Corrosion Behavior of 2205 Duplex Stainless Steels in HCl Solution Containing Sulfide
Abstract
:1. Introduction
2. Experimental Part
2.1. Material and Mass Loss Test
2.2. Electrochemical Experiments
2.3. Scanning Kelvin Probe
2.4. Morphological and Chemical Analysis
3. Results
3.1. Mass Loss Test
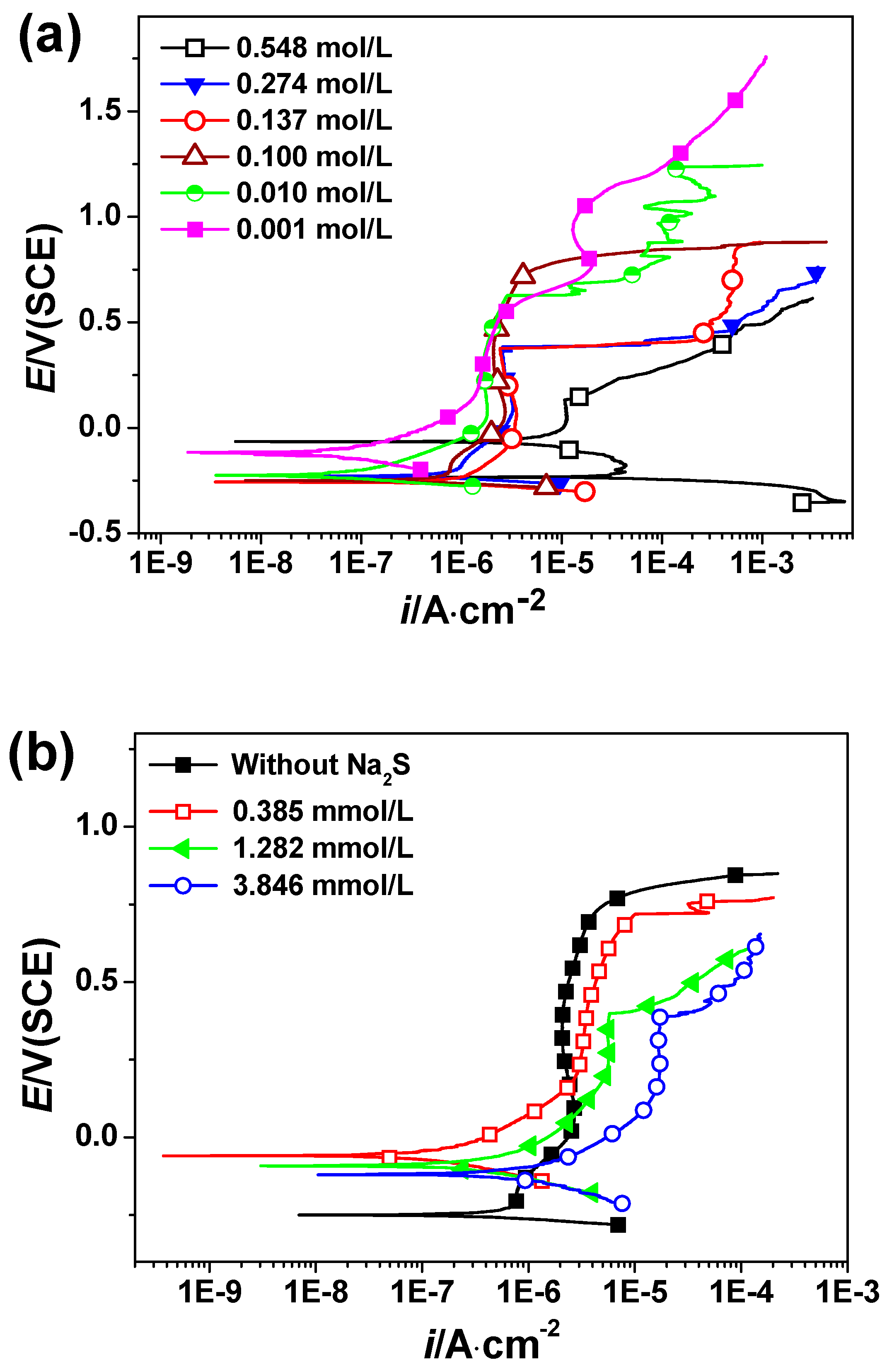
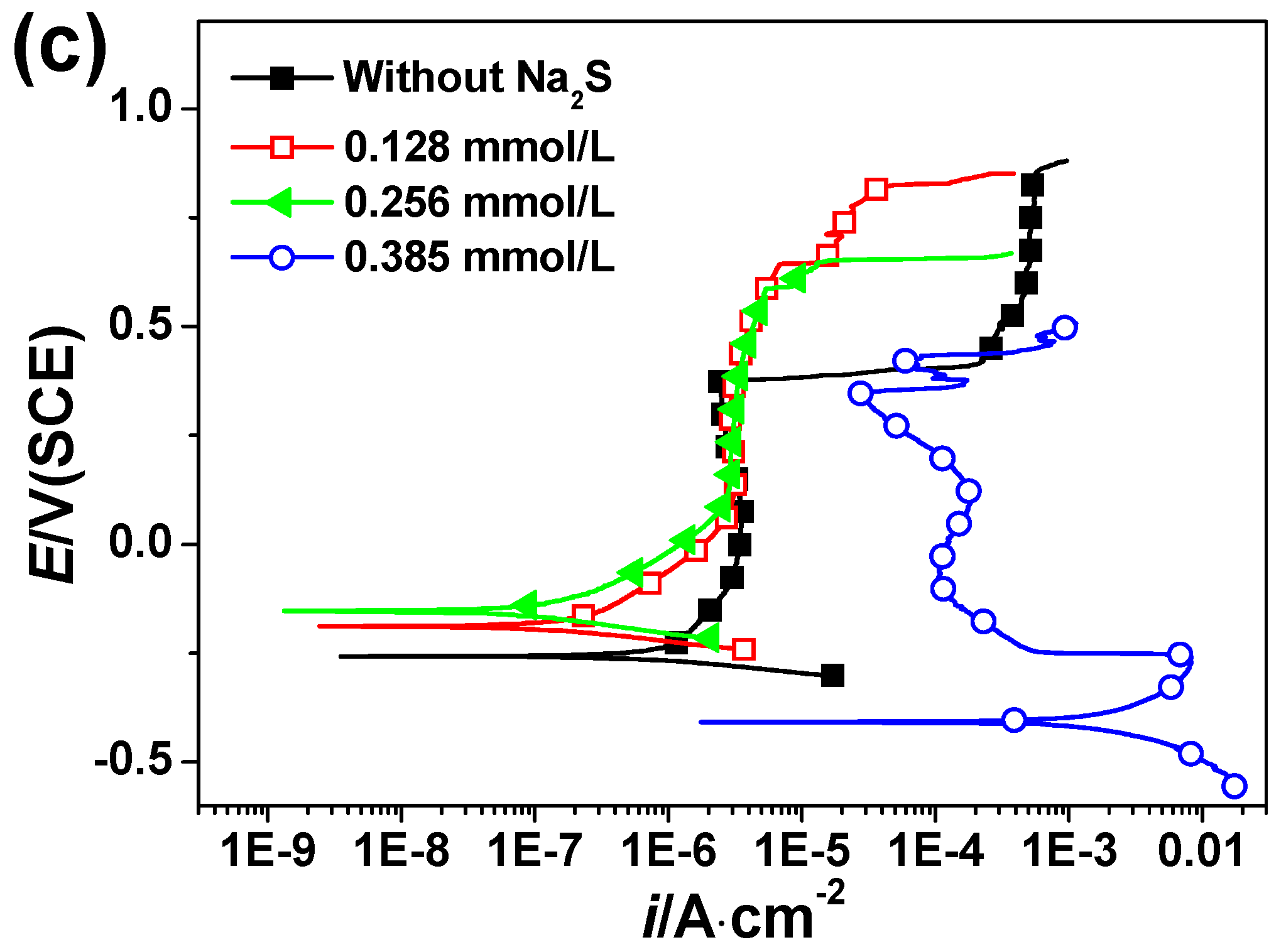
3.2. Polarization Curves Measurement
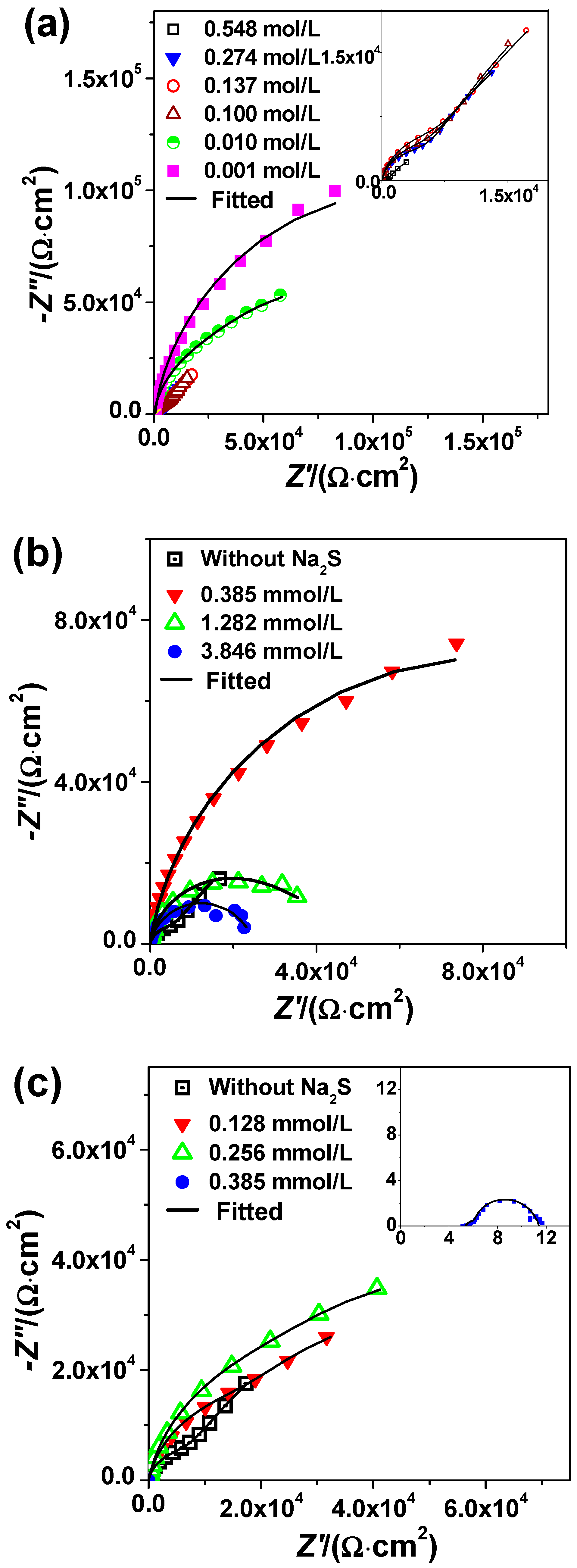
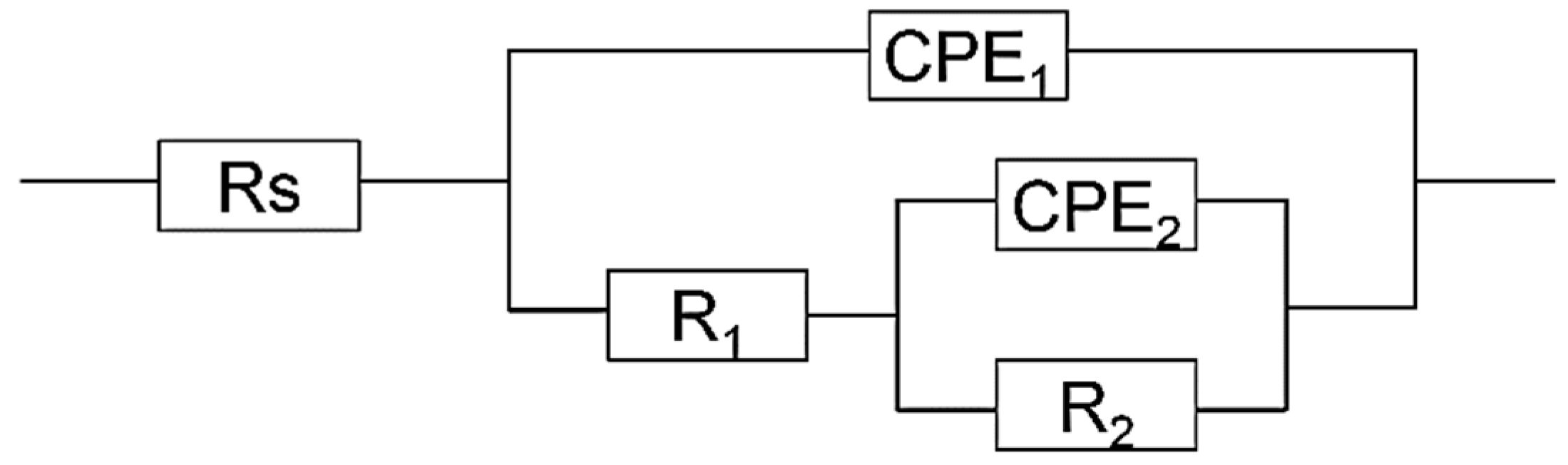
3.3. EIS Study
3.4. Scanning Kelvin Probe Mapping
3.5. Corrosion Micromorphology
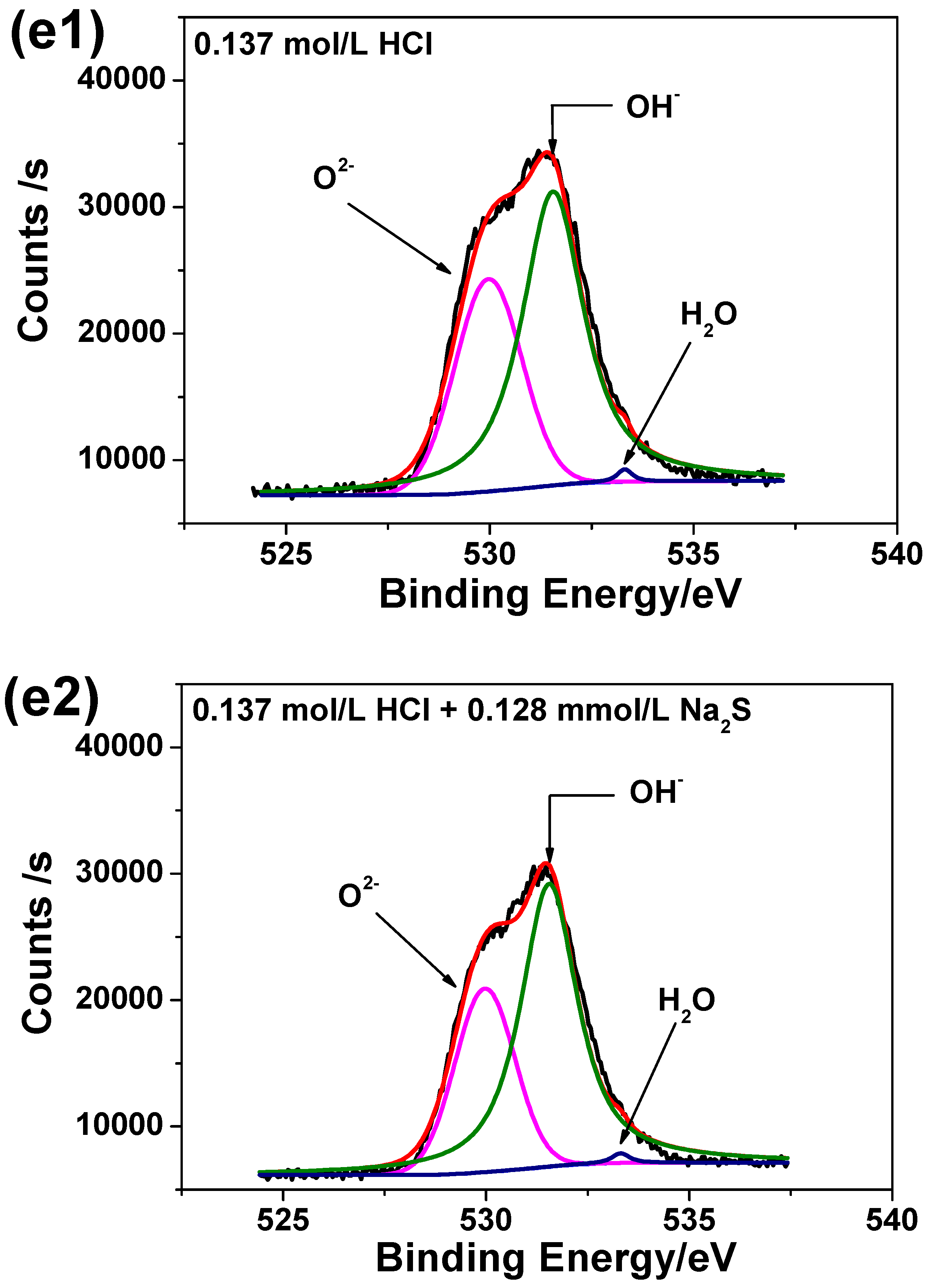
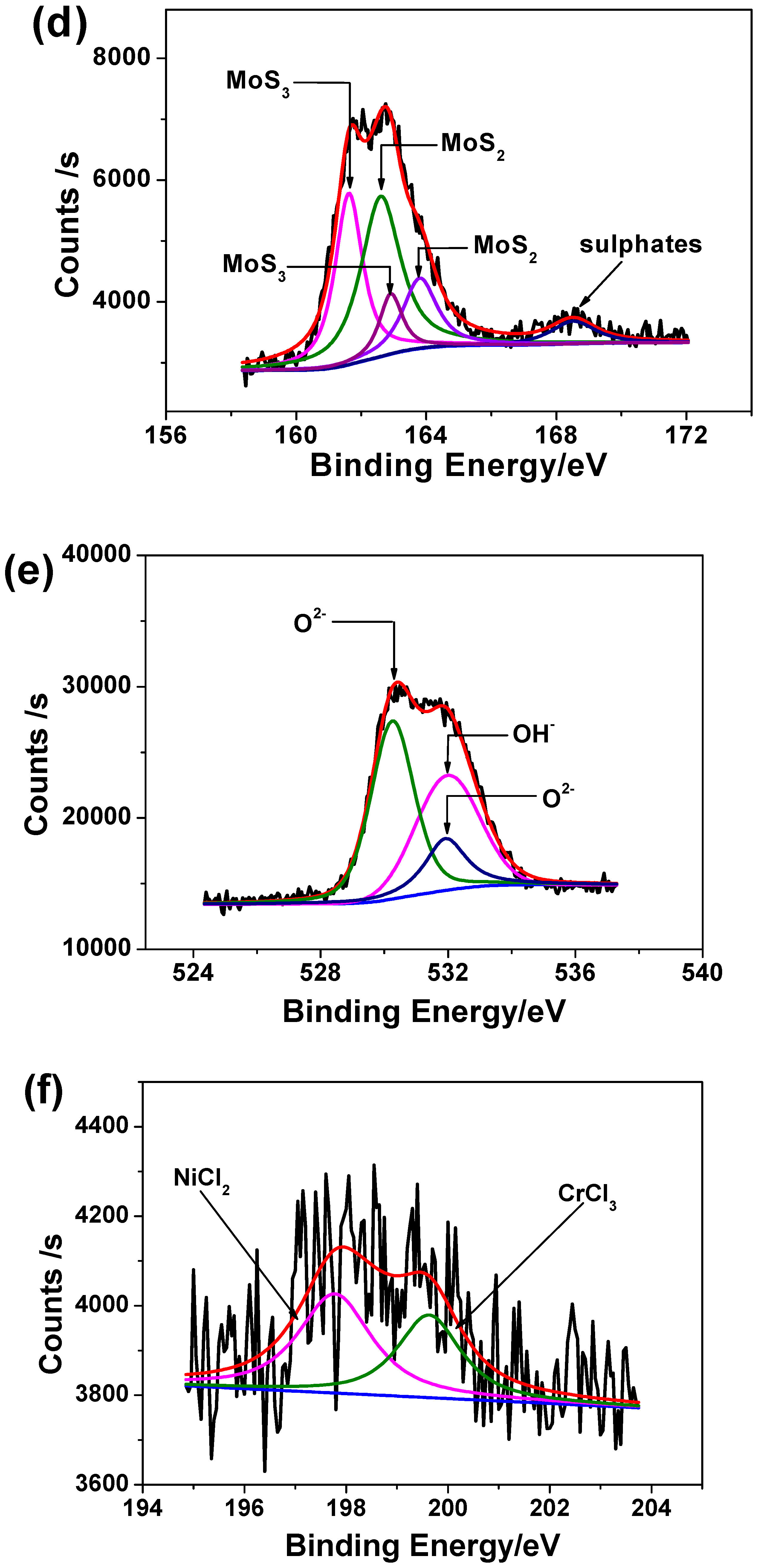
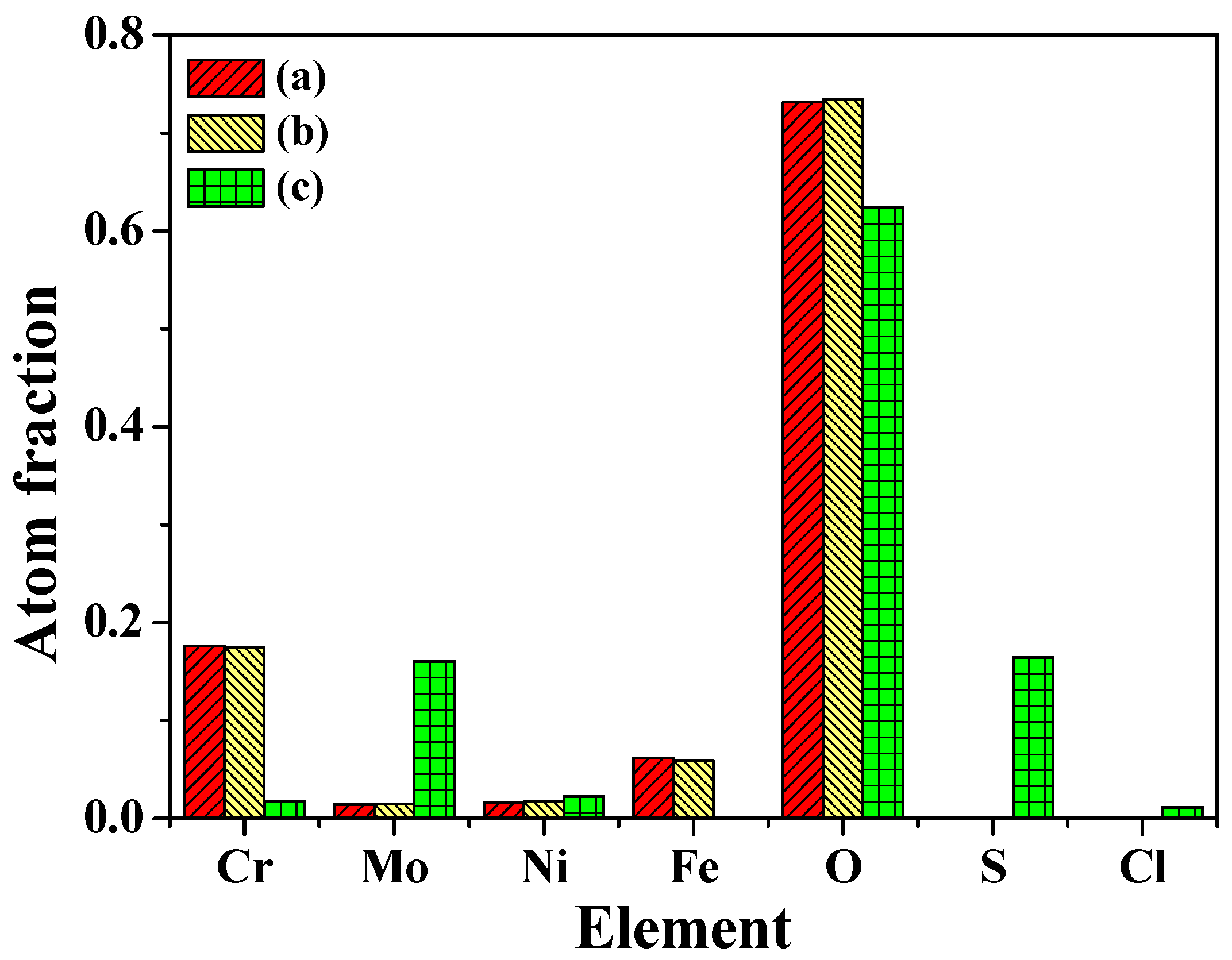
3.6. XPS Analysis
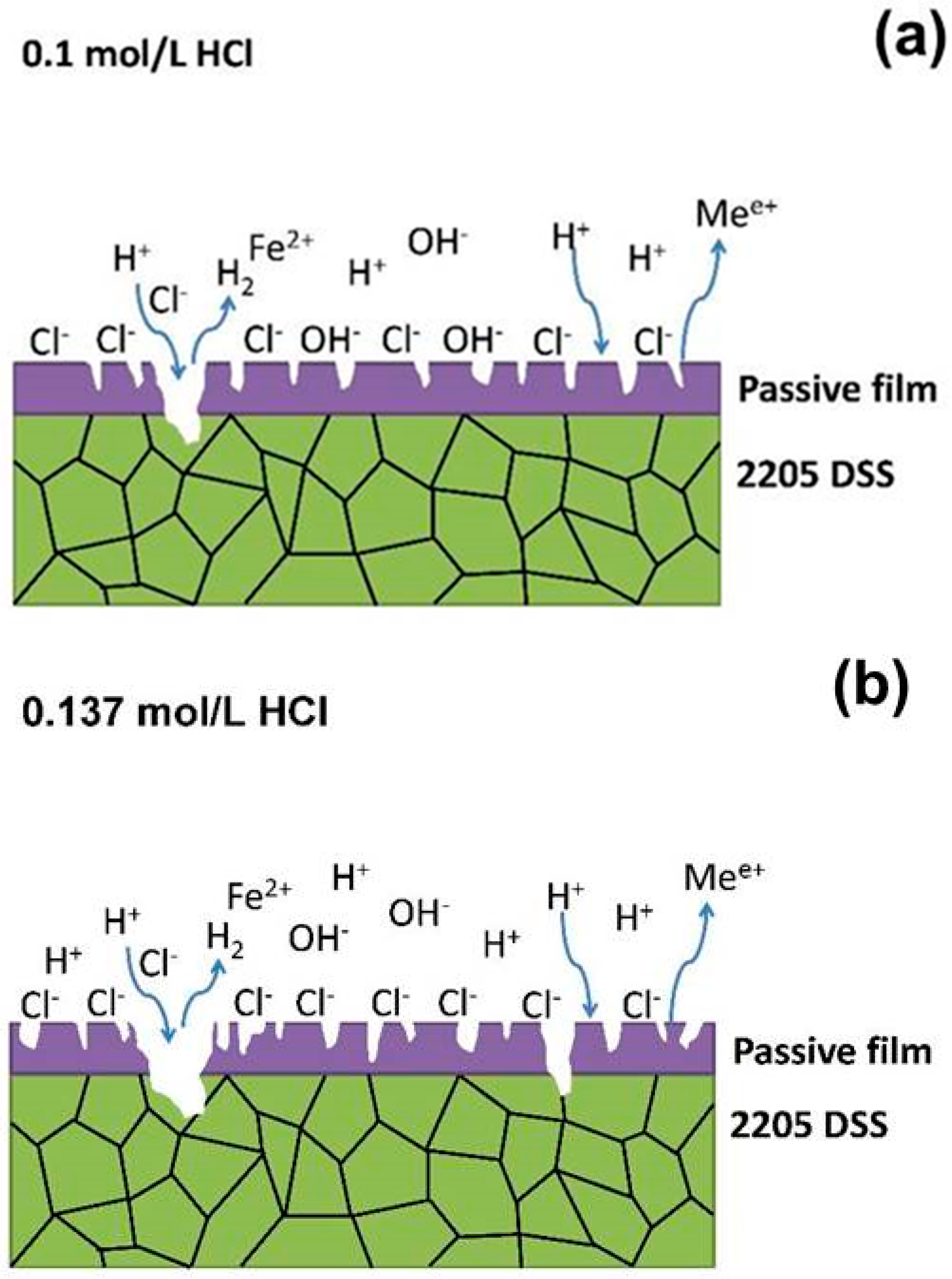
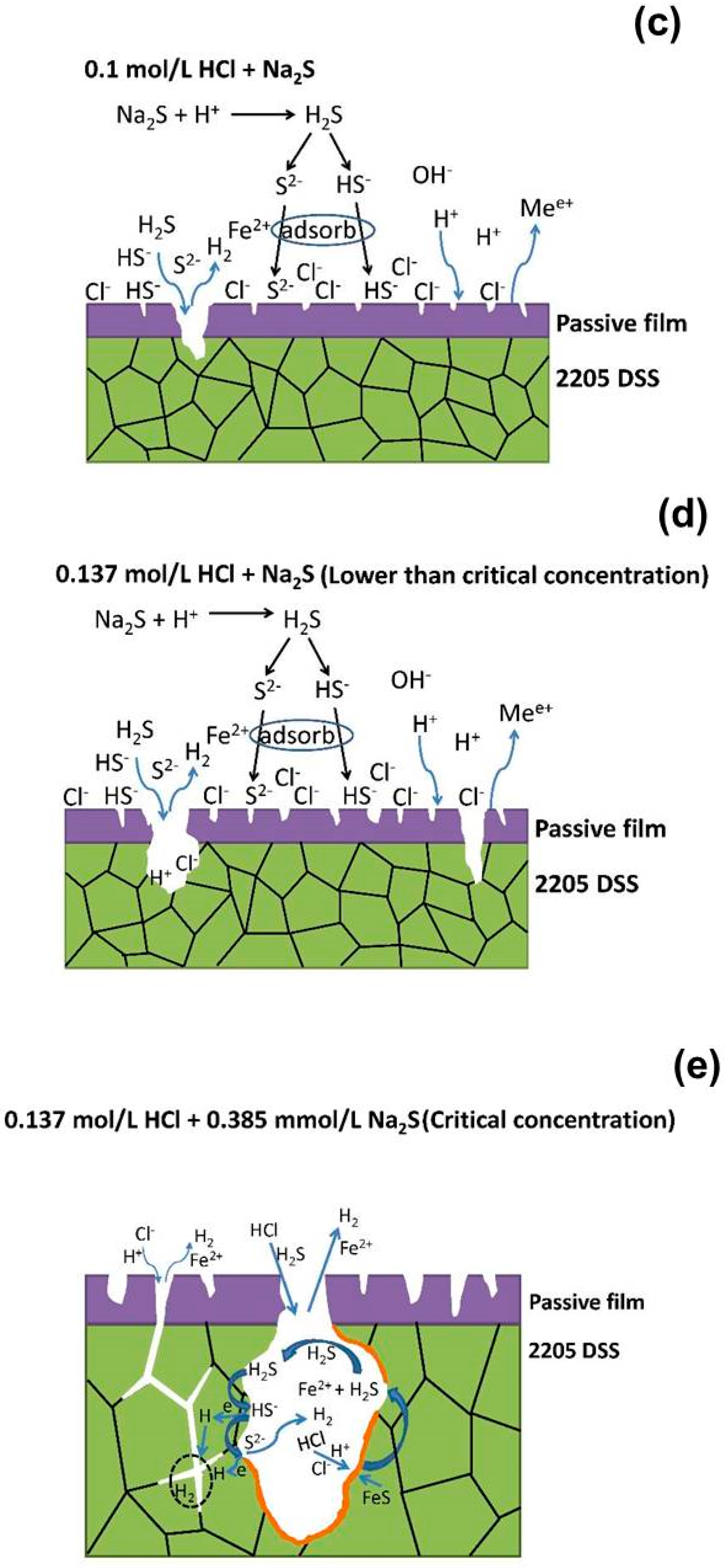
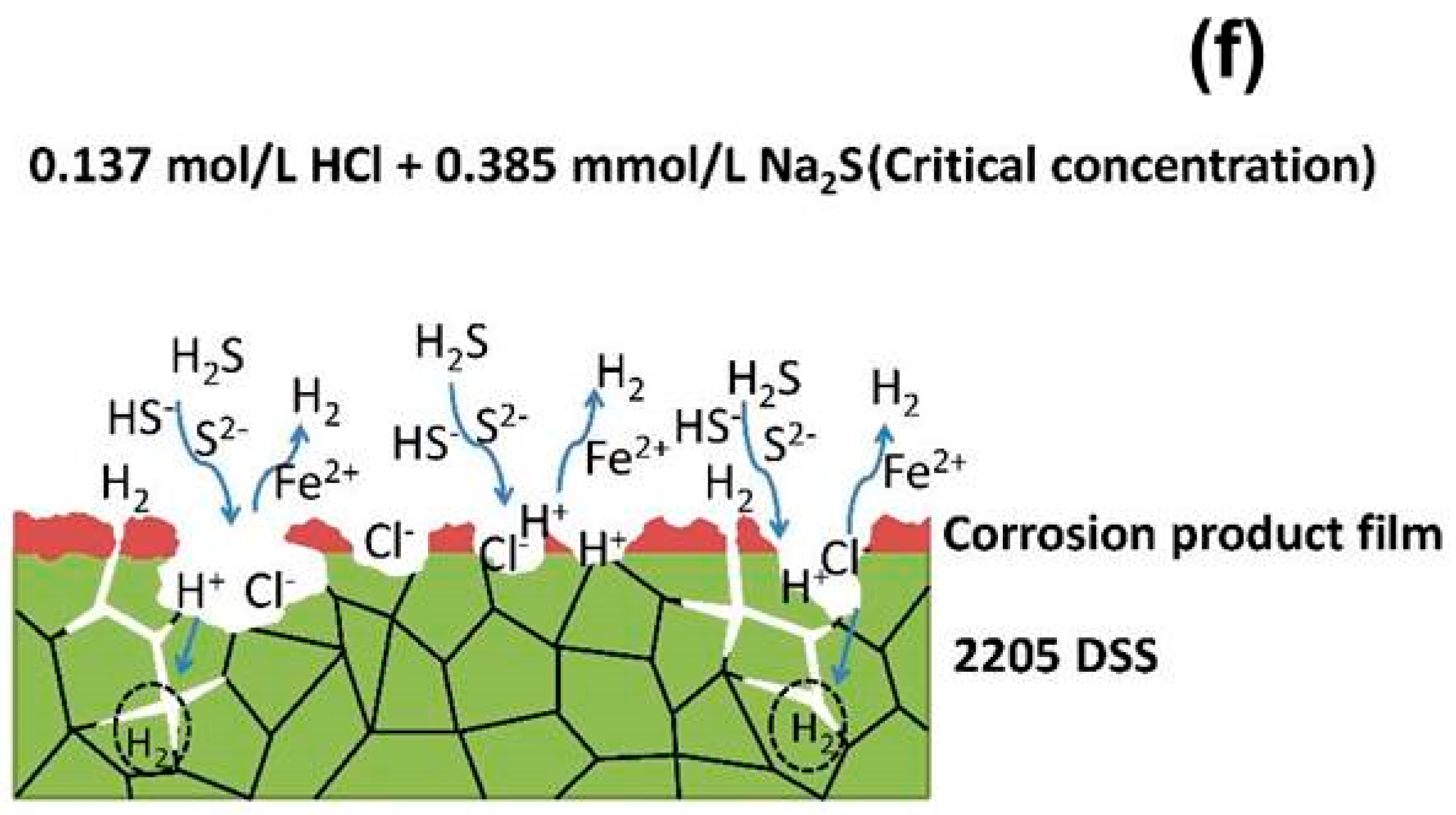
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tan, H.; Jiang, Y.; Deng, B.; Sun, T.; Xu, J.; Li, J. Effect of annealing temperature on the pitting corrosion resistance of super duplex stainless steel UNS S32750. Mater. Charact. 2009, 60, 1049–1054. [Google Scholar] [CrossRef]
- Antony, P.J.; Chongdar, S.; Kumar, P.; Raman, R. Corrosion of 2205 duplex stainless steel in chloride medium containing sulfate-reducing bacteria. Electrochim. Acta 2007, 52, 3985–3994. [Google Scholar] [CrossRef]
- Kangas, P.; Chai, G.C. Use of advanced austenitic and duplex stainless steels for applications in oil & gas and process industry. Adv. Mater. Res. 2013, 794, 645–669. [Google Scholar] [CrossRef]
- Huang, B.S.; Yin, W.F.; Sang, D.H.; Jiang, Z.Y. Applied surface science synergy effect of naphthenic acid corrosion and sulfur corrosion in crude oil distillation unit. Appl. Surf. Sci. 2012, 259, 664–670. [Google Scholar] [CrossRef]
- Cheng, Q.; Tao, B.; Song, L.; Zhang, W.; Liu, X.; Li, W.; Hou, B.; Liu, Q. Corrosion behaviour of Q235B carbon steel in sediment water from crude oil. Eval. Program Plan. 2016, 111, 61–71. [Google Scholar] [CrossRef]
