The Aqueous Electrochemical Response of TiC–Stainless Steel Cermets
Abstract
:1. Introduction
2. Experimental Procedure
2.1. Sample Preparation and Characterisation
2.2. Electrochemical Testing
3. Results and Discussion
3.1. Microstructural Development
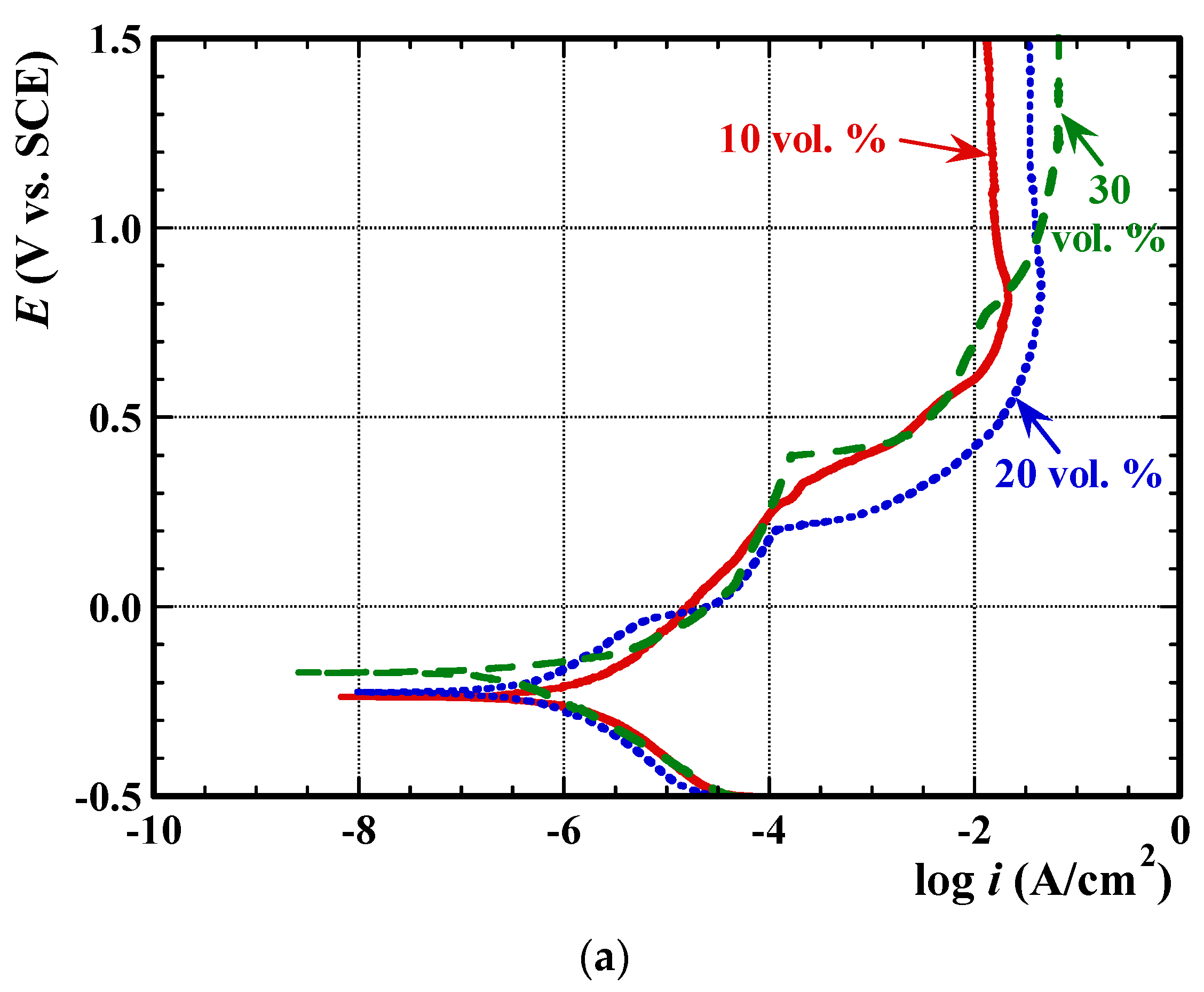
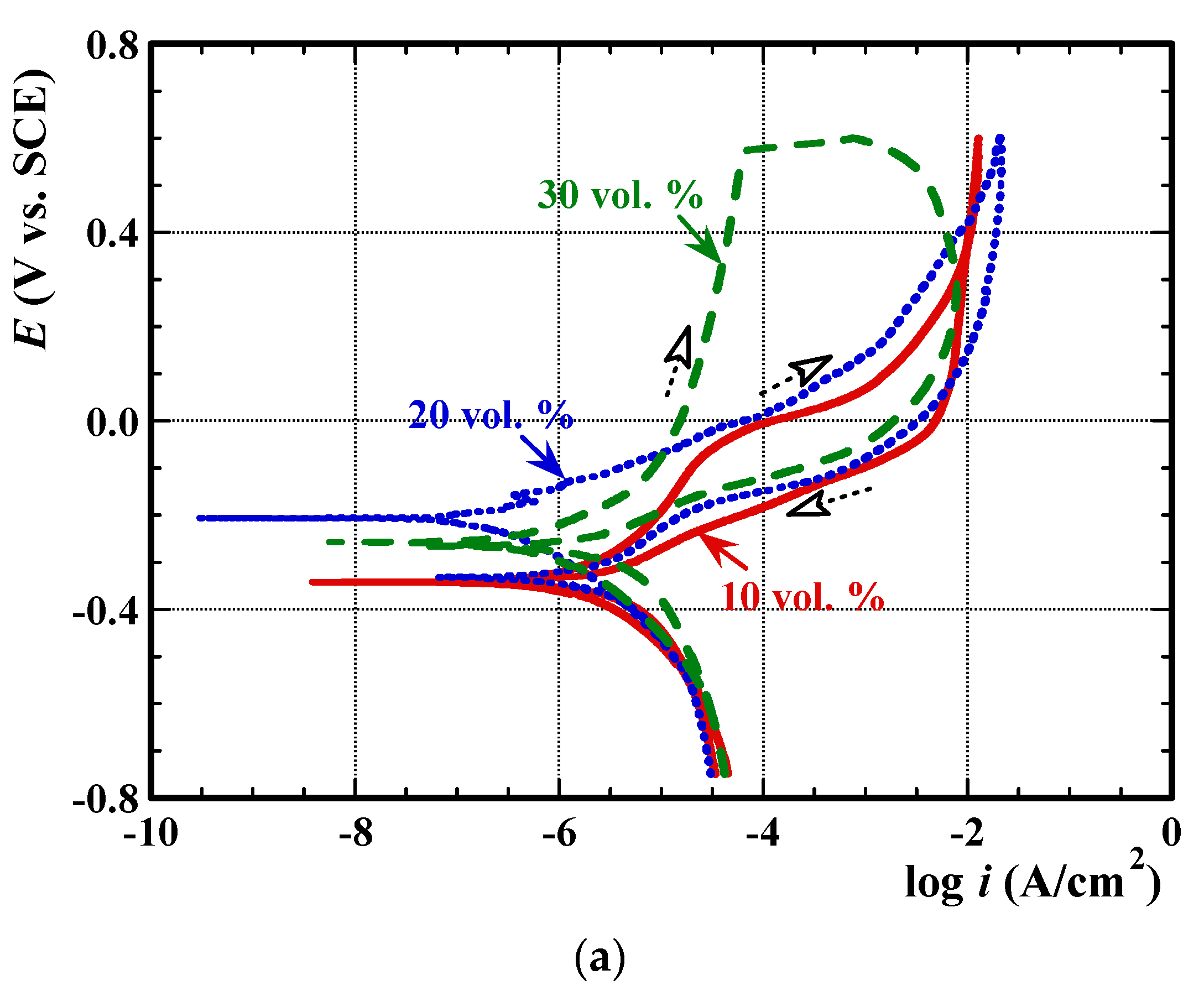
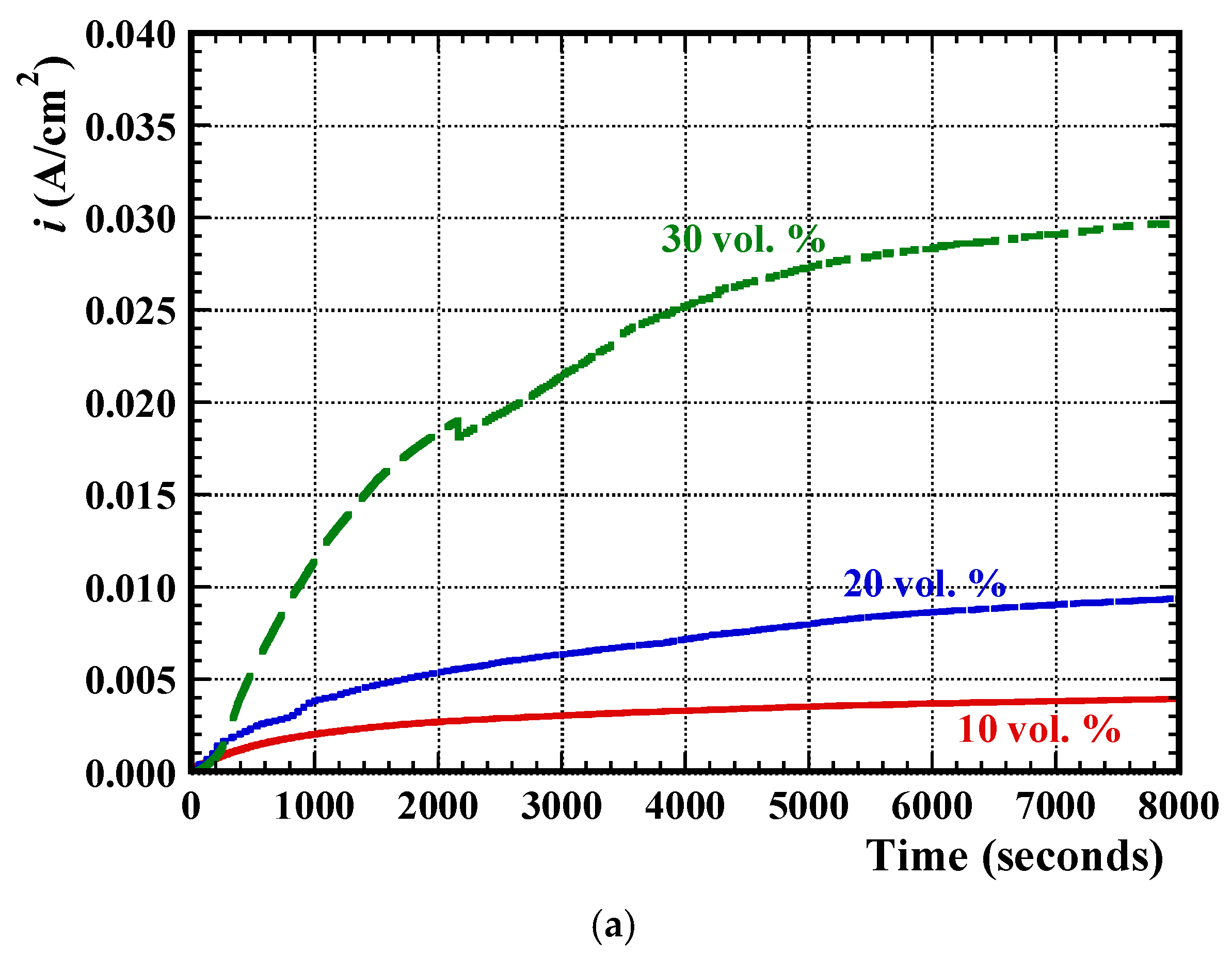
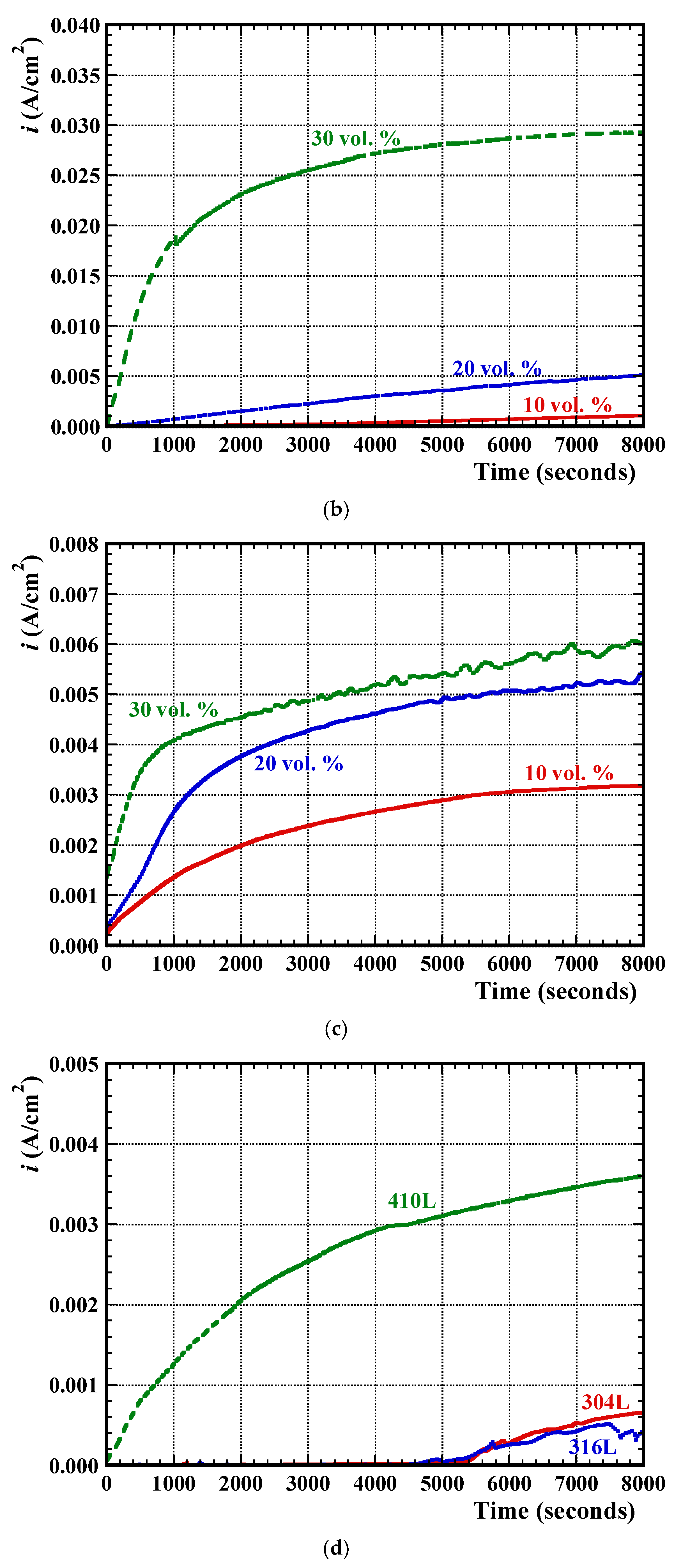
3.2. Electrochemical Measurements
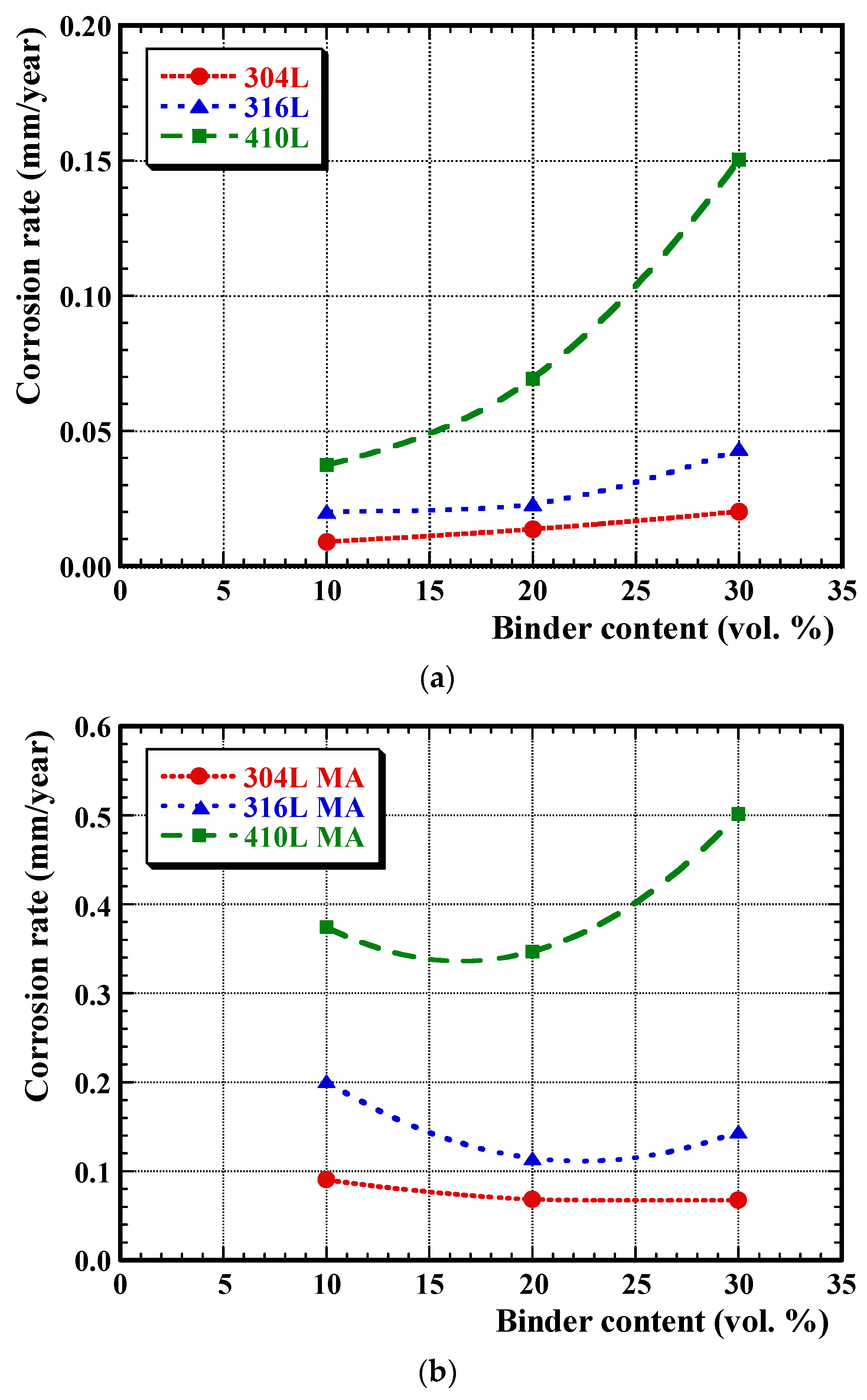
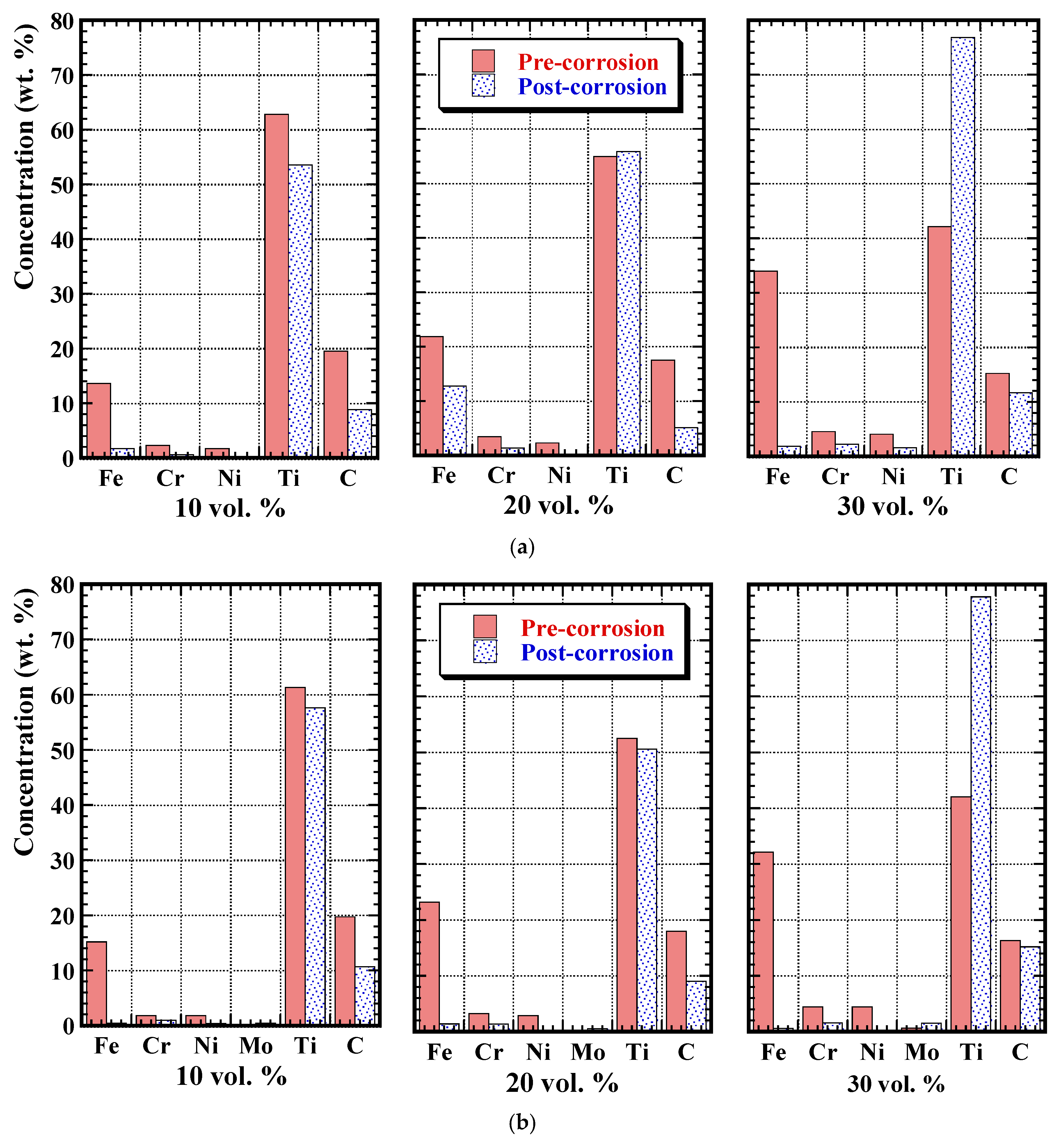
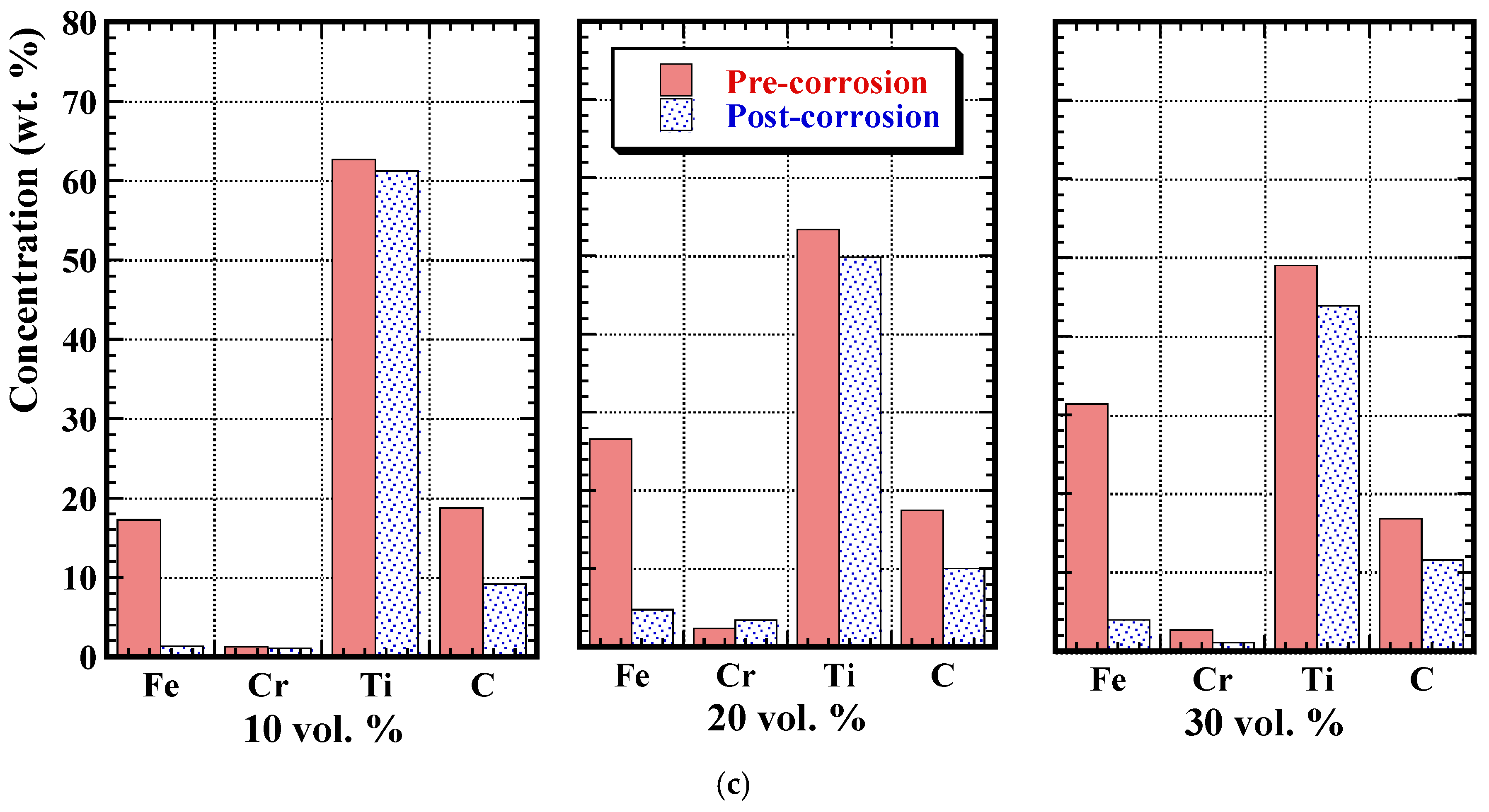
3.3. Post-Corrosion Sample Characterisation
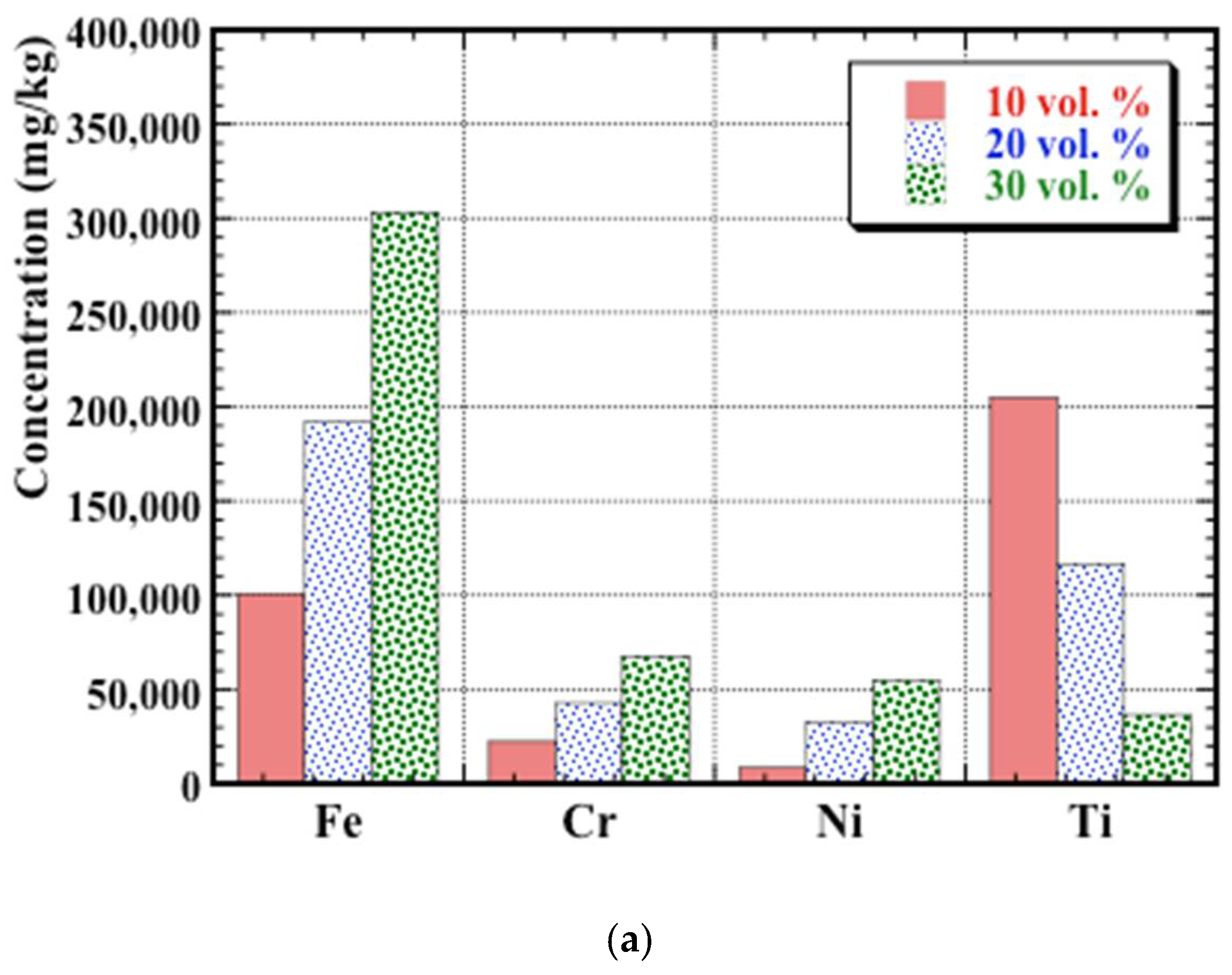
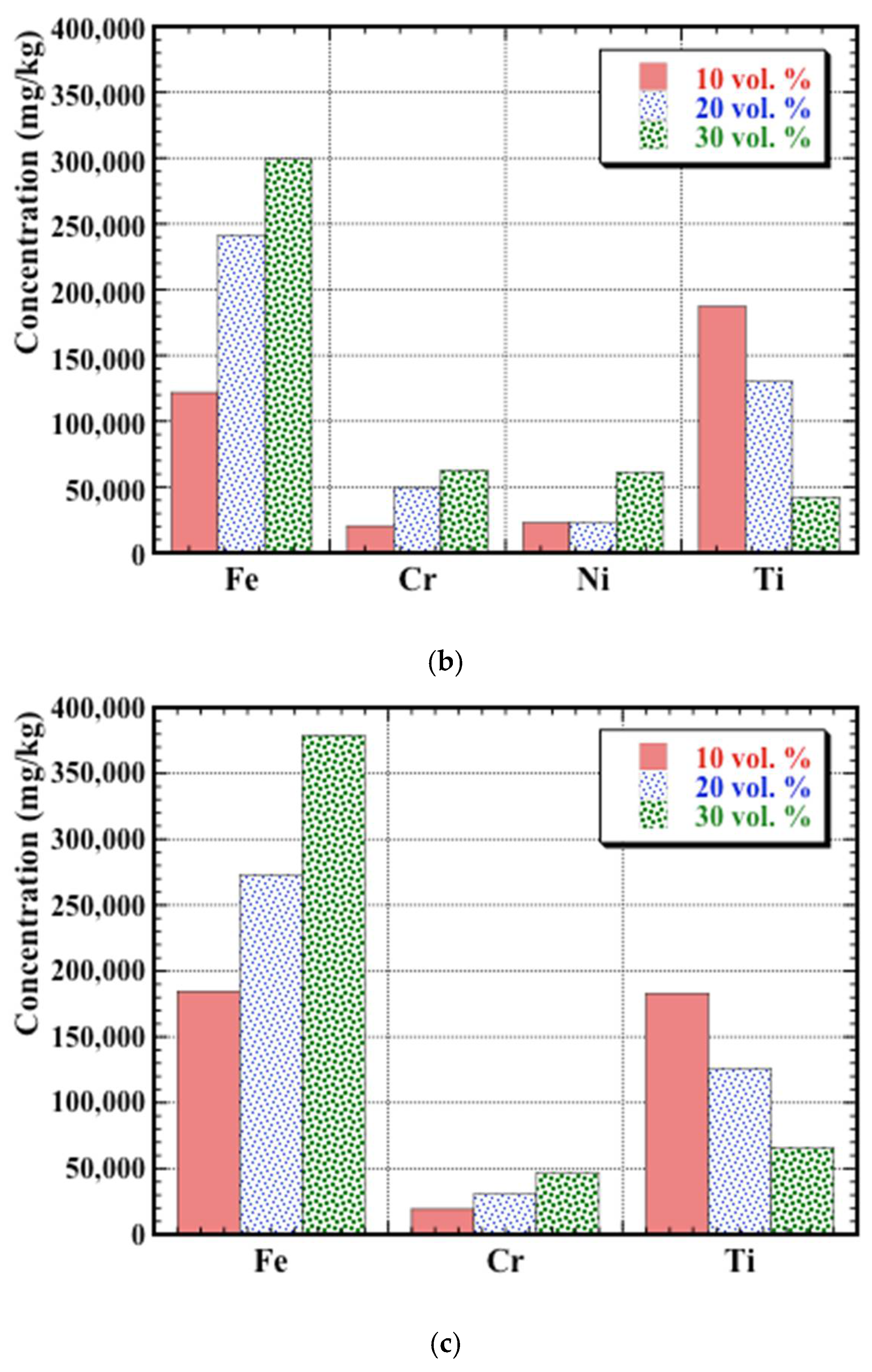
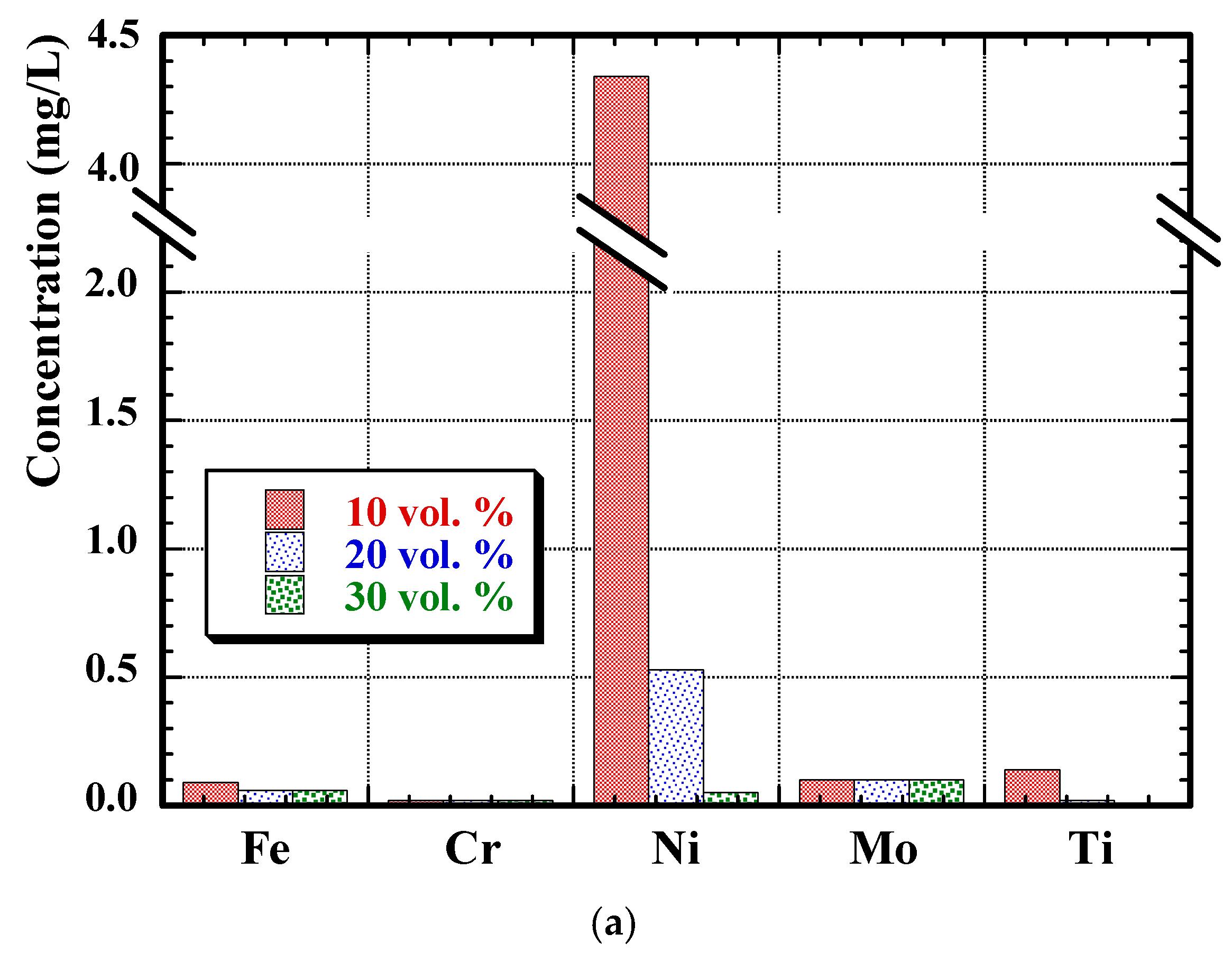
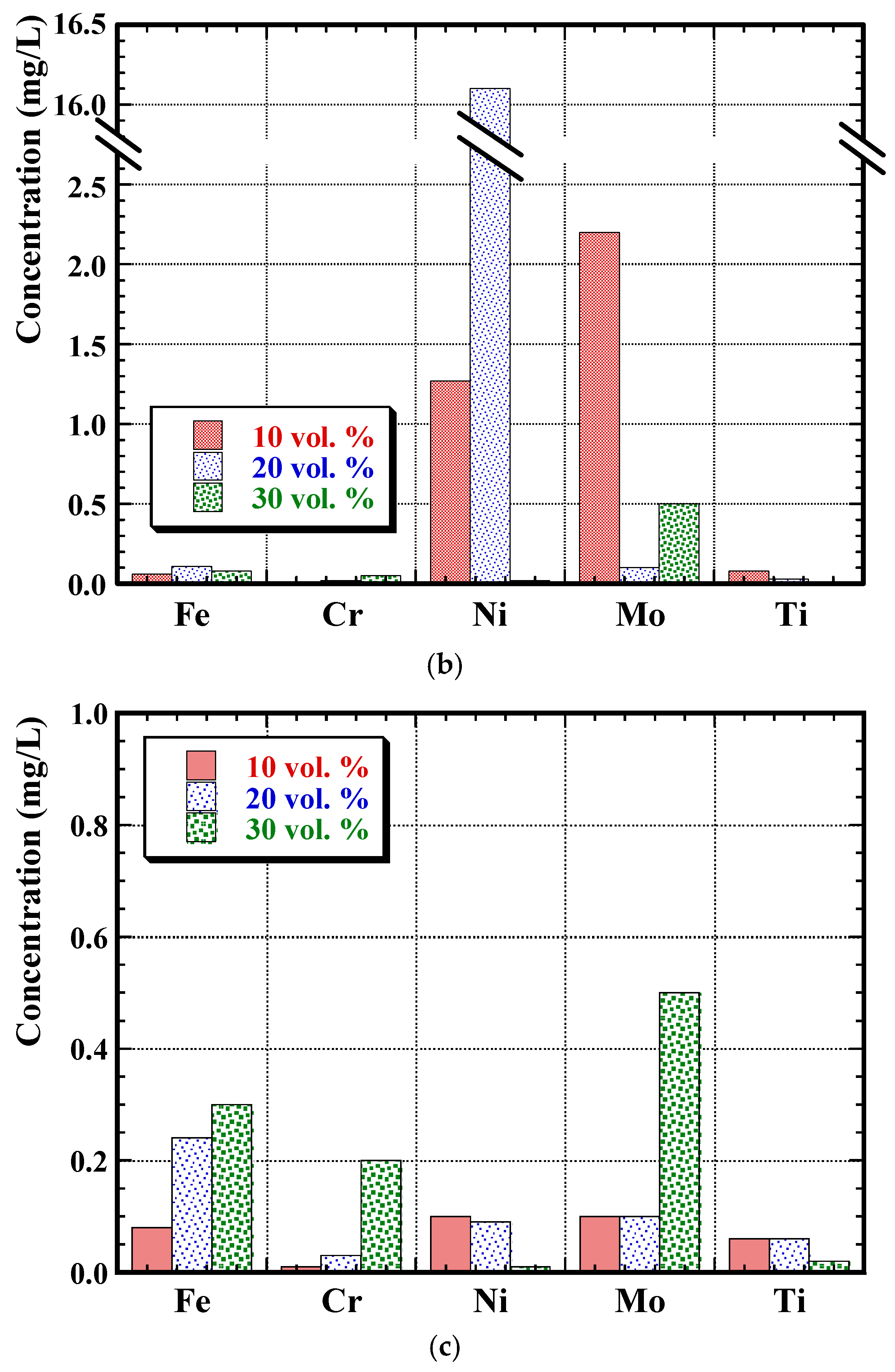
3.4. Post-Corrosion Chemical Analysis
4. Conclusions
Author Contributions
Acknowledgements
Conflicts of Interest
References
- Liu, H.Y.; Huang, J.H.; Yin, C.F.; Zhang, J.G.; Lin, G.B. Microstructure and properties of TiC-Fe cermets coatings by reactive flame spraying using asphalt as carbonaceous precursor. Ceram. Int. 2007, 33, 827–835. [Google Scholar] [CrossRef]
- Ettmayer, P.; Kolaska, H.; Dreyer, K. Effect of the sintering atmosphere on the properties of cermets. Powder Metall. Int. 1991, 23, 224–230. [Google Scholar]
- Pirso, J.; Viljus, M.; Letunovits, S. Sliding wear of TiC-NiMo cermets. Tribol. Int. 2004, 37, 817–824. [Google Scholar] [CrossRef]
- Pirso, J.; Viljus, M.; Juhani, K.; Letunovits, S. Two-body dry abrasive wear of cermets. Wear 2009, 266, 21–29. [Google Scholar] [CrossRef]
- Onuoha, C.C.; Farhat, Z.N.; Kipouros, G.J.; Plucknett, K.P. The effects of TiC grain size on the sliding wear behaviour of TiC-316L stainless steel cermets. Wear 2013, 303, 321–333. [Google Scholar] [CrossRef]
