New Technology to Produce 1 GPa Low Carbon Microalloyed Steels from Cast Strip
Abstract
1. Introduction
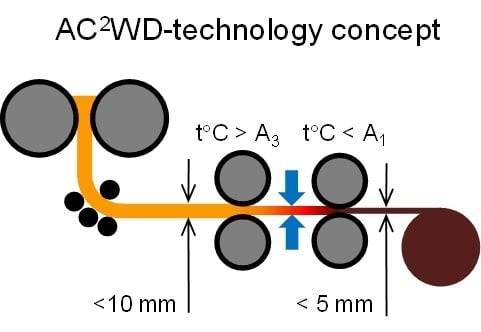
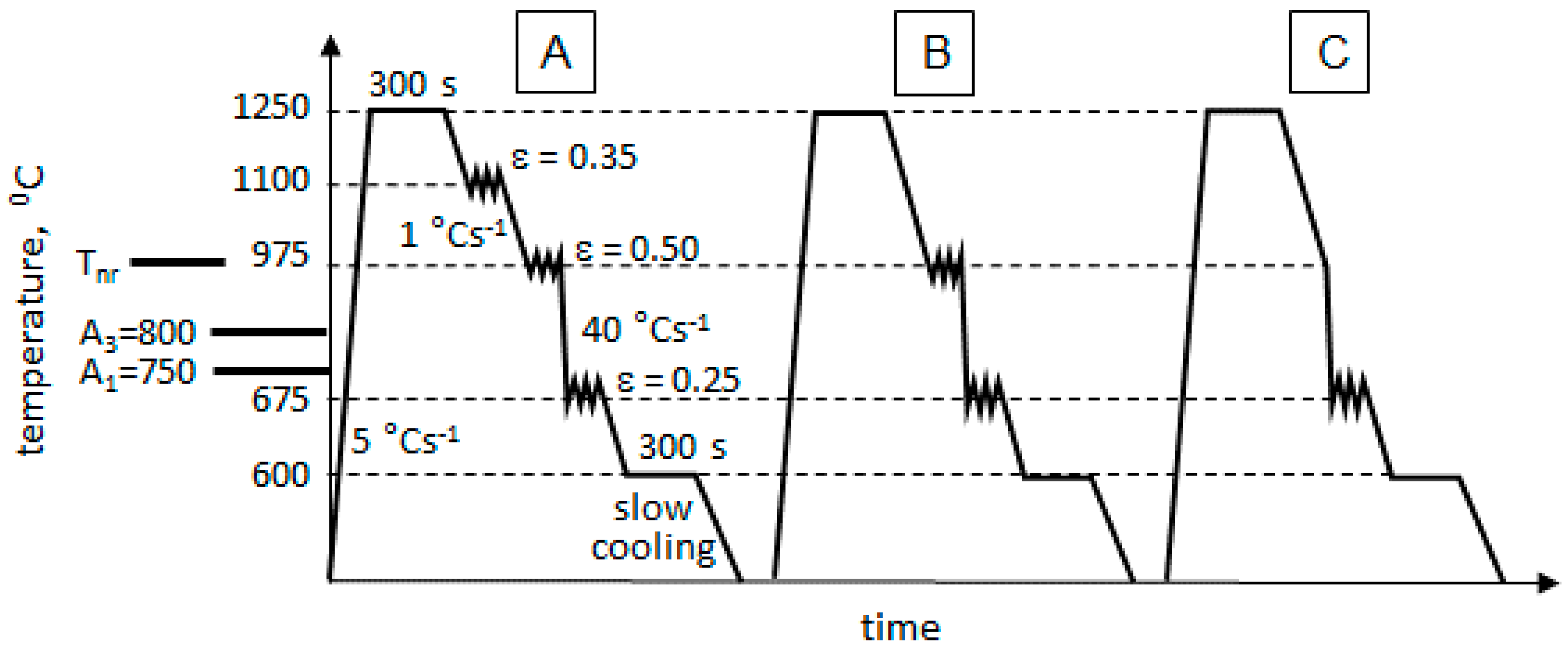
2. Technology Development
3. Effect of Processing on Microstructure
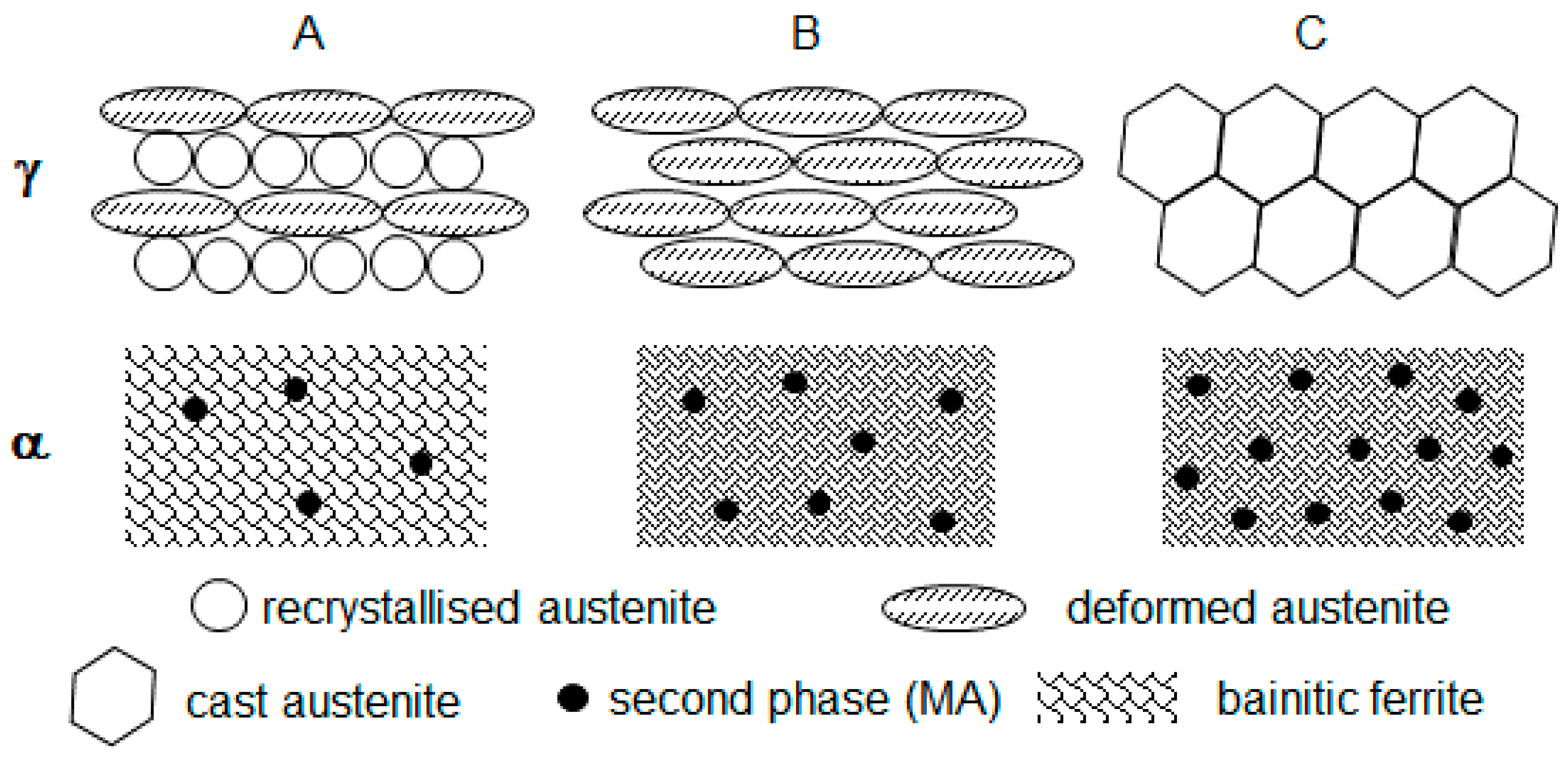
3.1. Grain Structure and Phase Balance
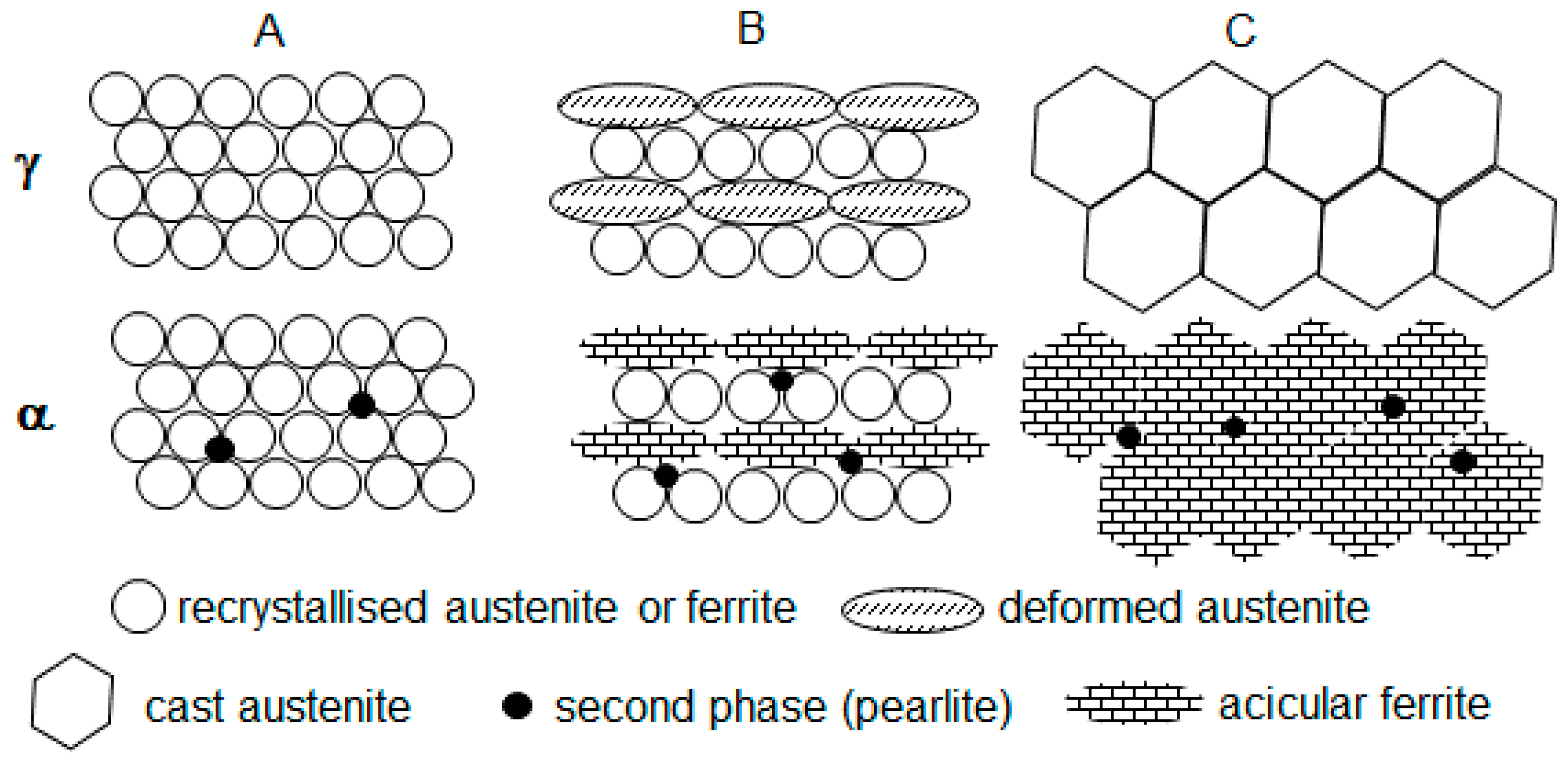
- Two high temperature deformations (route A) resulted in pronounced DRX and fine-grained austenite; during cooling the fine-grained austenite transformed to fine-grained ferrite (a substantial number of ferrite nucleation sites was determined by the large length of prior austenite grain boundaries); a significantly lower second phase fraction (2.5% in contrast to 10.9%, expected for 0.095 wt.% C on the basis of lever rule) formed as a result of accelerated cooling (40 C s−1); the ferrite solution was supersaturated with Carbon;
- One high temperature deformation (route B) resulted in partial DRX and formation of two types of austenite grains, fine recrystallised and large deformed non-recrystallised; during cooling the large deformed grains transformed to rather acicular ferrite grains (an increase in acicular ferrite fraction with an increase in austenite grain size was reported previously [48]) and fine recrystallised grains transformed to rather polygonal ferrite grains—this decreased the average measured value of ferrite grain size from 10 m for route A to 4.8 m for route B (Table 1); a slightly higher pearlite fraction, 3.2% for route B compared to 2.5% for route A, might be associated with slower strain induced precipitation of carbides and more Carbon available in solid solution for the pearlite reaction;
- Without high temperature deformation (route C), the large cast grains transformed to acicular ferrite (in addition to the effect of grain size, the austenite to ferrite transformation start temperature A3 could decrease with elimination of deformation [49,50], this would also facilitate the transformation to acicular ferrite instead of polygonal), although some polygonal ferrite formation could take place on austenite grain boundaries; a further minor increase in the pearlite fraction, from 3.2% for route B to 4.3% for route C, might be associated with an increase in remaining austenite fraction at the end of cooling, this austenite fraction would transform to pearlite during holding after the deformation (deformation was reported to facilitate the pearlite reaction [51]).
