Biocontrol of Soil-Borne Pathogens of Solanum lycopersicum L. and Daucus carota L. by Plant Growth-Promoting Actinomycetes: In Vitro and In Planta Antagonistic Activity
Abstract
1. Introduction
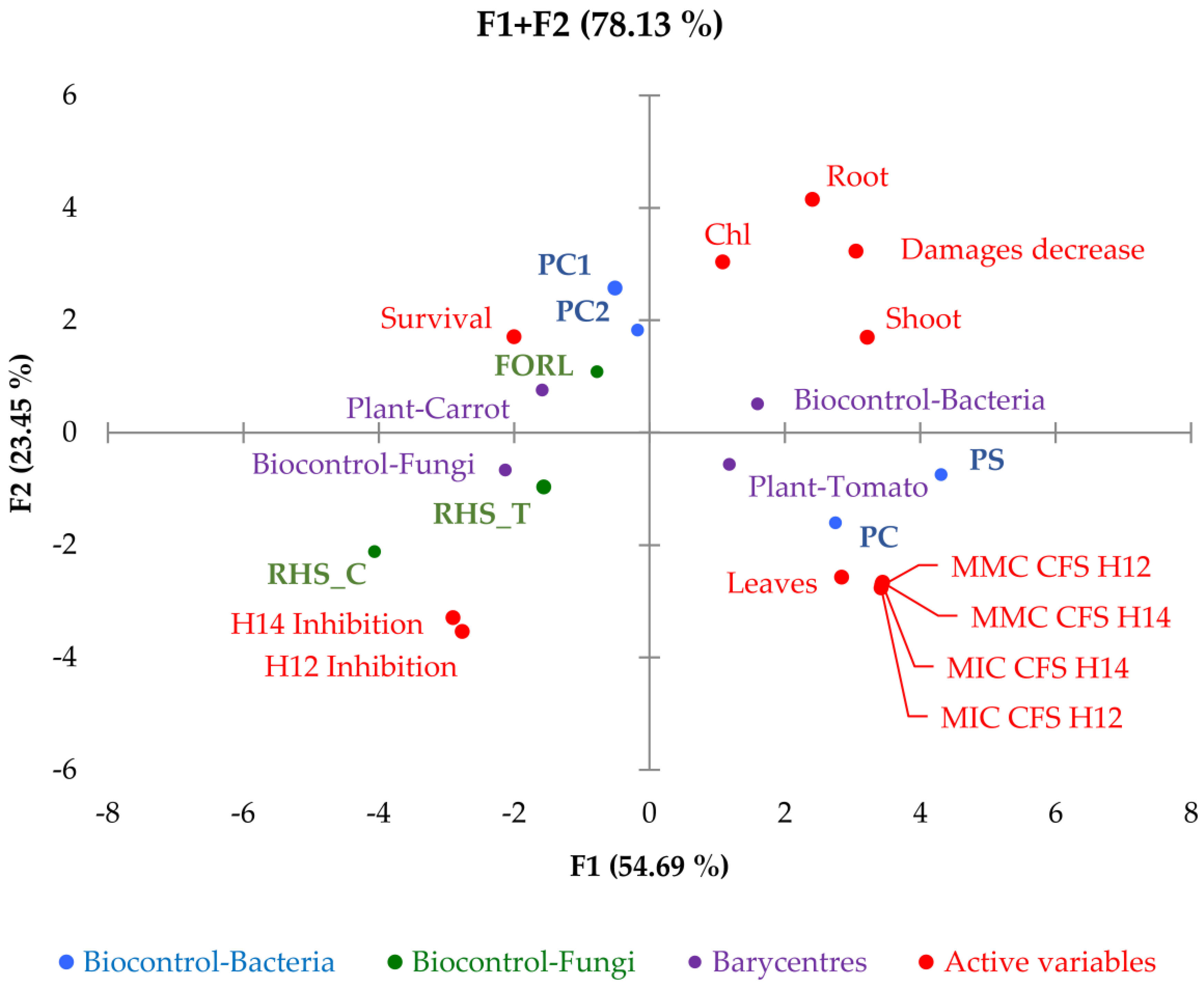
2. Results
2.1. Antibacterial Activity
2.1.1. In Vitro Antibacterial Activity of Selected Actinomycetes
2.1.2. Cell-Free Supernatants Minimum Inhibitory Concentration (MIC) and Maximum Bactericidal Concentration (MBC)
2.1.3. In Planta Biocontrol Activity
2.2. Antifungal Activity
2.2.1. In Vitro Antifungal Activity of Selected Actinomycetes
2.2.2. Actinomycetes Effects on the Hyphal Structure
2.2.3. Cell-Free Supernatants Minimum Inhibitory Concentration (MIC) and Minimum Fungicidal Concentration (MFC)
2.2.4. In Planta Biocontrol Activity
3. Discussion
4. Materials and Methods
- Nocardiopsis aegyptica H14 (MG597543).