- Manoj Kumar, B.V.; Balasubramanian, R.; Basu, B.J. Electrochemical behaviour of TiCN-Ni-based cermets. J. Am. Ceram. Soc. 2007, 90, 205–210. [Google Scholar] [CrossRef]
- Ghandehari, M.H. Anodic behaviour of cemented WC-6-percent Co alloy in phosphoric acid solutions. J. Electrochem. Soc. 1980, 127, 2144–2147. [Google Scholar] [CrossRef]
- Sutthiruangwong, S.; Mori, G.; Kosters, R. Passivity and pseudopassivity of cemented carbides. Int. J. Refract. Met. Hard Mater. 2005, 23, 129–136. [Google Scholar] [CrossRef]
- Sacks, N. The Wear and Corrosive-Wear Response of Tungsten Carbide-Cobalt Hardmetals under Woodcutting and Three Body Abrasion Conditions. Ph.D. Thesis, University of Erlangen-Nurnberg, Bavaria, Germany, 2002. [Google Scholar]
- Tomlinson, W.J.; Linzell, C.R. Anodic polarization and corrosion of cemented carbides with cobalt and nickel binders. J. Mater. Sci. 1988, 23, 914–918. [Google Scholar] [CrossRef]
- Human, A.M.; Northrop, I.T.; Luyckx, S.B.; James, N.M. A comparison between cemented carbides containing cobalt- and nickel-based binders. J. Hard Mater. 1991, 2, 245–256. [Google Scholar]
- Human, A.M.; Exner, H.E. Electrochemical behavior of tungsten carbide hard metals. Mater. Sci. Eng. A 1996, 209, 180–191. [Google Scholar] [CrossRef]
- Human, A.M.; Exner, H.E. The relationship between electrochemical behavior and in-service corrosion of WC based cemented carbides. Int. J. Refract. Met. Hard Mater. 1997, 15, 65–71. [Google Scholar] [CrossRef]
- Sutthiruangwong, S.; Mori, G. Corrosion properties of Co-based cemented carbides in acidic solutions. Int. J. Refract. Met. Hard Mater. 2003, 21, 135–145. [Google Scholar] [CrossRef]
- Banerjee, D.; Lal, G.K.; Upadhyaya, G.S. Effect of binder-phase modification and Cr3C2 addition on properties of WC-10Co cemented carbide. J. Mater. Eng. Perform. 1995, 4, 563–572. [Google Scholar] [CrossRef]
