Tensile Properties and Corrosion Resistance of Al-xFe-La Alloys for Aluminium Current Collector of Lithium-Ion Batteries
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussions
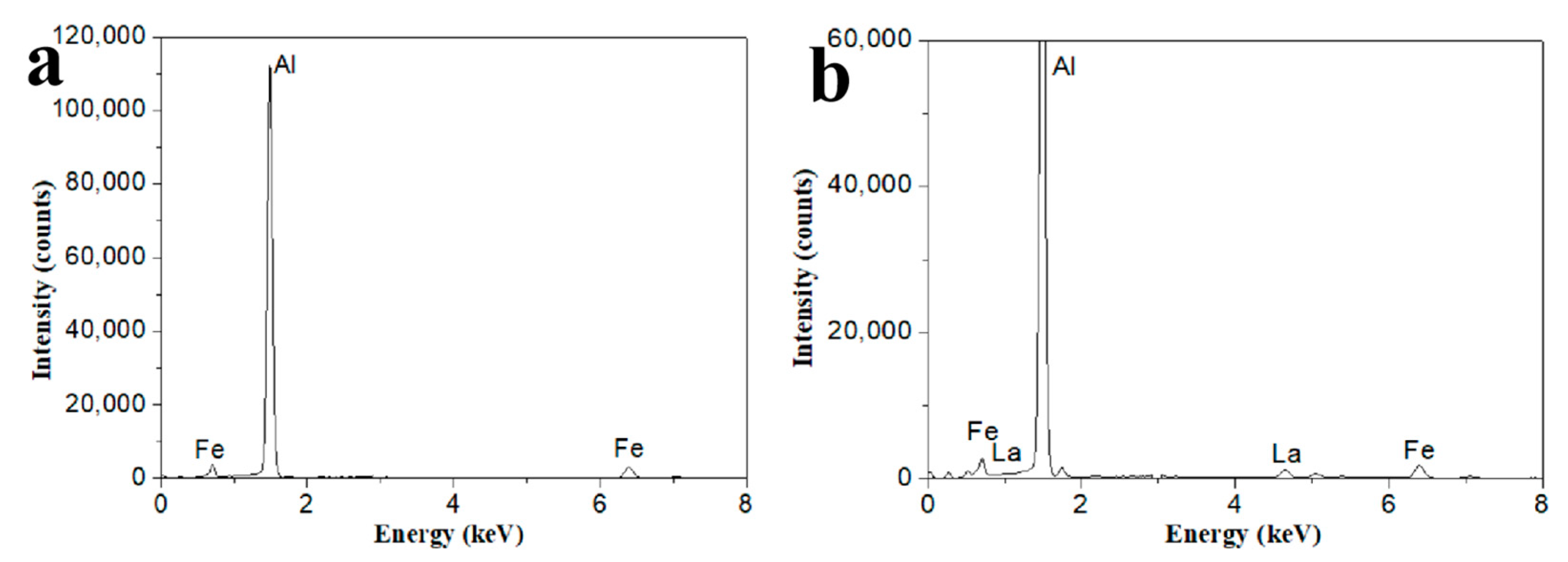
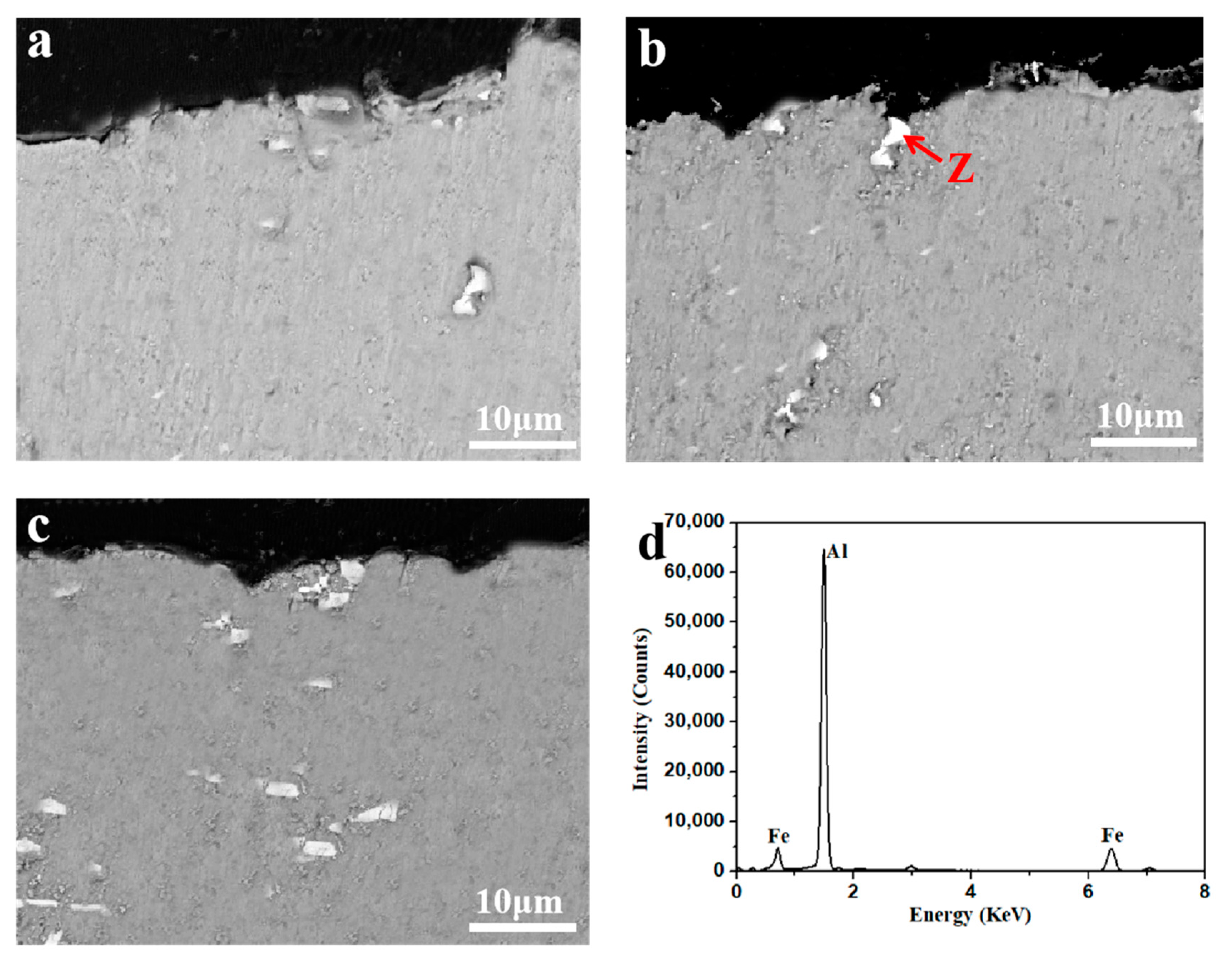
3.1. Microstructure
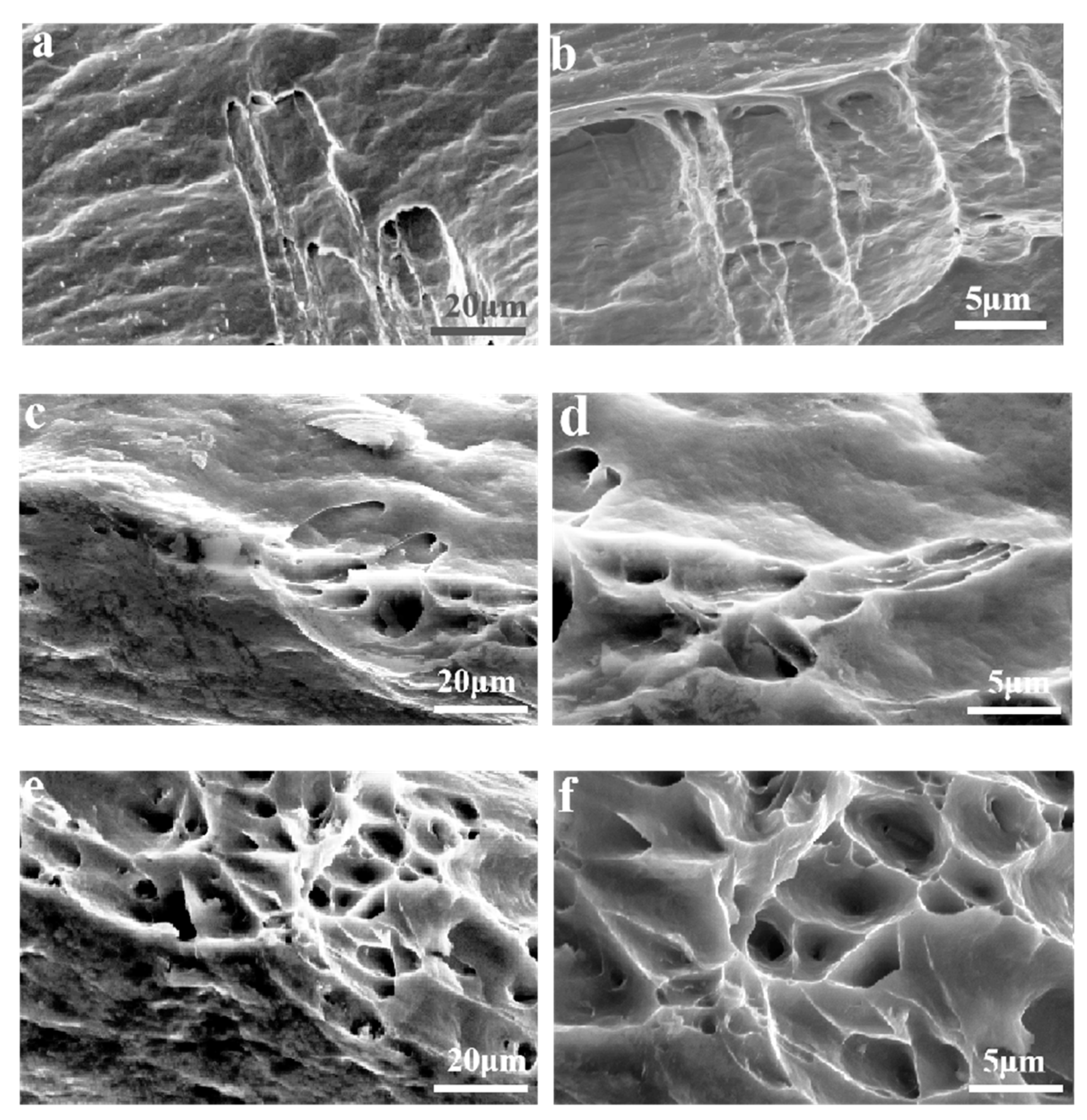
3.2. Tensile Properties
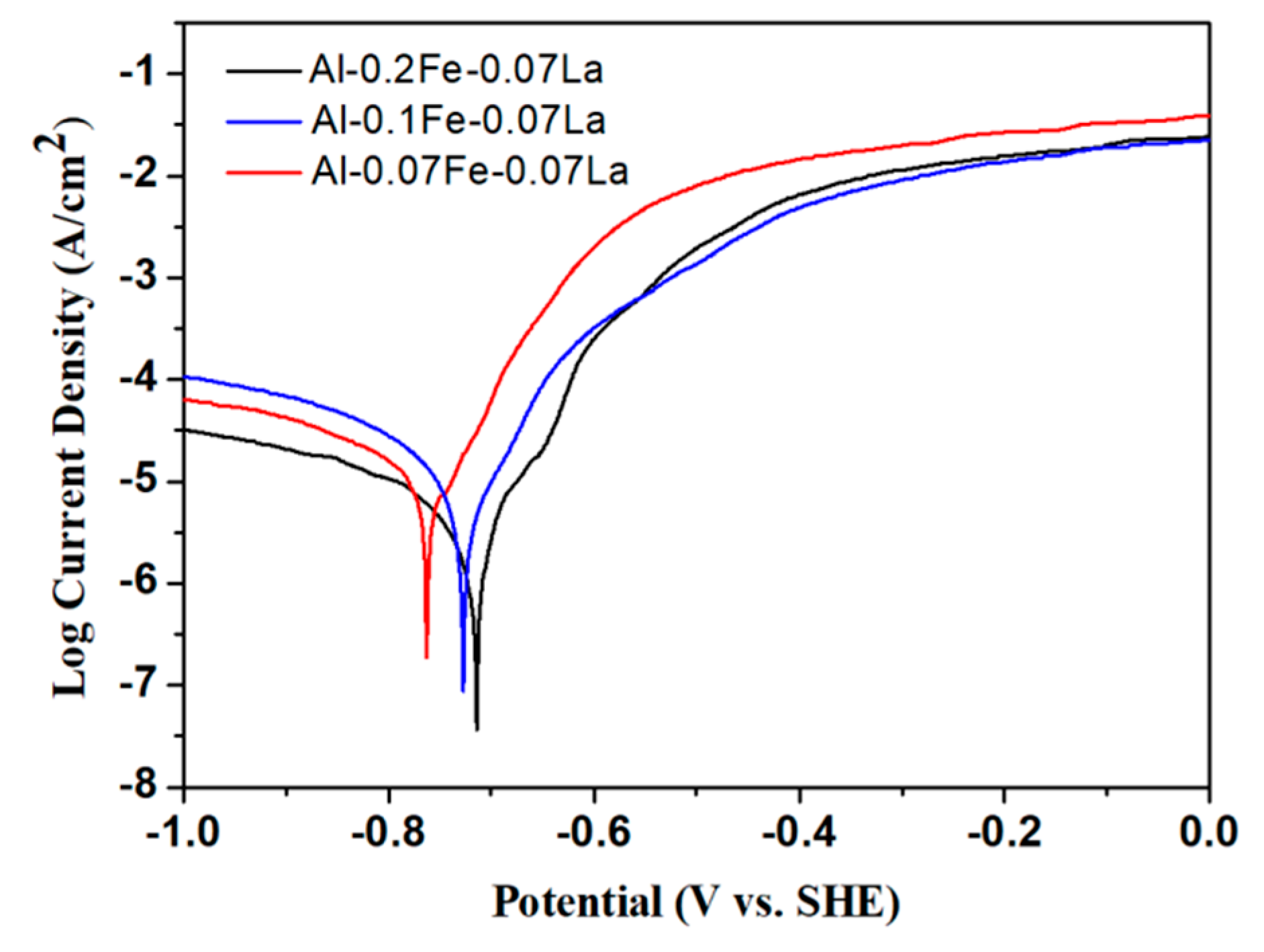
3.3. Electrochemical Performance
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Manthiram, A. Materials challenges and opportunities of lithium ion batteries. J. Phys. Chem. Lett. 2011, 2, 176–184. [Google Scholar] [CrossRef]
- Iwakura, C.; Fukumoto, Y.; Inoue, H.; Ohashi, S.; Kobayashi, S.; Tada, H.; Abe, M. Electrochemical characterization of various metal foils as a current collector of positive electrode for rechargeable lithium batteries. J. Power Sour. 1997, 68, 301–303. [Google Scholar] [CrossRef]
- Whitehead, A.H.; Schreiber, M. Current collectors for positive electrodes of lithium-based batteries. J. Electrochem. Soc. 2006, 152, A2105. [Google Scholar] [CrossRef]
- Church, B.C.; Kaminski, D.T.; Jiang, J. Corrosion of aluminum electrodes in aqueous slurries for lithium-ion batteries. J. Mater. Sci. 2014, 49, 3234–3241. [Google Scholar] [CrossRef]
