Enhanced Corrosion Resistance of SA106B Low-Carbon Steel Fabricated by Rotationally Accelerated Shot Peening
Abstract
:1. Introduction
2. Experimental Procedures
2.1. Material
2.2. Electrochemical Measures
3. Results and Discussion
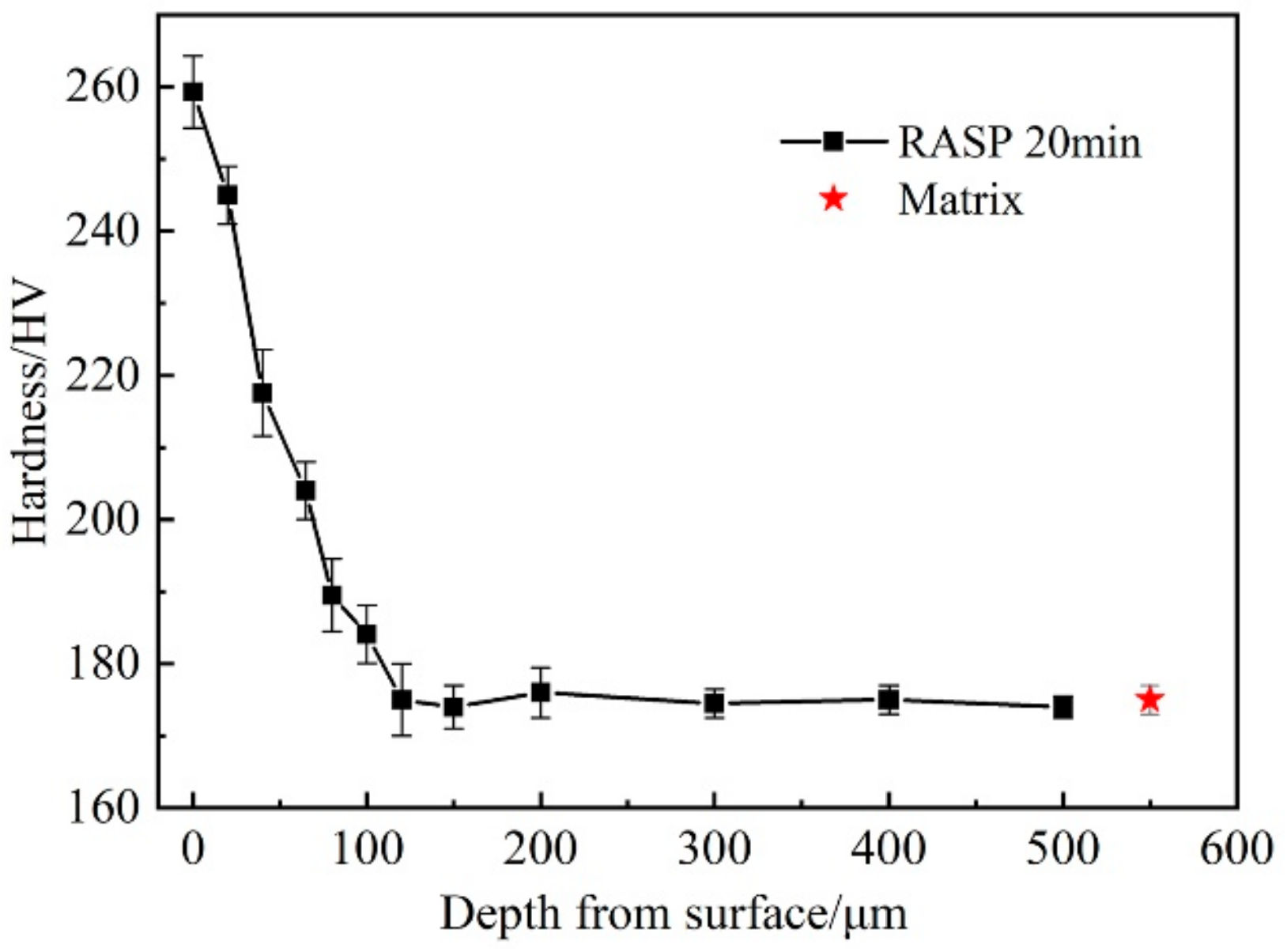
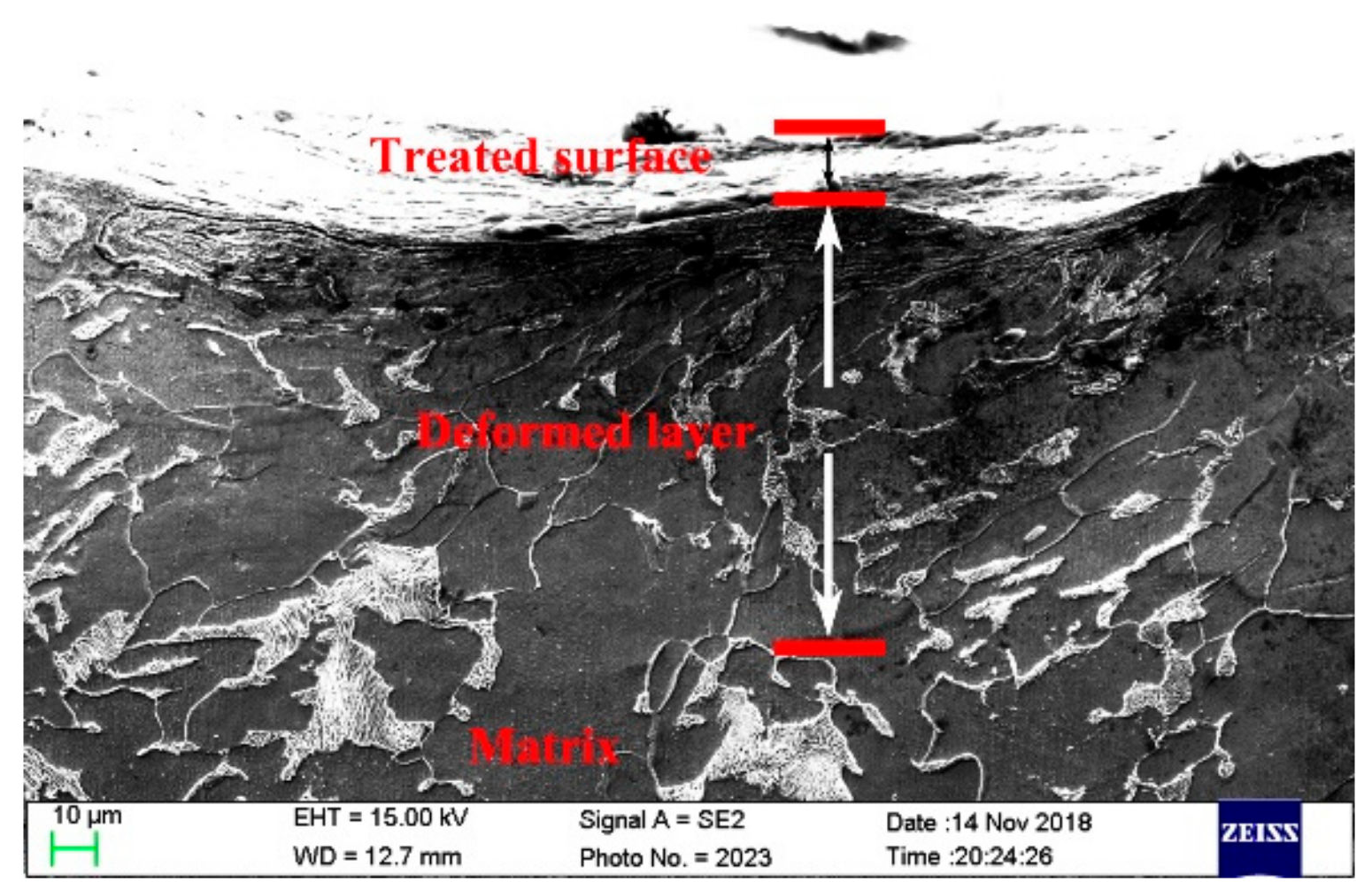
3.1. Microstructure Characterizations
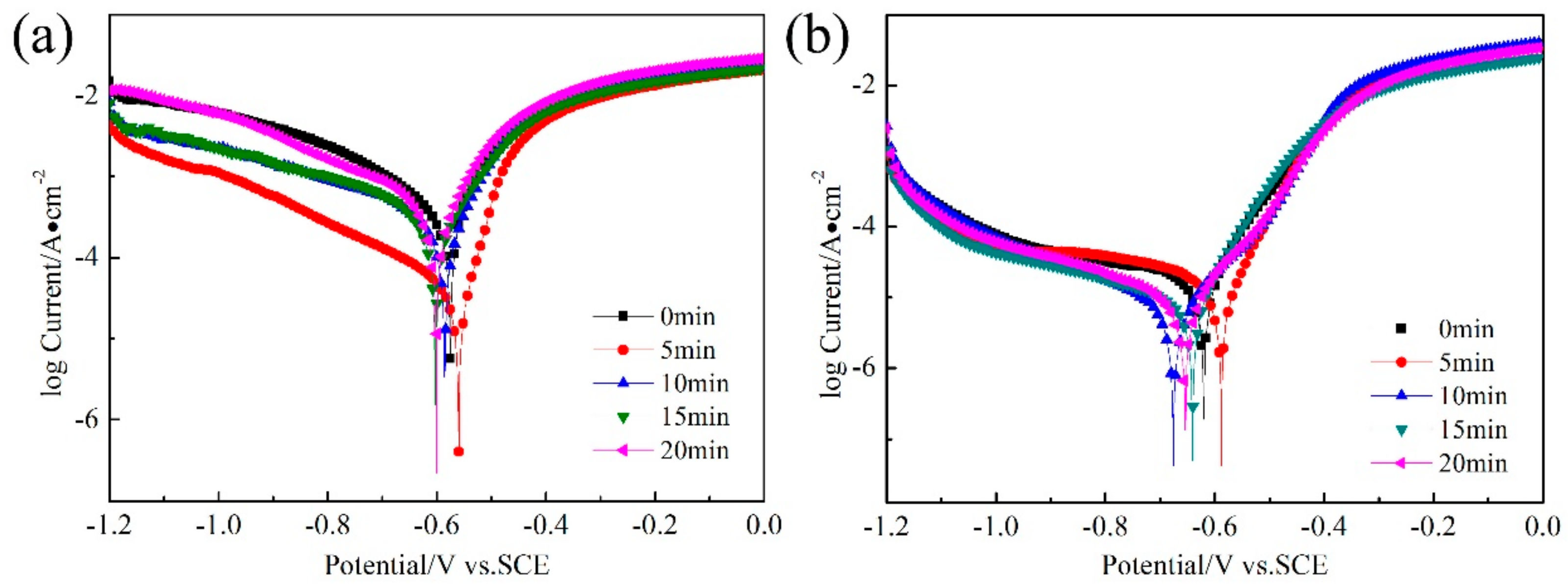
3.2. Potentiodynamic Polarization Results
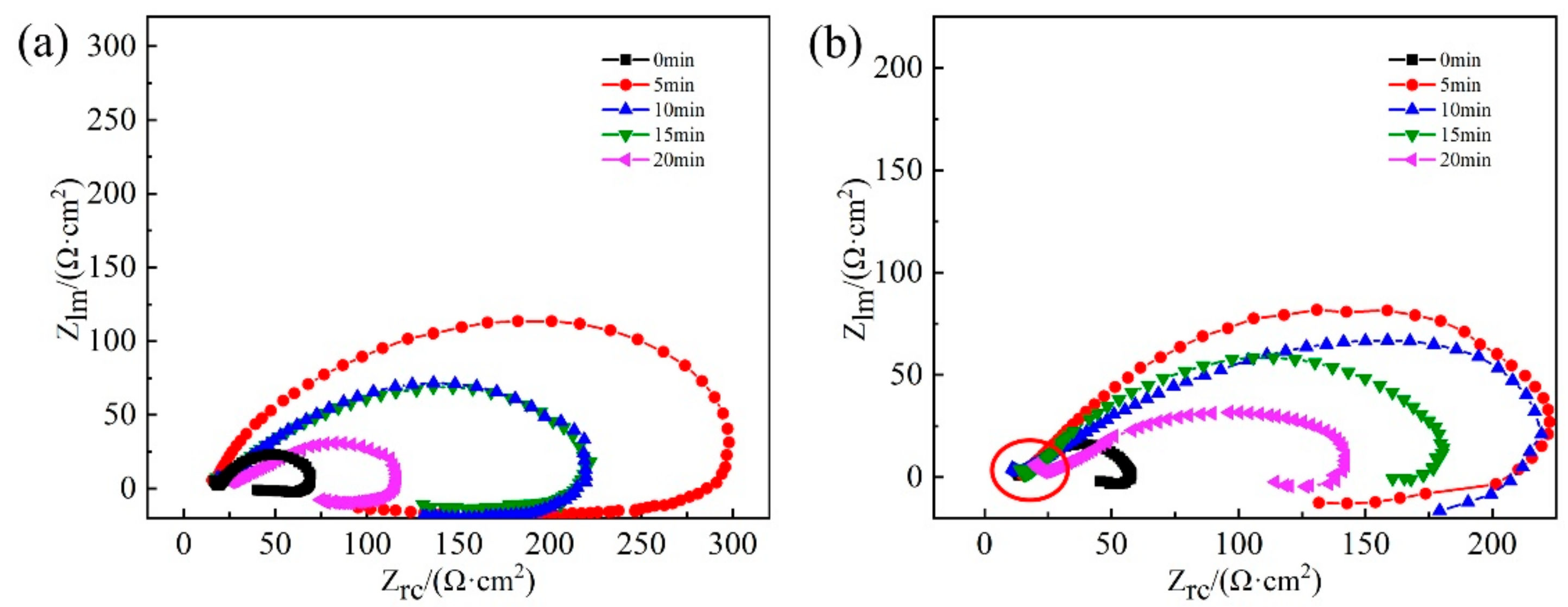
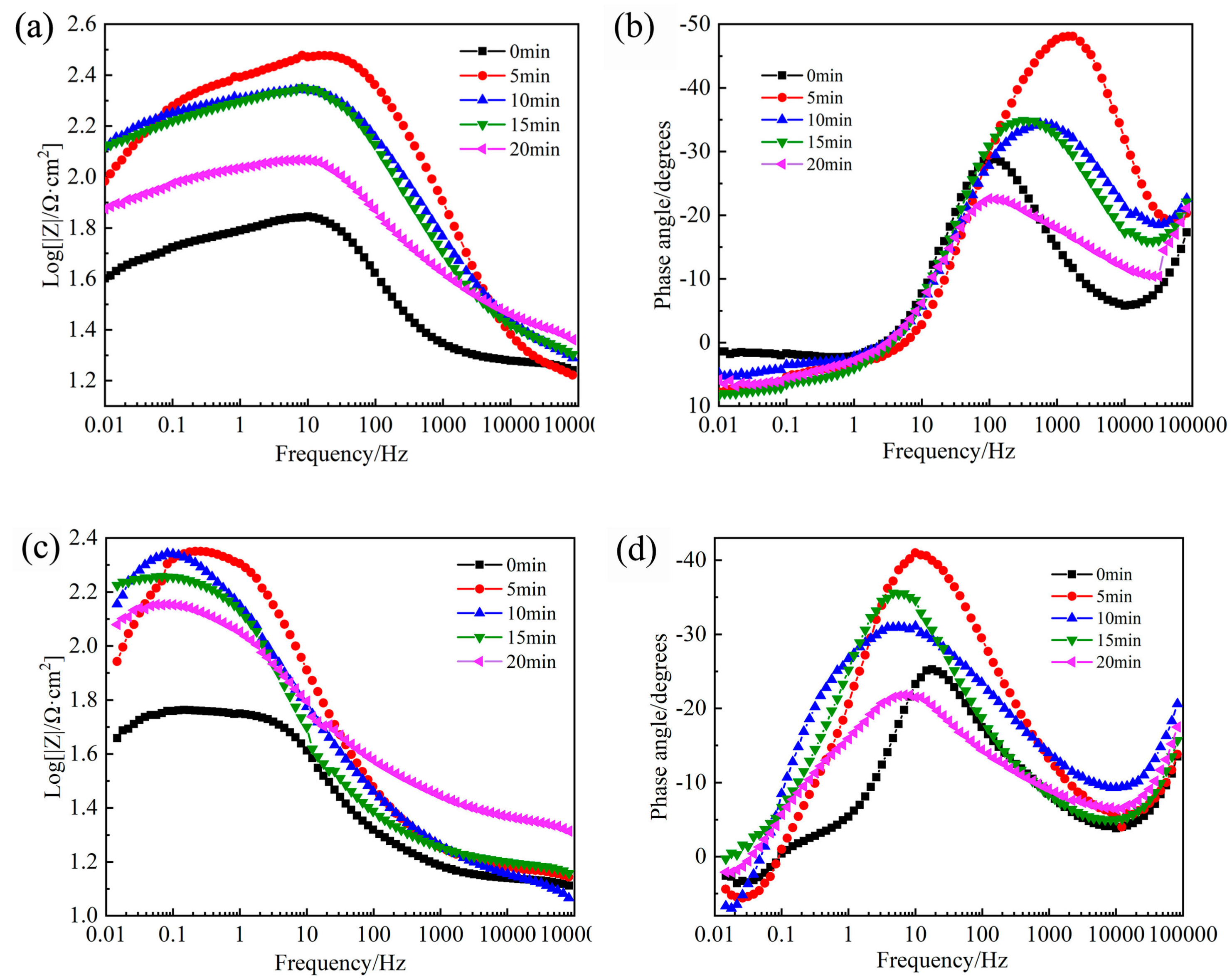
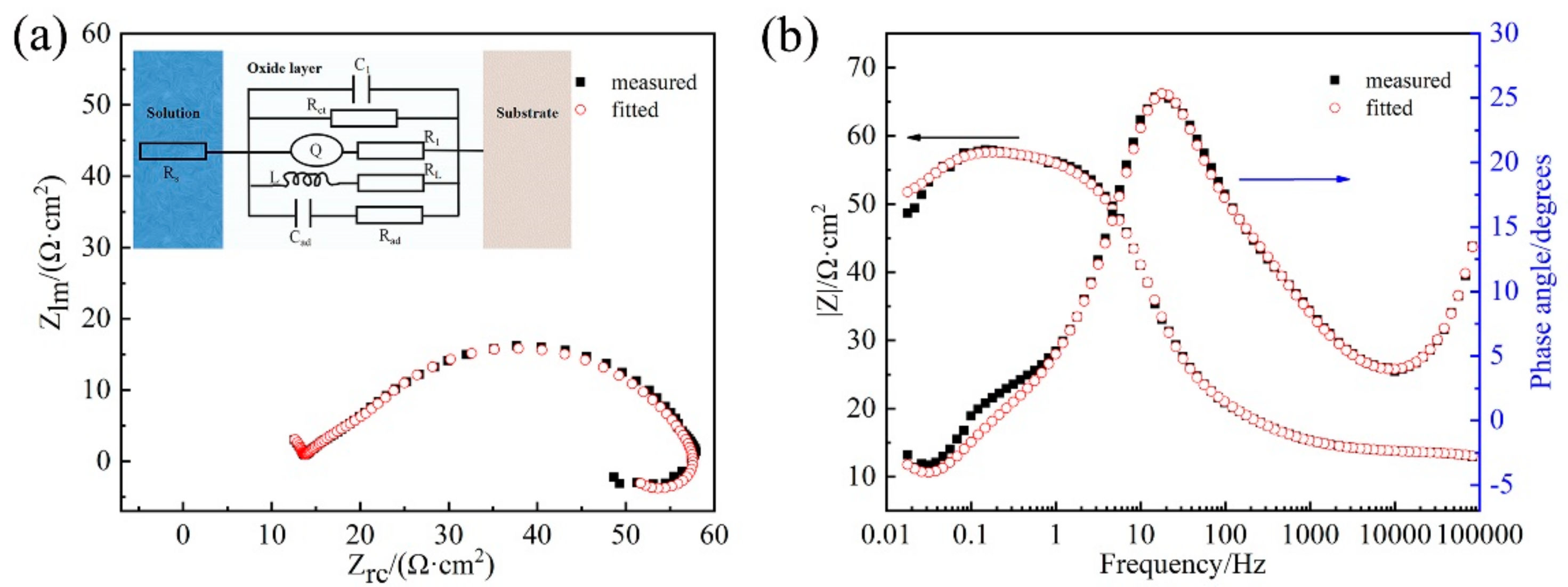
3.3. EIS Study
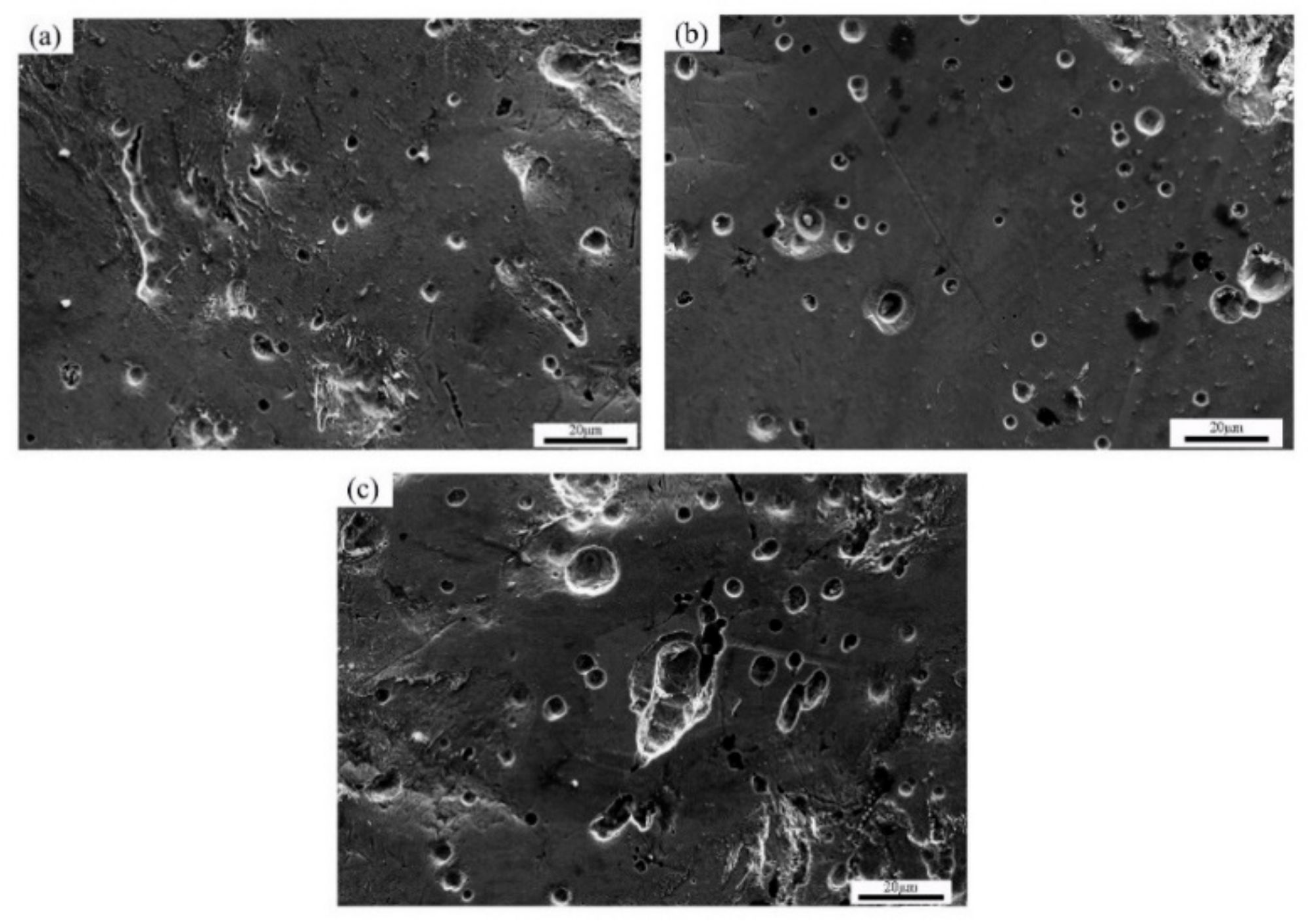
3.4. SEM Photomicrographs
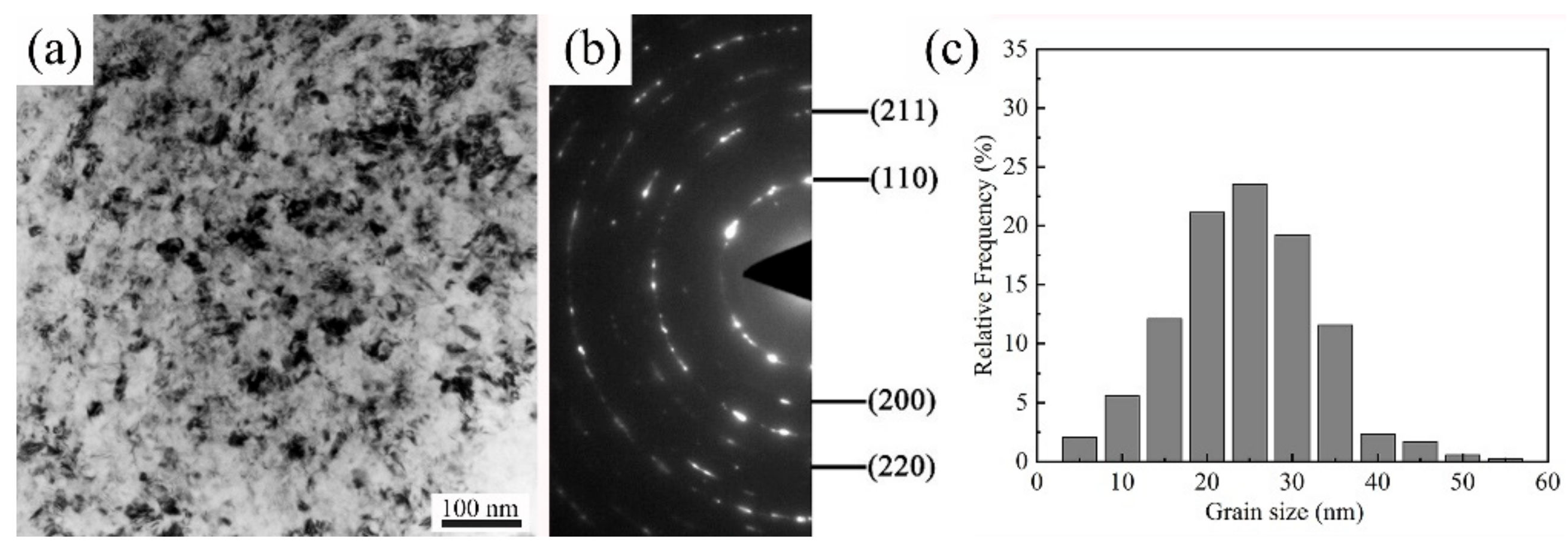
3.5. TEM Analysis
4. Conclusions
- (1)
- The RASP-processed SA106B carbon steel surface formed a deformation layer with a depth about 120 µm. The mean grain size is 25 nm in the top surface layer.
- (2)
- The sample RASP-processed for 5 min exhibited the best corrosion resistance due to the cementite dissolution and formation of nanograins.
- (3)
- Prominent micro-cracks and holes were produced in the steel when the RASP was processed more than 5 min, resulting in the decrease of corrosion resistance.
Author Contributions
Funding
Conflicts of Interest
References