- In spite of two high temperature deformations (route A) DRX was, probably, partial; this would correspond to a higher recrystallization stop temperature of the MoNbTi-steel (975 °C) than this of the Ti-steel (900 °C) [52]; during cooling the austenite grains of all types were transforming to bainitic ferrite, as a result of Mo [39,40,41,42] and Nb [43,44,45,46,47] promoting the bainite transformation; the second phase MA constituent formed from the austenite retained after bainitic ferrite transformation; transformation to MA occurred during the final cooling to ambient temperature after the warm deformation cycle;
- One high temperature deformation (route B) did not create conditions for DRX (the deformation temperature of 975 °C was equal to Tnr); therefore, large deformed non-recrystallised grains dominated; during cooling, the dislocation structure retained in austenite facilitated transformation to a finer bainitic ferrite (1 m of bainitic ferrite area width for route B in contrast to 2.5 m for route A, Table 1) via an increased nucleation rate (pre-deformation was observed increasing the bainite transformation rate [53]); however, the rate of bainitic ferrite growth might be reduced because of retained dislocation structure (stronger austenite slower transforms to bainite [54]); this resulted in an increased volume fraction of the second phase MA, 7.7% for route B compared to 5.5% for route A (the bainite volume fraction was observed decreasing with strain [55]);
- Without high temperature deformation (route C) large cast grains sustained, although their size could be smaller in the MoNbV-steel than in the Ti-steel due to the retardation of grain growth by Mo [56,57] and Nb [58,59,60] solute atoms and precipitates; during cooling large non-deformed austenitic grains could faster transform to bainite, due to the austenite being softer, and the bainite transformation could start at a higher temperature [61]—this would favour an increase in the bainite volume fraction; however, the potential number of nucleation sites was lower, and thus, a decreased bainitic ferrite fraction was observed for route C (MA fraction increased to 13%, Table 1).