- Moura, V.; Kina, A.Y.; Tavares, S.S.M.; Lima, L.D.; Mainier, F.B. Influence of stabilization heat treatments on microstructure, hardness and intergranular corrosion resistance of the AISI 321 stainless steel. J. Mater. Sci. 2008, 43, 536–540. [Google Scholar] [CrossRef]
- Vasconcelos, I.F.; Tavares, S.S.M.; Reis, F.E.U.; Abreu, H.F.G. Ageing effects on α’ precipitation and resistance to corrosion of a novel Cr-Mo stainless steel with high Mo content. J. Mater. Sci. 2009, 44, 293–299. [Google Scholar] [CrossRef]
- Moiseeva, L.S.; Aisin, A.E. Comparative tests of corrosion inhibitors in simulated media of the Samaraneftegaz oil fields. Prot. Met. 2007, 43, 84–86. [Google Scholar] [CrossRef]
- Silva, C.C.; Machado, J.P.S.E.; Sobral-Santiago, A.V.C.; de Sant’Ana, H.B.; Farias, J.P. High-temperature hydrogen sulfide corrosion on the heat-affected zone of the AISI 444 stainless steel caused by Venezuelan heavy petroleum. J. Pet. Sci. Eng. 2007, 59, 219–225. [Google Scholar] [CrossRef]
- Efremov, A.P.; Kim, S.K. Analysis of the corrosion and inhibitive protection of the Lukoil Komi oil-fields equipment. Prot. Met. 2006, 42, 194–200. [Google Scholar] [CrossRef]
- Mendoza-Canales, J.; Marín-Cruz, J. EIS characterization of corrosion processes of titanium and alloy UNS N10276 in sour environments. J. Solid State Electrochem. 2008, 12, 1637–1644. [Google Scholar] [CrossRef]
- Jeon, S.-H.; Kim, S.-T.; Lee, J.-S.; Lee, I.-S.; Park, Y.-S. Effects of sulfur addition on the formation of inclusions and the corrosion behavior of super duplex stainless steels in chloride solutions of different pH. Mater. Trans. 2012, 53, 1617–1626. [Google Scholar] [CrossRef]