- Moering, J.; Ma, X.; Chen, G. The role of shear strain on texture and microstructural gradients in low carbon steel processed by surface mechanical attritiontreatment. Scr. Mater. 2015, 108, 100–103. [Google Scholar] [CrossRef]
- Zhu, Y.T.; Lowe, T.C.; Langdon, T.G. Performance and applications of nanostructured materials produced by severe plastic deformation. Scr. Mater. 2004, 51, 826–830. [Google Scholar] [CrossRef]
- Fang, T.H.; Li, W.L.; Tao, N.R. Revealing extraordinary intrinsic tensile plasticity in gradient nano-grained copper. Science 2011, 331, 1587–1590. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Balasubramaniam, R. Effect of nanocrystalline grain size on the electrochemical and corrosion behavior of nickel. J. Corros. Sci. 2004, 46, 3019–3029. [Google Scholar] [CrossRef]
- González, J.; Peral, L.-B.; Colombo, C.; Fernández-Pariente, I. A study on the microstructural evolution of a low alloy steel by different shot peening treatments. Metals 2018, 8, 187. [Google Scholar] [CrossRef]
- Segal, V.M. Severe plastic deformation: simple shear versus pure shear. Mater. Sci. Eng. A 2002, 338, 331–344. [Google Scholar] [CrossRef]
- Lu, K.; Lu, J. Nanostructured surface layer on metallic materials induced by surface mechanical attrition treatment. Mater. Sci. Eng. A 2004, 375, 38–45. [Google Scholar] [CrossRef]