- Tomlinson, W.J.; Ayerst, N.J. Anodic polarization and corrosion of WC-Co hard metals containing small amounts of Cr3C2 and/or VC. J. Mater. Sci. 1989, 24, 2348–2354. [Google Scholar] [CrossRef]
- Human, A.M.; Roebuck, B.; Exner, H.E. Electrochemical polarisation and corrosion behaviour of cobalt and Co(W,C) alloys in 1N sulphuric acid. Mater. Sci. Eng. A 1998, 241, 202–210. [Google Scholar] [CrossRef]
- Monticelli, C.; Frignani, A.; Zucchi, F. Investigation on the corrosion process of carbon steel coated by HVOF WC/Co cermets in neutral solution. Corros. Sci. 2004, 46, 1225–1237. [Google Scholar] [CrossRef]
- Trueman, A.; Schweinsberg, D.P.; Hope, G.A. A study of the effect of cobalt additions on the corrosion of tungsten carbide/carbon steel metal matrix composites. Corros. Sci. 1999, 41, 1377–1389. [Google Scholar] [CrossRef]
- Wentzel, E.J.; Allen, C. The erosion-corrosion resistance of tungsten-carbide hard metals. Int. J. Refract. Met. Hard Mater. 1997, 15, 81–87. [Google Scholar] [CrossRef]
- Wood, R.J.K. Tribo-corrosion of coatings: A review. J. Phys. D Appl. Phys. 2007, 40, 5502–5521. [Google Scholar] [CrossRef]
- Hochstrasser-Kurz, S.; Mueller, Y.; Latkoczy, C.; Virtanen, S.; Schmutz, P. Analytical characterization of the corrosion mechanisms of WC-Co by electrochemical methods and inductively coupled plasma mass spectroscopy. Corros. Sci. 2007, 49, 2002–2020. [Google Scholar] [CrossRef]
- Cho, J.E.; Hwang, S.Y.; Kim, K.Y. Corrosion behaviour of thermal sprayed WC cermets coatings having various metallic binders in strong acidic environments. Surf. Coat. Technol. 2006, 200, 2653–2662. [Google Scholar] [CrossRef]
- Perry, J.M.; Neville, A.; Hodgkiss, T. A comparison of the corrosion behavior of WC-Co-Cr and WC-Co HVOF thermally sprayed coatings by in situ atomic force microscopy (AFM). J. Therm. Spray Technol. 2002, 11, 536–541. [Google Scholar] [CrossRef]
