Extraction of Mn from Black Copper Using Iron Oxides from Tailings and Fe2+ as Reducing Agents in Acid Medium
Abstract
:1. Introduction
2. Methodology
2.1. Black Oxide Samples
2.2. Ferrous Ions
2.3. Iron Oxide Tailings
2.4. Reagent and Leaching Test
2.5. The Effect of the Fe/MnO2 Ratio
2.6. The Effect of the Acid Concentration on the System
3. Results
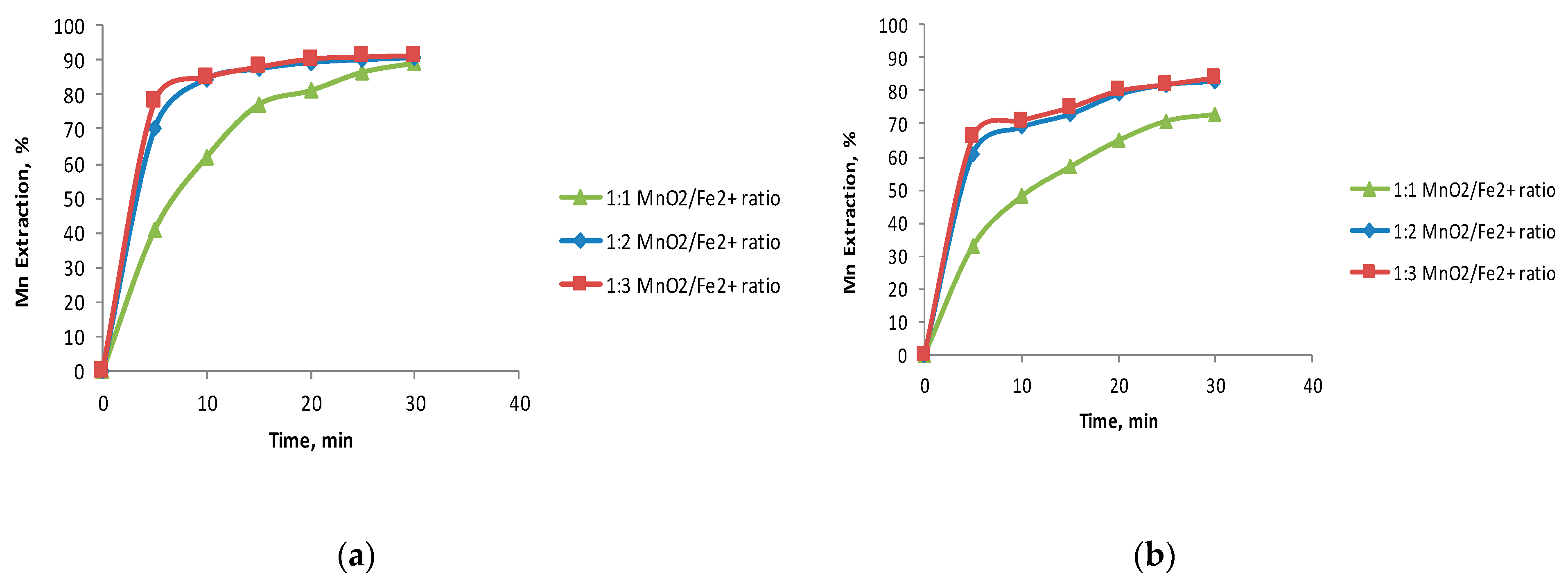
3.1. The Effect of the Fe2+/MnO2 Ratio
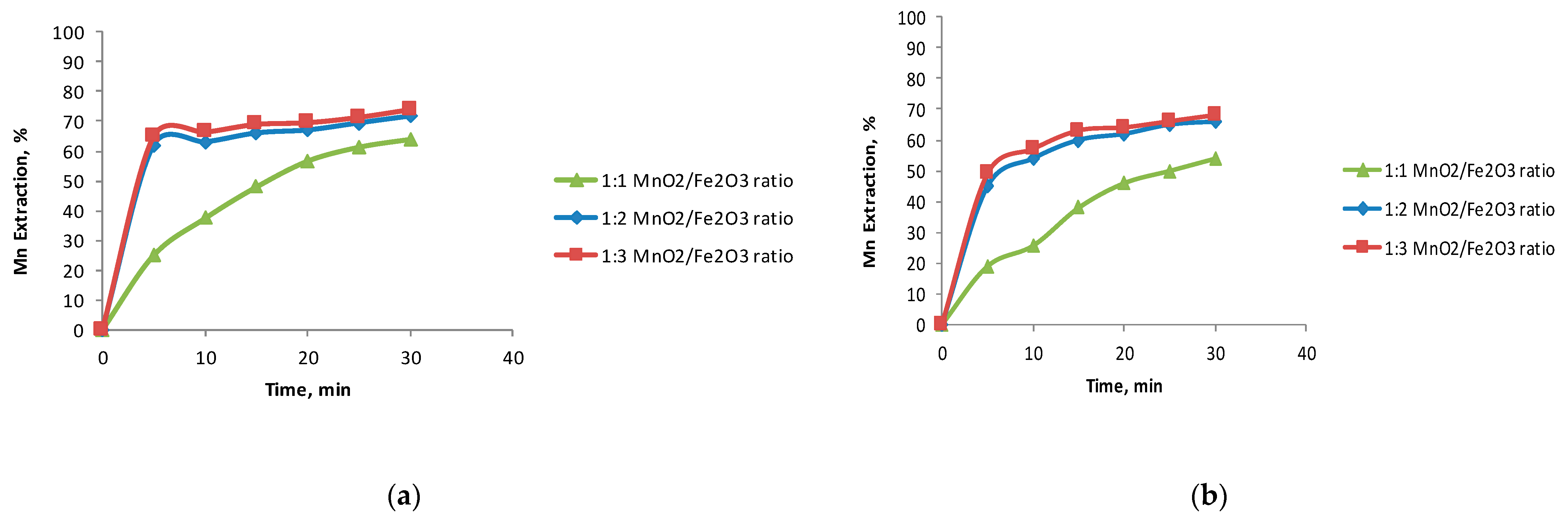
3.2. The Effect of the Fe2O3/MnO2 Ratio
3.3. The Effect of the H2SO4 Concentration
4. Conclusions
- (1)
- The ferrous ions were a better reducing agent than iron oxides to dissolve MnO2 in black copper.
- (2)
- The optimal reducing agent/black copper ratio was 2:1 for the studied reducing agents studied.
- (3)
- High concentrations of H2SO4 had a positive effect on the dissolution of Mn with the BCS-2 sample owing to the high content of clay (montmorillonite and kaolinite) and gangue (chlorite), which consume significant amounts of acid. The acid concentration was not significant with the BCS-1 sample.
- (4)
- The best results in this study were obtained working with the sample with fewer impurities (BCS-1), with an Fe2+/black copper ratio of 2:1, and 1 mol/L of sulfuric acid.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Servicio Nacional de Geología y Minería (SERNAGEOMIN). Anuario de la Minería de Chile; Servicio Nacional de Geología y Minería: Santiago, Chile, 2018; p. 274. [Google Scholar]
- Menzies, A.; Campos, E.; Hernández, V.; Sola, S.; Riquelme, R. Understanding Exotic-Cu Mineralisation Part II: Characterization of ‘Black Copper’ ore (‘Cobre Negro’). In Proceedings of the 13th SGA Biennial Meeting, Nancy, France, 24–27 August 2015; pp. 3–6. [Google Scholar]
- U.S. Geological Survey and U.S. Department of the Interior, Mineral. Commodity Summaries; U.S. Geological Survey: Reston, Virginia, 2018.