- Wan, T.; Xiao, N.; Shen, H.; Yong, X. Ultrasonics Sonochemistry The effect of chloride ions on the corroded surface layer of 00Cr22Ni5Mo3N duplex stainless steel under cavitation. Ultrason. Sonochem. 2016, 33, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Chaofang, D.; Hong, L.U.O.; Kui, X.; Ting, S.U.N.; Qian, L.I.U.; Xiaogang, L.I. Effect of temperature and Cl- concentration on pitting of 2205 duplex stainless steel. J. Wuhan Univ. Technol. 2011, 26, 641–647. [Google Scholar] [CrossRef]
- Lo, I.; Fu, Y.; Lin, C.; Tsai, W. Effect of electrolyte composition on the active-to-passive transition behavior of 2205 duplex stainless steel in H2SO4/HCl solutions. Corros. Sci. 2006, 48, 696–708. [Google Scholar] [CrossRef]
- Liu, Z.Y.; Dong, C.F.; Li, X.G.; Zhi, Q.; Cheng, Y.F. Stress corrosion cracking of 2205 duplex stainless steel in H2S-CO2 environment. J. Mater. Sci. 2009, 44, 4228–4234. [Google Scholar] [CrossRef]
- Liu, R.; Li, J.; Liu, Z.; Du, C.; Dong, C.; Li, X. Effect of pH and H2S concentration on sulfide stress corrosion cracking (SSCC) of API 2205 duplex stainless steel. Int. J. Mater. Res. 2015, 106, 608–613. [Google Scholar] [CrossRef]
- Tranchida, G.; Clesi, M.; di Franco, F.; di Quarto, F.; Santamaria, M. Electronic properties and corrosion resistance of passive films on austenitic and duplex stainless steels. Electrochim. Acta 2018, 273, 412–423. [Google Scholar] [CrossRef]
- Mohammadi, M.; Choudhary, L.; Gadala, I.M.; Alfantazia, A. Electrochemical and Passive Layer Characterizations of 304L, 316L, and Duplex 2205 Stainless Steels in Thiosulfate Gold Leaching Solutions. J. Electrochem. Soc. 2016, 163, C883–C894. [Google Scholar] [CrossRef]
- He, W.; Knudsen, O.Ø.; Diplas, S. Corrosion of stainless steel 316L in simulated formation water environment with CO2-H2S-Cl-. Corros. Sci. 2009, 51, 2811–2819. [Google Scholar] [CrossRef]
- Veloz, M.A.; Gonza, I. Electrochemical study of carbon steel corrosion in buffered acetic acid solutions with chlorides and H2S. Electrochim. Acta 2002, 48, 135–144. [Google Scholar] [CrossRef]