- He, J.W.; Ma, S.N.; Ba, D.M. Investigation of surface nanocrystallization by pre-forceing rolling technology. Adv. Mater. Res. 2013, 750, 1963–1966. [Google Scholar] [CrossRef]
- Sun, J.C.; Sheng, G.R.; Wang, Y.T. Surface self-nanocrystallization on industrial pure iron by high energy shot peening. (In Chinese). Heat Treat. Met. 2010, 35, 38–41. [Google Scholar]
- Wang, X.; Li, Y.S.; Zhang, Q. Gradient structured copper by rotationally accelerated shot peening. J. Mater. Sci. Technol. 2016, 7, 758–761. [Google Scholar] [CrossRef]
- Li, J.; Soh, A.K. Enhanced ductility of surface nano-crystallized materials by modulating grain size gradient. Modell. Simul. Mater. Sci. Eng. 2012, 20, 085002. [Google Scholar]
- Balusamy, T.; Sankara Narayanan, T.S.N.; Ravichandran, K. Influence of surface mechanical attrition treatment (SMAT) on the corrosion behaviour of AISI 304 stainless steel. Corros. Sci. 2013, 74, 332–344. [Google Scholar] [CrossRef]
- Ye, W.; Li, Y.; Wang, F. Effects of nanocrystallization on the corrosion behavior of 309 stainless steel. Electrochim. Acta 2006, 51, 4426–4432. [Google Scholar] [CrossRef]
