3. Experimental
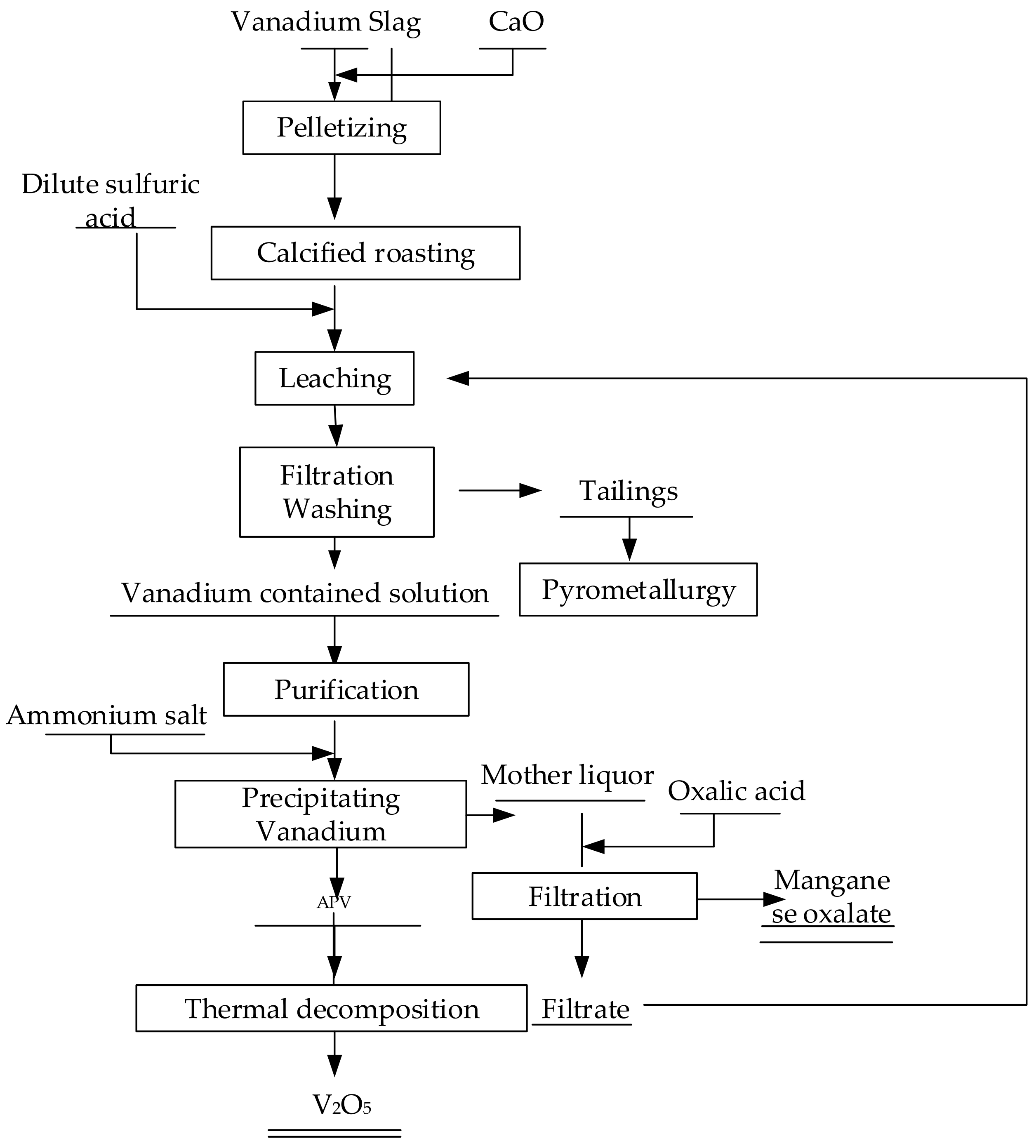
The whole flow chart for treating the converter vanadium slag was displayed as
Figure 2. Roasting experiments were carried out in a vertical tube furnace controlled by a Shinaden SR-53 temperature programmed instrument with molybdenum disilicide heating elements. Leaching, purification and vanadium precipitation were conducted in three-necked glass vessels centered in a thermostatic water bath (HH-4, China Changzhou Sino Instrument Co., LTD, Changzhou, China) and the pH values were measured by a pH-meter (Starter 3C). The operational parameters have been optimized in our previous studies [
22,
23,
24].
The vanadium slag (48–75 μm) was mixed with additive of CaO at a mass ratio (CaO)/(V2O5) of 0.42 and prepared into Ф 8–10 mm pellets. The pellets were heated to 850 °C at rate of 2 °C/min from room temperature and then held for 150 min. When the holding time was completed, the roasted pellets were quenched to room temperature and ground to fine powder. Roasting efficiency was expressed by vanadium recovery upon leaching, and the leaching conditions were liquid to solid ratio of 3:1, temperature of 55 °C, time of 30 min, pH of 2.5 and stirring speed of 500 rpm.
Because the lower selectivity of dilute acid leaching, some impurities of Ca, Mg, Si, Al, and Mn transferred into vanadium solution accompanying with V, and which in turn affected vanadium precipitation rate and quality of final product. According to analyses for occurrence of impurities and vanadium in leaching solution, neutralization separation could be adopted to remove Si and Al, during which the solution pH value was adjusted to 4.5 ± 0.05. Taking into consideration of decreasing the vanadium loss, Ca, Mg, Al and Si were removed simultaneously through adding a composite reagent of MnNH3F3 to precipitate Ca, Mg and adjusting the solution pH to remove Al and Si. The purification conditions were set as stirring speed of 200 rpm, reagent adding amount of 1.6 (molar ratio of 2n(F)/n(Ca + Mg)), temperature of 50 °C and time of 30 min. Besides, flocculant polyacrylamide and diatomaceous earth (DA-1) were used to improve sedimentation and filtration respectively during the purification process.
Mn in solution was not removed before vanadium precipitation step and ammonium salt was added to precipitate vanadium since this method can alleviate the side effect of Mn on the quality of final product compared with hydrolysis precipitation. Vanadium precipitation step was conducted at pH 2.00 ± 0.05 and temperature of 95 °C, ammonium sulfate was added at molar ratio (NH3)/(V) of 1.5, and the reaction time was controlled in 120 min. Some Vanadium precipitation was heated at 550 °C for 2 h and then saffron yellow powder of vanadium pentoxide was obtained. The vanadium precipitation and vanadium pentoxide were characterized by XRD (X’Pert Pro MPD, PAnalytical B.V., Almelo, Netherlands), SEM-EDS (SSX-550, Shimadzu Corporation, Kyoto, Japan) and laser particle size analyzer (Mastersizer 2000, Malvern Panalytical, Malvern, UK).
5. Evaluation of the Proposed Process and Conclusions
The proposed process for vanadium pentoxide production in this paper mainly includes five operational steps of calcified roasting, dilute acid leaching, chemical precipitation purification, vanadium precipitation with ammonium salt, thermal decomposition and recycle of mother liquor.