- Patel, M.; Saurabh, K.; Bhanu Prasad, V.V.; Subrahmanyam, J. High temperature C/C–SiC composite by liquid silicon infiltration: A literature review. Bull. Mater. Sci. 2012, 35, 63–73. [Google Scholar] [CrossRef]
- Levy, A.; Miriyev, A.; Elliott, A.; Babu, S.S.; Frage, N. Additive manufacturing of complex-shaped graded TiC/steel composites. Mater. Des. 2017, 118, 198–203. [Google Scholar] [CrossRef]
- Collier, R.B.; Plucknett, K.P. A comparison of anionic and cationic polyelectrolytes for the aqueous colloidal processing of titanium carbide ceramics. Int. J. Refract. Met. Hard Mater. 2011, 29, 298–305. [Google Scholar] [CrossRef]
- Boyer, H.E.; Gall, T.L. Metals Handbook: Desk Edition, 2nd ed.; Davis, J.R., Ed.; ASM International: Geauga County, OH, USA, 1998. [Google Scholar]
- Jin, C.; Onuoha, C.C.; Farhat, Z.N.; Kipouros, G.J.; Plucknett, K.P. Reciprocating wear behaviour of TiC-stainless steel cermets. Tribol. Int. 2016, 105, 250–263. [Google Scholar] [CrossRef]
- Plucknett, K.P.; Becher, P.F. Processing and microstructure development of titanium carbide-nickel aluminide composites prepared by melt-infiltration/sintering (MIS). J. Am. Ceram. Soc. 2001, 84, 55–61. [Google Scholar] [CrossRef]
- Mendelson, M.I. Average grain size in polycrystalline ceramics. J. Am. Ceram. Soc. 1969, 52, 443–446. [Google Scholar] [CrossRef]
- Gurland, J. The measurement of grain contiguity in 2-phase alloys. Trans. Metall. Soc. AIME 1958, 212, 452–455. [Google Scholar]
- Marquardt, D. An algorithm for least-squares estimation of nonlinear parameters. SIAM J. Appl. Math. 1963, 11, 431–441. [Google Scholar] [CrossRef]
