Figure 1.
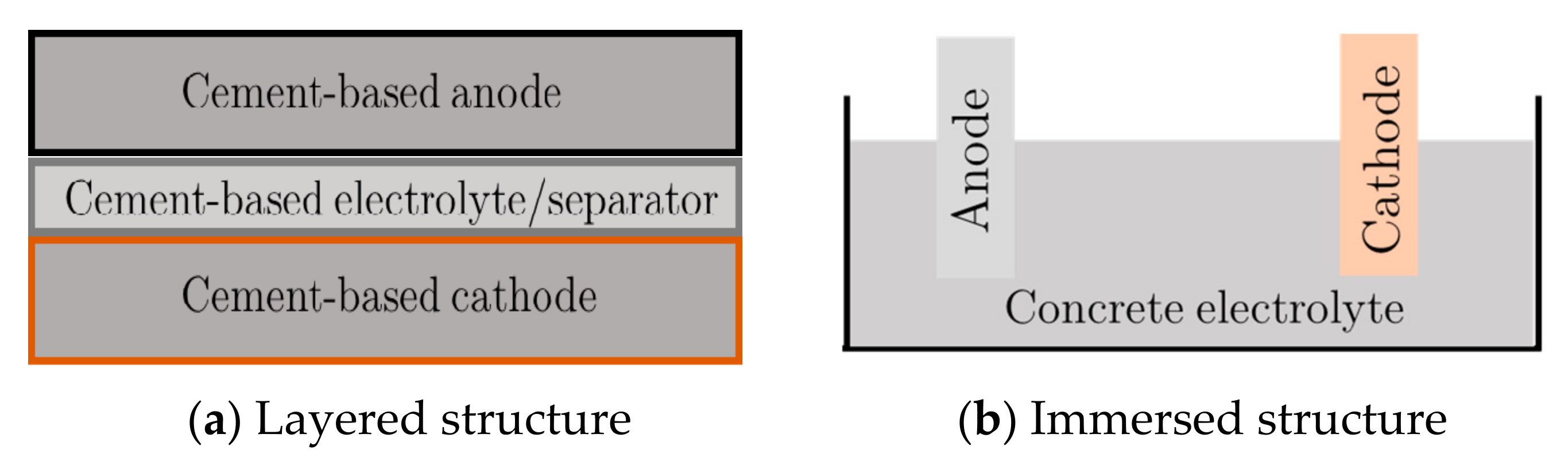
Two common cell arrangements of the concrete batteries: (a) the layered structure and (b) the immersed structure.
Figure 1.
Two common cell arrangements of the concrete batteries: (a) the layered structure and (b) the immersed structure.
Figure 2.
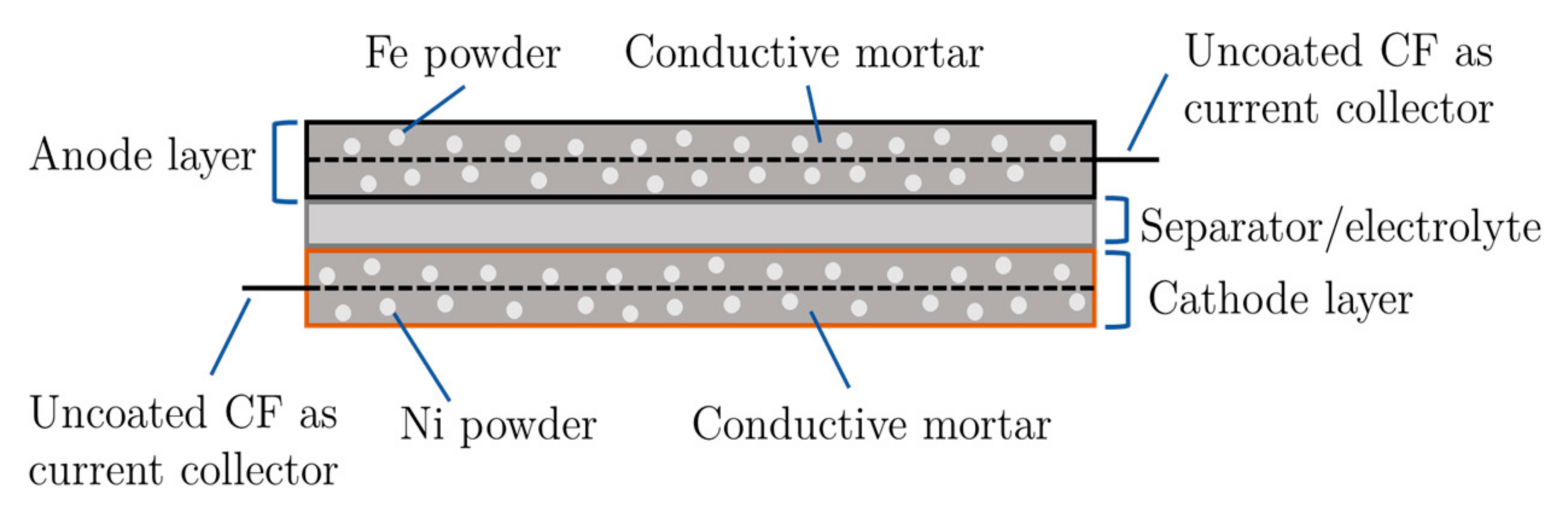
Schematic illustration of the design of the powder-mixing battery.
Figure 2.
Schematic illustration of the design of the powder-mixing battery.
Figure 3.
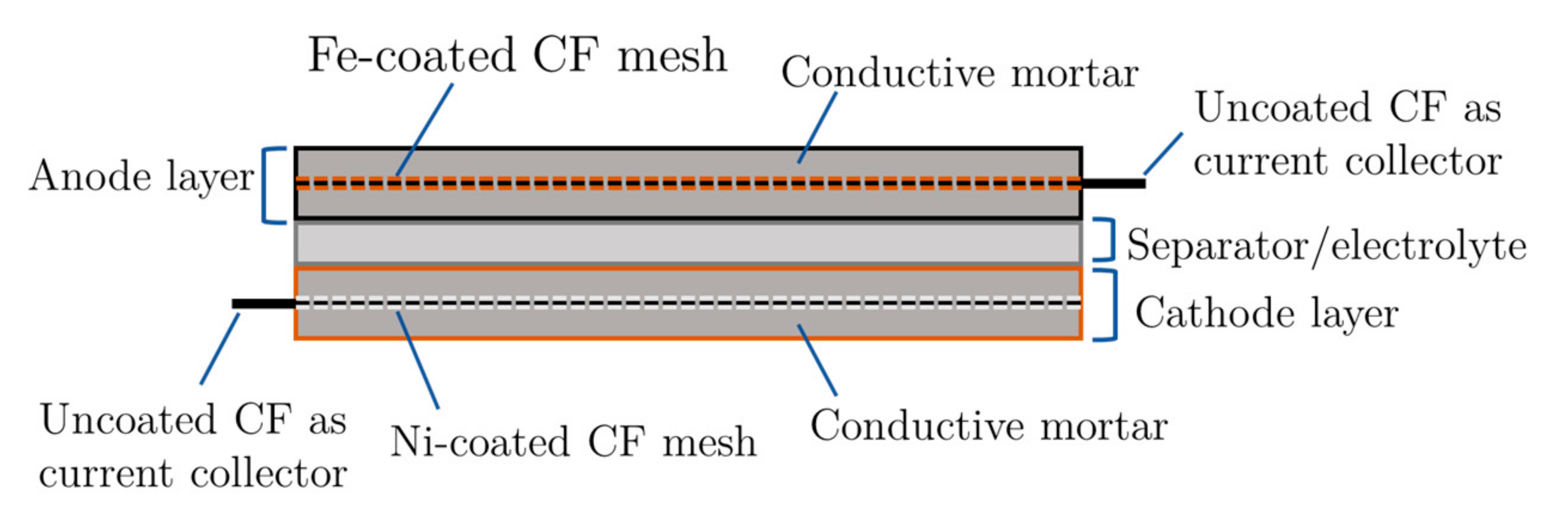
Schematic illustration of the design of the metal-coating battery.
