First Record of Trichobilharzia physellae (Talbot, 1936) in Europe, a Possible Causative Agent of Cercarial Dermatitis
Abstract
:1. Introduction
2. Results
2.1. Morphology
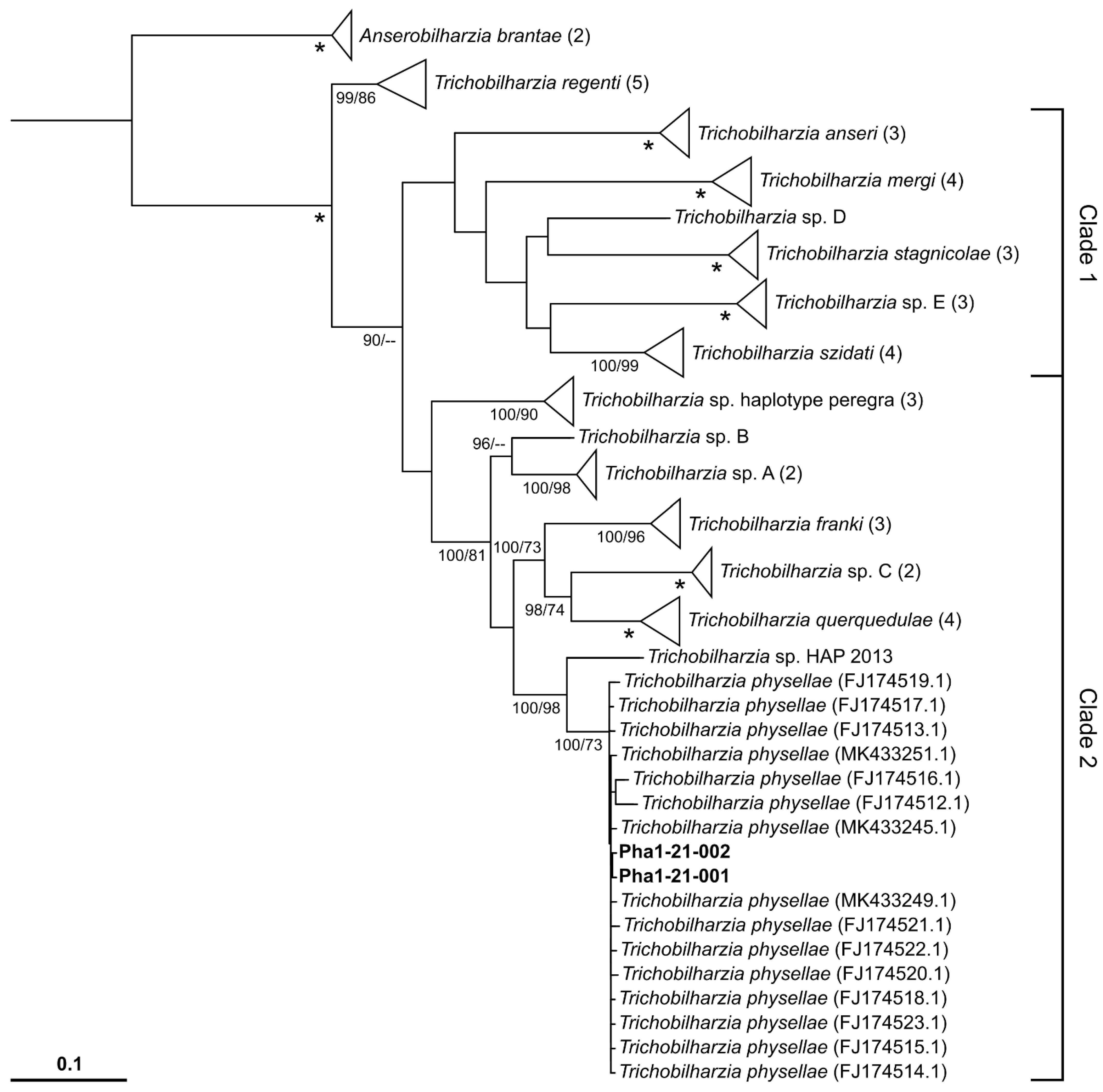
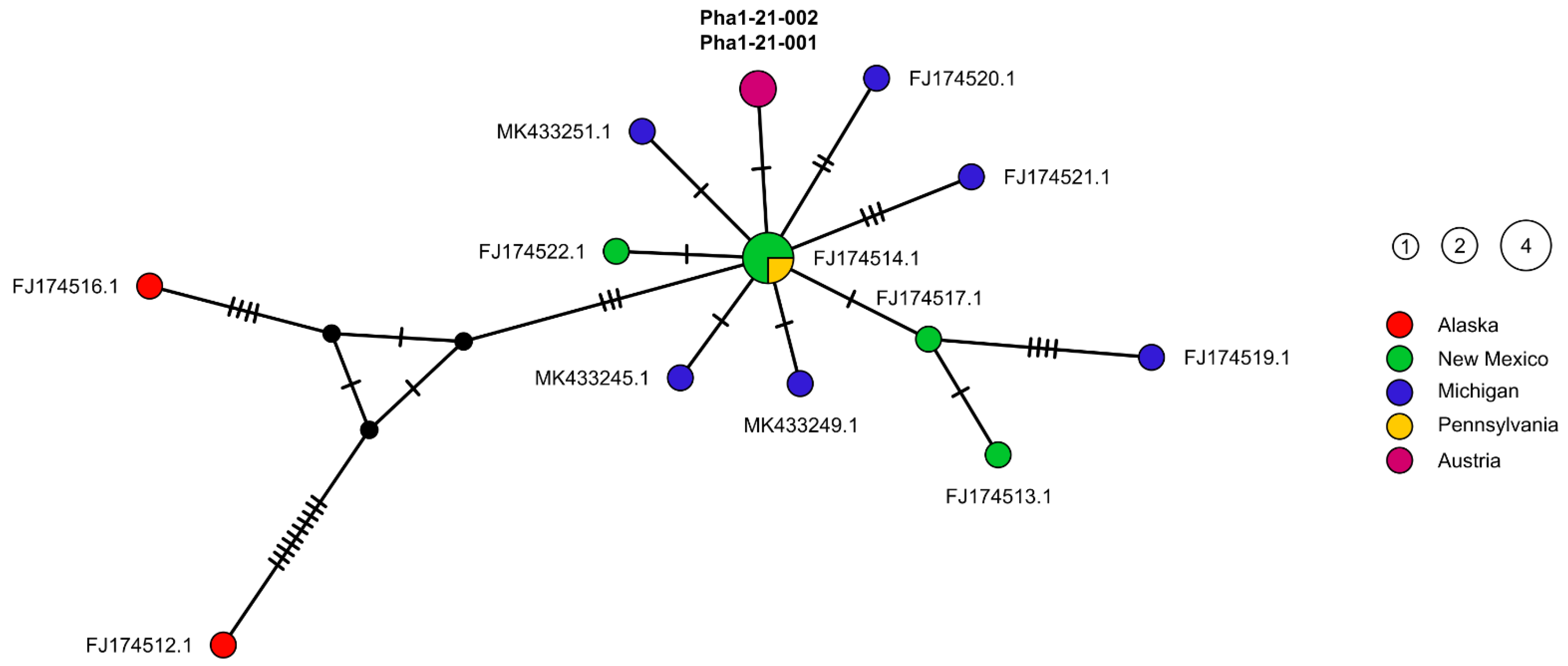
2.2. Molecular Genetic Results
3. Discussion
4. Materials and Methods
4.1. Sampling
4.2. Morphological Examination
4.3. Analysis of the Mitochondrial cytochrome c oxidase subunit 1 Gene
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Genus/ Species | Acc.No/ Lab-ID | Host | Life Cycle Stage | Locality | Reference |
|---|---|---|---|---|---|
| Anserobilharzia | |||||
| Anserobilharzia brantae | |||||
| MK433247.1 | Gyraulus sp. | cercariae | USA, Michigan | [37] | |
| KC570954.1 | Anser anser | egg | France, Der-Chantecoq Lake | [14] | |
| Trichobilharzia | |||||
| Trichobilharzia anseri | |||||
| KP901380.1 | Radix balthica | cercariae | Iceland, Family park, Reykjavik | [77] | |
| KP901382.1 | Anser anser | egg | France, Der-Chantecoq Lake | [77] | |
| KP901381.1 | Radix balthica | cercariae | Iceland, Family park, Reykjavik | [77] | |
| Trichobilharzia franki | |||||
| § FJ174530.1 | Radix sp. | cercariae | Czech Republic | [34] | |
| HM131200.1 | Radix auricularia | cercariae | France, Beauvais | [15] | |
| HM131197.1 | Radix auricularia | cercariae | France, Der-Chantecoq Lake | [15] | |
| Trichobilharzia mergi | |||||
| JQ681535.1 | Radix ampla | cercariae | Belarus, Naroch Lake | [78] | |
| JQ681536.1 | Radix ampla | cercariae | Belarus, Naroch Lake | [78] | |
| JX456172.1 | Mergus serrator | adult | Iceland, Botsvatn Lake | [79] | |
| JX456171.1 | Mergus serrator | adult | Iceland, Botsvatn Lake | [79] | |
| Trichobilharzia physellae | |||||
| FJ174512.1 | Aythya affinis | adult | USA, Alaska | [34] | |
| FJ174513.1 | Physa gyrina | cercaria | USA, New Mexico | [34] | |
| FJ174514.1 | Bucephala albeola | miracidia | USA, New Mexico | [34] | |
| FJ174515.1 | Aythya affinis | adult | USA, Pennsylvania | [34] | |
| FJ174516.1 | Clangula hyemalis | adult | USA, Alaska | [34] | |
| FJ174517.1 | Aythya collaris | adult | USA, New Mexico | [34] | |
| FJ174518.1 | Aythya affinis | adult | USA, New Mexico | [34] | |
| FJ174519.1 | Mergus merganser | miracidia | USA, Michigan | [34] | |
| § FJ174520.1 | Mergus merganser/Physa parkeri | miracidia/cercariae | USA, Michigan | [34] | |
| FJ174521.1 | Mergus merganser | miracidia | USA, Michigan | [34] | |
| FJ174522.1 | Aythya affinis | adult | USA, New Mexico | [34] | |
| FJ174523.1 | Physa gyrina | cercaria | USA, New Mexico | [34] | |
| MK433245.1 | Physa sp. | cercariae | USA, Michigan | [37] | |
| MK433249.1 | Common Merganser | miracidia | USA, Michigan | [37] | |
| MK433251.1 | Mallard | miracidia | USA, Michigan | [37] | |
| * Pha1-21-001/NHBP001-21/ OL434665 | Physella acuta | cercaria | Austria, Upper Austria | Present Study | |
| * Pha1-21-002/NHBP002-21/ OL434663 | Physella acuta | cercaria | Austria, Upper Austria | Present Study | |
