Effects of Different Parameters on Initiation and Propagation of Stress Corrosion Cracks in Pipeline Steels: A Review
Abstract
1. Introduction
2. Explanation of SCC and HIC
3. Role of Microstructure on the SCC Cracks
4. Role of Stress Intensity Factor (SIF) in SCC and HIC Crack Propagation
5. Role of Surface Films in SCC
6. Effect of Residual Stress on the SCC Crack Propagation
7. Role of AC Current on the SCC Susceptibility
8. Type of Environment on the SCC Behavior
9. Effect of Different Elements on SCC Susceptibility
10. Effect of Crystallographic Texture and Meso-Texture on SCC Susceptibility
11. Improvement of SCC Resistance
12. Conclusions
- (1)
- Microstructure of steel plays a key role on the SCC crack initiation and propagation. Nature of the SCC cracks, specially transgranular crack propagation, highly depends on the microstructure of steel. Other metallurgical factors including grain size and grain boundaries become determinative factors during intergranular cracking.
- (2)
- The effect of residual stresses in crack initiation and propagation should not be neglected. Tensile residual stress gives a high amount of mechanical driving force for crack initiation and short crack propagation.
- (3)
- AC current density may affect the crack propagation by two ways. First, AC current may break down the insulation layer of pipeline steel and destroy the cathodic protection system. Secondly, AC current density affects the nature of the SCC crack. When there is no AC current, the SCC cracks propagate through intergranular manner and its mechanism is the anodic dissolution. However, with the presence of AC current, the SCC susceptibility is increased and the SCC cracks propagate through transgranular manner at high current densities.
- (4)
- In a subsea environment, the permeation of sea water in the gaps will promote the peeling of the coating resulting in a complicated and more severe corrosion in pipeline steel by destroying the coating of pipeline steel.
- (5)
- Addition of some elements to the pipeline steel have a substantial effect on the SCC susceptibility. For instance, Carbon, manganese and phosphorus elements are segregated at the center of thickness of pipeline slabs and make some hard phases and structures such as bainite and martensite. Copper also makes a protective layer on the surface of steel and decrease the hydrogen diffusion inside the steel.
- (6)
- The SCC crack propagation near-neutral pH solution does not occur under constant loading and fluctuation loading is required to initiate and propagate the cracks. The time of crack initiation near-neutral environment under constant loading is decreased when preload is applied.
- (7)
- Crystallographic texture plays a key role in SCC crack propagation. Grains with {111}∥rolling plane and {110}∥rolling plane, coincidence site lattice boundaries and low angle grain boundaries are recognized as crack resistant paths while grains with high angle grain boundaries provides easy path for intergranular SCC crack propagation.
Acknowledgments
Conflicts of Interest
References
- Liu, Z.Y.; Li, X.G.; Du, C.W.; Lu, L.; Zhang, Y.R.; Cheng, Y.F. Effect of inclusions on initiation of stress corrosion cracks in X70 pipeline steel in an acidic soil environment. Corros. Sci. 2009, 51, 895–900. [Google Scholar] [CrossRef]
- Shi, X.B.; Yan, W.; Wang, W.; Zhao, L.Y.; Shan, Y.Y.; Yang, K. HIC and SSC behavior of high-strength pipeline steels. Acta Metall. Sinica 2015, 28, 799–808. [Google Scholar] [CrossRef]
- Hara, T.; Asahi, H.; Ogawa, H. Conditions of hydrogen-induced corrosion occurrence of X65 grade linepipe steels in sour environments. Corros. Sci. 2004, 60, 1113–1121. [Google Scholar] [CrossRef]
- Shi, X.B.; Yan, W.; Wang, W.; Zhao, L.Y.; Shan, Y.Y.; Yang, K. Effect of microstructure on hydrogen induced cracking behavior of a high deformability pipeline steel. J. Iron Steel Res. 2015, 22, 937–942. [Google Scholar] [CrossRef]
- Kim, W.K.; Koh, S.U.; Yang, B.Y.; Kim, K.Y. Effect of environment and metallurgical factors on hydrogen induced cracking. Corros. Sci. 2008, 50, 3336–3342. [Google Scholar] [CrossRef]
- Maciejewski, J. The effects of sulfide inclusions on mechanical properties and failures of steel components. J. Fail. Anal. Prev. 2015, 15, 169–178. [Google Scholar] [CrossRef]
- Mohtadi-Bonab, M.A.; Szpunar, J.A.; Razavi-Tousi, S.S. A comparative study of hydrogen induced cracking behavior in API 5L X60 and X70 pipeline steels. Eng. Fail. Anal. 2013, 33, 163–175. [Google Scholar] [CrossRef]
- Mohtadi-Bonab, M.A.; Eskandari, M.; Karimdadashi, R.; Szpunar, J.A. Effect of different microstructural parameters on hydrogen induced cracking in an API X70 pipeline steel. Met. Mater. Int. 2017, 23, 726–735. [Google Scholar] [CrossRef]
- Fan, Z.; Hu, X.; Liu, J.; Li, H.; Fu, J. Stress corrosion cracking of L360NS pipeline steel in sulfur environment. Petroleum 2017, 3, 377–383. [Google Scholar] [CrossRef]
- Mohtadi-Bonab, M.A.; Eskandari, M.; Ghaednia, H.; Das, S. Effect of microstructural parameters on fatigue crack propagation in an API X65 pipeline steel. J. Mater. Eng. Perform. 2016, 25, 4933–4940. [Google Scholar] [CrossRef]
- Arafin, M.A.; Szpunar, J.A. Effect of bainitic microstructure on the susceptibility of pipeline steels to hydrogen induced cracking. Mater. Sci. Eng. A 2011, 528, 4927–4940. [Google Scholar] [CrossRef]
- Arafin, M.A.; Szpunar, J.A. A new understanding of intergranular stress corrosion cracking resistance of pipeline steel through grain boundary character and crystallographic texture studies. Corros. Sci. 2009, 51, 119–128. [Google Scholar] [CrossRef]
- Wright, J. Inhibiting rust and corrosion to prevent machine failures. In Proceedings of the Machinary Lubrication Conference and Exhibition, Houston, TX, USA, 6–8 November 2018. [Google Scholar]
- National Energy Board. Stress Corrosion Cracking on Canadian Oil and Gas Pipelines; Report No. MH-2-95; National Energy Board: Calgary, AB, Canada, 1996.