- ASTM Standard G102-89. Standard Practice for Calculation of Corrosion Rates and Related Information from Electrochemical Measurements; ASTM International: West Conshohocken, PA, USA, 2010.
- Buccolieri, A.; Buccolieri, G.; Cardellicchio, N.; Dell’Atti, A.; Di Leo, A.; Maci, A.; Petronio, B.M. Distribution and speciation of metals in surface sediments of Taranto Gulf (Ionian Sea, Southern Italy). Ann. Chim. 2004, 94, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.C.; Chi, P.H.; Shiue, M.Y. Comparison of different digestion methods for total decomposition of siliceous and organic environmental samples. Anal. Sci. 2001, 17, 1395–1399. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Casey, J.F.; Gao, Y. A new sample preparation method for crude or fuel oils by mineralization utilizing single reaction chamber microwave for broader multi-element analysis by ICP techniques. Fuel 2017, 206, 64–79. [Google Scholar] [CrossRef]
- Isaacs, H.S. The localized breakdown and repair of passive surfaces during pitting. Corros. Sci. 1989, 29, 313–323. [Google Scholar] [CrossRef]
- Jones, D.A. Principles and Prevention of Corrosion, 1st ed.; Macmillan Publishing Company: Macmillan, NY, USA, 1991. [Google Scholar]
- Cowling, R.D.; Hintermann, H.E. Corrosion of titanium carbide. J. Electrochem. Soc. Electrochem. Technol. 1970, 117, 1447–1449. [Google Scholar] [CrossRef]
- Suguma, T. CVD-titanium carbonitride coatings as corrosion-preventing barriers for steel in acid-brine steam at 200 degrees C. Mater. Lett. 1999, 38, 227–234. [Google Scholar] [CrossRef]
- Application Note CORR-4. Electrochemistry and Corrosion: Overview and Techniques; Princeton Applied Research: Oak Ridge, TN, USA, 1999. [Google Scholar]
- Von Fieandt, L.; Johansson, K.; Lindahl, E.; Larsson, T.; Boman, M.; Rehnlund, D. Corrosion properties of CVD grown Ti(C,N) coatings in 3.5 wt. % NaCl environment. Corros. Eng. Sci. Technol. 2018, 53, 316–320. [Google Scholar] [CrossRef]