- Ding, J.; Zhang, L.; Lu, M.; Wang, J.; Wen, Z.; Hao, W. Applied surface science the electrochemical behaviour of 316L austenitic stainless steel in Cl− containing environment under different H2S partial pressures. Appl. Surf. Sci. 2014, 289, 33–41. [Google Scholar] [CrossRef]
- Case, R.P.; Rincon, H.E.; McIntyre, D.R. Analysis of pit stability in type 316L stainless steel exposed to H2S-saturated dilute chloride solutions above the critical pitting temperature. Corrosion 2012, 68, 1–12. [Google Scholar] [CrossRef]
- Tang, J.; Hu, Y.; Han, Z.; Wang, H.; Zhu, Y.; Wang, Y.; Nie, Z.; Wang, Y. Experimental and Theoretical Study on the Synergistic Inhibition Effect of Pyridine Derivatives and Sulfur-Containing Compounds on the Corrosion of Carbon Steel in CO2-Saturated 3.5 wt.% NaCl Solution. Molecules 2018, 23, 3270. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Tang, J.; Wang, H.; Wang, Y.; Qiao, J.; Apreutesei, M. Bernard Normand, Corrosion behavior of bulk (Zr58Nb3Cu16Ni13Al10)100−xYx (x = 0, 0.5, 2.5 at.%) metallic glasses in sulfuric acid. Corros. Sci. 2019. [Google Scholar] [CrossRef]
- Marcus, P. Surface science approach of corrosion phenomena. Electrochim. Acta 1998, 43, 109–118. [Google Scholar] [CrossRef]
- Marcus, P.; Maurice, V.; Strehblow, H.H. Localized corrosion (pitting): A model of passivity breakdown including the role of the oxide layer nanostructure. Corros. Sci. 2008, 50, 2698–2704. [Google Scholar] [CrossRef]
- Fredriksson, W.; Petrini, D.; Edström, K.; Björefors, F.; Nyholm, L. Corrosion resistances and passivation of powder metallurgical and conventionally cast 316L and 2205 stainless steels. Corros. Sci. 2013, 67, 268–280. [Google Scholar] [CrossRef]
- Zhang, G.A.; Zeng, Y.; Guo, X.P.; Jiang, F.; Shi, D.Y.; Chen, Z.Y. Electrochemical corrosion behavior of carbon steel under dynamic high pressure H2S/CO2 environment. Corros. Sci. 2012, 65, 37–47. [Google Scholar] [CrossRef]