- Aly, O.F.; Neto, M.M. Stress Corrosion Cracking, Developments in Corrosion Protection; Aliofkhazraei, M., Ed.; IntechOpen Limited: London, UK, 2014. [Google Scholar]
- Sutcliffe, J.M.; Fessler, R.R.; Boyd, W.K.; Parkins, R.N. Stress corrosion cracking of carbon steel in carbonate solutions. Corrosion 1972, 28, 313–320. [Google Scholar] [CrossRef]
- Beavers, J.A.; Harle, B.A. Mechanisms of high-pH and nearneutral-pH SCC of underground pipelines. Offshore Mech. Arct. Eng. 2003, 123, 147–151. [Google Scholar] [CrossRef]
- Charles, E.A.; Parkins, R.N. Generation of stress corrosion cracking environments at pipeline surfaces. Corrosion 1995, 51, 518–527. [Google Scholar] [CrossRef]
- Fang, B.Y.; Atrens, A.; Wang, J.Q.; Han, E.H.; Zhu, Z.Y.; Ke, W. Review of stress corrosion cracking of pipeline steels in “low” and “high” pH solutions. J. Mater. Sci. 2003, 38, 127–132. [Google Scholar] [CrossRef]
- Mohtadi-Bonab, M.A.; Ghesmati-Kucheki, H. Important Factors on the Failure of Pipeline Steels with Focus on Hydrogen Induced Cracks and Improvement of Their Resistance: Review Paper. Met. Mater. Int. 2019. [Google Scholar] [CrossRef]
- Kadhim, M.G.; Albdiry, M. A critical review on corrosion and its prevention in the oilfield equipment. J. Petrol. Res. Stud. 2017, 14, 162–189. [Google Scholar]
- Zhu, M.; Du, C.; Li, X.; Liu, Z.; Wang, S.; Zhao, T.; Jia, J. Effect of strength and microstructure on stress corrosion cracking behavior and mechanism of X80 pipeline steel in high pH carbonate/bicarbonate solution. J. Mech. Eng. Perform. 2014, 23, 1358–1365. [Google Scholar] [CrossRef]
- Gonzalez, J.; Gutierrez-Solana, F.; Varona, J.M. The effects of microstructure, strength level, and crack propagation mode on stress corrosion cracking behavior of 4135 steel. Met. Mater. Trans. A 1994, 27, 281–290. [Google Scholar] [CrossRef]
- Masouri, D.; Zafari, M.; Araghi, A. Sulfide stress cracking of pipeline-case history. In Proceedings of the NACE International, Corrosion 2008 Proceedings, New Orleans, LA, USA, 16–20 March 2008. [Google Scholar]
- Lancsater, J. Handbook of Structural Welding: Processes, Materials and Methods in the Welding of Major Structures, Pipelines and Process Plant; Woodhead Publishing: Sawston, UK, 2003. [Google Scholar]