- Raghavan, V. Al-Fe-La (Aluminum-Iron-Lanthanum). J. Phase Equilibria 2001, 22, 566–567. [Google Scholar] [CrossRef]
- Li, G.; Bian, X.; Song, K.; Guo, J.; Li, X.; Wang, C. Effect of Si addition on glass forming ability and thermal stability of Al–Fe–La alloys. J. Alloys Compd. 2009, 471, L47–L50. [Google Scholar] [CrossRef]
- Liang, Y.H.; Shi, Z.M.; Li, G.W.; Zhang, R.Y.; Zhao, G. Effects of Er addition on the crystallization characteristic and microstructure of Al-2wt%Fe cast alloy. J. Alloys Compd. 2019, 781, 235–244. [Google Scholar] [CrossRef]
- Lai, X.F.; Peng, H.P.; Wang, J.H.; Wu, C.J.; Tu, H.; Liu, Y.; Su, X.P. Effect of La on intermetallic layer of galvalume. Surf. Eng. 2013, 29, 390–395. [Google Scholar] [CrossRef]
- Miyake, J.; Fine, M.E. Electrical conductivity versus strength in a precipitation hardened alloy. Acta Metall. Mater. 1992, 40, 733–741. [Google Scholar] [CrossRef]
- Kino, T.; Endo, T.; Kawata, S. Deviations from matthiessen’s rule of the electrical resistivity of dislocations in aluminum. J. Phys. Soc. Jpn. 1974, 36, 698–705. [Google Scholar] [CrossRef]
- Han, K.; Walsh, R.P.; Ishmaku, A.; Toplosky, V.; Brandao, L.; Embury, J.D. High strength and high electrical conductivity bulk Cu. Philos. Mag. 2004, 84, 3705–3716. [Google Scholar] [CrossRef]