- Nocardiopsis aegyptica S2 (MG597572).
- Nocardiopsis alba J21 (MG597576).
- Nocardiopsis dassonvillei subsp. dassonvillei D14 (MG597514).
- Nocardiopsis dassonvillei subsp. dassonvillei T45 (MG597502).
- Streptomyces albidoflavus H12 (MG597552).
- Streptomyces ambofaciens J27 (MG597599).
- Streptomyces anulatus J13 (MG597579).
- Streptomyces iakyrus G10 (MG597593).
- Streptomyces thinghirensis K23 (MG597560).
- Streptomyces thinghirensis J4 (MG597590).
- Streptomyces xantholiticus K12 (MG597545).
- Streptomyces xantholiticus G22 (MG597582).
- Streptomyces xantholiticus G33 (MG597585).
4.1. Antibacterial Activity
4.1.1. In Vitro Antibacterial Activity of Selected Actinomycetes
4.1.2. Cell-Free Supernatants Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
4.1.3. In Planta Antibacterial Activity
- CTL: control, untreated plants.
- CONS: plants inoculated only with actinomycetes.
- CONS + PS/PC/PC1/PC2: plants treated with actinomycetes and infected with a bacterial pathogen.
- PS/PC/PC1/PC2: plants infected only with a bacterial pathogen.
4.2. Antifungal Activity
4.2.1. In Vitro Antifungal Activity of Selected Actinomycetes (Diffusible and Volatile Compounds)
4.2.2. Actinomycetes Effects on the Hyphal Structure
4.2.3. Cell-Free Supernatants Minimum Inhibitory Concentration (MIC) and Minimum Fungicidal Concentration (MFC)
4.2.4. In Planta Antifungal Activity
- CTL: control, untreated plants.
- CONS: plants inoculated only with actinomycetes.
- CONS + FORL/RHS: plants treated with actinomycetes and infected with a fungal pathogen.
- FORL/RHS: plants infected only with a fungal pathogen.
4.3. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Que, F.; Hou, X.-L.; Wang, G.-L.; Xu, Z.-S.; Tan, G.-F.; Li, T.; Wang, Y.-H.; Khadr, A.; Xiong, A.-S. Advances in Research on the Carrot, an Important Root Vegetable in the Apiaceae Family. Hortic. Res. 2019, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, M.; Ercole, C.; Di Zio, C.; Matteucci, F.; Pace, L.; Del Gallo, M. In Vitro and in Planta Antagonistic Effects of Plant Growth-Promoting Rhizobacteria Consortium against Soilborne Plant Pathogens of Solanum tuberosum and Solanum lycopersicum. FEMS Microbiol. Lett. 2020, 367, fnaa099. [Google Scholar] [CrossRef] [PubMed]
- Goudjal, Y.; Zamoum, M.; Sabaou, N.; Mathieu, F.; Zitouni, A. Potential of Endophytic Streptomyces Spp. for Biocontrol of Fusarium Root Rot Disease and Growth Promotion of Tomato Seedlings. Biocontrol Sci. Technol. 2016, 26, 1691–1705. [Google Scholar] [CrossRef]
- Rashad, Y.M.; Al-Askar, A.A.; Ghoneem, K.M.; Saber, W.I.; Hafez, E.E. Chitinolytic Streptomyces griseorubens E44G Enhances the Biocontrol Efficacy against Fusarium Wilt Disease of Tomato. Phytoparasitica 2017, 45, 227–237. [Google Scholar] [CrossRef]
- Goode, M.J.; Sasser, M. Prevention-the Key to Controlling Bacterial Spot and Bacterial Speck of Tomato. Plant Dis. 1980, 64, 831–834. [Google Scholar] [CrossRef]
- Zhang, W.; Luo, Q.; Zhang, Y.; Fan, X.; Ye, T.; Mishra, S.; Bhatt, P.; Zhang, L.; Chen, S. Quorum Quenching in a Novel Acinetobacter Sp. XN-10 Bacterial Strain against Pectobacterium carotovorum subsp. carotovorum. Microorganisms 2020, 8, 1100. [Google Scholar] [CrossRef]
