Performance of the LIAISON® SARS-CoV-2 Antigen Assay vs. SARS-CoV-2-RT-PCR
Abstract
1. Introduction
2. Results
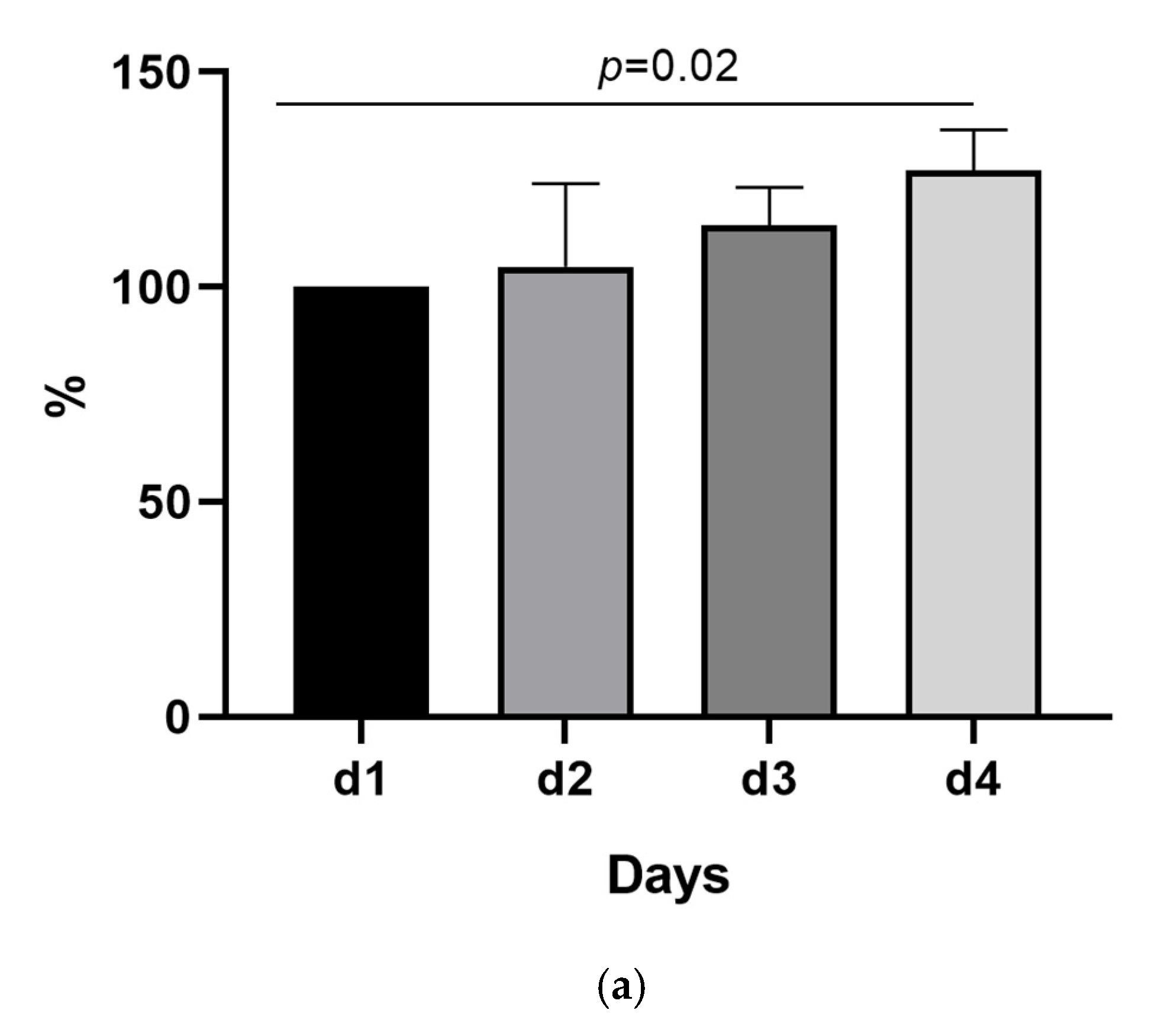
2.1. The SARS-CoV-2 Nucleocapsid Antigen Concentration Remains Stable over 3 Days
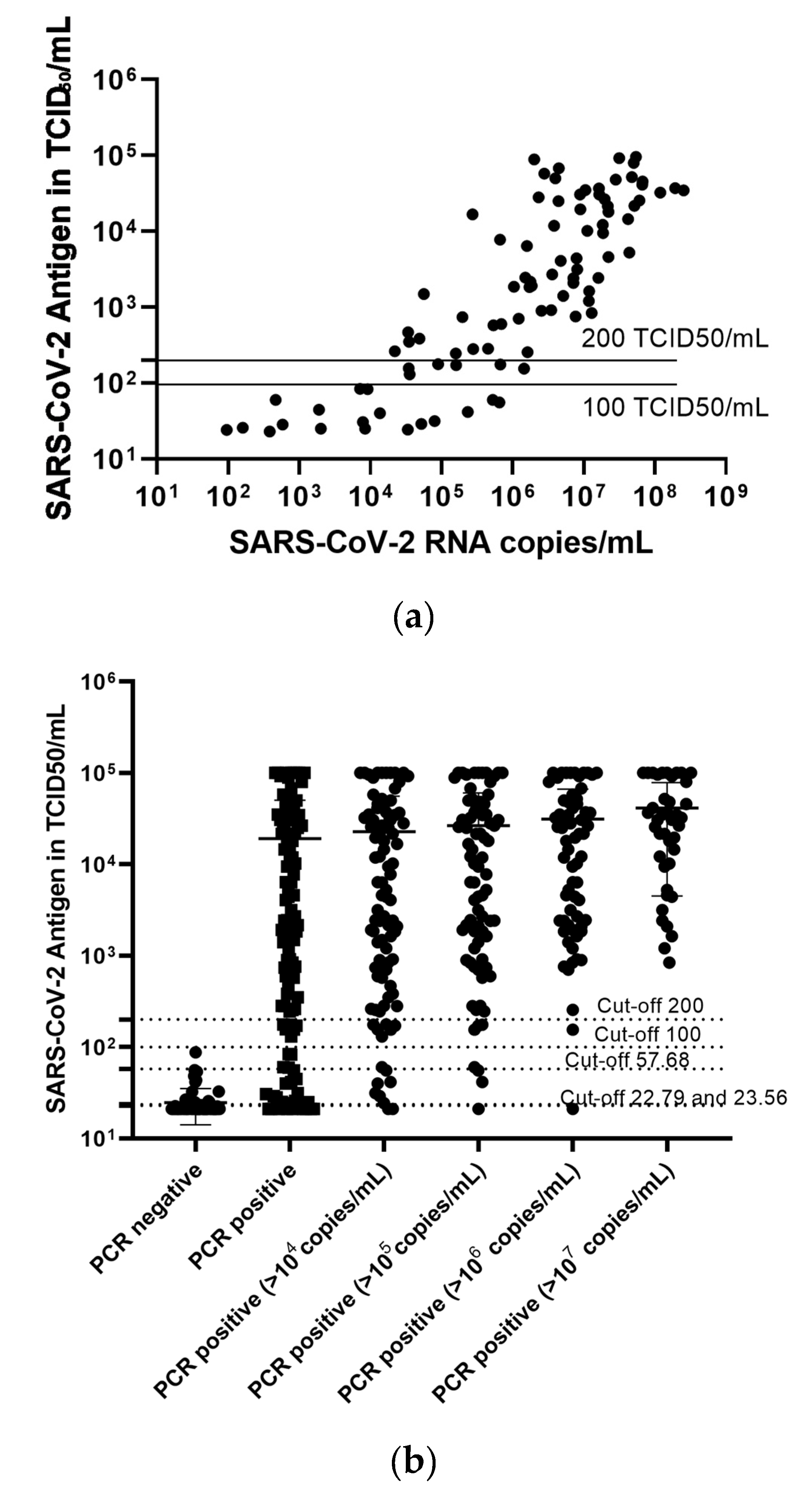
2.2. A Third of the Samples Was Falsely Classified as Negative Using the 200 TCID50/mL as Cut-Off
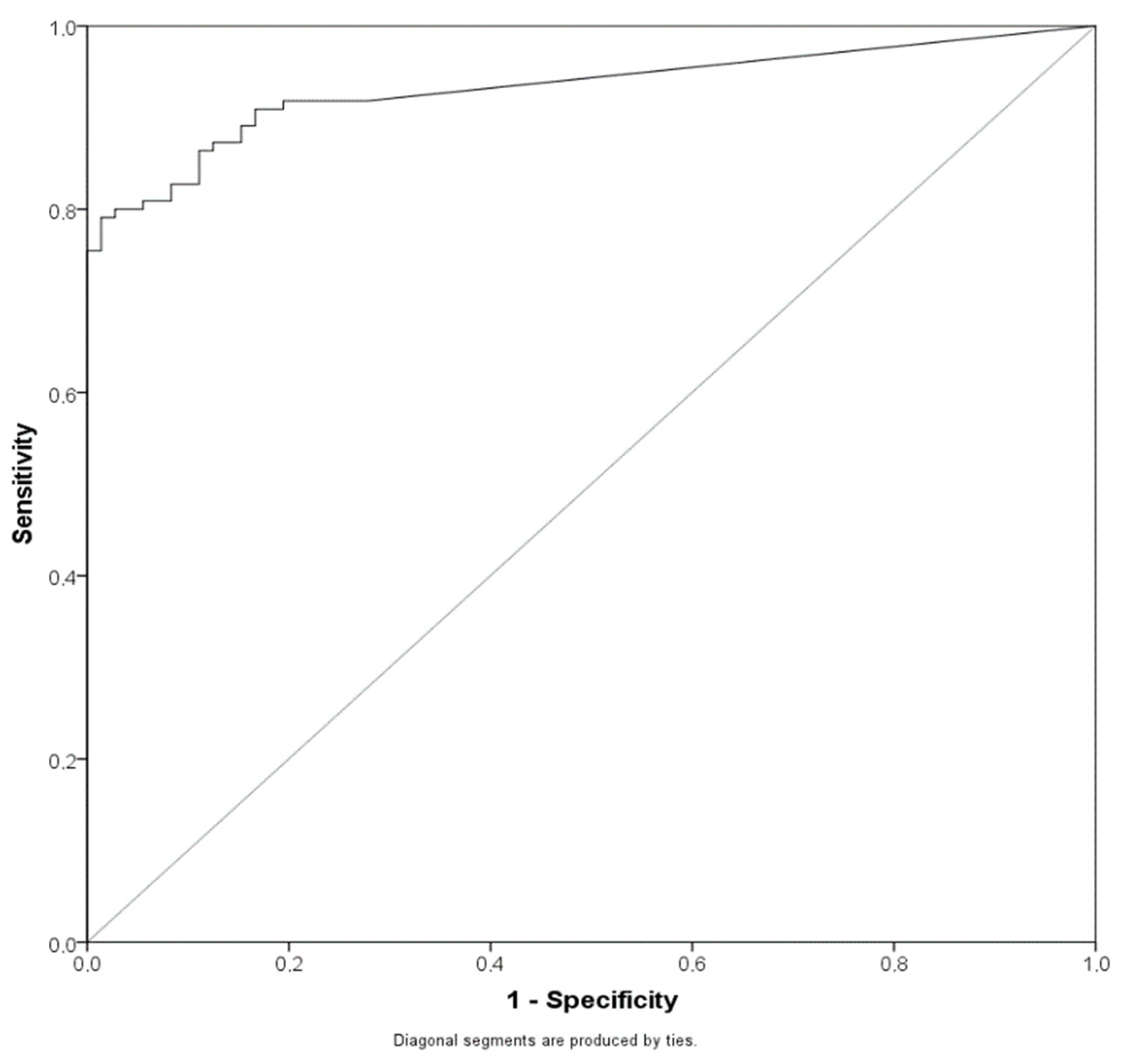
2.3. Lowering the Cut-Off for the Antigen Assay Can Significantly Increase the Sensitivity of the Method
2.4. The LIAISON® SARS-CoV-2 Antigen Assay Can Reliably Detect Samples with High SARS-CoV-2 Viral Load, While the Test Cannot Reliably Differentiate between Negative and Low Positive Samples
2.5. Testing Other Materials by LIAISON® SARS-CoV-2 Antigen Assay: Possible but Suboptimal