Compared with the sodium roasting process widely used in vanadium pentoxide production industry, this process performs best in environmental protection and resource utilization. The traditional vanadium extraction process of sodium roasting followed by water leaching has strict requirements for calcium content in raw vanadium slag. If the content of CaO is increased by 1%, vanadium leaching rate will decrease by 4.7–9% due to the formation of water-insoluble vanadate during roasting. On the contrary, powder CaO is applied as additives in the calcified roasting, and high content of calcium in the raw vanadium slag can decrease the adding amount of additive of CaO. During roasting step, poisonous gases, such as Cl2, HCl, SO2 and SO3 are discharged in the sodium roasting process, which results from the decomposition of additives of NaCl and Na2SO4 at high temperature, while calcified roasting can avoid this problem. Furthermore, calcified roasting can alleviate the phenomenon of burden bonding roasting furnace, which usually happens due to the generation of the low melting-point phase of sodium salts during sodium roasting process.
Since no sodium and potassium salt is introduced in the process, wastewater can be closed-circulating after removing the enriched impurities of P, Si, Ti and Cr with adding powder CaO. In contrast, it is well known that the wastewater from the sodium process contains high concentration of sodium, which is difficult to remove from the aqueous solution, resulting in enrichments of Na2SO4 which makes the viscosity of mother liquor increased and let the mother liquor not suitable for recycling to leach the roasted slag. In addition, because lots of chromium transfers into the vanadium-bearing solution as Cr6+ during the sodium roasting-water leaching process, the residual chromium in the mother liquor has to be treated with reduction and precipitation. In this proposed process, the valuable metals like Fe, Cr, Ti, Mn in the tailings after vanadium extraction are more feasible to be comprehensively recovered through conventional pyrometallurgical processes since little Na is contained. The product of vanadium pentoxide contains little alkaline metals of Na and K, it will be better to be used in the iron and steel industry.
In the proposed process, a chemical precipitation method was used to purifying the acidic solution. Unlike the conventional method of solvent extraction for purification of acidic vanadium-bearing solution, adding the composite reagent of Mn(NH4)F3 to remove the impurities from the acidic vanadium-bearing solution is easy to operation and convenient to apply in industry, meanwhile, does not bring forth any serious secondary pollution.
In the whole process, from roasting to thermal decomposition, the total vanadium recovery is more than 83% (> 91% × 95% ×96%), which is higher than 75% of that for the conventional sodium roasting process. Manganese resources can be simultaneously recovered as MnC2O4 with purity of 98.5% during the proposed process.
In conclusion, the final product of vanadium pentoxide prepared with this proposed process meets the standards of Grade 98 and the total vanadium recovery is higher than 83%, which is a relatively satisfactory vanadium resource utilization rate. In addition, during the whole process, little poisonous gas is discharged, wastewater almost realizes closed recycle, and the tailing after vanadium extraction is rich in Ca and favor to be treated with pyrometallurgy to recover other valuable metal resources.
Author Contributions
Conceptualization, J.Z.; Methodology, J.Z. and W.Z.; Software, J.Z. and W.Z., Validation, J.Z.; Formal Analysis, J.Z., W.Z. and Z.X.; Investigation, J.Z.; Resources, J.Z.; Data curation, J.Z., W.Z. and Z.X.; Writing-Original Draft Preparation, J.Z.; Writing-Review & Editing, W.Z. and Z.X.; Visualization, W.Z.; Supervision, Z.X.; Project Administration, Z.X.; Funding Acquisition, J.Z.
Funding
This project was financially supported by the National Natural Science Foundation of China (No. 51804230, 51804228) and the Scientific Research Project of Education Department of Hubei Province (No. Q20181108).
Acknowledgments
We gratefully acknowledge the Sichuan Weiyuan Iron & Steel Co., Ltd. for providing samples of vanadium slag and thank Aihua Liu for her contributions during collecting the data of particle size.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- Moskalyk, R.R.; Alfantazi, A.M. Processing of vanadium: A review. Miner. Eng. 2003, 16, 793–805. [Google Scholar] [CrossRef]
- Xu, J.Q.; Chen, G.R.; Guo, F.; Xie, J.Q. Development of wide-temperature vanadium-based catalysts for selective catalytic reducing of NOx with ammonia: Review. Chem. Eng. J. 2018, 353, 507–518. [Google Scholar] [CrossRef]