- Roffey, P.; Davies, E.H. The generation of corrosion under insulation and stress corrosion cracking due to sulphide stress cracking in an austenitic stainless steel hydrocarbon gas pipeline. Eng. Fail. Anal. 2014, 44, 148–157. [Google Scholar] [CrossRef]
- Elboujdaini, M.; Revie, R.W. Metallurgical factors in stress corrosion cracking (SCC) and hydrogen-induced cracking (HIC). J. Solid State Electrochem. 2009, 13, 1091–1099. [Google Scholar] [CrossRef]
- Chen, W. Modeling and prediction of stress corrosion cracking of pipeline steels, Trends in Oil and Gas Corrosion Research and Technologies; Woodhead Publishing Series in Energy; Woodhead Publishing: Sawston, UK, 2017; pp. 707–748. [Google Scholar]
- Hongliang, Y.; He, X.; Fuqiang, Y.; Lingyan, Z. Effect of film-induced stress on mechanical properties at stress corrosion cracking tip. Rare Met. Mater. Eng. 2017, 46, 3595–3600. [Google Scholar] [CrossRef]
- Austen, I.M. Effective stress intensities in stress corrosion cracking. Int. J. Fract. 1976, 12, 253–263. [Google Scholar] [CrossRef]
- Costin, W.L.; Lavigne, O.; Kotousov, A.; Ghomashchi, R.; Linton, V. Investigation of hydrogen assisted cracking in acicular ferrite using site-specific micro-fracture tests. Mater. Sci. Eng. A 2016, 651, 859–868. [Google Scholar] [CrossRef]
- Wang, J.Q.; Atrens, A. SCC initiation for X65 pipeline steel in the high pH carbonate/bicarbonate solution. Corros. Sci. 2003, 45, 2199–2217. [Google Scholar] [CrossRef]
- Chen, W.; van Boven, G.; Rogge, R. The role of residual stress in neutral pH stress corrosion cracking of pipeline steels—Part II: Crack dormancy. Acta Mater. 2007, 55, 43–53. [Google Scholar] [CrossRef]
- Zhu, M.; Du, C.; Li, X.; Liu, Z.; Li, H.; Zhang, D. Effect of AC on stress corrosion cracking behavior and mechanism of X80 pipeline steel in carbonate/bicarbonate solution. Corros. Sci. 2014, 87, 224–232. [Google Scholar] [CrossRef]
- Hosokawa, Y.; Kajiyama, F.; Fukuoka, T. Alternating current corrosion risk arising from alternating current-powered rail transit systems on cathodically protected buried steel pipelines and its measures. Corrosion 2004, 60, 408–413. [Google Scholar] [CrossRef]