- Valiev, R.Z.; Murashkin, M.Y.; Sabirov, I. A nanostructural design to produce high-strength Al alloys with enhanced electrical conductivity. Scr. Mater. 2014, 76, 13–16. [Google Scholar] [CrossRef]
- Sauvage, X.; Bobruk, E.V.; Murashkin, M.Y.; Nasedkina, Y.N.; Enikeev, A.R.; Valiev, Z. Optimization of electrical conductivity and strength combination by structure design at the nanoscale in Al-Mg-Si alloys. Acta Mater. 2015, 98, 355–366. [Google Scholar] [CrossRef]
- Hou, J.P.; Wang, Q.; Zhang, Z.J.; Tian, Y.Z.; Wu, X.M.; Yang, H.J.; Li, X.W.; Zhang, Z.F. Nano-scale precipitates: the key to high strength and high conductivity in Al alloy wire. Mater. Des. 2017, 132, 148–157. [Google Scholar] [CrossRef]
- Hou, J.P.; Wang, Q.; Zhang, Z.J.; Tian, Y.Z.; Wu, X.M.; Yang, H.J.; Li, X.W.; Zhang, Z.F. Breaking the trade-off relation of strength and electrical conductivity in pure Al wire by controlling texture and grain boundary. J. Alloys Compd. 2018, 769, 96–109. [Google Scholar] [CrossRef]
- Sherby, O.D.; Taleff, E.M. Influence of grain size, solute atoms and second-phase particles on creep behavior of polycrystalline solids. Mater. Sci. Eng. A 2002, 322, 89–99. [Google Scholar] [CrossRef]
- Yamanaka, A.; Okamoto, M. Grain growth in a system containing finely dispersed mobile second-phase particles: a GPU-accelerated multi-phase-field study. In Proceedings of the 6th International Conference on Recrystallization and Grain Growth (ReX&GG 2016), Pittsburgh, PA, USA, 17–21 July 2016. [Google Scholar]