- Toma, D.; Brandl, W.; Marginean, G. Wear and corrosion behaviour of thermally sprayed cermet coatings. Surf. Coat. Technol. 2001, 138, 149–158. [Google Scholar] [CrossRef]
- Rendon-Belmonte, M.; Perez-Quiroz, J.T.; Teran-Guillen, J.; Porcayo-Calderon, J.; Torres-Acosta, A.; Orozco-Gambia, G. Evaluation of a Cr3C2(NiCr) coating deposited on s4400 by means of an HVOF process and used for flow plates of PEM fuel. Int. J. Electrochem. Soc. 2012, 7, 1079–1092. [Google Scholar]
- Memarrashidi, Z.; Plucknett, K.P. Factors influencing the aqueous electrochemical response of TiC-Ni3Al Cermets. J. Mater. Res. 2017, 17, 3333–3343. [Google Scholar] [CrossRef]
- Wu, Q.L.; Li, W.; Zhong, N. Corrosion behavior of TiC particle-reinforced 304 stainless steel. Corros. Sci. 2011, 53, 4258–4264. [Google Scholar] [CrossRef]
- McCafferty, E. Introduction to Corrosion Science, 1st ed.; Springer-Verlag: Midtown Manhattan, NY, USA, 2010. [Google Scholar]
- Jin, C.; Plucknett, K.P. Microstructural instability in TiC-316L cermets. Int. J. Refract. Met. Hard Mater. 2016, 58, 74–83. [Google Scholar] [CrossRef]
- Wu, Q.L.; Li, W.; Yin, Y. Corrosion behavior of TiC particle-reinforced 2Cr13 stainless steel. Steel Res. Int. 2011, 82, 719–725. [Google Scholar] [CrossRef]







| Type | Nominal Steel Composition (Max. wt. %) | Density | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cr | Ni | C | Mn | Si | P | S | N | Mo | (g/cm3) | |
| 304-L | 18–20 | 8–12 | 0.03 | 2 | 1 | 0.045 | 0.03 | 0.1 | - | 8.03 |
| 316-L | 16–18 | 10–14 | 0.03 | 2 | 1 | 0.045 | 0.03 | 0.1 | 2–3 | 8.03 |
| 410-L | 11–13.5 | 0.75 | 0.03 | 1 | 1 | 0.04 | 0.03 | - | 0.75–1.25 | 7.75 |
| Binder Content (vol. %) | OCP (V vs. SCE) * | ||
|---|---|---|---|
| TiC-304 L | TiC-316 L | TiC-410 L | |