- Tan, T.C.; Chin, D.T. Ac corrosion of nickel in sulphate solutions. J. Appl. Electrochem. 1988, 18, 831–838. [Google Scholar] [CrossRef]
- Goidanich, S.; Lazzari, L.; Ormellese, M.; Pedeferri, M. Influence of AC on Corrosion Kinetics for Carbon Steel, Zinc and Copper; CORROSION/2005, Paper No. 05189; NACE International: Houston, TX, USA, 2005. [Google Scholar]
- Wendt, J.L.; Chin, D.T. The A.C. corrosion of stainless steel – II. The breakdown of passivity of ss304 in neutral aqueous solutions. Corros. Sci. 1985, 25, 889–900. [Google Scholar]
- Muralidharan, S.; Kim, D.K.; Ha, T.H.; Bae, J.H.; Ha, Y.C.; Lee, H.G.; Scantlebury, J. Influence of alternating, direct and superimposed alternating and direct current on the corrosion of mild steel in marine environments. Desalination 2007, 216, 103–115. [Google Scholar] [CrossRef]
- Vasudevan, S.; Lakshmi, J. Effects of alternating and direct current in electrocoagulation process on the removal of cadmium from waterea novel approach. Sep. Purif. Technol. 2011, 80, 643–651. [Google Scholar] [CrossRef]
- Jiang, Z.; Du, Y.; Lu, M.; Zhang, Y.; Tang, D.; Dong, L. New findings on the factors accelerating AC corrosion of buried pipeline. Corros. Sci. 2014, 81, 1–10. [Google Scholar] [CrossRef]
- Wan, H.; Song, D.; Liu, Z.; Du, C.; Zeng, Z.; Yang, X.; Li, X. Effect of alternating current on stress corrosion cracking behavior and mechanism of X80 pipeline steel in near-neutral solution. J. Nat. Gas Sci. Eng. 2017, 38, 458–465. [Google Scholar] [CrossRef]
- Luo, J.; Zhang, L.; Li, L.; Yang, F.; Ma, W.; Wang, K.; Zhao, X. Electrochemical corrosion behaviors of the X90 linepipe steel in NS4 solution. Nat. Gas. Ind. 2016, 3, 346–351. [Google Scholar] [CrossRef]
- Zhu, M.; Du, C.W. A new understanding on AC corrosion of pipeline steel in alkaline environment. J. Mater. Eng. Perform. 2017, 26, 221–228. [Google Scholar] [CrossRef]
- Zhu, M.; Du, C.; Li, X.; Liu, Z.; Wang, S.; Li, J.; Zhang, D. Effect of AC current density on stress corrosion cracking behavior of X80 pipeline steel in high pH carbonate/bicarbonate solution. Electrochem. Acta 2014, 117, 351–359. [Google Scholar] [CrossRef]