| * Pha2-21-001/NHBP003-21/ OL434664 | Physella acuta | cercaria | Austria, Upper Austria | Present Study | |
| * Pha2-21-002/NHBP004-21/ OL434662 | Physella acuta | cercaria | Austria, Upper Austria | Present Study | |
| Trichobilharzia querquedulae | |||||
| KU057181.1 | Anas rhynchotis | adult | New Zealand, South Island | [45] | |
| FJ174498.1 | Anas discors | adult | USA, Louisiana | [34] | |
| KU057183.1 | Anas rhynchotis | adult | New Zealand, South Island | [45] | |
| FJ174497.1 | Anas clypeata | adult | USA, Louisiana | [34] | |
| Trichobilharzia regenti | |||||
| MN337555.1 | Anas platyrhynchos | egg | Iran, Azbaran | unpublished | |
| MN337560.1 | Radix auricularia | cercariae | Iran, Azbaran | unpublished | |
| NC_009680.1 | Radix peregra | cercariae | Laboratory Snail | [80] | |
| MN337557.1 | Anas platyrhynchos domesticus | egg | Iran, Sari | unpublished | |
| HM439501.1 | Mergus merganser | adult | France, Annecy Lake | [81] | |
| Trichobilharzia sp. A | |||||
| FJ174527.1 | Anas americana | adult | USA, Alaska | [34] | |
| FJ174524.1 | Anas americana | adult | USA, New Mexico | [34] | |
| Trichobilharzia sp. B | |||||
| FJ174528.1 | Anas americana | adult | USA, Alaska | [34] | |
| Trichobilharzia sp. C | |||||
| KJ855996.1 | Aix sponsa | adult | USA | [35] | |
| FJ174529.1 | Lophodytes cucullatus | adult | USA | [34] | |
| Trichobilharzia sp. D | |||||
| FJ174485.1 | Stagnicola sp. | cercariae | Canada | [34] | |
| Trichobilharzia sp. E | |||||
| FJ174483.1 | Stagnicola sp. | cercariae | Canada | [34] | |
| FJ174487.1 | Anas acuta | adult | Canada | [34] | |
| FJ174486.1 | Stagnicola sp. | cercariae | Canada | [34] | |
| Trichobilharzia sp. HAP_2013 | |||||
| KJ855995.1 | Physa marmorata | cercariae | Brazil, Espírito Santo | [35] | |
| Trichobilharzia sp. haplotype peregra | |||||
| HM131205.1 | Radix peregra | cercariae | France, Annecy Lake | [15] | |
| HM131204.1 | Radix peregra | cercariae | France, Annecy Lake | [15] | |
| HM131203.1 | Radix peregra | cercariae | France, Annecy Lake | [15] | |
| Trichobilharzia stagnicolae | |||||
| FJ174488.1 | Stagnicola sp. | cercariae | USA, Montana | [34] | |
| KT831352.1 | Stagnicola elodes | cercariae | Canada, Alberta | [36] | |
| FJ174492.1 | Stagnicola sp. | cercariae | USA, New Mexico | [34] | |
| Trichobilharzia szidati | |||||
| NC_036411.1 | Lymnaea stagnalis | cercariae | Belarus, Naroch Lake | [82] | |
| MT708493.1 | Lymnaea stagnalis | cercariae | Belarus, Naroch Lake | [83] | |
| MG570047.1 | - | - | China | unpublished | |
| JF838200.1 | Lymnaea stagnalis | cercariae | Russia, Moscow, Olympiyskaya derevnya ponds | [47] | |
References
- Kearn, G.C. Parasitism and the Platyhelminths; Chapman & Hall: London, UK, 1998; ISBN 9780412804601. [Google Scholar]