Figure 3.
Schematic illustration of the design of the metal-coating battery.
Figure 4.
Images of metal-coated CF meshes (a–c) and an example of coated-metal CF mesh cast inside the cement-based electrolyte before demolding from the white plastic foam (d). The mixture of mortar is the same for the powder-mixing mortar.
Figure 4.
Images of metal-coated CF meshes (a–c) and an example of coated-metal CF mesh cast inside the cement-based electrolyte before demolding from the white plastic foam (d). The mixture of mortar is the same for the powder-mixing mortar.
Figure 5.
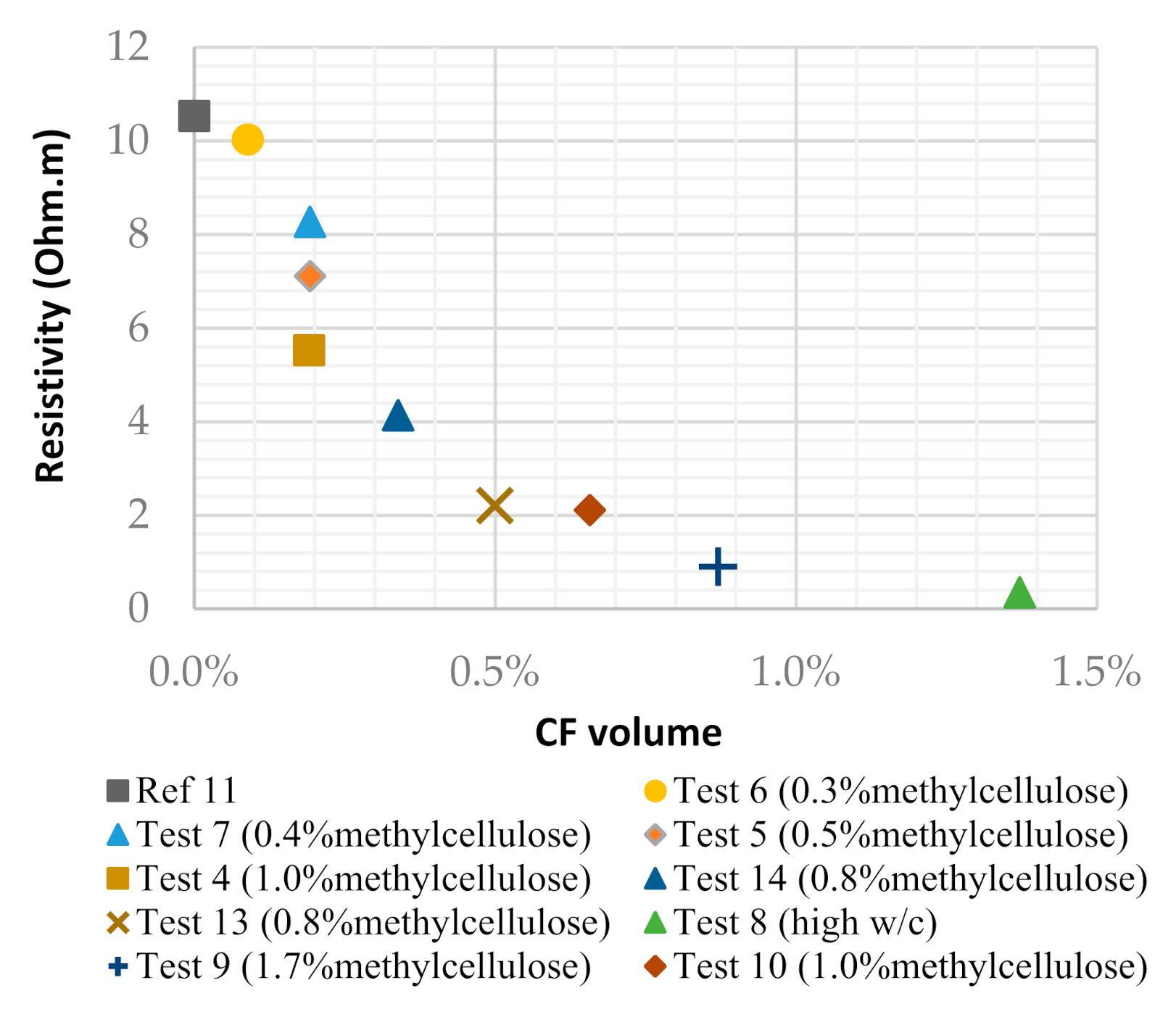
Resistivity of the cement-based solid electrolyte using different mixtures.
Figure 5.
Resistivity of the cement-based solid electrolyte using different mixtures.