- Rodríguez, J.J.S.; Hernández, F.J.S.; González, J.E.G. The effect of environmental and meteorological variables on atmospheric corrosion of carbon steel, copper, zinc and aluminium in a limited geographic zone with different types of environment. Corros. Sci. 2003, 45, 799–815. [Google Scholar] [CrossRef]
- Iakovleva, E.; Mäkilä, E.; Salonen, J.; Sitarz, M.; Sillanpää, M. Industrial products and wastes as adsorbents for sulphate and chloride removal from synthetic alkaline solution and mine process water. Chem. Eng. J. 2015, 259, 364–371. [Google Scholar] [CrossRef]
- Mustapha, A.; Charles, E.A.; Hardie, D. Evaluation of environment-assisted cracking susceptibility of a grade X100 pipeline steel. Corros. Sci. 2012, 54, 5–9. [Google Scholar] [CrossRef]
- Oskuie, A.A.; Shahrabi, T.; Shahriari, A.; Saebnoori, E. Electrochemical impedance spectroscopy analysis of X70 pipeline steel stress corrosion cracking in high pH carbonate solution. Corros. Sci. 2012, 61, 111–122. [Google Scholar] [CrossRef]
- Yan, M.C.; Xu, J.; Yu, L.B.; Wu, T.Q.; Sun, C.; Ke, W. EIS analysis on stress corrosion initiation of pipeline steel under disbonded coating in near-neutral pH simulated soil electrolyte. Corros. Sci. 2016, 110, 23–34. [Google Scholar]
- Kang, Y.W.; Chen, W.X.; Kania, R.; Boven, G.V.; Worthingham, R. Simulation of crack growth during hydrostatic testing of pipeline steel in near-neutral pH environment. Corros. Sci. 2011, 53, 968–975. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, J.W.; Liu, L.; Li, Y.; Wang, F.H. Study of the failure mechanism of an epoxy coating system under high hydrostatic pressure. Corros. Sci. 2013, 74, 59–70. [Google Scholar] [CrossRef]
- Suna, F.; Ren, S.; Li, Z.; Liu, Z.; Li, X.; Du, C. Comparative study on the stress corrosion cracking of X70 pipeline steel in simulated shallow and deep sea environments. Mater. Sci. Eng. A 2017, 685, 145–153. [Google Scholar] [CrossRef]
- Marshakov, A.I.; Ignatenko, V.E.; Bogdanov, R.I.; Arabey, A.B. Effect of electrolyte composition on crack growth rate in pipeline steel. Corros. Sci. 2014, 83, 209–216. [Google Scholar] [CrossRef]