- Jamieson, B.G.M. Schistosoma: Biology, Pathology, and Control; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Horák, P.; Mikeš, L.; Lichtenbergová, L.; Skála, V.; Soldánová, M.; Brant, S.V. Avian schistosomes and outbreaks of cercarial dermatitis. Clin. Microbiol. Rev. 2015, 28, 165–190. [Google Scholar] [CrossRef] [Green Version]
- Horák, P.; Kolářová, L. Snails, waterfowl and cercarial dermatitis. Freshw. Biol. 2010, 56, 779–790. [Google Scholar] [CrossRef]
- Auer, H.; Aspöck, H. Helminths and helminthoses in Central Europe: General overview and diseases caused by trematodes (flukes). Wien. Med. Wochenschr. 2014, 164, 405–413. [Google Scholar] [CrossRef]
- Bayssade-Dufour, C.; Martins, C.; Vuong, P.N. Histopathologie pulmonaire d’un modèle mammifère et dermatite cercarienne humaine. Méd. Mal. Infect. 2001, 31, 713–722. [Google Scholar] [CrossRef]
- Marszewska, A.; Cichy, A.; Heese, T.; Żbikowska, E. The real threat of swimmers’ itch in anthropogenic recreational water body of the Polish Lowland. Parasitol. Res. 2016, 115, 3049–3056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macháček, T.; Turjanicová, L.; Bulantová, J.; Hrdý, J.; Horák, P.; Mikeš, L. Cercarial dermatitis: A systematic follow-up study of human cases with implications for diagnostics. Parasitol. Res. 2018, 117, 3881–3895. [Google Scholar] [CrossRef]
- Lichtenbergová, L.; Horák, P. Pathogenicity of Trichobilharzia spp. for Vertebrates. J. Parasitol. Res. 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Soldánová, M.; Selbach, C.; Kalbe, M.; Kostadinova, A.; Sures, B. Swimmer’s itch: Etiology, impact, and risk factors in Europe. Trends Parasitol. 2013, 29, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Horák, P.; Kolářová, L.; Adema, C. Biology of the schistosome genus Trichobilharzia. Adv. Parasitol. 2002, 52, 155–233. [Google Scholar] [CrossRef] [PubMed]
- Brant, S.V. The occurrence of the avian schistosome Allobilharzia visceralis Kolarova, Rudolfova, Hampl et Skirnisson, 2006 (Schistosomatidae) in the tundra swan, Cygnus columbianus (Anatidae), from North America. Folia Parasitol. 2007, 54, 99–104. [Google Scholar] [CrossRef] [Green Version]
- Brant, S.V.; Loker, E.S. Discovery-based studies of schistosome diversity stimulate new hypotheses about parasite biology. Trends Parasitol. 2013, 29, 449–459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brant, S.V.; Jouet, D.; Ferté, H.; Loker, E.S. Anserobilharzia gen. n. (Digenea, Schistosomatidae) and redescription of A. brantae (Farr & Blankemeyer, 1956) comb. n. (syn. Trichobilharzia brantae), a parasite of geese (Anseriformes). Zootaxa 2013, 3670, 193–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jouet, D.; Skírnisson, K.; Kolářová, L.; Ferté, H. Molecular diversity of Trichobilharzia franki in two intermediate hosts (Radix auricularia and Radix peregra): A complex of species. Infect. Genet. Evol. 2010, 10, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, A.; Olsen, A.; Buchmann, K.; Kania, P.W.; Nejsum, P.; Vennervald, B. Molecular diversity of avian schistosomes in Danish freshwater snails. Parasitol. Res. 2015, 115, 1027–1037. [Google Scholar] [CrossRef]
