Removal of Mg and MgO By-Products through Magnesiothermic Reduction of Ti Powder in Self-Propagating High-Temperature Synthesis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Won, C.W.; Nersisyan, H.H.; Won, H.I. Titanium powder prepared by a rapid exothermic reaction. Chem. Eng. J. 2010, 157, 270–275. [Google Scholar] [CrossRef]
- Nersisyan, H.H.; Won, H.I.; Won, C.W.; Jo, A.; Kim, J.H. Direct magnesiothermic reduction of titanium dioxide to titanium powder through combustion synthesis. Chem. Eng. J. 2014, 235, 67–74. [Google Scholar] [CrossRef]
- Zheng, H.; Okabe, T.H. Selective chlorination of titanium ore and production of titanium powder by preform reduction process (PRP). In Proceedings of the 16th Iketani Conference, Tokyo, Japan, 12–16 November 2006. [Google Scholar]
- Zheng, H.; Okabe, T.H. Production of titanium powder directly from titanium ore by preform reduction process (PRP). In Proceedings of the 5th UT2 Graduate Student Workshop (University of Tokyo-University of Toronto), Tokyo, Japan, 7 June 2006. [Google Scholar]
- Chen, G.Z.; Fray, D.J.; Farthing, T.W. Direct electrochemical reduction of titanium dioxide to titanium in molten calcium chloride. Nature 2000, 47, 361–363. [Google Scholar] [CrossRef]
- Suzuki, R.O.; Ono, K.; Teranuma, K. Calciothermic reduction of titanium oxide and in-situ electrolysis in molten CaCl2. Metall. Mater. Trans. B 2003, 34, 287–295. [Google Scholar] [CrossRef]
- Okabe, T.H.; Oda, T.; Mitsuda, Y. Titanium powder production by preform reduction process (PRP). J. Alloys Compd. 2004, 364, 156–163. [Google Scholar] [CrossRef]
- Chen, W.; Yamamoto, Y.; Peter, W.H.; Gorti, S.B.; Sabau, A.S.; Clark, M.B.; Nunn, S.D.; Kiggans, J.O.; Blue, C.A.; Williams, J.C.; et al. Cold compaction study of Armstrong process Ti-6Al-4V powders. Powder Technol. 2011, 214, 194–199. [Google Scholar] [CrossRef]
- Chen, W.; Yamamoto, Y.; Peter, W.H.; Clark, M.B.; Nunn, S.D.; Kiggans, J.O.; Muth, T.R.; Blue, C.A.; Williams, J.C.; Akhtar, K. The investigation of die-pressing and sintering behavior of ITP CP-Ti and Ti-6Al-4V powders. J. Alloys Compd. 2012, 541, 440–447. [Google Scholar] [CrossRef]
- Xia, Y.; Fang, Z.Z.; Zhang, Y.; Lefler, H.; Zhang, T.; Sun, P.; Huang, Z. Hydrogen assisted magnesiothermic reduction (HAMR) of commercial TiO2 to produce titanium powder with controlled morphology and particle size. Mater. Trans. 2017, 58, 355–360. [Google Scholar] [CrossRef]
- Kapias, T.; Griffiths, R.F. Accidental releases of titanium tetrachloride (TiCl4) in the context of major hazards-spill behavior using REACTPOOL. J. Hazard. Mater. 2005, A119, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Okabe, T.H.; Hamanaka, Y.; Taninouchi, Y. Direct oxygen removal technique for recycling titanium using molten MgCl2 salt. Faraday Discuss. 2016, 190, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Ali, B.; Hyun, S.K.; Sim, J.J.; Choi, W.J.; Joo, W.; Lim, J.H.; Lee, Y.J.; Kim, T.S.; Park, K.T. Fabrication of a spherical titanium powder by combined combustion synthesis and DC plasma treatment. Arch. Metall. Mater. 2017, 62, 1057–1062. [Google Scholar] [CrossRef]
- Bolivar, R.; Friedrich, B. Synthesis of titanium via magnesiothermic reduction of TiO2 (pigment). In Proceedings of the EMC, Kyoto, Japan, 20–24 July 2009. [Google Scholar]
- HSC Chemistry Software ver. 8.0, Eh-pH Diagrams Module, Outotec. 2014. Available online: https://www.outotec.com (accessed on 20 November 2018).
- Brown, P.L.; Ekberg, C. Hydrolysis of Metal Ions; Wiley-VCH: Weinheim, Germany, 2016; pp. 180, 194. [Google Scholar]
- Won, Y.R.; Kim, D.S. Studies on the effect of temperature on lead ion in aqueous environment based on pourbaix diagram. J. Korea Soc. Waste Manag. 2013, 30, 60–67. [Google Scholar] [CrossRef]
- Qian, M.; Froes, F.H. Titanium Powder Metallurgy: Science, Technology and Applications; Elsevier Inc.: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Fang, Z.Z.; Middlemas, S.; Guo, J.; Fan, P. A new, energy-efficient chemical pathway for extracting Ti metal from Ti minerals. J. Am. Chem. Soc. 2013, 135, 18248–18251. [Google Scholar] [CrossRef] [PubMed]
- HSC Chemistry Software ver. 8.0, Reaction Equations Module, Outotec. 2014. Available online: https://www.outotec.com (accessed on 20 November 2018).