- Parkins, R.N.; Blanchard, W.K., Jr.; Delanty, B.S. Transgranular stress corrosion cracking of high-pressure pipelines in contact with solutions of near-neutral-pH. Corrosion 1994, 50, 394–408. [Google Scholar] [CrossRef]
- Lu, B.T.; Luo, J.L.; Norton, P.R. Environmentally assisted cracking mechanism of pipeline steel in near-neutral pH groundwater. Corros. Sci. 2010, 52, 1787–1795. [Google Scholar] [CrossRef]
- Lu, B.T.; Luo, J.L.; Norton, P.R.; Ma, H.Y. Effects of dissolved hydrogen and elastic and plastic deformation on active dissolution of pipeline steel in anaerobic groundwater of near-neutral pH. Acta Mater. 2009, 57, 41–49. [Google Scholar] [CrossRef]
- Liu, Z.Y.; Wang, X.Z.; Du, C.W.; Li, J.K.; Li, X.G. Effect of hydrogen-induced plasticity on the stress corrosion cracking of X70 pipeline steel in simulated soil environments. Mater. Sci. Eng. A 2016, 658, 348–354. [Google Scholar] [CrossRef]
- Mohtadi-Bonab, M.A.; Eskandari, M.; Szpunar, J.A. Role of cold rolled followed by annealing on improvement of hydrogen induced cracking resistance in pipeline steel. Eng. Fail. Anal. 2018, 91, 172–181. [Google Scholar] [CrossRef]
- Mohtadi-Bonab, M.A.; Eskandari, M.; Szpunar, J.A. Effect of arisen dislocation density and texture components during cold rolling and annealing treatments on hydrogen induced cracking susceptibility in pipeline steel. J. Mater. Res. 2016, 31, 3390–3400. [Google Scholar] [CrossRef]
- Fang, B.Y.; Han, E.H.; Zhu, Z.Y.; Wang, J.Q.; Ke, W. Stress corrosion cracking of pipeline steels. J. Mater. Sci. Technol. 2002, 18, 3–6. [Google Scholar]
- Lu, B.T.; Luo, J.L. Crack initiation and early propagation of X70 Steel in simulated near-neutral pH groundwater. Corrosion 2006, 62, 723–731. [Google Scholar] [CrossRef]
- Chen, W.X.; Kania, R.; Worthingham, R.; van Boven, G. Transgranular crack growth in the pipeline steels exposed to near-neutral pH soil aqueous solutions-The role of hydrogen. Acta Mater. 2009, 57, 6200–6214. [Google Scholar] [CrossRef]
- Chen, W.; Wang, S.H.; Chu, R.; King, F.; Jack, T.R.; Fessler, R.R. Effect of precyclic loading on stress-corrosion-cracking initiation in an X-65 pipeline steel exposed to near-neutral pH soil environment. Metall. Mater. Trans. A 2003, 34, 2601–2608. [Google Scholar] [CrossRef]
