Mortality Levels and Production Indicators for Suspicion of Highly Pathogenic Avian Influenza Virus Infection in Commercially Farmed Ducks
Abstract
:1. Introduction
2. Results
2.1. Broiler Ducks
2.1.1. Daily (Baseline) Mortality and Feed and Water Intake in Non-HPAIV-Infected Flocks
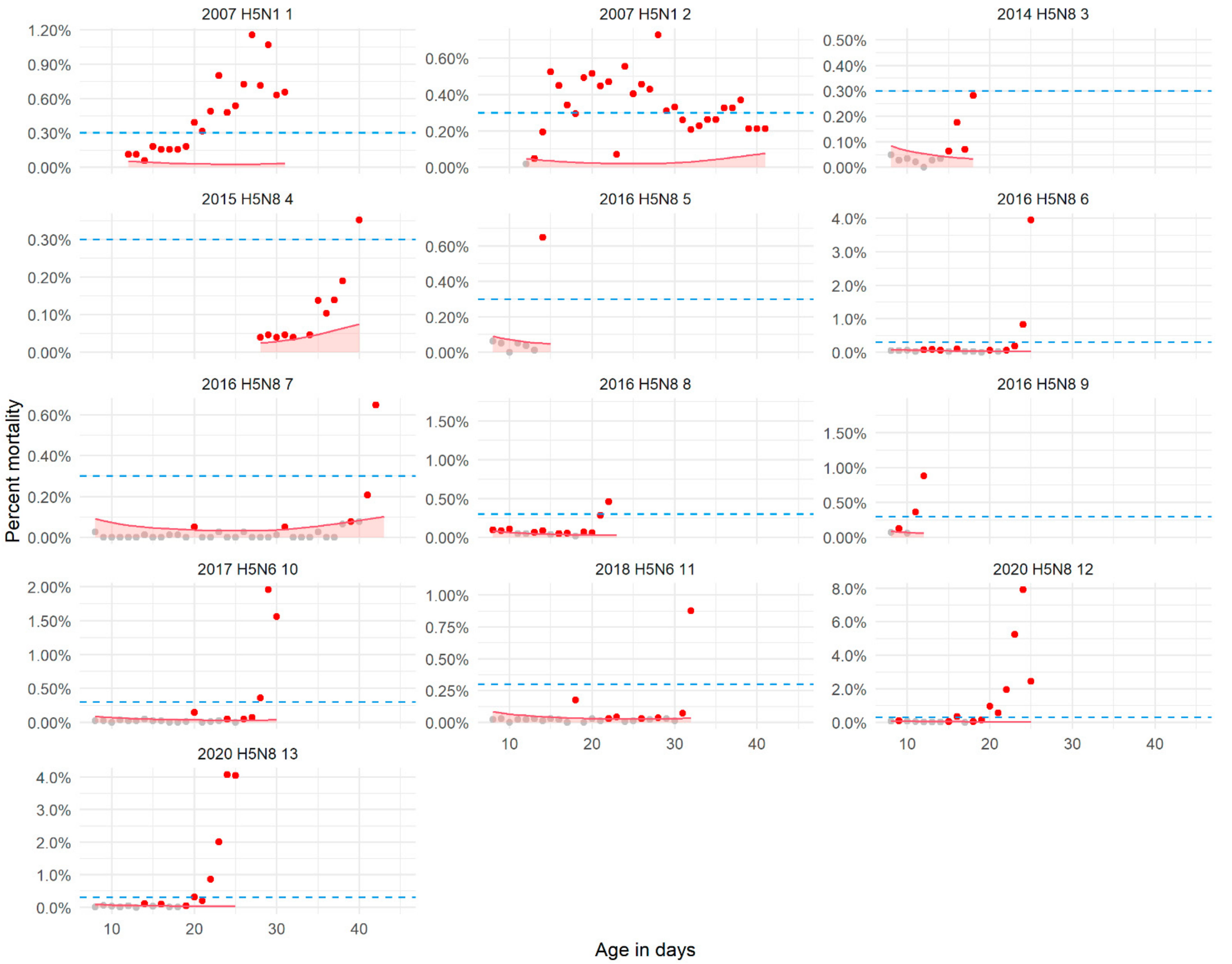
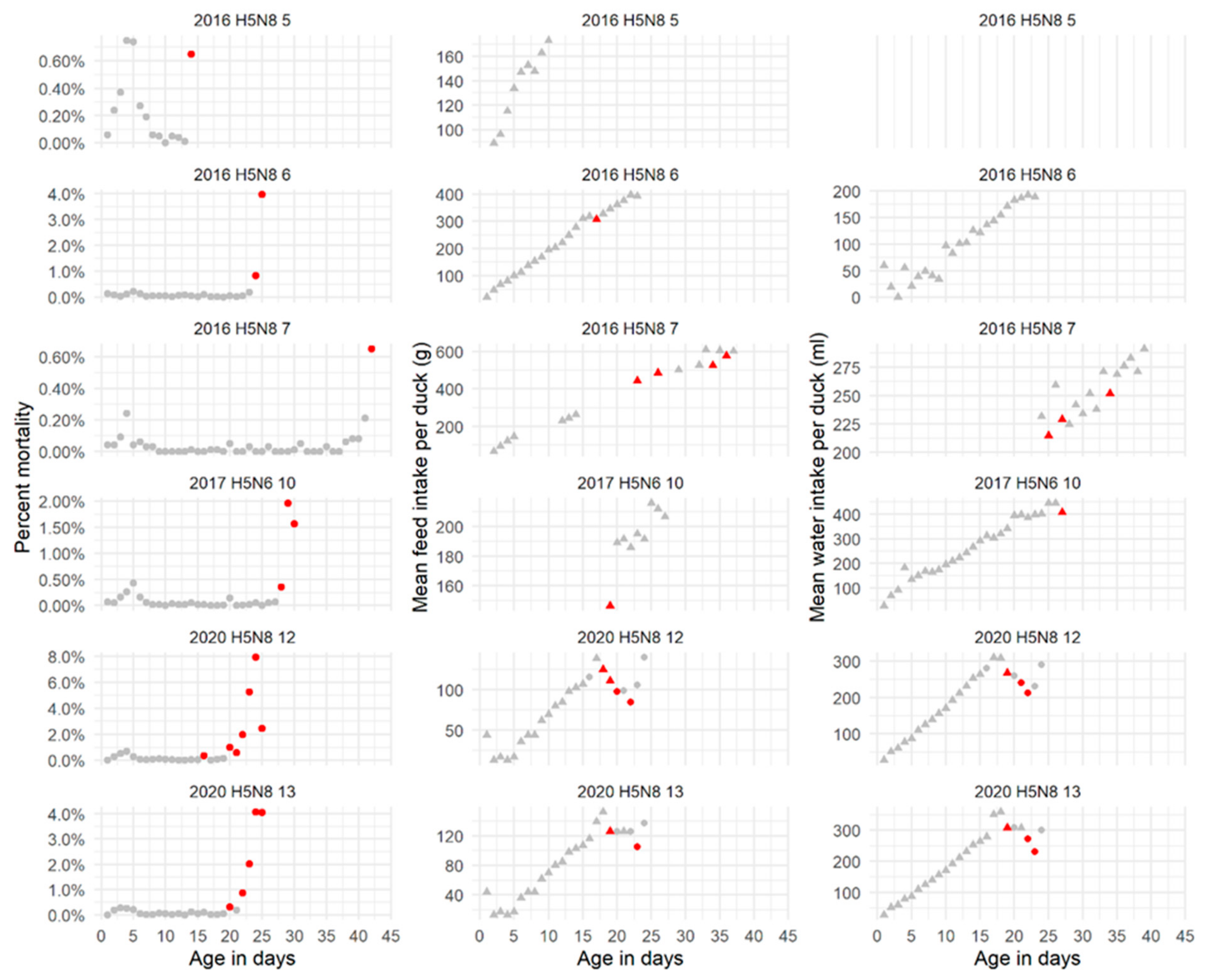
2.1.2. Mortality and Feed and Water Intake in HPAIV-Infected Broiler Flocks
2.1.3. Detection Thresholds
2.2. Duck Breeders
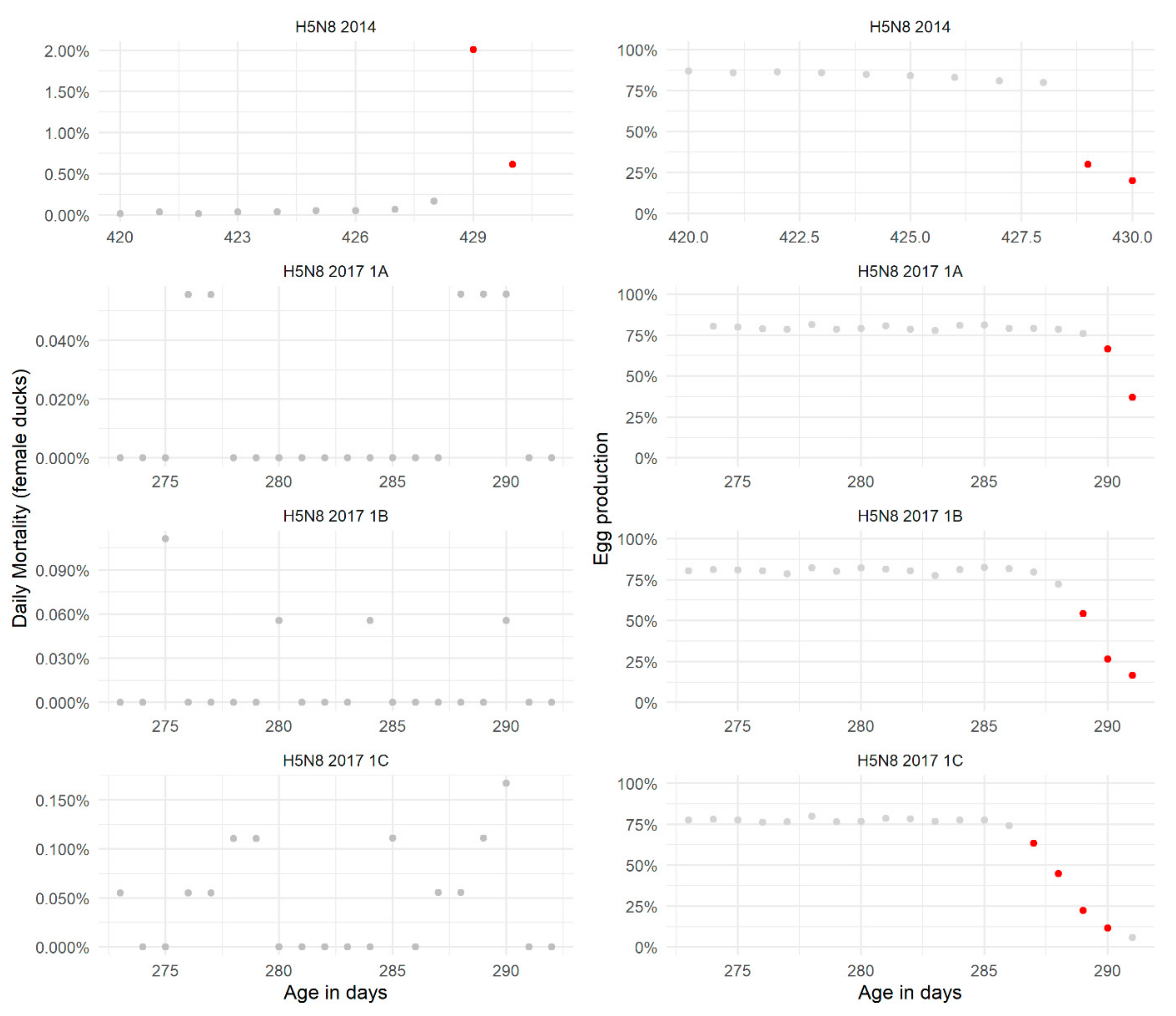
2.2.1. Daily (Baseline) Mortality and Egg Production in Non-HPAIV-Infected Flocks
2.2.2. Mortality and Egg Production in HPAIV-Infected Flocks and Performance of Detection Thresholds
3. Discussion
4. Materials and Methods
4.1. Data
4.1.1. Non-HPAIV-Infected Flocks
4.1.2. HPAIV-Infected Duck Flocks
4.2. Production Parameters and Detection Algorithms
4.2.1. Daily Mortality
- Fixed Mortality Thresholds
- Flock-Tailored Triggers
4.2.2. Feed and Water Intake
4.2.3. Egg Production