- Huang, R.; Han, Y. The effect of SMAT-induced grain refinement and dislocations on the corrosion behavior of Ti-25Nb-3Mo-3Zr-2Sn alloy. Mater. Sci. Eng. 2013, 33, 2353–2359. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, F.; Liu, G. Grain size effect on the electrochemical corrosion behavior of surface nanocrystallized low-carbon steel. Corros. Sci. 2004, 60, 891–896. [Google Scholar] [CrossRef]
- Zhang, L.; Ma, A.; Jiang, J. Electrochemical corrosion properties of the surface layer produced by supersonic fine-particles bombarding on low-carbon steel. Surf. Coat. Technol. 2013, 232, 412–418. [Google Scholar] [CrossRef]
- Chen, X.D.; Li, Y.S.; Zhu, Y.T. Improved corrosion resistance of 316LN stainless steel performed by rotationally accelerated shot peening. Appl. Surf. Sci. 2019, 481, 1305–1312. [Google Scholar] [CrossRef]
- Chabok, A.; Dehghani, K. Formation of nanograin in IF steels by friction stir processing. Mater. Sci. Eng. A 2010, 528, 309–313. [Google Scholar] [CrossRef]
- Duan, C.; Wang, M. A review of microstructural evolution in the adiabatic shear bands induced by high speed machining. Acta Metal. 2013, 26, 97–112. [Google Scholar] [CrossRef]
- Duan, Z.Q.; Li, S.X.; Huang, D.W. Microstructures and adiabatic shear bands formed by ballistic impact in steels and tungsten alloy. Fatigue. Fract. Eng. Mater. Struct. 2003, 26, 1119–1126. [Google Scholar] [CrossRef]
