesnais, C.B.; Bizet, C.; Campillo, J.T.; Njamnshi, W.Y.; Bopda, J.; Nwane, P.; Pion, S.D.; Njamnshi, A.K.; Boussinesq, M. A Second Population-Based Cohort Study in Cameroon Confirms the Temporal Relationship Between Onchocerciasis and Epilepsy. Open Forum Infect. Dis. 2020, 7, ofaa206. [Google Scholar] [CrossRef] [PubMed]
- Hotterbeekx, A.; Raimon, S.; Abd-Elfarag, G.; Carter, J.Y.; Sebit, W.; Suliman, A.; Fodjo, J.N.S.; De Witte, P.; Logora, M.Y.; Colebunders, R.; et al. Onchocerca volvulus is not detected in the cerebrospinal fluid of persons with onchocerciasis-associated epilepsy. Int. J. Infect. Dis. 2020, 91, 119–123. [Google Scholar] [CrossRef]
- Hotterbeekx, A.; Lammens, M.; Idro, R.; Akun, P.R.; Lukande, R.; Akena, G.; Nath, A.; Taylor, J.; Olwa, F.; Kumar-Singh, S.; et al. Neuroinflammation and Not Tauopathy Is a Predominant Pathological Signature of Nodding Syndrome. J. Neuropathol. Exp. Neurol. 2019, 78, 1049–1058. [Google Scholar] [CrossRef] [PubMed]
- Colebunders, R.; Hendy, A.; Nanyunja, M.; Wamala, J.F.; van Oijen, M. Nodding syndrome—A new hypothesis and new direction for research. Int. J. Infect. Dis. 2014, 27, 74–77. [Google Scholar] [CrossRef]
- Colebunders, R.; Mandro, M.; Mokili, J.L.; Mucinya, G.; Mambandu, G.; Pfarr, K.; Reiter-Owona, I.; Hoerauf, A.; Tepage, F.; Levick, B.; et al. Risk factors for epilepsy in Bas-Uélé Province, Democratic Republic of the Congo: A case–control study. Int. J. Infect. Dis. 2016, 49, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Föger, K.; Gora-Stahlberg, G.; Sejvar, J.; Ovuga, E.; Jilek-Aall, L.; Schmutzhard, E.; Kaiser, C.; Winkler, A.S. Nakalanga Syndrome: Clinical Characteristics, Potential Causes, and Its Relationship with Recently Described Nodding Syndrome. PLoS Neglect. Trop. Dis. 2017, 11, e0005201. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Terunuma, H.; Handema, R.; Sakamoto, M.; Kitamura, T.; Ito, M.; Akahane, Y. Higher prevalence and viral load of TT virus in saliva than in the corresponding serum: Another possible transmission route and replication site of TT virus. J. Med. Virol. 2000, 62, 531–537. [Google Scholar] [CrossRef]
- Goto, K.; Sugiyama, K.; Ando, T.; Mizutani, F.; Terabe, K.; Tanaka, K.; Nishiyama, M.; Wada, Y. Detection Rates of TT Virus DNA in Serum of Umbilical Cord Blood, Breast Milk and Saliva. Tohoku J. Exp. Med. 2000, 191, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, K.; Goto, K.; Ando, T.; Mizutani, F.; Terabe, K.; Kawabe, Y.; Yokoyama, T.; Wada, Y. Prevalence of TTV DNA among children with a histo-ry of transfusion or liver disease. J. Med. Virol. 2000, 60, 172–176. [Google Scholar] [CrossRef]
- Sugiyama, K.; Goto, K.; Ando, T.; Mizutani, F.; Terabe, K.; Yokoyama, T.; Wada, Y. TT Virus Infection in Japanese Children: Isolates from Genotype 1 are Overrepresented in Patients with Hepatic Dysfunction of Unknown Etiology. Tohoku J. Exp. Med. 2000, 191, 233–239. [Google Scholar] [CrossRef][Green Version]