- Jia, Y.Z.; Wang, J.Q.; Han, E.H.; Ke, W. Stress corrosion cracking of x80 pipeline steel in near-neutral pH environment under constant load tests with and without preload. J. Mater. Sci. Technol. 2011, 27, 1039–1046. [Google Scholar] [CrossRef]
- Ghosh, G.; Rostron, P.; Garg, R.; Panday, A. Hydrogen induced cracking of pipeline and pressure vessel steels: A review. Eng. Fract. Mech. 2018, 199, 609–618. [Google Scholar] [CrossRef]
- Dayal, R.K.; Grabke, H.J. Hydrogen induced stress corrosion cracking in low and high strength ferritic steels of different phosphorus content in acid media. Mater. Corros. 1987, 38, 409–416. [Google Scholar] [CrossRef]
- Mohtadi-Bonab, M.A.; Szpunar, J.A.; Basu, R.; Eskandari, M. The mechanism of failure by hydrogen induced cracking in an acidic environment for API 5L X70 pipeline steel. Int. J. Hydrogen Energy 2015, 40, 1096–1107. [Google Scholar] [CrossRef]
- Mohtadi-Bonab, M.A.; Eskandari, M. A focus on different factors affecting hydrogen induced cracking in oil and natural gas pipeline steel. Eng. Fail. Anal. 2017, 79, 351–360. [Google Scholar] [CrossRef]
- Mohtadi-Bonab, M.A.; Eskandari, M.; Sanayei, M.; Das, S. Microstructural aspects of intergranular and transgranular crack propagation in an API X65 steel pipeline related to fatigue failure. Eng. Fail. Anal. 2018, 94, 214–225. [Google Scholar] [CrossRef]
- Shi, X.; Yan, W.; Wang, W.; Shan, Y.; Yang, K. Novel Cu-bearing high-strength pipeline steels with excellent resistance to hydrogen-induced cracking. Mater. Des. 2016, 92, 300–305. [Google Scholar] [CrossRef]
- Baba, K.; Mizuno, M.D.; Yasuda, K.; Nakamichi, H.; Ishikawa, N. Effect of Cu addition in pipeline steels on prevention of hydrogen permeation in mildly sour environments. Corrosion 2016, 72, 1107–1115. [Google Scholar] [CrossRef]
- Bulger, J.T.; Lu, B.T.; Luo, J.L. Microstructural effect on near-neutral pH stress corrosion cracking resistance of pipeline steels. J. Mater. Sci. 2006, 41, 5001–5005. [Google Scholar] [CrossRef]