- Edgar, D.C.; Lino, H.; Carlos, C.; Valentina, V.C.; Lisseetle, J.; Vito, L.; Giuseppe, L. Vanadium: History, chemistry, interactions with α-amino acids and potential therapeutic applications. Coordin. Chem. Rev. 2018, 372, 117–140. [Google Scholar]
- Li, M.; Liu, B.; Zheng, S.L.; Wang, S.M.; Du, H.; Dreisinger, D.B.; Zhang, Y. A cleaner vanadium extraction method featuring non-salt roasting and ammonium bicarbonate leaching. J. Clean. Prod. 2017, 149, 206–217. [Google Scholar] [CrossRef]
- Wu, K.H.; Wang, Y.R.; Wang, X.R.; Wang, S.N.; Liu, B.; Zhang, Y.; Du, H. Co-extraction of vanadium and chromium from chromium containing vanadium slag by low-pressure liquid phase oxidation method. J. Clean. Prod. 2018, 203, 873–884. [Google Scholar] [CrossRef]
- Liu, B.; Du, H.; Wang, S.N. A novel method to extract vanadium and chromium from vanadium slag using molten NaOH-NaNO3 binary system. AIChE J. 2013, 59, 541–552. [Google Scholar] [CrossRef]
- Liu, F.; Ning, P.G.; Cao, H.B.; Zhang, Y. Measurement and modeling for vanadium extraction from the (NaVO3 + H2SO4 + H2O) system by primary amine N1923. J. Chem. Thermodyn. 2015, 80, 13–21. [Google Scholar] [CrossRef]
- Wang, Z.H.; Zheng, S.L.; Wang, S.N.; Qin, Y.L.; Du, H.; Zhang, Y. Electrochemical decomposition of vanadium slag in concentrated NaOH solution. Hydrometallurgy 2015, 151, 51–55. [Google Scholar] [CrossRef]
- Zhang, G.Q.; Zhang, T.A.; Lv, G.Z.; Zhang, Y.; Liu, Y.; Xie, G. Extraction of vanadium from LD converter slag by pressure leaching process with titanium white waste acid. Rare Metal Mat. Eng. 2015, 44, 1894–1898. [Google Scholar]
- Liu, Z.H.; Li, Y.; Chen, M.L.; Nueraihemaiti, A.; Du, J.; Fan, X.; Tao, C.Y. Enhanced leaching of vanadium slag in acidic solution by eletro-oxidation. Hydrometallurgy 2016, 159, 1–5. [Google Scholar] [CrossRef]
- Mirazimi, S.M.J.; Rashchi, F.; Saba, M. A new approach for direct leaching of vanadium from LD converter slag. Chem. Eng. Res. Des. 2015, 94, 131–140. [Google Scholar] [CrossRef]
- Zhang, G.Q.; Luo, D.M.; Deng, C.H.; Lv, L.; Liang, B.; Li, C. Simultaneous extraction of vanadium and titanium from vanadium slag using ammonium sulfate roasting-leaching process. J. Alloy Compd. 2018, 742, 504–511. [Google Scholar] [CrossRef]
- Zhang, J.H.; Zhang, W.; Xue, Z.L. Oxidation kinetics of vanadium slag roasting in the presence of calcium oxide. Min. Proc. Ext. Met. Rev. 2017, 38, 265–273. [Google Scholar] [CrossRef]
- Xiang, J.Y.; Huang, Q.Y.; Lv, X.W.; Bai, C.G. Extraction of vanadium from converter slag by two-step sulfuric acid leaching process. J. Clean. Prod. 2018, 170, 1089–1101. [Google Scholar] [CrossRef]
- Zhang, J.H.; Zhang, W.; Zhang, L.; Gu, S.Q. A critical review of technology for selective recovery of vanadium from leaching solution in V2O5 production. Solvent Extr. Ion Exc. 2014, 32, 221–248. [Google Scholar] [CrossRef]
- Sun, Z.H. Analysis on new vanadium technologies and prospects of vanadium industry. Iron Steel Vanadium Titan. 2012, 33, 1–7. (In Chinese) [Google Scholar]
- Chen, H.S. Study on the vanadium extraction from vanadium slag with lime roasting process. Iron Steel Vanadium Titan. 1992, 13, 1–9. (In Chinese) [Google Scholar]
- Cao, P. Research on vanadium slag roasted with calcium salt. Iron Steel Vanadium Titan. 2012, 33, 30–34. (In Chinese) [Google Scholar]
- Yin, D.F.; Peng, Y.; Sun, Z.H.; He, W.Y. Influencing factors of calcified roasting and thermal analysis to the process of vanadium slag produced from Pangang. Metal Mine 2012, 4, 91–94. (In Chinese) [Google Scholar]
- Fu, Z.B. Experimental research on vanadium extraction by calcified roasting and acid leaching. Iron Steel Vanadium Titan. 2014, 35, 1–6. (In Chinese) [Google Scholar]
- Wang, M.H.; Liu, Z.H.; Zhao, H.; Liu, Y.; Lou, T.P.; Sun, Z.H.; Tang, H.J.; Jing, H. Measurement of calcification of vanadium slag by XRD semi-quantitative method. Iron Steel Vanadium Titan. 2018, 39, 6–12. (In Chinese) [Google Scholar] [CrossRef]
- Zhang, J.H.; Zhang, W.; Zhang, L.; Gu, S.Q. Mechanism of vanadium slag roasting with calcium oxide. Int. J. Mine. Process. 2015, 138, 20–29. [Google Scholar] [CrossRef]
- Zhang, J.H.; Zhang, W.; Zhang, L.; Gu, S.Q. Effect of acid leaching on the vanadium leaching rate in process of vanadium extraction using calcium roasting. J. Northeast. Univ. Nat. Sci. 2014, 35, 1574–1578. (In Chinese) [Google Scholar]
- Zhang, J.H.; Yan, Z.F.; Zhang, L. Influence factors and kinetics of vanadium precipitation for the vanadium solution from calcified vanadium extraction. J. Process Eng. 2018, 18, 111–117. (In Chinese) [Google Scholar]
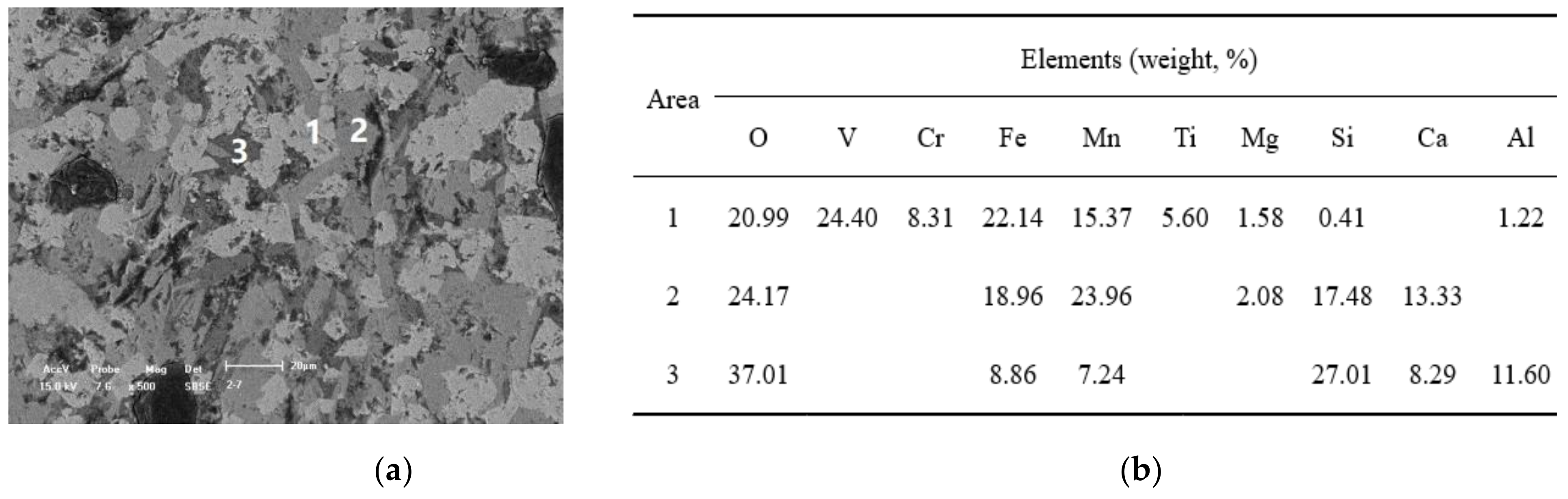
Figure 1.
Scanning electron microscope and energy dispersive spectrometer (SEM-EDS) analysis for the converter vanadium slag (
a) the scanning electron micrograph of the converter vanadium slag; (
b) the energy dispersive spectrum analysis for different areas in
Figure 1a.
Figure 2.
Flow chart for treating the converter vanadium slag.