- Goudjal, Y.; Toumatia, O.; Yekkour, A.; Sabaou, N.; Mathieu, F.; Zitouni, A. Biocontrol of Rhizoctonia solani Damping-off and Promotion of Tomato Plant Growth by Endophytic Actinomycetes Isolated from Native Plants of Algerian Sahara. Microbiol. Res. 2014, 169, 59–65. [Google Scholar] [CrossRef]
- Debasis, M.; Ganeshamurthy, A.N.; Komal, S.; Radha, T.K.; Rupa, T.R. Plant Growth Promotion and Biocontrol Activity of Some Typical Harsh Environment Rhizo-Microbes and Their Effects on Amaranthus cruentus Plants. Res. J. Biotechnol. Vol 2020, 15, 1. [Google Scholar]
- Berendsen, R.L.; Pieterse, C.M.; Bakker, P.A. The Rhizosphere Microbiome and Plant Health. Trends Plant Sci. 2012, 17, 478–486. [Google Scholar] [CrossRef]
- Cao, P.; Liu, C.; Sun, P.; Fu, X.; Wang, S.; Wu, F.; Wang, X. An Endophytic Streptomyces Sp. Strain DHV3-2 from Diseased Root as a Potential Biocontrol Agent against Verticillium dahliae and Growth Elicitor in Tomato (Solanum Lycopersicum). Antonie Van Leeuwenhoek 2016, 109, 1573–1582. [Google Scholar] [CrossRef]
- Marimuthu, S.; Karthic, C.; Mostafa, A.A.; Al-Enazi, N.M.; Abdel-Raouf, N.; Sholkamy, E.N. Antifungal Activity of Streptomyces Sp. SLR03 against Tea Fungal Plant Pathogen Pestalotiopsis theae. J. King Saud Univ. Sci. 2020, 32, 3258–3264. [Google Scholar] [CrossRef]
- Chaurasia, A.; Meena, B.R.; Tripathi, A.N.; Pandey, K.K.; Rai, A.B.; Singh, B. Actinomycetes: An Unexplored Microorganisms for Plant Growth Promotion and Biocontrol in Vegetable Crops. World J. Microbiol. Biotechnol. 2018, 34, 132. [Google Scholar] [CrossRef] [PubMed]
- Vurukonda, S.S.K.P.; Giovanardi, D.; Stefani, E. Plant Growth Promoting and Biocontrol Activity of Streptomyces Spp. as Endophytes. Int. J. Mol. Sci. 2018, 19, 952. [Google Scholar] [CrossRef]
- Doumbou, C.L.; Hamby Salove, M.K.; Crawford, D.L.; Beaulieu, C. Actinomycetes, Promising Tools to Control Plant Diseases and to Promote Plant Growth. Phytoprotection 2001, 82, 85–102. [Google Scholar] [CrossRef]
- Djebaili, R.; Pellegrini, M.; Smati, M.; Del Gallo, M.; Kitouni, M. Actinomycete Strains Isolated from Saline Soils: Plant-Growth-Promoting Traits and Inoculation Effects on Solanum lycopersicum. Sustainability 2020, 12, 4617. [Google Scholar] [CrossRef]
- Djebaili, R.; Pellegrini, M.; Rossi, M.; Forni, C.; Smati, M.; Del Gallo, M.; Kitouni, M. Characterization of Plant Growth-Promoting Traits and Inoculation Effects on Triticum Durum of Actinomycetes Isolates under Salt Stress Conditions. Soil Syst. 2021, 5, 26. [Google Scholar] [CrossRef]
- Alper, D.; Güven, K.; Şahin, N. Isolation, Plant Growth-Promoting Traits, Antagonistic Effects on Clinical and Plant Pathogenic Organisms and Identification of Actinomycetes from Olive Rhizosphere. Microb. Pathog. 2020, 143, 104134. [Google Scholar]
- Glöckner, F.O.; Zaichikov, E.; Belkova, N.; Denissova, L.; Pernthaler, J.; Pernthaler, A.; Amann, R. Comparative 16S RRNA Analysis of Lake Bacterioplankton Reveals Globally Distributed Phylogenetic Clusters Including an Abundant Group of Actinobacteria. Appl. Environ. Microbiol. 2000, 66, 5053–5065. [Google Scholar] [CrossRef]