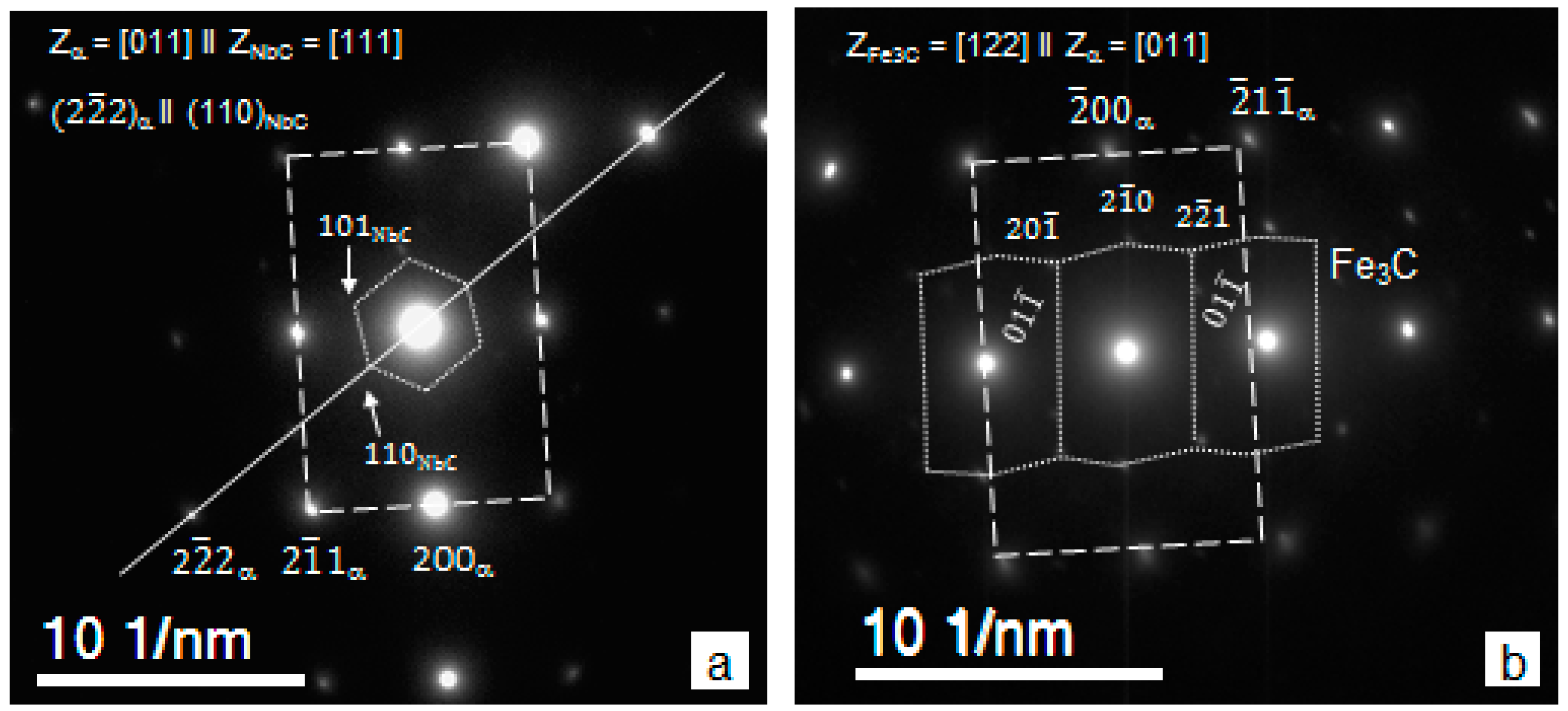
3.2. Particle Precipitation
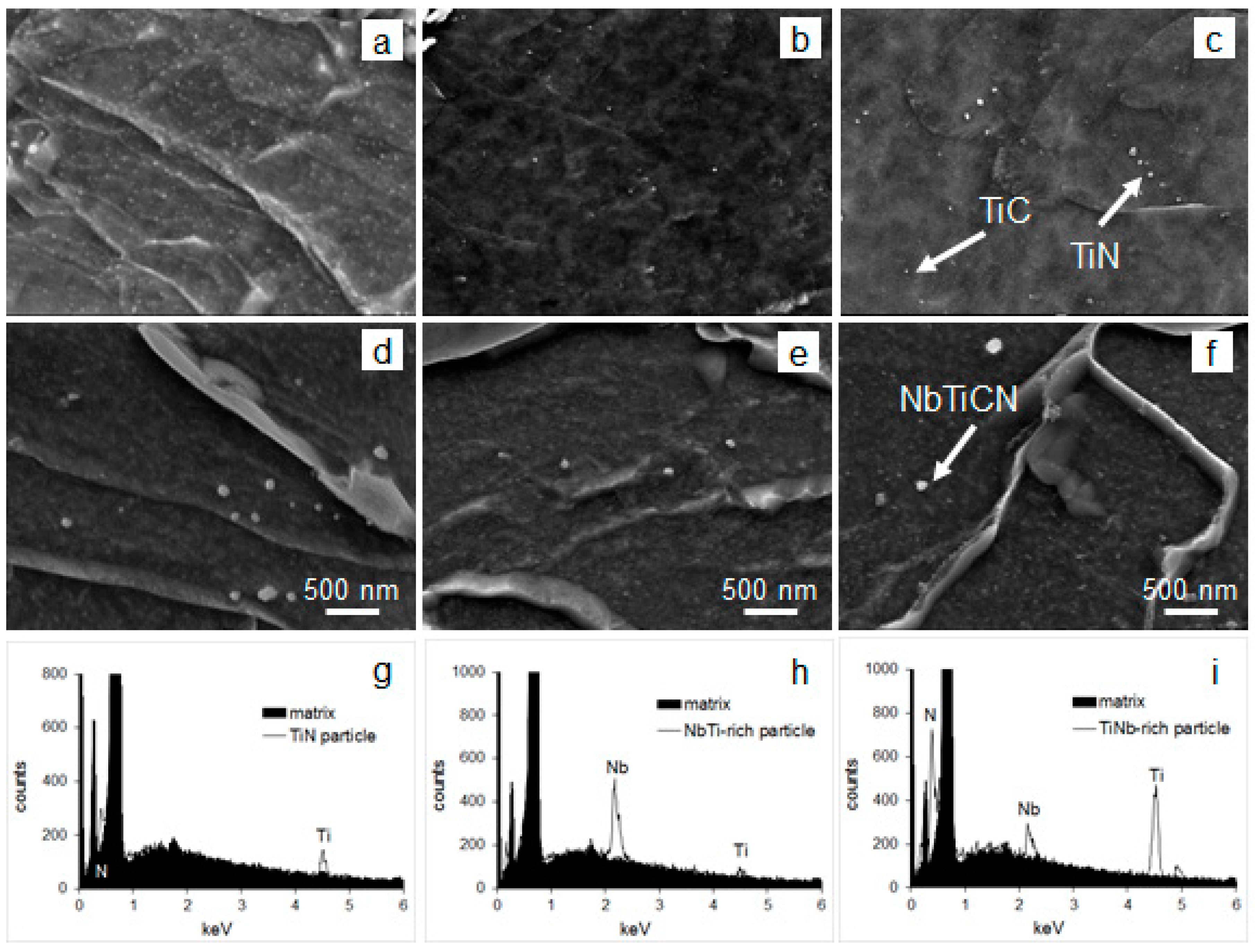
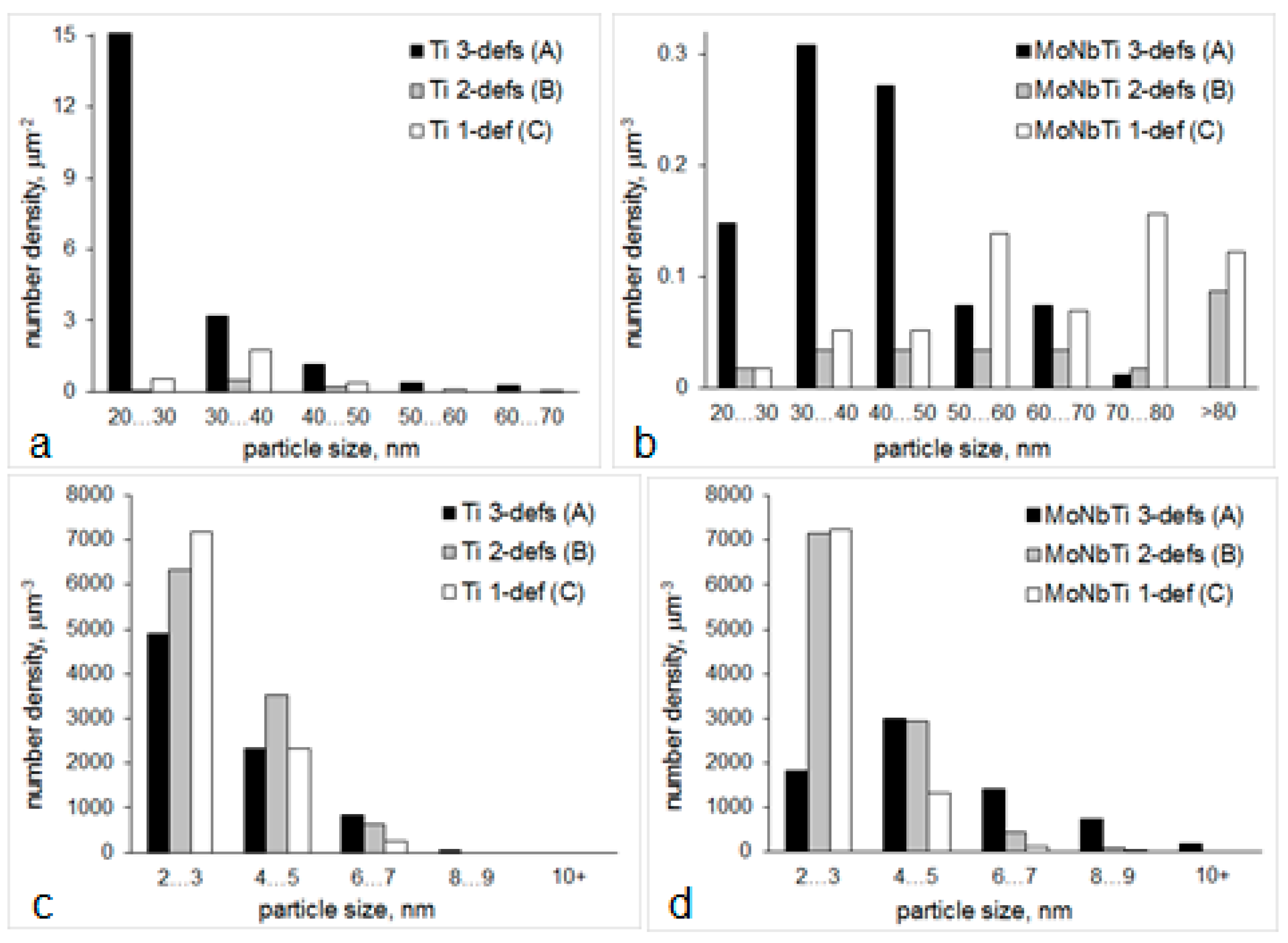
- The maximum of >20 nm particle number density and volume fraction, maximum of relative percentage of TiC in the >20 nm particle size range, absence of TiC in the <20 nm size range, and minimum of matrix unit cell size (associated with minimum concentration of solid solute atoms in the matrix [71]) were observed for route A. These data indicate a pronounced strain induced precipitation of TiC occurring in austenite when two high temperature deformations, at 1100 and 975 °C, were applied;
- With elimination of deformation at 1100 °C (route B), the >20 nm particle number density decreased by almost 30 times (note the particle size distribution variation in Figure 9a), the average particle size increased, relative amount of >20 nm TiC particles decreased to 25% (from 41% for route A), the <20 nm particle number density increased by 1.3 times (note the particle size distribution variation in Figure 9c), amongst <20 nm particles the TiC ones were identified in addition to Fe3C, the matrix unit cell size increased to 0.305 nm (from 0.296 nm for route A). These suggest a substantial decrease in strain-induced precipitation in austenite, compared to route A, with more Ti and C available for precipitation of <20 nm particles.
- Without high temperature deformation (route C) the number density of >20 nm particles increased due to static precipitation of TiN, although precipitation of TiC in austenite continued to decrease (note a decrease in the relative amount of TiC in the >20 nm particle size range), the number density and volume fraction of <20 nm particles slightly decreased (by about 6% compared to route B), although the number density of <20 nm particles was 22% higher than for route A, the matrix unit cell size was of intermediate value between routes A and B. These data mean that route C gives a “balanced distribution” of Ti between >20 nm, <20 nm particles and solid solution.