3. Discussion
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, D.; Ju, C.; Han, C.; Shi, R.; Chen, X.; Duan, D.; Yan, J.; Yan, X. Nanozyme chemiluminescence paper test for rapid and sensitive detection of SARS-CoV-2 antigen. Biosens. Bioelectron. 2020, 173, 112817. [Google Scholar] [CrossRef] [PubMed]
- WHO. Laboratory Testing for Coronavirus Disease 2019 (COVID-19) in Suspected Human Cases: Interim Guidance. 2 March 2020. Available online: https://apps.who.int/iris/handle/10665/331329 (accessed on 23 March 2021).
- Corman, V.M.; Haage, V.C.; Bleicker, T.; Schmidt, M.L.; Mühlemann, B.; Zuchowski, M.; Jo, W.K.; Tscheak, P.; Möncke-Buchner, E.; Müller, M.A.; et al. Comparison of seven commercial SARS-CoV-2 rapid point-of-care antigen tests: A single-centre laboratory evaluation study. Lancet Microbe 2021. [Google Scholar] [CrossRef]
- Ruopp, M.D.; Perkins, N.J.; Whitcomb, B.W.; Schisterman, E.F. Youden Index and optimal cut-point estimated from observations affected by a lower limit of detection. Biom. J. J. Math. Methods Biosci. 2008, 50, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Brümmer, L.E.; Katzenschlager, S.; Gaeddert, M.; Erdmann, C.; Schmitz, S.; Bota, M.; Grilli, M.; Larmann, J.; Weigand, M.A.; Pollock, N.R.; et al. The accuracy of novel antigen rapid diagnostics for SARS-CoV-2: A living systematic review and meta-analysis. medRxiv 2021. [Google Scholar] [CrossRef]
- Anastasiou, O.E.; Holtkamp, C.; Schäfer, M.; Schön, F.; Eis-Hübinger, A.M.; Krumbholz, A. Fast detection of SARS-CoV-2 RNA directly from respiratory samples using a loop-mediated isothermal amplification (LAMP) test. Viruses 2021, 13, 801. [Google Scholar] [CrossRef] [PubMed]
- Dao Thi, V.L.; Herbst, K.; Boerner, K.; Meurer, M.; Kremer, L.P.; Kirrmaier, D.; Freistaedter, A.; Papagiannidis, D.; Galmozzi, C.; Stanifer, M.L.; et al. A colorimetric RT-LAMP assay and LAMP-sequencing for detecting SARS-CoV-2 RNA in clinical samples. Sci. Transl. Med. 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- WHO. Antigen-Detection in the Diagnosis of SARS-CoV-2 Infection Using Rapid Immunoassays: Interim Guidance. 11 September 2020. Available online: https://apps.who.int/iris/handle/10665/334253 (accessed on 24 March 2021).