- Chen, Y.F.; Chen, X.D.; Dai, X. Effect of spinodal decomposition on the pitting corrosion resistance of Z3CN20.09M duplex stainless steel. Mater. Corros. 2017, 69, 527–535. [Google Scholar] [CrossRef]
- Yu, Y.C. Electrochemistry basics tutorial, 2nd ed.; Beijing Chemical Industry Press: Beijing, China, 2019; pp. 118–120. [Google Scholar]
- Guo, H.X.; Lu, B.T.; Luo, J.L. Interaction of mechanical and electrochemical factors in erosion-corrosion of carbon steel. Electrochim. Acta. 2005, 51, 315–323. [Google Scholar] [CrossRef]
- Kolawole, S.K.; Kolawole, F.O.; Enegela, O.P. Pitting corrosion of a low carbon steel in corrosive environments: experiments and models. Adv. Mater. Res. 2015, 1132, 349–365. [Google Scholar] [CrossRef]
- Mansfeld, F.; Kending, M.W.; Lorenz, W.J. Corrosion inhibition in neutral, aerated media. J. Electrochem. Soc. 1985, 132, 290–296. [Google Scholar] [CrossRef]
- Li, Y.R.; Lin, W.M.; Wei, Y.H. Electrochemical corrosion behavior of mechanical attrition treated surface layer with nanocrystallines on Cu-10Ni alloy. Corros. Sci. Prot. Technol. 2012, 24, 397–400. [Google Scholar]
- Clover, D.; Kinsella, B.; Pejcic, B. The influence of microstructure on the corrosion rate of various carbon steels. J. Appl. Electrochem. 2005, 35, 139–149. [Google Scholar] [CrossRef]