- Wang, L.; Xin, J.; Cheng, L.; Zhao, K.; Sun, B.; Li, J.; Wang, X.; Cui, Z. Influence of inclusions on initiation of pitting corrosion and stress corrosion cracking of X70 steel in near-neutral pH environment. Corros. Sci. 2019, 147, 108–127. [Google Scholar] [CrossRef]
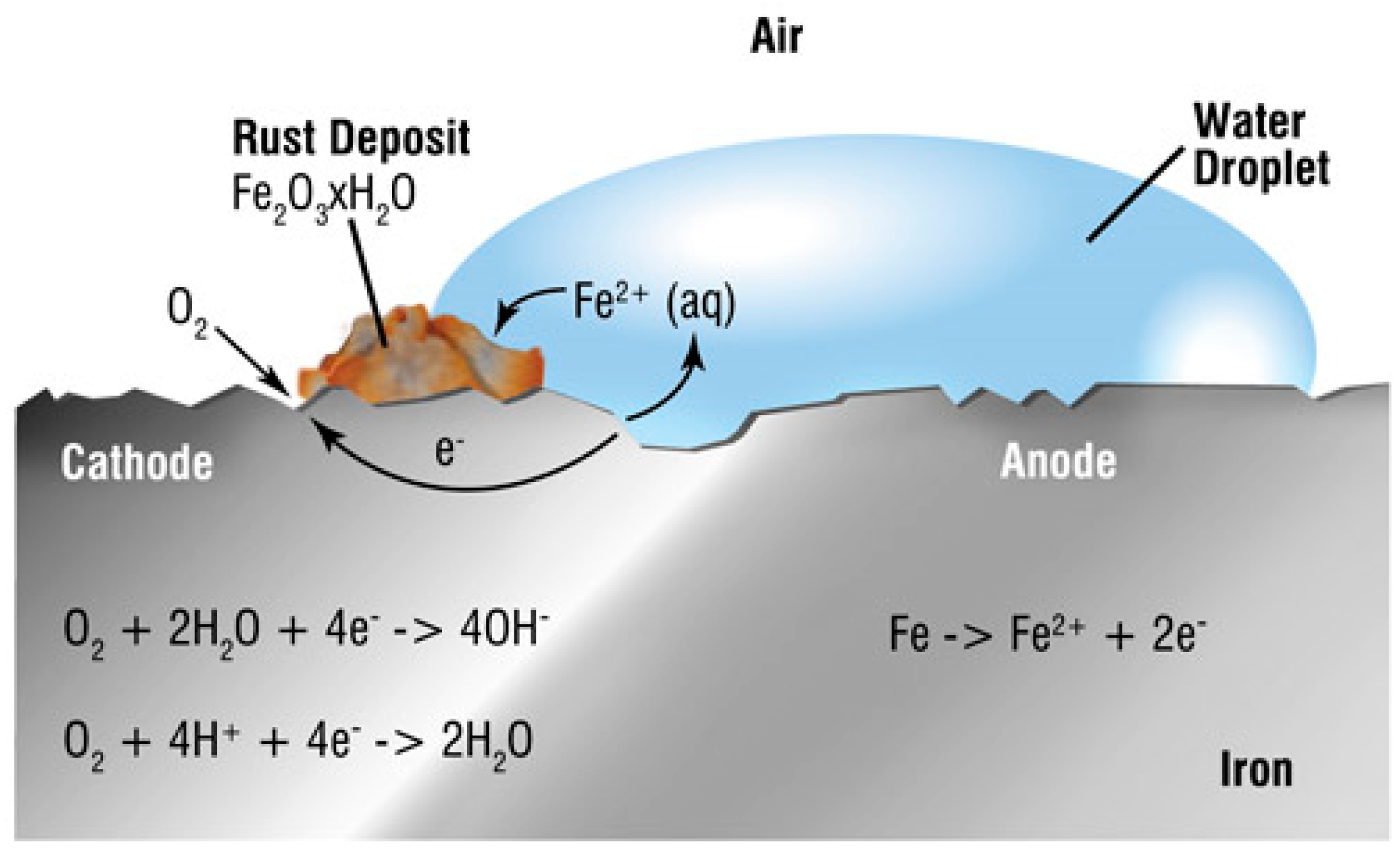
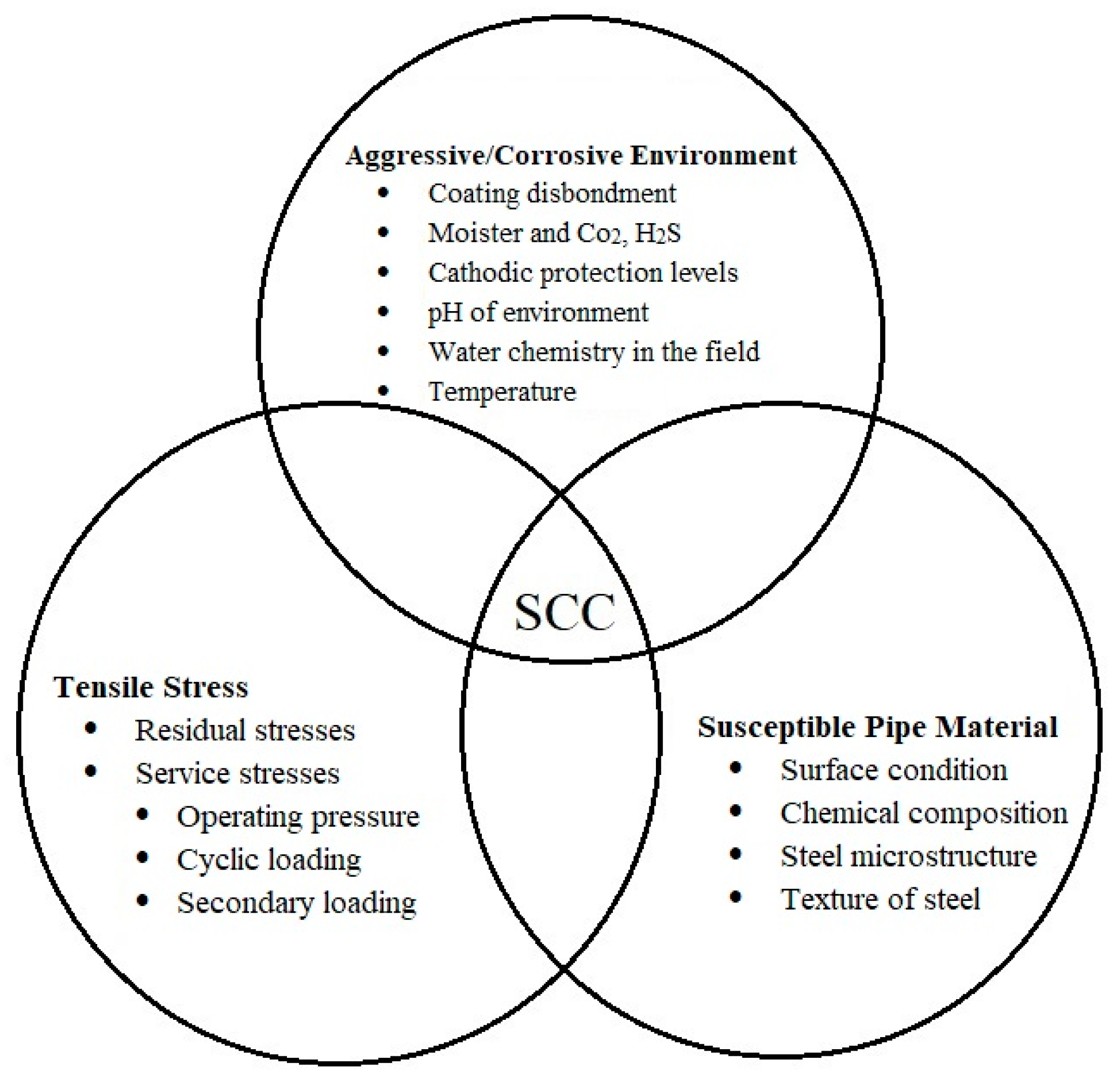


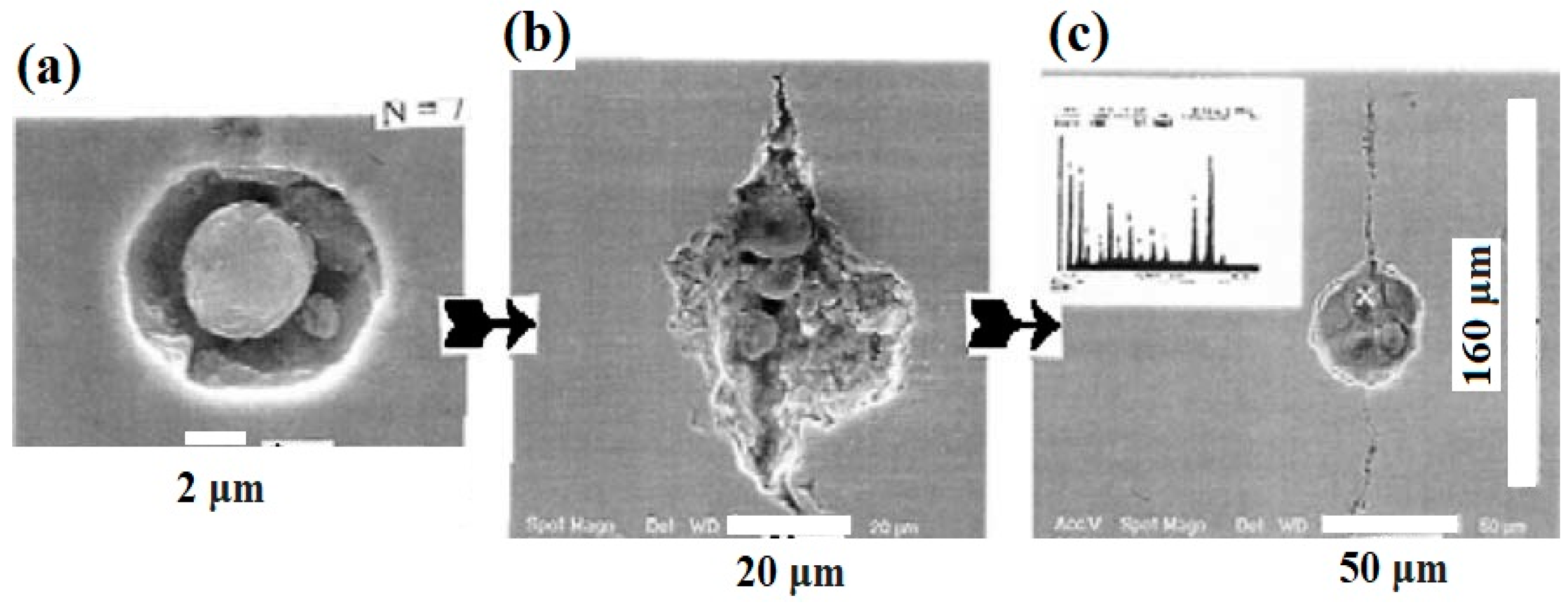
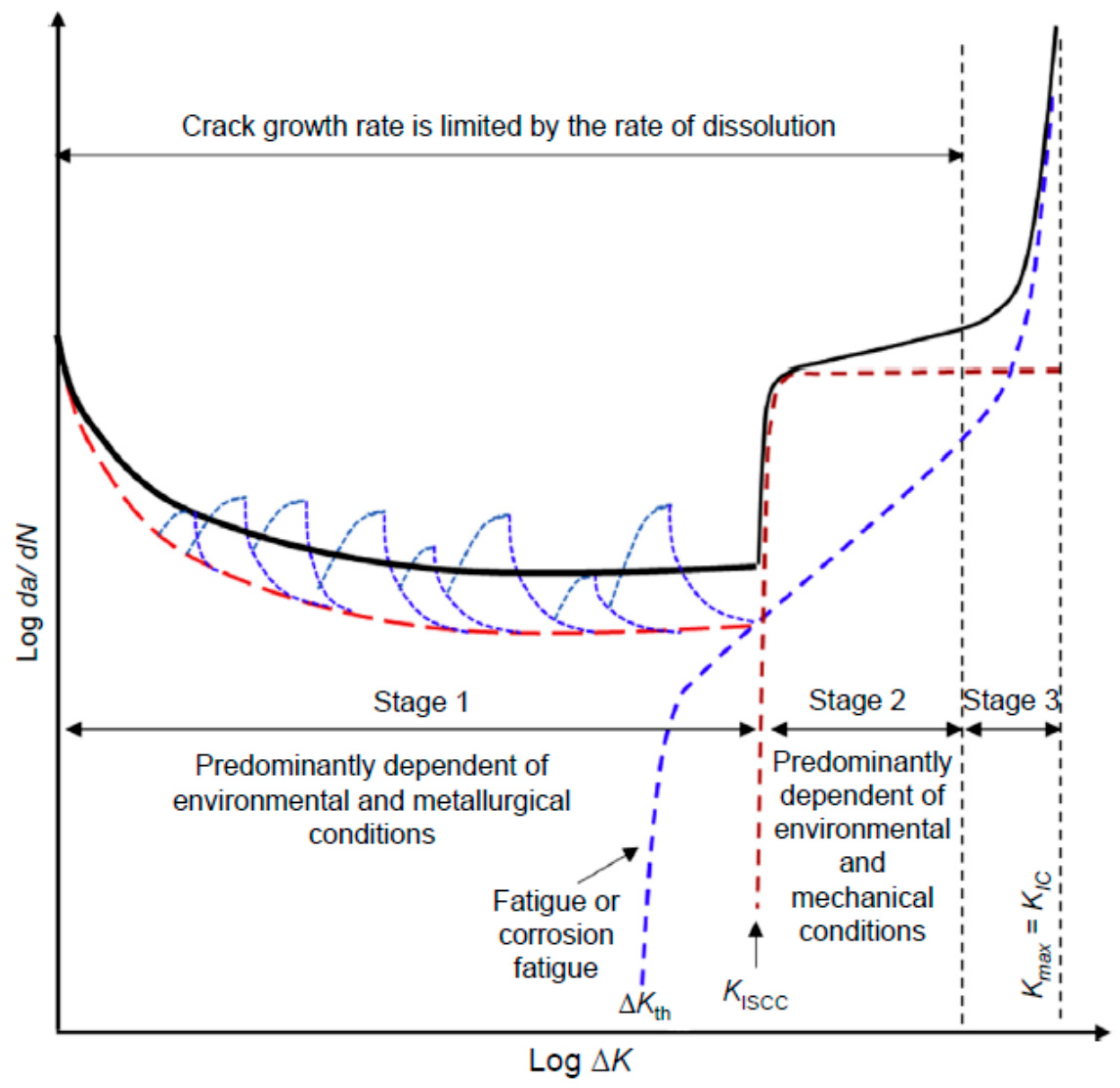

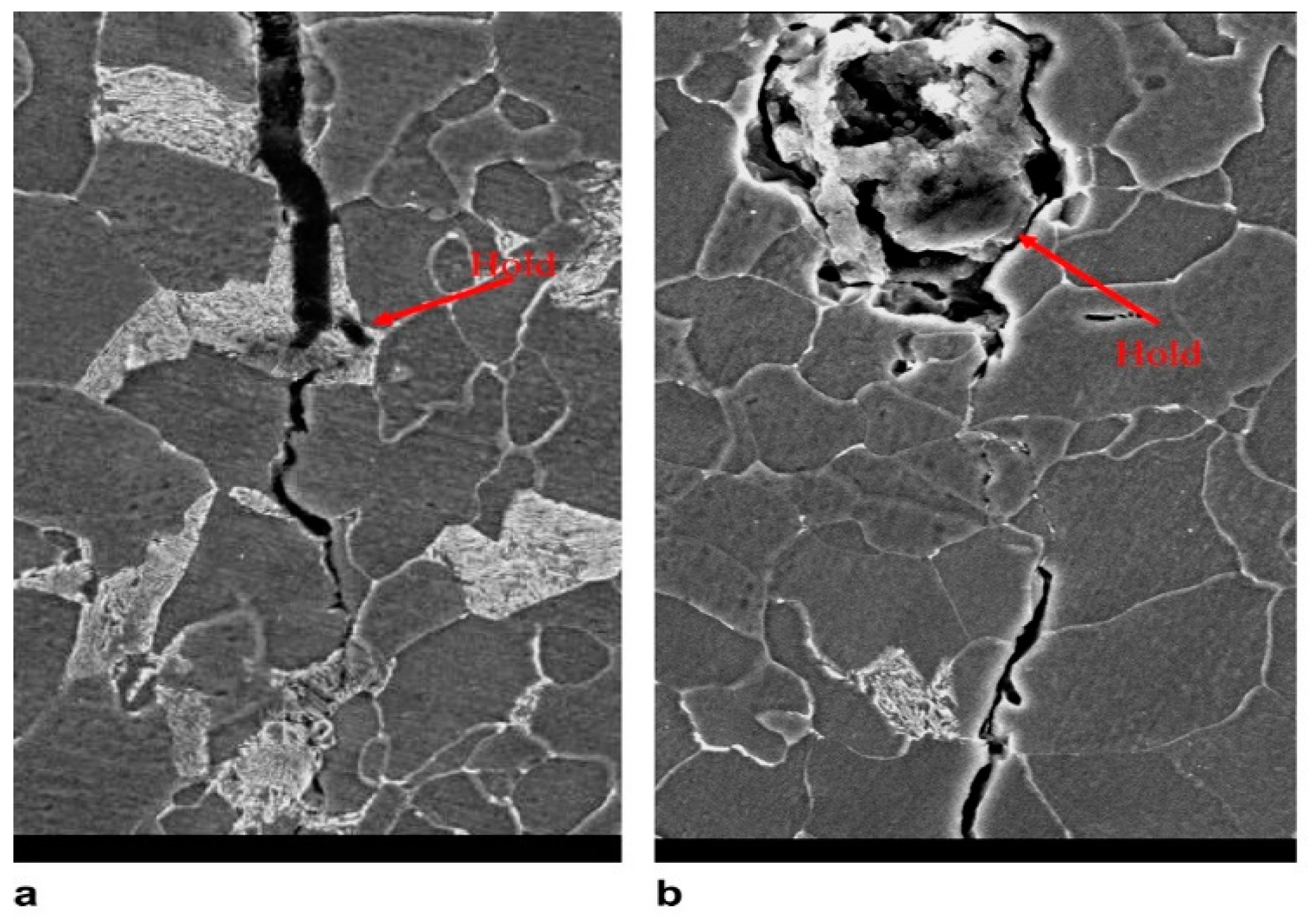

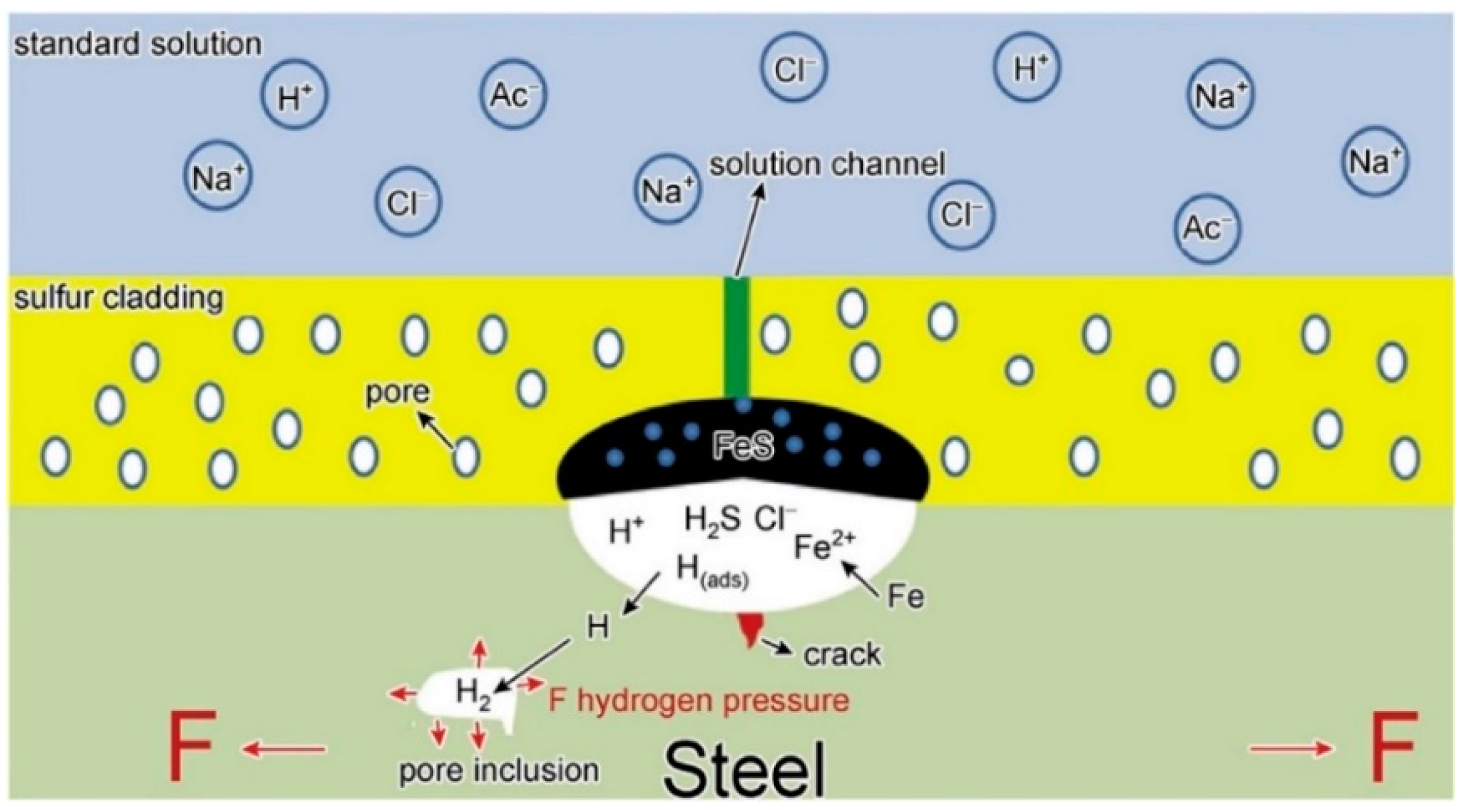

| Pipeline Steel | C | Mn | Si | Nb | Mo | Ti | Cr | Cu | Ni | V |
|---|---|---|---|---|---|---|---|---|---|---|
| X60 | 0.052 | 1.50 | 0.15 | 0.067 | 0.096 | 0.022 | 0.07 | 0.18 | 0.19 | 0.001 |
| X65 | 0.081 | 1.54 | 0.33 | 0.04 | - | 0.002 | - | 0.18 | - | 0.001 |
| X70 | 0.025 | 1.65 | 0.26 | 0.068 | 0.175 | 0.015 | 0.07 | 0.21 | 0.08 | 0.001 |
| X80 | 0.056 | 1.90 | 0.31 | 0.046 | 0.213 | 0.018 | - | 0.044 | 0.221 | - |
| L360NS | 0.12 | 1.50 | 0.25 | - | 0.02 | - | 0.07 | 0.08 | 0.04 | - |
| Pipeline Steel | Yield Strength (MPa) | Tensile Strength (MPa) | Elongation (%) |
|---|---|---|---|
| X60 | 520 | 610 | 33 |
| X65 | 568 | 650 | 32 |
| X70 | 615 | 720 | 29 |
| X80 | 640 | 780 | 25 |
| L360NS | 380 | 510 | 41 |
| NS4 solution | KCl | NaHCO3 | CaCl2·2H2O | MgSO4·7H2O |
|---|---|---|---|---|
| - | 0.122 | 0.483 | 0.181 | 0.131 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohtadi-Bonab, M.A. Effects of Different Parameters on Initiation and Propagation of Stress Corrosion Cracks in Pipeline Steels: A Review. Metals 2019, 9, 590. https://doi.org/10.3390/met9050590
Mohtadi-Bonab MA. Effects of Different Parameters on Initiation and Propagation of Stress Corrosion Cracks in Pipeline Steels: A Review. Metals. 2019; 9(5):590. https://doi.org/10.3390/met9050590
Chicago/Turabian StyleMohtadi-Bonab, M.A. 2019. "Effects of Different Parameters on Initiation and Propagation of Stress Corrosion Cracks in Pipeline Steels: A Review" Metals 9, no. 5: 590. https://doi.org/10.3390/met9050590
APA StyleMohtadi-Bonab, M. A. (2019). Effects of Different Parameters on Initiation and Propagation of Stress Corrosion Cracks in Pipeline Steels: A Review. Metals, 9(5), 590. https://doi.org/10.3390/met9050590