4.3. Evaluation of Performance
4.4. Data Analysis Software
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Elbers, A.R.W.; Koch, G.; Bouma, A. Performance of clinical signs in poultry for the detection of outbreaks during the avian influenza A (H7N7) epidemic in the Netherlands in 2003. Avian Pathol. 2005, 34, 181–187. [Google Scholar] [CrossRef]
- Gonzales, J.L.; Elbers, A.R.W. Effective thresholds for reporting suspicions and improve early detection of avian influenza outbreaks in layer chickens. Sci. Rep. 2018, 8, 8533. [Google Scholar] [CrossRef] [PubMed]
- Adlhoch, C.; Fusaro, A.; Gonzales, J.L.; Kuiken, T.; Marangon, S.; Niqueux, É.; Staubach, C.; Terregino, C.; Muño Guajardo, I.; Lima, E.; et al. Avian influenza overview December 2020–February 2021. EFSA J. 2021, 19, e06497. [Google Scholar]
- Swayne, D.E.; Suarez, D.L.; Simms, L.D. Influenza. In Diseases of Poultry, 14th ed.; Swayne, D.E., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2020; pp. 210–256. [Google Scholar]
- Capua, I.; Mutinelli, F. Mortality in Muscovy ducks (Cairina moschata) and domestic geese (Anser anser var. domestica) associated with natural infection with a highly pathogenic avian influenza virus of H7N1 subtype. Avian Pathol. 2001, 30, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.-K.; Joh, S.-J.; Kim, M.-C.; Sung, H.-W.; Lee, Y.-J.; Choi, J.-G.; Lee, E.-K.; Kim, J.-H. Highly pathogenic avian influenza (H5N1) in the commercial domestic ducks of South Korea. Avian Pathol. 2005, 34, 367–370. [Google Scholar] [CrossRef] [Green Version]
- Rhyoo, M.-Y.; Lee, K.-H.; Moon, O.-K.; Park, W.-H.; Bae, Y.-C.; Jung, J.-Y.; Yoon, S.-S.; Kim, H.-R.; Lee, M.-H.; Lee, E.-J.; et al. Analysis of signs and pathology of H5N1-infected ducks from the 2010-2011 Korean highly pathogenic avian influenza outbreak suggests the influence of age and management practices on severity of disease. Avian Pathol. 