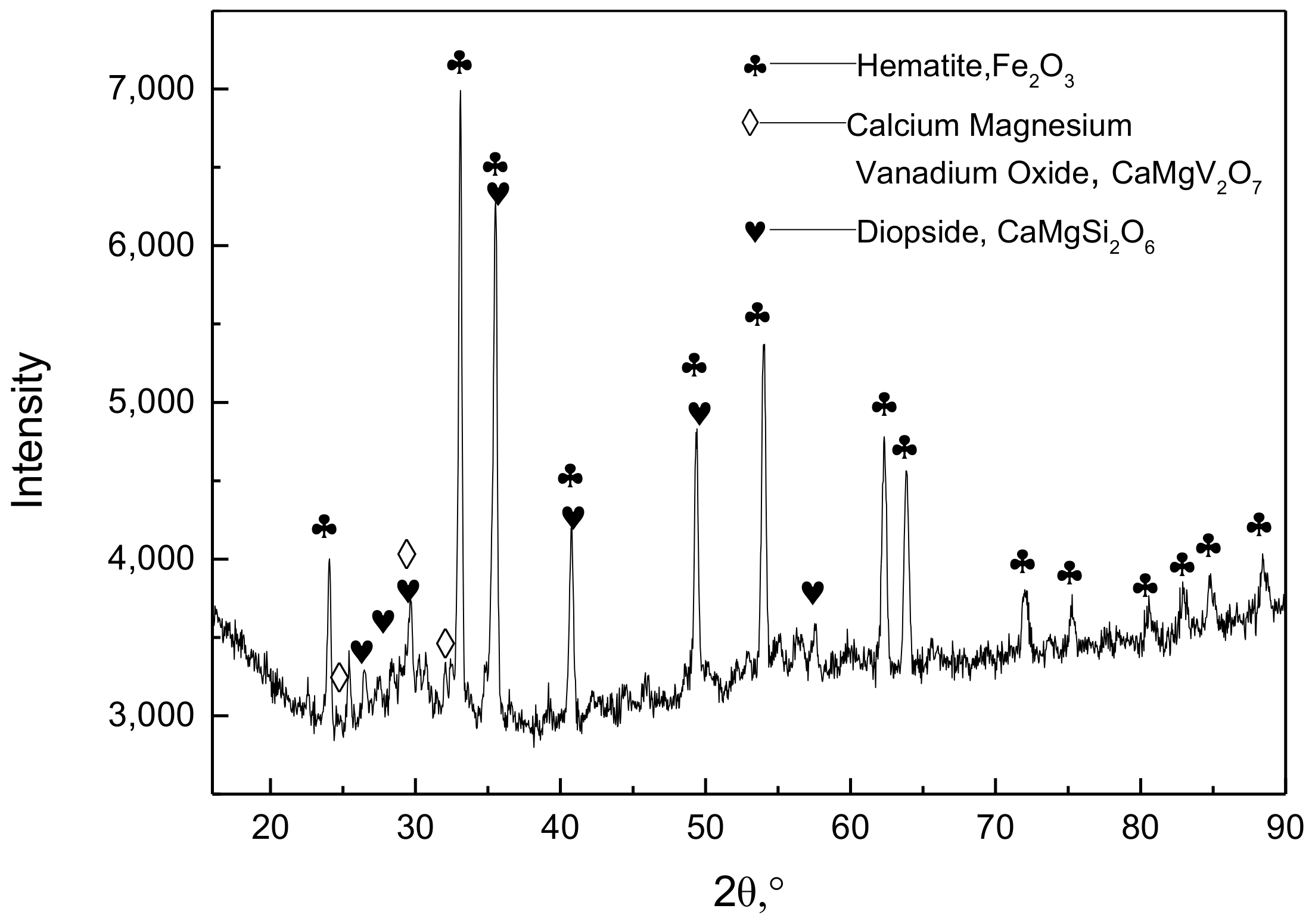
Figure 3.
X-ray diffraction pattern for vanadium slag roasted at 850 °C for 150 min.
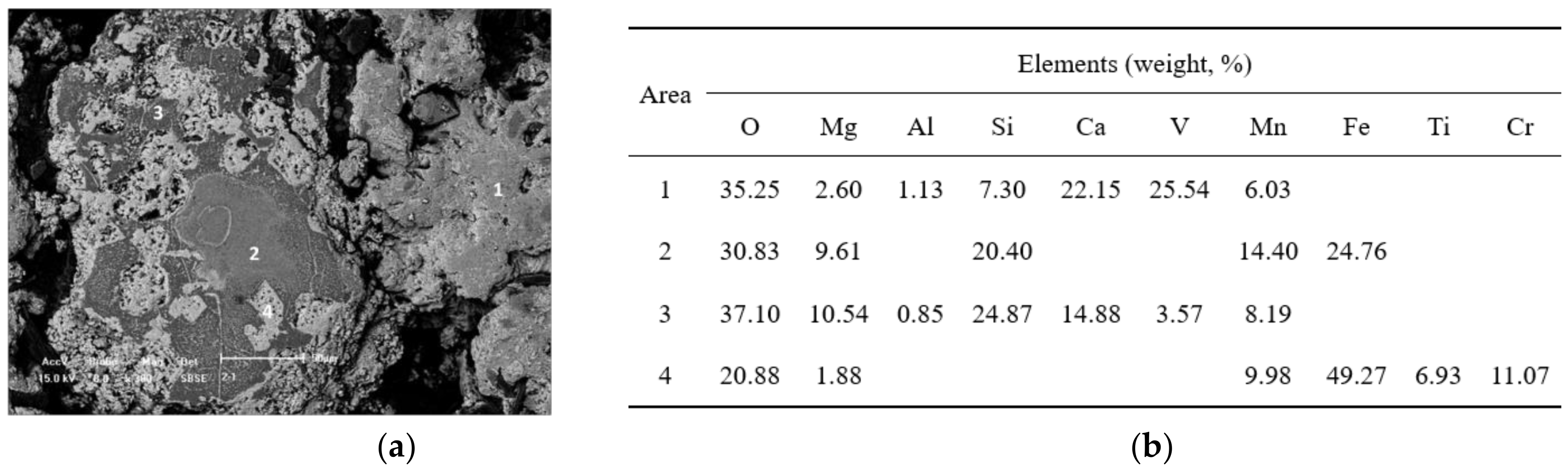
Figure 4.
SEM-EDS analysis for the sample roasted at 850 °C for 150 min (
a) the scanning electron micrograph of the roasted slag; (
b) the energy dispersive spectrum analysis for different areas in
Figure 4a.
Figure 5.