- Sattmann, H.; Hörweg, C.; Konecny, R. Zerkariendermatitis in Österreich–Rückblick und Perspektiven. Denisia 2004, 13, 457–461. [Google Scholar]
- Hörweg, C.; Sattmann, H.; Auer, H. Cercarial dermatitis in Austria: Questionnaires as useful tools to estimate risk factors? Wien. Klin. Wochenschr. 2006, 118, 77–80. [Google Scholar] [CrossRef]
- Graefe, G. Experimenteller Nachweis einer von Cercarien verursachten Dermatitis am Neusiedler See. Sitz. Akad. Wiss. Math. Klasse. Abt. 1 1971, 179, 73–79. [Google Scholar]
- Graefe, G.; Aspöck, H.; Picher, O. Auftreten von Bade-Dermatitis in Österreich und Möglichkeiten ihrer Bekämpfung. Zentralbl. Bakteriol. Orig. A. 1973, 225, 398–405. [Google Scholar]
- Dvořák, J.; Sattmann, H.; Horák, P.; Konecny, R. Bird schistosomes from freshwater snails in Austria, with some notes on current problems (Digenea, Schistosomatidae). Mitt. Österr. Ges. Tropenmed. Parasitol. 1999, 21, 69–76. [Google Scholar]
- Reier, S.; Haring, E.; Billinger, F.; Blatterer, H.; Duda, M.; Gorofsky, C.; Grasser, H.-P.; Heinisch, W.; Hörweg, C.; Kruckenhauser, L.; et al. First confirmed record of Trichobilharzia franki Müller & Kimmig, 1994, from Radix auricularia (Linnaeus, 1758) for Austria. Parasitol. Res. 2020, 119, 4135–4141. [Google Scholar] [CrossRef]
- Gaub, L.; Sattmann, H.; Hörweg, C.; Walochnik, J. Molecular validation of species determination of larval trematodes from freshwater snail hosts in Austria, with special emphasis on the genus Trichobilharzia Skrjabin & Zakharow, 1920. Arianta 2020, 8, 13–19. [Google Scholar]
- Auer, H.; Aspöck, H. “Vogelbilharzien” als Erreger einer Hautkrankheit: Die Zerkarien-Dermatitis. Denisia 2002, 6, 321–331. [Google Scholar]
- Lydeard, C.; Campbell, D.; Golz, M. Physa acuta Draparnaud, 1805 should be treated as a native of North America, not Europe. Malacologia 2016, 59, 347–350. [Google Scholar] [CrossRef]
- Vinarski, M.V. The history of an invasion: Phases of the explosive spread of the physid snail Physella acuta through Europe, Transcaucasia and Central Asia. Biol. Invasions 2017, 19, 1299–1314. [Google Scholar] [CrossRef]
- Ebbs, E.T.; Loker, E.S.; Brant, S.V. Phylogeography and genetics of the globally invasive snail Physa acuta Draparnaud 1805, and its potential to serve as an intermediate host to larval digenetic trematodes. BMC Evol. Biol. 2018, 18, 103. [Google Scholar] [CrossRef]
- Appleton, C.C. Alien and invasive fresh water Gastropoda in South Africa. Afr. J. Aquat. Sci. 2003, 28, 69–81. [Google Scholar] [CrossRef]
- Helmer, N.; Haring, E.; Hörweg, C.; Sattmann, H.; Szucsich, N. Sensitivität des eDNA-Nachweises von Zerkariendermatitis- Erregern in oberösterreichischen Gewässern; Wien. 2021. Available online: https://www.land-oberoesterreich.gv.at/Mediendateien/Formulare/DokumenteUWDAbt_WW/ProjektberichtZerkarieneDNA2020-v2.pdf (accessed on 1 September 2021).