- Ossandon, C.G.; Freraut, C.R.; Gustafson, L.B.; Lindsay, D.D.; Zentilli, M. Geology of the Chuquicamata Mine: A Progress Report. Econ. Geol. 2001, 96, 249–270. [Google Scholar] [CrossRef]
- Mote, T.I.; Becker, T.A.; Renne, P.; Brimhall, G.H. Chronology of Exotic Mineralization at El Salvador, Chile, by 40Ar/39Ar Dating of Copper Wad and Supergene Alunite. Econ. Geol. 2001, 96, 351–366. [Google Scholar] [CrossRef]
- Cuadra, C.P.; Rojas, S.G. Oxide mineralization at the Radomiro Tomic porphyry copper deposit, Northern Chile. Econ. Geol. 2001, 96, 387–400. [Google Scholar]
- Mora, R.; Artal, J.; Brockway, H.; Martinez, E.; Muhr, R. El Tesoro exotic copper deposit, Antofagasta region, northern Chile. Econ. Geol. Spec. Publ. 2004, 11, 187–197. [Google Scholar]
- Pinget, M.; Dold, B.; Fontboté, L. Exotic mineralization at Chuquicamata, Chile: Focus on the copper wad enigma. In Proceedings of the 10th Swiss Geoscience Meeting, Bern, Switzerland, 16–17 November 2012; pp. 88–89. [Google Scholar]
- Kojima, S.; Astudillo, J.; Rojo, J.; Tristá, D.; Hayashi, K.-I. Ore mineralogy, fluid inclusion, and stable isotopic characteristics of stratiform copper deposits in the coastal Cordillera of northern Chile. Miner. Deposita 2003, 38, 208–216. [Google Scholar] [CrossRef]
- Pincheira, M.; Dagnini, A.; Kelm, U.; Helle, S. Copper Pitch Y Copper Wad: Contraste Entre Las Fases Presentes En Las Cabezas Y En Los Ripios En Pruebas De Mina sur, Chuquicamata; X Congreso Geológico Chileno: Concepción, Chile, 2003; p. 10. [Google Scholar]