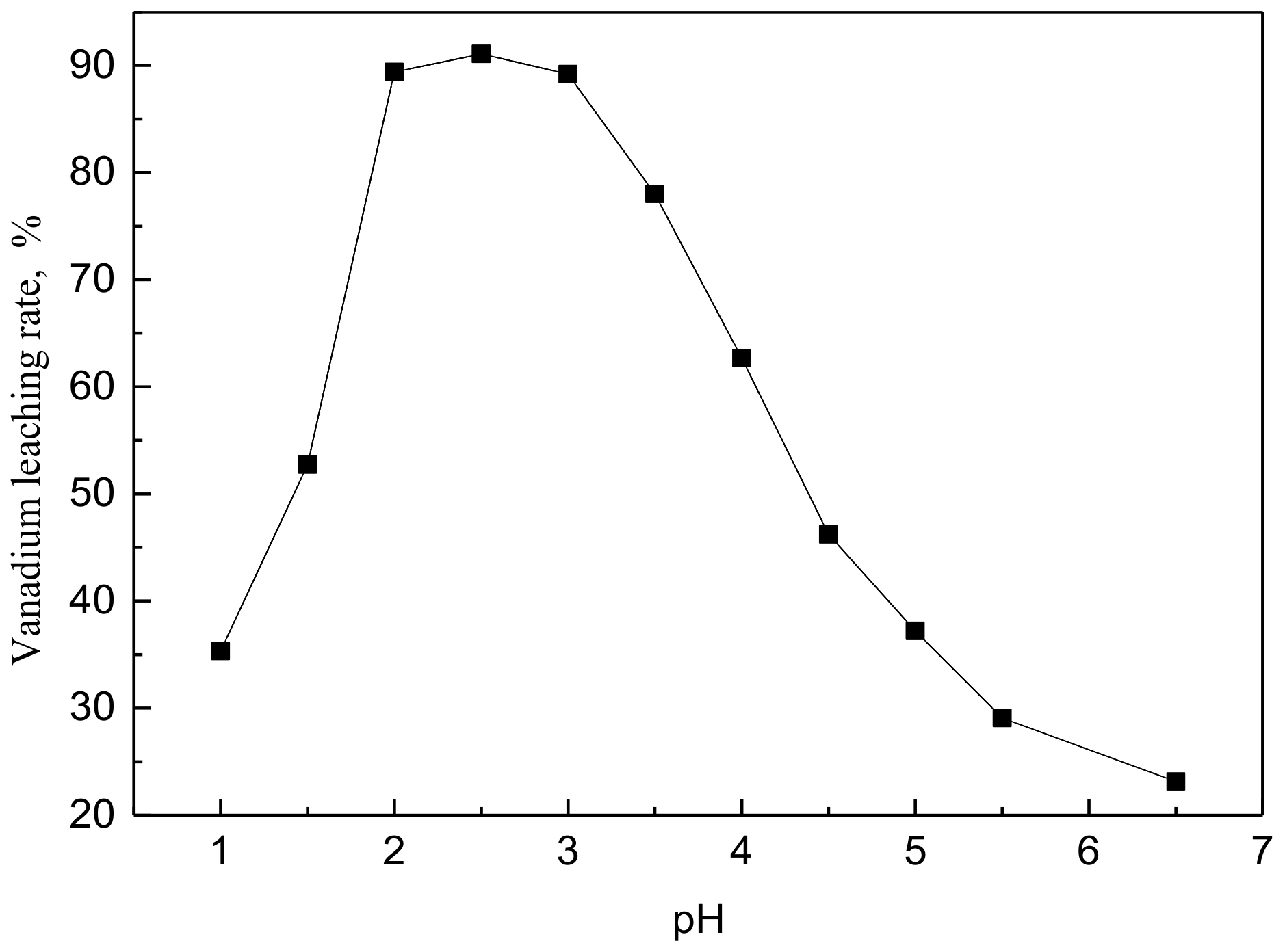
Effect of pH on the vanadium leaching rate (particle size of 75–96 μm, time of 60 min, T = 65 °C, L/S = 4, stirring speed of 500 rpm).
Figure 6.
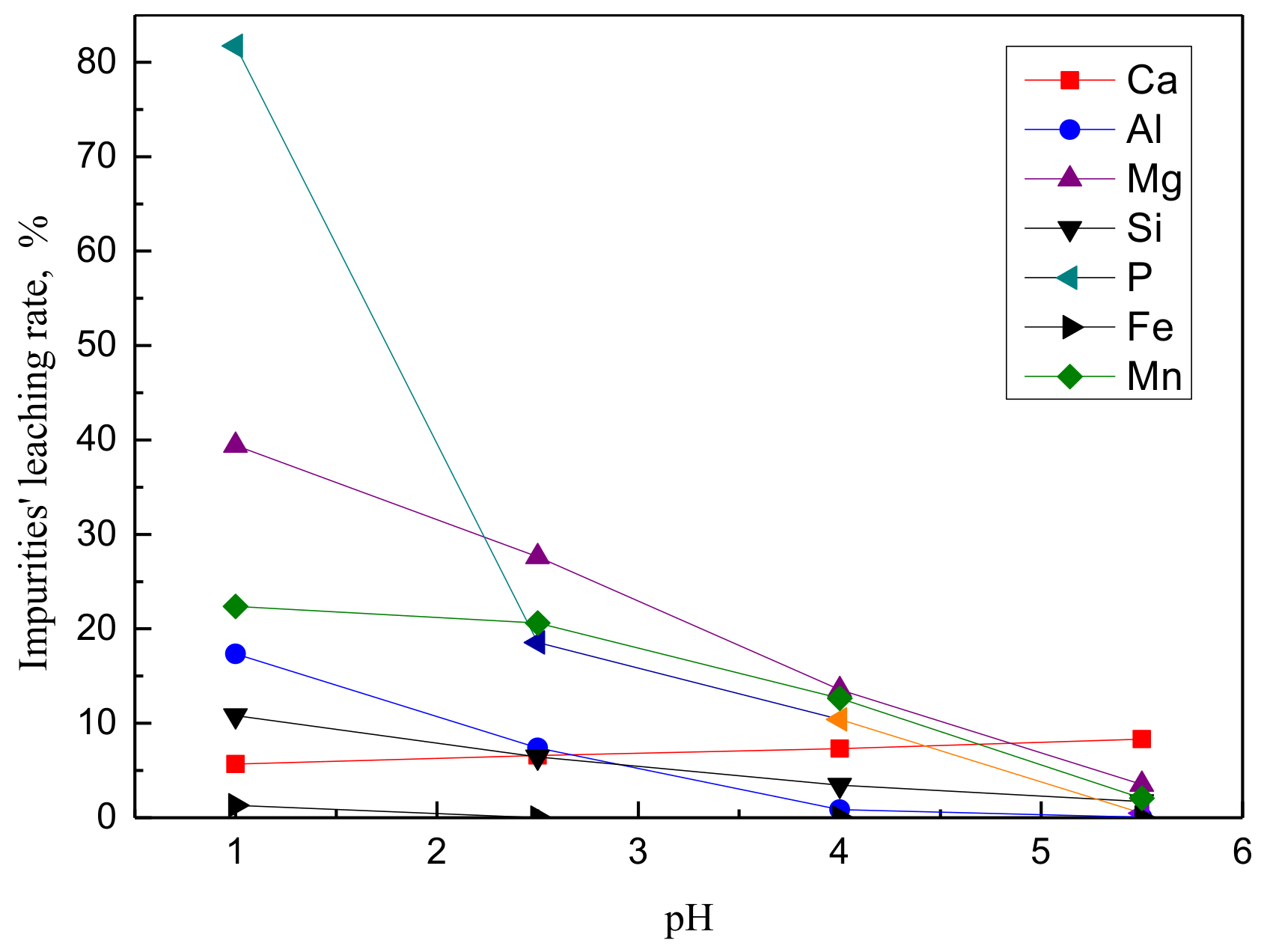
Effect of pH on the leaching rate of impurities (particle size of 75–96 μm, time of 60 min, T = 65 °C, L/S = 4, stirring speed of 500 rpm).
Figure 7.