- Herbsleb, G.; Poepperling, R.K. Corrosion properties of austenitic-ferritic duplex steel AF 22 in chloride and sulfide containing environments. Corrosion 1980, 36, 611–618. [Google Scholar] [CrossRef]
- Bhola, S.M.; Bhola, R.; Mishra, B.; Olson, D.L. Povidone-iodine as a corrosion inhibitor towards a low modulus beta Ti-45Nb implant alloy in a simulated body fluid. J. Mater. Sci. Mater. Med. 2011, 22, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Bhola, S.M.; Bhola, R.; Mishra, B.; Olson, D.L. Electrochemical impedance spectroscopic characterization of the oxide film formed over low modulus Ti-35.5Nb-7.3Zr-5.7Ta alloy in phosphate buffer saline at various potentials. J. Mater. Sci. 2010, 45, 6179–6186. [Google Scholar] [CrossRef]
- Qiao, Y.X.; Zheng, Y.G.; Ke, W.; Okafor, P.C. Electrochemical behaviour of high nitrogen stainless steel in acidic solutions. Corros. Sci. 2009, 51, 979–986. [Google Scholar] [CrossRef]
- Ma, H.; Cheng, X.; Li, G.; Chen, S.; Quan, Z.; Zhao, S.; Niu, L. The influence of hydrogen sulfide on corrosion of iron under different conditions. Corros. Sci. 2000, 42, 1669–1683. [Google Scholar] [CrossRef]
- Azevedo, C.; Bezerra, P.S.A.; Esteves, F.; Joia, C.J.B.M.; Mattos, O.R. Hydrogen permeation studied by electrochemical techniques. Electrochim. Acta 1999, 44, 4431–4442. [Google Scholar] [CrossRef]
- Ha, H.Y.; Jang, M.H.; Lee, T.H.; Moon, J. Understanding the Relation between Phase Fraction and Pitting Corrosion Resistance of UNS 32750 Stainless Steel; Elsevier B.V.: Amsterdam, The Netherlands, 2015. [Google Scholar] [CrossRef]
- Kittel, J.; Smanio, V.; Fregonese, M.; Garnier, L.; Lefebvre, X. Hydrogen induced cracking (HIC) testing of low alloy steel in sour environment: Impact of time of exposure on the extent of damage. Corros. Sci. 2010, 52, 1386–1392. [Google Scholar] [CrossRef]