- The route A resulted in the highest number density of >20 nm particles, the largest fraction of NbC in the >20 nm particle size range, and the lowest number density of <20 nm particles, although the average size of <20 nm particles and their volume fraction were the highest compared to other routes (remember that <20 nm NbC precipitated in austenite). Similarly, to Ti-steel, these indicate a substation strain-induced precipitation in austenite for route A, in particular precipitation of NbC in addition to NbTiCN;
- With elimination of deformation at 1100 °C (route B) the number density of >20 nm particles decreased by thre times (Figure 9b), although the average size and fraction of TiNbCN increased; the number density of <20 nm particles increased by almost one and a half times (and by almost four times in the 2–3 nm size range, Figure 9d); the matrix unit cell size showed a certain decrease (which may follow a decrease in the solid solute concentrations). These data for route B support a slower strain-induced precipitation in austenite, growth of Ti-core particles via deposition of Nb containing shell, and quite significant precipitation of <20 nm particles (NbC in austenite and Fe3C in the ferrite temperature field);
- Without high temperature deformation (route C), the average size of >20 nm particles further increased, their area fraction increased by 2.7 times compared to route B, the relative amount of coarse TiNbCN continued to rise; the average size, number density and volume fraction of <20 nm particles decreased compared to route B (although the number density of 2–3 nm particles was as high as for route B), and the matrix unit cell size slightly increased. These mean that static precipitation of NbTiCN and growth of TiNbCN particles in austenite were significant, some static precipitation of NbC in austenite took place, Fe3C precipitation was significant, and the solid solution might be enriched in Mo, Nb and C.