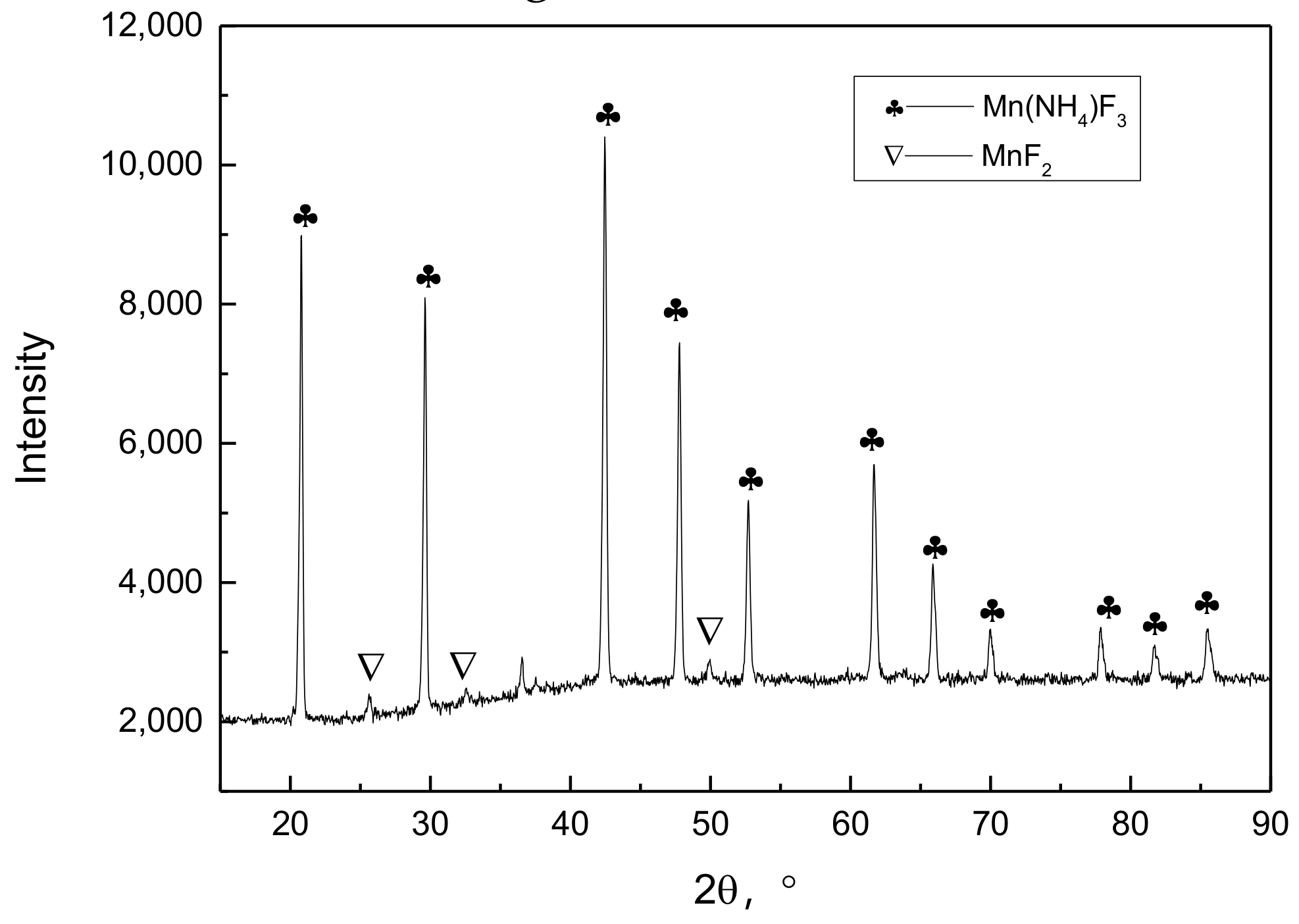
X-ray diffraction (XRD) pattern of the composite reagent for removing impurities.
Figure 8.
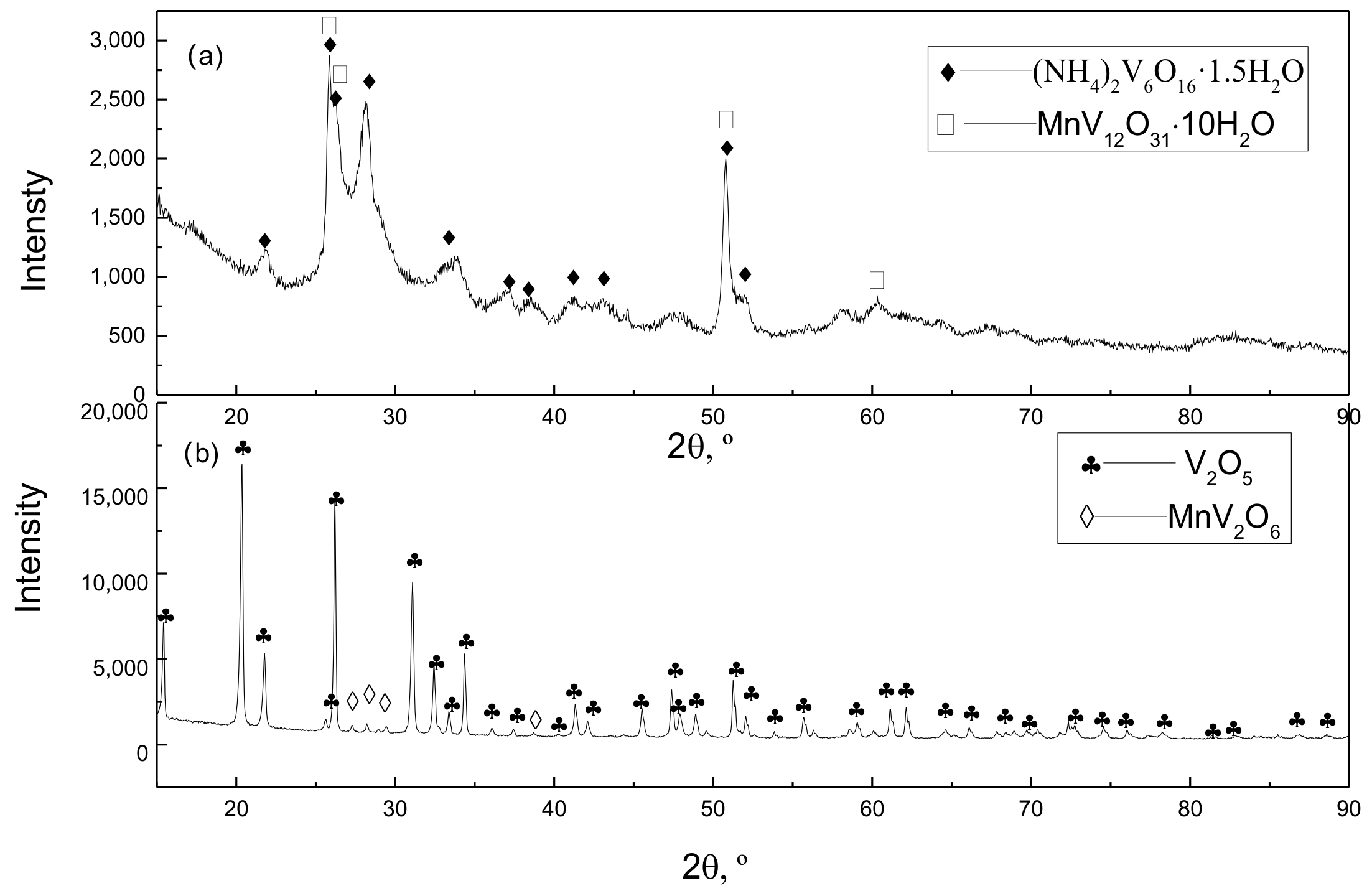
XRD pattern for products of vanadium precipitate (a) ammonium poly-vanadate; (b) vanadium pentoxide.