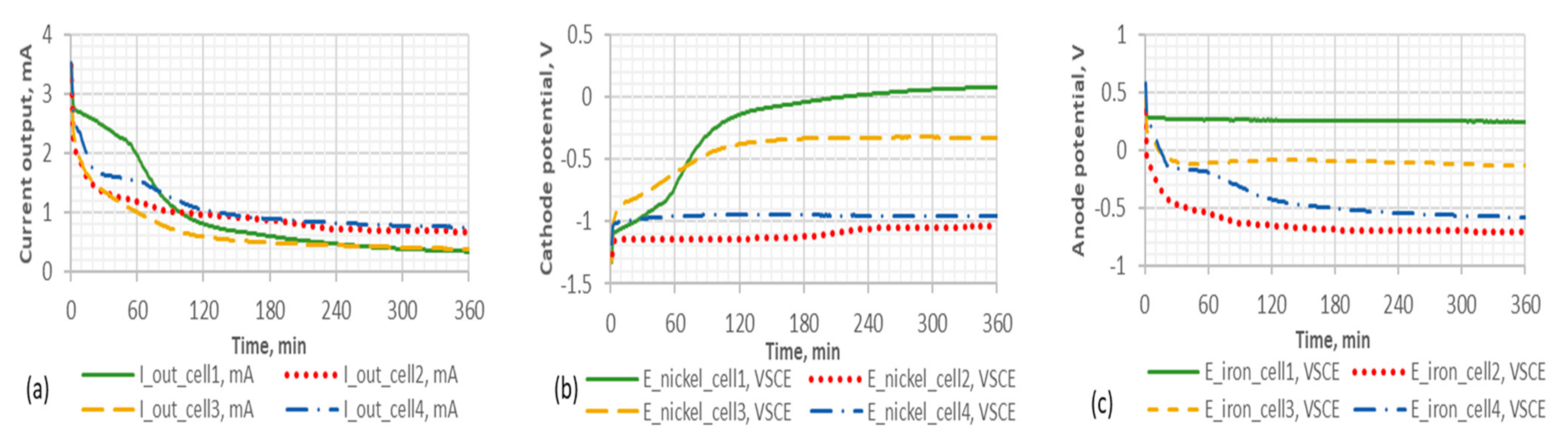
Figure 6.
Current and potential profiles of the powder-mixing electrodes through a 500 Ω resistor for each cell. The aarrangements of the cells are listed in
Table 10. Subfigure (
a) represents the current profile, (
b) for the cathode profile and (
c) for the anode profile.
Figure 6.
Current and potential profiles of the powder-mixing electrodes through a 500 Ω resistor for each cell. The aarrangements of the cells are listed in
Table 10. Subfigure (
a) represents the current profile, (
b) for the cathode profile and (
c) for the anode profile.
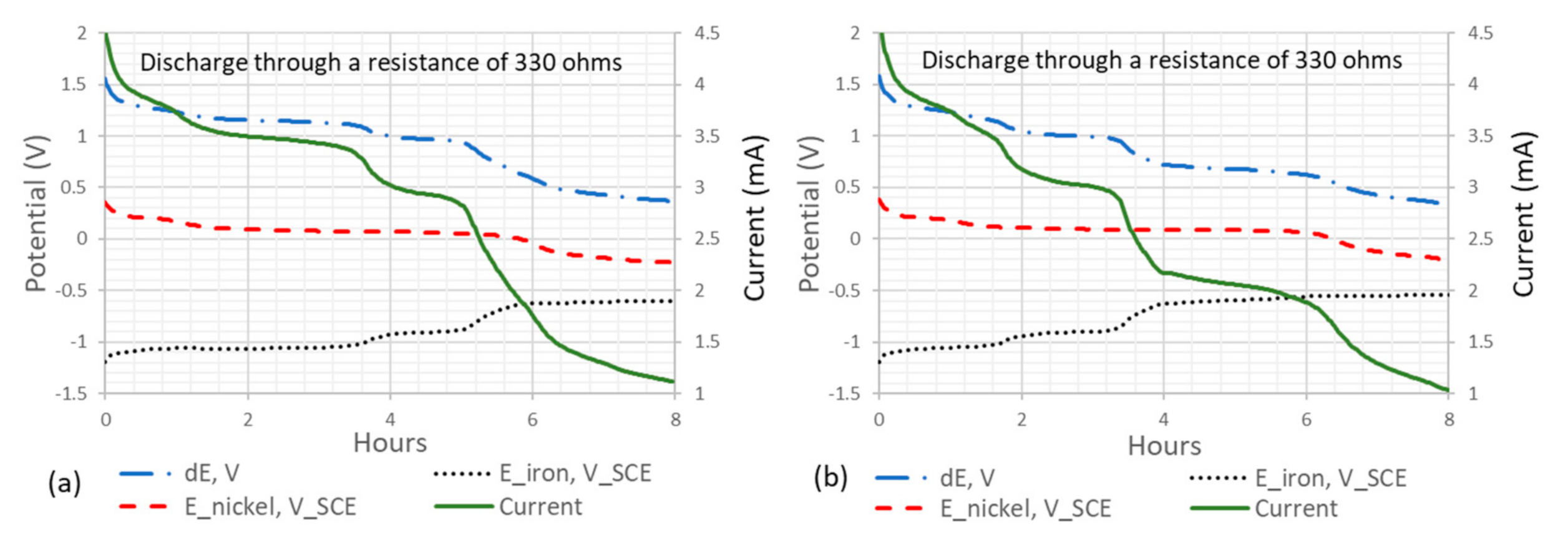
Figure 7.
Potential and current profiles of the metal-coating Ni-CF cement-based anode against a pure iron plate cathode over two 8-h discharge cycles. Measurements include the potential difference between the anode and cathode (dE), the potential of the iron plate as the cathode (E_iron, V_SCE), the potential of the Ni-CF anode (E_nickel, V_SCE), and the current with a 330 Ω resistor. Subfigure (a) represents discharge cycle one and (b) discharge cycle two.
Figure 7.
Potential and current profiles of the metal-coating Ni-CF cement-based anode against a pure iron plate cathode over two 8-h discharge cycles. Measurements include the potential difference between the anode and cathode (dE), the potential of the iron plate as the cathode (E_iron, V_SCE), the potential of the Ni-CF anode (E_nickel, V_SCE), and the current with a 330 Ω resistor. Subfigure (a) represents discharge cycle one and (b) discharge cycle two.