- Saback, F.L.; Palmer, T.E.; Sabino, R.R.; Carvalho, S.M.; Amorim, L.M.; Gaspar, A.M.; Oliveria, M.L.A.; Yoshida, C.F.T.; Niel, C. Infection with Hepatitis A and TT Viruses and Socioeconomic Status in Rio de Janeiro, Brazil. Scand. J. Infect. Dis. 2001, 33, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Saback, F.L.; Gomes, S.A.; Niel, C. High frequency of mixed TT virus infections in healthy adults and children detected by a simplified heteroduplex mobility assay. J. Virol. Methods 2002, 101, 117–125. [Google Scholar] [CrossRef]
- Kaczorowska, J.; van der Hoek, L. Human anelloviruses: Diverse, omnipresent and commensal members of the virome. FEMS Microbiol. Rev. 2020, 44, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Davidson, F.; Macdonald, D.; Mokili, J.L.K.; Prescott, L.E.; Graham, S.; Simmonds, P. Early Acquisition of TT Virus (TTV) in an Area Endemic for TTV Infection. J. Infect. Dis. 1999, 179, 1070–1076. [Google Scholar] [CrossRef]
- Phan, T.; Kapusinszky, B.; Wang, C.; Rose, R.K.; Lipton, H.L.; Delwart, E.L. The Fecal Viral Flora of Wild Rodents. PLoS Pathog. 2011, 7, e1002218. [Google Scholar] [CrossRef]
- Ng, T.F.F.; Marine, R.; Wang, C.; Simmonds, P.; Kapusinszky, B.; Bodhidatta, L.; Oderinde, B.S.; Wommack, K.E.; Delwart, E. High Variety of Known and New RNA and DNA Viruses of Diverse Origins in Untreated Sewage. J. Virol. 2012, 86, 12161–12175. [Google Scholar] [CrossRef]
- Fernandez-Cassi, X.; Timoneda, N.; Gonzales-Gustavson, E.; Abril, J.; Bofill-Mas, S.; Girones, R. A metagenomic assessment of viral contamination on fresh parsley plants irrigated with fecally tainted river water. Int. J. Food Microbiol. 2017, 257, 80–90. [Google Scholar] [CrossRef]
- Aguado-García, Y.; Taboada, B.; Morán, P.; Gutierrez, X.J.R.; Serrano-Vázquez, A.; Iša, P.; Rojas-Velázquez, L.; Pérez-Juárez, H.; López, S.; Torres, J.; et al. Tobamoviruses can be frequently present in the oropharynx and gut of infants during their first year of life. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Guo, L.; Hua, X.; Zhang, W.; Yang, S.; Shen, Q.; Hu, H.; Li, J.; Liu, Z.; Wang, X.; Wang, H.; et al. Viral metagenomics analysis of feces from coronary heart disease patients reveals the genetic diversity of the Microviridae. Virol. Sin. 2017, 32, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Tisza, M.J.; Pastrana, D.V.; Welch, N.L.; Stewart, B.; Peretti, A.; Starrett, G.; Pang, Y.-Y.S.; Krishnamurthy, S.R.; Pesavento, P.A.; McDermott, D.H.; et al. Discovery of several thousand highly diverse circular DNA viruses. eLife 2020, 9, e51971. [Google Scholar] [CrossRef] [PubMed]
- Mokili, J.L.; Rohwer, F.; Dutilh, B.E. Metagenomics and future perspectives in virus discovery. Curr. Opin. Virol. 2012, 2, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Mokili, J.L.; Dutilh, B.E.; Lim, Y.W.; Schneider, B.S.; Taylor, T.; Haynes, M.R.; Metzgar, D.; Myers, C.A.; Blair, P.J.; Nosrat, B.; et al. Identification of a Novel Human Papillomavirus by Metagenomic Analysis of Samples from Patients with Febrile Respiratory Illness. PLoS ONE 2013, 8, e58404. [Google Scholar] [CrossRef] [PubMed]