- Talbot, S.B. Studies on schistosome dermatitis: II. Morphological and life history studies on three dermatitis-producing schis-tosome cercariae, C. elvae Miller, 1923, C. stagnicolae n. sp., and C. physellae n. sp. Am. J. Epidemiol. 1936, 23, 372–384. [Google Scholar] [CrossRef]
- Tanaka, M. Studies on Trichobilharzia physellae in Oki Islands. 2. Four kinds of schistosome cercariae parasitic in Lymnaea japonica in Oki Island. Jpn. J. Parasitol. 1960, 9, 604–609. [Google Scholar]
- Pence, D.B.; Rhodes, M.J. Trichobilharzia physellae (Digenea: Schistosomatidae) from endemic waterfowl on the high plains of texas. J. Wildl. Dis. 1982, 18, 69–74. [Google Scholar] [CrossRef] [Green Version]
- Moore, A.C.; Burch, J.B.; Duda, T.F. Recognition of a highly restricted freshwater snail lineage (Physidae: Physella) in southeastern Oregon: Convergent evolution, historical context, and conservation considerations. Conserv. Genet. 2014, 16, 113–123. [Google Scholar] [CrossRef] [Green Version]
- Brant, S.V.; Loker, E.S. Molecular systematics of the avian schistosome genus Trichobilharzia (Trematoda: Schistosomatidae) in North America. J. Parasitol. 2009, 95, 941–963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinto, H.A.; Brant, S.V.; De Melo, A.L. Physa marmorata (Mollusca: Physidae) as a natural intermediate host of Trichobilharzia (Trematoda: Schistosomatidae), a potential causative agent of avian cercarial dermatitis in Brazil. Acta Trop. 2014, 138, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Gordy, M.A.; Kish, L.; Tarrabain, M.; Hanington, P.C. A comprehensive survey of larval digenean trematodes and their snail hosts in central Alberta, Canada. Parasitol. Res. 2016, 115, 3867–3880. [Google Scholar] [CrossRef] [PubMed]
- Rudko, S.P.; Turnbull, A.; Reimink, R.L.; Froelich, K.; Hanington, P.C. Species-specific qPCR assays allow for high-resolution population assessment of four species avian schistosome that cause swimmer’s itch in recreational lakes. Int. J. Parasitol. Parasit. Wildl. 2019, 9, 122–129. [Google Scholar] [CrossRef]
- Misaki, S. Studies on the fine structure of cercariae of Trichobilharzia physellae (Talbot 1936) and Trichobilharzia ocellata (La-Valette 1855). Jpn. J. Parasitol. 1960, 9, 744–759. [Google Scholar]
- Tanaka, M. Studies on Trichobilharzia physellae in Oki Island. 1. Trichobilharzia physellae found in wild ducks in Oki Island. Jpn. J. Parasitol. 1960, 9, 596–603. [Google Scholar]
- Dutt, S.C. The Occurrence of Trichobilharzia physellae in India. Sci. Cult. 1965, 31, 320. [Google Scholar]
- Blair, D.; Islam, K. The life-cycle and morphology of Trichobilharzia australis n. sp. (Digenea: Schistosomatidae) from the nasal blood vessels of the black duck (Anas superciliosa) in Australia, with a review of the genus Trichobilharzia. Syst. Parasitol. 1983, 5, 89–117. [Google Scholar] [CrossRef]
- Podhorský, M.; Huůzová, Z.; Mikeš, L.; Horák, P. Cercarial dimensions and surface structures as a tool for species determination of Trichobilharzia spp. Acta Parasitol. 2009, 54, 28–36. [Google Scholar] [CrossRef]
- Wethington, A.R.; Lydeard, C. A molecular phylogeny of Physidae (Gastropoda: Basommatophora) based on mitochondrial DNA sequences. J. Molluscan Stud. 2007, 73, 241–257. [Google Scholar] [CrossRef] [Green Version]
- Lawton, S.P.; Lim, R.M.; Dukes, J.P.; Cook, R.T.; Walker, A.J.; Kirk, R.S. Identification of a major causative agent of human cercarial dermatitis, Trichobilharzia franki (Müller and Kimmig 1994), in southern England and its evolutionary relationships with other European populations. Parasit. Vectors 2014, 7, 277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ebbs, E.T.; Loker, E.S.; Davis, N.E.; Flores, V.; Veleizan, A.; Brant, S.V. Schistosomes with wings: How host phylogeny and ecology shape the global distribution of Trichobilharzia querquedulae (Schistosomatidae). Int. J. Parasitol. 2016, 46, 669–677. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopatkin, A.A.; Chrisanfova, G.G.; Voronin, M.V.; Zazornova, O.P.; Beer, S.A.; Semyenova, S.K. Polymorphism of the cox1 gene in cercariae isolates of bird schistosomes (Trematoda: Schistosomatidae) from ponds of Moscow and Moscow region. Russ. J. Genet. 2010, 46, 873–880. [Google Scholar] [CrossRef]
- Korsunenko, A.; Chrisanfova, G.; Lopatkin, A.; Beer, S.A.; Voronin, M.; Ryskov, A.; Semyenova, S.K. Genetic differentiation of cercariae infrapopulations of the avian schistosome Trichobilharzia szidati based on RAPD markers and mitochondrial cox1 gene. Parasitol. Res. 2012, 110, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Duggan, I.C. The freshwater aquarium trade as a vector for incidental invertebrate fauna. Biol. Invasions 2010, 12, 3757–3770. [Google Scholar] [CrossRef]
- Facon, B.; Pointier, J.-P.; Glaubrecht, M.; Poux, C.; Jarne, P.; David, P. A molecular phylogeography approach to biological invasions of the New World by parthenogenetic Thiarid snails. Mol. Ecol. 2003, 12, 3027–3039. [Google Scholar] [CrossRef]
- Pinto, H.; de Melo, A. A checklist of trematodes (Platyhelminthes) transmitted by Melanoides tuberculata (Mollusca: Thiaridae). Zootaxa 2011, 2799, 15–28. [Google Scholar] [CrossRef] [Green Version]
- Graham, A.L. Effects of snail size and age on the prevalence and intensity of avian schistosome infection: Relating laboratory to field studies. J. Parasitol. 2003, 89, 458–463. [Google Scholar] [CrossRef]
- DAK Seltene Vögel in Deutschland 2013. 2014, pp. 2–39. Available online: https://www.dda-web.de/downloads/publications/statusreports/svid_2013_seltenheitenbericht.pdf (accessed on 11 August 2021).