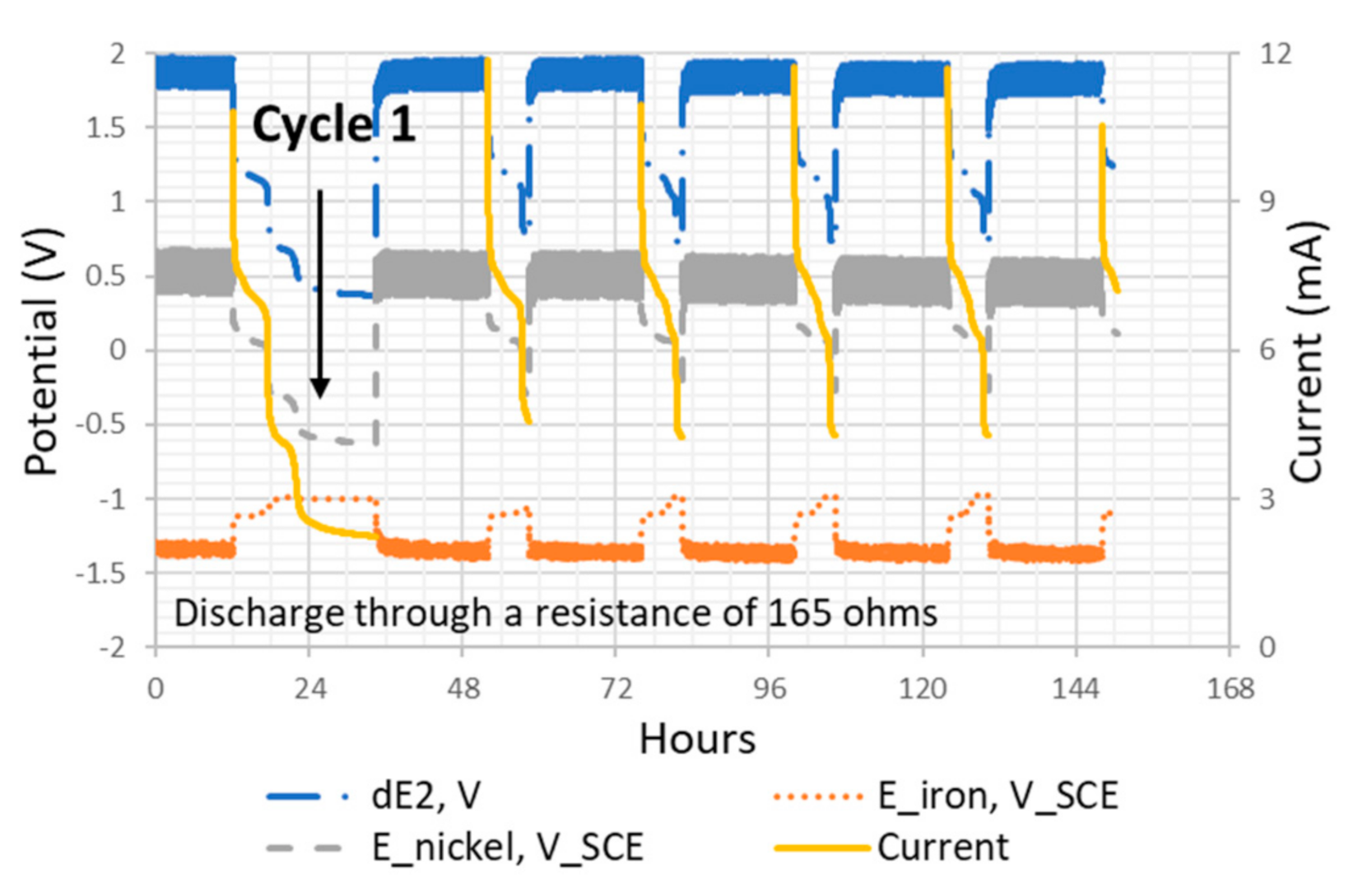
Figure 8.
The potential and current profiles of the Ni–Fe cell with a 165 Ω resistor. The first discharge is enlarged and presented in
Figure 9. Measurements include the potential difference between the cathode and anode (dE2), the potential of the iron plate as anode (E_iron, V_
SCE), the potential of the Ni–CF cathode (E_nickel, V_
SCE), and the current with a 165 Ω resistor. The arrow indicates the region of cycle one.
Figure 8.
The potential and current profiles of the Ni–Fe cell with a 165 Ω resistor. The first discharge is enlarged and presented in
Figure 9. Measurements include the potential difference between the cathode and anode (dE2), the potential of the iron plate as anode (E_iron, V_
SCE), the potential of the Ni–CF cathode (E_nickel, V_
SCE), and the current with a 165 Ω resistor. The arrow indicates the region of cycle one.
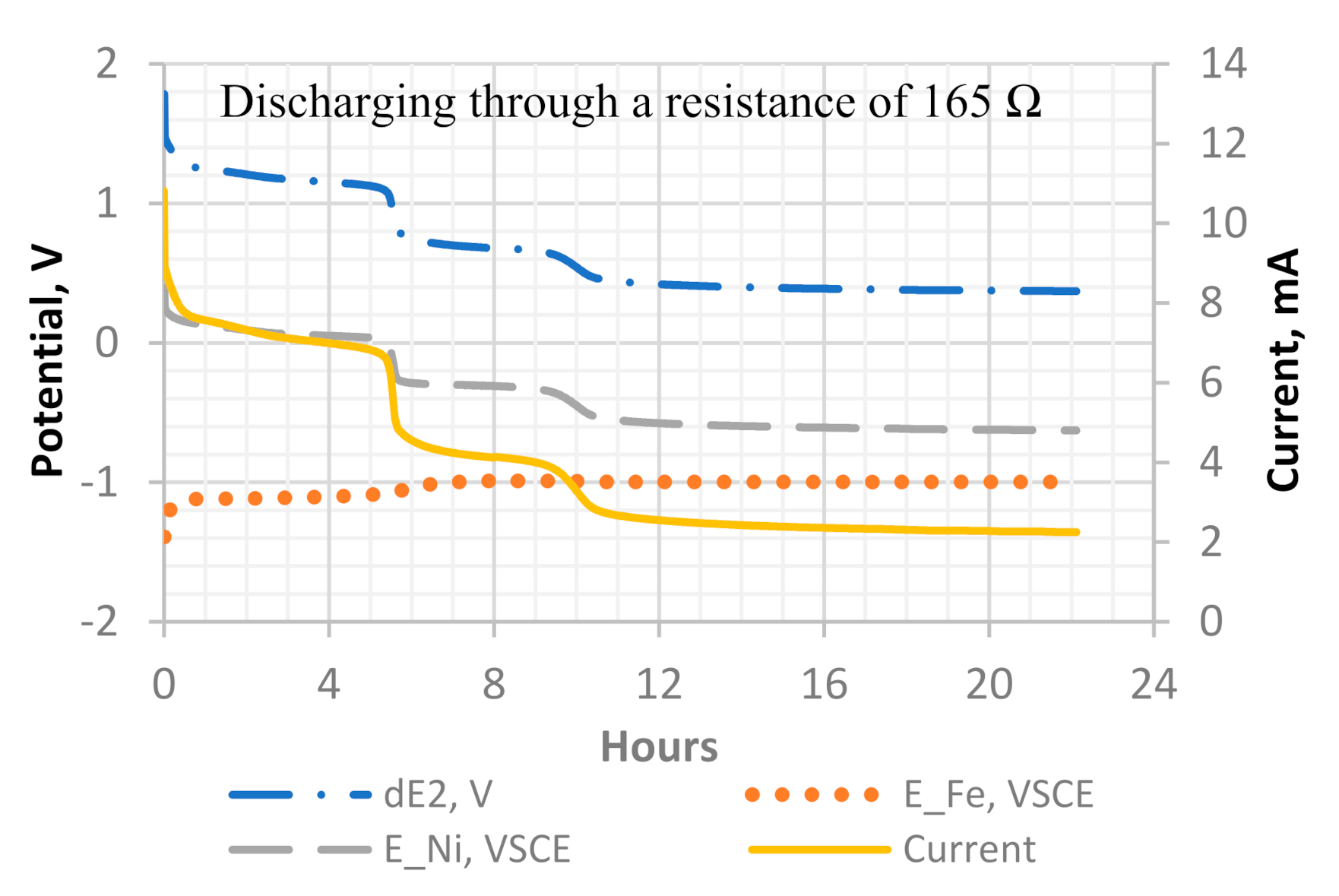
Figure 9.