- Dong, H.; Unluer, C.; Al-Tabbaa, A.; Yang, E.H. Characterization of MgO Calcined from Mg(OH)2 Produced from Reject Brine. In Proceedings of the Fourth International Conference on Sustainable Construction Materials and Technologies, Las Vegas, NV, USA, 7–11 August 2016. [Google Scholar]
- TOPAS Software ver. 5.0, Rietveld Refinement Module, Bruker. 2014. Available online: https://www.bruker.com/products/x-ray-diffraction-and-elemental-analysis/x-ray-diffraction/xrd-software/topas.html (accessed on 17 August 2018).
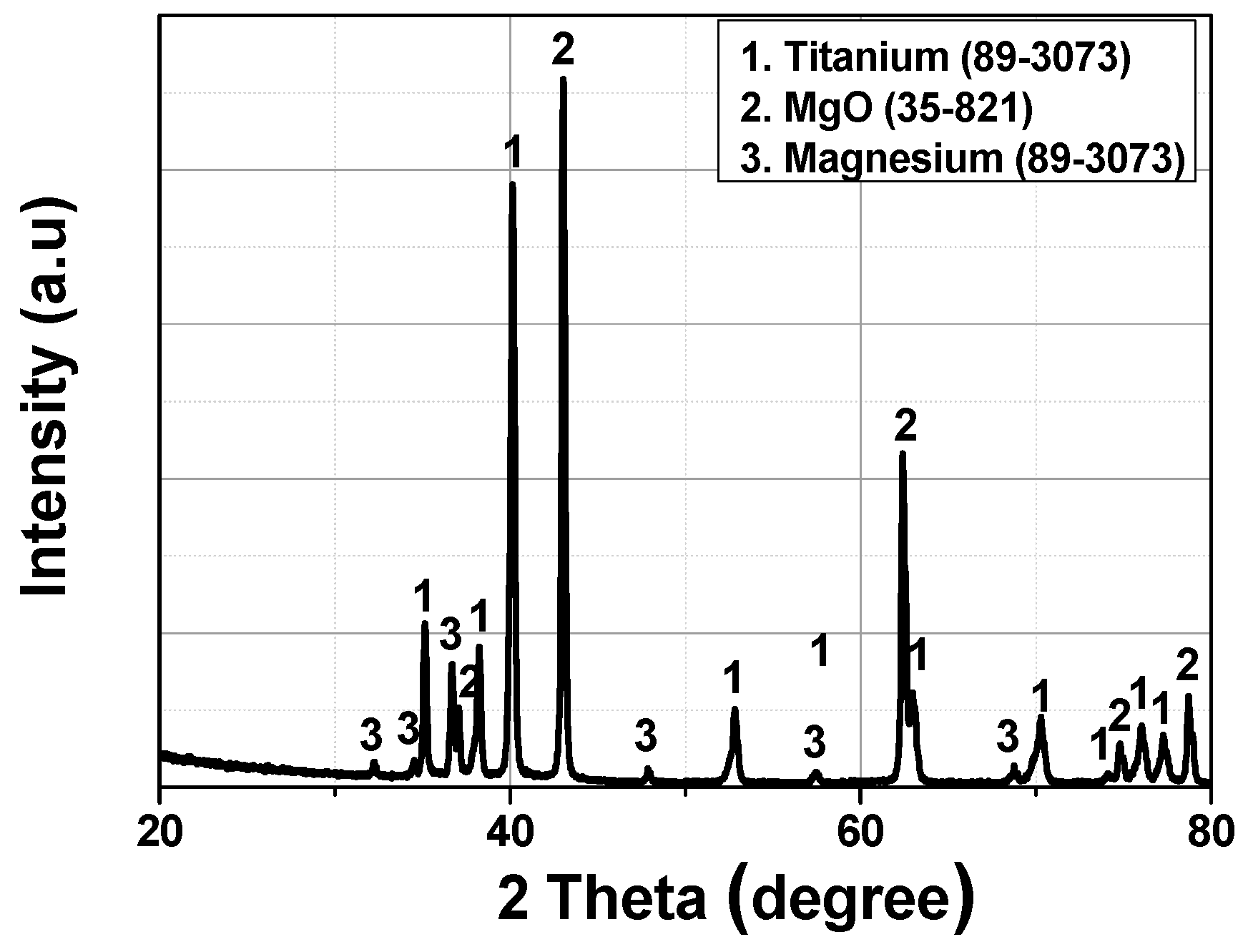
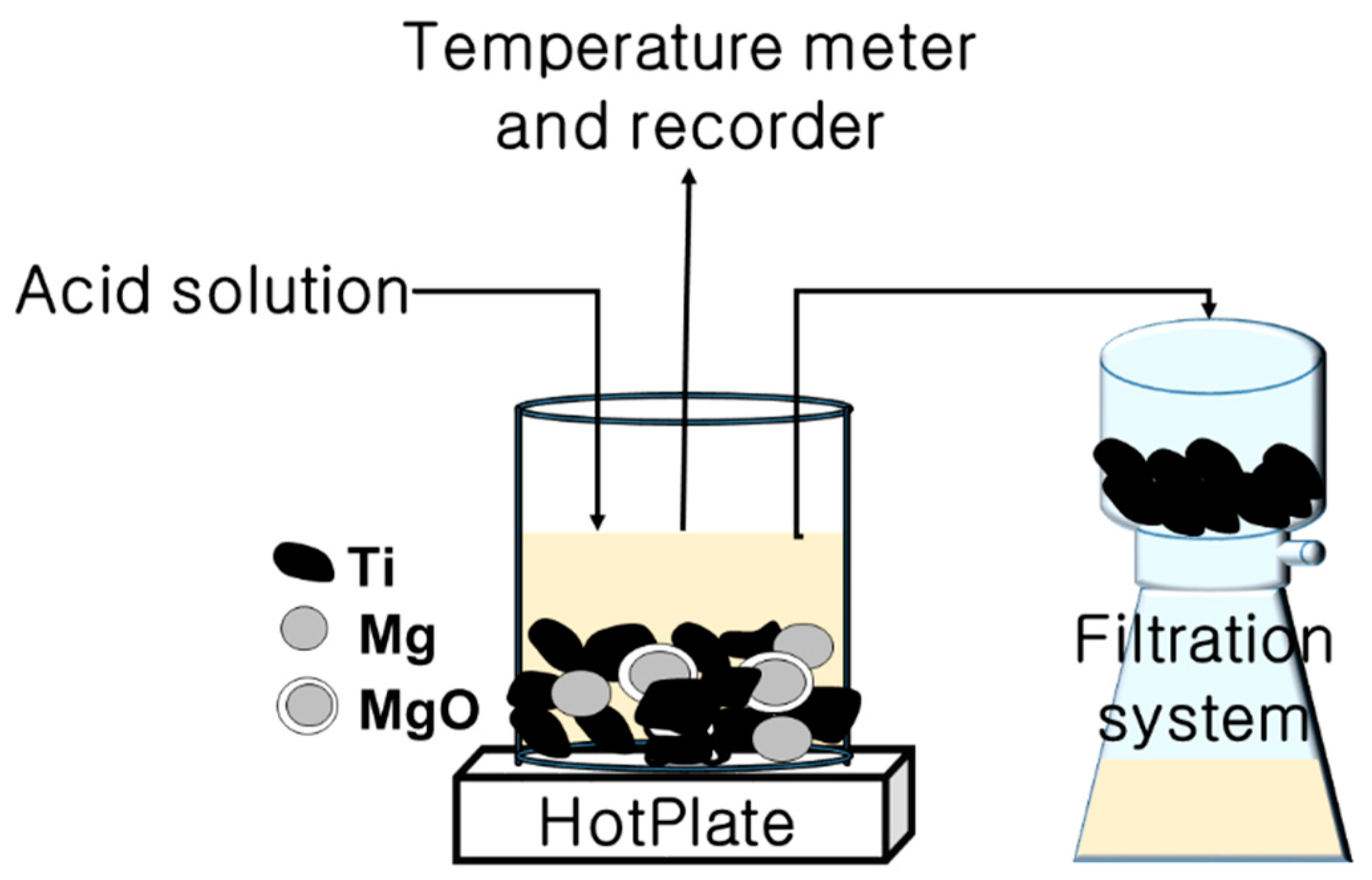
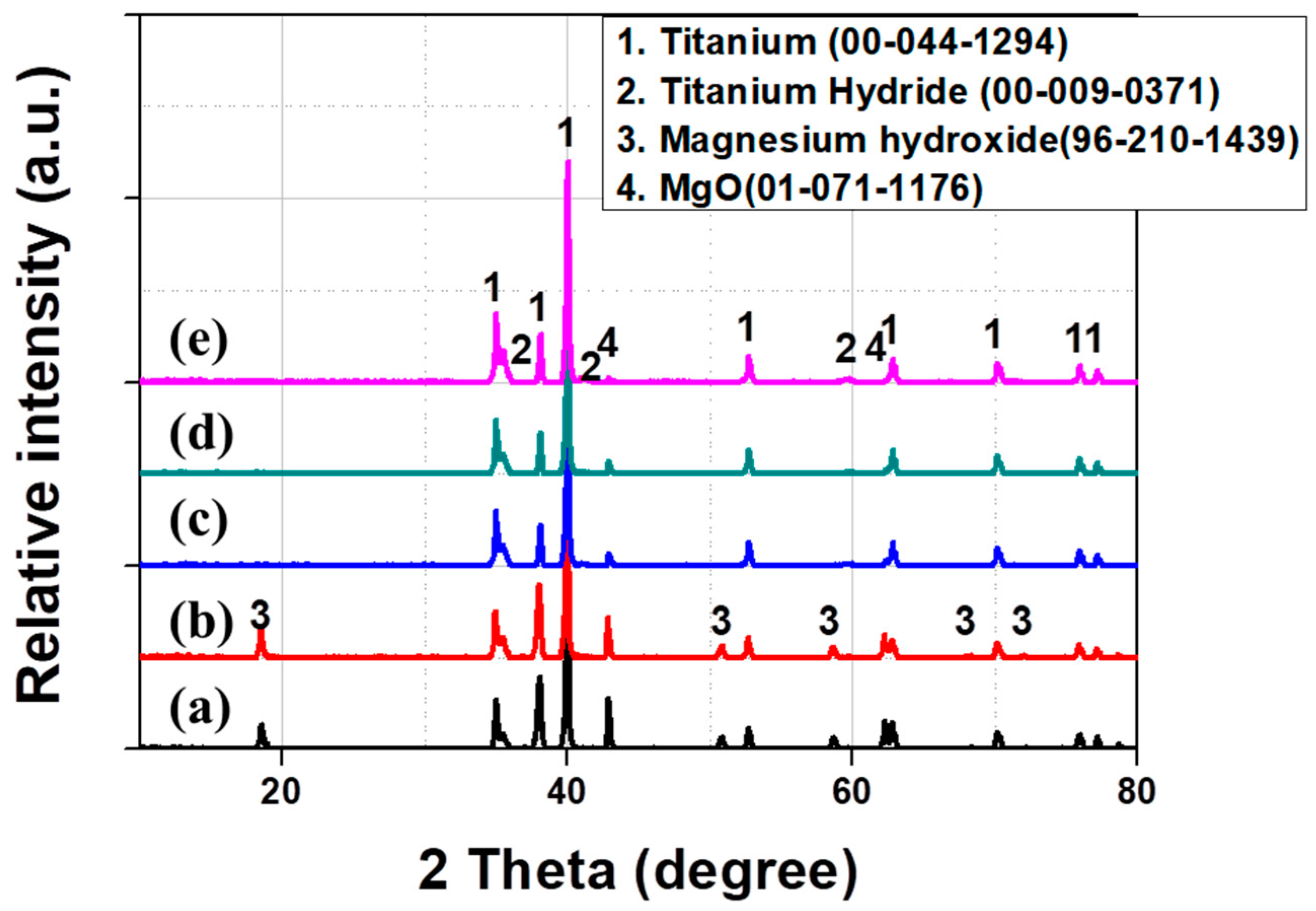
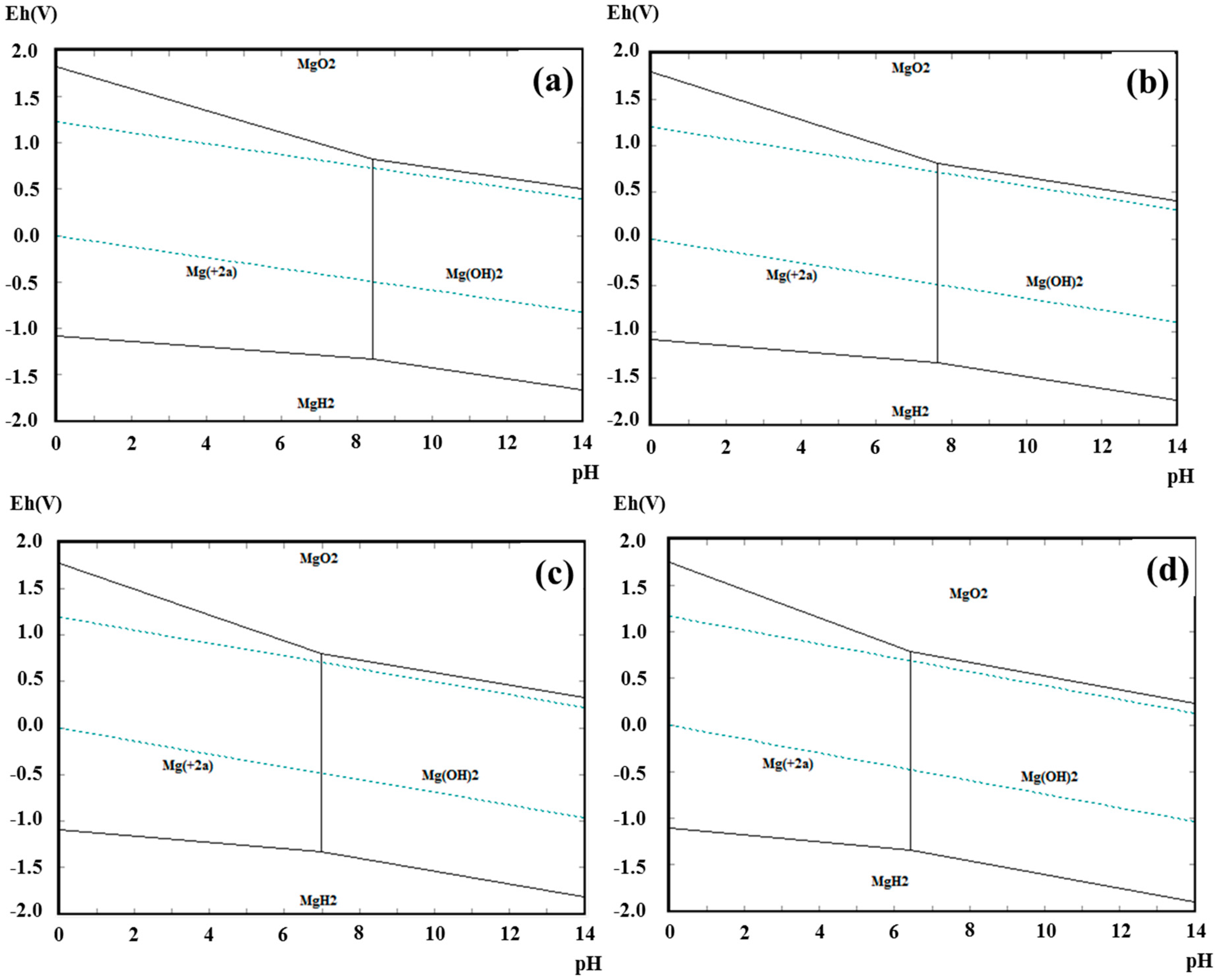
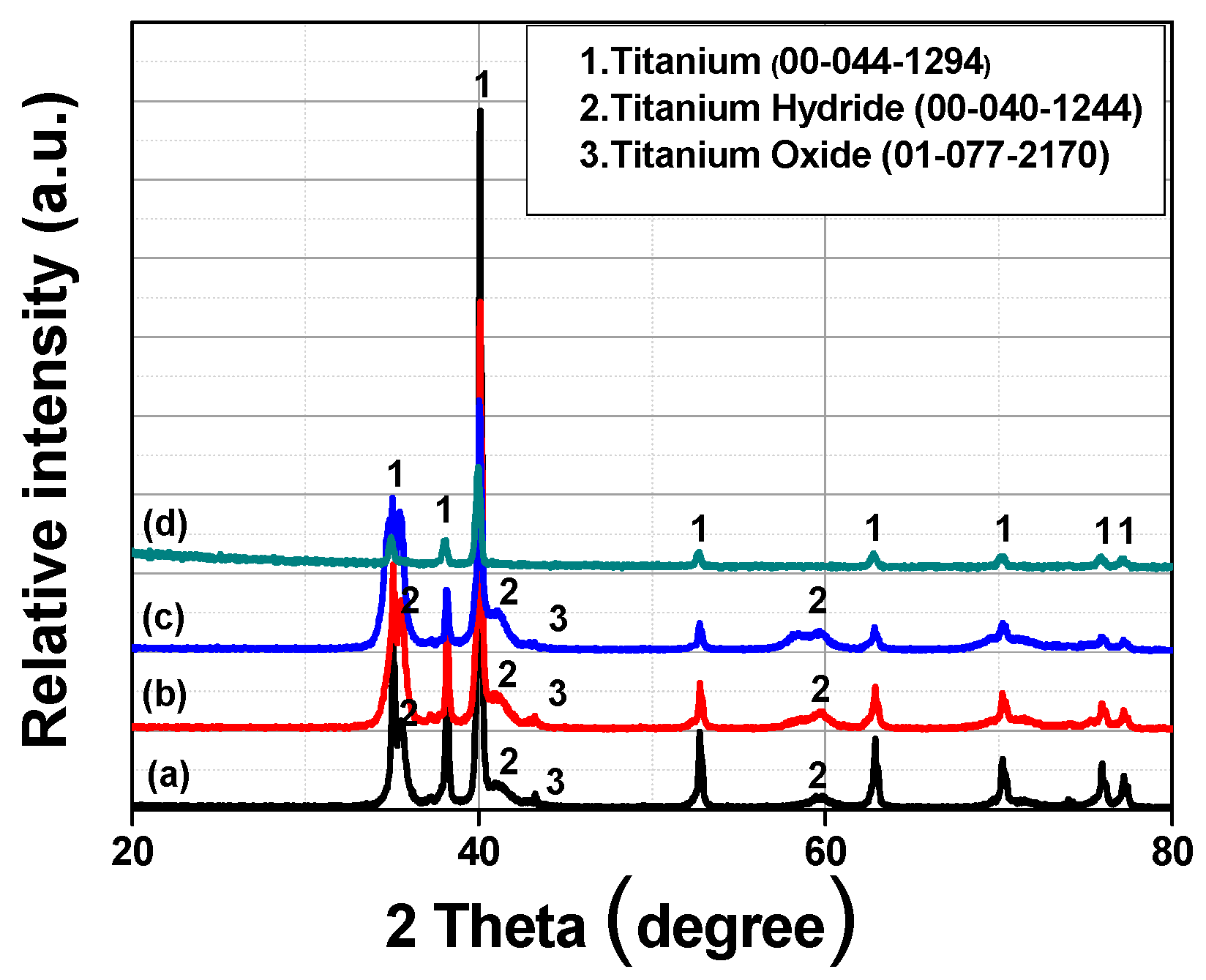
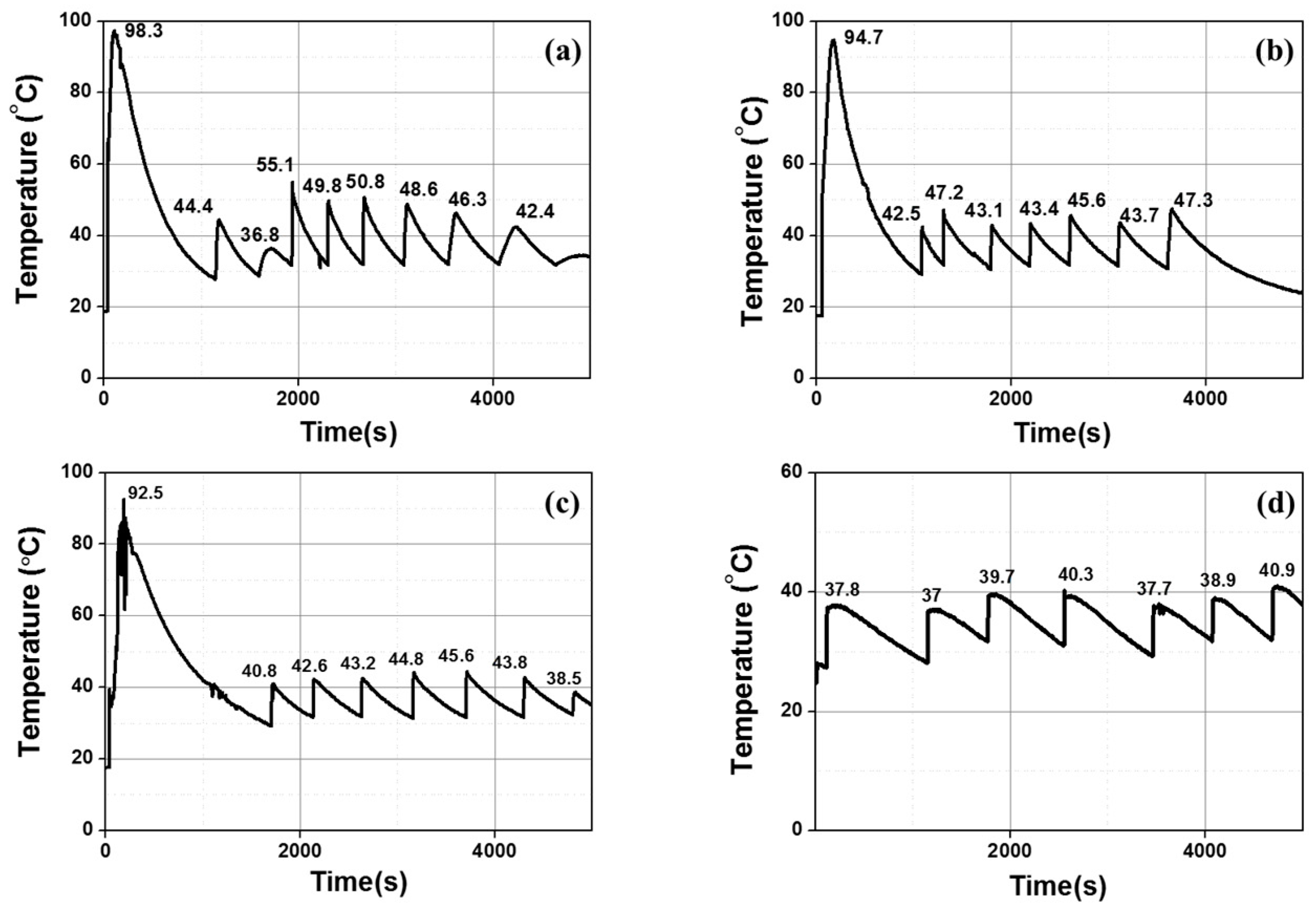
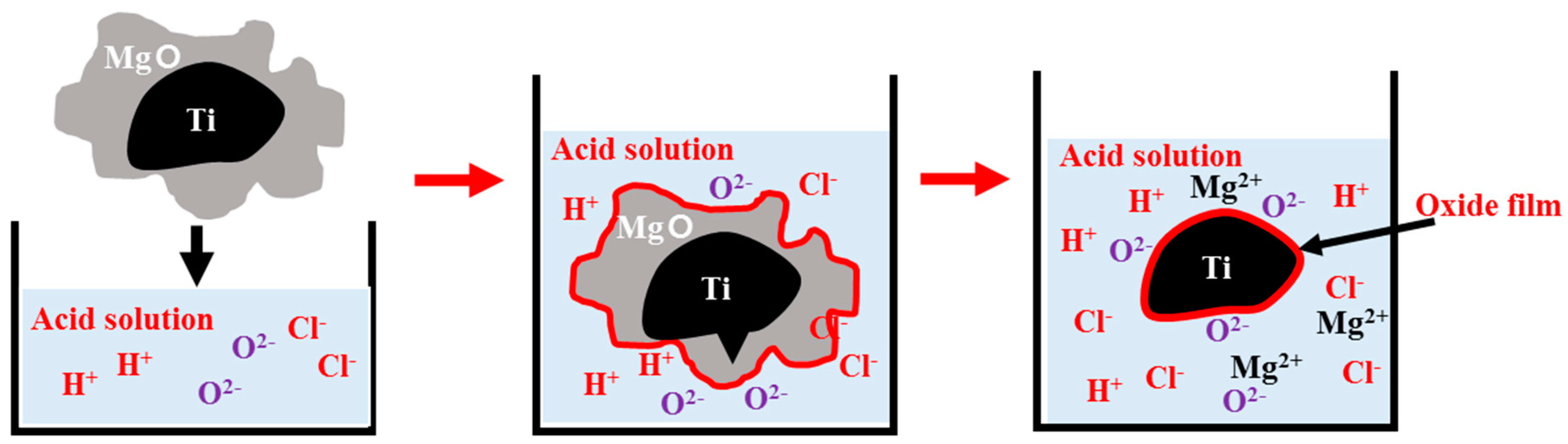

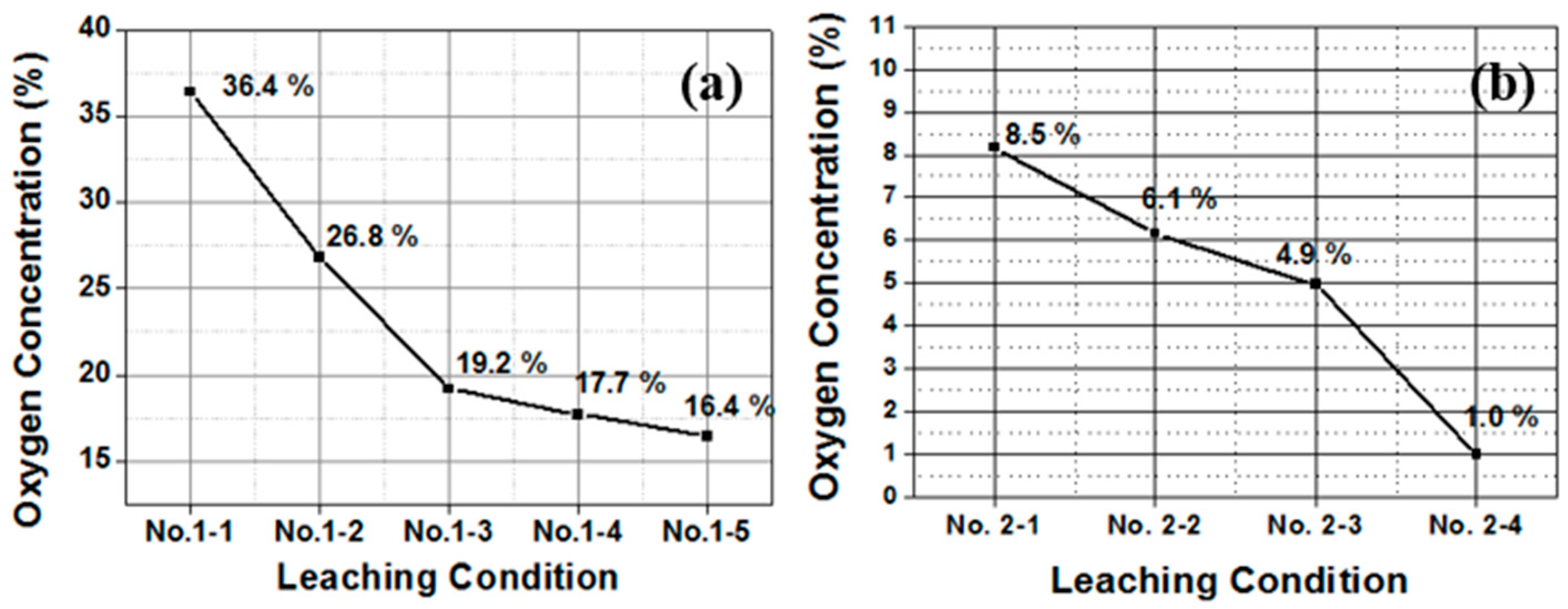
| Group Number | Experiment Number | Step | Solvent | Concentration (M) | Amount of Product (g) | Total Volume of Solution (mL) | Stirring Time (h) | Stirring Speed (rpm) |