Figure 9.
Scanning electron micrographs for ammonium poly-vanadate and vanadium pentoxide powder, (a) ammonium poly-vanadate × 500; (b) ammonium poly-vanadate × 4,000; (c) vanadium pentoxide powder × 10,000.
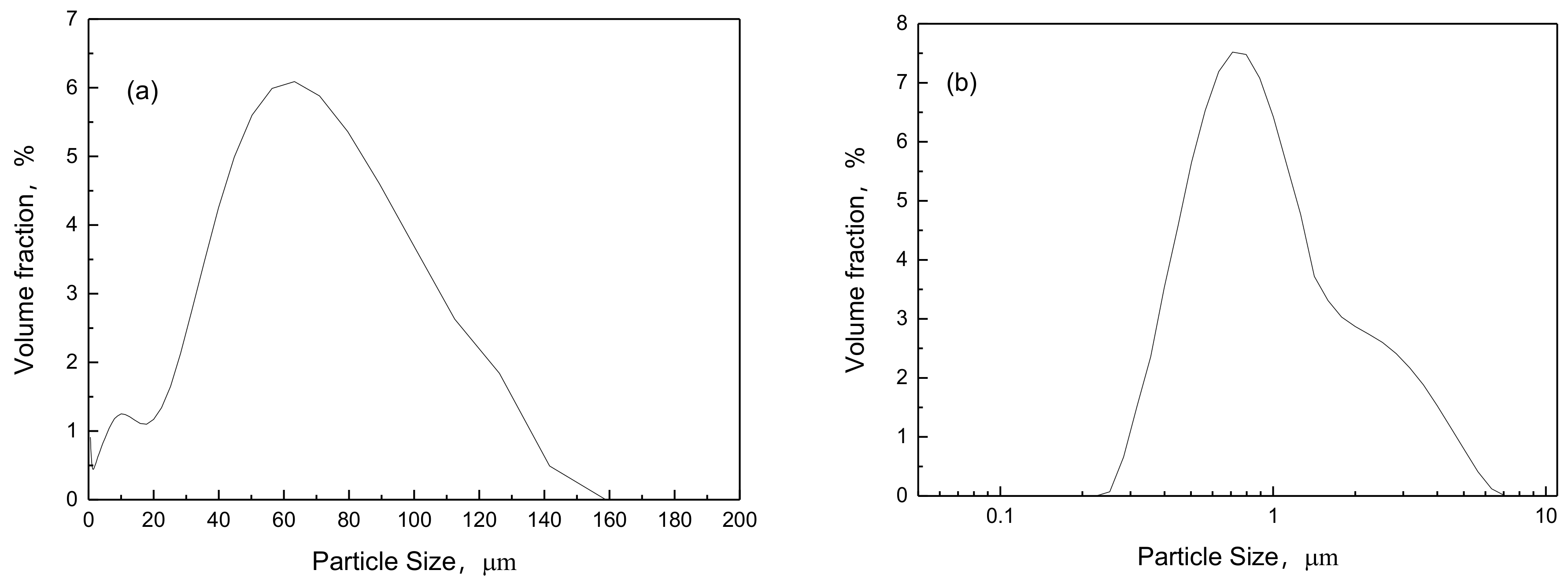
Figure 10.
Particle size distribution for ammonium poly-vanadate and vanadium pentoxide (a) ammonium poly-vanadate; (b) vanadium pentoxide.
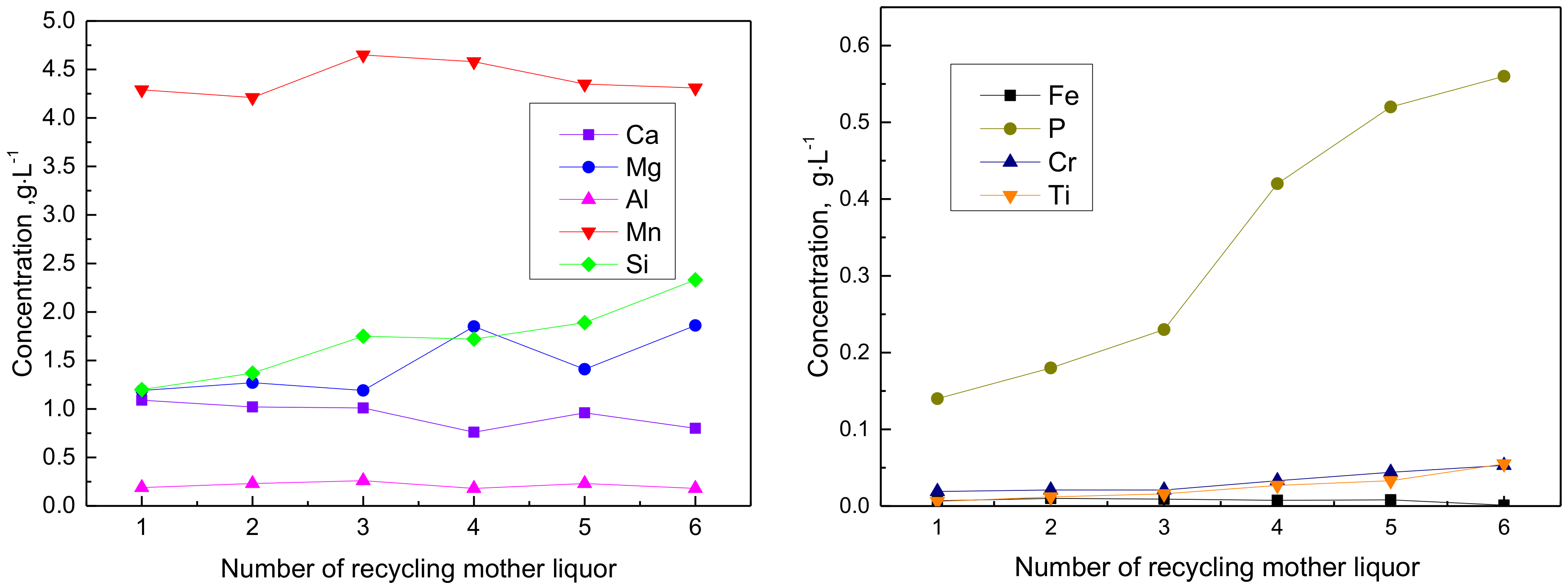
Figure 11.
Relationships between concentrations of impurities and the number of recycling mother liquor (a) variation of the concentration of main impurities of Ca, Mg, Al, Mn and Si as the recycle increases; (b) variation of the concentration of the minor impurities of Fe, P, Cr and Ti as the recycle increases.