- Brunner, J.G.; Birbilis, N.; Ralston, K.D.; Virtanen, S. Impact of ultrafine-grained microstructure on the corrosion of aluminium alloy AA2024. Corros. Sci. 2012, 57, 209–214. [Google Scholar] [CrossRef]
- Ralston, K.D.; Fabijanic, D.; Birbilis, N. Effect of grain size on corrosion of high purity aluminium. Electrochim. Acta 2011, 56, 1729–1736. [Google Scholar] [CrossRef]
- Mi, G.F.; Dong, C.F.; Zhao, D.W. Effect of different cooling speed and rare earth La on the microstructures and phase constitution of Al-Fe alloy. Appl. Mech. Mater. 2010, 44–47, 2126–2130. [Google Scholar] [CrossRef]
- Khan, M.; Amjad, M.; Khan, A.; Ud-Din, R.; Ahmad, I.; Subhani, T. Microstructural evolution, mechanical profile, and fracture morphology of aluminum matrix composites containing graphene nanoplatelets. J. Mater. Res. 2017, 32, 2055–2066. [Google Scholar] [CrossRef]
- Srivatsan, T.S.; Sriram, S.; Veeraraghavan, D.; Vasudevan, V.K. Microstructure, tensile deformation and fracture behaviour of aluminium alloy 7055. J. Mater. Sci. 1997, 32, 2883–2894. [Google Scholar] [CrossRef]