- Simoes, A.M.; Bastos, A.C.; Ferreira, M.G. Use of SVET and SECM to study the galvanic corrosion of an iron-zinc cell. Corros. Sci. 2007, 49, 726–739. [Google Scholar] [CrossRef]
- Wu, X.; Tao, N.; Hong, Y. Microstructure and evolution of mechanically-induced ultrafine grain in surface layer of AL-alloy subjected to USSP. Acta Mater. 2002, 50, 2075–2084. [Google Scholar] [CrossRef]
- Bang, C.W.; Seol, J.B.; Yang, Y.S. Atomically resolved cementite dissolution governed by the strain state in pearlite steel wires. Scr. Mater. 2015, 108, 151–155. [Google Scholar] [CrossRef]
- Danoix, F.; Julien, D.; Sauvage, X.; Copreaux, J. Direct evidence of cementite dissolution in drawn pearlitic steels observed by tomographic atom probe. Mater. Sci. Eng. A 1998, 250, 8–13. [Google Scholar] [CrossRef]
- Murayama, M.; Horita, Z.; Hono, K. Microstructure of two-phase Al-1.7 at% Cu alloy deformed by equal-channel angular pressing. J. Acta Mater. 2001, 49, 21–29. [Google Scholar] [CrossRef]



| C | Mn | Si | S | P | Cr | Mo | V | Fe |
|---|---|---|---|---|---|---|---|---|
| 0.21 | 0.53 | 0.26 | 0.008 | 0.011 | 0.02 | 0.01 | 0.01 | Balance |
| RASP Time (min) | Ecorr (mVSCE) | Icorr (μA/cm2) |
|---|---|---|
| 0 | −576 (±10) | 490 (±11) |
| 5 | −560 (±8) | 44 (±6) |
| 10 | −586 (±9) | 227 (±8) |
| 15 | −601 (±12) | 295 (±15) |