|---|---|---|---|---|---|---|---|---|
| Group 1 | 1–1 | 1 | HCl | 5 | 28 | 183 | 4 | 150 |
| 1–2 | 5.5 | |||||||
| 1–3 | 6.5 | |||||||
| 1–4 | 7 | |||||||
| 1–5 | 7.5 | |||||||
| Group 2 | 2–1 | 1 | CH3COOH | 8.75 | 183 | 6 | ||
| 2 | HCl | 6.5 | 183 | 1 | ||||
| 2–2 | 1 | HCl | 0.6 | 183 | 6 | |||
| 2 | 6.5 | 183 | 1 | |||||
| 2–3 | 1 | HCl | 0.6 | 183 | 6 | |||
| 2 | 7.5 | 183 | 1 | |||||
| 2–4 | 1 | HCl | 0.4 | 3000 | 6 | |||
| 2 | 7.5 | 183 | 1 |
| Ti + MgO + Mg + 4HCl → Ti + H2O(g) + H2(g) | Ti + MgO + Mg + 4CH3COOH → Ti + 2Mg(CH3COO)2 + H2O(g) + H2(g) | |||
|---|---|---|---|---|
| Temp. (°C) | ΔH (kJ) | ΔG (kJ) | ΔH (kJ) | ΔG (kJ) |
| 10 | −213.557 | −328.134 | −645.888 | −488.862 |
| 20 | −217.321 | −332.114 | −627.825 | −483.641 |
| 30 | −220.731 | −335.971 | −612.213 | −478.991 |
| 40 | −223.968 | −339.720 | −598.095 | −474.830 |
| 50 | −227.133 | −343.366 | −584.942 | −471.104 |