- Abd-Allah, E.F. Streptomyces Plicatus as a Model Biocontrol Agent. Folia Microbiol. 2001, 46, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Anitha, A.; Rabeeth, M. Control of Fusarium Wilt of Tomato by Bioformulation of Streptomyces griseus in Green House Condition. Afr. J. Basic Appl. Sci. 2009, 1, 9–14. [Google Scholar]
- Kim, H.-R.; Song, H.-G. Antifungal Activity of Streptomyces costaricanus HR391 against Some Plant-Pathogenic Fungi. Korean J. Microbiol. 2016, 52, 437–443. [Google Scholar] [CrossRef][Green Version]
- Kim, Y.J.; Kim, J.; Rho, J.-Y. Antifungal Activities of Streptomyces blastmyceticus Strain 12-6 Against Plant Pathogenic Fungi. Mycobiology 2019, 47, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Ningthoujam, S.; Sanasam, S.; Tamreihao, K.; Nimaich, S. Antagonistic Activities of Local Actinomycete Isolates against Rice Fungal Pathogens. Afr. J. Microbiol. Res. 2009, 3, 737–742. [Google Scholar]
- Song, L.; Jiang, N.; Wei, S.; Lan, Z.; Pan, L. Isolation, Screening, and Identification of Actinomycetes with Antifungal and Enzyme Activity Assays against Colletotrichum dematium of Sarcandra glabra. Mycobiology 2020, 48, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Tlemsani, M.; Fortas, Z.; Dib, S.; Bellahcen, M. In Vitro Antagonism between Actinomycete Isolates and Fusarium oxysporum f. sp. ciceri: The Causative Agent of Chickpea Vascular Wilt. South Asian J. Exp. Biol. 2020, 10, 255–267. [Google Scholar] [CrossRef]
- Errakhi, R.; Bouteau, F.; Lebrihi, A.; Barakate, M. Evidences of Biological Control Capacities of Streptomyces Spp. against Sclerotium rolfsii Responsible for Damping-off Disease in Sugar Beet (Beta Vulgaris L.). World J. Microbiol. Biotechnol. 2007, 23, 1503–1509. [Google Scholar] [CrossRef]
- Jayasinghe, B.D.; Parkinson, D. Actinomycetes as Antagonists of Litter Decomposer Fungi. Appl. Soil Ecol. 2008, 38, 109–118. [Google Scholar] [CrossRef]
- Benhadj, M.; Metrouh, R.; Menasria, T.; Gacemi-Kirane, D.; Slim, F.Z.; Ranque, S. Broad-Spectrum Antimicrobial Activity of Wetland-Derived Streptomyces Sp. ActiF450. EXCLI J. 2020, 19, 360. [Google Scholar]
- Wang, M.; Xue, J.; Ma, J.; Feng, X.; Ying, H.; Xu, H. Streptomyces Lydicus M01 Regulates Soil Microbial Community and Alleviates Foliar Disease Caused by Alternaria alternata on Cucumbers. Front. Microbiol. 2020, 11, 942. [Google Scholar] [CrossRef]
- Oskay, A.M.; Üsame, T.; Cem, A. Antibacterial Activity of Some Actinomycetes Isolated from Farming Soils of Turkey. Afr. J. Biotechnol. 2004, 3, 441–446. [Google Scholar]
- Zhao, J.; Han, L.; Yu, M.; Cao, P.; Li, D.; Guo, X.; Liu, Y.; Wang, X.; Xiang, W. Characterization of Streptomyces sporangiiformans sp. vov., a Novel Soil Actinomycete with Antibacterial Activity against Ralstonia solanacearum. Microorganisms 2019, 7, 360. [Google Scholar] [CrossRef] [PubMed]
- Pacios-Michelena, S.; Aguilar González, C.N.; Alvarez-Perez, O.B.; Rodriguez-Herrera, R.; Chávez-González, M.; Arredondo Valdés, R.; Ascacio Valdés, J.A.; Govea Salas, M.; Ilyina, A. Application of Streptomyces Antimicrobial Compounds for the Control of Phytopathogens. Front. Sustain. Food Syst. 2021, 5, 310. [Google Scholar] [CrossRef]