- Maumary, L.; Martinez, N. Seltene Vogelarten und ungewöhnliche Vogelbeobachtungen in der Schweiz im Jahre 2018. Der Ornithol. Beobachter 2019, 116, 241–274. [Google Scholar]
- Ranner, A.; Khil, L. Records of rare and remarkable bird species in Austria 2007–2009. Sixth report of the Avifaunistic Commission of BirdLife Austria. Egretta 2011, 52, 13–32. [Google Scholar]
- Šrank, V. The 6th Report of the Slovak Rarities Committee. Tichodroma 2006, 18, 145–147. [Google Scholar]
- MME Nomenclator Committee MME Nomenclator Committee Annual Report 2014 on the Occurrence of Rare Bird Species in Hungary. 2014, pp. 1–15. Available online: http://www.birding.hu/doc/NB2014.pdf (accessed on 11 August 2021).
- Fracasso, G.; Janni, O.; Fulco, E.; Liuzzi, C. Commissione Ornitologica Italiana (COI)—Report 27. Avocetta 2018, 42, 45–54. [Google Scholar]
- DAK Seltene Vögel in Deutschland 2017. 2019, pp. 2–34. Available online: https://www.dda-web.de/downloads/publications/statusreports/svid_2017_seltenheitenbericht.pdf (accessed on 11 August 2021).
- Marques, D.A.; Jaquier, S. Seltene Vogelarten und ungewöhnliche Vogelbeobachtungen in der Schweiz im Jahre 2019. Ornithol. Beob. 2020, 117, 312–337. [Google Scholar]
- Vavřík, M.; Šírek, J. FK Čso Rare birds in the Czech Republic in 2019. Sylvia 2020, 56, 93–114. [Google Scholar]
- Keller, V. The Goosander Mergus merganser population breeding in the Alps and its connections to the rest of Europe. Wildfowl 2009, special issue 2, 60–73. [Google Scholar]
- Weißmair, W. The goosander (Mergus merganser) in Upper-Austria–Breeding population in 2016/2017 and wintering population 1996–2016. Vogelkdl. Nachr. aus Oberösterr. Nat. Aktuell 2019, 27, 3–35. [Google Scholar]
- Lockyer, A.E.; Olson, P.D.; Østergaard, P.; Rollinson, D.; Johnston, D.A.; Attwood, S.W.; Southgate, V.R.; Horak, P.; Snyder, S.D.; LE, T.H.; et al. The phylogeny of the Schistosomatidae based on three genes with emphasis on the interrelationships of Schistosoma Weinland, 1858. Parasitology 2003, 126, 203–224. [Google Scholar] [CrossRef] [Green Version]
- Duda, M.; Schindelar, J.; Macek, O.; Eschner, A.; Kruckenhauser, L. First record of Trochulus clandestinus (Hartmann, 1821) in Austria (Gastropoda: Eupulmonata: Hygromiidae). Malacol. Bohemoslov. 2016, 16, 37–43. [Google Scholar]
- Nicholas, K.B.; Nicholas, H.B.J. GeneDoc: A tool for editing and annotating multiple sequence alignments. Embnet. News 1997, 4, 1–4. [Google Scholar]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K.; Battistuzzi, F.U. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2017, 20, 1160–1166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoang, D.T.; Chernomor, O.; Von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the Ultrafast Bootstrap Approximation. Mol. Biol. Evol. 2017, 35, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.; Von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; von Haeseler, A.; Lanfear, R. IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef] [Green Version]
- Chernomor, O.; Von Haeseler, A.; Minh, B.Q. Terrace aware data structure for phylogenomic inference from supermatrices. Syst. Biol. 2016, 65, 997–1008. [Google Scholar] [CrossRef] [Green Version]
- Ronquist, F.; Teslenko, M.; Van Der Mark, P.; Ayres, D.L.; Darling, A.; Hoehna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [Green Version]
- Letunic, I.; Bork, P. Interactive Tree of Life (iTOL) v5: An online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021, 49, W293–W296. [Google Scholar] [CrossRef]
- Bandelt, H.J.; Forster, P.; Rohl, A. Median-joining networks for inferring intraspecific phylogenies. Mol. Biol. Evol. 1999, 16, 37–48. [Google Scholar] [CrossRef]
- Rozas, J.; Ferrer-Mata, A.; Sánchez-DelBarrio, J.C.; Guirao-Rico, S.; Librado, P.; Ramos-Onsins, S.; Sánchez-Gracia, A. DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol. Biol. Evol. 2017, 34, 3299–3302. [Google Scholar] [CrossRef]
- Brant, S.V.; (University of New Mexico, Albuquerque, NM, USA). Personal communication, 2021.