2015, 44, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Banyai, K.; Bistyak, A.T.; Thuma, A.; Gyuris, E.; Ursu, K.; Marton, S.; Farkas, S.L.; Bacsadi, A.; Dan, A. Neuroinvasive influenza A (H5N8) in fattening ducks. Infect. Genet. Evol. 2016, 43, 418–423. [Google Scholar]
- Nunez, A.; Brookes, S.M.; Reid, S.M.; Garcia-Rueda, C.; Hicks, D.J.; Seekings, J.M.; Spencer, Y.I.; Brown, I.H. Highly pathogenic avian influenza H5N8 Clade 2.3.4.4. virus: Equivocal pathogenicity and implications for surveillance following natural infection in breeder ducks in the United Kingdom. Transbound. Emerg. Dis. 2016, 63, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Stoute, S.; Crossly, B.; Shivaprasad, H.L. Study of an outbreak of highly pathogenic avian influenza H5N8 in commercial Pekin ducks (Anas platyrhynchos domesticus) in California. Avian Dis. 2018, 62, 101–108. [Google Scholar] [CrossRef]
- Dutch Animal Health and Welfare Law. Regeling Preventie, Bestrijding en Monitoring van Besmettelijke Dierziekten en Zoönosen en TSE’s. Available online: https://wetten.overheid.nl/BWBR0018397/2021-01-01 (accessed on 20 November 2020).