A detailed potential and current profile of discharge cycle 1 of the Ni-Fe cell. Measurements include the potential difference between the cathode and anode (dE2), the potential of the iron plate as anode (E_Fe, V_SCE), the potential of the Ni-CF cathode (E_Ni, V_SCE), and the current with a 165 Ω resistor.
Figure 9.
A detailed potential and current profile of discharge cycle 1 of the Ni-Fe cell. Measurements include the potential difference between the cathode and anode (dE2), the potential of the iron plate as anode (E_Fe, V_SCE), the potential of the Ni-CF cathode (E_Ni, V_SCE), and the current with a 165 Ω resistor.
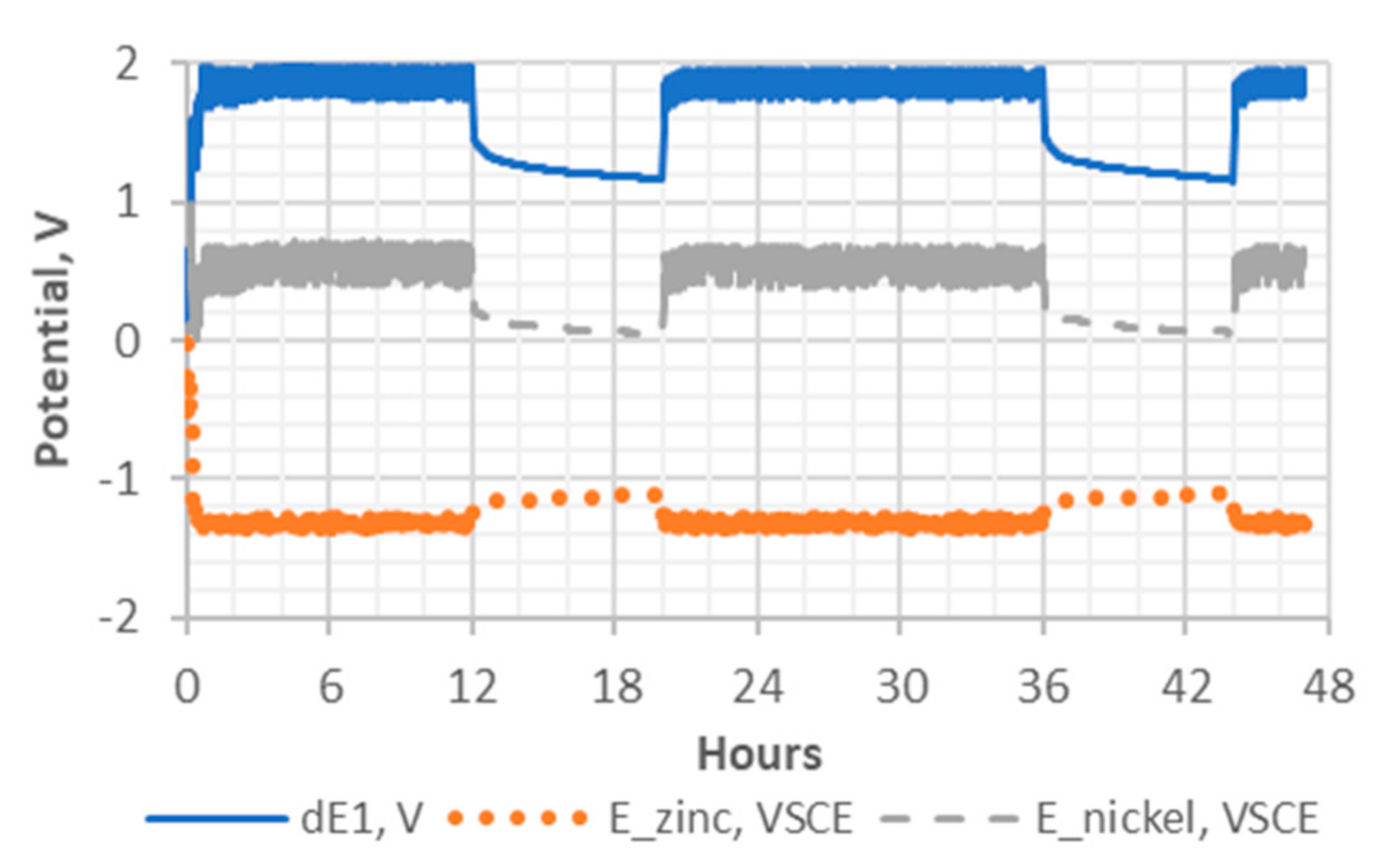
Figure 10.
Potential profiles of the Ni-Zn cell during charge and discharge cycles under a no-load condition. Curve dE1 represents the potential difference between the cathode and the anode. The potentials of the anode and cathode are indicated by E_zinc and E_nickel, respectively.
Figure 10.
Potential profiles of the Ni-Zn cell during charge and discharge cycles under a no-load condition. Curve dE1 represents the potential difference between the cathode and the anode. The potentials of the anode and cathode are indicated by E_zinc and E_nickel, respectively.