- Jouet, D.; Kolářová, L.; Patrelle, C.; Ferté, H.; Skírnisson, K. Trichobilharzia anseri n. sp. (Schistosomatidae: Digenea), a new visceral species of avian schistosomes isolated from greylag goose (Anser anser L.) in Iceland and France. Infect. Genet. Evol. 2015, 34, 298–306. [Google Scholar] [CrossRef]
- Chrisanfova, G.G.; Lopatkin, A.A.; Mishchenkov, V.A.; Kheidorova, E.E.; Dorozhenkova, T.E.; Zhukova, T.V.; Ryskov, A.; Semyenova, S.K. Genetic variability of bird schistosomes (class Trematoda, family Schistosomatidae) of Naroch Lake: Identification of a new species in the Trichobilharzia ocellata group. Dokl. Biochem. Biophys. 2009, 428, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Kolářová, L.; Skírnisson, K.; Ferté, H.; Jouet, D. Trichobilharzia mergi sp. nov. (Trematoda: Digenea: Schistosomatidae), a visceral schistosome of Mergus serrator (L.) (Aves: Anatidae). Parasitol. Int. 2013, 62, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Webster, B.L.; Rudolfová, J.; Horák, P.; Littlewood, D.T.J. The complete mitochondrial genome of the bird schistosome Trichobilharzia regenti (Platyhelminthes: Digenea), causative agent of cercarial dermatitis. J. Parasitol. 2007, 93, 553–561. [Google Scholar] [CrossRef]
- Jouet, D.; Skírnisson, K.; Kolářová, L.; Ferté, H. Final hosts and variability of Trichobilharzia regenti under natural conditions. Parasitol. Res. 2010, 107, 923–930. [Google Scholar] [CrossRef]
- Semyenova, S.; Chrisanfova, G.; Mozharovskaya, L.; Guliaev, A.; Ryskov, A. The complete mitochondrial genome of the causative agent of the human cercarial dermatitis, the visceral bird schistosome species Trichobilharzia szidati (platyhelminthes: Trematoda: Schistosomatidae). Mitochondrial DNA Part B 2017, 2, 469–470. [Google Scholar] [CrossRef]
- Chrisanfova, G.; Mozharovskaya, L.; Zhukova, T.; Nefedova, D.; Semyenova, S. Non-coding Regions of mitochondrial DNA and the cox1 gene reveal genetic variability among local Belarusian populations of the causative agent of cercarial dermatitis, bird schistosome Trichobilharzia szidati (Digenea: Schistosomatidae). Acta Parasitol. 2021, 1–11. [Google Scholar] [CrossRef] [PubMed]
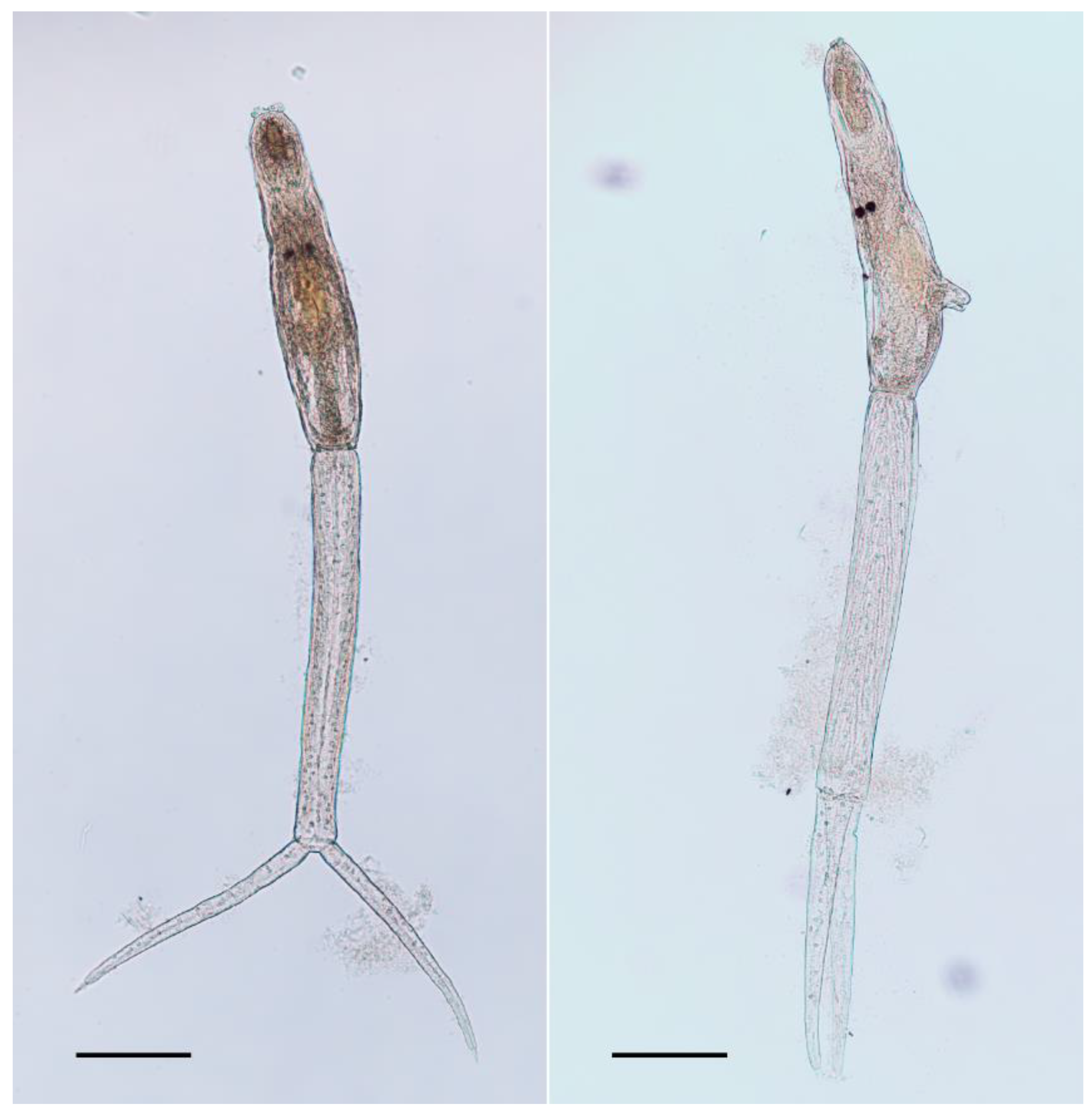
| Reference | Talbot (1936) | Tanaka (1960) | Pence and Rhodes (1982) | This Study |
|---|---|---|---|---|
| Host | Physella parkeri, P. magnalacustris | Radix japonica | Physa anatina | Physella acuta |
| Locality | MI, USA | OI, JPN | TX, USA | UA, AUT |
| Fixation | Formalin | Formalin | AFA | 96% EtOH |
| Length of body | 265 ± 8.4 | 281 | 244 ± 15 | 306.5 ± 3.1 |
| Width of body | 60 ± 4.5 | 51 | 65 ± 4 | 60.5 ± 1.4 |
| Diameter of ventral sucker | 29 ± 2.4 | 27 | 18 ± 3 | 29.2 ± 0.7 |
| Distance from ventral sucker to the posterior end of the body | 80 ± 5.2 | 95 | 68 ± 6 | 83.6 ± 1.2 |