3.3. Dislocation Structure
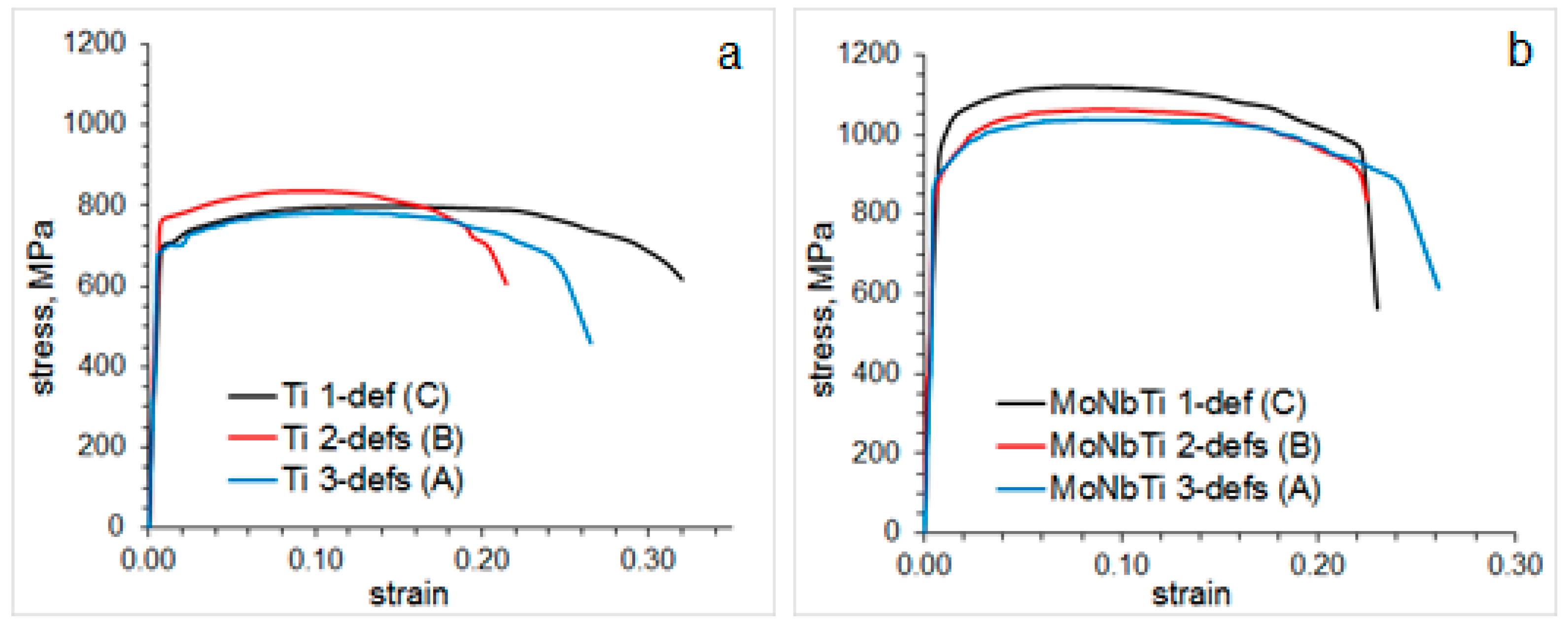
4. Effect of Processing on Mechanical Properties
5. Conclusions
- For the studied steel chemistry examples, new technology provided the yield stress in the range of 686–765 MPa and 880–950 MPa, ultimate tensile strength in the range of 783–836 MPa and 1037–1120 MPa, and simultaneously high elongation of 20–30% and 22–24% in the steels containing 0.095C-0.012Ti and 0.081C-0.1Mo-0.064Nb-0.021Ti (wt.%), respectively. This is 100–300 MPa superior to strength levels obtained using contemporary processing of steels with several times higher microalloying element contents. So remarkable property improvement can be related to minimal particle coarsening in austenite, optimum microalloying elements distribution between fine particles and solid solution, and formation of deformation bands and dislocation walls reducing the dislocation free-pass and contributing to the grain refinement effect.
- Processing route (in particular, two, one or no deformation passes in the austenite temperature field) leading to the maximum steel strength varied with steel composition. A lower microalloyed steel (Ti-steel) showed its maximum strength for the processing route with one pass of hot deformation of austenite and one pass of warm deformation of bainite. However, a higher microalloyed steel (MoNbTi-steel) showed its maximum strength for the processing route with no deformation of austenite. This is related to a variation in strengthening mechanisms in steels with different chemistry: in addition to grain refinement, critical for both compositions, precipitation strengthening was the second dominating in the Ti-steel, and the solid solution strengthening was the second in the MoNbTi-steel.
Author Contributions
Acknowledgments
Conflicts of Interest
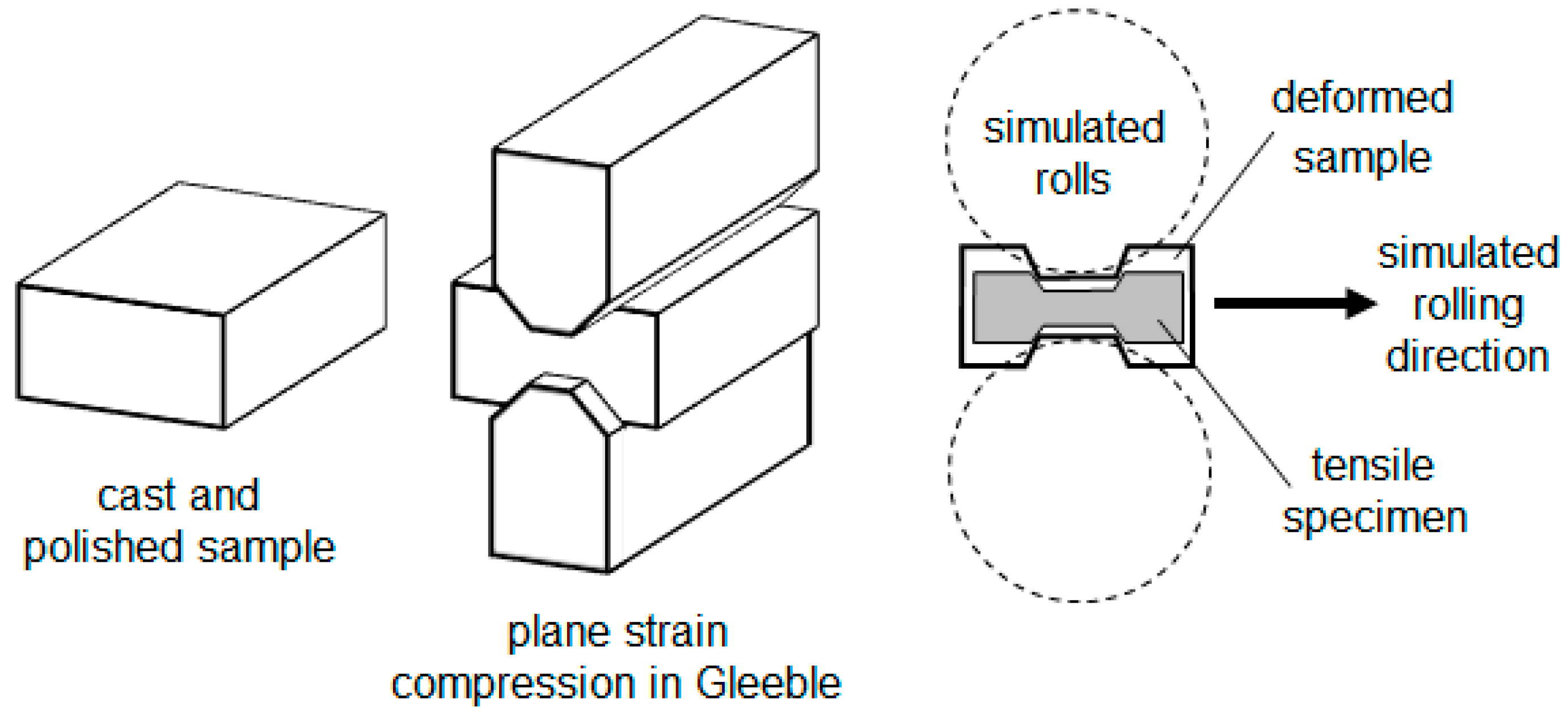
Appendix A. Materials and Methods

References
- Rankin, J. Energy Use in Metal Production. In Proceedings of the 4th Annual High Temperature Processing Symposium, Swinburne University, Melbourne, Australia, 6–7 February 2012. [Google Scholar]
- World Steel Association. Energy Use in the Steel Industry. Available online: http://www.worldsteel.org/publications/fact-sheets.html (accessed on 26 July 2017).
- Castrip LLC. The Castrip® Advantage. Available online: http://www.castrip.com/Advantage/advantage.html (accessed on 23 August 2018).