- Aoki, K.; Nagasawa, T.; Ishii, Y.; Yagi, S.; Okuma, S.; Kashiwagi, K.; Maeda, T.; Miyazaki, T.; Yoshizawa, S.; Tateda, K. Clinical validation of quantitative SARS-CoV-2 antigen assays to estimate SARS-CoV-2 viral loads in nasopharyngeal swabs. J. Infect. Chemother. 2021, 27, 613–616. [Google Scholar] [CrossRef] [PubMed]
- Wölfel, R.; Corman, V.M.; Guggemos, W.; Seilmaier, M.; Zange, S.; Müller, M.A.; Niemeyer, D.; Jones, T.C.; Vollmar, P.; Rothe, C.; et al. Virological assessment of hospitalized patients with COVID-2019. Nature 2020, 581, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Fung, J.; Lai, C.L.; Young, J.; Wong, D.K.; Yuen, J.; Seto, W.K.; Yuen, M.F. Stability of hepatitis B surface antigen over time: Implications for studies using stored sera. J. Med. Virol. 2011, 83, 1900–1904. [Google Scholar] [CrossRef] [PubMed]
- Basso, D.; Aita, A.; Navaglia, F.; Franchin, E.; Fioretto, P.; Moz, S.; Bozzato, D.; Zambon, C.F.; Martin, B.; Dal Prà, C.; et al. SARS-CoV-2 RNA identification in nasopharyngeal swabs: Issues in pre-analytics. Clin. Chem. Lab. Med. 2020, 58, 1579–1586. [Google Scholar] [CrossRef] [PubMed]
- D’Cruz, R.J.; Currier, A.W.; Sampson, V.B. Laboratory testing methods for novel severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2). Front. Cell Dev. Biol. 2020, 8, 468. [Google Scholar] [CrossRef] [PubMed]

| PCR-Negative n (%) | PCR-Positive n (%) | Total n (%) | |
|---|---|---|---|
| Antigen-negative | 72 (39.6%) | 33 (18.1%) | 105 (58.2%) |
| Antigen-positive | 0 (0%) | 77 (42.3%) | 77 (42.3%) |
| Total | 72 (39.6%) | 110 (60.4%) | 182 (100%) |
| Cut-Off (TCID50/mL) | AUC | Sensitivity | Specificity | PPV | NPV |
|---|---|---|---|---|---|
| 22.79 | 0.862 (0.8–0.923) | 92% (85–96%) | 81% (69–89%) | 0.88 (0.8–0.93) | 0.87 (0.76–0.93) |
| 23.56 | 0.871 (0.812–0.93) | 91% (84–95%) | 83% (72–91%) | 0.89 (0.82–0.94) | 0.86 (0.75–0.93) |
| 57.68 | 0.889 (0.839–0.938) | 79% (70–86%) | 99% (91–100%) | 0.99 (0.93–1) | 0.76 (0.65–0.84) |
| 100 | 0.877 (0.826–0.928) | 75% (66–83%) | 100% (94–100%) | 1 (0.94–1) | 0.73 (0.63–0.81) |
| 200 | 0.85 (0.794–0.906) | 70% (60–78%) | 100% (94–100%) | 1 (0.94–1) | 0.69 (0.59–0.77) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fiedler, M.; Holtkamp, C.; Dittmer, U.; Anastasiou, O.E. Performance of the LIAISON® SARS-CoV-2 Antigen Assay vs. SARS-CoV-2-RT-PCR. Pathogens 2021, 10, 658. https://doi.org/10.3390/pathogens10060658
Fiedler M, Holtkamp C, Dittmer U, Anastasiou OE. Performance of the LIAISON® SARS-CoV-2 Antigen Assay vs. SARS-CoV-2-RT-PCR. Pathogens. 2021; 10(6):658. https://doi.org/10.3390/pathogens10060658
Chicago/Turabian StyleFiedler, Melanie, Caroline Holtkamp, Ulf Dittmer, and Olympia E. Anastasiou. 2021. "Performance of the LIAISON® SARS-CoV-2 Antigen Assay vs. SARS-CoV-2-RT-PCR" Pathogens 10, no. 6: 658. https://doi.org/10.3390/pathogens10060658
APA StyleFiedler, M., Holtkamp, C., Dittmer, U., & Anastasiou, O. E. (2021). Performance of the LIAISON® SARS-CoV-2 Antigen Assay vs. SARS-CoV-2-RT-PCR. Pathogens, 10(6), 658. https://doi.org/10.3390/pathogens10060658