| 10 | −0.224 (±0.039) | −0.241 (±0.058) | −0.470 (±0.034) |
| 20 | −0.260 (±0.027) | −0.264 (±0.018) | −0.526 (±0.018) |
| 30 | −0.278 (±0.031) | −0.233 (±0.028) | −0.520 (±0.020) |
| Steel only | −0.105 (±0.021) | −0.123 (±0.040) | −0.455 (±0.030) |
| Sample | icrit (A/cm2) | ipass (A/cm2) |
|---|---|---|
| TiC-10 vol. % 410 L | 0.018 | 0.010 |
| TiC-20 vol. % 410 L | 0.030 | 0.020 |
| TiC-30 vol. % 410 L | 0.043 | 0.031 |
| TiC-10 vol. % 316 L | 0.024 | 0.013 |
| TiC-20 vol. % 316 L | 0.045 | 0.030 |
| TiC-30 vol. % 316 L | 0.062 | 0.056 |
| TiC-10 vol. % 304 L | 0.021 | 0.015 |
| TiC-20 vol. % 304 L | 0.044 | 0.028 |
| TiC-30 vol. % 304 L | 0.066 | 0.057 |
| Sample | Ecorr (V vs. SCE) | icorr (µA/cm2) | icorrMA (µA/cm2) |
|---|---|---|---|
| TiC-10 vol. % 304 L | −0.210 (±0.047) | 0.576 (±0.206) | 5.76 (±2.06) |
| TiC-20 vol. % 304 L | −0.288 (±0.023) | 0.922 (±0.033) | 4.61 (±0.165) |
| TiC-30 vol. % 304 L | −0.264 (±0.029) | 1.440 (±0.235) | 4.80 (±0.783) |
| 304 L stainless steel | −0.204 (±0.023) | 0.221 (±0.013) | NA |
| TiC-10 vol. % 316 L | −0.211 (±0.031) | 1.261 (±0.808) | 12.61 (±8.08) |
| TiC-20 vol. % 316 L | −0.266 (±0.028) | 1.522 (±0.230) | 7.61 (±1.15) |
| TiC-30 vol. % 316 L | −0.234 (±0.023) | 3.043 (±0.516) | 10.143 (±1.72) |
| 316 L stainless steel | −0.221 (±0.031) | 0.190 (±0.022) | NA |
| TiC-10 vol. % 410 L | −0.465 (±0.035) | 2.298 (±0.649) | 22.98 (±6.49) |
| TiC-20 vol. % 410 L | −0.499 (±0.001) | 4.483 (±0.163) | 22.415 (±0.815) |
| TiC-30 vol. % 410 L | −0.506 (±0.037) | 5.965 (±0.244) | 19.883 (±8.087) |
| 410 L stainless steel | −0.400 (±0.024) | 2.004 (±0.451) | NA |
| Sample | Epit (V vs. SCE) | Eprot (V vs. SCE) | ΔE (V vs. SCE) | Hysteresis Loop Analysis |
|---|---|---|---|---|
| TiC-10 vol. % 304 L | −0.068 | −0.339 | 0.271 | (−) |
| TiC-20 vol. % 304 L | 0.150 | No protection | NA | (+) |