- Shimizu, Y.; Iwatani, J.; Hasai, J. Compact hot strip mill for high quality strip production. Mitsubishi Heavy Ind. Tech. Rev. 2000, 37, 48–51. [Google Scholar]
- Hensger, K.-E. Processing of advanced structural steels on CSP plants. Metalurgija 2002, 41, 183–190. [Google Scholar]
- DeArdo, A.J.; Marraccini, R.; Hua, M.J.; Garcia, C.I. Optimization of Nb HSLA microstructure using advanced thermomechanical processing in a CSP plant. Mater. Sci. Forum 2007, 539–543, 28–35. [Google Scholar] [CrossRef]
- Stubbles, J.R. The Minimill Story. Metall. Mater. Trans. B 2009, 40, 134–144. [Google Scholar] [CrossRef]
- Rosenthal, D.; Kraemer, S.; Klein, C.; Geerkens, C.; Mueller, J. 20 years of CSP: Success story of an extraordinary technology. Stahl und Eisen 2009, 129, 73–89. [Google Scholar]
- Patra, P.; Sam, S.; Singhai, M.; Kant, N. Study on the Production of Ultra High Strength Steel (UHSS) in Thin Slab Caster. SAE Tech. Pap. 2014. [Google Scholar] [CrossRef]
- Ge, S.; Isac, M.; Guthrie, R.I.L. Progress of strip casting technology for steel: Historical developments. ISIJ Int. 2012, 52, 2109–2122. [Google Scholar] [CrossRef]
- Killmore, C.R.; Edelman, D.G.; Carpenter, K.R.; Kaul, H.R.; Williams, J.G.; Campbell, P.C.; Blejde, W.N. Recent product developments with ultra-thin cast strip products produced by the CASTRIP® Process. Mater. Sci. Forum 2010, 654–656, 198–201. [Google Scholar] [CrossRef]
- Xie, K.Y.; Zheng, T.; Cairney, J.M.; Kaul, H.; Williams, J.G.; Barbaro, F.J.; Killmore, C.R.; Ringer, S.P. Strengthening from Nb-rich clusters in a Nb-microalloyed steel. Scr. Mater. 2012, 66, 710–713. [Google Scholar] [CrossRef]
- Wang, Z.; Carpenter, K.; Chen, Z.; Killmore, C. The effect of cooling rate and coiling temperature on the niobium retention in Ultra-Thin Cast Strip steel. Mater. Sci. Eng. A 2017, 700, 234–240. [Google Scholar] [CrossRef]
- Xiong, Z.P.; Kostryzhev, A.G.; Stanford, N.E.; Pereloma, E.V. Effect of deformation on microstructure and mechanical properties of dual phase steel produced via strip casting simulation. Mater. Sci. Eng. A 2016, 651, 291–305. [Google Scholar] [CrossRef]
- Xiong, Z.P.; Kostryzhev, A.G.; Chen, L.; Pereloma, E.V. Microstructure and mechanical properties of strip cast TRIP steel subjected to thermo-mechanical simulation. Mater. Sci. Eng. A 2016, 677, 356–366. [Google Scholar] [CrossRef]
- Xiong, Z.P.; Saleh, A.A.; Kostryzhev, A.G.; Pereloma, E.V. Strain-induced ferrite formation and its effect on mechanical properties of a dual phase steel produced using laboratory simulated strip casting. J. Alloys Compd. 2017, 721, 291–306. [Google Scholar] [CrossRef]
- Smith, Y.E.; Siebert, C.A. Continuous cooling transformation kinetics of thermomechanically worked low-carbon austenite. Metall. Trans. 1971, 2, 1711–1725. [Google Scholar]
- Priestner, R.; Biring, M.S. Transformation of low-carbon austenite after small plastic strains. Met. Sci. 1973, 7, 60–64. [Google Scholar] [CrossRef]
- Park, J.K.; Kim, K.H.; Chung, J.H.; Ok, S.Y. Deformation-induced asutenite to ferrite massive transformation in medium carbon steel. Metall. Mater. Trans. A 2008, 39, 235–242. [Google Scholar] [CrossRef]
- Song, R.; Ponge, D.; Raabe, D.; Speer, J.G.; Matlock, D.K. Overview of processing, microstructure and mechanical properties of ultrafine grained bcc steels. Mater. Sci. Eng. A 2006, 441, 1–17. [Google Scholar] [CrossRef]
- DeArdo, A.J. Metallurgical basis for thermomechanical processing of microalloyed steels. Ironmak. Steelmak. 2001, 28, 138–144. [Google Scholar] [CrossRef]
- Cizek, P.; Wynne, B.P.; Davies, C.H.J.; Muddle, B.C.; Hodgson, P.D. Effect of composition and austenite deformation on the transformation characteristics of low-carbon and ultralow-carbon microalloyed steels. Metall. Mater. Trans. A 2002, 33, 1331–1349. [Google Scholar] [CrossRef]
- Bakkaloğlu, A. Effect of processing parameters on the microstructure and properties of an Nb microalloyed steel. Mater. Lett. 2002, 56, 200–209. [Google Scholar] [CrossRef]
- Sun, L.; Wynne, B.P.; Palmiere, E.J. Effect of austenite deformation on recrystallization behaviour in an X70 microalloyed steel. Adv. Mater. Res. 2010, 89–91, 721–726. [Google Scholar] [CrossRef]
- Hodgson, P.D.; Hickson, M.R.; Gibbs, R.K. Ultrafine ferrite in low carbon steel. Scr. Mater. 1999, 40, 1179–1184. [Google Scholar] [CrossRef]
- Mabuchi, H.; Hasegawa, T.; Ishikawa, T. Metallurgical features of steel plates with ultra fine grains in the surface layers and their formation mechanism. ISIJ Int. 1999, 39, 477–485. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, R. Formation of ultra-fine grain structure of plain low carbon steel through deformation induced ferrite transformation. ISIJ Int. 2003, 43, 761–766. [Google Scholar] [CrossRef]
- Hong, S.C.; Lim, S.H.; Hong, H.S.; Lee, K.J.; Shin, D.H.; Lee, K.S. Effects of Nb on strain induced ferrite transformation in C-Mn steel. Mater. Sci. Eng. A 2003, 355, 241–248. [Google Scholar] [CrossRef]
- Wang, R.Z.; Lei, T.C. Dynamic recrystallization of ferrite in a low carbon steel during hot rolling in the (F + A) two-phase range. Scr. Metall. Mater. 1994, 31, 1193–1196. [Google Scholar] [CrossRef]
- Ohmori, A.; Torizuka, S.; Nagai, K.; Koseki, N.; Kogo, Y. Effect of deformation temperature and strain rate on evolution of ultrafine grained structure through single-pass large-strain warm deformation in a low carbon steel. Mater. Trans. 2004, 45, 2224–2231. [Google Scholar] [CrossRef]
- Song, R.; Ponge, D.; Raabe, D. Mechanical properties of an ultrafine grained C–Mn steel processed by warm deformation and annealing. Acta Mater. 2005, 53, 4881–4892. [Google Scholar] [CrossRef]
- Li, L.; Yang, W.; Sun, Z. Dynamic recrystallization of ferrite in a low-carbon steel. Metall. Mater. Trans. A 2006, 37, 609–619. [Google Scholar]
- Hou, H.; Chen, Q.; Liu, Q.; Dong, H. Grain refinement of a Nb–Ti microalloyed steel through heavy deformation controlled cooling. J. Mater. Process. Technol. 2003, 137, 173–176. [Google Scholar] [CrossRef]