| 60 | −230.256 | −346.915 | −572.584 | −467.772 |
| 70 | −233.373 | −350.370 | −560.828 | −464.802 |
| 80 | −236.525 | −353.734 | −549.498 | −462.168 |
| 90 | −239.732 | −357.008 | −538.522 | −459.850 |
| 100 | −243.019 | −360.193 | −527.840 | −457.830 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, S.H.; Sim, J.J.; Lim, J.H.; Seo, S.-J.; Kim, D.-W.; Hyun, S.-K.; Park, K.-T. Removal of Mg and MgO By-Products through Magnesiothermic Reduction of Ti Powder in Self-Propagating High-Temperature Synthesis. Metals 2019, 9, 169. https://doi.org/10.3390/met9020169
Choi SH, Sim JJ, Lim JH, Seo S-J, Kim D-W, Hyun S-K, Park K-T. Removal of Mg and MgO By-Products through Magnesiothermic Reduction of Ti Powder in Self-Propagating High-Temperature Synthesis. Metals. 2019; 9(2):169. https://doi.org/10.3390/met9020169
Chicago/Turabian StyleChoi, Sang Hoon, Jae Jin Sim, Jae Hong Lim, Seok-Jun Seo, Dong-Wook Kim, Soong-Keun Hyun, and Kyoung-Tae Park. 2019. "Removal of Mg and MgO By-Products through Magnesiothermic Reduction of Ti Powder in Self-Propagating High-Temperature Synthesis" Metals 9, no. 2: 169. https://doi.org/10.3390/met9020169
APA StyleChoi, S. H., Sim, J. J., Lim, J. H., Seo, S.-J., Kim, D.-W., Hyun, S.-K., & Park, K.-T. (2019). Removal of Mg and MgO By-Products through Magnesiothermic Reduction of Ti Powder in Self-Propagating High-Temperature Synthesis. Metals, 9(2), 169. https://doi.org/10.3390/met9020169