- Hellé, S.; Kelm, U.; Barrientos, A.; Rivas, P.; Reghezza, A. Improvement of mineralogical and chemical characterization to predict the acid leaching of geometallurgical units from Mina Sur, Chuquicamata, Chile. Miner. Eng. 2005, 18, 1334–1336. [Google Scholar] [CrossRef]
- García, C.; Garcés, J.P.; Rojas, C.; Zárate, G. Efecto sinérgico del tratamiento de mezcla de minerales conteniendo copper wad y sulfuros secundarios. In Proceedings of the IV International Copper Hydrometallurgy Workshop, Viña del Mar, Chile, 16–18 May 2007. [Google Scholar]
- Zambra, J.; Kojima, S.; Espinoza, S.; Definis, A. Angélica Copper Deposit: Exotic Type Mineralization in the Tocopilla Plutonic Complex of the Coastal Cordillera, Northern Chile. Resour. Geol. 2007, 57, 427–434. [Google Scholar] [CrossRef]
- Consejo Minero. Guía Metodológica sobre Drenaje en la Industria Minera; Subsecretaría de economía Consejo Nacional de Producción Limpia: Santiago, Chile, 2002. [Google Scholar]
- Sequeira, R. A note on the consumption of acid through cation exchange with clay minerals in atmospheric precipitation. Atmos. Environ. Part. A Gen. Top. 1991, 25, 487–490. [Google Scholar] [CrossRef]
- Helle, S.; Pincheira, M.; Jerez, O.; Kelm, U. Sequential extraction to predict the leaching potential of refractory. In Proceedings of the XV Balkan Mineral Processing Congress, Sozopol, Bulgaria, 12–16 June 2013; pp. 109–111. [Google Scholar]
- Hernández, M.C.; Benavente, O.; Melo, E.; Núñez, D. Copper leach from black copper minerals. In Proceedings of the 6th International Seminar on Copper Hydrometallurgy, Viña del Mar, Chile, 6–8 July 2011; pp. 1–10. [Google Scholar]
- Randhawa, N.S.; Hait, J.; Jana, R.K. A brief overview on manganese nodules processing signifying the detail in the Indian context highlighting the international scenario. Hydrometallurgy 2016, 165, 166–181. [Google Scholar] [CrossRef]