- Sobol, O.; Holzlechner, G.; Nolze, G.; Wirth, T.; Eliezer, D.; Boellinghaus, T.; Wolfgang, E.S. Unger Time-of-Flight Secondary Ion Mass Spectrometry (ToF-SIMS) imagingof deuterium assisted cracking in a 2205 duplex stainless steel microstructure. Mater. Sci. Eng. A 2016, 676, 271–277. [Google Scholar] [CrossRef]
- Bellezze, T.; Giuliani, G.; Viceré, A.; Roventi, G. Study of stainless steels corrosion in a strong acid mixture. Part 2: Anodic selective dissolution, weight loss and electrochemical impedance spectroscopy tests. Corros. Sci. 2018, 130, 12–21. [Google Scholar] [CrossRef]
- Femenia, M.; Pan, J.; Leygraf, C.; Luukkonen, P. In situ study of selective dissolution of duplex stainless steel 2205 by electrochemical scanning tunnelling microscopy. Corros. Sci. 2001, 43, 1939–1951. [Google Scholar] [CrossRef]
- Luo, H.; Dong, C.F.; Xiao, K.; Li, X.G. Characterization of passive film on 2205 duplex stainless steel in sodium thiosulphate solution. Appl. Surf. Sci. 2011, 258, 631–639. [Google Scholar] [CrossRef]
- Liu, C.T.; Wu, J.K. Influence of pH on the passivation behavior of 254SMO stainless steel in 3.5% NaCl solution. Corros. Sci. 2007, 49, 2198–2209. [Google Scholar] [CrossRef]
- Sun, W.; Nesic, S. A mechanistic model of H2S corrosion of mild steel. In Proceedings of the NACE Proceedings, Nashville, TN, USA, 11–15 March 2007. [Google Scholar]
- Tang, J.; Shao, Y.; Guo, J.; Zhang, T.; Meng, G.; Wang, F. The effect of H2S concentration on the corrosion behavior of carbon steel at 90 °C. Corros. Sci. 2010, 52, 2050–2058. [Google Scholar] [CrossRef]
- Zimer, A.M.; Rios, E.C.; Mendes, P.D.; Gonçalves, W.N.; Bruno, O.M.; Pereira, E.C.; Mascaro, L.H. Investigation of AISI 1040 steel corrosion in H2S solution containing chloride ions by digital image processing coupled with electrochemical techniques. Corros. Sci. 2011, 53, 3193–3201. [Google Scholar] [CrossRef]
- Shoesmith, D.W.; Bailey, M.G.; Ikeda, B. Electrochemical formation of mackinawite in alkaline sulphide solutions. Electrochim. Acta 1978, 23, 1329–1339. [Google Scholar] [CrossRef]