| 20 | −603 (±5) | 380 (±26) |
| RASP Time (min) | Ecorr (mVSCE) | Icorr (μA/cm2) |
|---|---|---|
| 0 | −620 (±11) | 19.80 (±1.79) |
| 5 | −588 (±15) | 7.78 (±0.37) |
| 10 | −676 (±12) | 8.50 (±0.29) |
| 15 | −641 (±9) | 8.90 (±0.31) |
| 20 | −645 (±12) | 9.55 (±0.43) |
| RASP Time (min) | Rs (Ω·cm2) | Rct (Ω·cm2) | Q Parameters | C1 (F/cm2) | Cad (F/cm2) | R1 (Ω·cm2) | Rad (Ω·cm2) | RL (Ω·cm2) | L (H·cm2) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Y0 (µF/cm2) | α | |||||||||
| 0 | 4.1 | 63.4 | 20.83 | 0.91 | 5.59 × 10−8 | 2.16 × 10−5 | 33.3 | 45.1 | 168.2 | 84.3 |
| 5 | 8.3 | 282.1 | 2.73 | 0.92 | 1.20 × 10−7 | 1.42 × 10−6 | 12.7 | 248.1 | 320.3 | 398.4 |
| 10 | 10.3 | 202 | 11.63 | 0.79 | 1.23 × 10−7 | 3.05 × 10−6 | 15.8 | 222.2 | 534.6 | 718.6 |
| 15 | 10.2 | 201.7 | 12.93 | 0.83 | 1.15 × 10−7 | 3.99 × 10−6 | 16.8 | 164.4 | 416.6 | 483.2 |
| 20 | 11.0 | 101.9 | 16.75 | 0.78 | 8.55 × 10−8 | 1.10 × 10−5 | 23.9 | 95.5 | 231.4 | 381.5 |
| RASP Time (min) | Rs (Ω·cm2) | Rct (Ω·cm2) | Q Parameters | C1 (F/cm2) | Cad (F/cm2) | R1 (Ω·cm2) | Rad (Ω·cm2) | RL (Ω·cm2) | L (H·cm2) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Y0 (µF/cm2) | α | |||||||||
| 0 | 1.8 | 56.9 | 1394 | 0.64 | 4.40 × 10−8 | 0.000102 | 19.4 | 58.1 | 321.9 | 1656 |