- Elbers, A.R.W.; Fabri, T.; De Vries, T.S.; De Wit, J.J.; Pijpers, A.; Koch, G. The highly pathogenic Avian Influenza A (H7N7) virus epidemic in the Netherlands: Lessons learned from the first five outbreaks. Avian Dis. 2004, 48, 691–705. [Google Scholar] [CrossRef]
- Bouwstra, R.J.; Koch, G.; Heutink, R.; Harders, F.; van der Spek, A.N.; Elbers, A.R.W.; Bossers, A. Full genome sequence of HPAI H5N8 outbreak strains provide evidence for four separate introductions and one between-poultry farm transmission in the Netherlands, 2014. Euro Surveill. 2015, 20, 21174. [Google Scholar] [CrossRef] [Green Version]
- Beerens, N.; Heutink, R.; Bergervoet, S.A.; Harders, F.; Bossers, A.; Koch, G. Multiple Reassorted Viruses as Cause of Highly Pathogenic Avian Influenza A(H5N8) Virus Epidemic, the Netherlands, 2016. Emerg. Infect. Dis. 2017, 23, 1974–1981. [Google Scholar] [CrossRef] [PubMed]
- Beerens, N.; Koch, G.; Heutink, R.; Harders, F.; Vries, D.P.E.; Ho, C.; Bossers, A.; Elbers, A.R.W. Novel Highly Pathogenic Avian Influenza A(H5N6) Virus in the Netherlands, December 2017. Emerg. Infect. Dis. 2018, 24, 770–773. [Google Scholar] [CrossRef]
- Guinat, C.; Artois, J.; Bronner, A.; Guerin, J.L.; Gilbert, M.; Paul, M.C. Duck production systems and highly pathogenic avian influenza H5N8 in France, 2016–2017. Sci. Rep. 2019, 9, 6177. [Google Scholar] [CrossRef]
- Lemetayer, M. France Plans Mass Duck Slaughter as Bird Flu Hits Foie Gras. Available online: https://phys.org/news/2021-01-france-duck-cull-bird-flu.html (accessed on 28 June 2021).
- Xin, H.; Berry, I.L.; Barton, T.L.; Tabler, G.T. Feed and water consumption, growth and mortality of male broilers. Poult. Sci. 1994, 73, 610–616. [Google Scholar] [CrossRef]
- Tabler, G.T.; Berry, I.L.; Mendenhall, A.M. Mortality Patterns Associated with Commercial Broiler Production. The Poultry Site, 13 December 2004. Available online: https://www.thepoultrysite.com/articles/mortality-patterns-associated-with-commercial-broiler-production(accessed on 2 May 2021).
- Yassin, H.; Velthuis, A.G.J.; Boerjan, M.; van Riel, J. Field study on broilers’ first-week mortality. Poult. Sci. 2009, 88, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Yerpes, M.; Llonch, P.; Manteca, X. Factors associated with cumulative first-week mortality in broiler chicks. Animals 2020, 10, 310. [Google Scholar] [CrossRef] [Green Version]
- Heier, B.T.; Hogasen, H.R.; Jarp, J. Factors associated with mortality in Norwegian broiler flocks. Prev. Vet. Med. 2002, 53, 147–158. [Google Scholar] [CrossRef]
- Elbers, A.R.W.; Gorgievski-Duijvesteijn, M.J.; Zarafshani, K.; Koch, G. To report or not to report: A psychosocial investigation aimed at improving early detection of Avian Influenza outbreaks. Rev. Sci. Tech. OIE 2010, 29, 435–449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beltrán-Alcrudo, D.; Carpenter, T.E.; Cardona, C. A flock-tailored early warning system for low pathogenic avian influenza (LPAI) in commercial egg laying flocks. Prev. Vet. Med. 2009, 92, 324–332. [Google Scholar] [CrossRef]
- Gonzales, J.L.; Elbers, A.R.W.; Bouma, A.; Koch, G.; de Wit, J.J.; Stegeman, J.A. Low-pathogenic notifiable avian influenza serosurveillance and the risk of infection in poultry—A critical review of the European Union active surveillance programme (2005–2007). Influenza Other Respir. Dis. 2010, 4, 91–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutwagner, L.C.; Thompson, W.W.; Seeman, G.M.; Treadwell, T. A simulation model for assessing aberration detection methods used in public health surveillance for systems with limited baselines. Stat. Med. 2005, 24, 543–550. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 13 May 2021).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Knowles, J.E.; Frederick, C. merTools: Tools for Analyzing Mixed Effect Regression Models. R package version 0.5.2. 2020. Available online: https://CRAN.R-project.org/package=merTools (accessed on 9 April 2021).