- Park, K.-T.; Han, S.Y.; Ahn, B.D.; Shin, D.H.; Lee, Y.K.; Um, K.K. Ultrafine grained dual phase steel fabricated by equal channel angular pressing and subsequent intercritical annealing. Scr. Mater. 2004, 51, 909–913. [Google Scholar] [CrossRef]
- Ueji, R.; Tsuji, N.; Minamino, Y.; Koizumi, Y. Ultragrain refinement of plain low carbon steel by cold-rolling and annealing of martensite. Acta Mater. 2002, 50, 4177–4189. [Google Scholar] [CrossRef]
- Azevedo, G.; Barbosa, R.; Pereloma, E.V.; Santos, D.B. Development of an ultrafine grained ferrite in a low C–Mn and Nb–Ti microalloyed steels after warm torsion and intercritical annealing. Mater. Sci. Eng. A 2005, 402, 98–108. [Google Scholar] [CrossRef]
- Tsuji, N.; Ito, Y.; Saito, Y.; Minamino, Y. Strength and ductility of ultrafine grained aluminum and iron produced by ARB and annealing. Scr. Mater. 2002, 47, 893–899. [Google Scholar] [CrossRef]
- Kostryzhev, A.G.; Marenych, O.O. Superior mechanical properties of microalloyed steels processed via a new technology based on austenite conditioning followed by warm deformation. Mater. Sci. Eng. A 2017, 688, 16–19. [Google Scholar] [CrossRef]
- Aaronson, H.I.; Reynolds, W.T.; Purdy, G.R. The incomplete transformation phenomenon in steel. Metall. Mater. Trans. A 2006, 37, 1731–1745. [Google Scholar] [CrossRef]
- Humphreys, E.S.; Fletcher, H.A.; Hutchins, J.D.; Garratt-Reed, A.J.; Reynolds, W.T.; Aaronson, H.I.; Purdy, G.R.; Smith, G.D.W. Molybdenum accumulation at ferrite: Austenite interfaces during isothermal transformation of an Fe-0.24 pct C-0.93 pct Mo alloy. Metall. Mater. Trans. A 2004, 35, 1223–1235. [Google Scholar] [CrossRef]
- Kong, J.; Xie, C. Effect of molybdenum on continuous cooling bainite transformation of low-carbon microalloyed steel. Mater. Des. 2006, 27, 1169–1173. [Google Scholar] [CrossRef]
- Hu, H.; Xu, G.; Zhou, M.; Yuan, Q. Effect of Mo content on microstructure and property of low-carbon bainitic steels. Metals 2016, 6, 173. [Google Scholar] [CrossRef]
- Mintz, B.; Banerjee, J.R.; Banks, K.M. Regression equation for Ar3 temperature for coarse grained as cast steels. Ironmak. Steelmak. 2011, 38, 197–203. [Google Scholar] [CrossRef]
- Lee, Y.K.; Hong, J.-M.; Choi, C.S.; Lee, J.K. Continuous cooling transformation temperatures and microstructures of Niobium bearing microalloyed steels. Mater. Sci. Forum 2005, 475–479, 65–68. [Google Scholar]
- Chen, Y.; Zhang, D.; Liu, Y.; Li, H.; Xu, D. Effect of dissolution and precipitation of Nb on the formation of acicular ferrite/bainite ferrite in low-carbon HSLA steels. Mater. Charact. 2013, 84, 232–239. [Google Scholar] [CrossRef]
- Tamehiro, H.; Murata, M.; Habu, R.; Nagumo, M. Optimum microalloying of Niobium and Boron in HSLA Steel for thermomechanical processing. Trans. Iron Steel Inst. Jpn. 1987, 27, 120–129. [Google Scholar] [CrossRef]
- Carpenter, K.R.; Killmore, C.R. The effect of Nb on the continuous cooling transformation curves of ultra-thin strip CASTRIP© steels. Metals 2015, 5, 1857–1877. [Google Scholar] [CrossRef]
- Barbaro, F.J.; Krauklis, P.; Easterling, K.E. Formation of acicular ferrite at oxide particles in steels. Mater. Sci. Technol. 1989, 5, 1057–1068. [Google Scholar] [CrossRef]
- Tamura, I.; Ouchi, C.; Tanaka, T.; Sekine, H. Thermomechanical Processing of High Strength Low Alloy Steels; Butterworths: London, UK, 1988. [Google Scholar]
- Grajcar, A.; Opiela, M. Influence of plastic deformation on CCT-diagrams of low-carbon and medium-carbon TRIP-steels. J. Achiev. Mater. Manuf. Eng. 2008, 29, 71–78. [Google Scholar]
- Wang, X.; Du, L.; Xie, H.; Di, H.; Gu, D. Effect of deformation on continuous cooling phase transformation behaviors of 780 MPa Nb-Ti ultra-high strength steel. Steel Res. Int. 2011, 82, 1417–1424. [Google Scholar] [CrossRef]
- AlShahrani, A.M. The Effects of Niobium Segregation and Dynamic Recrystallisation on Microstructural Homogeneity of Microalloyed Steels. Ph.D. Thesis, University of Wollongong, Wollongong, Australia, 2013. [Google Scholar]
- Lambers, H.-G.; Tschumak, S.; Maier, H.J.; Canading, D. Role of austenitization and pre-deformation on the linetics of the isothermal bainitic transformation. Metall. Mater. Trans. A 2009, 40, 1355–1366. [Google Scholar] [CrossRef]
- Bhadeshia, H.K.D.H. Bainite in Steels; IOM Communications: London, UK, 2001; pp. 23–25. [Google Scholar]
- Larn, R.H.; Yang, J.R. The effect of compressive deformation of austenite on the bainitic ferrite transformation in Fe-Mn-Si-C steels. Mater. Sci. Eng. A 2000, 278, 278–291. [Google Scholar] [CrossRef]
- Andrade, H.L.; Akben, M.G.; Jonas, J.J. Effect of molybdenum, niobium, and vanadium on static recovery and recrystallization and on solute strengthening in microalloyed steels. Metall. Trans. A 1983, 14, 1967–1977. [Google Scholar] [CrossRef]
- Schambron, T.; Chen, L.; Gooch, T.; Dehghan-Manshadi, A.; Pereloma, E.V. Effect of Mo concentration on dynamic recrystallization behavior of low carbon microalloyed steels. Steel Res. Int. 2013, 84, 1191–1195. [Google Scholar] [CrossRef]
- Kwon, O.; DeArdo, A.J. Interactions between recrystallization and precipitation in hot-deformed microalloyed steels. Acta Metall. Mater. 1991, 39, 529–538. [Google Scholar] [CrossRef]
- Medina, S.F.; Quispe, A. Improved model for static recrystallization kinetics of hot deformed austenite in low alloy and Nb/V microalloyed steels. ISIJ Int. 2001, 41, 774–781. [Google Scholar] [CrossRef]
- Vervynckt, S.; Verbeken, K.; Thibaux, P.; Liebeherr, M.; Houbaert, Y. Austenite recrystallization—precipitation interaction in Niobium microalloyed steels. ISIJ Int. 2009, 49, 911–920. [Google Scholar] [CrossRef]
- Kang, S.; Yoon, S.; Lee, S.-J. Prediction of bainite start temperature in alloy steels with different grain sizes. ISIJ Int. 2014, 54, 997–999. [Google Scholar] [CrossRef]
- Teresiak, A.; Kubsch, H. X-ray investigations of high energy ball milled transition metal carbides. Nanostruct. Mater. 1995, 6, 671–674. [Google Scholar] [CrossRef]