- Dutilh, B.E.; Cassman, N.; McNair, K.; Sanchez, S.E.; Silva, G.G.Z.; Boling, L.; Barr, J.; Speth, D.R.; Seguritan, V.; Aziz, R.; et al. A highly abundant bacteriophage discovered in the unknown sequences of human faecal metagenomes. Nat. Commun. 2014, 5, 4498. [Google Scholar] [CrossRef]
- Benedek, G.; El Latif, M.A.; Miller, K.; Rivkin, M.; Lasu, A.A.R.; Riek, L.P.; Lako, R.; Edvardson, S.; Alon, S.-A.; Galun, E.; et al. Protection or susceptibility to devastating childhood epilepsy: Nodding Syndrome associates with immunogenetic fingerprints in the HLA binding groove. PLoS Neglect. Trop. Dis. 2020, 14, e0008436. [Google Scholar] [CrossRef]
- Colebunders, R.; Tepage, F.; Rood, E.; Mandro, M.; Abatih, E.N.; Musinya, G.; Mambandu, G.; Kabeya, J.; Komba, M.; Levick, B.; et al. Prevalence of River Epilepsy in the Orientale Province in the Democratic Republic of the Congo. PLoS Neglect. Trop. Dis. 2016, 10, e0004478. [Google Scholar] [CrossRef]
- De Vries, M.; Deijs, M.; Canuti, M.; Van Schaik, B.D.C.; Faria, N.R.; Van De Garde, M.D.B.; Jachimowski, L.C.M.; Jebbink, M.F.; Jakobs, M.; Luyf, A.C.M.; et al. A Sensitive Assay for Virus Discovery in Respiratory Clinical Samples. PLoS ONE 2011, 6, e16118. [Google Scholar] [CrossRef]
- Boom, R.; Sol, C.J.; Salimans, M.M.; Jansen, C.L.; Dillen, P.M.W.-V.; van der Noordaa, J. Rapid and simple method for purification of nucleic acids. J. Clin. Microbiol. 1990, 28, 495–503. [Google Scholar] [CrossRef]
- Endoh, D. Species-independent detection of RNA virus by representational difference analysis using non-ribosomal hexanucleotides for reverse transcription. Nucleic Acids Res. 2005, 33, e65. [Google Scholar] [CrossRef]
- Cotten, M.; Munnink, B.O.; Canuti, M.; Deijs, M.; Watson, S.J.; Kellam, P.; Van Der Hoek, L. Full Genome Virus Detection in Fecal Samples Using Sensitive Nucleic Acid Preparation, Deep Sequencing, and a Novel Iterative Sequence Classification Algorithm. PLoS ONE 2014, 9, e93269. [Google Scholar] [CrossRef] [PubMed]
- Steinegger, M.; Söding, J. MMseqs2 enables sensitive protein sequence searching for the analysis of massive data sets. Nat. Biotechnol. 2017, 35, 1026–1028. [Google Scholar] [CrossRef]
- Vassetzky, N.S.; Kramerov, D.A. SINEBase: A database and tool for SINE analysis. Nucleic Acids Res. 2012, 41, D83–D89. [Google Scholar] [CrossRef]
- Schneider, V.A.; Graves-Lindsay, T.; Howe, K.; Bouk, N.; Chen, H.-C.; Kitts, P.A.; Murphy, T.D.; Pruitt, K.; Thibaud-Nissen, F.; Albracht, D.; et al. Evaluation of GRCh38 and de novo haploid genome assemblies demonstrates the enduring quality of the reference assembly. Genome Res. 2017, 27, 849–864. [Google Scholar] [CrossRef]
- The UniProt Consortium. UniProt: The universal protein knowledgebase in 2021. Nucleic Acids Res. 2021, 49, D480–D489. [Google Scholar] [CrossRef]