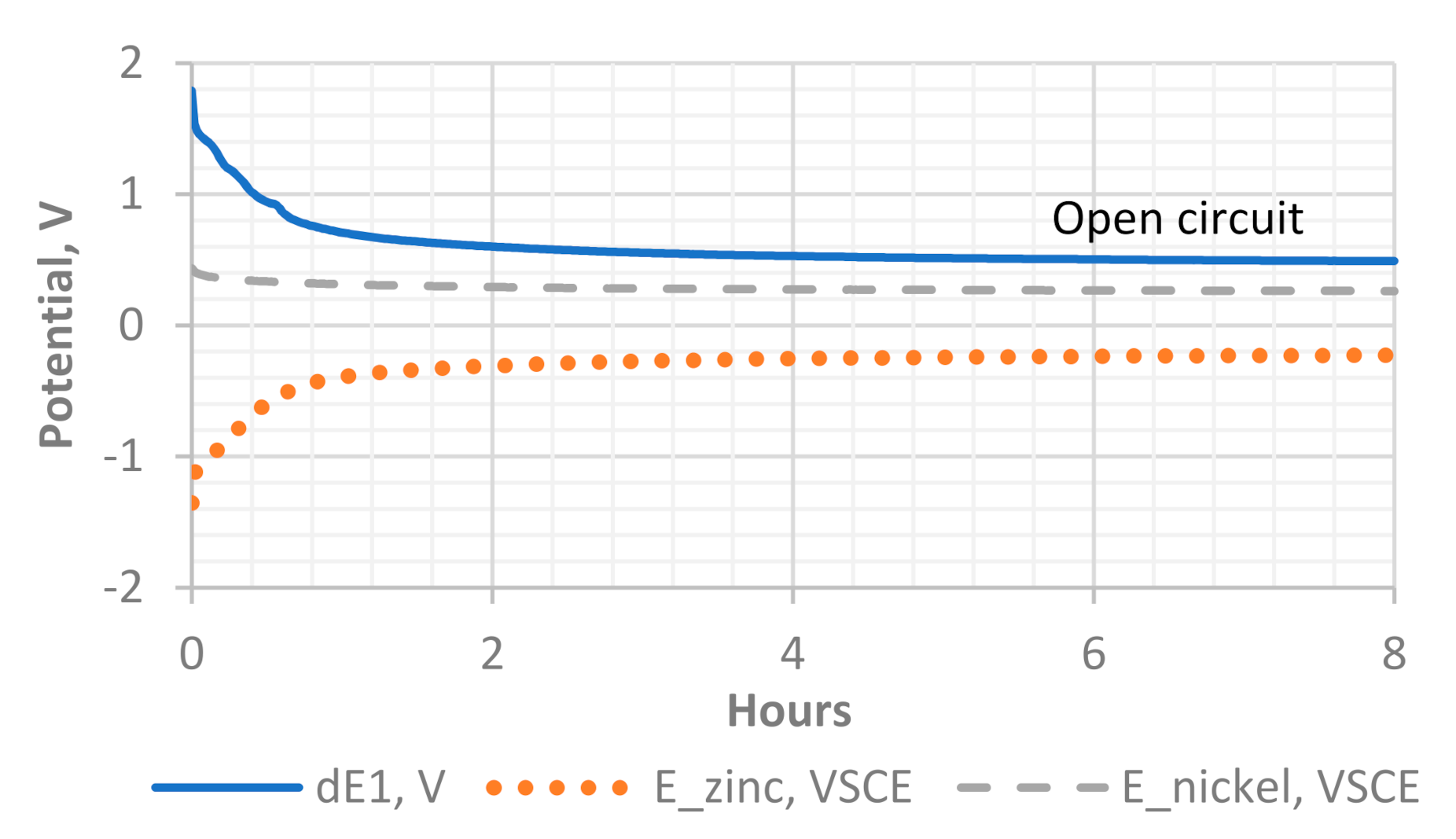
Figure 11.
The open-circuit potential of the Ni–Zn cell during discharge. Curve dE1 represents the potential difference between the cathode and the anode. The potentials of the anode and cathode are indicated by E_zinc and E_nickel, respectively.
Figure 11.
The open-circuit potential of the Ni–Zn cell during discharge. Curve dE1 represents the potential difference between the cathode and the anode. The potentials of the anode and cathode are indicated by E_zinc and E_nickel, respectively.
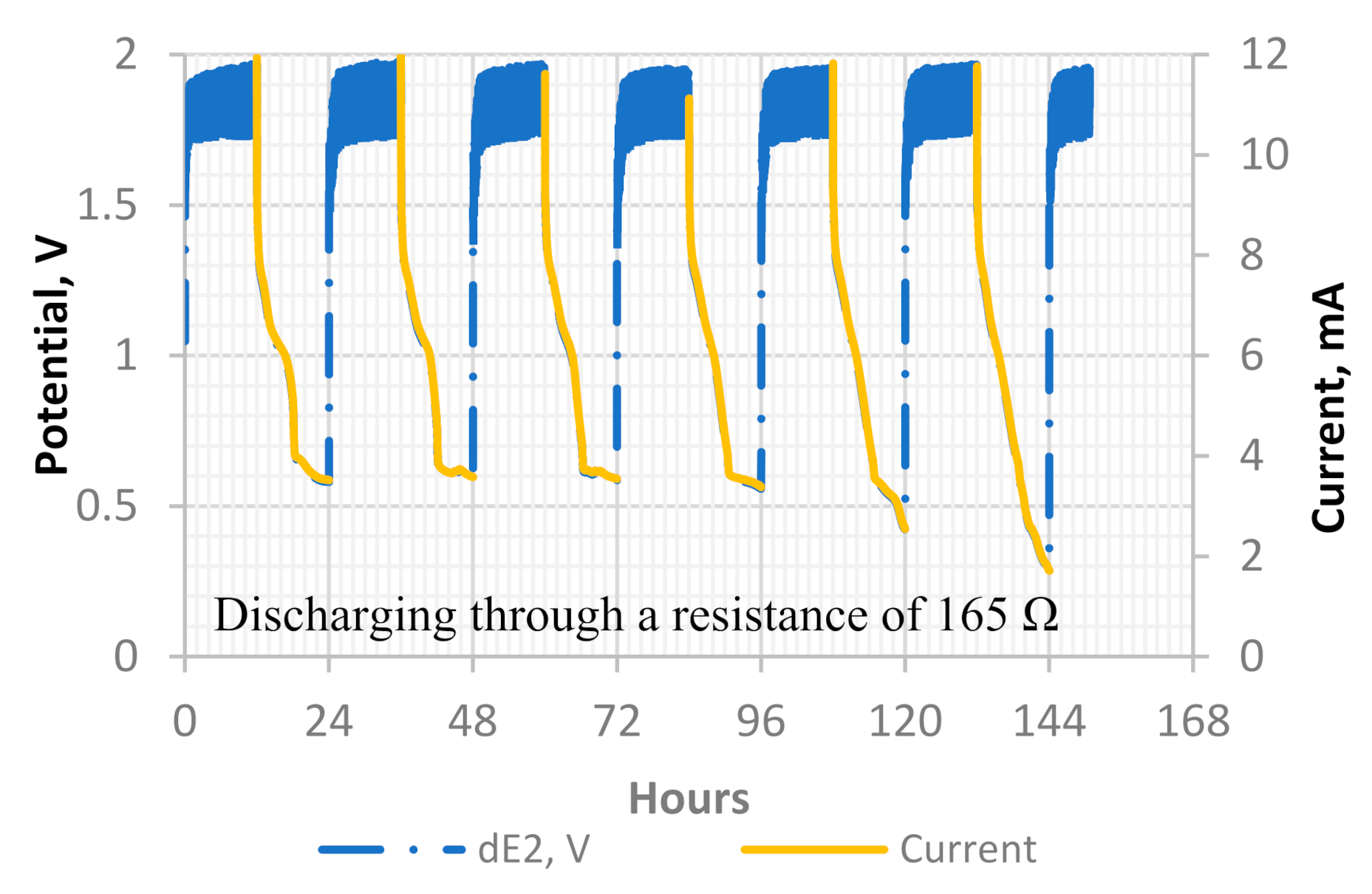
Figure 12.