- Getha, K.; Vikineswary, S. Antagonistic Effects of Streptomyces violaceusniger Strain G10 on Fusarium oxysporum f. sp. cubense Race 4: Indirect Evidence for the Role of Antibiosis in the Antagonistic Process. J. Ind. Microbiol. Biotechnol. 2002, 28, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Ma, Z.; Xu, X.; Yu, X. Isolation and Identification of Biocontrol Agent Streptomyces Rimosus M527 against Fusarium oxysporum f. sp. cucumerinum. J. Basic Microbiol. 2016, 56, 929–933. [Google Scholar] [CrossRef] [PubMed]
- Manasa, M.; Yashoda, K.; Pallavi, S.; Vivek, M.N.; Onkarappa, R.; Kekuda, T.R.P. Biocontrol Potential of Streptomyces Species against Fusarium oxysporum f. sp. zingiberi (Causal Agent of Rhizome Rot of Ginger). J. Adv. Sci. Res. 2013, 4, 1–3. [Google Scholar]
- Saengnak, V.; Chaisiri, C.; Nalumpang, S. Antagonistic Streptomyces Species Can Protect Chili Plants against Wilt Disease Caused by Fusarium. J. Agric. Technol. 2013, 9, 1895–1908. [Google Scholar]
- Wei, Y.; Zhao, Y.; Zhou, D.; Qi, D.; Li, K.; Tang, W.; Chen, Y.; Jing, T.; Zang, X.; Xie, J. A Newly Isolated Streptomyces Sp. YYS-7 With a Broad-Spectrum Antifungal Activity Improves the Banana Plant Resistance to Fusarium oxysporum f. sp. cubense Tropical Race 4. Front. Microbiol. 2020, 11, 1712. [Google Scholar] [CrossRef]
- Cao, L.; Qiu, Z.; You, J.; Tan, H.; Zhou, S. Isolation and Characterization of Endophytic Streptomyces Strains from Surface-Sterilized Tomato (Lycopersicon esculentum) Roots. Lett. Appl. Microbiol. 2004, 39, 425–430. [Google Scholar] [CrossRef]
- Khair, A. In Vitro Antifungal Activity of Streptomyces Spororaveus RDS28 against Some Phytopathogenic Fungi. Afr. J. Agric. Res. 2011, 6, 2835–2842. [Google Scholar]
- Patil, H.J.; Srivastava, A.K.; Singh, D.P.; Chaudhari, B.L.; Arora, D.K. Actinomycetes Mediated Biochemical Responses in Tomato (Solanum lycopersicum) Enhances Bioprotection against Rhizoctonia solani. Crop Prot. 2011, 30, 1269–1273. [Google Scholar] [CrossRef]
- Patil, H.J.; Srivastava, A.K.; Kumar, S.; Chaudhari, B.L.; Arora, D.K. Selective Isolation, Evaluation and Characterization of Antagonistic Actinomycetes against Rhizoctonia solani. World J. Microbiol. Biotechnol. 2010, 26, 2163–2170. [Google Scholar] [CrossRef]
- Singh, S.P.; Gupta, R.; Gaur, R.; Srivastava, A.K. Antagonistic Actinomycetes Mediated Resistance in Solanum lycopersicon Mill. against Rhizoctonia solani Kühn. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2017, 87, 789–798. [Google Scholar] [CrossRef]
- Yandigeri, M.S.; Malviya, N.; Solanki, M.K.; Shrivastava, P.; Sivakumar, G. Chitinolytic Streptomyces vinaceusdrappus S5MW2 Isolated from Chilika Lake, India Enhances Plant Growth and Biocontrol Efficacy through Chitin Supplementation against Rhizoctonia solani. World J. Microbiol. Biotechnol. 2015, 31, 1217–1225. [Google Scholar] [CrossRef]
- Soltanzadeh, M.; Soltani Nejad, M.; Shahidi Bonjar, G.H. Application of Soil-Borne Actinomycetes for Biological Control against Fusarium Wilt of Chickpea (Cicer Arietinum) Caused by Fusarium solani fsp pisi. J. Phytopathol. 2016, 164, 967–978. [Google Scholar] [CrossRef]