- Tiwari, G.P.; Bose, A.; Chakravartty, J.K.; Wadekar, S.L.; Totlani, M.K.; Arya, R.N.; Fotedar, R.K. A study of internal hydrogen embrittlement of steels. Mater. Sci. Eng. A 2000, 286, 269–281. [Google Scholar] [CrossRef]
- González, J.; Gutiérrez-Solana, F.; Varona, J.M. The effects of microstructure, strength level, and crack propagation mode on stress corrosion cracking behavior of 4135 steel. Metall. Mater. Trans. A 1996, 27, 281–290. [Google Scholar] [CrossRef]
- Sozańska, M.; Kłyk-Spyra, K. Investigation of hydrogen induced cracking in 2205 duplex stainless steel in wet H2S environments after isothermal treatment at 675, 750 and 900 °C. Mater. Charact. 2006, 56, 399–404. [Google Scholar] [CrossRef]
- Sobol, O.; Nolze, G.; Saliwan-Neumann, R.; Eliezer, D.; Boellinghaus, T.; Unger, W.E. Novel approach to image hydrogen distribution and related phase transformation in duplex stainless steels at the sub-micron scale. Int. J. Hydrogen Energy 2017, 42, 25114–25120. [Google Scholar] [CrossRef]
- Mancia, F. The effect of environmental modification on the sulphide stress corrosion cracking resistance of 13Cr martensitic stainless steel in H2S-CO2-Cl-systems. Corros. Sci. 1987, 27, 1225–1237. [Google Scholar] [CrossRef]




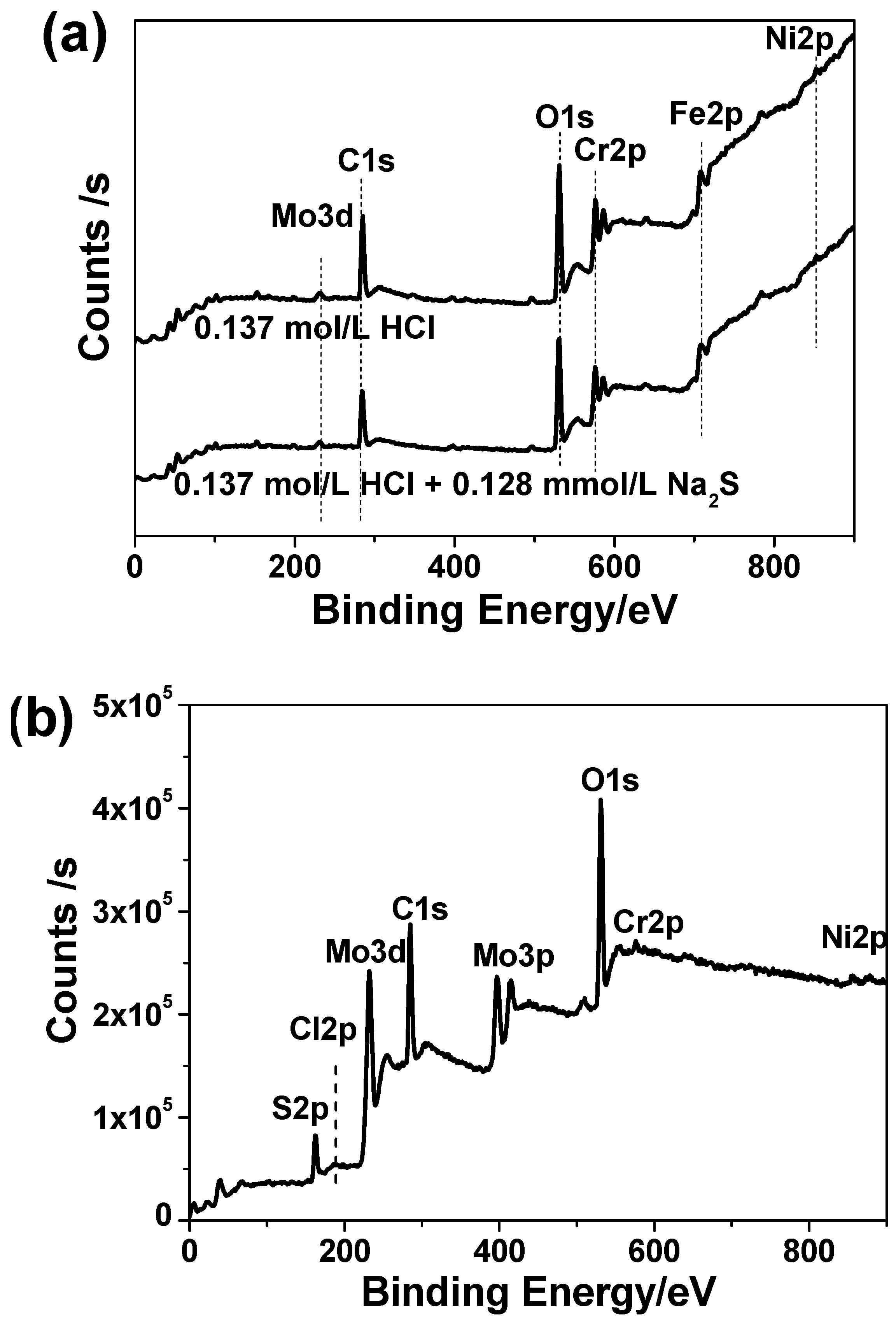
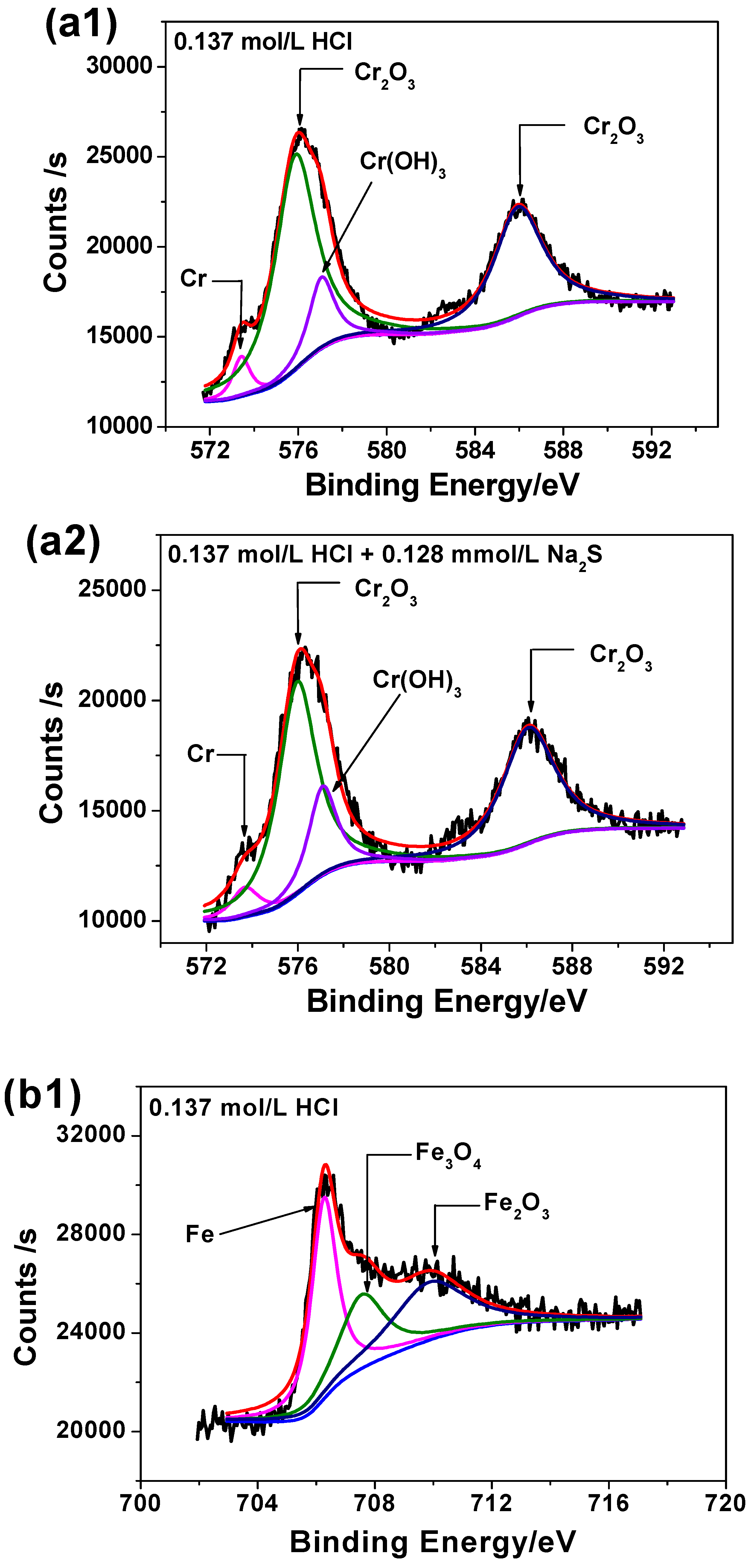
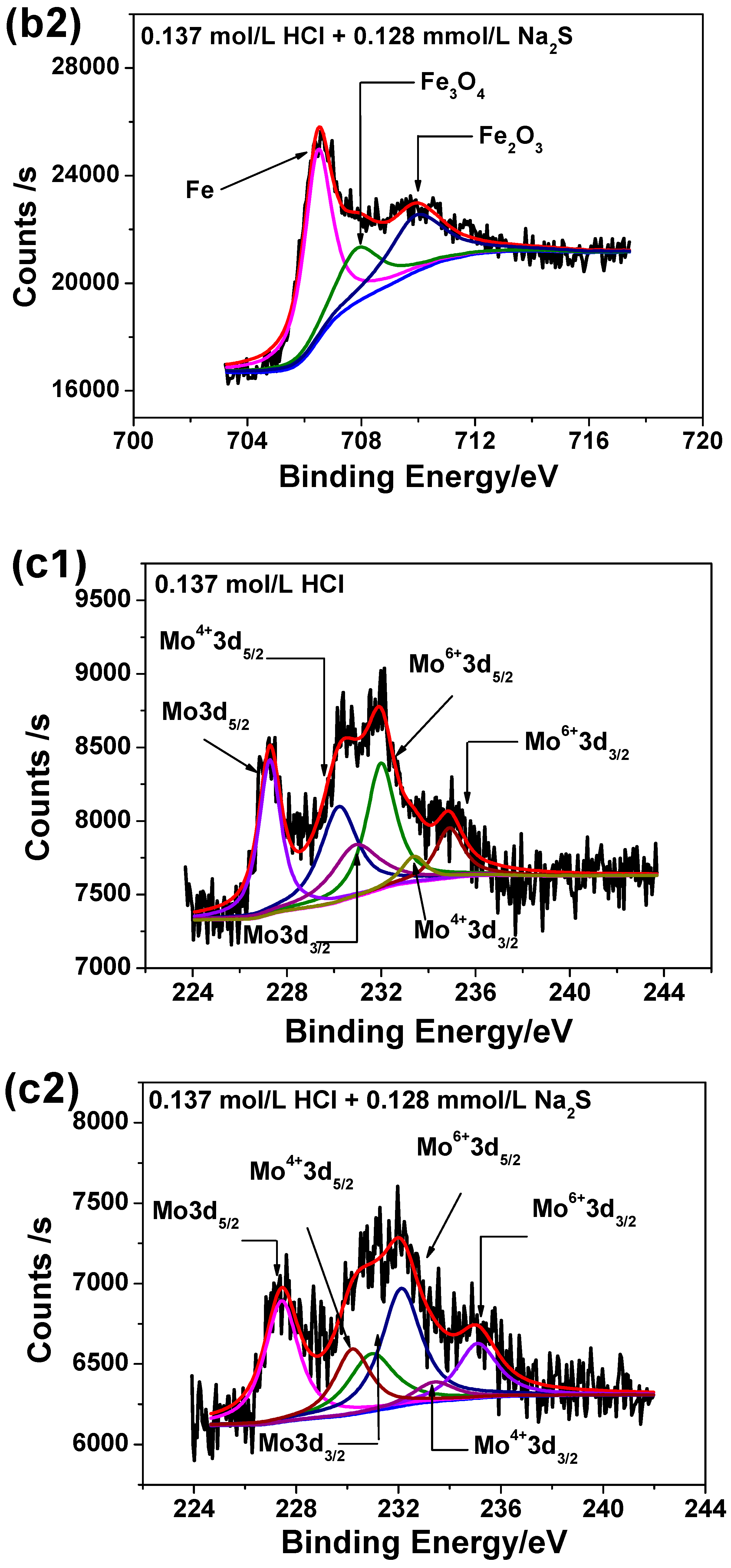
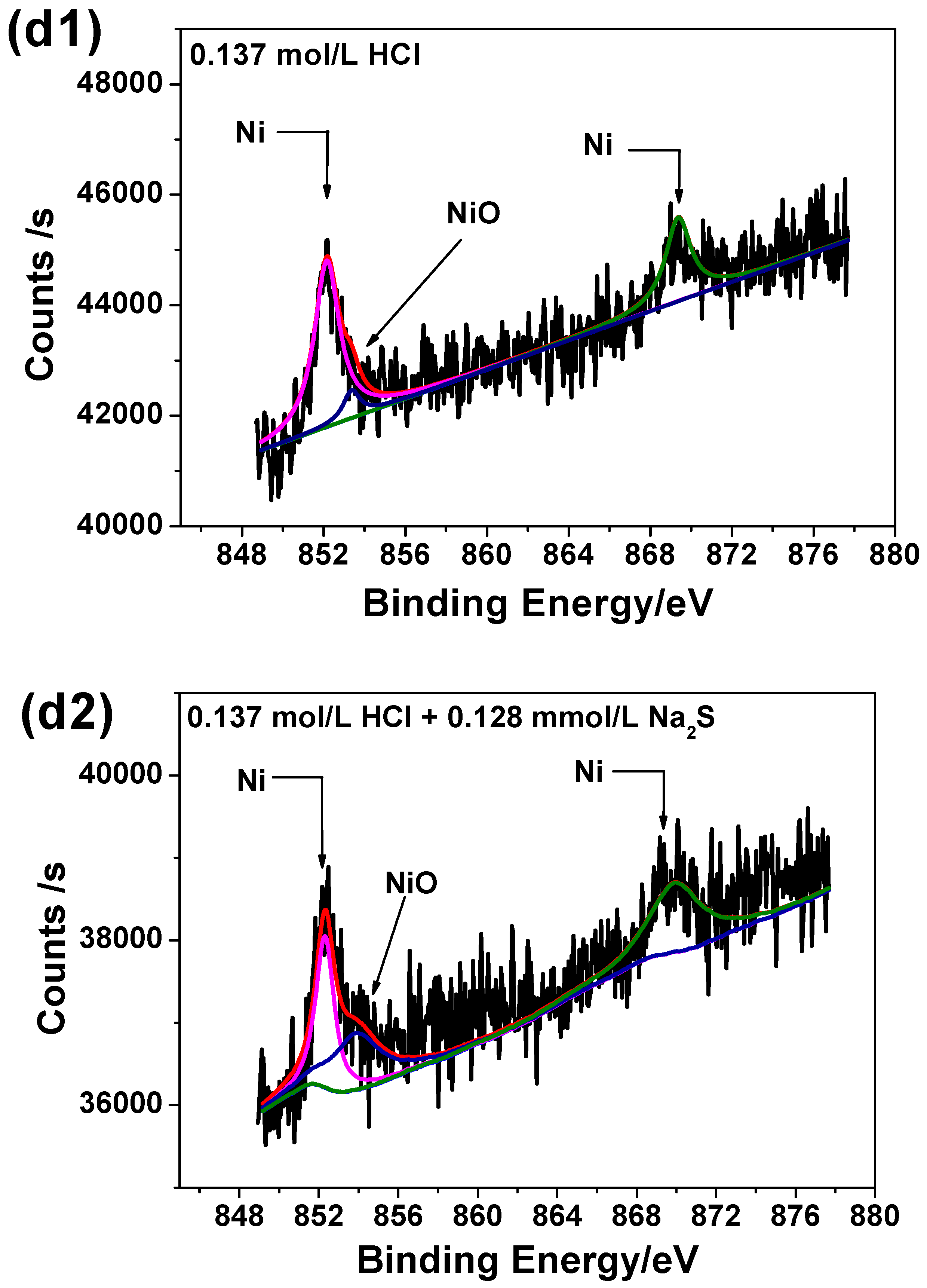


| CHCl (mol/L) | Corrosion Rate without Na2S (mm/a) | Χ12 | Corrosion Rate with 0.385 mmol/L Na2S (mm/a) | Χ22 | Growth Ratio (%) |
|---|---|---|---|---|---|
| 0.001 | 0.0095 | 5.4 × 10−2 | 0.0091 | 7.6 × 10−2 | −4 |
| 0.01 | 0.0097 | 6.9 × 10−2 | 0.0093 | 6.5 × 10−2 | −4 |
| 0.1 | 0.0113 | 7.3 × 10−2 | 0.0111 | 6.7 × 10−2 | −2 |
| 0.137 | 0.0114 | 6.5 × 10−2 | 12.364 | 2.2 × 10−2 | 108,549 |
| 0.274 | 0.0130 | 8.1 × 10−2 | 24.506 | 7.0 × 10−2 | 188,551 |
| 0.548 | 52.734 | 7.4 × 10−2 | 81.252 | 9.8 × 10−3 | 54 |