- Zhang, J.C.; Ding, D.Y.; Xu, X.L.; Gao, Y.J.; Chen, G.Z.; Chen, W.G.; You, X.H.; Huang, Y.W.; Tang, J.S. Effect of Ce addition on the mechanical and electrochemical properties of a lithium battery shell alloy. J. Alloys Compd. 2014, 617, 665–669. [Google Scholar] [CrossRef]
- Zhang, R.; Ding, D.Y.; Zhang, W.L.; Gao, Y.J.; Wu, Z.L.; Chen, G.Z.; Chen, R.Z.; Huang, Y.W.; Tang, J.S. Al-1.5Fe-xLa alloys for lithium-ion battery package. Metals (Basel) 2018, 8, 890. [Google Scholar] [CrossRef]
- Seri, O.; Furumata, K. Effect of Al-Fe-Si intermetallic compound phases on initiation and propagation of pitting attacks for aluminum 1100. Mater. Corros. 2002, 53, 111–120. [Google Scholar] [CrossRef]
- Park, J.O. Influence of Fe-Rich Intermetallic inclusions on pit initiation on aluminum alloys in aerated NaCl. J. Electrochem. Soc. 1999, 146, 517. [Google Scholar] [CrossRef]
- Nişancioğlu, K. Electrochemical behavior of aluminum-base intermetallics containing iron. J. Electrochem. Soc. 2006, 137, 69. [Google Scholar] [CrossRef]



| Alloy | σ0.2 (MPa) | σb (MPa) | δ (%) |
|---|---|---|---|
| Al-0.07Fe-0.07La | 129.31 (±1) | 134.40 (±1) | 0.69 (±0.05) |
| Al-0.1Fe-0.07La | 169.67 (±2) | 180.33 (±2) | 1.07 (±0.05) |
| Al-0.2Fe-0.07La | 195.50 (±1.5) | 213.00 (±1) | 1.30 (±0.01) |
| Alloy | Ecorr (mV vs. SCE) | Icorr (μA/cm2) |
|---|---|---|
| Al-0.07Fe-0.07La | −763 (±4) | 14.71 (±0.1) |