| Threshold | Sensitivity (n = 13) | Median (Range) Days Earlier Detection | Specificity (n = 53) | False Alarm Rate (95% Confidence Interval) | ||||
|---|---|---|---|---|---|---|---|---|
| 1 day | 2 days | 1 day | 2 days | 1 day | 2 days | 1 day | 2 days | |
| 0.1% | 1.0 | 0.769 | 2 (0–6) | 1 (0–5) | 0.245 | 0.491 | 0.104 (0.091–0.119) | 0.066 (0.056–0.078) |
| 0.17% | 1.0 | 0.769 | 2 (0–7) | 1 (0–4) | 0.547 | 0.698 | 0.041 (0.033–0.051) | 0.022 (0.016–0.029) |
| 0.3% | 0.923 | 0.538 | 1 (0–5) | 1 (0–5) | 0.811 | 0.962 | 0.009 (0.005–0.014) | 0.001 (0.000–0.004) |
| GLMM a | 1.0 | 0.923 | 9 (3–11) | 3 (2–11) | 0.792 | 0.962 | 0.009 (0.006–0.014) | 0.002 (0.001–0.004) |
| CUSUM-1 b | 1.0 | 1.0 | 5 (1–6) | 3 (0–4) | 0.038 | 0.170 | 0.216 (0.198–0.234) | 0.109 (0.096–0.123) |
| CUSUM-2 c | 1.0 | 1.0 | 5 (1–6) | 3 (0–4) | 0.057 | 0.226 | 0.206 (0.189–0.225) | 0.104 (0.091–0.119) |
| Threshold | Specificity (n = 10) | False Alarm Rate (95% CI) | ||
|---|---|---|---|---|
| 1 Day | 2 Days | 1 Day | 2 Days | |
| Mortality | ||||
| 0.07% | 0.000 | 0.000 | 0.2847 (0.2704–0.3000) | 0.1142 (0.1044–0.1250) |
| 0.2% | 0.000 | 0.600 | 0.0348 (0.0294–0.0412) | 0.0043 (0.0026–0.0070) |
| 0.3% | 0.400 | 0.900 | 0.0088 (0.0063–0.0124) | 0.0005 (0.0001–0.0019) |
| Drop in egg production | ||||
| >9% | 0.636 | 1.0 | 0.0016 (0.0007–0.0038) | 0.0000 (0.0000–0.0012) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elbers, A.R.W.; Gonzales, J.L. Mortality Levels and Production Indicators for Suspicion of Highly Pathogenic Avian Influenza Virus Infection in Commercially Farmed Ducks. Pathogens 2021, 10, 1498. https://doi.org/10.3390/pathogens10111498
Elbers ARW, Gonzales JL. Mortality Levels and Production Indicators for Suspicion of Highly Pathogenic Avian Influenza Virus Infection in Commercially Farmed Ducks. Pathogens. 2021; 10(11):1498. https://doi.org/10.3390/pathogens10111498
Chicago/Turabian StyleElbers, Armin R. W., and José L. Gonzales. 2021. "Mortality Levels and Production Indicators for Suspicion of Highly Pathogenic Avian Influenza Virus Infection in Commercially Farmed Ducks" Pathogens 10, no. 11: 1498. https://doi.org/10.3390/pathogens10111498
APA StyleElbers, A. R. W., & Gonzales, J. L. (2021). Mortality Levels and Production Indicators for Suspicion of Highly Pathogenic Avian Influenza Virus Infection in Commercially Farmed Ducks. Pathogens, 10(11), 1498. https://doi.org/10.3390/pathogens10111498