- Pellegrini, M.; Pagnani, G.; Bernardi, M.; Mattedi, A.; Spera, D.M.; Gallo, M.D. Cell-Free Supernatants of Plant Growth-Promoting Bacteria: A Review of Their Use as Biostimulant and Microbial Biocontrol Agents in Sustainable Agriculture. Sustainability 2020, 12, 9917. [Google Scholar] [CrossRef]
- Gu, L.; Zhang, K.; Zhang, N.; Li, X.; Liu, Z. Control of the Rubber Anthracnose Fungus Colletotrichum gloeosporioides Using Culture Filtrate Extract from Streptomyces deccanensis QY-3. Antonie Van Leeuwenhoek 2020, 113, 1573–1585. [Google Scholar] [CrossRef]
- Faheem, M.; Raza, W.; Zhong, W.; Nan, Z.; Shen, Q.; Xu, Y. Evaluation of the Biocontrol Potential of Streptomyces goshikiensis YCXU against Fusarium oxysporum f. sp. niveum. Biol. Control 2015, 81, 101–110. [Google Scholar] [CrossRef]
- Li, Y.; Guo, Q.; He, F.; Li, Y.; Xue, Q.; Lai, H. Biocontrol of Root Diseases and Growth Promotion of the Tuberous Plant Aconitum carmichaelii Induced by Actinomycetes Are Related to Shifts in the Rhizosphere Microbiota. Microb. Ecol. 2020, 79, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Alekhya, G.; Gopalakrishnan, S. Characterization of Antagonistic Streptomyces as Potential Biocontrol Agent against Fungal Pathogens of Chickpea and Sorghum. Philipp. Agric. Sci. 2014, 97, 191–198. [Google Scholar]
- Lyu, A.; Liu, H.; Che, H.; Yang, L.; Zhang, J.; Wu, M.; Chen, W.; Li, G. Reveromycins A and B from Streptomyces sp. 3–10: Antifungal Activity against Plant Pathogenic Fungi In Vitro and in a Strawberry Food Model System. Front. Microbiol. 2017, 8, 550. [Google Scholar] [CrossRef]
- Bennur, T.; Kumar, A.R.; Zinjarde, S.; Javdekar, V. Nocardiopsis Species: Incidence, Ecological Roles and Adaptations. Microbiol. Res. 2015, 174, 33–47. [Google Scholar] [CrossRef]
- Intra, B.; Mungsuntisuk, I.; Nihira, T.; Igarashi, Y.; Panbangred, W. Identification of Actinomycetes from Plant Rhizospheric Soils with Inhibitory Activity against Colletotrichum spp., the Causative Agent of Anthracnose Disease. BMC Res. Notes 2011, 4, 98. [Google Scholar] [CrossRef] [PubMed]
- Sabu, R.; Soumya, K.R.; Radhakrishnan, E.K. Endophytic Nocardiopsis sp. from Zingiber Officinale with Both Antiphytopathogenic Mechanisms and Antibiofilm Activity against Clinical Isolates. 3 Biotech 2017, 7, 115. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.-Z.; Pu, X.; Luo, G.; Zhao, L.-X.; Xu, L.-H.; Li, W.-J.; Luo, Y. Isolation and Characterization of New P-Terphenyls with Antifungal, Antibacterial, and Antioxidant Activities from Halophilic Actinomycete Nocardiopsis gilva YIM 90087. J. Agric. Food Chem. 2013, 61, 3006–3012. [Google Scholar] [CrossRef] [PubMed]
- Bora, L.C.; Lohit, K.; Kuldeep, T.; Parveen, K.; Joli, D.; Rinku, S. Yield Enhancement and Bacteiral Wilt Suppression in Bhut Jolokia (Capsicum Assamicum) Using Consortia of Microbial Antagonists. Biopestic. Int. 2016, 12, 165–171. [Google Scholar]