- Nartowski, A.M.; Parkin, I.P.; MacKenzie, M.; Craven, A.J.; MacLeod, I. Solid state metathesis routes to transition metal carbides. J. Mater. Chem. 1999, 9, 1275–1281. [Google Scholar] [CrossRef]
- Morales, E.V.; Gallego, J.; Kestenbach, H.-J. On coherent carbonitride precipitation in commercial microalloyed steels. Philos. Mag. Lett. 2003, 83, 79–87. [Google Scholar] [CrossRef]
- Fors, D.H.R.; Wahnström, G. Theoretical study of interface structure and energetics in semicoherent Fe(001)/MX(001) systems (M = Sc, Ti, V, Cr, Zr, Nb, Hf, Ta; X = C or N). Phys. Rev. B 2010, 82, 195410. [Google Scholar] [CrossRef]
- Tirumalasetty, G.K.; van Huis, M.A.; Fang, C.M.; Xu, Q.; Tichelaar, F.D.; Hanlon, D.N.; Sietsma, J.; Zandbergen, H.W. Characterization of NbC and (Nb, Ti)N nanoprecipitates in TRIP assisted multiphase steels. Acta Mater. 2011, 59, 7406–7415. [Google Scholar] [CrossRef]
- He, Y.; Godet, S.; Jonas, J.J. Observations of the Gibeon meteorite and the inverse Greninger–Troiano orientation relationship. J. Appl. Cryst. 2006, 39, 72–81. [Google Scholar] [CrossRef]
- Shiozawa, H.; Bachmatiuk, A.; Stangl, A.; Cox, D.C.; Silva, S.R.; Rümmeli, M.H.; Pichler, T. Microscopic insight into the bilateral formation of carbon spirals from a symmetric iron core. Sci. Rep. 2013, 3, 1840. [Google Scholar] [CrossRef] [PubMed]
- Kostryzhev, A.G.; Slater, C.D.; Marenych, O.O.; Davis, C.L. Effect of solidification rate on microstructure evolution in dual phase microalloyed steel. Sci. Rep. 2016, 6, 35715. [Google Scholar] [CrossRef] [PubMed]
- Andrews, K.W. Tabulation of interplanar spacings of cementite Fe3C. Acta Crystallogr. 1963, 16, 68. [Google Scholar] [CrossRef]
- Agrawal, B.K. Introduction to Engineering Materials; Tata McGraw-Hill: New Delhi, India, 1988. [Google Scholar]
- Mukherjee, S.; Timokhina, I.; Zhu, C.; Ringer, S.P.; Hodgson, P.D. Clustering and precipitation processes in a ferritic titanium-molybdenum microalloyed steel. J. Alloys Compd. 2017, 690, 621–632. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, H.; Guo, C.; Liu, W.; Yang, Z.; Sun, X.; Zhang, Z.; Jiang, F. Effect of molybdenum addition on the precipitation of carbides in the austenite matrix of titanium micro-alloyed steels. J. Mater. Sci. 2016, 51, 4996–5007. [Google Scholar] [CrossRef]
- Akben, M.G.; Bacroix, B.; Jonas, J.J. Effect of vanadium and molybdenum addition on high temperature recovery, recrystallization and precipitation behaviour of niobium-based microalloyed steels. Acta Metall. 1983, 31, 161–174. [Google Scholar] [CrossRef]
- Yi, H.-L.; Liu, Z.-Y.; Wang, G.-D.; Wu, D. Development of Ti-Microalloyed 600 MPa Hot Rolled High Strength Steel. J. Iron Steel Res. Int. 2010, 17, 54–58. [Google Scholar] [CrossRef]
- Xia, J.; Huo, X.; Li, L.; Peng, Z.; Chen, S. Development of Ti microalloyed high strength steel plate by controlling thermo-mechanical control process schedule. Mater. Res. Express 2017, 4, 126504. [Google Scholar] [CrossRef]
- Tang, S.; Liu, Z.Y.; Wang, G.D.; Misra, R.D.K. Microstructural evolution and mechanical properties of high strength microalloyed steels: Ultra Fast Cooling (UFC) versus Accelerated Cooling (ACC). Mater. Sci. Eng. A 2013, 580, 257–265. [Google Scholar] [CrossRef]
- Funakawa, Y.; Shiozaki, T.; Tomita, K.; Yamamoto, T.; Maeda, E. Development of high strength hot-rolled sheet steel consisting of ferrite and nanometer-sized carbides. ISIJ Int. 2004, 44, 1945–1951. [Google Scholar] [CrossRef]





| Parameters | Ti-Steel | MoNbTi-Steel | |||||
|---|---|---|---|---|---|---|---|
| A | B | C | A | B | C | ||
| Optical and scanning electron microscopy | |||||||
| ferrite grain size/ bainitic ferrite area width (m) | 10.0 ± 6.4 | 4.8 ± 3.0 | 4.7 ± 2.5 | 2.5 ± 1.8 | 1.0 ± 0.5 | 0.9 ± 0.4 | |
| second phase | average size (m) | 3.3 | 1.7 | 1.9 | 1.2 | 1.1 | 1.8 |
| Fraction (%) | 2.5 | 3.2 | 4.3 | 5.5 | 7.7 | 13.0 | |
| Scanning electron microscopy | |||||||
| >20 nm particles | average size (nm) | 24 ± 8 | 30 ± 6 | 30 ± 10 | 37 ± 12 | 66 ± 26 | 71 ± 26 |
| number density (µm−2) | 20.1 | 0.7 | 2.8 | 0.9 | 0.3 | 0.6 | |
| area fraction | 0.0097 | 0.0005 | 0.0022 | 0.0010 | 0.0010 | 0.0027 | |
| chemistry | 59%TiN 41%TiC | 75%TiN 25%TiC | 87%TiN 13%TiC | 38%TiNb 43%NbTi 19%NbC | 62%TiNb 38%NbTi | 83%TiNb 17%NbTi | |
| Transmission electron microscopy | |||||||
| <20 nm particles | average size (nm) | 3.4 ± 1.2 | 3.0 ± 1.1 | 2.8 ± 0.9 | 4.6 ± 1.9 | 3.0 ± 1.1 | 2.5 ± 0.9 |
| number density (µm−3) | 8098 | 10502 | 9888 | 7154 | 10604 | 8721 | |
| volume fraction | 0.00024 | 0.00023 | 0.00015 | 0.00057 | 0.00022 | 0.00011 | |
| chemistry | Fe3C | TiC Fe3C | TiC Fe3C | NbC Fe3C | NbC Fe3C | NbC Fe3C | |
| Matrix unit cell size (nm) | 0.296 | 0.305 | 0.302 | 0.298 | 0.294 | 0.302 | |
| Dislocation density (1014 m−2) | 1.9 ± 0.2 | 2.6 ± 0.2 | 2.0 ± 0.3 | 4.2 ± 0.3 | 3.2 ± 0.3 | 4.3 ± 0.2 | |
| Parameters | Ti-Steel | MoNbTi-Steel | ||||
|---|---|---|---|---|---|---|
| A | B | C | A | B | C | |
| YS (Mpa) | 686 | 765 | 700 | 880 | 900 | 950 |
| UTS (Mpa) | 783 | 836 | 800 | 1037 | 1062 | 1120 |
| Elongation (%) | 24 | 20 | 30 | 24 | 22 | 22 |
| Toughness # (MPa%) | 18,792 | 16,720 | 24,000 | 24,888 | 23,364 | 24,640 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kostryzhev, A.; Marenych, O. New Technology to Produce 1 GPa Low Carbon Microalloyed Steels from Cast Strip. Metals 2018, 8, 662. https://doi.org/10.3390/met8090662
Kostryzhev A, Marenych O. New Technology to Produce 1 GPa Low Carbon Microalloyed Steels from Cast Strip. Metals. 2018; 8(9):662. https://doi.org/10.3390/met8090662
Chicago/Turabian StyleKostryzhev, Andrii, and Olexandra Marenych. 2018. "New Technology to Produce 1 GPa Low Carbon Microalloyed Steels from Cast Strip" Metals 8, no. 9: 662. https://doi.org/10.3390/met8090662
APA StyleKostryzhev, A., & Marenych, O. (2018). New Technology to Produce 1 GPa Low Carbon Microalloyed Steels from Cast Strip. Metals, 8(9), 662. https://doi.org/10.3390/met8090662