- Saldaña, M.; Toro, N.; Castillo, J.; Hernández, P.; Trigueros, E.; Navarra, A. Development of an Analytical Model for the Extraction of Manganese from Marine Nodules. Metals 2019, 9, 903. [Google Scholar] [CrossRef]
- Kanungo, S. Rate process of the reduction leaching of manganese nodules in dilute HCl in presence of pyrite. Hydrometallurgy 1999, 52, 313–330. [Google Scholar] [CrossRef]
- Kanungo, S.; Das, R. Extraction of metals from manganese nodules of the Indian Ocean by leaching in aqueous solution of sulphur dioxide. Hydrometallurgy 1988, 20, 135–146. [Google Scholar] [CrossRef]
- Su, H.; Liu, H.; Wang, F.; Lu, X.; Wen, Y.-X. Kinetics of Reductive Leaching of Low-grade Pyrolusite with Molasses Alcohol Wastewater in H2SO4. Chin. J. Chem. Eng. 2010, 18, 730–735. [Google Scholar] [CrossRef]
- Bafghi, M.S.; Zakeri, A.; Ghasemi, Z.; Adeli, M. Reductive dissolution of manganese ore in sulfuric acid in the presence of iron metal. Hydrometallurgy 2008, 90, 207–212. [Google Scholar] [CrossRef]
- Toro, N.; Saldaña, M.; Gálvez, E.; Cánovas, M.; Trigueros, E.; Castillo, J.; Hernández, P.C. Optimization of Parameters for the Dissolution of Mn from Manganese Nodules with the Use of Tailings in An Acid Medium. Minerals 2019, 9, 387. [Google Scholar] [CrossRef]
- Zakeri, A.; Bafghi, M.; Shahriari, S. Dissolution Kinetics of Manganese Dioxide Ore in Sulfuric Acid in the Presence of Ferrous Ion. Iran. J. Mater. Sci. Eng. 2007, 4, 22–27. [Google Scholar]
- Toro, N.; Saldaña, M.; Castillo, J.; Higuera, F.; Acosta, R. Leaching of Manganese from Marine Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Tailings. Minerals 2019, 9, 289. [Google Scholar] [CrossRef]