- El_Komy, M.H.; Hassouna, M.G.; Abou-Taleb, E.M.; Al-Sarar, A.S.; Abobakr, Y. A Mixture of Azotobacter, Azospirillum, and Klebsiella Strains Improves Root-Rot Disease Complex Management and Promotes Growth in Sunflowers in Calcareous Soil. Eur. J. Plant Pathol. 2020, 156, 713–726. [Google Scholar] [CrossRef]
- Saikia, K.; Bora, L.C. Exploring Actinomycetes and Endophytes of Rice Ecosystem for Induction of Disease Resistance against Bacterial Blight of Rice. Eur. J. Plant Pathol. 2020, 159, 1–13. [Google Scholar] [CrossRef]
- Verma, P.P.; Shelake, R.M.; Sharma, P.; Kim, J.-Y.; Das, S.; Kaur, M. Microbes in Crop Production: Formulation and Application. In Microbial Diversity, Interventions and Scope; Springer: Berlin/Heidelberg, Germany, 2020; pp. 51–73. [Google Scholar]
- Hata, E.M.; Sijam, K.; Ahmad, Z.A.M.; Yusof, M.T.; Azman, N.A. In Vitro Antimicrobial Assay of Actinomycetes in Rice Against Xanthomonas oryzae pv. oryzicola and as Potential Plant Growth Promoter. Braz. Arch. Biol. Technol. 2015, 58, 821–832. [Google Scholar] [CrossRef]
- El-Shatoury, S.A.; Ameen, F.; Moussa, H.; Wahid, O.A.; Dewedar, A.; AlNadhari, S. Biocontrol of Chocolate Spot Disease (Botrytis cinerea) in Faba Bean Using Endophytic Actinomycetes Streptomyces: A Field Study to Compare Application Techniques. PeerJ 2020, 8, e8582. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; Van Wees, S.C.M.; Bakker, P.A.H.M. Induced Systemic Resistance by Beneficial Microbes. Annu. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef]
- Smati, M.; Kitouni, M. Diversity of Actinobacteria in the Marshes of Ezzemoul and Djendli in Northeastern Algeria. Eur. J. Ecol. 2019, 5, 41–53. [Google Scholar] [CrossRef][Green Version]
- Wayne, P.A. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing. Inf. Suppl. 2011, 31, 100–121. [Google Scholar]
- Teh, C.H.; Nazni, W.A.; Norazah, A.; Lee, H.L. Determination of Antibacterial Activity and Minimum Inhibitory Concentration of Larval Extract of Fly via Resazurin-Based Turbidometric Assay. BMC Microbiol. 2017, 17, 36. [Google Scholar] [CrossRef] [PubMed]



| SiG10 | SxG33 | SaH12 | NaH14 | SaJ13 | SaJ27 | NaS2 | |
|---|---|---|---|---|---|---|---|
| P. syringae | + | + | +++ | +++ | + | − | − |
| P. corrugata | ++ | ++ | +++ | +++ | ++ | − | ++ |
| Pc. carotovorum PC1 | ++ | ++ | +++ | +++ | ++ | + | − |
| Pc. carotovorum PC2 | ++ | ++ | +++ | +++ | ++ | + | ++ |
| Pathogenic Bacteria | SaH12 CFS | NaH14 CFS | SaH12 + NaH14 CFS | |||
|---|---|---|---|---|---|---|
| MIC | MBC | MIC | MBC | MIC | MBC | |
| P. syringae | 25.5 | 50.0 | 50.0 | 50.0 | 25.0 | 50.0 |
| P. corrugata | 25.5 | 50.0 | 50.0 | 50.0 | 25.0 | 50.0 |
| Pc. carotovorum PC1 | - | - | - | - | 50.0 | - |
| Pc. carotovorum PC2 | 0.8 | 0.8 | 0.8 | 1.6 | 0.8 | 0.8 |
| Survival (%) | Leaves (n°) | Damage (Grade) | Root (cm) | Shoot (cm) | Chlorophyll (mg g FW−1) | |
|---|---|---|---|---|---|---|
| Tomato | ||||||
| CTL | 85.90 b | 3.40 c | - | 2.30 c | 7.04 c | 0.94 b |
| CONS | 98.80 a | 5.30 a | - | 3.99 a | 11.09 a | 1.08 a |
| CONS + PS | 64.70 c | 4.80 a,b | 2.80 c | 2.96 b | 9.32 b | 1.08 a |
| CONS + PC | 87.00 b | 4.60 b | 3.50 b | 2.79 b | 8.89 b | 1.00 b |
| PS | 60.70 e | 2.70 d | 2.50 a | 2.02 d | 6.06 e | 0.62 c |