Six charge and discharge cycles of the Ni–Fe concrete battery, through a resistance of 165 Ω. The cell potential is represented by dE2, which is the potential difference between the Ni cathode and the Fe anode.
Figure 12.
Six charge and discharge cycles of the Ni–Fe concrete battery, through a resistance of 165 Ω. The cell potential is represented by dE2, which is the potential difference between the Ni cathode and the Fe anode.
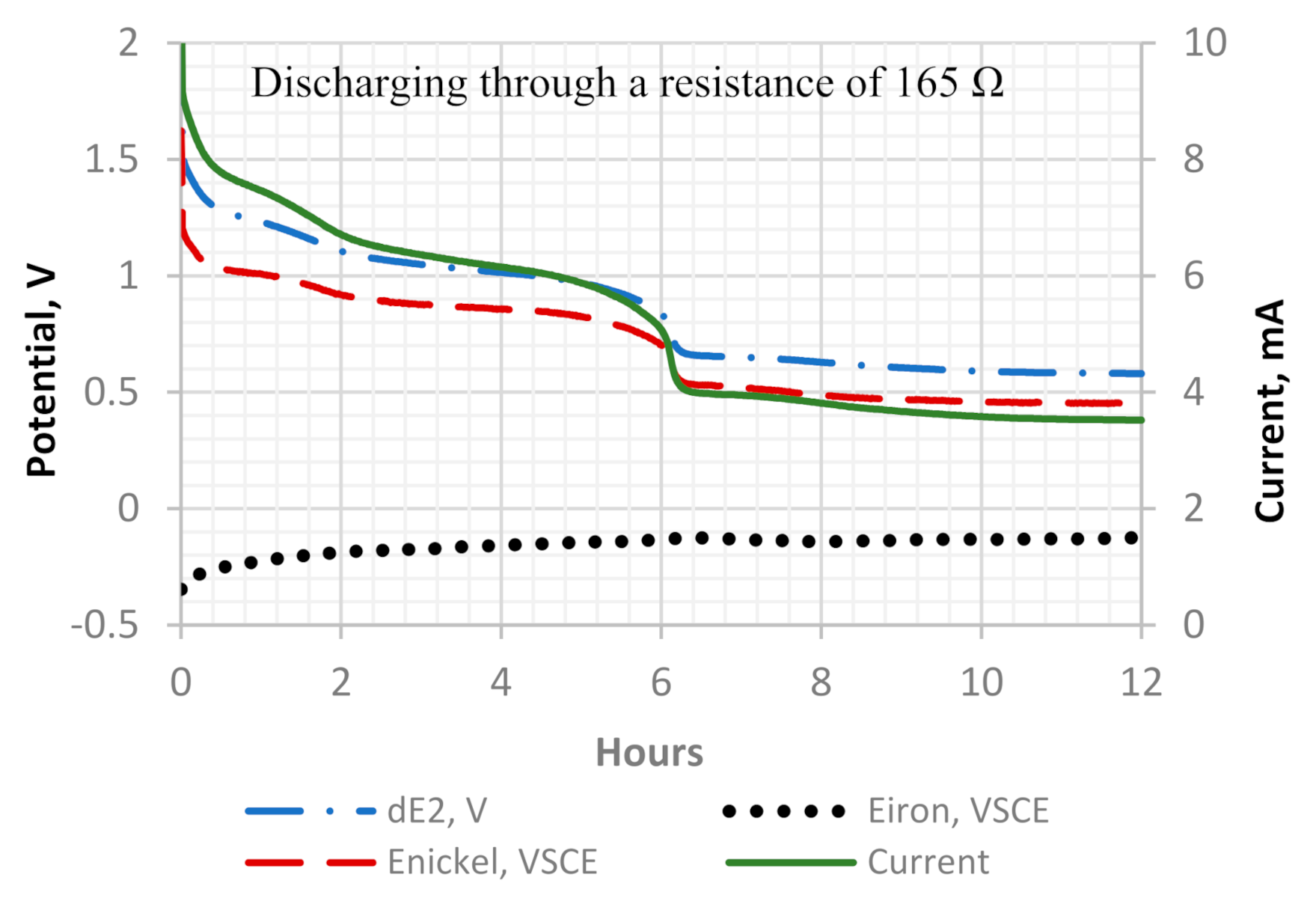
Figure 13.
A detailed potential and current development of discharge cycle one. The cell potential is represented by dE2, which is the potential difference between the Ni cathode and the Fe anode.
Figure 13.
A detailed potential and current development of discharge cycle one. The cell potential is represented by dE2, which is the potential difference between the Ni cathode and the Fe anode.
Figure 14.
An image of the single-cell cement-based structural battery. A multimeter shows the cell potential of 1.24 V after a 3-h discharge.
Figure 14.
An image of the single-cell cement-based structural battery. A multimeter shows the cell potential of 1.24 V after a 3-h discharge.
Table 1.
Possible anode materials and their half-cell reactions.
Table 1.
Possible anode materials and their half-cell reactions.
| Metal | Half-Cell Reaction | Ered° (V) |
|---|
| Iron | Fe(OH)2(s) + 2e− → Fe(s) + 2OH−(aq) | −0.89 |
| Zinc | Zn(OH)42−(aq) + 2e− → Zn(s) + 4OH−(aq) | −1.20 |
Table 2.
Possible cathode materials and their half-cell reactions.
Table 2.
Possible cathode materials and their half-cell reactions.
| Metal | Half-Cell Reaction | Ered° (V) |
|---|
| Nickel oxyhydroxide | NiOOH + H2O + e− → Ni(OH)2 + OH− | +0.52 [16] |
Table 3.
Mix proportion of the cement-based electrolyte separator.
Table 3.
Mix proportion of the cement-based electrolyte separator.
| Cement (g) | Fine Sand (g) | Alkaline Solution (g) | Ion Exchange Resin (g) | Total Volume (cm3) |
|---|
| 22 | 15 | 12 | 8 | 35 |
Table 4.
Compositions of the alkaline solution.
Table 4.
Compositions of the alkaline solution.
| KOH (85.6%) (g) | LiOH∙H2O (>98%) (g) | Deionized Water (g) |
|---|
| 245.3 | 12.6 | 756.3 |
Table 5.
Proportions for the cement-based conductive mortar and their resistivity.
Table 5.