| TiC-30 vol. % 304 L | 0.567 | No protection | NA | (+) |
| 304 L stainless steel | 0.101 | No protection | NA | (+) |
| TiC-10 vol. % 316 L | −0.132 | −0.335 | 0.203 | (−) |
| TiC-20 vol. % 316 L | −0.186 | No protection | NA | (+) |
| TiC-30 vol. % 316 L | 0.513 | No protection | NA | (+) |
| 316 L stainless steel | 0.014 | No protection | NA | (+) |
| TiC-10 vol. % 410 L | −0.167 | −0.027 | 0.140 | (−) |
| TiC-20 vol. % 410 L | −0.276 | −0.145 | 0.131 | (−) |
| TiC-30 vol. % 410 L | −0.283 | 0.0035 | 0.286 | (−) |
| 410 L stainless steel | −0.238 | No protection | NA | (+) |
| TiC | −0.26 | 1.12 | 0.86 | (−) |
| Sample | Polarisation Voltage (V vs. SCE) | ||
|---|---|---|---|
| 304 L | 316 L | 410 L | |
| Single metallic phase (steel) | 0.10725 | 0.16475 | −0.175 |
| 10 vol. % steel | 0.02075 | −0.0505 | −0.127 |
| 20 vol. % steel | 0.2475 | 0.01725 | −0.2275 |
| 30 vol. % steel | 0.575 | 0.52 | −0.195 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Onuoha, C.; Russell, Z.; Kipouros, G.; Farhat, Z.; Plucknett, K. The Aqueous Electrochemical Response of TiC–Stainless Steel Cermets. Metals 2018, 8, 398. https://doi.org/10.3390/met8060398
Onuoha C, Russell Z, Kipouros G, Farhat Z, Plucknett K. The Aqueous Electrochemical Response of TiC–Stainless Steel Cermets. Metals. 2018; 8(6):398. https://doi.org/10.3390/met8060398
Chicago/Turabian StyleOnuoha, Chukwuma, Zhila Russell, Georges Kipouros, Zoheir Farhat, and Kevin Plucknett. 2018. "The Aqueous Electrochemical Response of TiC–Stainless Steel Cermets" Metals 8, no. 6: 398. https://doi.org/10.3390/met8060398
APA StyleOnuoha, C., Russell, Z., Kipouros, G., Farhat, Z., & Plucknett, K. (2018). The Aqueous Electrochemical Response of TiC–Stainless Steel Cermets. Metals, 8(6), 398. https://doi.org/10.3390/met8060398