| Al-0.1Fe-0.07La | −728 (±10) | 8.66 (±0.1) |
| Al-0.2Fe-0.07La | −715 (±6) | 2.26 (±0.2) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, X.; Ding, D.; Xu, Y.; Zhang, W.; Gao, Y.; Wu, Z.; Chen, G.; Chen, R.; Huang, Y.; Tang, J. Tensile Properties and Corrosion Resistance of Al-xFe-La Alloys for Aluminium Current Collector of Lithium-Ion Batteries. Metals 2019, 9, 706. https://doi.org/10.3390/met9060706
Yang X, Ding D, Xu Y, Zhang W, Gao Y, Wu Z, Chen G, Chen R, Huang Y, Tang J. Tensile Properties and Corrosion Resistance of Al-xFe-La Alloys for Aluminium Current Collector of Lithium-Ion Batteries. Metals. 2019; 9(6):706. https://doi.org/10.3390/met9060706
Chicago/Turabian StyleYang, Xin, Dongyan Ding, Yawu Xu, Wenlong Zhang, Yongjin Gao, Zhanlin Wu, Guozhen Chen, Renzong Chen, Yuanwei Huang, and Jinsong Tang. 2019. "Tensile Properties and Corrosion Resistance of Al-xFe-La Alloys for Aluminium Current Collector of Lithium-Ion Batteries" Metals 9, no. 6: 706. https://doi.org/10.3390/met9060706
APA StyleYang, X., Ding, D., Xu, Y., Zhang, W., Gao, Y., Wu, Z., Chen, G., Chen, R., Huang, Y., & Tang, J. (2019). Tensile Properties and Corrosion Resistance of Al-xFe-La Alloys for Aluminium Current Collector of Lithium-Ion Batteries. Metals, 9(6), 706. https://doi.org/10.3390/met9060706