| 5 | 8.2 | 222.3 | 684 | 0.67 | 1.65 × 10−7 | 0.000107 | 7.2 | 141.2 | 186.5 | 867 |
| 10 | 7.0 | 208.7 | 837.6 | 0.61 | 2.64 × 10−7 | 0.000200 | 7.9 | 154.8 | 386.0 | 1867 |
| 15 | 5.5 | 199.9 | 1171 | 0.58 | 8.59 × 10−8 | 0.000142 | 12.2 | 74.4 | 1308.0 | 3746 |
| 20 | 3.5 | 172.1 | 1939 | 0.53 | 3.51 × 10−8 | 0.000064 | 21.3 | 78.5 | 520.9 | 2847 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lei, C.; Chen, X.; Li, Y.; Chen, Y.; Yang, B. Enhanced Corrosion Resistance of SA106B Low-Carbon Steel Fabricated by Rotationally Accelerated Shot Peening. Metals 2019, 9, 872. https://doi.org/10.3390/met9080872
Lei C, Chen X, Li Y, Chen Y, Yang B. Enhanced Corrosion Resistance of SA106B Low-Carbon Steel Fabricated by Rotationally Accelerated Shot Peening. Metals. 2019; 9(8):872. https://doi.org/10.3390/met9080872
Chicago/Turabian StyleLei, Chaonan, Xudong Chen, Yusheng Li, Yuefeng Chen, and Bin Yang. 2019. "Enhanced Corrosion Resistance of SA106B Low-Carbon Steel Fabricated by Rotationally Accelerated Shot Peening" Metals 9, no. 8: 872. https://doi.org/10.3390/met9080872
APA StyleLei, C., Chen, X., Li, Y., Chen, Y., & Yang, B. (2019). Enhanced Corrosion Resistance of SA106B Low-Carbon Steel Fabricated by Rotationally Accelerated Shot Peening. Metals, 9(8), 872. https://doi.org/10.3390/met9080872