- O’Leary, N.A.; Wright, M.W.; Brister, J.R.; Ciufo, S.; Haddad, D.; McVeigh, R.; Rajput, B.; Robbertse, B.; Smith-White, B.; Ako-Adjei, D.; et al. Reference sequence (RefSeq) database at NCBI: Current status, taxonomic expansion, and functional annotation. Nucleic Acids Res. 2016, 44, D733–D745. [Google Scholar] [CrossRef] [PubMed]
- Cochrane, G.R.; Galperin, M.Y. The 2010 Nucleic Acids Research Database Issue and online Database Collection: A community of data resources. Nucleic Acids Res. 2009, 38, D1–D4. [Google Scholar] [CrossRef] [PubMed]
| Clinical Characteristics | Cases (n = 16) | Controls (n = 9) | p-Value |
|---|---|---|---|
| Age, median (IQR) | 14.5 (11.7–19.7) | 16.0 (10.5–24.5) | 0.820 |
| Male n (%) | 7 (43.7) | 3 (42.9) | 0.778 |
| Body weight (kg) mean (S.D.) | 39.0 (14.0) | 40.7 (14.9) * | 1.000 |
| Height (cm), mean (S.D.) | 145.0 (19.0) | 150.5 (16.0) * | 0.673 |
| Body mass index, mean (S.D.) | 17.7 (3.5) | 17.3 (2.7) * | 0.923 |
| Age of epilepsy onset, median (IQR) | 10.0 (9.0–13.0) | NA | NA |
| Only generalized tonic–clonic seizures n (%) | 10 (62.3) | NA | NA |
| Generalized tonic–clonic seizures and absences n (%) | 5 (31.2) | NA | NA |
| Only absences n (%) | 1 (6.2) | NA | NA |
| Mental disorder n (%) | 8 (50) | NA | NA |
| Stunting n (%) | 3 (18.7) | NA | NA |
| Onchocerciasis skin lesion (“leopard skin legions” both legs) n (%) | 3 (18.75) | NA | NA |
| Itching n (%) | 10 (62.5) | 1 (11.1) * | 0.061 |
| Onchocerciasis nodules | 2 (12.5) | ||
| Skin snip O. volvulus PCR positive n (%) | 14 (77.8) | 1/5 (20) | 0.009 |
| O. volvulus IgG4, n (%) | 10/15 (66.6) | NA | NA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roach, M.; Cantu, A.; Vieri, M.K.; Cotten, M.; Kellam, P.; Phan, M.; Hoek, L.v.d.; Mandro, M.; Tepage, F.; Mambandu, G.; et al. No Evidence Known Viruses Play a Role in the Pathogenesis of Onchocerciasis-Associated Epilepsy. An Explorative Metagenomic Case-Control Study. Pathogens 2021, 10, 787. https://doi.org/10.3390/pathogens10070787
Roach M, Cantu A, Vieri MK, Cotten M, Kellam P, Phan M, Hoek Lvd, Mandro M, Tepage F, Mambandu G, et al. No Evidence Known Viruses Play a Role in the Pathogenesis of Onchocerciasis-Associated Epilepsy. An Explorative Metagenomic Case-Control Study. Pathogens. 2021; 10(7):787. https://doi.org/10.3390/pathogens10070787
Chicago/Turabian StyleRoach, Michael, Adrian Cantu, Melissa Krizia Vieri, Matthew Cotten, Paul Kellam, My Phan, Lia van der Hoek, Michel Mandro, Floribert Tepage, Germain Mambandu, and et al. 2021. "No Evidence Known Viruses Play a Role in the Pathogenesis of Onchocerciasis-Associated Epilepsy. An Explorative Metagenomic Case-Control Study" Pathogens 10, no. 7: 787. https://doi.org/10.3390/pathogens10070787
APA StyleRoach, M., Cantu, A., Vieri, M. K., Cotten, M., Kellam, P., Phan, M., Hoek, L. v. d., Mandro, M., Tepage, F., Mambandu, G., Musinya, G., Laudisoit, A., Colebunders, R., Edwards, R., & Mokili, J. L. (2021). No Evidence Known Viruses Play a Role in the Pathogenesis of Onchocerciasis-Associated Epilepsy. An Explorative Metagenomic Case-Control Study. Pathogens, 10(7), 787. https://doi.org/10.3390/pathogens10070787