Proportions for the cement-based conductive mortar and their resistivity.
| | Ref11 | Test 4 | Test 5 | Test 6 | Test 7 | Test 8 | Test 9 | Test 10 | Test 13 | Test 14 |
|---|
| Cement * (g) | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 |
| Sand (0–1 mm) (g) | 80 | 80 | 80 | 80 | 80 | 80 | 80 | 80 | 80 | 80 |
| CF (3 mm) (g) | 0 | 0.2 | 0.2 | 0.1 | 0.2 | 2.3 | 1.1 | 0.8 | 0.6 | 0.4 |
| Deionized water (g) | 24 | 24 | 24 | 24 | 24 | 48 | 24 | 24 | 24 | 24 |
| Methylcellulose (g) | 0 | 0.25 | 0.12 | 0.07 | 0.10 | 1.38 | 0.39 | 0.24 | 0.19 | 0.19 |
| Superplasticizer ** (g) | 0 | 0.9 | 0.4 | 0.2 | 0.5 | 1.1 | 2.6 | 1.0 | 0.7 | 0.6 |
| Water-cement ratio | 0.6 | 0.6 | 0.6 | 0.6 | 0.6 | 1.2 | 0.6 | 0.6 | 0.6 | 0.6 |
| CF volume (%) | 0% | 0.2% | 0.2% | 0.1% | 0.2% | 1.4% | 0.9% | 0.7% | 0.5% | 0.3% |
| Resistivity (Ω∙m) | 10.53 | 5.52 | 7.11 | 10.03 | 8.27 | 0.35 | 0.91 | 2.11 | 2.21 | 4.14 |
Table 6.
Mix proportions of the “powder-mixing” cathodes.
Table 6.
Mix proportions of the “powder-mixing” cathodes.
| Cathode Mix Number | Ni-1 | Ni-2 | Ni-3 | Ni-4 |
|---|
| Cement 42.5R (g) | 15 | 10 | 20 | 6 |
| Deionized water (g) | 10.8 | 11.0 | 10.0 | 10.0 |
| CF (3 mm) (g) | 0.8 | 0.8 | 0.8 | - |
| CF powder (0.2 mm) (g) | 0.35 | 0.35 | 0.35 | 1.80 |
| w/c ratio | 0.79 | 1.20 | 0.55 | 1.76 |
| CF volume (%) | 2.7% | 2.7% | 2.8% | 6.2% |
| Ni(OH)2 powder (g) | 22.5 | 30 | 15 | 10 |
| Metal volume (%) | 24% | 31% | 16% | 15% |
| Superplasticizer (g) | 1.5 | 1.5 | 1.5 | 0.8 |
Table 7.
Mix proportions of the “powder-mixing” anodes.
Table 7.
Mix proportions of the “powder-mixing” anodes.
| Anode Mix Number | Fe-1 | Fe-2 | Fe-3 | Fe-4 | Fe-5 |
|---|
| Cement (g) | 12 | 14 | 6 | 6 | 6 |
| Alkaline solution (g) | 15.8 | 14.0 | 6.8 | 10.1 | 10.2 |
| CF (3 mm) (g) | - | 0.8 | - | - | 0.8 |
| CF powder (0.2 mm) (g) | - | 0.35 | 0.60 | 1.60 | 1.60 |
| w/c ratio | 1.12 | 0.87 | 0.95 | 1.39 | 1.42 |
| CF volume (%) | 0% | 2.8% | 3.3% | 6.7% | 9.5% |
| Fe powder (g) | 60 | 35 | 12 | 12 | 12 |
| Metal volume (%) | 29% | 20% | 15% | 12% | 12% |
| Superplasticizer (g) | 1.4 | 1.7 | 0.5 | 0.6 | 0.7 |
Table 8.
Details of electroplating metals on carbon fiber.
Table 8.
Details of electroplating metals on carbon fiber.
| Cathode | Anode | Bath Solution | Current and Duration |
|---|
| CF mesh | Ni plate | 250 g/L NiSO4∙7H2O
20 g/L NiCl2∙6H2O
25 g/L H3BO3 | 1.0A for 4 h |
| CF mesh | Zn plate | 74 g Zn granulate
1 L acetic acid
20 g NaCl
15 g sugar | 1.0A for 4 h |
| CF mesh | Fe plate | 180 g/L FeSO4∙7H2O | 1.2A for 6 h |
Table 9.
Weight gained of metal-coated carbon fiber meshes.
Table 9.
Weight gained of metal-coated carbon fiber meshes.
| | Ni–CF Mesh | Zn–CF Mesh | Fe–CF Mesh |
|---|
| Initial weight (g) | 2.433 | 3.577 | 2.433 |
| Final weight (g) | 4.955 | 6.311 | 8.714 |
| Gained weight (g) | 2.522 | 2.734 | 6.281 |
| Theoretical thickness of coating (µm) | 23 | 30 | 60 |
Table 10.
Cell arrangement of powder-mixing electrodes.
Table 10.
Cell arrangement of powder-mixing electrodes.
| | Cell 1 | Cell 2 | Cell 3 | Cell 4 |
|---|
| Anode | Ni-1 | Ni-2 | Ni-3 | Ni-4 |
| Cathode | Fe-3 | Fe-4 | Fe-5 | Pure Fe plate |
Table 11.
Cell arrangement of metal-coating electrodes.
Table 11.
Cell arrangement of metal-coating electrodes.
| | Cell A | Cell B | Cell C |
|---|
| Anode | Mortar with Ni–CF mesh | Mortar with Ni–CF mesh | Mortar with Ni–CF mesh |
| Cathode | Iron plate | Mortar with Fe–CF mesh | Mortar with Zn–CF mesh |
Table 12.
Summary of cell performance tested in alkali solution as electrolyte.
Table 12.
Summary of cell performance tested in alkali solution as electrolyte.
| Cell | Discharge Cycles | Battery Capacity (mAh) | Energy (mWh) | Energy Density * (Wh/m2) |
|---|
| Ni-1/Fe-3 (Powder-mixing) | 1 (at 6 h) | 5.6 | 4.3 | 0.5 |
| Ni–CF/Fe plate | 1 (at 8 h) | 22 | 23 | 2.8 |
| 2 (at 8 h) | 20 | 19 | 2.3 |
| Ni–CF/Fe–CF | 1 (at 6 h) | 42 | 50 | 6.2 |
| 1 (at 8 h) | 51 | 56 | 6.9 |
| 1 (at 12) | 64 | 64 | 7.9 |
| 1 (at 22) | 89 | 73 | 9.0 |
| 2 (at 6 h) | 44 | 52 | 6.4 |
| 3 (at 6 h) | 43 | 49 | 6.1 |
| 4 (at 6 h) | 43 | 49 | 6.1 |
| 5 (at 6 h) | 43 | 49 | 6.1 |
Table 13.
Battery performance of Ni-Fe concrete battery. Each discharge cycle is 12 h.
Table 13.
Battery performance of Ni-Fe concrete battery. Each discharge cycle is 12 h.
| Discharging Cycles | Battery Capacity (mAh) | Energy (mWh) | Energy Density * (Wh/m2) |
|---|
| 1 | 62 | 57.6 | 7.10 |
| 2 | 62 | 58.5 | 7.23 |
| 3 | 61 | 57.3 | 7.07 |
| 4 | 61 | 56.3 | 6.95 |
| 5 | 59 | 53.7 | 6.63 |
| 6 | 55 | 49.5 | 6.11 |