| PC | 50.40 d | 2.20 d | 4.70 c | 1.25 c | 4.15 d | 0.62 c |
| LSD | 1.24 | 0.63 | 0.47 | 0.37 | 0.88 | 0.06 |
| Carrot | ||||||
| CTL | 88.00 B | 2.00 B | - | 1.79 B | 2.60 D,E | 0.89 C |
| CONS | 98.60 A | 3.70 A | - | 3.35 A | 4.90 A | 1.10 A |
| CONS + PC1 | 86.80 B | 2.00 B | 2.50 B | 3.08 A | 4.27 B | 1.08 A |
| CONS + PC2 | 40.80 C | 2.00 B | 2.50 B | 3.07 A | 3.78 C | 0.99 B |
| PC1 | 40.20 C | 2.00 B | 4.80 A | 1.40 B | 2.65 D | 0.61 D |
| PC2 | 27.40 D | 2.00 B | 4.50 A | 1.63 B | 2.16 E | 0.61 D |
| LSD | 1.40 | 0.18 | 0.37 | 0.40 | 0.48 | 0.07 |
| Pathogenic Bacteria | SaH12 CFS | NaH14 CFS | SaH12 + NaH14 CFS | |||
|---|---|---|---|---|---|---|
| MIC | MFC | MIC | MFC | MIC | MFC | |
| Fusarium oxysporum | 0.8 | 0.8 | 0.4 | 0.4 | 0.4 | 0.4 |
| Rhizoctonia solani | 0.8 | 0.8 | 0.2 | 0.4 | 0.4 | 0.4 |
| Survival (%) | Leaves (n°) | Damage (Grade) | Root (cm) | Shoot (cm) | Chlorophyll (mg g FW−1) | |
|---|---|---|---|---|---|---|
| Tomato | ||||||
| CONS | 98.40 a | 6.00 a | - | 3.85 a | 10.71 a | 1.10 a |
| CTL | 86.40 b | 3.60 b,c | - | 2.98 c | 7.71 b | 0.94 b |
| CONS + FORL | 78.50 c | 4.00 b | 2.60 b | 3.51 b | 6.15 c | 1.07 a |
| CONS + RHS | 60.30 d | 3.40 c | 2.60 b | 3.37 b | 5.90 c | 0.72 c |
| FORL | 53.60 e | 2.60 d | 4.60 a | 1.76 e | 4.69 d | 0.39 d |
| RHS | 49.60 f | 2.00 e | 4.60 a | 2.23 d | 3.93 e | 0.42 d |
| LSD | 1.30 | 0.56 | 0.38 | 0.25 | 0.51 | 0.09 |
| Carrot | ||||||
| CONS | 98.70 A | 3.70 A | - | 3.10 A | 4.89 A | 0.84 A |
| CTL | 88.00 B | 2.00 C | - | 2.30 B | 3.20 B | 0.94 A |
| CONS + RHS | 44.30 C | 2.50 B | 2.70 B | 2.40 B | 4.80 A | 1.06 A |
| RHS | 20.20 D | 1.50 D | 4.80 A | 1.43 C | 2.60 C | 0.57 B |
| LSD | 2.13 | 0.40 | 0.36 | 0.28 | 0.52 | 0.24 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Djebaili, R.; Pellegrini, M.; Ercole, C.; Farda, B.; Kitouni, M.; Del Gallo, M. Biocontrol of Soil-Borne Pathogens of Solanum lycopersicum L. and Daucus carota L. by Plant Growth-Promoting Actinomycetes: In Vitro and In Planta Antagonistic Activity. Pathogens 2021, 10, 1305. https://doi.org/10.3390/pathogens10101305
Djebaili R, Pellegrini M, Ercole C, Farda B, Kitouni M, Del Gallo M. Biocontrol of Soil-Borne Pathogens of Solanum lycopersicum L. and Daucus carota L. by Plant Growth-Promoting Actinomycetes: In Vitro and In Planta Antagonistic Activity. Pathogens. 2021; 10(10):1305. https://doi.org/10.3390/pathogens10101305
Chicago/Turabian StyleDjebaili, Rihab, Marika Pellegrini, Claudia Ercole, Beatrice Farda, Mahmoud Kitouni, and Maddalena Del Gallo. 2021. "Biocontrol of Soil-Borne Pathogens of Solanum lycopersicum L. and Daucus carota L. by Plant Growth-Promoting Actinomycetes: In Vitro and In Planta Antagonistic Activity" Pathogens 10, no. 10: 1305. https://doi.org/10.3390/pathogens10101305
APA StyleDjebaili, R., Pellegrini, M., Ercole, C., Farda, B., Kitouni, M., & Del Gallo, M. (2021). Biocontrol of Soil-Borne Pathogens of Solanum lycopersicum L. and Daucus carota L. by Plant Growth-Promoting Actinomycetes: In Vitro and In Planta Antagonistic Activity. Pathogens, 10(10), 1305. https://doi.org/10.3390/pathogens10101305