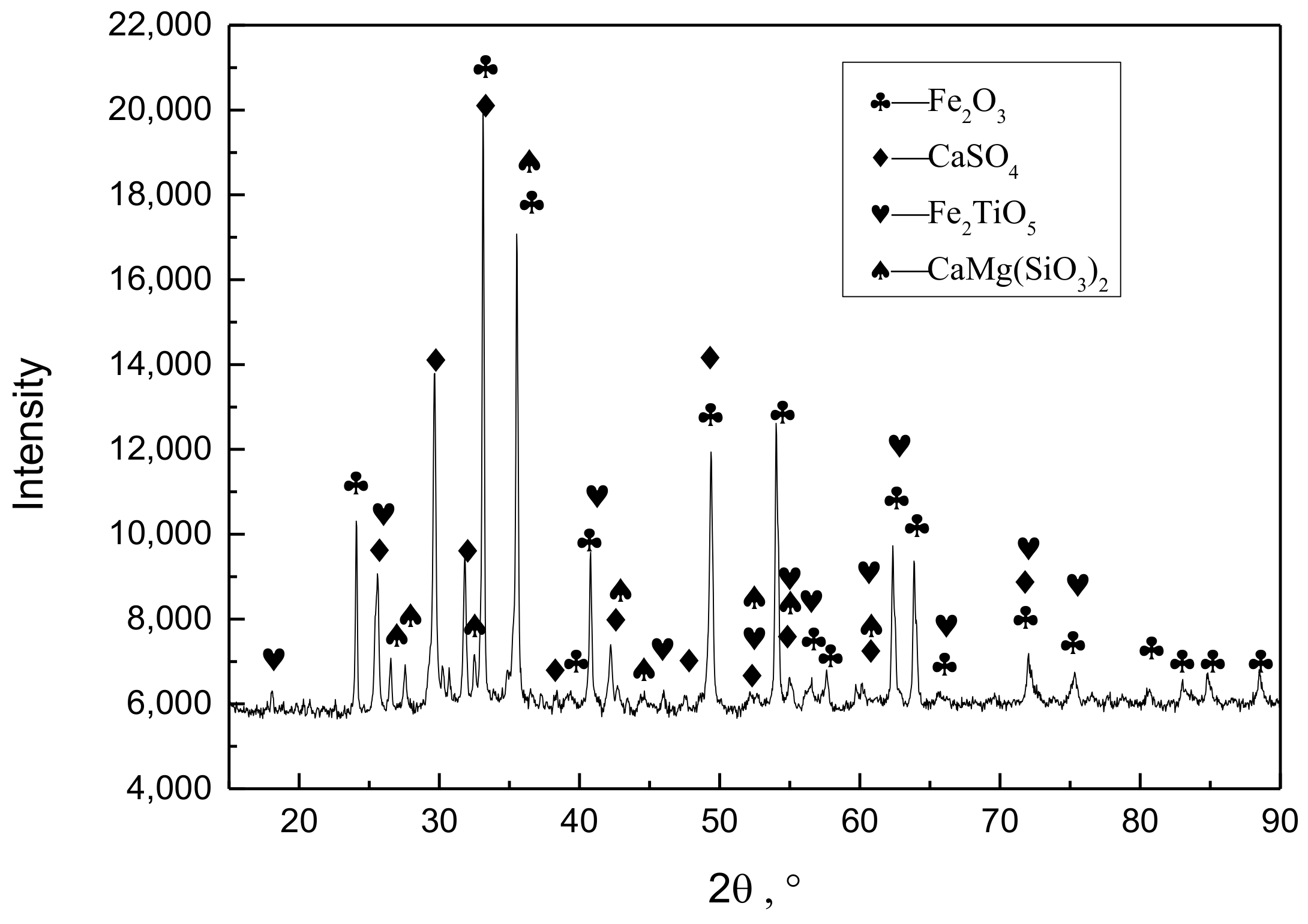
Figure 12.
XRD pattern for tailings of vanadium extraction.
Table 1.
Concentrations of V and main impurities contained in the of vanadium-bearing solution (g·L−1).
| Elements | V | Mn | Si | Al | Ca | Fe | Mg | Cr | Ti | P |
|---|
| Before Purification | 21.4 | 5.99 | 1.49 | 0.20 | 1.02 | 0.010 | 1.59 | 0.010 | 0.0057 | 0.12 |
| After Purification | 19.8 | 10.1 | 0.88 | 0.009 | 0.033 | 0.009 | 0.079 | 0.010 | 0.004 | 0.089 |
Table 2.
Chemical composition of the vanadium pentoxide product (wt. %).
| V2O5 | Mn | S | Si | Fe | Al | P | Ca | Na2O | K2O |
|---|
| 98.63 | 0.85 | 0.011 | 0.031 | 0.012 | 0.011 | 0.016 | 0.007 | - | 0.011 |
Table 3.
Concentrations of V and the main impurities contained in mother liquor after vanadium precipitation (g·L−1).
| V | Mn | Ca | Mg | Al | Ti | Cr | P | Si |
|---|
| 0.69 | 9.82 | 0.016 | 0.061 | 0.006 | 0.003 | 0.010 | 0.055 | 0.70 |
Table 4.
Effect of the number of recycling mother liquor on the quality of final product (wt. %).
| Number of Recycle | V2O5 | Mn | Si | Cr | Ti | Fe | S | Al | P | Ca | Mg | K2O |
|---|
| 1 | 98.73 | 0.39 | 0.052 | - | - | 0.022 | - | - | 0.014 | 0.020 | - | - |
| 2 | 98.63 | 0.73 | 0.023 | - | - | 0.024 | - | 0.010 | 0.021 | 0.034 | - | 0.012 |
| 3 | 98.04 | 0.92 | 0.079 | - | 0.035 | 0.019 | 0.007 | 0.011 | 0.022 | 0.011 | 0.005 | 0.020 |
| 4 | 97.30 | 1.09 | 0.21 | - | 0.060 | 0.019 | 0.044 | 0.038 | 0.078 | 0.011 | 0.011 | 0.018 |
| 5 | 91.65 | 0.53 | 2.41 | 0.164 | 0.114 | 0.024 | 0.132 | 0.035 | 0.308 | 0.064 | 0.040 | 0.031 |
| 6 | 88.00 | 0.50 | 4.55 | 0.137 | 0.258 | 0.021 | 0.196 | 0.024 | 0.279 | 0.050 | 0.034 | 0.033 |
Table 5.
Chemical composition of tailings after vanadium extraction with calcified roasting-acid leaching (wt. %).
| V2O5 | Fe2O3 | SiO2 | CaO | MnO | TiO2 | Cr2O3 | MgO | Al2O3 |
|---|
| 1.20 | 33.24 | 10.97 | 15.91 | 6.45 | 7.08 | 4.70 | 2.33 | 1.54 |
| P2O5 | SO3 | Nb2O5 | | | | | | |
| 0.49 | 16.05 | 0.047 | | | | | | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).