- Benavente, O.; Hernández, M.C.; Melo, E.; Núñez, D.; Quezada, V.; Zepeda, Y. Copper Dissolution from Black Copper Ore under Oxidizing and Reducing Conditions. Metals 2019, 9, 799. [Google Scholar] [CrossRef]
- Toro, N.; Herrera, N.; Castillo, J.; Torres, C.M.; Sepúlveda, R. Initial Investigation into the Leaching of Manganese from Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Foundry Slag—Part, I. Minerals 2018, 8, 565. [Google Scholar] [CrossRef]
- Helle, S.; Kelm, U. Experimental leaching of atacamite, chrysocolla and malachite: Relationship between copper retention and cation exchange capacity. Hydrometallurgy 2005, 78, 180–186. [Google Scholar] [CrossRef]
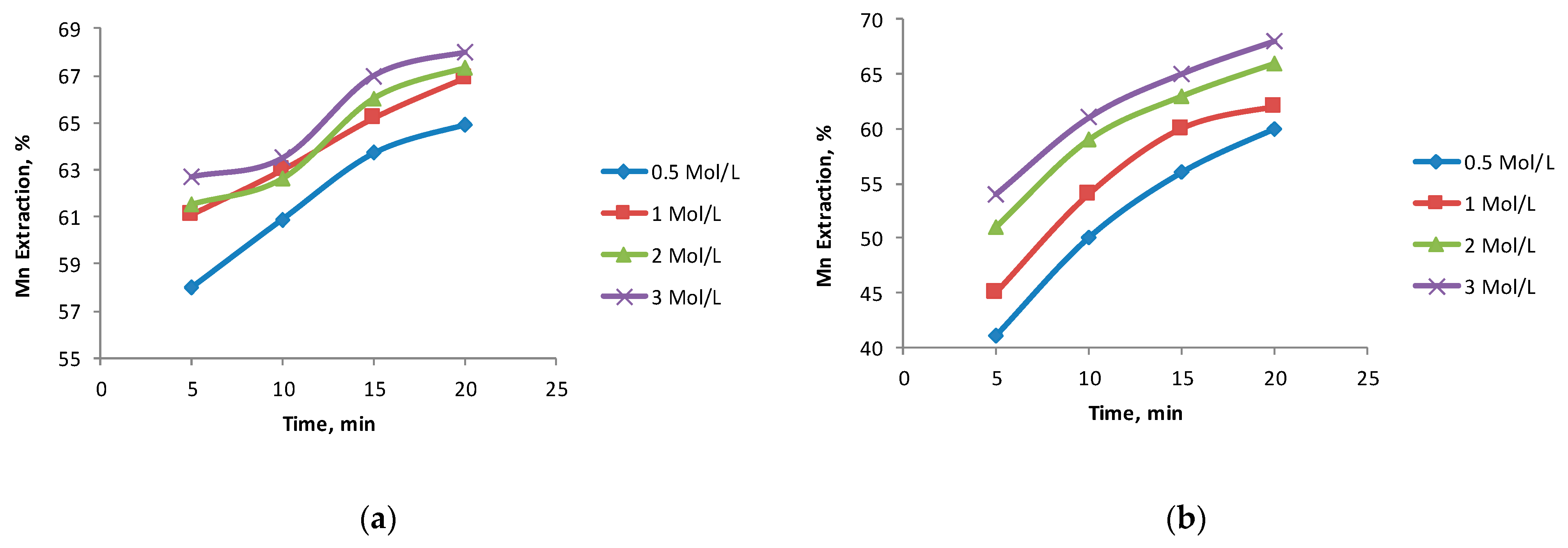
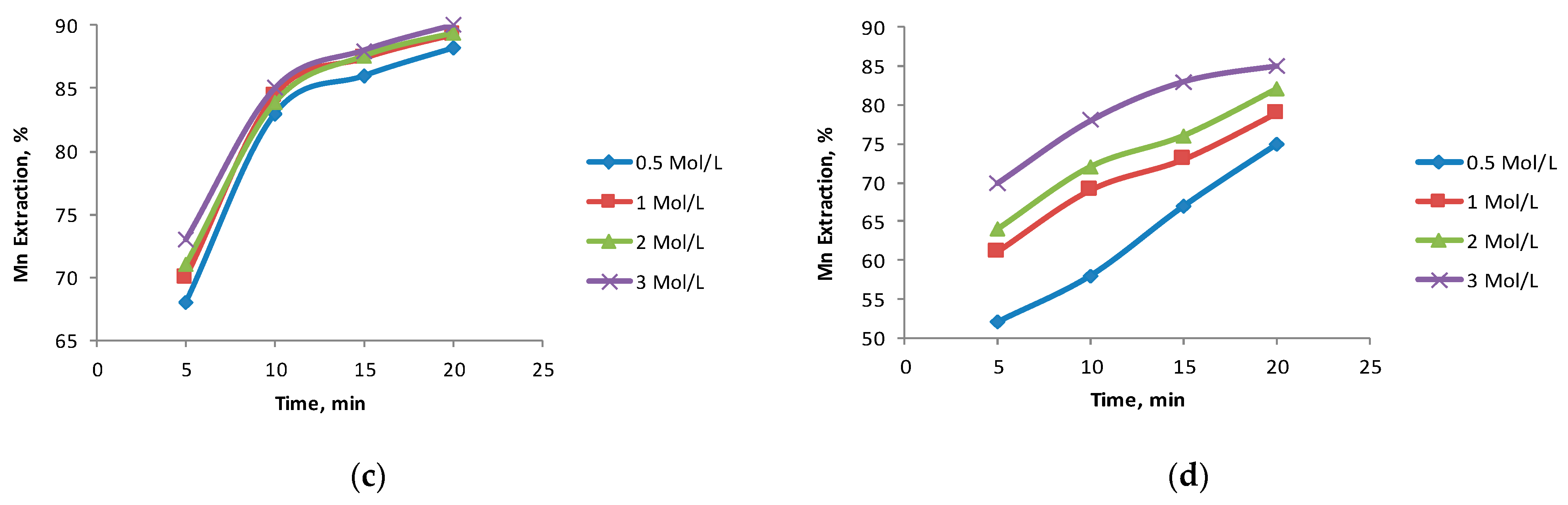
| Sample | Mn (%) | Fe (%) |
|---|---|---|
| Black Copper Sample-1 | 22.01 | 7.92 |
| Black Copper Sample-2 | 0.51 | 3.88 |
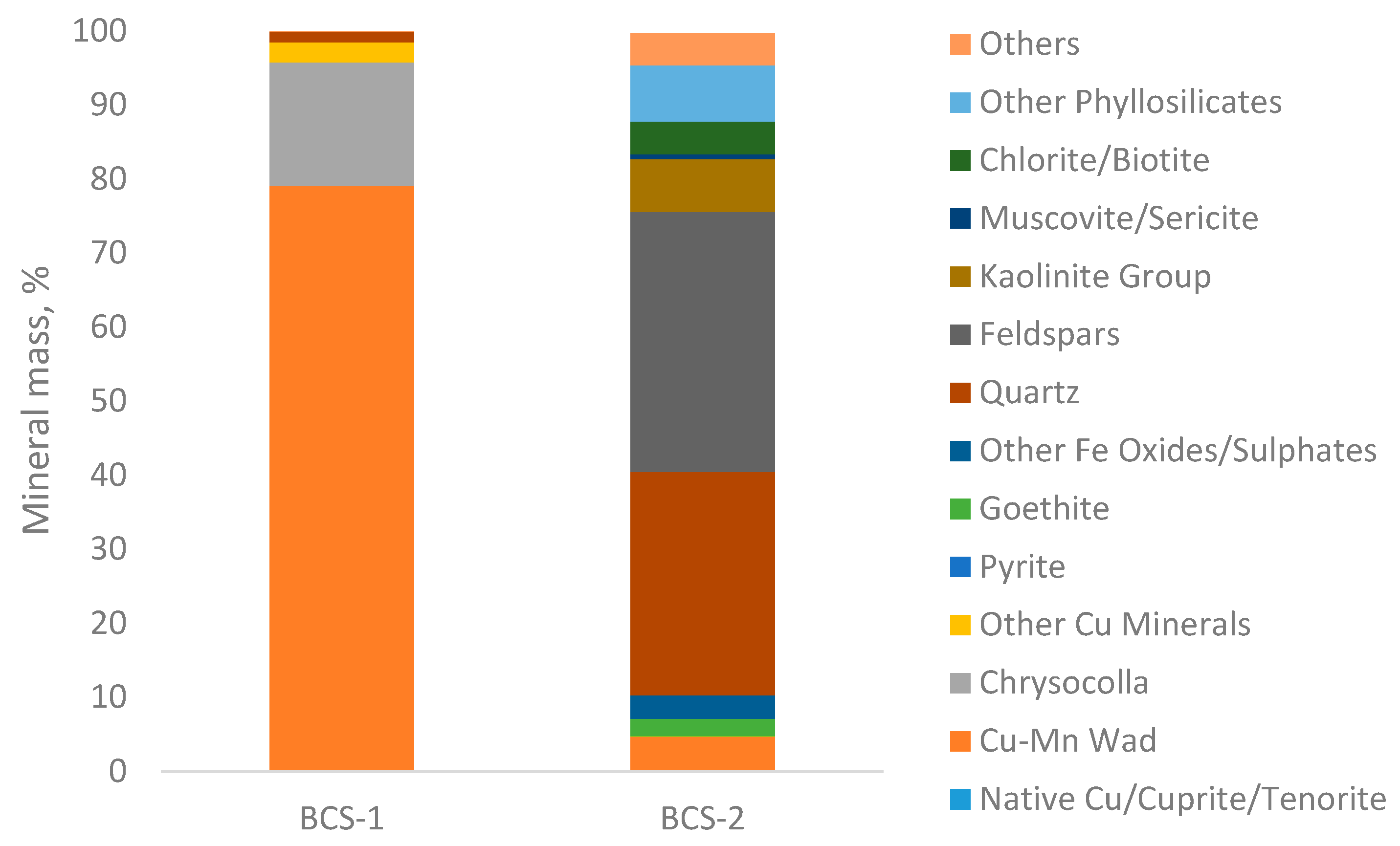
| Mineral (% Mass) | Black Copper Sample-1 | Black Copper Sample-2 |
|---|---|---|
| Native Cu/Cuprite/Tenorite | 0.12 | 0.00 |
| Cu-Mn Wad | 78.90 | 4.64 |
| Chrysocolla | 16.72 | 0.03 |
| Other Cu Minerals | 2.69 | 0.03 |
| Pyrite | 0.00 | 0.01 |
| Goethite | 0.01 | 2.39 |
| Other Fe Oxides/Sulphates | 0.00 | 3.15 |
| Quartz | 1.41 | 30.20 |
| Feldspars | 0.02 | 35.11 |
| Kaolinite Group | 0.01 | 7.08 |
| Muscovite/Sericite | 0.01 | 0.67 |
| Chlorite/Biotite | 0.01 | 4.45 |
| Montmorillonite | 0.00 | 4.56 |
| Others | 0.09 | 7.35 |
| Total | 100 | 100 |
| Mineral | Amount % (w/w) |
|---|---|
| Chalcopyrite/Bornite | 0.47 |
| Tennantite/Tetrahedrite | 0.03 |