| Length of tail stem | 374 ± 10.6 | 361 | 301 ± 7 | 343.8 ± 3.1 |
| Width of tail stem | 40 ± 3.6 | 35 | 36 ± 4 | 43.5 ± 0.8 |
| Length of tail furca | 196 ± 7.8 | 221 | 157 ± 4 | 225.9 ± 1.8 |
| Width of tail furca | 32 ± 0.9 | 39 | 18 ± 1 | 26.3 ± 0.8 |
| Name | Sequence 5′–3′ | Reference |
|---|---|---|
| Schisto-COI-5-Fw | TCTTTRGATCATAAGCG | [63] |
| Schisto-COI-3-Rv | TAATGCATMGGAAAAAAACA | [63] |
| Tricho_tRNA_fw | GGTTGTCGCTGCTAACGA | This study |
| Tricho_tRNA_rv_2 | CCATATAAAACATTGAAGGAACC | This study |
| Cox1_schist_5_trich | GTTRGTTTCTTTGGATCATAAGCG | This study |
| CO1560R_modif | GCAGTACCAAATTTTCGATC | This study |
| Tricho_Fw2 | GGTTCTGTAAAATTTATAACTAC | This study |
| Tricho_Rv2_2 | CCTAACATATACAACCAAG | This study |
| ZDOE-COI-fw | TAGTTTGTGCTATGGGTTCTATAGT | This study |
| Tricho_rev_20 | GCATTCCTAAATAATGCATAGG | This study |
| LCO1490_ABOL_Moll_1 | TCAACAAAYCATAARGAYATTGG | [64] |
| HCO2198_ABOL_Moll_1 | TAAACTTCTGGRTGACCAAAAAAYCA | [64] |
| Primer Combination | Amplicon Length | Tann/Elongation Time |
|---|---|---|
| Cox1_schist_5k/Cox1_schist_3k | 1244 bp | 50 °C/90 s |
| Tricho_tRNA_fw/Tricho_tRNA_rv_2 | 401–482 bp | 54 °C/60 s |
| Cox1_schist_5_trich/CO1560R_modif | 612 bp | 52 °C/60 s |
| Tricho_Fw2/Tricho_Rv2_2 | 491 bp | 49 °C/60 s |
| ZDOE-COI-fw/Tricho_rev_20 | 486 bp | 53 °C/60 s |
| LCO1490_ABOL_Moll_1/HCO2198_ABOL_Moll _1 | 704 bp | 50 °C/60 s |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Helmer, N.; Blatterer, H.; Hörweg, C.; Reier, S.; Sattmann, H.; Schindelar, J.; Szucsich, N.U.; Haring, E. First Record of Trichobilharzia physellae (Talbot, 1936) in Europe, a Possible Causative Agent of Cercarial Dermatitis. Pathogens 2021, 10, 1473. https://doi.org/10.3390/pathogens10111473
Helmer N, Blatterer H, Hörweg C, Reier S, Sattmann H, Schindelar J, Szucsich NU, Haring E. First Record of Trichobilharzia physellae (Talbot, 1936) in Europe, a Possible Causative Agent of Cercarial Dermatitis. Pathogens. 2021; 10(11):1473. https://doi.org/10.3390/pathogens10111473
Chicago/Turabian StyleHelmer, Nikolaus, Hubert Blatterer, Christoph Hörweg, Susanne Reier, Helmut Sattmann, Julia Schindelar, Nikolaus U. Szucsich, and Elisabeth Haring. 2021. "First Record of Trichobilharzia physellae (Talbot, 1936) in Europe, a Possible Causative Agent of Cercarial Dermatitis" Pathogens 10, no. 11: 1473. https://doi.org/10.3390/pathogens10111473
APA StyleHelmer, N., Blatterer, H., Hörweg, C., Reier, S., Sattmann, H., Schindelar, J., Szucsich, N. U., & Haring, E. (2021). First Record of Trichobilharzia physellae (Talbot, 1936) in Europe, a Possible Causative Agent of Cercarial Dermatitis. Pathogens, 10(11), 1473. https://doi.org/10.3390/pathogens10111473