| HCl (mol/L) | Na2S (mmol/L) | Rs (Ω·cm2) | CPE1 (μΩ−1·cm−2·s) | n1 | R1 (KΩ·cm2) | CPE2 (μΩ−1·cm−2·sn) | n2 | R2 (KΩ·cm2) | Χ2 |
|---|---|---|---|---|---|---|---|---|---|
| 0.100 | 0 | 9.67 | 71.71 | 0.90 | 11.04 | 278.70 | 0.80 | 109.70 | 2.5 × 10−4 |
| 0.385 | 7.94 | 62.81 | 0.90 | 13.12 | 4.43 | 0.80 | 155.90 | 2.7 × 10−4 | |
| 1.282 | 7.35 | 60.21 | 0.90 | 36.78 | 1103.00 | 0.80 | 10.60 | 3.8 × 10−4 | |
| 3.846 | 6.97 | 66.58 | 0.88 | 22.86 | 3746.00 | 0.80 | 1.50 | 2.3 × 10−4 | |
| 0.137 | 0 | 5.99 | 75.61 | 0.89 | 10.79 | 293.3 | 0.80 | 66.74 | 1.6 × 10−4 |
| 0.128 | 5.82 | 78.89 | 0.87 | 33.38 | 239.1 | 0.80 | 67.48 | 5.0 × 10−4 | |
| 0.256 | 5.54 | 80.16 | 0.87 | 53.51 | 203.2 | 0.80 | 61.65 | 1.4 × 10−4 | |
| 0.385 | 5.41 | 5545.00 | 0.80 | 0.0012 | 9038.00 | 0.97 | 0.0048 | 3.3 × 10−4 |
| Samples (2205 DSS) | ΔESKP (mV) |
|---|---|
| Before immersion | 146 |
| After immersion | 160 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, J.; Yang, X.; Wang, Y.; Wang, H.; Xiao, Y.; Apreutesei, M.; Nie, Z.; Normand, B. Corrosion Behavior of 2205 Duplex Stainless Steels in HCl Solution Containing Sulfide. Metals 2019, 9, 294. https://doi.org/10.3390/met9030294
Tang J, Yang X, Wang Y, Wang H, Xiao Y, Apreutesei M, Nie Z, Normand B. Corrosion Behavior of 2205 Duplex Stainless Steels in HCl Solution Containing Sulfide. Metals. 2019; 9(3):294. https://doi.org/10.3390/met9030294
Chicago/Turabian StyleTang, Junlei, Xi Yang, Yingying Wang, Hu Wang, Ying Xiao, Mihai Apreutesei, Zhen Nie, and Bernard Normand. 2019. "Corrosion Behavior of 2205 Duplex Stainless Steels in HCl Solution Containing Sulfide" Metals 9, no. 3: 294. https://doi.org/10.3390/met9030294
APA StyleTang, J., Yang, X., Wang, Y., Wang, H., Xiao, Y., Apreutesei, M., Nie, Z., & Normand, B. (2019). Corrosion Behavior of 2205 Duplex Stainless Steels in HCl Solution Containing Sulfide. Metals, 9(3), 294. https://doi.org/10.3390/met9030294