| Other Cu Minerals | 0.63 |
| Cu–Fe Hydroxides | 0.94 |
| Pyrite | 0.12 |
| Magnetite | 58.52 |
| Specular Hematite | 0.89 |
| Hematite | 4.47 |
| Ilmenite/Titanite/Rutile | 0.04 |
| Siderite | 0.22 |
| Chlorite/Biotite | 3.13 |
| Other Phyllosilicates | 11.61 |
| Fayalite | 4.59 |
| Dicalcium Silicate (Si) | 8.30 |
| Kirschsteinite (CaFeSi) | 3.40 |
| Forsterite (Si) | 2.30 |
| Barite (BaSO4) | 0.08 |
| Zinc Oxide (ZnO) | 0.02 |
| Lead Oxide (PbO) | 0.01 |
| Sulfate (S) | 0.20 |
| Others | 0.03 |
| Total | 100.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez, K.; Toro, N.; Campos, E.; González, J.; Jeldres, R.I.; Nazer, A.; Rodriguez, M.H. Extraction of Mn from Black Copper Using Iron Oxides from Tailings and Fe2+ as Reducing Agents in Acid Medium. Metals 2019, 9, 1112. https://doi.org/10.3390/met9101112
Pérez K, Toro N, Campos E, González J, Jeldres RI, Nazer A, Rodriguez MH. Extraction of Mn from Black Copper Using Iron Oxides from Tailings and Fe2+ as Reducing Agents in Acid Medium. Metals. 2019; 9(10):1112. https://doi.org/10.3390/met9101112
Chicago/Turabian StylePérez, Kevin, Norman Toro, Eduardo Campos, Javier González, Ricardo I. Jeldres, Amin Nazer, and Mario H. Rodriguez. 2019. "Extraction of Mn from Black Copper Using Iron Oxides from Tailings and Fe2+ as Reducing Agents in Acid Medium" Metals 9, no. 10: 1112. https://doi.org/10.3390/met9101112
APA StylePérez, K., Toro, N., Campos, E., González, J., Jeldres, R. I., Nazer, A., & Rodriguez, M. H. (2019). Extraction of Mn from Black Copper Using Iron Oxides from Tailings and Fe2+ as Reducing Agents in Acid Medium. Metals, 9(10), 1112. https://doi.org/10.3390/met9101112








